Summary
Reelin is a glycoprotein that is critical for proper layering of neocortex during development as well as dynamic regulation of glutamatergic postsynaptic signaling in mature synapses. Here we show that Reelin also acts presynaptically, resulting in robust rapid enhancement of spontaneous neurotransmitter release without affecting properties of evoked neurotransmission. This effect of Reelin requires a modest but significant increase in presynaptic Ca2+ initiated via ApoER2 signaling. The specificity of Reelin action on spontaneous neurotransmitter release is encoded at the level of vesicular SNARE machinery as it requires VAMP7 and SNAP-25 but not synaptobrevin2, VAMP4 or vti1a. These results uncover a novel presynaptic regulatory pathway that utilizes the heterogeneity of synaptic vesicle associated SNAREs and selectively augments action potential-independent neurotransmission.
Introduction
Synaptic vesicles (SVs) within individual presynaptic nerve terminals are divided into distinct pools with respect to their relative propensities for fusion (Alabi and Tsien, 2012). Putative segregation of SV populations giving rise to action potential (AP) evoked versus spontaneous neurotransmitter release is a key functional outcome of this vesicle heterogeneity (Chung et al., 2010; Fredj and Burrone, 2009; Sara et al., 2005). Recent studies have demonstrated that the heterogeneous distribution of SV associated SNARE (soluble N-ethylmaleimide-sensitive factor attachment protein receptors) proteins underlies this functional diversity among SVs (Hua et al., 2011; Raingo et al., 2012; Ramirez et al., 2012). In central synapses, synaptobrevin2 (syb2, also called VAMP2) is the predominant SV SNARE protein that interacts with the plasma membrane SNAREs SNAP-25 and syntaxin1 to execute exocytosis (Sudhof and Rothman, 2009). However, while neurons lacking syb2 have a nearly complete absence of evoked neurotransmission they still maintain significant levels of spontaneous neurotransmitter release (Schoch et al., 2001). SVs in central synapses contain lower levels of alternative vesicular SNARE proteins such as VAMP4, VAMP7 (also called tetanus-insensitive or TI-VAMP) and Vps10p tail interactor 1 a (Vti1a), with structures similar to that of syb2 (Takamori et al., 2006). Recent evidence suggests that these alternative vesicular SNAREs maintain neurotransmission independently of syb2 (Raingo et al., 2012; Ramirez et al., 2012). Moreover, they also constitute molecular tags for independently functioning SV populations and provide a potential molecular basis for selective regulation of distinct forms of neurotransmitter release (Ramirez and Kavalali, 2012). Earlier work has provided several examples where spontaneous or evoked neurotransmission is differentially sensitive to neuromodulatory signaling cascades (Phillips et al., 2008; Pratt et al., 2011; Ramirez and Kavalali, 2011; Vyleta and Smith, 2011), however, the SV-associated substrates that link this differential regulation to vesicle pool heterogeneity have not yet been identified. Despite the accumulating functional and molecular evidence in support of this SNARE-dependent vesicle pool diversity, the physiological role of this functional specialization, in particular the biological significance of the residual syb2-independent forms of neurotransmitter release remains poorly understood.
Here, we examined the presynaptic effects of Reelin, a glycoprotein critical for proper layering of neocortex as well as dynamic regulation of glutamatergic postsynaptic signaling in mature synapses (D'Arcangelo et al., 1995; Herz and Chen, 2006). During development, Reelin is secreted by Cajal-Retzius cells in the marginal zone of embryonic brain where it guides the migration of newly generated neurons from the ventricular zone to the marginal zone, thus forming a properly layered structure in the adult brain (Knuesel, 2010; Kubo et al., 2002; Soriano and Del Rio, 2005; Trommsdorff et al., 1999; Tissir and Goffinet, 2003). Reelin is a ligand for both apolipoprotein receptor 2 (ApoER2) and very low density lipoprotein receptor (VLDLR), which are required for its developmental role (Hiesberger et al., 1999; Trommsdorff et al., 1999). After development, the production of Reelin is dramatically decreased but remains prominent in GABAergic interneurons (Alcantara et al., 1998) of the cortex and hippocampus (Pesold et al., 1998). In mature neuronal circuitry, Reelin modulates AMPA and NMDA receptor activity by postsynaptic activation of ApoER2 and VLDLR (Beffert et al., 2005; Qiu et al., 2006). The interaction between Reelin and its receptors leads to a signaling cascade initiated by phosphorylation of disabled-1 (Dab-1) which in turn leads to activation of Src, Fyn or PI-3 kinases (Kuo et al., 2005; Trommsdorff et al., 1999). Here, we demonstrate that Reelin also acts presynaptically in mature neurons to rapidly enhance spontaneous neurotransmitter release without detectable alterations in the properties of evoked neurotransmission. This action of Reelin depended on the function of the vesicular SNARE protein VAMP7 but not syb2, VAMP4 or vti1a. This finding demonstrates a novel example where an endogenous neuromodulator relies on the diversity of SV pool associated SNAREs and selectively mobilizes a subset of vesicles independent of electrical activity.
Results
Reelin causes an increase in spontaneous neurotransmitter release
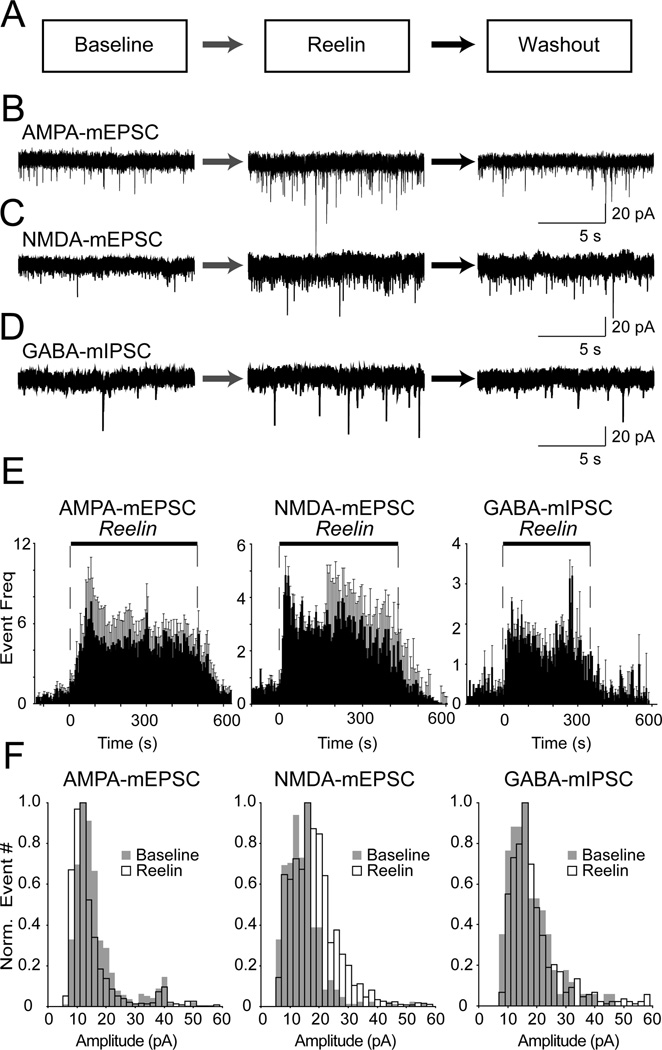
To assess the effect of Reelin on neurotransmitter release, we applied Reelin (5 nM) to hippocampal neurons and recorded spontaneous miniature postsynaptic currents in the presence of TTX to block APs. Using whole-cell voltage clamp recordings, we monitored pharmacologically isolated excitatory postsynaptic currents (mEPSCs) generated by activation of AMPA or NMDA receptors as well as GABAergic miniature inhibitory postsynaptic currents (mIPSCs) for 5 minutes in normal Tyrode’s solution. Reelin was then perfused into the chamber and mPSCs were measured for at least 5 minutes followed by washout of Reelin (Figure 1). Reelin robustly increased the frequency of spontaneous AMPA mEPSCs (Figure 1B) from 0.8 ± 0.1 Hz up to 4.8 ± 0.2 Hz during Reelin (~6-fold increase with t1/2 = 67.6 ± 14.4 s). This effect was dependent on acute Reelin application as upon Reelin removal, spontaneous event frequency returned to baseline levels (0.9 ± 0.2 Hz with t1/2 = 75.1 ± 23.3 s). Similarly, Reelin increased the frequency of both NMDA-derived mEPSCs (from 0.7 ± 0.1 Hz before Reelin to 3.2 ± 0.3 Hz during Reelin and 0.7 ± 0.1 Hz after Reelin washout, ~ 4.5-fold increase with a rise time of: t1/2 = 43.1 ± 21.0 s and a decay time of: t1/2 = 26.1 ± 7.5 s) and GABA-mediated mIPSCs (from 0.4 ± 0.04 Hz before Reelin to 1.7 ± 0.1 Hz during Reelin and 0.4 ± 0.1 Hz after Reelin washout, a ~4-fold increase with a rise time of: t1/2 = 17.0 ± 4.8 s and decay time of: t1/2 = 37.1 ± 15.1 s) (Figure 1C, D). In all cases the elevated spontaneous release frequency was sustained for longer than 5 minutes in the presence of Reelin (Figure 1E). Furthermore, the Reelin-dependent increase in spontaneous mEPSC frequency was present as early as 6 days in vitro (DIV) (Supplementary Figure 1) at a developmental time point when synapses are not capable of robust evoked release (Mozhayeva et al., 2002). This finding suggests an early developmental significance for the Reelin effect. Previous studies have shown that Reelin can augment the amplitude of AP-evoked NMDA receptor mediated currents (Chen et al., 2005). We found a modest, but significant, increase in NMDA mEPSC amplitudes from 13.3 ± 1.8 pA at baseline to 16.9 ± 1.6 pA after Reelin application (Figure 1F and see Supplementary Figure 2 for additional details). This observation could account for the previously reported increase in evoked NMDA receptor mediated synaptic responses (Chen et al., 2005). Under the same conditions, the amplitude of AMPA mEPSCs showed a slight decrease from 16.2 ± 1.3 pA before Reelin to 14.5 ± 1.3pA in the presence of Reelin, while GABA mEPSC amplitude was relatively unchanged (Figure 1F). Although Reelin action on spontaneous release may have a transient component, within the time frame of our experiments, our data did not reveal a statistically significant difference between the initial and later phases of Reelin action. Moreover, it is important to note that as Reelin is a large protein delivered at nM concentrations, it is difficult to ensure the consistency of Reelin concentrations during application.
Figure 1. Reelin increases the frequency of spontaneous neurotransmission.
(A) Whole-cell voltage clamp recordings of AMPA-mEPSCs, NMDA-mEPSCs as well as GABA- mIPSCs were performed for 5 minutes in normal Tyrode’s solution (baseline), followed by at least 5 minute-long Reelin application and subsequent 5 minutes during washout of Reelin.
(B, C and D) Example traces of spontaneous neurotransmission derived from recordings of (B) AMPA-mEPSC, (C) NMDA-mEPSC and (D) GABA-mIPSCs before Reelin application (left), during 5 minutes of Reelin application (middle), after Reelin washout (Right).
(E) The mEPSC and mIPSC frequency for AMPA-, NMDA- and GABA-mediated spontaneous events increases in response to Reelin (AMPA n = 6; NMDA n = 8; GABA n = 5). AMPA mEPSC frequency increased from 0.8 ± 0.1 Hz before Reelin to 4.8 ± 0.2 Hz during Reelin and decreased to 0.9 ± 0.2 Hz after Reelin washout. Reelin similarly affected both NMDA- mEPSCs (from 0.7 ± 0.1 Hz before Reelin to 3.2 ± 0.3 Hz during Reelin and 0.7 ± 0.1 Hz after Reelin washout) as well as GABA-mediated mIPSCs (from 0.4 ± 0.04 Hz before Reelin to 1.7 ± 0.1 Hz during Reelin and 0.4 ± 0.1 Hz after Reelin washout)
(F) Distribution of AMPA-mEPSC, NMDA-mEPSC and GABA-mIPSC amplitudes before Reelin (Baseline, grey bars) and during Reelin (white bars) application. Overall averages of AMPA mEPSC amplitude showed a slight decrease during Reelin treatment (p < 0.05 paired student’s T-test, n = 7) while NMDA mEPSC amplitude increased (P < 0.01 paired student’s T-test, n = 10). GABA mIPSC amplitudes did not reveal a marked change after Reelin application (p > 0.3, n = 4). All error bars represent standard errors of the mean.
Reelin selectively augments spontaneous transmission
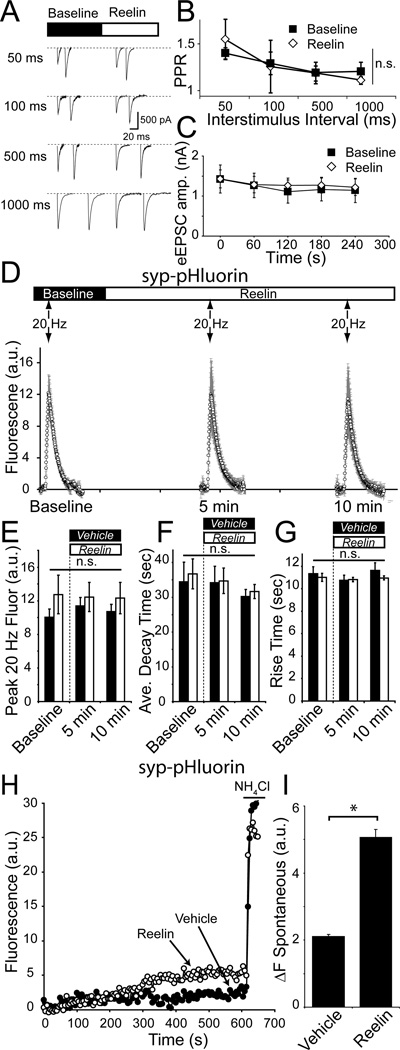
To assess the effect of Reelin on evoked SV fusion probability, we measured paired-pulse facilitation of evoked AMPA receptor mediated synaptic responses in hippocampal neurons in the presence of Reelin (within ~5 minutes of treatment). Neurons were stimulated using single APs with increasing inter-stimulus intervals of 50 ms, 100 ms, 500 ms and 1000 ms (Figure 2A). These experiments did not reveal a significant difference in the ratio of synaptic responses to paired-pulse stimulation in the presence or absence of Reelin, suggesting that Reelin does not alter AP-dependent release probability (Figure 2B), in agreement with earlier observations (Qiu et al., 2006). In addition, absolute amplitudes of evoked AMPA-EPSCs were stable and did not show a significant difference before or during Reelin application (Figure 2C; before Reelin: 1223.3 ± 130.7 pA; after Reelin: 1292.2 ± 88.0 pA; p>0.7).
Figure 2. Reelin selectively augments spontaneous SV trafficking without altering the properties of evoked neurotransmission.
(A) Example of paired-pulse ratio traces for AMPA receptor mediated events before Reelin and after Reelin application.
(B) Paired-pulse ratio before and after Reelin application was unaltered compared to Vehicle control (Vehicle n = 5; Reelin n = 5).
(C) Single AP stimulation delivered at 60 s intervals did not alter AMPA-EPSC amplitude in the absence (closed squares) or presence (open diamonds) of Reelin (No Reelin n = 7, Reelin = 13)
(D) Optical measurements of cells infected with synaptophysin-pHluorin. Cells were electrically stimulated with 200 APs delivered at 20 Hz before, 5 minutes after, and 10 minutes after Reelin application.
(E, F and G) Reelin did not affect 20 Hz stimulation driven fluorescence peak amplitude, decay time or rise time (Vehicle n = 5; Reelin n = 6). (E) Peak fluorescence amplitudes before Vehicle 10.1 ± 0.9 a.u., after 5 minutes in Vehicle 11.4 ± 1.0 a.u., and after 10 minutes in Vehicle 10.7 ± 0.9 a.u., compared to 12.7 ± 2.3 a.u. before Reelin, 12.4 ± 1.7 a.u. after 5 minutes of Reelin application, and 12.3 ± 1.9 a.u. 10 minutes after Reelin application. (F) Fluorescence decay kinetics were not different with 34.5 ± 5.7 s before Vehicle, 34.2 ± 4.8 s 5 minutes after Vehicle, and 30.2 ± 2.1 s after 10 minutes in Vehicle compared to 35.7 ± 3.5 s before Reelin application, 34.6 ± 3.0 s after 5 minutes in Reelin and 32.1 ± 1.8 s after 10 minutes in Reelin. Finally, fluorescence rise time (G) in Reelin was not significantly different from Vehicle (11.3 ± 0.6 s before Vehicle, 10.8 ± 0.4 s after 5 minutes in Vehicle, and 11.6 ± 0.7 10 minutes after Vehicle compared to 11.0 ± 0.4 s before Reelin application, 10.8 ± 0.2 s after 5 minutes in Reelin and 11.0 ± 0.2 s after 10 minutes in Reelin). There were no significant differences between any of the groups, analyzed by ANOVA (p > 0.2 between all groups).
(H and I) In the presence of the vacuolar ATPase inhibitor, folimycin, syp-pHluorin fluorescence accumulates reflecting spontaneous vesicle trafficking. (H) Example traces of fluorescence increase in Vehicle (closed circles) and Reelin (open circles) followed by NH4Cl application to visualize the total syp-pHluorin pool. (I) In the presence of Reelin, spontaneous syp-pHluorin fluorescence increased to 5.06 ± 0.24 a.u. after 10 minutes compared to 2.11 ± 0.05 a.u. after 10 minutes in Vehicle (Vehicle, n = 701 synapses from 6 experiments; Reelin, n = 917 synapses from 6 experiments; p-value <0.001). Here and all subsequent figures asterisks denote statistical significance. All error bars represent standard errors of the mean.
To directly examine the effect of Reelin on preSV trafficking, we turned to optical monitoring of a SV associated protein, synaptophysin, tagged with pH-sensitive GFP within the vesicle lumen (synaptophysinpHluorin, syp-pH). Exogenous expression of syp-pH typically leads to its wide distribution across SV pools (Kwon and Chapman, 2011). Neurons were stimulated using a bipolar electrode delivering 200 APs at 20 Hz, before, 5 minutes after, and 10 minutes after Reelin application (Figure 2D). In this setting, we did not detect a significant change in peak fluorescence amplitude (Figure 2E), fluorescence decay kinetics (Figure 2F) or fluorescence rise time (Figure 2G), indicating that under these conditions Reelin did not significantly alter activity-evoked SV trafficking.
In the presence of the vacuolar ATPase inhibitor folimycin, SVs cannot be re-acidified after endocytosis, thus trapping the pHluorin molecules present in vesicles that have undergone fusion in a fluorescent state (Sara et al., 2005). Therefore, in the presence of folimycin, the AMPAR antagonist CNQX, the NMDA antagonist AP-5, and TTX to prevent AP firing, spontaneous transmission can be directly monitored as an accumulation of fluorescence at rest (Atasoy et al., 2008; Hua et al., 2011; Ramirez et al., 2012). Under these conditions, Reelin application increased spontaneous trafficking of syp-pH more than two-fold from 2.11 ± 0.05 a.u. after 10 minutes in Vehicle to 5.06 ± 0.24 a.u. after 10 minutes in Reelin (Figure 2H, I). Together these data indicate that Reelin selectively facilitates spontaneous neurotransmission while leaving evoked neurotransmission and SV trafficking relatively unaffected.
The effect of Reelin requires the ApoE type 2 receptor, subsequent PI3K activity and Ca2+ signaling
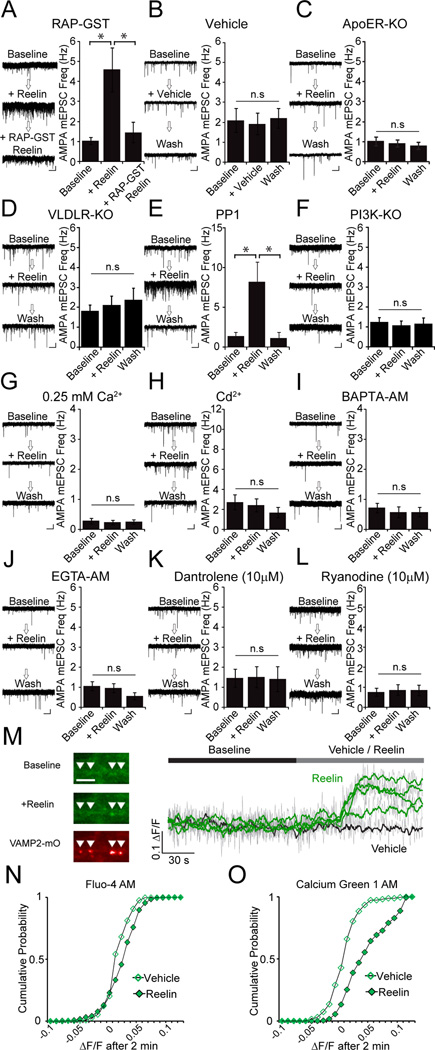
Reelin is known to bind two members of the large-density lipoprotein receptor (LDLR) family, ApoER2 and VLDLR, leading to their clustering (Herz and Chen, 2006). To test the hypothesis that Reelin binds to a member of the LDLR family to induce the observed increase in spontaneous neurotransmission frequency, we monitored spontaneous AMPA-mEPSC frequency in the presence of Reelin and a high-affinity receptor-associated protein fused to GST (GST-RAP). GST-RAP acts as a non-receptor subtype specific ligand for the LDLR-related proteins (LRPs) and thus a competitive antagonist of ApoER2 (Figure 3A) (Herz et al., 1991). In this setting, Reelin again produced a robust increase in mEPSC frequency; however, the application of GST-RAP in the presence of Reelin decreased mEPSC frequency to near baseline levels within minutes. To confirm that the binding of Reelin to a member of the LRP family is sufficient to elicit an increase in spontaneous transmission, we again monitored mEPSC frequency after addition of the Reelin purification media lacking Reelin (vehicle) (Figure 3B). Vehicle perfusion did not alter spontaneous mEPSC frequency. To confirm that ApoER2 and VLDLR are required for the Reelin-dependent increase in spontaneous transmission, we monitored AMPA-mEPSC frequency in neurons from mice lacking ApoER2 (ApoER2 −/−) or VLDLR (VLDLR −/−) (Figure 3C and D). Reelin did not elicit an increase in spontaneous transmission frequency in ApoER2- or VLDLR-deficient neurons, suggesting that Reelin requires both ApoER2 and VLDLR to facilitate spontaneous transmission. We next analyzed the cellular localization of ApoER2 relative to the SV marker, synapsin to probe if ApoER2 is expressed on the presynaptic terminal. We found that while ApoER2 is predominantly detected in postsynaptic structures, there was a small but significant level of overlap between ApoER2 and synapsin, suggesting ApoER2 is also present on the presynaptic surface (Supplementary Figure 3).
Figure 3. Reelin acts through ApoER2 and VLDLR to activate PI3K and triggers presynaptic Ca2+ signaling.
(A) Application of a soluble antagonist for ApoER, GST-RAP, inhibits the Reelin effect. Cell exhibited a robust increase in mEPSC frequnecy (from 1.1 ± 0.2 Hz before Reelin to 4.6 ± 1.2 Hz after Reelin (p < 0.05, n=4), while application of GST-RAP in the presence of Reelin returned mEPSC frequency to near baseline levels (1.5 ± 0.57 Hz) (p > 0.5 compared to baseline).
(B) Reelin and not another factor in Reelin culture medium is responsible for the observed increase in spontaneous transmission, as medium lacking Reelin has no effect (2.0 ± 0.7 Hz in Vehicle, compared to 1.8 ± 0.6 Hz after application of Reelin; p > 0.5, n=6)
(C) ApoER2 KO cells do not respond to Reelin (1.0 ± 0.2 Hz before Reelin, and 0.9 ± 0.2 Hz after; p > 0.5, n=7)
(D) VLDLR KO cells do not respond to Reelin (1.8 ± 0.3 Hz before Reelin, and 2.1 ± 0.5 Hz after; p > 0.3, n=5)
(E) The Src kinase inhibitor PP1 leaves the Reelin effect intact (1.4 ± 0.5 Hz before and 8.1 ± 2.5 Hz; p < 0.001, n=6)
(F) Reelin requires the activation of PI3K to augment spontaneous transmission as PI3K KO cells do not respond to Reelin (1.2 ± 0.2 Hz before and 1.1± 0.2 Hz after Reelin; p > 0.1, n=5; n=4 for wild type positive controls data not shown).
(G, H, I and J) The Reelin effect was attenuated after decreasing the extracellular Ca2+ concentration to 0.25 mM (0.3 ± 0.1 Hz before and 0.2 ± 0.1 Hz after Reelin; p > 0.5, n=7) or addition of Cd2+ (200 µM) to the extracellular solution (2.7 ± 0.8 Hz before and 2.2 ± 0.6 Hz after Reelin; p > 0.5, n=6). Cells incubated in 20 µM BAPTA-AM (0.7 ± 0.2 Hz before Reelin and 0.6 ± 0.2 Hz after Reelin; p > 0.5, n=9) or 10 µM EGTA-AM (1.1 ± 0.2 Hz before and 1.0 ± 0.2 after Reelin application; p > 0.5, n=9) did not respond to Reelin.
(K and L) Prior incubation with Dantrolene (10 µM) and Ryanodine (10 µM) also abolished the Reelin effect (1.5 ± 0.4 Hz before and 1.5 ± 0.5 Hz after Reelin and 1.1 ± 0.2 Hz before and 1.1 ± 0.2 Hz after Reelin for Dantrolene (n=6) and Ryanodine (n=9), respectively. In both cases, p > 0.5)
(M) Example traces of Calcium Green-1 images identified by co-labeling with the presynaptic marker syb2-mOrange. Scale bar is 5 µm. The presence of negative ΔF/F values are due to baseline fluorescence fluctuations as well as mild photobleaching.
(N) Cumulative probability histogram showing Fluo-4 ΔF/F over 2 min in Vehicle (N=5, 410 puncta) and Reelin (N=7, 529 puncta). K-S test shows significance p < 0.0001 with Dmax value = 0.195.
(O) Cumulative probability histogram showing Calcium Green-1 ΔF/F over 2 min in vehicle (N=3, 259 synapses) and Reelin (N=4, 309 synapses). K-S test shows significance p < 0.0001, Dmax value = 0.449. Unless noted otherwise, all p-values were calculated using paired Student’s T-test. All error bars represent standard errors of the mean.
ApoER2 is upstream of both Src kinase dependent signaling pathways as well as phosphoinositide 3-kinase (PI3K) (Bock et al., 2003a; Bock et al., 2003b; Herz and Chen, 2006). To determine which pathway is involved in the Reelin-dependent enhancement of spontaneous neurotransmission, we preincubated neurons for 1 hr in the Src inhibitor PP1 (10 µM), or applied Reelin to neurons deficient in p110α and p110β isoforms of PI3K (p110α −/−, p110β−/−) (Utermark et al., 2012) (Figure 3E and F). Cells preincubated in PP1 still exhibited a Reelin-dependent increase in mEPSC frequency (Figure 3E), whereas neurons lacking the two commonly expressed PI3K isoforms did not respond to Reelin (Figure 3F). These data suggest that PI3K, and not Src kinase, activity is required for the Reelin-dependent increase in spontaneous neurotransmission frequency. Furthermore, when neurons were preincubated in the PI3K inhibitors LY294002 and wortmannin, Reelin was unable to increase AMPA mEPSC frequency (Supplementary Figure 4A–C). However, when LY294002 and wortmannin were included only in the patch pipette, and not the extracellular solution, Reelin did increase mEPSC frequency (Supplementary Figure 4D–F). These data reinforce the notion that PI3K activation, specifically in the presynaptic neuron, is required for the effect of Reelin and suggest that Reelin acts presynaptically to increase mEPSC frequency.
Earlier studies have linked PI3K activation to increases in canonical transient receptor potential (TRPC) channel activity leading to membrane depolarization and Ca2+ influx (Bezzerides et al., 2004; Williams et al., 2011). Therefore, we next tested whether an increase in presynaptic Ca2+ signaling is required for the Reelin mediated augmentation of spontaneous neurotransmitter release. When we applied Reelin to neurons in low extracellular Ca2+ medium (0.25 mM) or in the presence of cadmium (Cd2+; 200 µM) to block Ca2+ influx (Figure 3G and H), both manipulations precluded Reelin from increasing mEPSC frequency, although low extracellular Ca2+ medium noticeably reduced the baseline spontaneous neurotransmission (0.29 ± 0.09 Hz before and 0.24 ± 0.08 Hz after Reelin in 0.25 mM extracellular Ca2+). Taken together, these results suggest that Reelin binds to ApoER2 to initiate PI3K signaling which leads to Ca2+ influx to facilitate spontaneous neurotransmission.
To test if the Reelin-dependent augmentation of spontaneous transmission requires internal Ca2+, we measured mEPSC frequency after 30-min incubation with membrane permeable Ca2+ chelators, BAPTA-AM (30 µM) or EGTA-AM (10 µM) (Figure 3I and J). Reelin had no effect on spontaneous neurotransmission in cells that were preincubated in BAPTA-AM or EGTA-AM. (Figure 3I, J). However, when BAPTA (1 mM) was included only in the patch pipette, Reelin still caused an increase in spontaneous transmission, suggesting that the suppression of the Reelin effect by BAPTA-AM and EGTA-AM are due to presynaptic Ca2+ sequestration and not postsynaptic effects (Supplementary Figure 5). We also tested whether the Ca2+ signal required for the presynaptic effect of Reelin depended on a rise in internal Ca2+ levels via Ca2+-induced Ca2+ release by activation of ryanodine receptors (RyR). In this scenario, the effect of a small amount of Ca2+ influx can be swiftly amplified giving rise to an increase in neurotransmitter release independent of APs as previously documented in hippocampal synapses (Sharma and Vijayaraghavan, 2003; Xu et al., 2009). To test if Reelin alters cytosolic Ca2+ levels through RyRs, we preincubated neurons in the ryanodine receptor blockers, dantrolene (10 µM) or ryanodine (10 µM). Preincubation in either dantrolene or ryanodine abolished the effect of Reelin (Figure 3K, L, respectively). Together these data suggest that Ca2+-induced Ca2+ release is necessary for the Reelin-dependent increase in spontaneous neurotransmitter release.
In the next set of experiments, we attempted to visualize the Reelin mediated Ca2+ signal predicted by the results of the experiments manipulating Ca2+ signaling within presynaptic boutons described above. For this purpose, we infected neurons with a red-shifted pH sensitive fluorescent protein, mOrange, fused to the luminal end of syb2 (syb2-mOrange) to identify presynaptic terminals (Raingo et al., 2012; Ramirez et al., 2012). Cells were incubated with the Ca2+ indicator Fluo-4 AM (Ca2+ KD ~ 335 nM) or the higher affinity indicator Calcium Green 1 (Ca2+ KD ~ 190 nM). After washing out extracellular dye, cells were imaged for 2 minutes in the presence of blockers to silence APs (TTX), ionotropic AMPA receptors (NBQX) and NMDA receptors (AP-5). In presynaptic terminals co-labeled with syb2-mOrange, application of Reelin, as opposed to vehicle perfusion, caused a small but significant increase in intracellular Ca2+ that was observable across nearly all boutons (Figure 3M). The Reelin-induced rise in presynaptic Ca2+ was particularly robust when monitored with Calcium Green 1 while the lower affinity dye Fluo-4 was not as effective in detecting the Reelin-induced Ca2+ signal (Figure 3 N,O). The more pronounced shift in the distribution of Ca2+ increases observed with Calcium Green 1 suggests that Reelin application results in a modest increase in presynaptic Ca2+ which in turn increases baseline spontaneous SV release rates (Lou et al., 2005; Sun et al., 2007).
Reelin-dependent facilitation of spontaneous neurotransmission requires SNAP-25 but not syb2
Our results so far suggest that Reelin acting via its canonical receptors ApoER2 and VLDLR causes a modest but significant increase in presynaptic Ca2+ which in turn augments resting neurotransmitter release rate without significantly altering the properties of evoked neurotransmitter release. At synaptic terminals, SNARE protein interactions are largely responsible for vesicle fusion and neurotransmitter release. The canonical synaptic SNARE complex composed of syb2 on the SV and syntaxin 1 and SNAP-25, both on the target plasma membrane, mediates rapid exocytosis. Syb2 is the most abundant vesicle associated SNARE and is essential for fast synchronous neurotransmission (Schoch et al. 2001; Takamori et al., 2006). Therefore, in order to examine the presynaptic fusion machinery that underlies this effect, we tested the effect of Reelin on neurons deficient in the canonical synaptic SNARE proteins SNAP-25 and syb2 (Figure 4A). Although neurons from their wild type littermates showed swift responses to Reelin application (Figure 4B), neurons lacking SNAP-25 (SNAP25 −/−) showed no response to Reelin and had an overall lower mEPSC frequency (Figure 4C) (Bronk et al., 2007). Here, it is important to note that SNAP-25 deficient synapses respond to other secretagogues such as hypertonic sucrose, ionomycin or α-latrotoxin (Bronk et al., 2007; Deak et al., 2009). These data indicate that Reelin causes an increase in SV fusion frequency that requires the function of the plasma membrane associated SNARE, SNAP-25, in agreement with an earlier study suggesting a SNAP-25 dependent role for Reelin in presynaptic function (Hellwig et al., 2011). To test if the SV SNARE syb2 is also required for the Reelin dependent augmentation in transmission, we added Reelin to neurons deficient in syb2 (syb2 −/−) (Figure 4A, D). Surprisingly, neurons lacking syb2 still responded to Reelin despite their low basal mEPSC frequency (Figure 4D). Taken together, these results suggest that the Reelin-dependent increase in spontaneous transmission requires a SNARE complex that contains SNAP-25 but does not require the vesicle associated protein syb2.
Figure 4. Reelin-induced increase in spontaneous release requires SNAP-25 but not syb2.
(A) Example traces of AMPAR mEPSCs from WT, SNAP-25 KO and Syb2 KO neurons.
(B, C and D) Reelin increases mEPSC frequency in WT cells (0.6 ± 0.2 Hz before Reelin, 2.2 ± 0.6 Hz during Reelin and 0.4 ± 0.1 Hz after Reelin washout in WT cells; asterisks p < 0.005 ) while SNAP-25 KO cells (C), do not respond to Reelin (0.06 ± 0.01 Hz before Reelin, 0.05 ± 0.02 Hz during Reelin and 0.06 ± 0.00 Hz after Reelin washout; p > 0.5). However, Reelin still caused an increase in mEPSC frequency in syb2 KO cells (D) (0.31 ± 0.04 Hz before Reelin compared to 1.1 ± 0.1 Hz after Reelin application and 0.4 ± 0.1 Hz after washout; p < 0.005). All p-values were calculated by paired Student’s T-test. All error bars represent standard errors of the mean.
Reelin facilitates trafficking of VAMP7 expressing SVs
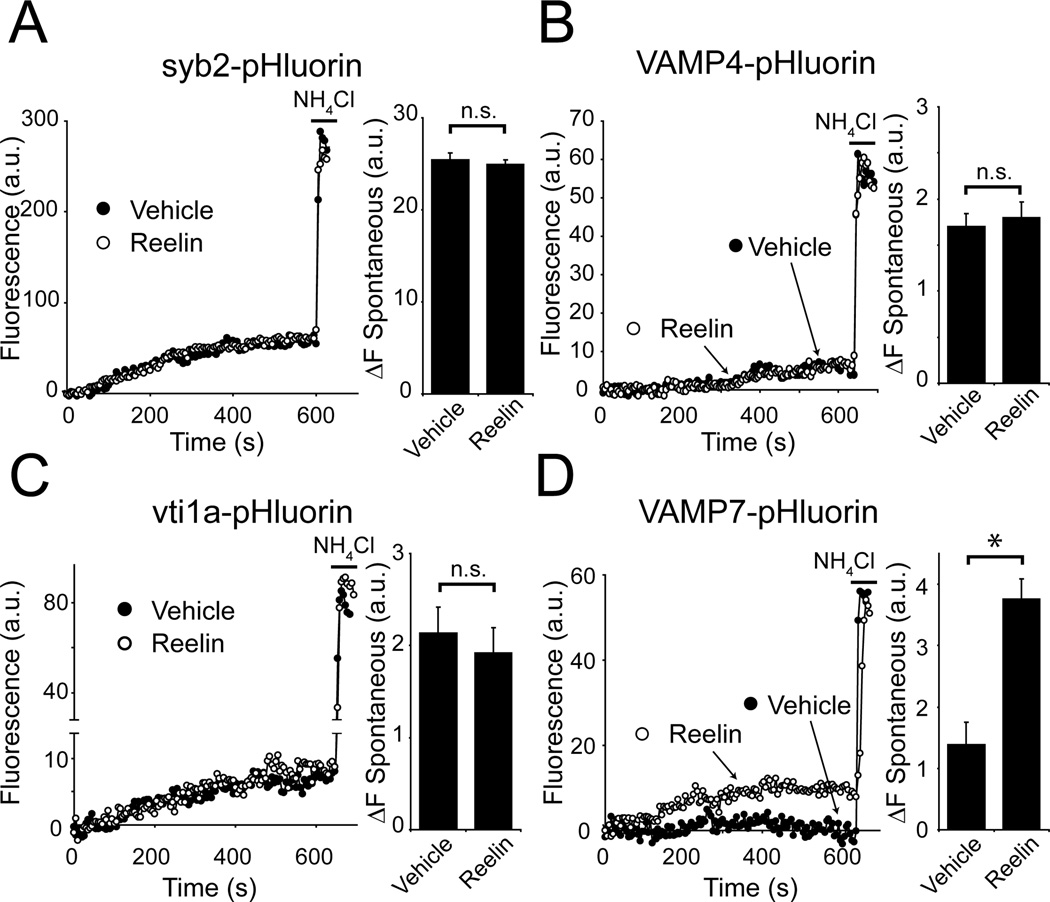
The ability of Reelin to increase spontaneous release in the absence of syb2 but not SNAP-25 suggests that the presynaptic Reelin effect requires an alternative vesicular SNARE. This observation is rather surprising as we detected a modest Reelin-dependent increase in presynaptic Ca2+ levels in presynaptic terminals identified via co-expression of syb2-mOrange in our Ca2+ imaging experiments (Figure 3M, N). These findings suggest that Reelin can signal to presynaptic terminals expressing syb2 but its effect on neurotransmitter release does not require syb2 function. To identify the alternative vesicular SNARE that mediates the observed Reelin-elicited exocytosis, we monitored the fluorescence of wild type neurons expressing one of four vesicular SNAREs (syb2, VAMP4, vti1a, or VAMP7) tagged with pHluorin at their C-terminal ends in the SV lumen. Using the same setting as in Figure 2G, we took advantage of the vacuolar ATPase inhibitor, folimycin, to prevent SV re-acidification at rest, and monitored spontaneous fusion of vesicles tagged with the four vesicular SNAREs. In this setting, we measured the increase in fluorescence after 10 minutes of Reelin application. Under these conditions, syb2-pHluorin (Figure 5A), VAMP4-pHluorin (Figure 5B) or vti1a-pHluorin (Figure 5C) trafficking did not respond to Reelin when compared to vehicle. Reelin did, however, robustly increase the cumulative rate of spontaneous fusion of vesicles labeled with VAMP7-pHluorin from 0.14 ± 0.04 a.u. per min. (in vehicle) to 0.38 ± 0.03 a.u. per min. (in Reelin) (Figure 5D). The rise in VAMP7-pHluorin fluorescence was in striking contrast to the very limited trafficking of VAMP7 under normal conditions compared to other vesicular SNAREs (Figure 5D). This finding is consistent with our earlier observations (Ramirez et al., 2012) as well as the earlier proposal that VAMP7 primarily resides within the resting SV pool (Hua et al., 2011). These results suggest that Reelin facilitates spontaneous neurotransmitter release specifically through mobilization of VAMP7-containing SVs.
Figure 5. Reelin selectively mobilizes a pool of vesicles tagged with VAMP7.
(A–D) Example traces of spontaneous pHluorin-tagged SV trafficking in 2 mM Ca2+ followed by application of NH4Cl is shown on left, and quantification of fluorescence change after 10 minutes in Vehicle or Reelin is shown on right.
(A) Reelin does not alter syb2-pHluorin trafficking (25.4 ± 0.7 a.u. after 10 minutes for Vehicle compared to 24.9 ± 0.5 a.u. in Reelin; Vehicle: n = 576 synapses, 5 experiments; Reelin: n = 689 synapses 5 experiments; p > 0.5).
(B) Reelin does not alter VAMP4-pHluorin trafficking (1.7 ± 0.1 a.u over 10 minutes in Vehicle compared to 1.8 ± 0.2 a.u in Reelin; Vehicle: n = 350 synapses, 4 experiments; Reelin: n = 466 synapses, 5 experiments; p > 0.5).
(C) Reelin does not affect vti1a-pHluorin spontaneous trafficking (2.1 ± 0.3 a.u. after 10 minutes in Vehicle compared to 1.9 ± 0.3 a.u. in Reelin; Vehicle: n = 340 synapses, 4 experiments; Reelin: n = 512 synapses, 5 experiments; p > 0.5).
(D) VAMP7-pHluroin trafficking is increased more than 2-fold in the presence of Reelin compared to Vehicle (1.4 ± 0.4 a.u. after 10 minutes in vehicle to 3.8 ± 0.3 a.u. in Reelin; Vehicle: n = 209 synapses, 7 experiments; Reelin: n = 263 synapses, 7 experiments; p < 0.001). The p-values reported were calculated by unpaired Student’s T-test. All error bars represent standard errors of the mean.
Reelin selectively acts on VAMP7 expressing SVs within an individual synaptic terminal
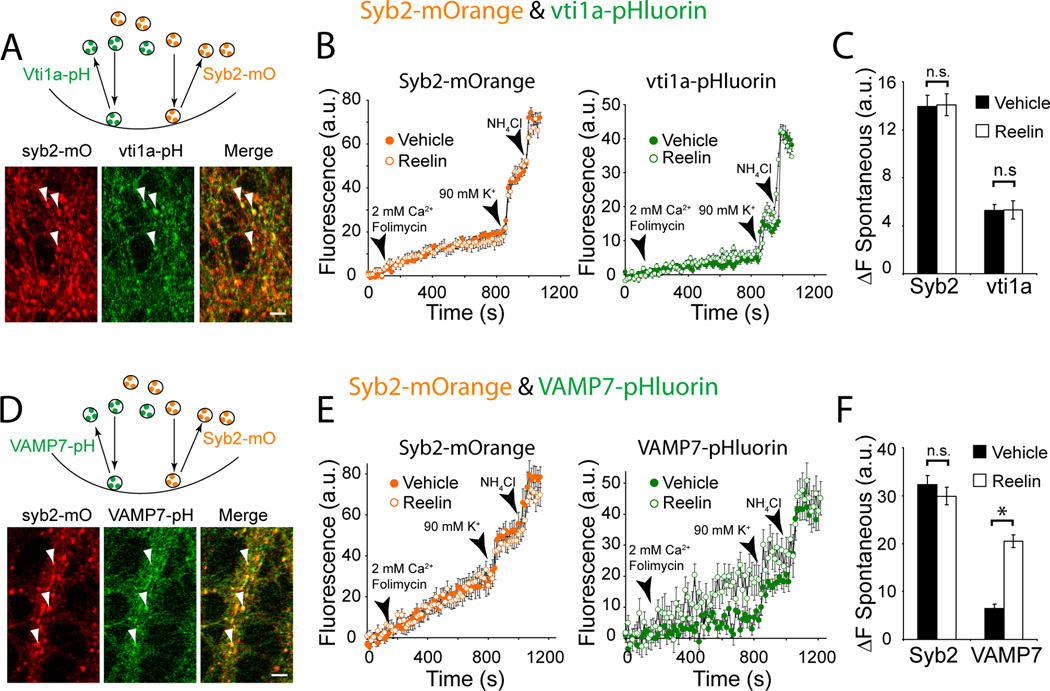
To evaluate the premise that in a single presynaptic terminal, VAMP7-containing SVs are selectively mobilized over those containing other vesicular SNAREs in response to Reelin, we utilized dual color imaging to compare relative vesicular SNARE trafficking within individual nerve terminals (Raingo et al., 2012; Ramirez et al., 2012). We co-infected cells with syb2-mOrange and either vti1a- or VAMP7-pHluorin. Synaptic boutons were then selected based on syb2-mOrange fluorescence and fluorescence changes in both channels were measured. Under these conditions, we detected significant fluorescent co-localization of syb2-mOrange and vti1a-pHluorin as well as syb2-mOrange and VAMP7-pHluorin (Figure 6A and D) (also see Ramirez et al., 2012). However, addition of Reelin did not alter syb2-mOrange or vti1a-pHluorin trafficking (Figure 6B and C). In contrast, Reelin could selectively increase the rate of trafficking of VAMP7-pHluorin-containing vesicles, while syb2-mOrange labeled vesicles in the same boutons were unaffected (Figure 6E and F). Collectively, these data demonstrate that within a given presynaptic terminal, Reelin selectively mobilizes a subset of VAMP7-containing SVs and leaves vesicles harboring other vesicular SNAREs relatively unaffected.
Figure 6. Dual color analysis of vesicular SNARE trafficking reveals that VAMP7 specifically responds to Reelin application.
(A) Neurons were co-infected with syb2-mOrange and vti1a-pHluorin. (Top) Cartoon depicting the segregation of these fluorescent signals to distinct SV pools. (Bottom) Co-localization of syb2-mOrange and vti1a-pHluorin fluorescence within individual synaptic boutons. Arrows indicate representative regions of interest selected for analysis. Scale bars indicate 5 µm.
(B) In synapses co-labeled with syb2-mOrange and vti1a-pHluorin, application of Reelin did not alter the trafficking of either vesicular SNARE compared to Vehicle application.
(C) In Vehicle syb2-mOrange fluorescence increased to 13.9 ± 0.9 a.u. after 10 minutes in Vehicle compared to 14.1 ± 0.9 a.u. after Reelin application (p > 0.5). In the case of vti1a-pHluorin, during Vehicle application fluorescence reached to 5.3 ± 0.5 a.u. after 10 minutes, whereas in Reelin vti1a-pHluorin fluorescence was leveled at 5.3 ± 0.8 a.u. (Vehicle: n =382 synapses, 5 experiments vs. Reelin: n = 351 synapses, 5 experiments; p > 0.5)
(D) Neurons were co-infected with syb2-mOrange and VAMP7-pHluorin. (Top) Cartoon depicting the segregation of these fluorescent signals to distinct SV pools. (Bottom) Co-localization of syb2-mOrange and VAMP7-pHluorin fluorescence within individual synaptic boutons. Arrows indicate representative regions of interest selected for analysis.
(E) Reelin selectively enhances VAMP7 trafficking in synapses that co-express syb2-mOrange and VAMP7-pHluorin.
(F) During application of Vehicle, syb2-mOrange fluorescence reached to 32.3 ± 1.9 a.u. in 10 minutes. Under the same condition, VAMP7-pHluorin fluorescence reached to 6.4 ± 0.9 a.u. after 10 minutes. In the presence of Reelin, however, syb2-mOrange fluorescence was relatively unaffected (29.9 ± 1.9 a.u. in 10 minutes; p > 0.2), whereas VAMP7-pHluorin fluorescence reached 20.5 ± 1.3 a.u. in 10 minutes (Vehicle: n = 108 synapses, 8 experiments; Reelin n = 77 synapses, 6 experiments; p<0.001). All error bars represent standard errors of the mean.
VAMP7 knockdown abolishes the effect of Reelin
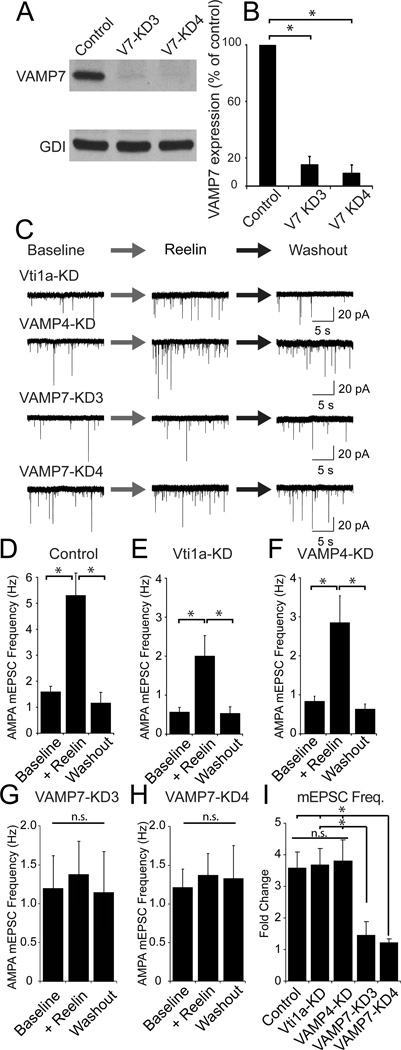
To further examine whether VAMP7 expression is indeed required for the effect of Reelin on spontaneous neurotransmitter release, we infected neurons with shRNA constructs directed against VAMP7, VAMP4 or vti1a (Figure 7, see also Supplementary Figure 6). In addition to the constructs against VAMP4 (Raingo et al 2011) and vti1a (Ramirez et al 2012) we reported earlier, we developed two VAMP7-knock down (KD) constructs (VAMP7-KD3 and VAMP7-KD4), which effectively reduced endogenous VAMP7 protein levels as detected by western blot (Figure 7A, B). Uninfected control neurons and neurons infected with the vti1a- and VAMP4-KD constructs still responded to Reelin (Figure 7C–F, I), while knockdown of VAMP7 using either the VAMP7-KD3 or VAMP7-KD4 construct abolished the effect of Reelin (Figure 7G–I). Moreover, insufficient knockdown of VAMP4 or vti1a did not attenuate the effect of Reelin on AMPA mEPSC frequency whereas cells with substantial amounts of VAMP7 expression remaining still exhibited increased AMPA-mEPSC frequency in the presence of Reelin (Supplementary Figure 6). Taken together, these results provide further support that Reelin mobilizes a specific subset of SVs containing VAMP7 to facilitate spontaneous neurotransmission.
Figure 7. VAMP7 knockdown is necessary and sufficient to abolish the effect of Reelin.
(A and B) Immunoblot and quantification of blots for the knockdown (KD) of VAMP7 (n = 4) (KD3 15.2 ± 5.9% of control (p < 0.01) KD4 is 9.3 ± 6.3% (p < 0.01)) in hippocampal neurons.
(C) Example traces of AMPAR mEPSCs from, vit1a-KD, VAMP4-KD and two different constructs of VAMP7-KD.
(D–H) Quantification of AMPAR mEPSCs data in control cells, and after knockdown of vit1a, VAMP4, and VAMP-7 using two different constructs (VAMP7-KD3 and VAMP7-KD4).
(D) In control cells, Reelin resulted in a large increase in spontaneous AMPAR mEPSC frequency (from 1.5 ± 0.2 Hz to 5.2 ± 0.8 Hz during reelin, returning to 1.1 ± 0.4 Hz after washout; p < 0.05) .
(E) Cells in which Vti1a was knocked down still responded to reelin (0.6 ± 0.1 Hz in control compared to 2.0 ± 0.5 Hz in the presence of Reelin and 0.50 ± 0.2 Hz after Reelin washout; p < 0.05).
(F) Similarly, VAMP-4 knockdown did not alter the Reelin response (0.8 ± 0.1 Hz before Reelin, 2.8 ± 0.7 Hz in the presence of Reelin and 0.6 ± 0.1 Hz after Reelin washout; p < 0.05).
(G and H) Cells infected with either VAMP-7 KD construct, -KD3 (G) or -KD4 (H) did not respond to Reelin (0.7 ± 0.2 Hz before Reelin to 0.9 ± 0.3 Hz in the presence of Reelin and 0.6 ± 0.2 after Reelin washout, and 1.3 ± 0.2 Hz prior to Reelin application to 1.5 ± 0.3 Hz during Reelin application and 1.7 ± 0.3 Hz after Reelin washout for VAMP7-KD3 and VAMP7-KD4, respectively (for all conditions in either VAMP7-KD construct, p > 0.2).
(I) AMPAR-mEPSC frequency calculated as fold change for Control (3.5 ± 0.5 fold, n = 7), Vti1a-KD (3.6 ± 0.5 fold, n = 8), VAMP4-KD (3.7 ± 0.7 fold, n = 10), VAMP7-KD3 (1.4 ± 0.4 fold, n = 7), and VAMP7-KD4 (1.1 ± 0.1 fold, n = 7) (p <0.05, one-way ANOVA; Holm-Sidak’s post hoc except the difference between Control and VAMP7-KD3 did not reach significance; p = 0.053). All error bars represent standard errors of the mean.
Discussion
In this study, we examined the presynaptic effect of the secreted glycoprotein Reelin and found that Reelin increases spontaneous neurotransmitter release from excitatory as well as inhibitory synaptic terminals without significantly altering the properties of evoked neurotransmission. This effect of Reelin is initiated by the ApoER2 and VLDLR signaling pathway(s) leading to activation of PI3 kinase and an increase in presynaptic Ca2+, via Ca2+-induced Ca2+ release. The Reelin-induced Ca2+ signal and the subsequent increase in SV fusion was widely distributed across synaptic boutons indicating that the presynaptic action of Reelin was not restricted to a small subpopulation of synapses. Although our results from synaptophysin-pHluorin trafficking (Figure 2G, H) and presynaptic Ca2+ imaging (Figure 3M –O) experiments indicate a robust effect of Reelin on the majority of synaptic boutons in our hippocampal cultures, the degree to which presynaptic Ca2+ levels increased after Reelin application varied across synapses examined (Figure 3N–O). This variability may suggest a heterogeneous ability to respond to Reelin across synaptic boutons. Such heterogeneity may also agree with the earlier work showing relative enrichment of VAMP7 expression in the mossy fiber terminals that originate from dentate granule cells (Scheuber et al., 2006).
The selective increase in spontaneous neurotransmitter release was dependent on the plasma membrane associated SNARE protein SNAP-25, consistent with an earlier study that proposed a SNAP-25 dependent role for Reelin in presynaptic function (Hellwig et al., 2011). Surprisingly, however, the effect of Reelin persisted in neurons deficient in syb2, which is the most abundant vesicular SNARE protein in the central nervous system (Schoch et al., 2001; Takamori et al., 2006). A functional survey of alternative SV associated SNAREs VAMP4, vti1a and VAMP7 (Hua et al., 2011; Raingo et al., 2012; Ramirez et al., 2012; Takamori et al., 2006) revealed that Reelin mediated signaling selectively targeted VAMP7 to augment spontaneous release. Dual color imaging at individual synaptic boutons showed that Reelin application could selectively mobilize vesicles tagged with VAMP7-pHluorin but spare vesicles tagged with syb2- or vti1a-pHluorin. Importantly, loss-of-function experiments showed that the Reelin mediated increase in spontaneous release was absent after shRNA mediated knockdown of VAMP7 (Figure 7 and Supplementary Figure 7). These results support the premise that low-level sustained increases in baseline presynaptic Ca2+ triggered by Reelin can selectively mobilize a subset of SVs that are dependent on VAMP7 for their exocytosis.
In central synapses, vesicles that reside within the resting pool, which normally do not respond to AP stimulation (Sudhof, 2000) but may contribute to spontaneous neurotransmitter release (Fredj and Burrone, 2009), are enriched in VAMP7 expression (Hua et al., 2011). The selectivity of Reelin action to VAMP7 suggests that sustained increases in baseline Ca2+ levels can specifically mobilize these dormant vesicles and augment spontaneous release. Previous studies have demonstrated that intramolecular binding of the N-terminal longin domain of VAMP7 with its SNARE motif can negatively regulate SNARE complex formation (Pryor et al., 2008). Accordingly, VAMP7-pHluorin lacking the longin domain has an increased rate of spontaneous exocytosis compared to full-length VAMP7 (Hua et al., 2011). Therefore, downstream components of the Reelin-mediated ApoER2 and VLDLR signaling cascade, such as PI3 kinase, may relieve VAMP7 autoinhibition by its longin domain to promote VAMP7-containing vesicles to a fusion competent state. Alternatively, increases in baseline Ca2+ levels may act to overcome autoinhibition of VAMP7 to activate VAMP7-mediated neurotransmission. This reaction may be transduced by a high affinity Ca2+ sensor, such as Doc2, that may selectively interact with VAMP7 (Groffen et al., 2010; Pang et al., 2011; Yao et al., 2011). However, our loss-of-function analysis failed to show an impairment in the Reelin-mediated increase in spontaneous neurotransmitter release in cells with reduced levels of all four isoforms of the Doc2 protein family (Doc2A, Doc2B, Doc2G, and rabphilin) (Supplementary Figure 7) (Pang et al., 2011). Our results cannot exclude a potential role for an unidentified alternative Ca2+ sensor in transducing slow elevations in baseline Ca2+ levels to activation of VAMP7-mediated SV fusion. Indeed, earlier experiments performed at the calyx of Held demonstrated that the increase in baseline vesicular release rate induced by modest rises in presynaptic Ca2+ (<400 nM) was relatively unaffected by the loss of synaptotagmin2, which is the primary Ca2+ sensor for evoked synchronous neurotransmitter release at this synapse (Sun et al., 2007). Therefore, selective association of VAMP7 driven SNARE complexes with an alternative Ca2+ sensor may explain the anomalous behavior of VAMP7 tagged SVs that do not effectively respond to individual APs and require strong stimulation for mobilization (Figure 6 and see Ramirez et al. 2012). However, most presynaptic terminals are expected to harbor multiple Ca2+ sensors that can mobilize vesicles in response to distinct forms of stimuli. Accordingly, in several preparations elevation of resting Ca2+ augments both spontaneous release and evoked responses to single APs (Awatramani et al. 2005) albeit via different signaling pathways (Virmani et al., 2005; Bouhours et al. 2011; Chu et al. 2012). These earlier results can be reconciled with our observations if Reelin-induced Ca2+ signals and subsequent mobilization of VAMP7-enriched vesicles are spatially sequestered within individual presynaptic terminals or selectively localized to a subset of synaptic terminals. We cannot currently exclude either of these scenarios, as the Reelin effect may be partly compartmentalized to synapses that are enriched in VAMP7 such as mossy fiber terminals (Scheuber et al., 2006; Hua et al., 2011), which are present in our culture preparation (Kavalali et al., 1999). Furthermore, it also remains possible that a downstream component of Reelin signaling besides Ca2+ may act negatively to suppress the elevation of evoked release probability.
These findings provide new insight into regulation of neurotransmitter release by identifying a physiological neuromodulatory cascade that selectively targets syb2-independent neurotransmission. In view of the abundance of syb2 in central presynaptic terminals, the physiological significance of the residual release remaining after genetic deletion of syb2 has remained elusive (Scales et al., 2001; Schoch et al., 2001). The finding that Reelin signaling can act independent of syb2 function to augment neurotransmitter release strongly suggests that the syb2-independent release is functionally significant. The residual spontaneous neurotransmission present in syb2-deficient synapses could not be explained by the expression of the closely related vesicular SNARE cellubrevin (also called VAMP3) (Deak et al., 2006; Schoch et al., 2001), although it appears to be partially mediated by vti1a, an alternative SNARE molecule localized to SVs (Ramirez et al., 2012). However, in this study, we show that vti1a-mediated neurotransmission is largely unresponsive to Reelin induced Ca2+ signaling, whereas VAMP7 tagged vesicles robustly respond to the same Ca2+ signal. These results are consistent with earlier work suggesting the segregation of vesicles containing vti1a to a spontaneously recycling pool and those containing VAMP7 to a resting pool (Hua et al., 2011; Ramirez et al., 2012). The molecular specificity of the actions of Reelin on VAMP7-containing vesicles offers a striking contrast to potentiation of neurotransmitter release in response to other secretagogues such as α-latrotoxin, which could elicit release independent of several key release machinery components including SNAP-25 (Deak et al., 2009) or lanthanides that specifically rely on syb2 to facilitate the rate of neurotransmitter release at rest (Chung et al., 2008).
The action of neuromodulators is critical in shaping the function of neuronal circuits in multiple species (Bargmann, 2012; Marder, 2012). Although several endogenous neuromodulators can modify all forms of neurotransmitter release, some factors can specifically target AP-independent forms of neurotransmission (Peters et al., 2010; Ramirez and Kavalali, 2011; Sharma and Vijayaraghavan, 2003; Vyleta and Smith, 2011; Zucker, 2003). The present findings indicate that the endogenous neuromodulator Reelin may selectively mobilize a subset of SVs by specifically targeting their SNARE machinery. Therefore, we propose that presynaptic vesicular SNARE heterogeneity forms a molecular substrate for selective neuromodulation and enables regulation of neuronal output without alterations in AP-dependent information processing. Such segregation of AP-dependent and AP-independent presynaptic regulatory mechanisms may be particularly significant in circumstances where evoked release probability is low (Borst, 2010) and the two types of synaptic signals are difficult to differentiate. Under these circumstances, selective augmentation of spontaneous neurotransmitter release may facilitate neurotrophic, homeostatic or other signaling functions of released neurotransmitter substances without compromising their function in AP-evoked information transfer. Recent studies suggest that the AP-independent forms of neurotransmitter release are critical in the regulation of behavioral outcomes such as nociception, memory processing and response to antidepressants (Andresen et al., 2012; Autry et al., 2011; Jin et al., 2012; Kavalali and Monteggia, 2012; Xu et al., 2012, Nosyreva et al., 2013). This premise is consistent with the recent behavioral analysis of VAMP7 knockout mice which revealed a deficit in anxiety-related behaviors (Danglot et al., 2012). In this way, identification of the vesicular release machineries and neuromodulators that specifically modify AP-independent forms of neurotransmission may provide novel avenues to manipulate neurotransmission without altering AP-dependent information processing. These types of approaches provide a promising strategy to uncover the functional roles of these unconventional forms of neuronal communication in the regulation of behavior in normal as well as in disease states.
Experimental Procedures
Cell culture and mouse lines
Dissociated hippocampal cultures were prepared from postnatal day 0–3 Sprague Dawley rats of either sex as described previously (Kavalali et al., 1999). For syb2 KO and SNAP-25 KO experiments, dissociated hippocampal cultures were prepared from embryonic day 18 mice constitutively deficient in syb2 (syb2 −/−) or SNAP-25 (SNAP-25 −/−) as well as their littermate controls (Schoch et al., 2001; Washbourne et al., 2002). ApoER2 KO and VLDLR KO cultures were prepared from mice generated by constitutive deletion of Apoer2 (Trommsdorff et al., 1999) and vldlr genes (Frykman et al., 1995). To generate neurons deficient in p110α and p110β isoforms of PI3K (gift of Drs. Joel Elmquist, UTSW and Jean Zhao, Dana-Farber Cancer Institute), hippocampal cultures from mice expressing conditionals alleles of p110α and p110β genes were infected with lentivirus expressing Cre. Lentiviral expression system shows high infection efficacy (>90%) as previously demonstrated by full rescue of synaptic transmission in syb2−/− cultures by lentiviral expression of syb2 (Deak et al., 2006). All experiments were performed on 14–21 days in vitro (DIV) cultures. All experiments were performed in accordance with protocols approved by the UT Southwestern Institutional Animal Care and Use Committee.
Lentiviral preparations
The short hairpin sequences used to knockdown the vesicular SNARE proteins are as follows: for Vti1a, sense 5’-tgtacagcaacagaatgagttcaagagactcattctgttgctgtaca-3’, VAMP4, sense 5’-tcgaggagaatattaccaaggtaattcaagagattaccttggtaatattctctta-3’. For VAMP7 knockdown, two constructs were used: VAMP7 KD3 5’-ctgaagcatcactccgagattcaagagatctcggagtgatgcttcag -3’ and VAMP7 KD4 5’-ctgaaaggcatcatggtcattcaagagatgaccatgatgcctttcag-3’. In all cases, shRNA sequences were inserted into Xhol through Xbal cloning sites in the L307 lentiviral transfer vector, downstream of the human H1 promoter. Lentiviruses encoding pHluorin-tagged syb2, VAMP4, vti1a, VAMP7, mOrange-tagged syb2, and all shRNA constructs were prepared by transfection of human embryonic kidney (HEK) 293-T cells with FUGENE 6 and necessary viral coat and packaging protein constructs (pVSVG, pRsv-Rev, and pPRE). Three days after transfection, virus was harvested from HEK 293-T cell-conditioned media and added to neuronal media at 4 DIV. Lentiviral constructs to decrease expression of all four isoforms of the Doc2 protein family (Doc2A, Doc2B, Doc2G, and rabphilin) were a gift of Dr. Thomas C. Südhof (Stanford Univ.) (Pang et al., 2011).
Electrophysiology
Electrophysiological experiments were performed on dissociated hippocampal neurons as in (Nosyreva and Kavalali, 2010) (see Supplemental Experimental Procedures for further details).
Reelin and pharmacology
Reelin was prepared and purified as described previously (Beffert et al., 2002) (see Supplemental Experimental Procedures for further details).
Single color imaging
Single-wavelength experiments were performed using an Andor iXon+ back-illuminated EMCCD camera using MetaFluor 7.6 software as described previously (Leitz and Kavalali, 2011) (see Supplemental Experimental Procedures for further details).
Dual color imaging
Dual color experiments were performed using a Zeiss LSM510 confocal microscope using LSM5 software as described previously (Ramirez et al., 2012; Raingo et al., 2012) (see Supplemental Experimental Procedures for further details).
Statistical analysis
All error bars represent standard errors of the mean (S.E.M) and all statistical analyses were done using Microsoft Excel Software or Sigma Plot (see Supplemental Experimental Procedures for further details).
Supplementary Material
Highlights.
Reelin selectively augments spontaneous synaptic transmission
The effect of Reelin requires ApoER2 and VLDLR activation, PI3K activity and Ca2+
Reelin selectively acts on VAMP7 containing synaptic vesicles within a synapse
VAMP7 knockdown abolishes the effect of Reelin
Acknowledgements
We thank Drs. Megumi Adachi, Elena Nosyreva, Catherine Wasser for discussions and comments on the manuscript. We also thank Brent Trauterman for his excellent technical assistance. We are grateful to Drs. Mikhail Khvotchev and Yildirim Sara for their insight at early stages of this project. This work was supported by NIH grants MH066198 (ETK), NS075499 (ETK), T32 NS069562 (JL), MH070727 (LMM) and HL063762 (JH) as well as funding from the from the Brain & Behavior Research Foundation (LMM and ETK) and International Mental Health Research Organization (LMM).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Alabi AA, Tsien RW. Synaptic vesicle pools and dynamics. Cold Spring Harbor perspectives in biology. 2012;4:a013680. doi: 10.1101/cshperspect.a013680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alcantara S, Ruiz M, D'Arcangelo G, Ezan F, de Lecea L, Curran T, Sotelo C, Soriano E. Regional and cellular patterns of reelin mRNA expression in the forebrain of the developing and adult mouse. J Neurosci. 1998;18:7779–7799. doi: 10.1523/JNEUROSCI.18-19-07779.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andresen MC, Hofmann ME, Fawley JA. Invited Review: The un-silent majority - TRPV1 drives "spontaneous" transmission of unmyelinated primary afferents within cardiorespiratory NTS. Am J Physiol Regul Integr Comp Physiol. 2012;303:R1207–R1216. doi: 10.1152/ajpregu.00398.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atasoy D, Ertunc M, Moulder KL, Blackwell J, Chung C, Su J, Kavalali ET. Spontaneous and evoked glutamate release activates two populations of NMDA receptors with limited overlap. J Neurosci. 2008;28:10151–10166. doi: 10.1523/JNEUROSCI.2432-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Autry AE, Adachi M, Nosyreva E, Na ES, Los MF, Cheng PF, Kavalali ET, Monteggia LM. NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature. 2011;475:91–95. doi: 10.1038/nature10130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Awatramani GB, Price GD, Trussell LO. Modulation of transmitter release by presynaptic resting potential and background calcium levels. Neuron. 2005;48:109–121. doi: 10.1016/j.neuron.2005.08.038. [DOI] [PubMed] [Google Scholar]
- Bargmann CI. Beyond the connectome: how neuromodulators shape neural circuits. Bioessays. 2012;34:458–465. doi: 10.1002/bies.201100185. [DOI] [PubMed] [Google Scholar]
- Beffert U, Morfini G, Bock HH, Reyna H, Brady ST, Herz J. Reelin-mediated signaling locally regulates protein kinase B/Akt and glycogen synthase kinase 3beta. The Journal of biological chemistry. 2002;277:49958–49964. doi: 10.1074/jbc.M209205200. [DOI] [PubMed] [Google Scholar]
- Beffert U, Weeber EJ, Durudas A, Qiu S, Masiulis I, Sweatt JD, Li WP, Adelmann G, Frotscher M, Hammer RE, et al. Modulation of synaptic plasticity and memory by Reelin involves differential splicing of the lipoprotein receptor Apoer2. Neuron. 2005;47:567–579. doi: 10.1016/j.neuron.2005.07.007. [DOI] [PubMed] [Google Scholar]
- Bezzerides VJ, Ramsey IS, Kotecha S, Greka A, Clapham DE. Rapid vesicular translocation and insertion of TRP channels. Nature cell biology. 2004;6:709–720. doi: 10.1038/ncb1150. [DOI] [PubMed] [Google Scholar]
- Bock HH, Jossin Y, Liu P, Förster E, May P, Goffinet AM, Herz J. Phosphatidylinositol 3-kinase interacts with the adaptor protein Dab1 in response to Reelin signaling and is required for normal cortical lamination. J Biol Chem. 2003a;278:38772–38779. doi: 10.1074/jbc.M306416200. [DOI] [PubMed] [Google Scholar]
- Bock HH, Herz J. Reelin activates SRC family tyrosine kinases in neurons. Curr Biol. 2003b;13:18–26. doi: 10.1016/s0960-9822(02)01403-3. [DOI] [PubMed] [Google Scholar]
- Borst JG. The low synaptic release probability in vivo. Trends in neurosciences. 2010;33:259–266. doi: 10.1016/j.tins.2010.03.003. [DOI] [PubMed] [Google Scholar]
- Bouhours B, Trigo FF, Marty A. Somatic depolarization enhances GABA release in cerebellar interneurons via a calcium/protein kinase C pathway. J Neurosci. 2011;31:5804–5815. doi: 10.1523/JNEUROSCI.5127-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bronk P, Deak F, Wilson MC, Liu X, Sudhof TC, Kavalali ET. Differential effects of SNAP-25 deletion on Ca2+-dependent and Ca2+-independent neurotransmission. Journal of neurophysiology. 2007;98:794–806. doi: 10.1152/jn.00226.2007. [DOI] [PubMed] [Google Scholar]
- Chen Y, Beffert U, Ertunc M, Tang TS, Kavalali ET, Bezprozvanny I, Herz J. Reelin modulates NMDA receptor activity in cortical neurons. J Neurosci. 2005;25:8209–8216. doi: 10.1523/JNEUROSCI.1951-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu Y, Fioravante D, Thanawala M, Leitges M, Regehr WG. Calcium-dependent isoforms of protein kinase C mediate glycine-induced synaptic enhancement at the calyx of Held. J Neurosci. 2012;32:13796–13804. doi: 10.1523/JNEUROSCI.2158-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung C, Barylko B, Leitz J, Liu X, Kavalali ET. Acute dynamin inhibition dissects synaptic vesicle recycling pathways that drive spontaneous and evoked neurotransmission. J Neurosci. 2010;30:1363–1376. doi: 10.1523/JNEUROSCI.3427-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung C, Deak F, Kavalali ET. Molecular substrates mediating lanthanide-evoked neurotransmitter release in central synapses. Journal of neurophysiology. 2008;100:2089–2100. doi: 10.1152/jn.90404.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Arcangelo G, Miao GG, Chen SC, Soares HD, Morgan JI, Curran T. A protein related to extracellular matrix proteins deleted in the mouse mutant reeler. Nature. 1995;374:719–723. doi: 10.1038/374719a0. [DOI] [PubMed] [Google Scholar]
- Danglot L, Zylbersztejn K, Petkovic M, Gauberti M, Meziane H, Combe R, Champy MF, Birling MC, Pavlovic G, Bizot JC, et al. Absence of TI-VAMP/Vamp7 leads to increased anxiety in mice. J Neurosci. 2012;32:1962–1968. doi: 10.1523/JNEUROSCI.4436-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deak F, Liu X, Khvotchev M, Li G, Kavalali ET, Sugita S, Sudhof TC. Alpha-latrotoxin stimulates a novel pathway of Ca2+-dependent synaptic exocytosis independent of the classical synaptic fusion machinery. J Neurosci. 2009;29:8639–8648. doi: 10.1523/JNEUROSCI.0898-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deak F, Shin OH, Kavalali ET, Sudhof TC. Structural determinants of synaptobrevin 2 function in synaptic vesicle fusion. J Neurosci. 2006;26:6668–6676. doi: 10.1523/JNEUROSCI.5272-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fredj NB, Burrone J. A resting pool of vesicles is responsible for spontaneous vesicle fusion at the synapse. Nature neuroscience. 2009;12:751–758. doi: 10.1038/nn.2317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frykman PK, Brown MS, Yamamoto T, Goldstein JL, Herz J. Normal plasma lipoproteins and fertility in gene-targeted mice homozygous for a disruption in the gene encoding very low density lipoprotein receptor. Proc Natl Acad Sci U S A. 1995;92:8453–8457. doi: 10.1073/pnas.92.18.8453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groffen AJ, Martens S, Diez Arazola R, Cornelisse LN, Lozovaya N, de Jong AP, Goriounova NA, Habets RL, Takai Y, Borst JG, et al. Doc2b is a high-affinity Ca2+ sensor for spontaneous neurotransmitter release. Science. 2010;327:1614–1618. doi: 10.1126/science.1183765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hellwig S, Hack I, Kowalski J, Brunne B, Jarowyj J, Unger A, Bock HH, Junghans D, Frotscher M. Role for Reelin in neurotransmitter release. J Neurosci. 2011;31:2352–2360. doi: 10.1523/JNEUROSCI.3984-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herz J, Goldstein JL, Strickland DK, Ho YK, Brown MS. 39-kDa protein modulates binding of ligands to low density lipoprotein receptor-related protein/alpha 2-macroglobulin receptor. J Biol Chem. 1991;266:21232–21238. [PubMed] [Google Scholar]
- Herz J, Chen Y. Reelin, lipoprotein receptors and synaptic plasticity. Nature reviews neuroscience. 2006;7:850–859. doi: 10.1038/nrn2009. [DOI] [PubMed] [Google Scholar]
- Hiesberger T, Trommsdorff M, Howell BW, Goffinet A, Mumby MC, Cooper JA, Herz J. Direct binding of Reelin to VLDL receptor and ApoE receptor 2 induces tyrosine phosphorylation of disabled-1 and modulates tau phosphorylation. Neuron. 1999;24:481–489. doi: 10.1016/s0896-6273(00)80861-2. [DOI] [PubMed] [Google Scholar]
- Hua Z, Leal-Ortiz S, Foss SM, Waites CL, Garner CC, Voglmaier SM, Edwards RH. v-SNARE composition distinguishes synaptic vesicle pools. Neuron. 2011;71:474–487. doi: 10.1016/j.neuron.2011.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin I, Puthanveettil S, Udo H, Karl K, Kandel ER, Hawkins RD. Spontaneous transmitter release is critical for the induction of long-term and intermediate-term facilitation in Aplysia. Proceedings of the National Academy of Sciences of the United States of America. 2012;109:9131–9136. doi: 10.1073/pnas.1206914109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kavalali ET, Klingauf J, Tsien RW. Activity-dependent regulation of synaptic clustering in a hippocampal culture system. Proceedings of the National Academy of Sciences of the United States of America. 1999;96:12893–12900. doi: 10.1073/pnas.96.22.12893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kavalali ET, Monteggia LM. Synaptic Mechanisms Underlying Rapid Antidepressant Action of Ketamine. The American Journal of Psychiatry. 2012;169:1150–1156. doi: 10.1176/appi.ajp.2012.12040531. [DOI] [PubMed] [Google Scholar]
- Knuesel I. Reelin-mediated signaling in neuropsychiatric and neurodegenerative diseases. Progress in neurobiology. 2010;91:257–274. doi: 10.1016/j.pneurobio.2010.04.002. [DOI] [PubMed] [Google Scholar]
- Kubo K, Mikoshiba K, Nakajima K. Secreted Reelin molecules form homodimers. Neuroscience research. 2002;43:381–388. doi: 10.1016/s0168-0102(02)00068-8. [DOI] [PubMed] [Google Scholar]
- Kuo G, Arnaud L, Kronstad-O'Brien P, Cooper JA. Absence of Fyn and Src causes a reeler-like phenotype. J Neurosci. 2005;25:8578–8586. doi: 10.1523/JNEUROSCI.1656-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon SE, Chapman ER. Synaptophysin regulates the kinetics of synaptic vesicle endocytosis in central neurons. Neuron. 2011;70:847–854. doi: 10.1016/j.neuron.2011.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leitz J, Kavalali ET. Ca2+ influx slows single synaptic vesicle endocytosis. J Neurosci. 2011;31:16318–16326. doi: 10.1523/JNEUROSCI.3358-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lou X, Scheuss V, Schneggenburger R. Allosteric modulation of the presynaptic Ca2+ sensor for vesicle fusion. Nature. 2005;435:497–501. doi: 10.1038/nature03568. [DOI] [PubMed] [Google Scholar]
- Marder E. Neuromodulation of neuronal circuits: back to the future. Neuron. 2012;76:1–11. doi: 10.1016/j.neuron.2012.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mozhayeva MG, Sara Y, Liu X, Kavalali ET. Development of vesicle pools during maturation of hippocampal synapses. J Neurosci. 2002;22:654–665. doi: 10.1523/JNEUROSCI.22-03-00654.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nosyreva E, Kavalali ET. Activity-dependent augmentation of spontaneous neurotransmission during endoplasmic reticulum stress. J Neurosci. 2010;30:7358–7368. doi: 10.1523/JNEUROSCI.5358-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nosyreva E, Szabla K, Autry AE, Ryazanov AG, Monteggia LM, Kavalali ET. Acute suppression of spontaneous neurotransmission drives synaptic potentiation. J Neurosci. 2013;33:6990–7002. doi: 10.1523/JNEUROSCI.4998-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pang ZP, Bacaj T, Yang X, Zhou P, Xu W, Sudhof TC. Doc2 supports spontaneous synaptic transmission by a Ca(2+)-independent mechanism. Neuron. 2011;70:244–251. doi: 10.1016/j.neuron.2011.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pesold C, Impagnatiello F, Pisu MG, Uzunov DP, Costa E, Guidotti A, Caruncho HJ. Reelin is preferentially expressed in neurons synthesizing gamma-aminobutyric acid in cortex and hippocampus of adult rats. Proc Natl Acad Sci U S A. 1998;95:3221–3226. doi: 10.1073/pnas.95.6.3221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters JH, McDougall SJ, Fawley JA, Smith SM, Andresen MC. Primary afferent activation of thermosensitive TRPV1 triggers asynchronous glutamate release at central neurons. Neuron. 2010;65:657–669. doi: 10.1016/j.neuron.2010.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phillips CG, Harnett MT, Chen W, Smith SM. Calcium-sensing receptor activation depresses synaptic transmission. J Neurosci. 2008;28:12062–12070. doi: 10.1523/JNEUROSCI.4134-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pratt KG, Zhu P, Watari H, Cook DG, Sullivan JM. A novel role for {gamma}-secretase: selective regulation of spontaneous neurotransmitter release from hippocampal neurons. J Neurosci. 2011;31:899–906. doi: 10.1523/JNEUROSCI.4625-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pryor PR, Jackson L, Gray SR, Edeling MA, Thompson A, Sanderson CM, Evans PR, Owen DJ, Luzio JP. Molecular basis for the sorting of the SNARE VAMP7 into endocytic clathrin-coated vesicles by the ArfGAP Hrb. Cell. 2008;134:817–827. doi: 10.1016/j.cell.2008.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qiu S, Zhao LF, Korwek KM, Weeber EJ. Differential reelin-induced enhancement of NMDA and AMPA receptor activity in the adult hippocampus. J Neurosci. 2006;26:12943–12955. doi: 10.1523/JNEUROSCI.2561-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raingo J, Khvotchev M, Liu P, Darios F, Li YC, Ramirez DM, Adachi M, Lemieux P, Toth K, Davletov B, et al. VAMP4 directs synaptic vesicles to a pool that selectively maintains asynchronous neurotransmission. Nature neuroscience. 2012;15:738–745. doi: 10.1038/nn.3067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramirez DM, Kavalali ET. Differential regulation of spontaneous and evoked neurotransmitter release at central synapses. Current opinion in neurobiology. 2011;21:275–282. doi: 10.1016/j.conb.2011.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramirez DM, Kavalali ET. The role of non-canonical SNAREs in synaptic vesicle recycling. Cellular logistics. 2012;2:20–27. doi: 10.4161/cl.20114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramirez DM, Khvotchev M, Trauterman B, Kavalali ET. Vti1a identifies a vesicle pool that preferentially recycles at rest and maintains spontaneous neurotransmission. Neuron. 2012;73:121–134. doi: 10.1016/j.neuron.2011.10.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sara Y, Virmani T, Deak F, Liu X, Kavalali ET. An isolated pool of vesicles recycles at rest and drives spontaneous neurotransmission. Neuron. 2005;45:563–573. doi: 10.1016/j.neuron.2004.12.056. [DOI] [PubMed] [Google Scholar]
- Scales SJ, Finley MF, Scheller RH. Cell biology. Fusion without SNAREs? Science. 2001;294:1015–1016. doi: 10.1126/science.1066728. [DOI] [PubMed] [Google Scholar]
- Scheuber A, Rudge R, Danglot L, Raposo G, Binz T, Poncer JC, Galli T. Loss of AP-3 function affects spontaneous and evoked release at hippocampal mossy fiber synapses. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:16562–16567. doi: 10.1073/pnas.0603511103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoch S, Deak F, Konigstorfer A, Mozhayeva M, Sara Y, Sudhof TC, Kavalali ET. SNARE function analyzed in synaptobrevin/VAMP knockout mice. Science. 2001;294:1117–1122. doi: 10.1126/science.1064335. [DOI] [PubMed] [Google Scholar]
- Sharma G, Vijayaraghavan S. Modulation of presynaptic store calcium induces release of glutamate and postsynaptic firing. Neuron. 2003;38:929–939. doi: 10.1016/s0896-6273(03)00322-2. [DOI] [PubMed] [Google Scholar]
- Soriano E, Del Rio JA. The cells of cajal-retzius: still a mystery one century after. Neuron. 2005;46:389–394. doi: 10.1016/j.neuron.2005.04.019. [DOI] [PubMed] [Google Scholar]
- Sudhof TC. The synaptic vesicle cycle revisited. Neuron. 2000;28:317–320. doi: 10.1016/s0896-6273(00)00109-4. [DOI] [PubMed] [Google Scholar]
- Sudhof TC, Rothman JE. Membrane fusion: grappling with SNARE and SM proteins. Science. 2009;323:474–477. doi: 10.1126/science.1161748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun J, Pang ZP, Qin D, Fahim AT, Adachi R, Südhof TC. A dual-Ca2+-sensor model for neurotransmitter release in a central synapse. Nature. 2007;450:676–682. doi: 10.1038/nature06308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takamori S, Holt M, Stenius K, Lemke EA, Gronborg M, Riedel D, Urlaub H, Schenck S, Brugger B, Ringler P, et al. Molecular anatomy of a trafficking organelle. Cell. 2006;127:831–846. doi: 10.1016/j.cell.2006.10.030. [DOI] [PubMed] [Google Scholar]
- Tissir F, Goffinet AM. Reelin and brain development. Nat Rev Neurosci. 2003;4:496–505. doi: 10.1038/nrn1113. [DOI] [PubMed] [Google Scholar]
- Trommsdorff M, Gotthardt M, Hiesberger T, Shelton J, Stockinger W, Nimpf J, Hammer RE, Richardson JA, Herz J. Reeler/Disabled-like disruption of neuronal migration in knockout mice lacking the VLDL receptor and ApoE receptor 2. Cell. 1999;97:689–701. doi: 10.1016/s0092-8674(00)80782-5. [DOI] [PubMed] [Google Scholar]
- Utermark T, Rao T, Cheng H, Wang Q, Lee SH, Wang ZC, Iglehart JD, Roberts TM, Muller WJ, Zhao JJ. The p110α and p110β isoforms of PI3K play divergent roles in mammary gland development and tumorigenesis. Genes Dev. 2012;26:1573–1586. doi: 10.1101/gad.191973.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Virmani T, Ertunc M, Sara Y, Mozhayeva M, Kavalali ET. Phorbol esters target the activity-dependent recycling pool and spare spontaneous vesicle recycling. J Neurosci. 2005;25:10922–10929. doi: 10.1523/JNEUROSCI.3766-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vyleta NP, Smith SM. Spontaneous glutamate release is independent of calcium influx and tonically activated by the calcium-sensing receptor. J Neurosci. 2011;31:4593–4606. doi: 10.1523/JNEUROSCI.6398-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Washbourne P, Thompson PM, Carta M, Costa ET, Mathews JR, Lopez-Bendito G, Molnar Z, Becher MW, Valenzuela CF, Partridge LD, et al. Genetic ablation of the t-SNARE SNAP-25 distinguishes mechanisms of neuroexocytosis. Nature neuroscience. 2002;5:19–26. doi: 10.1038/nn783. [DOI] [PubMed] [Google Scholar]
- Williams KW, Sohn JW, Donato J, Jr, Lee CE, Zhao JJ, Elmquist JK, Elias CF. The acute effects of leptin require PI3K signaling in the hypothalamic ventral premammillary nucleus. J Neurosci. 2011;31:13147–13156. doi: 10.1523/JNEUROSCI.2602-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J, Pang ZP, Shin OH, Sudhof TC. Synaptotagmin-1 functions as a Ca2+ sensor for spontaneous release. Nature neuroscience. 2009;12:759–766. doi: 10.1038/nn.2320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu W, Morishita W, Buckmaster PS, Pang ZP, Malenka RC, Sudhof TC. Distinct neuronal coding schemes in memory revealed by selective erasure of fast synchronous synaptic transmission. Neuron. 2012;73:990–1001. doi: 10.1016/j.neuron.2011.12.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao J, Gaffaney JD, Kwon SE, Chapman ER. Doc2 is a Ca2+ sensor required for asynchronous neurotransmitter release. Cell. 2011;147:666–677. doi: 10.1016/j.cell.2011.09.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zucker RS. Can a synaptic signal arise from noise? Neuron. 2003;38:845–846. doi: 10.1016/s0896-6273(03)00363-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.