Abstract
Background
Heart failure (HF) is a leading cause of hospitalization and mortality. Plasma B-type natriuretic peptide (BNP) is an established diagnostic and prognostic ambulatory HF biomarker. We hypothesized that increased perioperative BNP independently associates with HF hospitalization or HF death up to 5 yr after coronary artery bypass graft surgery.
Methods
The authors conducted a two-institution, prospective, observational study of 1,025 subjects (mean age = 64 ± 10 yr SD) undergoing isolated primary coronary artery bypass graft surgery with cardiopulmonary bypass. Plasma BNP was measured preoperatively and on postoperative days 1–5. The study outcome was hospitalization or death from HF, with HF events confirmed by reviewing hospital and death records. Cox proportional hazards analyses were performed with multivariable adjustments for clinical risk factors. Preoperative and peak postoperative BNP were added to the multivariable clinical model in order to assess additional predictive benefit.
Results
One hundred five subjects experienced an HF event (median time to first event = 1.1 yr). Median follow-up for subjects who did not have an HF event = 4.2 yr. When individually added to the multivariable clinical model, higher preoperative and peak postoperative BNP concentrations each, independently associated with the HF outcome (log10 preoperative BNP hazard ratio = 1.93; 95% CI, 1.30–2.88; P = 0.001; log10 peak postoperative BNP hazard ratio = 3.38; 95% CI, 1.45–7.65; P = 0.003).
Conclusions
Increased perioperative BNP concentrations independently associate with HF hospitalization or HF death during the 5 yr after primary coronary artery bypass graft surgery. Clinical trials may be warranted to assess whether medical management focused on reducing preoperative and longitudinal postoperative BNP concentrations associates with decreased HF after coronary artery bypass graft surgery.
Heart failure (HF) is a major cause of hospitalization, poor health-related quality of life (HRQL), mortality, and healthcare expense.1,2 In the United States alone, 5.7 million people suffer from HF, with coronary artery disease known to be a major HF risk factor.1,3,4 Plasma B-type natriuretic peptide (BNP) is an established diagnostic and prognostic biomarker in ambulatory HF and acute coronary syndrome patients. BNP is secreted primarily by cardiac ventricular myocytes in response to ventricular pressure and volume overload and ischemia.5–10 Several studies of ambulatory chronic HF patients suggest that medical management designed to reduce increased plasma BNP (active fragment) or N-terminal-proBNP (NT-proBNP; inactive fragment) concentrations may reduce associated adverse cardiovascular events11–14 and patient care costs,15 improve HF hospitalization-free survival13,16,17 and HRQL,13 and have favorable effects on cardiac remodeling.13
Each year almost a quarter of a million Americans undergo coronary artery bypass graft (CABG) surgery.1 Guidelines from the American College of Cardiology and the American Heart Association state that CABG surgery is indicated not only to improve survival of coronary artery disease patients, but also to enhance longer-term postoperative HRQL.18 Particularly with advances in percutaneous coronary interventions and medical management of coronary artery disease delaying need for CABG surgery until progressively later in life, improving postoperative HRQL is arguably as important as postoperative survival. Although many CABG surgical patients experience improved postoperative HRQL, 7–24% of CABG patients experience significant declines in HRQL during the years after surgery.19–22 Thus, there remains an important need to identify modifiable perioperative risk factors that strongly associate with the postoperative outcomes that most adversely impact both HRQL and survival after CABG surgery. Existing models for perioperative risk stratification are modestly useful for predicting all-cause mortality or composite morbidity outcomes after CABG surgery.23–25 However, predicting all-cause mortality or composite morbidity outcomes does little to guide tailored medical interventions targeted to prevent and treat specific outcomes, such as long-term postoperative HF, an outcome that has substantial potential impact on both long-term HRQL and survival after CABG surgery.
We have previously demonstrated that increased preoperative and peak postoperative BNP independently associate with longer postoperative hospital stays and early cardiac ventricular dysfunction, as well as longer-term decreased physical functioning and all-cause mortality after primary CABG surgery.26–28 However, the association between increased perioperative BNP and development of significant longer-term HF after CABG surgery has not been assessed. We, therefore, conducted this study to evaluate the hypothesis that even after adjusting for important clinical risk factors, increased preoperative and peak postoperative plasma BNP associate with shorter postoperative time to first HF event (hospitalization or death) within the 5 yr after primary CABG surgery.
Materials and Methods
Study Population
Between August 2001 and September 2006, 1,519 patients (age 20–89 yr), scheduled for CABG surgery with cardiopulmonary bypass at either Brigham and Women’s Hospital, Boston, Massachusetts, or the Texas Heart Institute, Saint Luke’s Episcopal Hospital, Houston, Texas, were enrolled prospectively into an ongoing study called the CABG Genomics Program.††27 Before subject enrollment, the respective institutional review board approvals were obtained and all subjects provided written informed consent. Exclusion criteria for enrollment into the CABG Genomics Program include inability to give written informed consent, receiving leukocyte-rich blood product transfusion within 30 days before surgery, or having a preoperative hematocrit less than 25%. CABG Genomics Program subjects were then prospectively excluded from the current study if they had one or more of the following criteria: emergency surgery; concurrent valve surgery; previous cardiac surgery; preoperative intraaortic balloon pump, ventricular assist device or inotrope support; CABG surgery without cardiopulmonary bypass or without an aortic cross-clamp; preoperative hemodialysis or serum creatinine more than 3 mg/dl (severe renal dysfunction and perioperative dialysis can variably influence plasma BNP concentrations);29,30 or missing preoperative or peak postoperative plasma BNP measurements. Of the 1,183 subjects eligible for study analysis, 158 subjects (13.4%) were additionally excluded from analysis because they could not be contacted for follow-up after discharge from their primary surgical hospitalization. Seventy-seven percent of the subjects analyzed were enrolled at Brigham and Women’s Hospital and 23% were enrolled at the Texas Heart Institute.
Data and Blood Collection
Data were prospectively collected for each subject during primary CABG surgery hospitalization using a detailed, standardized case report form. This subject data included information regarding: (1) demographics; (2) preoperative medications, medical history, and co-morbidities; (3) surgical events and characteristics; and (4) in-hospital postoperative course.
The follow-up period for this study was through 5 yr after CABG surgery or until this study’s end date (May 7, 2009). Postoperative hospitalizations were identified using subjects’ responses to mailed questionnaires (6 week, 6 month, and annually for years 1–5 after surgery) and to telephone interviews, and through review of the electronic medical records systems available at the enrolling institutions. Hospital records were obtained for all subjects who were identified to have had postoperative hospitalizations requiring overnight stays for probable or possible cardiac or pulmonary reasons. Subjects who died during study follow-up were identified by responses to mailed questionnaires (family, friend, caregiver responses) or telephone interviews and by using the Social Security Death Index. Death certificates and/or National Death Index cause of death information was obtained for all subjects identified to have died during the study follow-up period. Postoperative HF hospitalization or death outcomes were identified by two investigators, who reviewed hospital records (Drs. Fox and Nascimben) and by two investigators, who reviewed death records (Drs. Fox and Muehlschlegel). If between-investigator discrepancies occurred when identifying an HF event, a third investigator adjudicated identification of an HF event. Investigators were blinded to BNP data when reviewing hospitalization and death records.
Plasma samples obtained preoperatively and daily on postoperative days 1–5 were stored in vapor-phase liquid nitrogen until analysis. BNP and cardiac troponin I were measured for all samples at a single-core laboratory using sandwich immunoassays (Biosite Triage®, San Diego, CA). These biomarker assessments were performed in a batch research analysis and were not available for clinical care of the subjects.
Definitions
The study outcome was defined prospectively as time to first HF hospitalization or HF death (if a preceding HF hospitalization was not identified) occurring up to 5 yr after primary CABG surgery. Although HF severity can be difficult to adjudicate in ambulatory populations, we felt that HF resulting in hospitalization or death could be well adjudicated as a study outcome. Five years was prospectively chosen as the maximum interval for postoperative follow-up based on consensus among investigators that even elderly subjects in the study cohort should reasonably expect enhanced HRQL and survival during this postoperative timeframe. Investigators developed criteria defining HF hospitalization and HF death before reviewing hospitalization and death records (Supplemental Digital Content 1, Methods, http://links.lww.com/ALN/A945).
Peak postoperative BNP was defined as the highest BNP measures of postoperative days 1–5, with all analyzed subjects having successful BNP measurements for at least 3 postoperative days. Previously, we reported that plasma BNP concentrations tend to increase significantly during the first 3 days after primary CABG surgery and then plateau or begin to decline on postoperative days 4 and 5.26,27 Therefore, even for subjects discharged on postoperative day 4, assessing the peak of at least three postoperative BNP assessments should closely approximate the peak BNP concentration during the first 5 postoperative days.
Postoperative creatinine clearance was estimated using the highest creatinine measured as part of each subject’s routine primary postoperative hospitalization and the four variable equation from the Modification of Diet in Renal Disease Study Group.31 Postoperative creatinine clearance less than 30 ml·min−1·1.73m−2 was used for multivariable modeling for postoperative HF, as this is an accepted threshold for severe renal dysfunction and could potentially confound association between increased plasma BNP and postoperative HF.31,32 Consistent with previous studies of our cohort, postoperative ventricular dysfunction was defined as new postoperative need for an intraaortic balloon pump, or for two or more inotropes (continuous infusion of amrinone, milrinone, dobutamine, dopamine more than 5 µg·kg−1·min−1, epinephrine, norepinephrine, isoproterenol, or vasopressin) in the operating room after separating from cardiopulmonary bypass or in the postoperative intensive care unit.26,33
Statistical Analysis
Statistical analyses were performed using SAS (version 9.2; SAS Institute, Cary, NC) and R (version 2.13.1; R Foundation for Statistical Computing, Vienna, Austria). P values for all study analyses were two-tailed. Distributions of both preoperative and peak postoperative plasma BNP data were right skewed. Wilcoxon rank sum tests were used to compare BNP concentrations for subjects who did and did not experience the study’s HF outcome. Wilcoxon signed-rank tests were used to compare preoperative versus peak postoperative BNP concentrations within the group of subjects who experienced a postoperative HF event and within the group of subjects who did not experience a postoperative HF event. Continuous BNP data were log10 transformed to normalize distributions before additional analyses. Pearson correlation was calculated between the preoperative and peak postoperative BNP variables. Table 1 covariates were selected a priori as potentially important risk factors for postoperative HF events. Cox proportional hazards regression was used to assess univariate associations of clinical and BNP variables with time to first postoperative HF event. In all Cox proportional hazards regression analyses, subjects were censored at the time of postoperative loss to follow-up, if loss to follow-up occurred before the end of the study period. Otherwise subjects were censored at the end of the study period. Given that we obtained follow-up data for 1,025 subjects, assuming 80% power and a type I error rate = 0.025, we estimate a minimum detectable unadjusted hazard ratio (HR) of 1.25 for the association between a 1 unit change in log10 preoperative BNP and risk of HF event during 5 yr postoperative follow-up, and we estimated a minimum detectable unadjusted HR of 2.37 for a 1 unit change in log10 peak postoperative BNP.34
Table 1.
Univariate Associations between Perioperative Clinical Characteristics and Time to Heart Failure Hospitalization or Heart Failure Mortality after Primary Coronary Artery Bypass Graft Surgery (n = 1,025; 105 subjects experienced heart failure hospitalization or heart failure death events)
| Clinical Variables | Mean or Proportion |
Hazard Ratio (95% CI) | P Value |
|---|---|---|---|
| Preoperative clinical characteristics | |||
| Age ≥65 yr | 487 (47.5%) | 2.25 (1.50–3.38) | <0.001 |
| Female sex | 196 (19.1%) | 1.95 (1.28–2.95) | 0.002 |
| Ethnicity (minority) | 139 (13.6%) | 1.08 (0.62–1.90) | 0.79 |
| Obesity (BMI >30 kg/m2) | 395 (38.5%) | 1.51 (1.03–2.22) | 0.03 |
| Diabetes mellitus (n = 1,024) | 296 (28.9%) | 2.48 (1.69–3.63) | <0.001 |
| Hypertension (n = 1,023) | 764 (74.7%) | 1.88 (1.10–3.20) | 0.02 |
| Hypercholesterolemia (n = 1,020) | 773 (75.8%) | 0.82 (0.53–1.27) | 0.37 |
| Smoking, >30 pack-year history (n = 981) | 262 (26.7%) | 2.36 (1.59–3.49) | <0.001 |
| Preoperative creatinine clearance (ml·min−1·1.73m−2) | 74.3 (SD, 20.6) | 0.97 (0.96–0.98) for 1 ml·min−1·1.73m−2 increase |
<0.001 |
| Myocardial infarction ≤2 wk preoperatively (n = 1,024) | 180 (17.6%) | 1.73 (1.12–2.68) | 0.01 |
| Left ventricular ejection fraction (%; n = 990) | 53 (SD, 12) | 0.97 (0.95–0.98) for 1% increase | <0.001 |
| Coronary artery regions with >50% stenosis | 0.82 | ||
| 0–1 region | 73 (7.1%) | 1.18 (0.56–2.46) | |
| 2 regions | 339 (33.1%) | 1.12 (0.74–1.69) | |
| 3 regions | 613 (59.8%) | 1.00 | |
| Mitral insufficiency (moderate or severe; n = 988) | 24 (2.4%) | 3.96 (1.84–8.55) | <0.001 |
| History of preoperative arrhythmia treatment | 103 (10.1%) | 2.99 (1.90–4.72) | <0.001 |
| Anemia (n = 1,024) | 342 (33.4%) | 3.36 (2.26–4.99) | <0.001 |
| Preoperative cTnI >0.1 µg/l | 151 (14.7%) | 1.28 (0.78–2.10) | 0.33 |
| Preoperative medications | |||
| ACE-inhibitor (n = 1,024) | 475 (46.4%) | 1.52 (1.03–2.23) | 0.03 |
| Diuretic (n = 1,024) | 218 (21.3%) | 2.94 (1.99–4.33) | <0.001 |
| Statin | 794 (77.5%) | 1.32 (0.80–2.17) | 0.27 |
| Digoxin | 27 (2.6%) | 2.68 (1.17–6.10) | 0.02 |
| β blocker | 806 (78.6%) | 1.32 (0.80–2.19) | 0.28 |
| Calcium channel blocker (n = 1,024) | 145 (14.2%) | 1.70 (1.06–2.71) | 0.03 |
| Aspirin | 788 (76.9%) | 0.75 (0.49–1.15) | 0.19 |
| Nonaspirin platelet inhibitor (n = 1,024) | 215 (21.0%) | 1.54 (1.01–2.37) | 0.05 |
| Nitroglycerin intravenous (n = 1,020) | 106 (10.4%) | 0.86 (0.45–1.66) | 0.66 |
| Heparin intravenous | 236 (23.0%) | 1.87 (1.25–2.79) | 0.002 |
| Surgical characteristics | |||
| Urgent surgery (n = 1,024) | 562 (54.9%) | 1.89 (1.25–2.86) | 0.002 |
| Cardiopulmonary bypass time >120 min | 231 (22.5%) | 0.96 (0.60–1.52) | 0.85 |
| Number of coronary grafts (n = 1,024) | 0.04 | ||
| <3 grafts | 147 (14.4%) | 1.91 (1.08–3.36) | |
| 3 grafts | 469 (45.8%) | 1.61 (1.03–2.51) | |
| >3 grafts | 408 (39.8%) | 1.00 | |
| In-hospital postoperative characteristics | |||
| Ventricular dysfunction* | 120 (11.7%) | 2.95 (1.89–4.60) | <0.001 |
| New onset atrial fibrillation | 308 (30.1%) | 1.76 (1.19–2.59) | 0.005 |
| Postoperative creatinine clearance (ml·min−1·1.73m−2; n = 1,024) |
67.8 (SD, 21.4) | 0.97 (0.96–0.98) for 1 ml·min·1.73m−2 increase |
<0.001 |
| Postoperative creatinine clearance <30 ml·min· 1.73m−2; n = 1,024) |
38 (3.7%) | 5.12 (2.86–9.16) | <0.001 |
| Peak postoperative cTnI, µg/l | 3.82 (SD, 8.56) | 1.52 (1.09–2.12) for log10 increase | 0.01 |
Data are shown as n (%) for categorical variables and mean ± SD for continuous variables.
Defined as new postoperative need for two or more inotropes or an intraaortic balloon pump.
ACE = angiotensin-converting enzyme; BMI = body mass index; cTnI = cardiac troponin I.
A multivariable clinical model for association with time to first postoperative HF event was created using step-wise selection with Cox proportional hazards regression. Age 65 yr or more, sex, study institution, and preoperative left ventricular ejection fraction were locked into the multivariable model before step-wise selection from the variables shown in table 1. P value thresholds for entry and exit into the multivariable model during step-wise selection were 0.15 and 0.05, respectively. Continuous preoperative and peak postoperative BNP data were entered into the final multivariable clinical model separately and then together to assess additional predictive benefit, and Akaike information criteria (AIC) were used to compare goodness of fit of these multivariable models. Proportional hazards assumptions for the variables in the final multivariable model (including preoperative and peak postoperative BNP assessed as continuous variables) were evaluated by using the Schoenfeld residuals method to confirm that the residuals for each variable were not correlated (P > 0.05) with the ordering of times to postoperative HF events.
Harrell time-censored C-indices (R Hmisc package; version 3.8.3) were used to assess the prognostic discriminatory relationship between perioperative BNP variables and time to first postoperative HF event.35 Harrell’s C-indices were calculated for preoperative and peak postoperative BNP data and for the final multivariable clinical model with and without the BNP variables added. C-indices for different models represent the probability of concordance between predicted and observed survival, with c = 1 for a perfectly discriminating model. U statistics were then computed to test whether multivariable clinical models including BNP variables had a higher fraction of concordant pairs than the multivariable clinical model that did not include BNP variables. Additionally, to assess the internal validity of the multivariable clinical models: (1) that did not include perioperative BNP variables; (2) that included preoperative BNP; (3) that included peak postoperative BNP, we computed C-indices for these models after performing bootstrap resampling (n = 100; R rms package; version 3.6.3).
Lastly, preoperative and peak postoperative BNP data were dichotomized at their top quartiles (preoperative BNP ≥50.8 pg/ml and peak postoperative BNP ≥314.1 pg/ml) and were entered separately into the final multivariable clinical model in order to assess additional predictive benefit. The dichotomized preoperative and peak postoperative BNP variables were also assessed together by classifying subjects into one of four groups according to whether preoperative and peak postoperative BNP concentrations were in the top quartile (high) or the bottom three quartiles (low). In other words, preoperative and peak postoperative BNP both low; preoperative BNP low and peak postoperative BNP high; preoperative BNP high and peak postoperative BNP low; preoperative BNP high and peak postoperative BNP high. AIC were used to compare goodness of fit of the multivariable models with and without dichotomized BNP variables. Kaplan–Meier survival curves were constructed to compare HF event-free survival in subjects with preoperative BNP in the top quartile as compared with subjects with preoperative BNP concentration in the bottom three quartiles. Kaplan–Meier survival curves were similarly created for peak postoperative BNP dichotomized at the top quartile. Log rank tests were used to assess for significant differences between stratified curves.
Results
Patient Characteristics
Table 1 describes clinical and surgical characteristics of the 1,025 subjects included in the study analysis along with univariate associations between each of these variables and time to postoperative first HF event. One hundred five subjects (10.2%) experienced at least one HF event within the 5-yr postoperative follow-up period; 99 subjects were hospitalized for HF and 12 subjects died from HF. Median time to first HF event was 1.1 yr (range 6 days to 4.8 yr). Clinical and surgical characteristics of subjects who experienced HF events are shown in Supplemental Digital Content 1 (table 2, http://links.lww.com/ALN/A945). For the 920 subjects who did not experience HF events, median follow-up time was 4.2 yr (range 5 days to 5 yr; this includes 68 subjects who died of non-HF causes during follow-up).
Table 2.
Multivariable Adjusted Associations between BNP Assessments and Time to Heart Failure Hospitalization or Heart Failure Mortality after Primary Coronary Artery Bypass Graft Surgery*
| Perioperative BNP Variable; n = 947†; 99 Subjects with Heart Failure Events |
Hazard Ratio (95% CI) | P Value | AIC of Multivariable Model* with BNP Included in Model |
|---|---|---|---|
| Preoperative BNP (pg/ml) | 1.93 (1.30–2.88) for log10 increase | 0.001 | 1,185.25 |
| Peak postoperative BNP (pg/ml) | 3.38 (1.45–7.65) for log10 increase | 0.003 | 1,190.24 |
| Preoperative BNP (pg/ml) together with | 1.67 (1.09–2.56) for log10 increase | 0.02 | 1,185.28 |
| Peak postoperative BNP (pg/ml) | 1.96 (0.77–4.98) for log10 increase | 0.16 |
Variables in multivariable clinical model are shown in Supplemental Digital Content 1, table 1, http://links.lww.com/ALN/A945. The AIC of the Supplemental Digital Content 1, table 1 model = 1,196.90 before adding BNP data to the model.
Seventy-eight subjects missing one or more of the variables in Supplemental Digital Content 1, table 1, http://links.lww.com/ALN/A945, are not included in the multivariable analysis.
AIC = Akaike information criterion; BNP = B-type natriuretic peptide.
Perioperative BNP Concentrations and Unadjusted Associations with Postoperative HF Hospitalization or Mortality
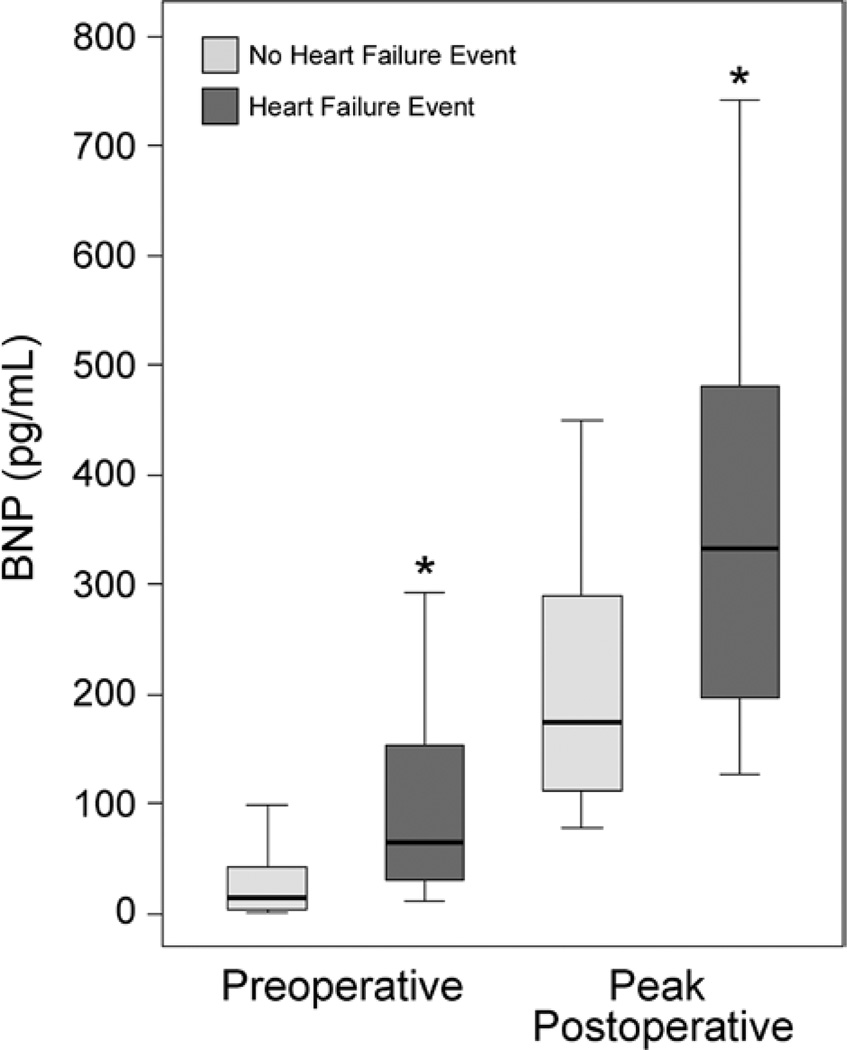
Median preoperative BNP concentration was 17.2 pg/ml (25th to 75th percentile range: 4.9–50.8 pg/ml). Median peak postoperative BNP concentration was 184.1 pg/ml (25th to 75th percentile range: 116.9–314.1 pg/ml). Preoperative BNP concentrations significantly correlated with peak postoperative BNP concentrations (r2 = 0.25; P < 0.001). Preoperative and peak postoperative BNP were significantly higher in the group of subjects who experienced HF hospitalization or HF death events during follow-up when compared with the group of subjects who did not experience HF events (P < 0.001; fig. 1). In both the subjects who did and did not develop postoperative HF events, peak postoperative BNP was significantly increased when compared with preoperative measures (P < 0.001).
Fig. 1.
Box and whisker plots of preoperative and peak postoperative plasma B-type natriuretic peptide (BNP) concentrations for 1,025 subjects undergoing primary coronary artery bypass graft surgery. BNP concentrations are stratified by the group of 920 subjects who did not experience heart failure (HF) events and the group of 105 subjects who did experience HF events during 5-yr postoperative follow-up. The 10th (lower whisker), 25th (lower box end), 50th (middle line), 75th (upper box end), and 90th (upper whisker) percentile BNP values are shown for each group for both the preoperative and peak postoperative (postoperative days 1–5) assessments. *P < 0.001 HF event compared with the no-HF event group at that perioperative time point.
In Cox proportional hazards analyses, both increased preoperative and peak postoperative BNP concentrations had strong univariate associations with shorter time to HF event during up to 5 yr postoperative follow-up (HR for log10 increase in preoperative BNP = 3.20; 95% CI, 2.33– 4.40; P < 0.001; HR for log10 increase in peak postoperative BNP = 9.78; 95% CI, 5.54–17.28; P < 0.001). When the preoperative and peak postoperative BNP were considered together, despite significant correlation between preoperative and peak postoperative BNP, both remained significant predictors of time to first HF event (HR for log10 increase in preoperative BNP = 2.10; 95% CI, 1.42–3.09; P < 0.001; HR for log10 increase in peak postoperative BNP = 3.36; 95% CI, 1.56–7.25; P = 0.002).
Multivariable Adjusted Associations between BNP Concentrations and Postoperative HF Hospitalization or Mortality
The study’s multivariable clinical model for time to postoperative HF event included age of 65 yr or more, sex, institution, preoperative left ventricular ejection fraction, diabetes mellitus, more than 30 pack-yr history of smoking, history of preoperative arrhythmia treatment, preoperative anemia, preoperative diuretic use, postoperative ventricular dysfunction, and postoperative creatinine clearance less than 30 ml·min−1·1.73m−2 (Supplemental Digital Content 1, table 1, http://links.lww.com/ALN/A945). When preoperative and peak postoperative BNP were added separately to the multivariable clinical model, both BNP variables significantly associated with shorter time to postoperative HF event (P ≤ 0.003; table 2). When both preoperative and peak postoperative BNP were added simultaneously into the multivariable model, preoperative BNP remained significantly associated with shorter time to first HF event (P = 0.02; table 2), but peak postoperative BNP was no longer significantly associated (P = 0.16; table 2). The goodness of fit of the clinical multivariable model was improved (lower AIC) by the individual additions of preoperative BNP (AIC = 1,185.25) or peak postoperative BNP data (AIC = 1,190.24). However, simultaneously adding preoperative BNP and peak postoperative BNP to the clinical multivariable model resulted in a model AIC that was essentially unchanged from the AIC of the multivariable clinical model that incorporated preoperative BNP alone (table 2).
Time-censored Harrell C-indices were calculated for both continuous preoperative and peak postoperative BNP data in relation to occurrence of HF events during the 5 yr after CABG surgery. Preoperative BNP demonstrated good prognostic discrimination with a C-index = 0.753, 95% CI, 0.706–0.801. Peak postoperative BNP had a C-index = 0.721 (95% CI, 0.674–0.769). The C-index for the study’s multivariable clinical model (without BNP data added) = 0.803 (95% CI, 0.760–0.845). When preoperative BNP was added to the study’s multivariable clinical model, the multivariable model’s C-index increased significantly (P = 0.01) to 0.820 (95% CI, 0.780–0.861). When peak postoperative BNP was added to the multivariable clinical model, the multivariable model’s C-index increased to 0.812 (95% CI, 0.771–0.854), but this C-index increase was not statistically significant (P = 0.12).
After performing bootstrap resampling from the data-set, C-indices were 0.784 for the multivariable clinical model, which did not include BNP variables, 0.805 for the multivariable clinical model, which added the preoperative BNP variable, and 0.796 for the multivariable clinical model, which added the peak postoperative BNP variable.
Perioperative BNP Concentrations Dichotomized at the Top Quartile and Postoperative HF Hospitalization or Mortality
Before adjusting for clinical risk factors, subjects in the top quartiles for preoperative or peak postoperative BNP concentrations were at significantly higher risk for HF events during the 5 yr after CABG surgery (P ≤ 0.003; table 3), with subjects in the top quartiles for both preoperative and peak postoperative BNP at highest risk when compared with subjects in the lower three quartiles for both BNP assessments (HR = 7.27; 95% CI, 4.61–11.46; P < 0.001). When added separately into the study’s multivariable clinical model, the top quartiles of both preoperative (P < 0.001) and peak postoperative BNP (P = 0.02) remained significantly associated with shorter time to HF event during the 5 yr after CABG surgery (table 3). After adjusting for the multivariable clinical model, subjects in the top quartile for preoperative BNP (regardless of whether peak postoperative BNP was in the top or lower quartiles) remained at significantly increased risk for HF events when compared with subjects in the lower three quartiles for both preoperative and peak postoperative BNP (P < 0.001; table 3). However, subjects in the top quartile for peak postoperative BNP but in the lower three quartiles for preoperative BNP were no longer at significantly increased risk for HF events when compared with subjects in the lower three quartiles for both preoperative and peak postoperative BNP (P = 0.17; table 3).
Table 3.
Univariate (n = 1,025) and Multivariable Adjusted (n = 947)*† Associations between BNP Variables Dichotomized at the Top Quartile and Time to Heart Failure Hospitalization or Heart Failure Mortality after Primary Coronary Artery Bypass Graft Surgery
| Perioperative BNP Variable | Proportion, n (%) |
Unadjusted Hazard Ratio (95% CI) |
Unadjusted P Value |
Multivariable Adjusted† Hazard Ratio (95% CI) |
Multivariable Adjusted† P Value |
AlC of Multivariable Model* with BNP Included in Model*† |
|
|---|---|---|---|---|---|---|---|
| Top quartile preoperative BNP | 257(25.1) | 4.93 (3.34–7.27) | <0.001 | 2.60(1.63–4.14) | <0.001 | 1,182.75 | |
| Top quartile peak postoperative BNP | 256 (25.0) | 4.01 (2.73–5.89) | <0.001 | 1.74(1.08–2.78) | 0.02 | 1,193.65 | |
| Preoperative BNP and Peak Postoperative BNP | 1,184.93 | ||||||
| Lower 3 quartiles | Lower 3 quartiles | 669 (65.3) | 1.00 | Reference | 1.00 | Reference | |
| Lower 3 quartiles | Top quartile | 99 (9.7) | 2.70(1.39–5.27) | 0.003 | 1.66(0.81–3.40) | 0.17 | |
| Top quartile | Lower 3 quartiles | 100(9.7) | 3.96(2.17–7.24) | <0.001 | 3.05(1.58–5.89) | <0.001 | |
| Top quartile | Top quartile | 157(15.3) | 7.27(4.61–11.46) | <0.001 | 3.06(1.72–5.44) | <0.001 | |
Multivariable clinical model is shown in table 1 of Supplemental Digital Content 1, http://links.lww.com/ALN/A945. The AIC of the multivariable clinical model before adding BNP into the model = 1,196.90.
Seventy-eight subjects missing one or more of the variables in Supplemental Digital Content, table 1, http://links.lww.com/ALN/A945, are not included in the multivariable analysis.
AIC = Akaike information criterion; BNP = B-type natriuretic peptide.
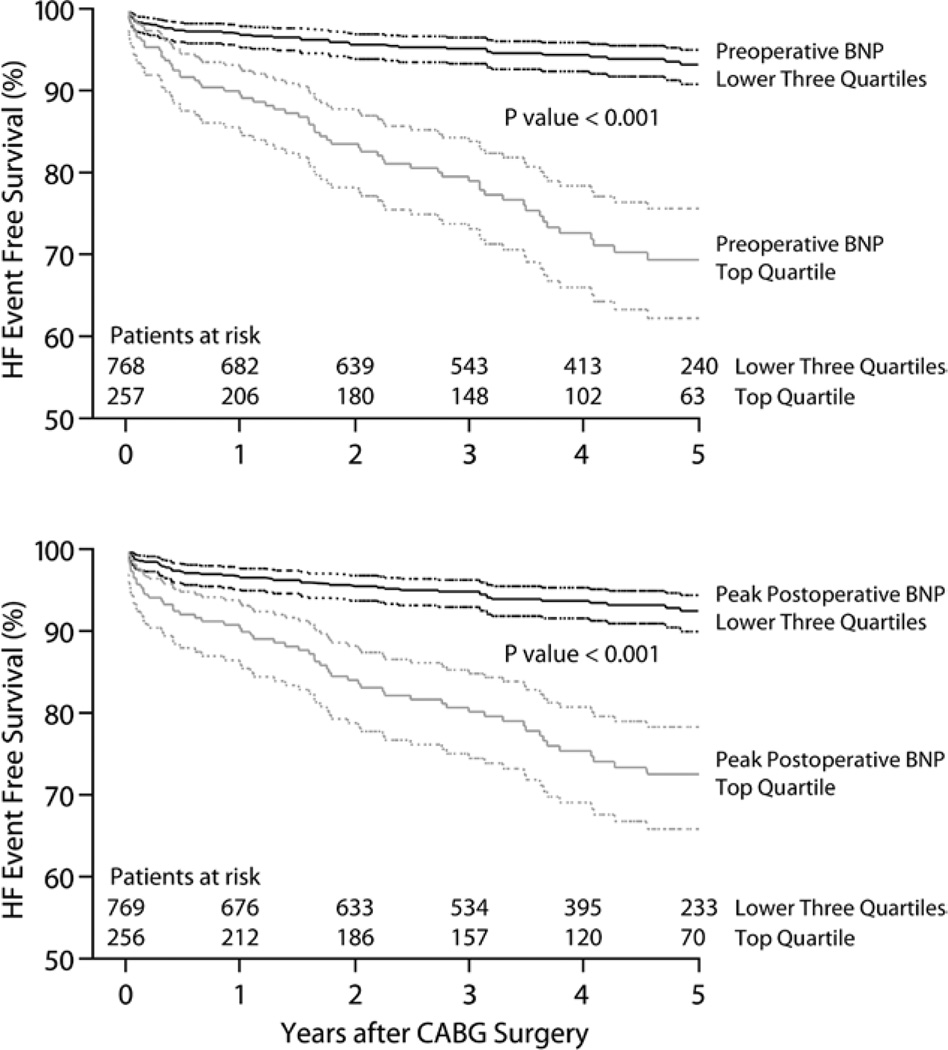
Kaplan—Meier survival curves comparing HF event-free survival for subjects in the top quartile versus the lower three quartiles for preoperative BNP (fig. 2A) and for subjects in the top quartile versus the lower three quartiles for peak postoperative BNP (fig. 2B) assessments indicate significantly improved (P < 0.001) HF event-free survival for subjects with the lower values of both BNP assessments.
Fig. 2.
Kaplan–Meier survival curves for 1,025 primary coronary artery bypass graft (CABG) surgical patients followed up to 5 yr after surgery. Curves are stratified according to the top versus bottom three quartiles of (A) preoperative B-type natriuretic peptide (BNP) concentrations and (B) the peak of postoperative day 1–5 BNP concentrations. Curves signified by dotted lines signify 95% CIs for heart failure (HF) event-free survival estimates during 5 yr of postoperative follow-up.
Discussion
Despite undergoing nonemergency, isolated primary CABG surgery, more than 10% of the 1,025 subjects followed in this study during the 5 yr after surgery, experienced HF severe enough to result in hospitalization or death. Both increased preoperative and peak postoperative BNP concentrations individually associated with significantly decreased time to first HF event after CABG surgery. This was true even after adjusting for multiple demographic and clinical HF risk factors, including preoperative left ventricular ejection fraction and development of in-hospital postoperative ventricular dysfunction requiring multiple inotropes or an intraaortic balloon pump.
The American College of Cardiology/American Heart Association’s perioperative evaluation guidelines highlight patients’ functional status as a key factor for deciding who should undergo preoperative cardiac testing or optimization, yet surgical patients are frequently elderly and have limited preoperative physical activity levels.36 Therefore, identification of a biomarker such as BNP, which could identify surgical patients with subclinical as well as clinically symptomatic HF or ischemia5 could potentially improve perioperative risk stratification and optimization. Several meta-analyses of studies of noncardiac surgical patients indicate that increased preoperative BNP (or NT-proBNP) significantly associates with higher incidence of postoperative adverse cardiovascular events and mortality.37–40
Our study expands on studies of noncardiac surgical patients, as we hypothesized that in patients with known preoperative coronary artery disease presenting for primary CABG surgery, increased preoperative and peak postoperative BNP concentrations significantly associate with occurrence of HF resulting in hospitalization or death during the 5 yr after CABG surgery. Two meta-analyses of studies assessing the relationship between increased preoperative BNP and adverse outcomes after cardiac surgery report a significant association between higher preoperative BNP concentrations and increased all-cause mortality after cardiac surgery.40,41 However, the majority of studies in these meta-analyses included subjects undergoing emergency cardiac surgery, valve surgery, or a combination of valve and CABG surgery. Consequently, associations observed between increased preoperative BNP and adverse postoperative outcomes may reflect increased clinical cardiac distress that occurs in these cardiac surgical groups already known to be at high risk for adverse postoperative events. Furthermore, many of these studies enrolled small subject numbers, and thus, did not adjust observed BNP associations for additional clinical risk factors.40,41 A strength of our study is that we assessed more than 1,000 subjects undergoing nonemergency, isolated primary CABG surgery, thereby, focusing on a patient population that presents for surgery with reasonable expectation of good long-term postoperative HRQL and survival. This cardiac surgical group is frequently not at obvious risk for developing adverse postoperative outcomes, and therefore, could derive considerable potential benefit from knowledge of risk factors that can be modified to help avoid adverse long-term postoperative cardiovascular morbidities such as HF.
In previous studies of primary CABG patients, we have shown that increased perioperative BNP concentrations significantly associate with increased postoperative in-hospital ventricular dysfunction,26 longer primary hospital stays,26,27 lower longer-term physical function,28 and increased all-cause mortality.26,27 However, these adverse outcomes do not have clearly defined approaches for their prevention or mitigation (i.e., it is difficult to design perioperative interventions to prevent or treat all-cause death). In ambulatory patients, HF is clearly linked with reduced HRQL and survival, and there are clear medical practice guidelines for HF prevention, surveillance, and treatment.1,2,42,43 Because BNP is an established diagnostic and prognostic biomarker in ambulatory HF patients,6–8 there is biology to suggest that increased perioperative BNP concentrations should independently associate with long-term HF after CABG surgery. Indeed, our study found that even after adjusting for clinical risk factors, increased preoperative and peak postoperative BNP concentrations strongly associate with significantly decreased time to first HF event during the 5 yr after CABG surgery. In previous studies of BNP and cardiac surgery, although increased BNP significantly associated with all-cause mortality, the prognostic discrimination of the BNP biomarker by itself for identifying patients at risk of dying after CABG surgery was not very robust (i.e., reported C-indices <0.70).26,27,41 The current study’s assessment of occurrence of HF hospitalization or HF death after CABG surgery identified substantially higher C-indices for individual assessments of preoperative (C-index = 0.75) and peak postoperative BNP (C-index = 0.72). Furthermore, adding preoperative BNP data to the study’s multivariable clinical model improved the prognostic discrimination of the multivariable model as determined by a statistically significant increase in the multivariable model’s C-index. These findings further support the potential clinical usefulness of perioperative BNP assessments for identifying patients at increased risk of developing HF after CABG surgery.
What still needs to be determined is how perioperative BNP measurements in CABG patients can be used to prevent adverse long-term postoperative outcomes such as HF. Some studies of ambulatory HF patients have found that medical management (e.g., frequent clinic visits coupled with careful and persistent titration of medications such as β-blockers and angiotensin-converting enzyme inhibitors) targeted toward gradually lowering serial follow-up measurements of BNP or NT-proBNP concentrations may reduce associated adverse cardiovascular events11–14 and improve HF hospitalization-free survival,13,16,17 HRQL,13 cardiac structure and function,13 healthcare costs,15 and overall mortality.44 What is intriguing about evaluation of BNP concentrations in primary CABG patients is the potential to use knowledge of increased perioperative BNP levels to guide medical surveillance and management aimed at decreasing postoperative HF and related sequelae. To date, there have been no studies of BNP-guided preoperative or postoperative surveillance and management of CABG surgical patients. However, it seems plausible that CABG surgery patients who have increased perioperative BNP concentrations and who receive perioperative medical interventions targeted toward identifying the cause(s) of increased BNP and lowering serial postoperative BNP measurements, could experience fewer adverse postoperative cardiac events (particularly HF events), than patients who are discharged to “usual” medical follow-up after CABG surgery. This hypothesis could be tested in future appropriately designed clinical trials.
In this study, when preoperative and peak postoperative BNP were considered together along with clinical risk factors for postoperative HF, increased preoperative BNP remained an independent predictor of HF hospitalization or death during the 5 yr after CABG surgery, whereas peak postoperative BNP did not remain an independent predictor. This is likely because peak postoperative BNP is significantly correlated with preoperative BNP and has a wider range of values when compared with preoperative BNP. However, our results suggest that increased peak postoperative BNP could be a useful biomarker for predicting development of significant HF after primary CABG surgery, particularly, if a preoperative BNP assessment is unavailable. Furthermore, because there is a significant BNP response to the intraoperative insults of CABG surgery itself, the peak postoperative BNP measurement provides a potentially important starting assessment for monitoring and treating those patients deemed at highest risk for developing long-term HF after CABG surgery. It is also possible that in a larger cohort study, peak postoperative BNP would also be an independent predictor of adverse outcome.
Several potential limitations of our study should be considered. First, it should be recognized that this is a study of patients undergoing isolated nonemergency primary CABG surgery with cardiopulmonary bypass, and our results may not apply to patients undergoing other types of cardiac surgery. For example, patients presenting with aortic stenosis for aortic valve replacement surgery may have higher preoperative BNP concentrations than patients undergoing CABG surgery, possibly because of relatively increased ventricular pressure overload. Future studies should validate our study’s observed associations in other primary CABG surgery cohorts and assess the utility of perioperative BNP measures in patients undergoing other types of cardiac surgeries. Second, there are multiple commercially available assays for measuring BNP and NT-proBNP, and although these assessments correlate well with each other, there is often between-assay variability in absolute values.45 Furthermore, although both BNP and NT-proBNP have been associated with adverse postoperative outcomes,37–41 NT-proBNP is metabolized differently than BNP, and NT-proBNP values are typically higher than BNP values.45 Absolute cutpoints in perioperative BNP concentrations should be considered specific to this study’s subject population and BNP assay. Third, we were unable to obtain postoperative follow-up data for 13% of our subjects, and therefore, cannot rule out the possibility of information bias influencing our results. Fourth, this is an analysis of an existing cohort for which we have previously assessed and reported associations between perioperative BNP and outcomes that include early postoperative ventricular dysfunction,26 primary hospital length of stay,26,27 longer-term physical function,28 and up to 5 yr postoperative all-cause mortality.26,27 The current study’s assessment of occurrence of HF hospitalization or HF death during the 5 yr after primary CABG surgery did not adjust for previous analyses of these other outcomes in the CABG Genomics Program cohort. Finally, our study’s HF outcome did not delineate specific etiologies of observed postoperative HF events (e.g., systolic vs. diastolic dysfunction or left atrial enlargement). Future studies are needed to describe cardiac structural changes that may exist preoperatively or occur over time in CABG patients, who have increased perioperative BNP concentrations. Such future studies could incorporate detailed echocardiographic characterization of cardiac structure and function, with corresponding delineation of the correlations between these echocardiographic assessments and perioperative and longer-term postoperative BNP concentrations.
Conclusion
After adjusting for clinical risk factors such as low preoperative left ventricular ejection fraction, increased preoperative and peak postoperative BNP strongly associate with HF-related hospitalization or death during the 5 yr after nonemergency isolated primary CABG surgery. Future studies are warranted to assess whether medical management, to reduce increased perioperative BNP in CABG surgical patients, decreases incidence of HF-related postoperative admissions and mortality.
What We Already Know about This Topic
Plasma B-type natriuretic peptide increase before and after coronary artery bypass graft surgery is associated with an in-crease in all-cause mortality postoperatively
Plasma B-type natriuretic peptide is a diagnostic and prognostic factor in ambulatory heart failure, but whether preoperative B-type natriuretic peptide increase predicts postoperative heart failure after coronary artery bypass graft surgery is not known
What This Article Tells Us That Is New
In a secondary analysis of an observational study of 1,025 coronary artery bypass graft surgical patients, increased perioperative plasma B-type natriuretic peptide independently predicted 5-yr heart failure hospitalization and heart failure death
Acknowledgments
Funding was provided by Biosite Incorporated, San Diego, California; Dennis W. Jahnigen Career Development Scholars Award, American Geriatrics Society, New York (to Principal Investigator: Dr. Fox); National Institutes of Health, Bethesda, Maryland (K23HL068774 and R01HL098601; to Principal Investigator: Dr. Body); National Institute on Aging, Bethesda, Maryland (K24AG035075; to Principal Investigator: Dr. Marcantonio); Division of Cardiovascular Anesthesia at the Texas Heart Institute, Saint Luke’s Episcopal Hospital, Houston, Texas (to Principal Investigator: Dr. Collard); and the Department of Anesthesiology, Perioperative and Pain Medicine, Brigham and Women’s Hospital, Boston, Massachusetts.
The authors thank the outstanding contributory efforts of the coronary artery bypass graft (CABG) Genomics Program’s research staff who worked diligently on data collection for this study: Kutjim Bodinaku, M.D. (Research Assistant, Department of Anesthesiology, Perioperative and Pain Medicine, Brigham and Women’s Hospital, Harvard Medical School, Boston, Massachusetts), Svetlana Gorbatov, M.P.H. (Research Assistant, Department of Anesthesiology, Perioperative and Pain Medicine, Brigham and Women’s Hospital, Harvard Medical School), James Gosnell, R.N. (Research Nurse, Department of Anesthesiology, Perioperative and Pain Medicine, Brigham and Women’s Hospital, Harvard Medical School), Sejfudin Kavazovic, C.R.A. (Research Assistant, Division of Cardiovascular Anesthesia, Texas Heart Institute, Saint Luke’s Episcopal Hospital, Houston, Texas), Adrienne Kicza, B.S. (Research Assistant, Department of Anesthesiology, Perioperative and Pain Medicine, Brigham and Women’s Hospital, Harvard Medical School), and Charles Wellington, R.N., B.S.N., C.C.R.C. (Research Nurse, Division of Cardiovascular Anesthesia, Texas Heart Institute, Saint Luke’s Episcopal Hospital). Finally the authors thank all the individuals who have participated in the CABG Genomics Program. We would like to credit the Texas Department of State Health Services as the source of death certificate information for study subjects who died in Texas.
Footnotes
Presented as an Oral Poster Discussion at the annual meeting of the American Society of Anesthesiologists, October 16, 2011, Chicago, Illinois.
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are available in both the HTML and PDF versions of this article. Links to the digital files are provided in the HTML text of this article on the Journal’s Web site (www.anesthesiology.org).
http://clinicaltrials.gov/show/NCT00281164. Accessed May 21, 2013.
References
- 1.Roger VL, Go AS, Lloyd-Jones DM, Adams RJ, Berry JD, Brown TM, Carnethon MR, Dai S, de Simone G, Ford ES, Fox CS, Fullerton HJ, Gillespie C, Greenlund KJ, Hailpern SM, Heit JA, Ho PM, Howard VJ, Kissela BM, Kittner SJ, Lackland DT, Lichtman JH, Lisabeth LD, Makuc DM, Marcus GM, Marelli A, Matchar DB, McDermott MM, Meigs JB, Moy CS, Mozaffarian D, Mussolino ME, Nichol G, Paynter NP, Rosamond WD, Sorlie PD, Stafford RS, Turan TN, Turner MB, Wong ND, Wylie-Rosett J. American Heart Association Statistics Committee and Stroke Statistics Subcommittee: Heart disease stroke statistics-2011 update A report from the American Heart Association. Circulation. 2011;123:e18. doi: 10.1161/CIR.0b013e3182009701. 09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Juenger J, Schellberg D, Kraemer S, Haunstetter A, Zugck C, Herzog W, Haass M. Health related quality of life in patients with congestive heart failure Comparison with other chronic diseases and relation to functional variables. Heart. 2002;87:235–241. doi: 10.1136/heart.87.3.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.He J, Ogden LG, Bazzano LA, Vupputuri S, Loria C, Whelton PK. Risk factors for congestive heart failure in US men and women: NHANES I epidemiologic follow-up study. Arch Intern Med. 2001;161:996–202. doi: 10.1001/archinte.161.7.996. [DOI] [PubMed] [Google Scholar]
- 4.Fonarow GC, Abraham WT, Albert NM, Stough WG, Gheorghiade M, Greenberg BH, O’Connor CM, Pieper K, Sun JL, Yancy CW, Young JB. OPTIMIZE-HF Investigators and Hospitals: Factors identified as precipitating hospital admissions for heart failure and clinical outcomes Findings from OPTIMIZE-HF. Arch Intern Med. 2008;168:847–854. doi: 10.1001/archinte.168.8.847. [DOI] [PubMed] [Google Scholar]
- 5.Daniels LB, Maisel AS. Natriuretic peptides. J Am Coll Cardiol. 2007;50:2357–2368. doi: 10.1016/j.jacc.2007.09.021. [DOI] [PubMed] [Google Scholar]
- 6.Maisel A, Hollander JE, Guss D, McCullough P, Nowak R, Green G, Saltzberg M, Ellison SR, Bhalla MA, Bhalla V, Clopton P, Jesse R. Rapid Emergency Department Heart Failure Outpatient Trial investigators: Primary results of the Rapid Emergency Department Heart Failure Outpatient Trial (REDHOT). A multicenter study of B-type natriuretic peptide levels, emergency department decision making, and outcomes in patients presenting with shortness of breath. J Am Coll Cardiol. 2004;44:1328–1333. doi: 10.1016/j.jacc.2004.06.015. [DOI] [PubMed] [Google Scholar]
- 7.Maisel AS, Krishnaswamy P, Nowak RM, McCord J, Hollander JE, Duc P, Omland T, Storrow AB, Abraham WT, Wu AH, Clopton P, Steg PG, Westheim A, Knudsen CW, Perez A, Kazanegra R, Herrmann HC, McCullough PA. Breathing Not Properly Multinational Study Investigators: Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N Engl J Med. 2002;347:161–167. doi: 10.1056/NEJMoa020233. [DOI] [PubMed] [Google Scholar]
- 8.Doust JA, Pietrzak E, Dobson A, Glasziou P. How well does B-type natriuretic peptide predict death and cardiac events in patients with heart failure: Systematic review. BMJ. 2005;330:625. doi: 10.1136/bmj.330.7492.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mega JL, Morrow DA, De Lemos JA, Sabatine MS, Murphy SA, Rifai N, Gibson CM, Antman EM, Braunwald E. B-type natriuretic peptide at presentation and prognosis in patients with ST-segment elevation myocardial infarction: An ENTIRE-TIMI-23 substudy. J Am Coll Cardiol. 2004;44:335–339. doi: 10.1016/j.jacc.2004.04.033. [DOI] [PubMed] [Google Scholar]
- 10.de Lemos JA, Morrow DA, Bentley JH, Omland T, Sabatine MS, McCabe CH, Hall C, Cannon CP, Braunwald E. The prognostic value of B-type natriuretic peptide in patients with acute coronary syndromes. N Engl J Med. 2001;345:1014–1021. doi: 10.1056/NEJMoa011053. [DOI] [PubMed] [Google Scholar]
- 11.Bhardwaj A, Rehman SU, Mohammed A, Baggish AL, Moore SA, Januzzi JL., Jr Design and methods of the pro-B type natriuretic peptide outpatient tailored chronic heart failure therapy (PROTECT) study. Am Heart J. 2010;159:532–538. doi: 10.1016/j.ahj.2010.01.005. e1. [DOI] [PubMed] [Google Scholar]
- 12.Troughton RW, Frampton CM, Yandle TG, Espiner EA, Nicholls MG, Richards AM. Treatment of heart failure guided by plasma aminoterminal brain natriuretic peptide (N-BNP) concentrations. Lancet. 2000;355:1126–1130. doi: 10.1016/s0140-6736(00)02060-2. [DOI] [PubMed] [Google Scholar]
- 13.Januzzi JL, Jr, Rehman SU, Mohammed AA, Bhardwaj A, Barajas L, Barajas J, Kim HN, Baggish AL, Weiner RB, Chen-Tournoux A, Marshall JE, Moore SA, Carlson WD, Lewis GD, Shin J, Sullivan D, Parks K, Wang TJ, Gregory SA, Uthamalingam S, Semigran MJ. Use of amino-terminal pro-B-type natriuretic peptide to guide outpatient therapy of patients with chronic left ventricular systolic dysfunction. J Am Coll Cardiol. 2011;58:1881–1889. doi: 10.1016/j.jacc.2011.03.072. [DOI] [PubMed] [Google Scholar]
- 14.Berger R, Moertl D, Peter S, Ahmadi R, Huelsmann M, Yamuti S, Wagner B, Pacher R. N-terminal pro-B-type natriuretic peptide-guided, intensive patient management in addition to multidisciplinary care in chronic heart failure a 3-arm, prospective, randomized pilot study. J Am Coll Cardiol. 2010;55:645–653. doi: 10.1016/j.jacc.2009.08.078. [DOI] [PubMed] [Google Scholar]
- 15.Adlbrecht C, Huelsmann M, Berger R, Moertl D, Strunk G, Oesterle A, Ahmadi R, Szucs T, Pacher R. Cost analysis and cost-effectiveness of NT-proBNP-guided heart failure specialist care in addition to home-based nurse care. Eur J Clin Invest. 2011;41:315–322. [Google Scholar]
- 16.Jourdain P, Jondeau G, Funck F, Gueffet P, Le Helloco A, Donal E, Aupetit JF, Aumont MC, Galinier M, Eicher JC, Cohen-Solal A, Juillière Y. Plasma brain natriuretic peptide-guided therapy to improve outcome in heart failure: The STARS-BNP Multicenter Study. J Am Coll Cardiol. 2007;49:1733–1739. doi: 10.1016/j.jacc.2006.10.081. [DOI] [PubMed] [Google Scholar]
- 17.Pfisterer M, Buser P, Rickli H, Gutmann M, Erne P, Rickenbacher P, Vuillomenet A, Jeker U, Dubach P, Beer H, Yoon SI, Suter T, Osterhues HH, Schieber MM, Hilti P, Schindler R, Brunner-La Rocca HP. TIME-CHF Investigators: BNP-guided vs symptom-guided heart failure therapy: The Trial of Intensified vs Standard Medical Therapy in Elderly Patients With Congestive Heart Failure (TIME-CHF) randomized trial. JAMA. 2009;301:383–392. doi: 10.1001/jama.2009.2. [DOI] [PubMed] [Google Scholar]
- 18.Eagle KA, Guyton RA, Davidoff R, Edwards FH, Ewy GA, Gardner TJ, Hart JC, Herrmann HC, Hillis LD, Hutter AM, Jr, Lytle BW, Marlow RA, Nugent WC, Orszulak TA. ACC/AHA 2004 guideline update for coronary artery bypass graft surgery: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1999 Guidelines for Coronary Artery Bypass Graft Surgery) Circulation. 2004;110:e340. 37. [PubMed] [Google Scholar]
- 19.Hawkes AL, Mortensen OS. Up to one third of individual cardiac patients have a decline in quality of life post-intervention. Scand Cardiovasc J. 2006;40:214–218. doi: 10.1080/14017430600784343. [DOI] [PubMed] [Google Scholar]
- 20.Rumsfeld JS, Magid DJ, O’Brien M, McCarthy M, Jr, MaWhinney S, Scd, Shroyer AL, Moritz TE, Henderson WG, Sethi GK, Grover FL, Hammermeister KE. Department of Veterans Affairs Cooperative Study in Health Services: Processes, Structures and Outcomes of Care in Cardiac Surgery: Changes in health-related quality of life following coronary artery bypass graft surgery. Ann Thorac Surg. 2001;72:2026–2032. doi: 10.1016/s0003-4975(01)03213-1. [DOI] [PubMed] [Google Scholar]
- 21.Vaccarino V, Lin ZQ, Kasl SV, Mattera JA, Roumanis SA, Abramson JL, Krumholz HM. Gender differences in recovery after coronary artery bypass surgery. J Am Coll Cardiol. 2003;41:307–314. doi: 10.1016/s0735-1097(02)02698-0. [DOI] [PubMed] [Google Scholar]
- 22.Lindsay GM, Hanlon P, Smith LN, Wheatley DJ. Assessment of changes in general health status using the short-form 36 questionnaire 1 year following coronary artery bypass grafting. Eur J Cardiothorac Surg. 2000;18:557–564. doi: 10.1016/s1010-7940(00)00542-x. [DOI] [PubMed] [Google Scholar]
- 23.Higgins TL, Estafanous FG, Loop FD, Beck GJ, Blum JM, Paranandi L. Stratification of morbidity and mortality outcome by preoperative risk factors in coronary artery bypass patients. A clinical severity score. JAMA. 1992;267:2344–2348. [PubMed] [Google Scholar]
- 24.Nilsson J, Algotsson L, Höglund P, Lührs C, Brandt J. Comparison of 19 preoperative risk stratification models in open-heart surgery. Eur Heart J. 2006;27:867–874. doi: 10.1093/eurheartj/ehi720. [DOI] [PubMed] [Google Scholar]
- 25.Kurki TS, Järvinen O, Kataja MJ, Laurikka J, Tarkka M. Performance of three preoperative risk indices; CABDEAL, EuroSCORE and Cleveland models in a prospective coronary bypass database. Eur J Cardiothorac Surg. 2002;21:406–410. doi: 10.1016/s1010-7940(02)00007-6. [DOI] [PubMed] [Google Scholar]
- 26.Fox AA, Shernan SK, Collard CD, Liu KY, Aranki SF, DeSantis SM, Jarolim P, Body SC. Preoperative B-type natriuretic peptide is as independent predictor of ventricular dysfunction and mortality after primary coronary artery bypass grafting. J Thorac Cardiovasc Surg. 2008;136:452–461. doi: 10.1016/j.jtcvs.2007.12.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fox AA, Muehlschlegel JD, Body SC, Shernan SK, Liu KY, Perry TE, Aranki SF, Cook EF, Marcantonio ER, Collard CD. Comparison of the utility of preoperative versus postoperative B-type natriuretic peptide for predicting hospital length of stay and mortality after primary coronary artery bypass grafting. Anesthesiology. 2010;112:842–851. doi: 10.1097/ALN.0b013e3181d23168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fox AA, Marcantonio ER, Collard CD, Thoma M, Perry TE, Shernan SK, Muehlschlegel JD, Body SC. Increased peak postoperative B-type natriuretic peptide predicts decreased longer-term physical function after primary coronary artery bypass graft surgery. Anesthesiology. 2011;114:807–816. doi: 10.1097/ALN.0b013e31820ef9c1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Das SR, Abdullah SM, Leonard D, Drazner MH, Khera A, McGuire DK, de Lemos JA. Association between renal function and circulating levels of natriuretic peptides (from the Dallas Heart Study) Am J Cardiol. 2008;102:1394–1398. doi: 10.1016/j.amjcard.2008.07.018. [DOI] [PubMed] [Google Scholar]
- 30.Wahl HG, Graf S, Renz H, Fassbinder W. Elimination of the cardiac natriuretic peptides B-type natriuretic peptide (BNP) and N-terminal proBNP by hemodialysis. Clin Chem. 2004;50:1071–1074. doi: 10.1373/clinchem.2003.030692. [DOI] [PubMed] [Google Scholar]
- 31.Foundation NK. K/DOQI clinical practice guidelines for chronic kidney disease Evaluation, classification, and stratification. Am J Kidney Dis. 2002;39:S1–S266. [PubMed] [Google Scholar]
- 32.Vickery S, Price CP, John RI, Abbas NA, Webb MC, Kempson ME, Lamb EJ. B-type natriuretic peptide (BNP), amino-terminal proBNP in patients with CKD: Relationship to renal function and left ventricular hypertrophy. Am J Kidney Dis. 2005;46:610–620. doi: 10.1053/j.ajkd.2005.06.017. [DOI] [PubMed] [Google Scholar]
- 33.Fox AA, Collard CD, Shernan SK, Seidman CE, Seidman JG, Liu KY, Muehlschlegel JD, Perry TE, Aranki SF, Lange C, Herman DS, Meitinger T, Lichtner P, Body SC. Natriuretic peptide system gene variants are associated with ventricular dysfunction after coronary artery bypass grafting. Anesthesiology. 2009;110:738–747. doi: 10.1097/aln.0b013e31819c7496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hsieh FY, Lavori PW. Sample-size calculations for the Cox proportional hazards regression model with nonbinary covariates. Control Clin Trials. 2000;21:552–560. doi: 10.1016/s0197-2456(00)00104-5. [DOI] [PubMed] [Google Scholar]
- 35.Harrell FE, Jr, Lee KL, Mark DB. Multivariable prognostic models: Issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med. 1996;15:361–387. doi: 10.1002/(SICI)1097-0258(19960229)15:4<361::AID-SIM168>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 36.Fleisher LA, Beckman JA, Brown KA, Calkins H, Chaikof E, Fleischmann KE, Freeman WK, Froehlich JB, Kasper EK, Kersten JR, Riegel B, Robb JF, Smith SC, Jr, Jacobs AK, Adams CD, Anderson JL, Antman EM, Buller CE, Creager MA, Ettinger SM, Faxon DP, Fuster V, Halperin JL, Hiratzka LF, Hunt SA, Lytle BW, Nishimura R, Ornato JP, Page RL, Tarkington LG, Yancy CW. ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery) Developed in collaboration with the American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Rhythm Society, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, and Society for Vascular Surgery. Circulation. 2007;116:e418–e499. doi: 10.1161/CIRCULATIONAHA.107.185699. [DOI] [PubMed] [Google Scholar]
- 37.Ryding AD, Kumar S, Worthington AM, Burgess D. Prognostic value of brain natriuretic peptide in noncardiac surgery: A meta-analysis. Anesthesiology. 2009;111:311–319. doi: 10.1097/ALN.0b013e3181aaeb11. [DOI] [PubMed] [Google Scholar]
- 38.Karthikeyan G, Moncur RA, Levine O, Heels-Ansdell D, Chan MT, Alonso-Coello P, Yusuf S, Sessler D, Villar JC, Berwanger O, McQueen M, Mathew A, Hill S, Gibson S, Berry C, Yeh HM, Devereaux PJ. Is a preoperative brain natriuretic peptide or N-terminal pro-B-type natriuretic peptide measurement an independent predictor of adverse cardiovascular outcomes within 30 days of noncardiac surgery? A systematic review and meta-analysis of observational studies. J Am Coll Cardiol. 2009;54:1599–1606. doi: 10.1016/j.jacc.2009.06.028. [DOI] [PubMed] [Google Scholar]
- 39.Rodseth RN, Lurati Buse GA, Bolliger D, Burkhart CS, Cuthbertson BH, Gibson SC, Mahla E, Leibowitz DW, Biccard BM. The predictive ability of preoperative B-type natriuretic peptide in vascular patients for major adverse cardiac events: An individual patient data meta-analysis. J Am Coll Cardiol. 2011;58:522–529. doi: 10.1016/j.jacc.2011.04.018. [DOI] [PubMed] [Google Scholar]
- 40.Lurati Buse GA, Koller MT, Burkhart C, Seeberger MD, Miodrag F. Predictive value of preoperative natriuretic peptide concentrations in adults undergoing surgery: Systematic review and meta-analysis. Anesth Analg. 2011;112:1019–1033. doi: 10.1213/ANE.0b013e31820f286f. [DOI] [PubMed] [Google Scholar]
- 41.Litton E, Ho KM. The use of preoperative brain natriuretic peptides as a predictor of adverse outcomes after cardiac surgery: A systematic review and meta-analysis. Eur J Cardiothorac Surg. 2012;41:525–534. doi: 10.1093/ejcts/ezr007. [DOI] [PubMed] [Google Scholar]
- 42.Hunt SA, Abraham WT, Chin MH, Feldman AM, Francis GS, Ganiats TG, Jessup M, Konstam MA, Mancini DM, Michl K, Oates JA, Rahko PS, Silver MA, Stevenson LW, Yancy CW, Antman EM, Smith SC, Jr, Adams CD, Anderson JL, Faxon DP, Fuster V, Halperin JL, Hiratzka LF, Jacobs AK, Nishimura R, Ornato JP, Page RL, Riegel B. ACC/AHA 2005 Guideline Update for the Diagnosis and Management of Chronic Heart Failure in the Adult: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation and Management of Heart Failure): Developed in collaboration with the American College of Chest Physicians and the International Society for Heart and Lung Transplantation: Endorsed by the Heart Rhythm Society. Circulation. 2005;112:e154. doi: 10.1161/CIRCULATIONAHA.105.167586. 35. [DOI] [PubMed] [Google Scholar]
- 43.Hunt SA, Abraham WT, Chin MH, Feldman AM, Francis GS, Ganiats TG, Jessup M, Konstam MA, Mancini DM, Michl K, Oates JA, Rahko PS, Silver MA, Stevenson LW, Yancy CW. 2009 Focused update incorporated into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines Developed in Collaboration With the International Society for Heart and Lung Transplantation. J Am Coll Cardiol. 2009;53:e1–e90. doi: 10.1016/j.jacc.2008.11.013. [DOI] [PubMed] [Google Scholar]
- 44.Felker GM, Hasselblad V, Hernandez AF, O’Connor CM. Biomarker-guided therapy in chronic heart failure: A meta-analysis of randomized controlled trials. Am Heart J. 2009;158:422–430. doi: 10.1016/j.ahj.2009.06.018. [DOI] [PubMed] [Google Scholar]
- 45.Apple FS, Wu AH, Jaffe AS, Panteghini M, Christenson RH. NACB Committee; IFCC C-SMCD: National Academy of Clinical Biochemistry and IFCC Committee for Standardization of Markers of Cardiac Damage Laboratory Medicine Practice Guidelines: Analytical issues for biomarkers of heart failure. Clin Biochem. 2008;41:222–226. doi: 10.1016/j.clinbiochem.2007.07.001. [DOI] [PubMed] [Google Scholar]