Abstract
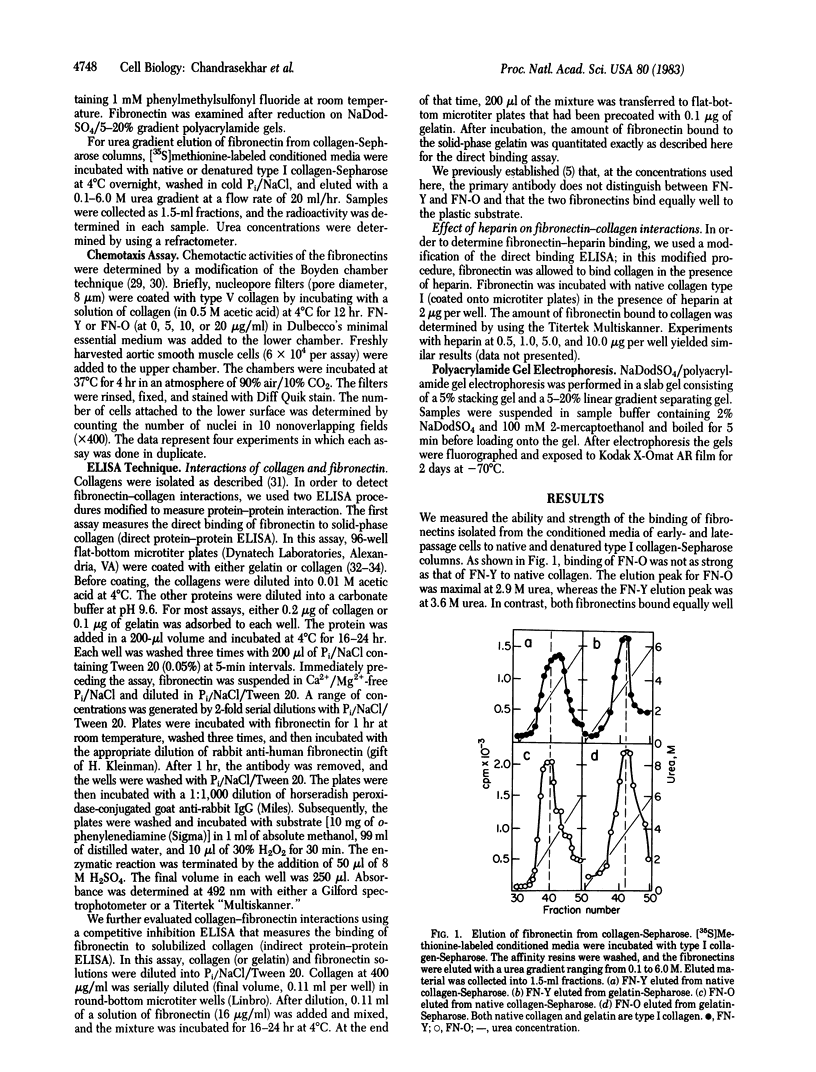
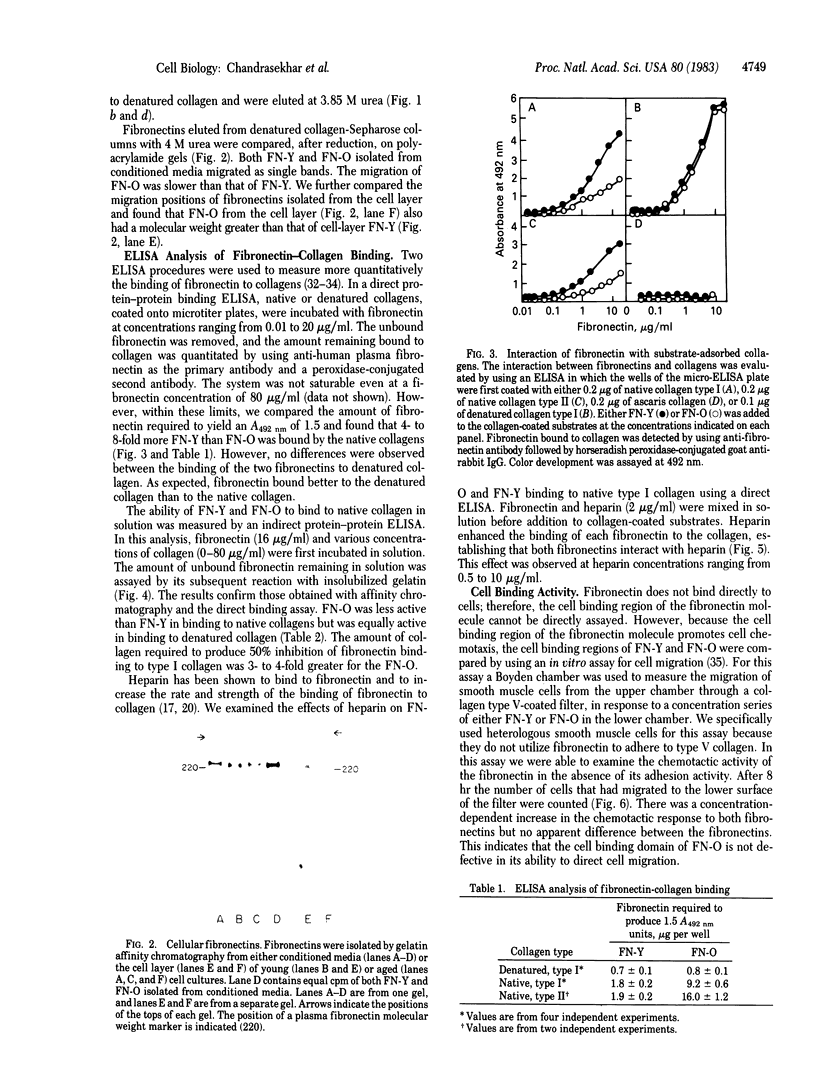
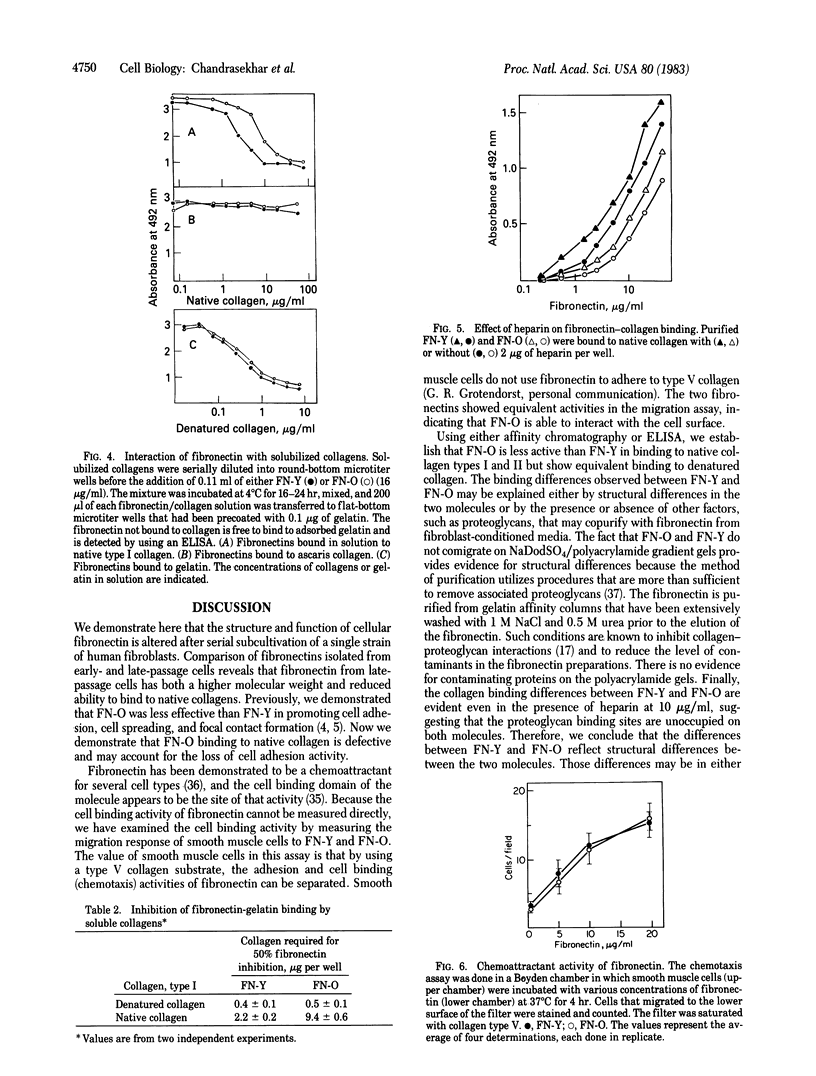
Fibronectins isolated fro early-passage and late-passage (in vitro aged) human fibroblasts were shown to differ in their ability to support cell adhesion and to influence cell morphology. Because fibroblast adhesion requires interactions between fibronectin, the cell surface, and the component of the extracellular matrix, we examined those functions in isolated cellular fibronectin. In comparison to fibronectin isolated from early-passage cells, fibronectin from late-passage cells bound poorly to native collagen types I and II. No differences were observed in the binding of the two fibronectins to denatured collagen. The binding of both fibronectins to native collagen was similarly promoted by heparin. Cell binding activity was evaluated by using a Boyden chamber assay to measure chemotaxis in response to either fibronectin. No differences were detected in cell binding. Comparisons of molecular weights by NaDodSO4/polyacrylamide gel electrophoresis reveals that fibronectin from late-passage cells is larger than that from early-passage cells. That difference is observed both in fibronectins isolated from conditioned media and in fibronectins isolated from the cell layer. These data support the hypothesis that late-passage cells produce a structurally and functionally distinct fibronectin. The defective binding to native collagen may account for some aspects of the aged phenotype.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arneson M. A., Hammerschmidt D. E., Furcht L. T., King R. A. A new form of Ehlers-Danlos syndrome. Fibronectin corrects defective platelet function. JAMA. 1980 Jul 11;244(2):144–147. [PubMed] [Google Scholar]
- Atherton B. T., Hynes R. O. A difference between plasma and cellular fibronectins located with monoclonal antibodies. Cell. 1981 Jul;25(1):133–141. doi: 10.1016/0092-8674(81)90237-3. [DOI] [PubMed] [Google Scholar]
- Azencott R., Courtois Y. Age-related differences in intercellular adhesion for chick fibroblasts cultured in vitro. Exp Cell Res. 1974 May;86(1):69–74. doi: 10.1016/0014-4827(74)90649-1. [DOI] [PubMed] [Google Scholar]
- Baumann H., Eldredge D. Dexamethasone increases the synthesis and secretion of a partially active fibronectin in rat hepatoma cells. J Cell Biol. 1982 Oct;95(1):29–40. doi: 10.1083/jcb.95.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandrasekhar S., Millis A. J. Fibronectin from aged fibroblasts is defective in promoting cellular adhesion. J Cell Physiol. 1980 Apr;103(1):47–54. doi: 10.1002/jcp.1041030108. [DOI] [PubMed] [Google Scholar]
- Chandrasekhar S., Norton E., Millis A. J., Izzard C. S. Functional changes in cellular fibronectin from late passage fibroblasts in vitro. Cell Biol Int Rep. 1983 Jan;7(1):11–21. doi: 10.1016/0309-1651(83)90100-5. [DOI] [PubMed] [Google Scholar]
- Crouch E., Balian G., Holbrook K., Duksin D., Bornstein P. Amniotic fluid fibronectin. Characterization and synthesis by cells in culture. J Cell Biol. 1978 Sep;78(3):701–715. doi: 10.1083/jcb.78.3.701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dessau W., Adelmann B. C., Timpl R. Identification of the sites in collagen alpha-chains that bind serum anti-gelatin factor (cold-insoluble globulin). Biochem J. 1978 Jan 1;169(1):55–59. doi: 10.1042/bj1690055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunham J. S., Hynes R. O. Differences in the sulfated macromolecules synthesized by normal and transformed hamster fibroblasts. Biochim Biophys Acta. 1978 Jan 19;506(2):242–255. doi: 10.1016/0005-2736(78)90395-4. [DOI] [PubMed] [Google Scholar]
- Engvall E., Perlmann P. Enzyme-linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry. 1971 Sep;8(9):871–874. doi: 10.1016/0019-2791(71)90454-x. [DOI] [PubMed] [Google Scholar]
- Engvall E., Ruoslahti E. Binding of soluble form of fibroblast surface protein, fibronectin, to collagen. Int J Cancer. 1977 Jul 15;20(1):1–5. doi: 10.1002/ijc.2910200102. [DOI] [PubMed] [Google Scholar]
- Grinnell F., Minter D. Attachment and spreading of baby hamster kidney cells to collagen substrata: effects of cold-insoluble globulin. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4408–4412. doi: 10.1073/pnas.75.9.4408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grotendorst G. R., Seppä H. E., Kleinman H. K., Martin G. R. Attachment of smooth muscle cells to collagen and their migration toward platelet-derived growth factor. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3669–3672. doi: 10.1073/pnas.78.6.3669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi M., Yamada K. M. Differences in domain structures between plasma and cellular fibronectins. J Biol Chem. 1981 Nov 10;256(21):11292–11300. [PubMed] [Google Scholar]
- Hayashi M., Yamada K. M. Differences in domain structures between plasma and cellular fibronectins. J Biol Chem. 1981 Nov 10;256(21):11292–11300. [PubMed] [Google Scholar]
- Hedman K., Johansson S., Vartio T., Kjellén L., Vaheri A., Hök M. Structure of the pericellular matrix: association of heparan and chondroitin sulfates with fibronectin-procollagen fibers. Cell. 1982 Mar;28(3):663–671. doi: 10.1016/0092-8674(82)90221-5. [DOI] [PubMed] [Google Scholar]
- Jilek F., Hörmann H. Fibronectin (cold-insoluble globulin), VI. Influence of heparin and hyaluronic acid on the binding of native collagen. Hoppe Seylers Z Physiol Chem. 1979 Apr;360(4):597–603. doi: 10.1515/bchm2.1979.360.1.597. [DOI] [PubMed] [Google Scholar]
- Kelley R. O., Vogel K. G., Crissman H. A., Lujan C. J., Skipper B. E. Development of the aging cell surface. Reduction of gap junction-mediated metabolic cooperation with progressive subcultivation of human embryo fibroblasts (IMR-90). Exp Cell Res. 1979 Mar 1;119(1):127–143. doi: 10.1016/0014-4827(79)90342-2. [DOI] [PubMed] [Google Scholar]
- Kleinman H. K., Klebe R. J., Martin G. R. Role of collagenous matrices in the adhesion and growth of cells. J Cell Biol. 1981 Mar;88(3):473–485. doi: 10.1083/jcb.88.3.473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleinman H. K., McGoodwin E. B., Martin G. R., Klebe R. J., Fietzek P. P., Woolley D. E. Localization of the binding site for cell attachment in the alpha1(I) chain of collagen. J Biol Chem. 1978 Aug 25;253(16):5642–5646. [PubMed] [Google Scholar]
- Laterra J., Culp L. A. Differences in hyaluronate binding to plasma and cell surface fibronectins. Requirement for aggregation. J Biol Chem. 1982 Jan 25;257(2):719–726. [PubMed] [Google Scholar]
- Linsenmayer T. F., Gibney E., Toole B. P., Gross J. Cellular adhesion to collagen. Exp Cell Res. 1978 Oct 15;116(2):470–474. doi: 10.1016/0014-4827(78)90473-1. [DOI] [PubMed] [Google Scholar]
- McDonald J. A., Broekelmann T. J., Kelley D. G., Villiger B. Gelatin-binding domain-specific anti-human plasma fibronectin Fab' inhibits fibronectin-mediated gelatin binding but not cell spreading. J Biol Chem. 1981 Jun 10;256(11):5583–5587. [PubMed] [Google Scholar]
- McDonald J. A., Kelley D. G. Degradation of fibronectin by human leukocyte elastase. Release of biologically active fragments. J Biol Chem. 1980 Sep 25;255(18):8848–8858. [PubMed] [Google Scholar]
- Millis A. J., Hoyle M., Field B. Human fibroblast conditioned media contains growth-promoting activities for low density cells. J Cell Physiol. 1977 Oct;93(1):17–24. doi: 10.1002/jcp.1040930104. [DOI] [PubMed] [Google Scholar]
- Mosesson M. W., Amrani D. L. The structure and biologic activities of plasma fibronectin. Blood. 1980 Aug;56(2):145–158. [PubMed] [Google Scholar]
- Mosher D. F. Fibronectin. Prog Hemost Thromb. 1980;5:111–151. [PubMed] [Google Scholar]
- Pearlstein E., Gold L. I., Garcia-Pardo A. Fibronectin: a review of its structure and biological activity. Mol Cell Biochem. 1980 Feb 8;29(2):103–128. doi: 10.1007/BF00220304. [DOI] [PubMed] [Google Scholar]
- Pearlstein E. Substrate activation of cell adhesion factor as a prerequisite for cell attachment. Int J Cancer. 1978 Jul 15;22(1):32–35. doi: 10.1002/ijc.2910220108. [DOI] [PubMed] [Google Scholar]
- Perkins M. E., Ji T. H., Hynes R. O. Cross-linking of fibronectin to sulfated proteoglycans at the cell surface. Cell. 1979 Apr;16(4):941–952. doi: 10.1016/0092-8674(79)90109-0. [DOI] [PubMed] [Google Scholar]
- Postlethwaite A. E., Snyderman R., Kang A. H. The chemotactic attraction of human fibroblasts to a lymphocyte-derived factor. J Exp Med. 1976 Nov 2;144(5):1188–1203. doi: 10.1084/jem.144.5.1188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rennard S. I., Berg R., Martin G. R., Foidart J. M., Robey P. G. Enzyme-linked immunoassay (ELISA) for connective tissue components. Anal Biochem. 1980 May 1;104(1):205–214. doi: 10.1016/0003-2697(80)90300-0. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E., Engvall E. Complexing of fibronectin glycosaminoglycans and collagen. Biochim Biophys Acta. 1980 Aug 13;631(2):350–358. doi: 10.1016/0304-4165(80)90308-6. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E., Engvall E., Hayman E. G. Fibronectin: current concepts of its structure and functions. Coll Relat Res. 1981;1(1):95–128. doi: 10.1016/s0174-173x(80)80011-2. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E., Hayman E. G. Two active sites with different characteristics in fibronectin. FEBS Lett. 1979 Jan 15;97(2):221–224. doi: 10.1016/0014-5793(79)80088-5. [DOI] [PubMed] [Google Scholar]
- Sekiguchi K., Hakomori S. Functional domain structure of fibronectin. Proc Natl Acad Sci U S A. 1980 May;77(5):2661–2665. doi: 10.1073/pnas.77.5.2661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seppä H. E., Yamada K. M., Seppä S. T., Silver M. H., Kleinman H. K., Schiffmann E. The cell binding fragment of fibronectin is chemotactic for fibroblasts. Cell Biol Int Rep. 1981 Aug;5(8):813–819. doi: 10.1016/0309-1651(81)90253-8. [DOI] [PubMed] [Google Scholar]
- Stathakis N. E., Mosesson M. W. Interactions among heparin, cold-insoluble globulin, and fibrinogen in formation of the heparin-precipitable fraction of plasma. J Clin Invest. 1977 Oct;60(4):855–865. doi: 10.1172/JCI108840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teng M. H., Rifkin D. B. Fibronectin from chicken embryo fibroblasts contains covalently bound phosphate. J Cell Biol. 1979 Mar;80(3):784–791. doi: 10.1083/jcb.80.3.784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaheri A., Mosher D. F. High molecular weight, cell surface-associated glycoprotein (fibronectin) lost in malignant transformation. Biochim Biophys Acta. 1978 Sep 18;516(1):1–25. doi: 10.1016/0304-419x(78)90002-1. [DOI] [PubMed] [Google Scholar]
- Yamada K. M., Kennedy D. W. Fibroblast cellular and plasma fibronectins are similar but not identical. J Cell Biol. 1979 Feb;80(2):492–498. doi: 10.1083/jcb.80.2.492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada K. M., Kennedy D. W., Kimata K., Pratt R. M. Characterization of fibronectin interactions with glycosaminoglycans and identification of active proteolytic fragments. J Biol Chem. 1980 Jul 10;255(13):6055–6063. [PubMed] [Google Scholar]
- Yamada K. M., Weston J. A. Isolation of a major cell surface glycoprotein from fibroblasts. Proc Natl Acad Sci U S A. 1974 Sep;71(9):3492–3496. doi: 10.1073/pnas.71.9.3492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto K., Yamamoto M., Ooka H. Cell surface changes associated with aging of chick embryo fibroblasts in culture. Exp Cell Res. 1977 Aug;108(1):87–93. doi: 10.1016/s0014-4827(77)80013-x. [DOI] [PubMed] [Google Scholar]




