Abstract
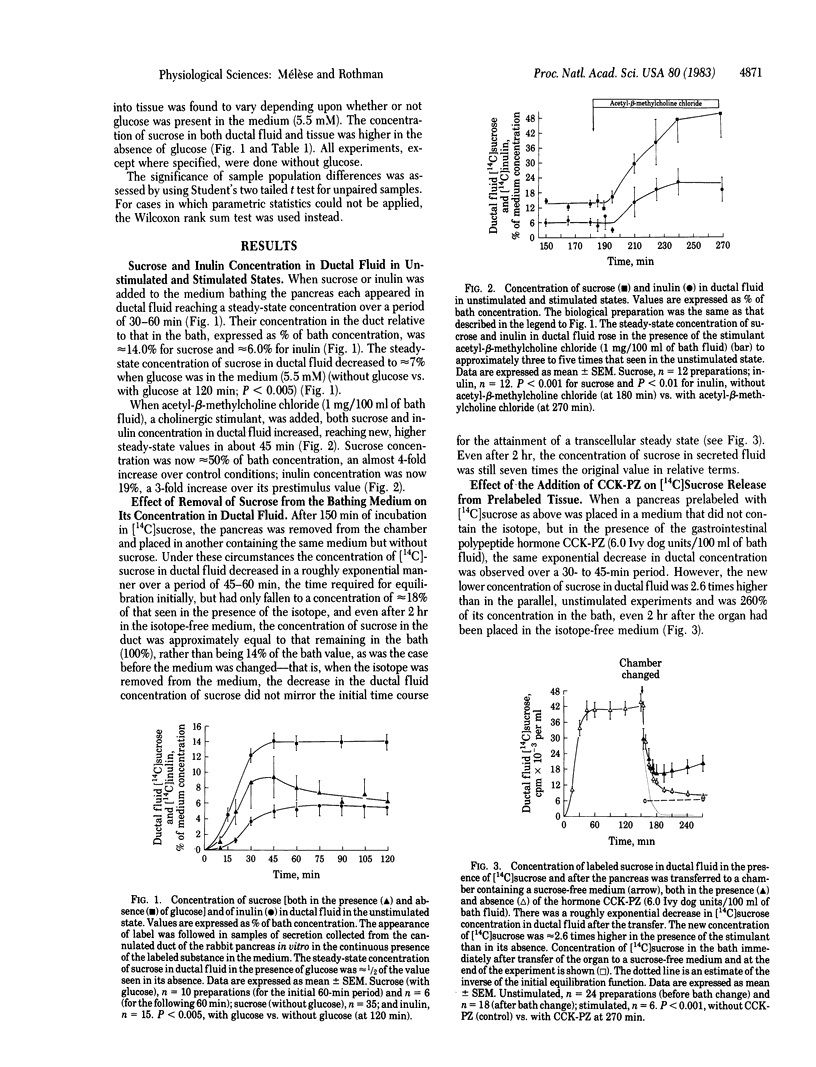
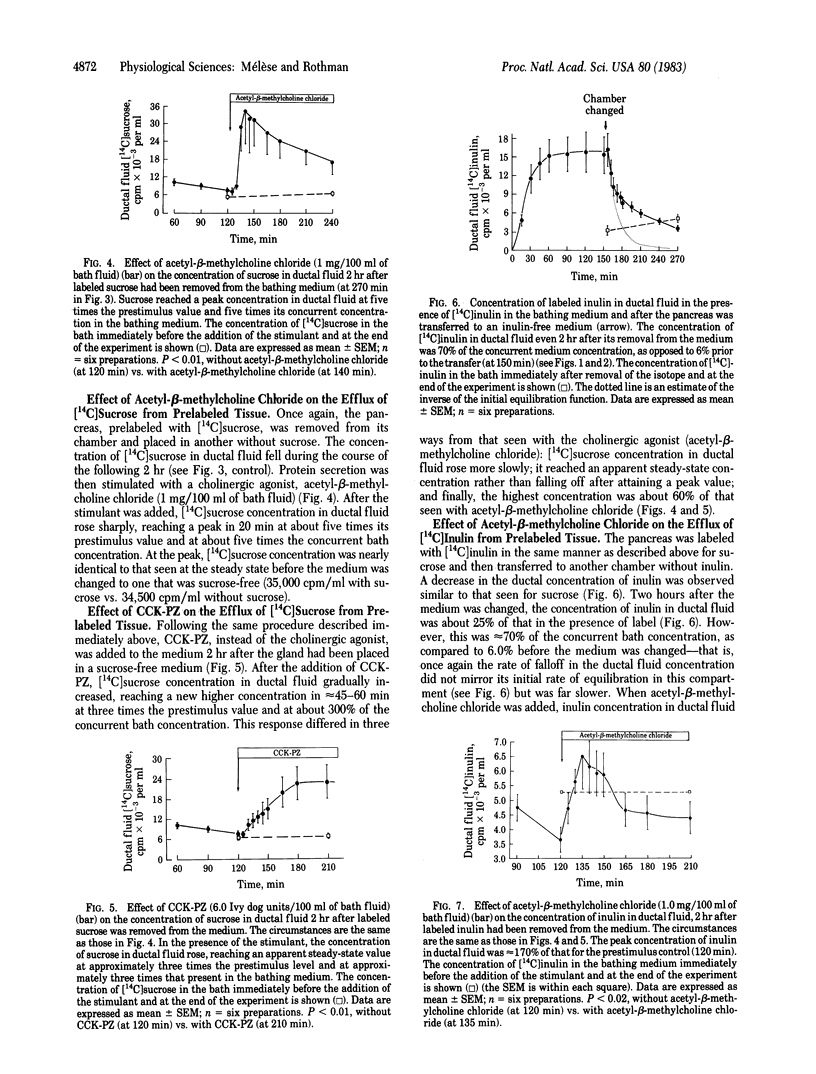
The pancreatic epithelium is permeable to both sucrose and inulin and becomes more permeable when protein secretion by the gland is stimulated. Because these molecules are not thought to enter cells, it has been assumed that their movement across the epithelium from interstitial to ductal fluid, as well as the increase in that flux that is observed during augmented protein secretion, is due to their passage through paracellular shunts. In the present experiments we have considered the alternative possibility that sucrose and inulin travel through the cells of the secretory epithelium instead of, or in addition to, their passage through paracellular shunts. The data support this view and suggest that the pancreas is unusually permeable to water soluble molecules of substantial size by means of a transcellular pathway.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Augustus J., Bijman J., van Os C. H. Electrical resistance of rabbit submaxillary main duct: a tight epithelium with leaky cell membranes. J Membr Biol. 1978 Oct 19;43(2-3):203–226. doi: 10.1007/BF01933479. [DOI] [PubMed] [Google Scholar]
- EAGLE H. Amino acid metabolism in mammalian cell cultures. Science. 1959 Aug 21;130(3373):432–437. doi: 10.1126/science.130.3373.432. [DOI] [PubMed] [Google Scholar]
- Jansen J. W., de Pont J. J., Bonting S. L. Transepithelial permeability in the rabbit pancreas. Biochim Biophys Acta. 1979 Feb 20;551(1):95–108. doi: 10.1016/0005-2736(79)90356-0. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- ROTHMAN S. S., BROOKS F. P. ELECTROLYTE SECRETION FROM RABBIT PANCREAS IN VITRO. Am J Physiol. 1965 Jun;208:1171–1176. doi: 10.1152/ajplegacy.1965.208.6.1171. [DOI] [PubMed] [Google Scholar]
- ROTHMAN S. S. EXOCRINE SECRETION FROM THE ISOLATED RABBIT PANCREAS. Nature. 1964 Oct 3;204:84–85. doi: 10.1038/204084a0. [DOI] [PubMed] [Google Scholar]
- Rossier M., Rothman S. S. Kinetics of Na+ uptake and transcellular transit by the pancreas. Am J Physiol. 1975 Apr;228(4):1199–1205. doi: 10.1152/ajplegacy.1975.228.4.1199. [DOI] [PubMed] [Google Scholar]
- Rothman S. S. Passage of proteins through membranes--old assumptions and new perspectives. Am J Physiol. 1980 May;238(5):G391–G402. doi: 10.1152/ajpgi.1980.238.5.G391. [DOI] [PubMed] [Google Scholar]
- Rothman S. S., Wells H. Selective effects of dietary egg white trypsin inhibitor on pancreatic enzyme secretion. Am J Physiol. 1969 Mar;216(3):504–507. doi: 10.1152/ajplegacy.1969.216.3.504. [DOI] [PubMed] [Google Scholar]
- Swanson C. H., Solomon A. K. A micropuncture investigation of the whole tissue mechanism of electrolyte secretion by the in vitro rabbit pancreas. J Gen Physiol. 1973 Oct;62(4):407–429. doi: 10.1085/jgp.62.4.407. [DOI] [PMC free article] [PubMed] [Google Scholar]



