Abstract
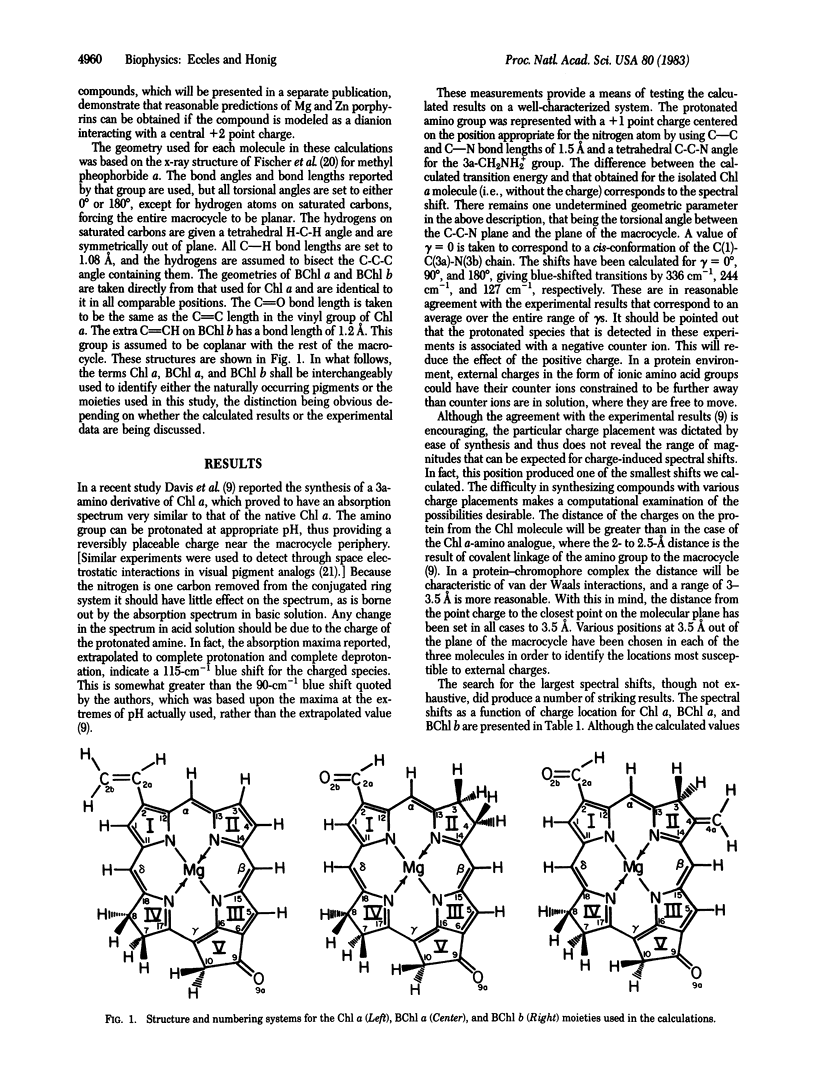
In this paper we propose that the large spectroscopic red shifts observed for chlorophyll (Chl) and bacteriochlorophyll (BChl) in vivo may be due to charged amino acids in the binding site. Molecular orbital calculations of the transition energies of Chl in the field of external charges are carried out. The calculated wavelength shifts induced by these charges are comparable in magnitude to those observed in vivo. Moreover the size of the shifts increases in the order BChl b > BChl a > Chl a, which is the observed trend. The ability of the calculations to account for both the absolute and relative magnitudes of the wavelength shifts argues for the validity of the model. Further indirect support comes from the recent demonstration that charged amino acids are responsible for the colors of visual pigments and bacteriorhodopsin. In addition to their effects on spectra the presence of external charges induces large changes in the ionization potential of Chl molecules and thus might explain the in vivo alteration of the oxidation potentials in reaction centers.
Keywords: photosynthesis, electrostatic effects
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Clayton R. K., Clayton B. J. B850 pigment-protein complex of Rhodopseudomonas sphaeroides: Extinction coefficients, circular dichroism, and the reversible binding of bacteriochlorophyll. Proc Natl Acad Sci U S A. 1981 Sep;78(9):5583–5587. doi: 10.1073/pnas.78.9.5583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fajer J., Davis M. S., Brune D. C., Spaulding L. D., Borg D. C., Forman A. Chlorophyll radicals and primary events. Brookhaven Symp Biol. 1976 Jun 7;(28):74–104. [PubMed] [Google Scholar]
- Honig B., Ebrey T., Callender R. H., Dinur U., Ottolenghi M. Photoisomerization, energy storage, and charge separation: a model for light energy transduction in visual pigments and bacteriorhodopsin. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2503–2507. doi: 10.1073/pnas.76.6.2503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honig B., Greenberg A. D., Dinur U., Ebrey T. G. Visual-pigment spectra: implications of the protonation of the retinal Schiff base. Biochemistry. 1976 Oct 19;15(21):4593–4599. doi: 10.1021/bi00666a008. [DOI] [PubMed] [Google Scholar]
- Kakitani T., Honig B., Crofts A. R. Theoretical studies of the electrochromic response of carotenoids in photosynthetic membranes. Biophys J. 1982 Jul;39(1):57–63. doi: 10.1016/S0006-3495(82)84490-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthews B. W., Fenna R. E., Bolognesi M. C., Schmid M. F., Olson J. M. Structure of a bacteriochlorophyll a-protein from the green photosynthetic bacterium Prosthecochloris aestuarii. J Mol Biol. 1979 Jun 25;131(2):259–285. doi: 10.1016/0022-2836(79)90076-7. [DOI] [PubMed] [Google Scholar]
- Norris J. R., Uphaus R. A., Crespi H. L., Katz J. J. Electron spin resonance of chlorophyll and the origin of signal I in photosynthesis. Proc Natl Acad Sci U S A. 1971 Mar;68(3):625–628. doi: 10.1073/pnas.68.3.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parson W. W. Photosynthetic bacterial reaction centers: interactions among the bacteriochlorophylls and bacteriopheophytins. Annu Rev Biophys Bioeng. 1982;11:57–80. doi: 10.1146/annurev.bb.11.060182.000421. [DOI] [PubMed] [Google Scholar]
- Pearlstein R. M., Hemenger R. P. Bacteriochlorophyll electronic transition moment directions in bacteriochlorophyll a-protein. Proc Natl Acad Sci U S A. 1978 Oct;75(10):4920–4924. doi: 10.1073/pnas.75.10.4920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rafferty C. N., Bolt J., Sauer K., Clayton R. K. Photooxidation of antenna bacteriochlorophyll in chromatophores from carotenoidless mutant Rhodopseudomonas sphaeroides and the attendant loss of dimeric exciton interaction. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4429–4432. doi: 10.1073/pnas.76.9.4429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wasielewski M. R., Norris J. R., Shipman L. L., Lin C. P., Svec W. A. Monomeric chlorophyll a enol: Evidence for its possible role as the primary electron donor in photosystem I of plant photosynthesis. Proc Natl Acad Sci U S A. 1981 May;78(5):2957–2961. doi: 10.1073/pnas.78.5.2957. [DOI] [PMC free article] [PubMed] [Google Scholar]



