Abstract
Current cancer treatments may create profound iatrogenic outcomes. The adverse effects of these treatments still remain, as the serious problems that practicing physicians have to cope with in clinical practice. Although, non-specific cytotoxic agents constitute an effective treatment modality against cancer cells, they also tend to kill normal, quickly dividing cells. On the other hand, therapies targeting the genome of the tumors are both under investigation, and some others are already streamlined to clinical practice. Several approaches have been investigated in order to find a treatment targeting the cancer cells, while not affecting the normal cells.
Suicide gene therapy is a therapeutic strategy, in which cell suicide inducing transgenes are introduced into cancer cells. The two major suicide gene therapeutic strategies currently pursued are: cytosine deaminase/5-fluorocytosine and the herpes simplex virus/ganciclovir. The novel strategies include silencing gene expression, expression of intracellular antibodies blocking cells’ vital pathways, and transgenic expression of caspases and DNases. We analyze various elements of cancer cells’ suicide inducing strategies including: targets, vectors, and mechanisms. These strategies have been extensively investigated in various types of cancers, while exploring multiple delivery routes including viruses, non-viral vectors, liposomes, nanoparticles, and stem cells. We discuss various stages of streamlining of the suicide gene therapy into clinical oncology as applied to different types of cancer.
Moreover, suicide gene therapy is in the center of attention as a strategy preventing cancer from developing in patients participating in the clinical trials of regenerative medicine. In oncology, these clinical trials are aimed at regenerating, with the aid of stem cells, of the patients’ organs damaged by pathologic and/or iatrogenic factors. However, the stem cells carry the risk of neoplasmic transformation. We discuss cell suicide inducing strategies aimed at preventing stem cell-originated cancerogenesis.
Keywords: Suicide gene therapy, Cancer, Transporters, Viral vectors, Non-viral vectors, Regenerative medicine
Introduction
The cornerstone of cancer treatment still remains the integrated multimodality approach with chemotherapy, radiotherapy and surgery. Neo-adjuvant and adjuvant treatments are applicable in several cancer types and complete surgical resection offers prolonged survival. However, chemotherapy and radiotherapy have iatrogenic adverse effects associated with the current mode of administration. As an example may serve cisplatin based chemotherapy. It may cause nausea and emesis to the degree, which may require reduction of the doses below those most effective or interruption of the therapy. As another example may serve radiotherapy of. In addition to the iatrogenic symptoms of radiation sickness, the patients’ treatment has may have to be postponed until the bone marrow recovers [1]. Moreover, bone marrow suppression in many cases requires hospitalization with additional costs for the national health system [2,3].
In an effort to reduce the adverse effects, novel routes of administration were investigated in many types of cancer with different strategies, with the main concept being to localize the treatment [4-14]. Until now, the approved cancer treatment modality for chemotherapy drugs is intravenous and in some cases the oral route. However, breakthroughs in revealing the genome mutations of tumors have indicated specific mutated populations. The era that we are currently experiencing is called “the era of the genome” with multiple genetic mutations and pathways being discovered in populations of people [15]. In these populations certain pathways were altered and pharmaceuticals were designed in order to target these mutations [16-18]. Pharmacogenetics links specific genetic traits of with efficacy of specific drugs [19]. The major limitations of this treatment approach are small populations of people carrying with specific gene defined high susceptibility to cancer. Continued investigations of cancer evolution keep revealing mechanisms that maybe utilized in order to block chemotherapy resistance and sensitize tumors to chemotherapy and radiotherapy [20].
The major breakthrough in gene therapy modality was designing suicide gene therapy. The safety and effectiveness of cancer treatment became reality through this modality. In the suicide gene therapy, the therapeutic transgenes have the ability to convert a non-toxic pro-drug, which penetrates the tumor, into cytotoxic drug or to express the toxic gene expression product. This action is taking place inside the tumor with the help of an administered viral or bacterial cell suicide inducing gene. The normal cells are not affected [21-23]. The suicide gene therapy has been investigated in several cancer types; a) colon [8,24,25], b) liver [9,26,27], c) lung [28,29], d) medulloblastomas [30], e) neuroendocrine [31], f) spinal cord tumors [32], g) prostate [33], h) breast [34,35], i) bladder [36], j) brain [37], k) gliomas [38-40], l) head and neck [41], m) sarcomas [42], and ovaries [108,111-112]. The suicide gene therapy has been investigated as; a) anti-vascular endothelial treatment [43,44], b) immune stimulation with interleukin- 7 (IL-7) [45] and interleukin-12 (IL-12) [46]. Furthermore, suicide gene therapy has been proven to be more efficient in chemotherapy resistant cancer cell lines [47] and to enhance radiotherapy [48]. Additional control of micrometastases, through the efficient local control of microenvironment, has also been achieved with suicide gene therapy [49]. Finally, suicide gene therapy has been explored by using the state of the art technology of nanoparticles, either as a mode to passively transport the suicide gene formulation, or as a mode to efficiently display the diffusion of the formulation [43,50,51].
Basic Strategies to Induce Cancer Cell Suicide
Cytosine deaminase and thymidine kinase
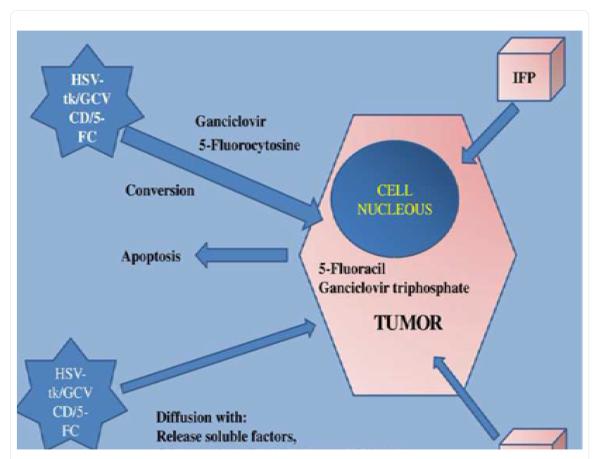
The basic concept of suicide gene therapy is the introduction of viral or bacterial genes into tumors cells, which in turn convert a non-toxic pro-drug to a toxic one (Figure1). There are systems that have been extensively investigated: the cytosine deaminase gene (CD) of Escherechia coli, which converts the pro-drug 5-Fluorocytosine (5-FC) to 5-Fluorouracil (5-FU) and the herpes simplex virus thymidine kinase gene (HSV-tk), which converts ganciclovir (GCV) to ganciclovir monophosphate, converted by the cancer cells’ enzymes to ganciclovir triphosphate. The HSV-tk/GCV system induced a delay in the S phase and G2-phase and the apoptosis observed is not a direct result of the system activation, but a result of the delayed proliferation process. In addition, mitochondrial damage was observed to be induced [55] and caspase-8, Chk1 activation was associated with extensive cell death [56,57].
Figure 1.
IFP; Interstitial fluid pressure is increased inside the tumor and prevents drug formulation/molecules from efficiently diffusing within the tumor, HAS-tk; Herpes simplex virus, CD; cytosine deaminase, 5-FC; 5-Fluorocytosine, GCV; Ganciclovir and EPR; Enhanced permeability effect.
The cytosine deaminase can be found in fungi and bacteria, but not in mammalian cells. The non-toxic 5-FC is transformed to the toxic 5-FU. The cellular enzymes will then further process the formulation to three cytotoxic antimetabolites: 1) 5-FdUTP, 2) 5-FUTP and 3) 5-FdUMP. The CD suicide therapy cytotoxic effects are based on three properties of the system: a) formation of (5-FU) RNA - b) 5-FU DNA complexes and c) thymidylate synthase. The mitochondrial pathways are down-regulated through the control of Bcl-2 [58] and heat shock protein 90-beta is activated, contributing to tumor regression [59]. Another methodology investigated was to target the erb-2 oncogene over-expression with CD-5-FU over [60]. The cytosine deaminase/5-FU system has been improved by including the gene uracil phosphoribosyltransferase (UPRT) [61]. Another method was to combine the UPRT with the yeast enzymatic activities of CD [28]. Finally, the combination of both systems delivered simultaneously has also been achieved [62,29]. Furthermore, there are other systems that are not widely used such as: a) Varicella-Zoster virus thymidine kinase (VZV-tk), b) Purine nucleoside phosphorylase (PNP) , c) Carboxypeptidase A, d) Carboxypeptidase G2, e) Linamarase, f) Beta-Galatosidase, g) Nitroreductase and h) Hepatic cytochrome P450-2B1
Transport systems
There are currently two main vehicles utilized to deliver cell suicide genes: viral vectors and non-viral vectors. The viral vectors have been derived from poxviruses, herpes simplex virus, lentivirus, retroviruses, vaccinia virus, adenovirus and adeno-associated viruses. Oncolytic viruses have been also used and they are capable of bearing tumor specificity. However, the major problem is generation of neutralizing antibodies (nAbs), which are formed due to the immunogenicity and inflammatory toxicity. The formation of nAbs is dependent on the viral vector load and viral vector type [63,64]. The non-viral vectors are widely used with three different approaches: 1) naked DNA, 2) physical methods and 3) chemical methods. Non-viral vectors include the cationic liposomes and cationic polymers. These two delivery systems have a positive charge, which interacts with the negative charge of the plasmid DNA. These are capable of transporting large amounts of DNA and can be modified in numerous ways according to the investigators’ design. Additional delivery systems have been designed for these vectors [65,66]. However; safety concerns of non-viral vectors are still under consideration for several cell lines and toxicity is correlated with the kDa size of the polymers [67]. Moreover, other non-viral have been investigated with: 1) exosomes, 2) erythrocyte ghosts, 3) bacteria, 4) bacteriophages, 5) virus-like particles, 6) antibodies, 7) immuno-liposomes. The bacteria that are usually used are; Clostridia, Salmonelle and Bifidibacteria. They have the ability to infect the hypoxic cancer cells and have presented efficient antitumor activity against chemo-resistant cancer cell lines [34,36].
Cellular vehicles, that have been proposed as transport systems, are summarized as follows: a) bone marrow stromal cells (MSCs) which exhibit the ability of homing to the tumor sites [39,68], b) neural stem cells (NSCs) which have an inherent tumor tropism [38,37], c) endothelial progenitor precursor and d) blood outgrowth endothelial cells. The last two target the vascular tumor tissue and are under investigation for clinical applications [43,44].
Currently nanoparticles are being investigated as efficient gene transporters for suicide gene therapy. They have the ability to efficiently diffuse within the tumor tissue with passive transportation. Their enhanced tumor accumulation is based on the enhanced permeability and retention effect (EPR) [69]. The nanocomplexes are diffused within the tumor due to the increased permeability of the tumor vessels, however; the low clearance prohibits the formulations from getting out. This methodology is mostly used for siRNA but not for DNA plasmid delivery.
An additional advantage of suicide gene therapy is the ability to display the distribution and transgene expression as the therapeutic gene can be used as a reporter gene. The suicide gene therapy system used for this application is the HSV-tk/GCV. The radiolabelled fluoro-nucleoside analogues of thymidine such as 1-(2-deoxy-2-fluoro-D-arabinofuranosyl)-5-iodouracil (FIAU) and radiolabelled 9-[4-fluoro-3-(hydroxymethyl)butyl]guanine (FHBG) can be used for HSV-tk substrates [70,71].
From the clinical practice point of view, of the great importance is the feature of the CD system, in which the conversion of the 5-FC to 5-FU can be quantified with the nuclear magnetic resonance spectroscopy (NMRS) [73,74].
Targeted delivery of suicide inducing genes
Several receptors on living cells have been used as targets for suicide gene therapy, such as; carcino-embryonic antigen (CEA) [29] and vascular endothelial growth factor receptor (VEGFR) [44,43]. Additional receptors that can targeted are: a) epidermal growth factor receptor (EGFR) [98], b) cluster of differentiation 44s (CD44s) [99], c) cluster of differentiation 133 (CD133 aka AC133) [100], c) folate receptor (FR) [101], d) transferrin receptor (TfR) or cluster differentiation 71 (CD71) [102], e) mucins [103], stage specific embryonic antigen 4 (SSEA-4) [104] and f) tumor resistance antigen 1-60 (TRA-1-60) [105]. Using promoters as a method to target specific over-expressed pathways has been used for; a) EGFR [104], b) carcino-embryonic antigen (CEA) [29], c) prostate specific antigen (PSA) [106], d) cyclooxygenase (Cox) [107], e) transferrin receptor (TfR) [108], f) Cytokeratin 18 and 19 [109] and g) Telomerase-hTERT [110]. Further investigation towards targeted suicide gene therapy is warranted and several efforts are currently in process [22,111] (Table 2).
Table 2.
Pathways and Promoters.
| Targeted Pathways | Promoters |
|---|---|
| Epidermal Growth Factor Receptors | Epidermal Growth Factor Receptors |
| Vascular Endothelial Growth Factor | Transferrin receptor |
| Carcino-embryonic antigen | Carcino-embryonic antigen |
| Transferrin receptor | Prostate specific antigen |
| Mucins | Telomerase-Htert |
| Cluster differentiation 44 | Cycloxiganase |
| Cluster differentiation 133 | Cytokeratin 18-19 |
| Stage specific embryonic antigen 4 | |
| Tumor resistance antigen 1-60 |
Bystander effect
In the bystander effect, the therapeutic consequences spread beyond the transfected tumor cells. In addition, distant tumor cells or tumor lesions have been observed to regress, although not directly transduced. There are currently five mechanisms investigated for the regression of untransfected cells due to bystander effects: a) release of soluble formulations, b) passage through gap junctions, c) passive transportation, d) stimulation of local microenvironment and e) endocytosis of apoptotic vesicles.
The CD/5-FU system demonstrates stronger local bystander effect than the HV-tk/GCV. The 5-FU diffuses efficiently within the tumor cells, and does not require cell to cell contact. The GCV trisphophate mediated bystander effect uses different mechanisms. An additional, mechanism which the HSV-tk/GCV exploits, is the local inflammation and devascularisation, which probably enhance the vascular permeability and the formulation diffuses more efficiently [52]. The distant tumor regression effect was initially attributed to the immune system stimulation [53]. However, tumor metastasis regression was also observed in immunodeficient SCID mice, which was associated with the release of soluble factors from the primary site of transfection [54]. Regarding the CD/5-FC system the distant tumor control has been linked to the stimulation of natural killer cells, CD4+ and CD8+. In summary, the CD/5-FC system does not use the cell to cell contact to diffuse efficiently, while the HSV-tk/GCV mainly uses the five previously mentioned mechanisms.
Gene-Immuno-therapy
In the study by Bondanza A. et al. [45] the interleukin -7 and -15 (IL-7, -15) were used in a leukemia model with herpes simplex virus thymidine kinase (TK). It was observed that thymidine kinase-positive HA-1- and H-Y- specific cytotoxic T cells differentiated into potent anti-leukemia effectors in vitro and in vivo in mouse model. Increased stimulation and infiltration of tumor with CD8+ was also observed.
In the study by Xu Y et al. [46] Lewis lung cancer cell lines and A549 were obtained and used for evaluation of a suicide gene therapy model as immuno-therapy (in vitro/in vivo). The recombinant adenovirus expressing horseradish peroxidase (HRP) mediated by human telomerase reverse transcriptase (hTERT) promoter (AdhTERTHRP) and murine interleukin-12 (mIL-12) under the control of Cytomegalovirus (CMV) promoter (AdCMVmIL-12) were developed for this study. The administration of Ad.hTERTHRP/indole-3-acetic acid (IAA) treatment and Ad.CMVmIL-12 resulted in enhanced tumor inhibition and survival improvement compared to Ad.hTERTHRP/IAA administration. Additional, evaluation of safety was performed with the collection of blood samples and measurement of alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatine (Cr) and blood urea nitrogen (BUN). No adverse effects were observed due to the suicide gene therapy. The same project investigated with or without adenovirus-mediated interleukin 12 (IL-12), a novel dual radiation-inducible and tumor-specific promoter system. This consisted of CArG elements and the human telomerase reverse transcriptase (hTERT) promoter in combination with adenovirus-mediated horseradish peroxidase (HRP)/indole-3-acetic (IAA) administration. The system was effectively activated with γ-irradiation. However, side effects were observed.
In the study by Finocchiaro L. M.E et al. [42] suicide gene therapy was investigated as a method of local immunostimulation after surgery. Canine sarcomas were treated surgically, while followed by lipoplexes carrying interferon-β (cIFN-β) and suicide genes co-administered with ganciclovir (ISG) were injected locally after surgerical excision. An additional boost was achieved with a vaccine with engineered hGM-CSF and hIL-2 secreting xenogeneic cells. This methodology was initially proposed for melanoma [80]. The concept is to control the microenvironment after surgery by two ways: firstly, to kill remaining cancer cells; and secondly, to prevent recurrence. Additionally, the authors observed control of distant metastases.
Further investigation of combined suicide gene therapy with immunotherapy was investigated by Marukawa Y. et. al. [27] were the herpes simplex virus thymidine kinase/ganciclovir (HSV-tk/GCV) was combined with monocyte chemo-attractant protein-1 (MCP-1). Additionally a recombinant adenovirus vector (rAd) harboring human MCP-1 gene and the membrane-spanning domain of the Chemokine (C-X3-C motif) ligand 1 (CX3CL1) gene were used. Increased numbers Mac-1, CD8a-positive, CD4+ and tumor necrosis factor (TNF) almost five-fold higher than with soluble MCP-1 were observed. These results indicate that membrane-bound form of monocyte chemo-attractant protein-1 enhances antitumor effects of suicide gene therapy.
Moreover, the MCP-1 was investigated with chemokine ligand 2 to form the chemokine (C-C motif) ligand 2 [Homo sapiens (human) (CCL2)/MCP-1 in order to enhance the antitumor effect of HSV-tk/GCV suicide gene therapy. This study was performed in mice. The results indicated that an adequate amount of CCL2/MCP-1 together with HSV-tk/GCV can induce T helper 1-polarized antitumor effects. Effective control of tumor microenvironment and metastasis was also observed [49] (Table 1).
Table 1.
Suicide Gene Therapy Studies.
| Author | Cells lines | Design | Result | Transport | Ref |
|---|---|---|---|---|---|
| Walther W. et al. |
MCF-7 HCT116 SKMel-5 Panc-1 |
In Vitro
In Vivo |
Effective against MCF-7 HCT116 (overexpressing claudin-3-, -4-) |
wtCPE optCPE |
[81] |
| Michaelsen S. R. et al. |
GLC-14, GLC-16, GLC-19, NCI-H69, H69-VP, H69-CPR, H69-DAU, H69-BCNU |
In Vitro
In Vivo |
Effective both in chemosensitive and chemoresistant cell lines |
INSM1 promoter- driven SG |
[47] |
| Mader R.M. et al. |
CCL227 (with low and Intermediate phenotypes) |
In Vitro | Effective with 100% Activation |
Adenoviral cosmids | [8] |
| Bondanza A. et al. |
Leukemia (mouse) |
In Vitro
In Vivo |
Effective with IL-7 receptor expression (HA-1-, H-Y-) |
Herpes simplex virus Thymidine kinase (tk) |
[45] |
| Xu Y. et al. | Lewis Lung Cancer A549 |
In Vitro
In Vivo |
Combination IL-12 and suicide gene therapy enhances the antitumor effect as a factor modifying the tumor microenvironment |
AdCMV(−), AdhTERTHRP, AdCMVmIL-12 |
[46] |
| Sia KC. et al. | HCC 26-1004 |
In Vitro
In Vivo |
Effective HSV-1 amplicon viral vector and 5-FU administration |
HSV-1 amplicon viral Vector coupled with yCD |
[9] |
| Li S. et al. | C17.2 NSC line |
In Vitro
In Vivo |
ATRA enhanced the HSV-tk/GCV | HSVtk/GCV | [30] |
| Finocchiaro M.E.L. et al. |
sarcoma | In Vivo | Effective Microenvironment Control and Distant metastasis |
Lipid-complexed plasmid Bearing IFN-β and suicide genes co-administered with ganciclovir (ISG) |
[42] |
| Leng A. et. al. |
Human colon carcinoma (Lovo) cell line |
In Vitro
In Vivo |
Anti-VEGF-A- Suicide gene therapy |
5-FC, CPNP-shVEGF-CDTK | [43] |
| Liu T. et. al. | SGC7901 human gastric Cancer cell line |
In vitro
In Vivo |
Anti-VEGF- Suicide gene therapy |
5-FC, triple gene vector ExpressingVEGF-shRNA and fusion suicide gene yCDglyTK delivered by CPNPs |
[44] |
| Finzi et. al. | Human HT29 and murine DHDK12 pro-b |
In Vitro
In Vivo |
MTX, aphidicolin andara-C. The rate of apoptosis increased two-fold in MTX-treated DHDK12 cells after treatment with GCV. |
HSVtk-GCV | [25] |
| Niu H. et. al. | VX2 liver cancer | In Vivo | Effective with lipiodol embolism and WTp53 |
TK/CD plus intraperitoneal Injection of GCV at 100mg/(kg.d) and 5-FC at 500mg/(kg.d) |
[26] |
| Marukawa Y. et. al. |
HCC |
In vitro
In Vivo |
Effective Mac-1, CD4, CD8a-positive and TNF increase |
-HSV-tk/GCV and MCP-1 -rAd harboring human MCP-1 and the membrane-spanning domain of the tumor cell surface |
[27] |
| Kosaka H. t. al. |
9L rat glioma cells and 293 cells |
In vitro
In Vivo |
MSC-EGFP or MSC-CD-5-FCresulted in significant prolongation of survival |
AdexCAEGFP AdexCACD |
[39] |
| Schmidt M. et. al. |
Head and Neck squamous carcinoma cell line FADU |
In Vitro | Effective with deletion Mutant of ETA as a Targetgene |
Gene Switch System | [41] |
| Cottin S. et. al. |
Glioblastoma | In Vitro | Effective against Cx43 cytoplasmic localization |
Lentiviral delivery of HSV-tk/GCV |
[40] |
| Kakinoki K. et. al. |
HCC |
In vitro
In Vivo |
Effective against metastasis and control of microenvironment |
CCL2/MCP-1 HSV-tk/GCV |
[49] |
| Sun X. et. al. | R3327-AT rat prostate Carcinoma cells |
In Vitro
In Vivo |
Effective against hypoxic cells |
Bifunctionalcytosine deaminase (CD) and uracil phosphoribosyltransferase (UPRT) with 5-FC and radiotherapy |
[48] |
| Amano S. et. al. |
C6 glioma cells |
In Vitro
In Vivo |
Safety evaluation of the Stem cell therapy in brain tissue |
Rat MSCtk/GCV | [65] |
| Zhao Y. et. al. |
U87 glioma and H4 cells |
In Vitro
In Vivo |
Effective as cellular Vehicle for targeted suicide genetherapy |
Tumor-tropic neural stem cells, HSV-tk/GCV |
[38] |
| Wang C. et. al. |
NCI-H460-GFP cells |
In Vitro
In Vivo |
Effective brain metastasis treatment |
NSC line expressing CD and TK |
[37] |
| Yin X. et. al. | Bladder cancer with N-methyl-nitrosourea perfusion |
In Vitro
In Vivo |
Effective both in extrisinc and intrisincpapoptosis pathways |
BI-HSV-tk/GCV | [36] |
| Leveille S. et. al. |
Prostate PC3, Breast MCF7, TSA mammary Adenocarcinoma, B-Lymphoma Karpas-422, Melanoma B16-F10 |
In Vitro
In Vivo |
Synergistic effect of (VSV)-MΔ51 and 5FC |
Combination (VSV)-MΔ51- Expressing(CD::UPRT)-5FC |
[85] |
| Cramer F. et. al. |
SCLC: GLC16, DMS53 andNCI-H69 and NSCLC cancer lines: H1299 and A549 |
In Vitro | Improved plasmid nucleardelivery |
NFnB-DTS in an YCD-YUPRT (SCD) |
[28] |
| Li J. et. al. | SGC7901 human gastric cancer cell line |
In Vitro | Effective Cell apoptosis | YCDglyTK and Htert with CPNPs /Novel vector pcDNA3.1(-)hTERT- ShRNA / YCDglyTK |
77 |
| Duan X. et. al. |
C-26 |
In Vitro
In Vivo |
DMP Delivered Survivin- T34A gene DMP/S-T34A) which induced apoptosis |
DOTAP and MPEG-PCL hybrid micelles (DMP) |
[50] |
| Tang Q et. al. |
Human hepatic cell line (HepG2) |
In Vitro
In Vivo |
Survival time and apoptosis |
Intratumorally injected KDR-TK, AFP-TK and microbubble Contrast agent with 5-FC and GCV |
[51] |
| Zarogoulidis P.et. al. |
Lewis lung cancer, SCLC, NSCLC patients |
Animals Humans |
Survival and malignant pleural effusion control with higher efficiency observed for SCLC. |
Ad.CD+5-FC | [6] |
| Yi B. et. al. | Review | Review | Review | Review | [35] |
| Qiu Y. et. al. |
A549, 16HBE, SPC-A-1 AndNCI-H520 |
In Vitro | Specific CA-positive Target gene expression |
CEA promoter and double suicide genes TK and CD. pCEA-TK/CD |
[29] |
| Won Y. et. al. |
C6, U87, F98 and 9L |
In Vitro
In Vivo |
Tumor growth Suppressionand locomotor function maintenance |
rPOA/HSV-tk/GCV | [32] |
| Akerstrom V. et. al. |
Neuroendocrine tumors: NCI-H69, NCI-H1155, NCI-H727, DMS53, U87MG,IMR-32, S-N-SH, SK-N-BE(2),Y79, WERI-Rb1, HeLa, ANC-1, BEAS, RIN, D283 Med, HepG2 |
In Vitro
In Vivo |
Enhanced antitumor activity over the RSV control |
INSM1 promoter, HSV-tk to generate Ad-K5 virus |
[31] |
| Lu M. et. al. | Prostate | Human | Initiatedand recruiting at the time of publication |
Replication-Competent Adenovirus- mediated suicide gene therapy |
[33] |
| Ma S. et. al. | MCF-7 and MDA-MB-231 Breast cell lines |
In Vitro
In Vivo |
Effective antitumor control |
Drosophila melanogaster (Dm-dNK) |
[34] |
| Preuss E. et. al. |
G62 human glioblastoma cell Line, A549 human lung Carcinoma, SW620 human Colorectal adenocarcinoma Cell line and IPC298 HumanMelanoma cell line |
In Vitro
In Vivo |
Continuous complete remission |
TK.007 novel suicide gene | [95] |
| Ahn Y. et. al. | CT26 murine colon adenocarcinoma cells and AGS human gastric adenocarcinoma cells |
In Vitro
In Vivo |
Effective combination Suicide immune therapy |
shRNA-lentivirus and Ad5.CMV.HSV.tk |
[24] |
| Gruber C. et. al. |
SCC |
In Vitro
In Vivo |
Efficient transfection of RDEB SCC |
SLO=PTM | [96] |
| Xiong J. et. al. |
HeLa human cervical cancer Cells, A549 human lung Adenocarcinoma cells, MHCC97 human hepatoma Carcinoma cells and hEL Normal human embryoniv Lung cells |
In Vitro | Combinational cancer Promoter responsive to irradiation |
Chimeric hTERT promoters, CArG |
[82] |
| Luo X. et. al. | SGC7901 human gastric Cancer cell lines |
In Vitro
In Vivo |
Higher efficiency with double suicide gene therapy CD/TK |
Double suicide gene therapy Ad-survivin/GFP and Ad-survivin/CD/TK |
[59] |
| Freytag S. O. et. al. |
Prostate cancer | Human | Transgene expression up to 3 weeks, PSA decline, Acute urinary and gastrointestinal toxicities |
Cytosine deaminasa(CD)/herpes simplexvirus thymidine kinase (HSV-1 TK) and 3D-CRT |
[92] |
| Pandha H. S. et. al. |
Breast cancer | Human | Efficient selectivity against erb-2 |
Therapeutic cassette that contains the Escherichia colicytosine deaminase gene drivan by the tumor-specific erb-2 promoter |
[57] |
| Li N. et. al. | HCC cancer | Human | Recurrence free survival | Adjuvant ADV-TK | [91] |
| Voges J. et. al. | Glioblastoma | Human | Inhomogeneity of tissue formulation distribution |
HSV-1-tk liposomal vector | [88] |
| Nasu Y. et. al. |
Prostate | Human | No serum cytokine changes after treatment, decreased PSA values, Increased CD8+/HLA-DR+ This study confirmed the safety profile at the surrogate marker of HSV-tk gene therapy. |
Ad.HSV-tk/GCV | [89] |
| Rainov N.G. et. al. |
Glioblastoma | Human | Surgical resection and Radiotherapy or standard therapy plus adjuvant gene therapy during surgery. Progression-free median survival in the gene group was 180 days compared with 1 83 days of control group |
RV-HSV-tk/GCV | [76] |
| Xu F. et. al. | Head and Neck | Human | Local response | Intratumoral RV-HSV-tk/GCV | [90] |
| Nemunaitis J. et. al. |
Refractory cancer patients | Human |
Salmonella bacterium can be utilized as a delivery vehicle of the cytosine deaminase gene to malignant tissue with low dose 3 × 107 CFU/m2efficiently. |
TAPET-CD | [94] |
| Freytag S. O. et. al. |
Pancreas | Human | Augments radiotherapy treatment of pancreatic cancer without excessive toxicity |
Ad5-yCD/mutTKSR39rep-ADP HSV-1 TKSR39 |
[93] |
INSM1;insulinoma-associated 1 gene, IFN-β; Interferon-β, GCV;ganciclovir, CEA;Carcinoembryonic antigen, ELISA;Enzyme-linked immuno sorbent assay, IL-7; Interleukin-7, wtCPE; wild type Clostridium perfringens enterotoxin, optCPE; translation-optimisedClostridium perfringens enterotoxin, IL-12; interleukin-12, HSV-1; herpes simplex virus-1, 5-FC; 5-Fluorocytosine, VEGF; vascular endothelial growth factor, CPNPs;bioresorbable calcium phosphate nanoparticles, HSV-tk;herpes simplex virus-thymidine kinase, yCDglyTK; fusion gene therapy of cytosine deaminase and thymidine kinase, Mac-1;Macrophage-1 antigen, CD4+; T lymphocytes, referring to those that carry the CD4 antigen, CD8+; T lymphocytes, referring to those that carry the CD8 antigen, CCl2;chemokine (C-C motif) ligand 2 [Homo sapiens (human), rAd; recombinant adenovirus, MCP-1;Monocyte chemoattractant protein-1, ETA; ETA receprtors, WTp53; wild type p53, Cx43;integral membrane protein of the connexin family, alpha-type (group II) subfamily, MSC;mesenchymal stem cells, AdexCAEGFP; MSC- adenovirus carrying either enhanced green fluorescent protein gene, AdexCACD; MSC- cytosine deaminase gene, UPRT; uracil phosphoribosyltransferase, NSCs; neural stem cells, hTERT; human telomerase reverse transcriptase, HRP; expressing horseradich peroxidase, IAA; idole-3-acetic acid, CArG;Smooth muscle alpha-actin CArG elements, BI;Bifidobacteriuminfantis, VSV; vesicular stomatitis virus, NFnB; nuclear factor B, DOTAP;N-[1-(2,3-dioleoyloxy)propyl]-N,N,N-trimethylammonium methyl sulfate,MPEG-PCL;monomethoxy poly(ethylene glycol)–poly(3-caprolactone , DMP; DOTAP–MPEG–PCL, KDR; kinase insert domain receptor, AFP;α-fetoprotein, rPOA; poly (oligo-D-arginine), DM;Drosophila melanogaster,PET; positronemission tomography, Ad5.CMV.HSV.tk;adenoviruse 5 harboring the herpes simplex virus thymidine kinase gene,SLO=PTM; toxin Streptolysin O-3′ pre-trans-splicing molecules, RDEB-SCC; recessive dystrophic epidermolysisbullosa squamous cell carcinoma, PTM screen; 3′ pre-trans-splicing molecules, PSA; prostate specific antigen, GDEPT; gene-directed enzyme pro-drug therapy, TAPET-CD; pCVD442-msb B-VNP20009
Inducing Cancer Cell Suicide by Targeted, Transgenic Expression of Antibodies against Anti-Oxidative Enzymes
Within the realm of the suicide gene therapy is also an entirely novel strategy for inducing the cancer cells’ deaths through transgenic expression of intracellular antibodies [112-113]. In this strategy, the sequences coding for antibodies are cloned into plasmids or viruses under the targeted-cancer-specific promoters. For example, to be used in the epidermal growth factor receptor (EGFR) over-expressing ovarian cancers, the cds are engineered under the EGFR promoters. Their delivery is guided by the genetically engineered antibodies targeting them to the cancer cells only. Therefore, for example, for the ovarian cancers expressing deletion mutation of the EGFR gene (EGFRvIII), the vectors are bioengineered to target only the cells displaying on their surfaces EGFRvIII. The genetically engineered antibodies are expressed inside the ovarian, testicular, or brain cancer cells, target, and block antioxidative enzymes: Cu, Zn superoxide dismutase (SOD Cu, Zn), Mn superoxide dismutase (SOD Mn), catalase, glutathione reductase, and glutathione peroxidase. This pushes the cancer cells into deepening oxidative stress, with the speed contingent upon the intensity of cancer cells’ metabolism, which generates free radicals. Importantly, this strategy of the cancer suicide gene therapy has two critical safe-guards. First, the transgenes are delivered to the targeted cancer cells only. Therefore, it does not inflict any harm onto the healthy cells. In the clinical trials, it should translate into minimizing side effects. Associated risks include immunization due to presence of genetically engineered antibodies after multiple applications, which are inherent with using any antibodies. This potential task is minimized by genetic engineering of Nano-Antibodies [111]. Second, the promoters are driving expression of the therapeutic transgenes only in the cells expressing cancer or tissue specific genes. Although, some of genes may be over-expressed and result in the receptors over-displayed on cancer cells, they may be also present in smaller quantities on healthy cells. For example epidermal growth factor receptors are over-expressed on some cancer cells. This is the basis for using anti-EGFR antibody-Cetuximab (Erbitux) (e.g., for treatment of KRAS wild type colon cancers), which blocks the receptor and sparks immune response aimed at eliminating cancer cells. However, these EGFRs are also present on healthy cells, thus some of the side effects may occur: fevers, rash, anaphylaxis. Although, they will bind to the healthy cells in approximately two orders of magnitude smaller quantities. Therefore, if the transgenes are guided to EGFR, then they are delivered in the quantities saturating proportional numbers of the receptors onto these healthy cells. Moreover, if internalized, they are expressed in the cells. For these reasons, using very specific targeting guides, as well as the choice of the promoters is of critical importance in this endeavor.
Inducing Cancer Cell Suicide by Transgenic Expression of caspases and DNases
In clinical practice, chemotherapy and radiotherapy trigger activation of multiple signaling pathways leading to apoptosis and complete destruction of the nuclear and mitochondrial DNA. In fact, HSV/TK/ganciclovir induces apoptosis through the ligand-independent death receptor aggregation, cytochrome release, and activation of caspases, including caspase-3 and -6 [114-115]. These killing mechanisms can further be enhanced by dual specific suicide genes in hypoxic conditions [116]. Human DNase I family DNases, DNase I, DNase X, DNase gamma, and DNAS1L2 are the main executors of death. However, the cancer cells develop mechanisms creating resistance to therapies and / or can reverse these processes [117]. These include expulsion of therapeutics by the family of the ABC transporters expressed in cancer cells [118-120]. However, these include also activating repair mechanisms in those cells [121-122].
Aforementioned phenomena prompted research on therapies, which would place the therapeutics closer to the effectors along the signaling pathway. As the caspases are directly responsible for activation of enzymes fragmenting DNA, they are the subjects of experimental therapies [123,124]. In that line of research, the constitutively active transgenes for the human caspase-8 were effectively killing cells in retrovirus and lentivirus based systems [125]. They were engineered into various inducible system including those activated by Tet-On and muristerone. Alternatively, caspase-9 was engineered into the adenovirus and expressed in prostate cancer cells effectively triggering their apoptotic pathways [126]. In other studies, recombinant caspase-1 or caspase-3 were dimerized into the active forms through chemical inducers to exert their effects onto the cancer cells [127]. Another route of research explored possibilities of using small molecules for activating nascent enzymes. A recently identified molecule - PAC-1, was capable for converting procaspase-3 into the active caspase-3, which was initiating apoptotic cascade and exerted therapeutic effects [128].
The most direct induction of cancer cell suicide was attained by the transgenic expression of the recombinant, human DNase I in the cancers of the ovaries and testes [109]. The transgenes were controlled by the switchable, ovary or testis specific promoters. The vector was equipped with the synthetic, nano-antibodies guiding it selectively into the ovarian cancer cells with the unique cell surface display of EGFRvIII – the epidermal growth factor receptor mutated gene expression product. Moreover, the vector was equipped with the nuclear localization signals driving it into the cell nuclei, thus assuring high levels of expression. Therefore, not only the suicide gene was delivered exclusively to the ovarian cancer cells, but also the cancer suicide inducing gene was expressed efficiently in those ovarian cancer cells only. This approach assured doubled safeguarding measures, as well as efficient expression of the therapeutic transgenes; thus therapeutic effects.
Inducing Cancer Cell Suicide by Silencing Transcripts and Blocking Gene Products for Vascular Endothelial Growth Factor
In the study by Leng A. et al. [43] the authors investigated the concept of anti-vascular endothelial growth factor suicide gene therapy in a human colon cell line mouse model. They designed a cassette to silence the VEGF-A expression by delivering DNA carrying shVEGF-CDTK into the cancer cells by calcium phosphate nanoparticles (CPNPs). This is one of the first studies using nanoparticles as a transport for suicide gene therapy. Additionally, the method was safe as no adverse effects were attributed to the methodology used.
In the study by Liu et. al. [44], the authors investigated a model of suicide gene therapy as anti-VEGF with a triple-gene vector expressing vascular endothelial growth factor VEGF-shRNA and fusion suicide gene yCDglyTK. The target cancer cell line was human gastric cancer cell line SGC7901. The formulation was again delivered by calcium phosphate nanoparticles. Increased apoptosis was observed within 24hours of administration. Finally, the study demonstrated that VEGF-targeted RNAi with suicide genes were an effective combination and probably with the addition of CPNPs a future concept for suicide gene therapy (Table 1).
Suicide Gene Therapy Delivered with the Aid of Stem Cells
Stem cells have been used for suicide gene therapy targeting many pathways [35]. In a recent study they were used as a suicide gene therapy by inoculation, expressing cytosine deaminase 5-FC [39]. Two strategies were used, where the mesenchymal stem cells (MSCs) were transfected with an adenovirus carrying enhanced green fluorescent protein gene (AdexCAEGFP) or the cytosine deaminase gene (AdexCACD). Two different types of MSCs were constructed the MSC-CD and MSC-EGFP. The first were used in combination with intraperitoneal 5-FC and demonstrated prolonged survival, however; it showed poor distribution in comparison to MSC-EGFP. In other studies, the stem cells were used as carrier of the transgenes. Moreover, the safety of MSCtk stem cell suicide gene therapy was investigated by Amano S. et al. in a C6 glioma cell mouse model [68]. The study was performed to evaluate the safety of this treatment modality by intracranial and intraperitoneal administration. No histological abnormality was observed at the injection site and normal brain tissue.
Tumor-tropic neural stem cells (NSCs F3.CD-TK) were investigated to express dual suicide gene therapy of CD and TK [37]. This methodology was investigated in a lung cancer brain metastasis model. The intravenously administered F3 cells migrated to the lung cancer metastatic lesions and with the co-administration of 5-FC and GCV efficient antitumor effect were observed (Table 1).
However, risks of oncogenesis, which are associated with using stem cells have to be addressed, as discussed further.
Interventional Suicide Gene Therapy
The concept of interventional suicide gene therapy has been also investigated in liver cancer model in rabbits [26]. The concepts of double gene therapy were investigated: a) wild type p53 and b) thymidine kinase/cytosine deaminase with GCV and 5FC. In total there were five experimental groups. Among them, 2 groups investigated interventional method for vascular tumor embolization in combination with single gene therapy and in 1 group interventional embolization with double gene therapy was investigated. The control group (0.9%NaCl) and the group receiving only interventional embolization did not present any favorable results. The combination of double gene therapy and interventional embolization presented efficient tumor control and an enxouraging concept for the future local treatment (Table 1).
Overexpression or Silencing Specific Genes
In the study by Walther W et al. [81] four different cancer cell lines were included in a mouse model. The cell lines were; a) MCF-7 (breast cancer), b) Panc-1 (pancreatic cancer), c) SKMel-5 (melanoma) and d) HCT116 (hepatocellular cancer). The study design was based on the concept, in which cancer cells expressing claudin -3- and -4- will be more sensitive to the proposed suicide gene therapy of wild type Clostridium pefringens enterotoxin cDNA (CPE) (wtCPE) or translation-optimized CPE cDNA (opt CPE). Indeed the MCF-7 and HCT116 cells lines, which overexpressed claudin -3- and -4, showed increased effects upon the tumors.In the Panc-1 minimal effects were observed and in SKMel-5 the therapy was not efficient.
In the study by Ahn Y et al. [24], the Signal Transducer and Activator of transcription 3 (STAT3) silencing was observed to enhance the suicide gene therapy positive antitumor activity. The STAT3 pathway has been associated with uncontrolled tumor proliferation and immune system suppression. The STAT3 was silenced using a shRNA-lentivirus and additionally Ad5.CMV.HSV.tk was administered to promote suicide gene therapy. The dual treatment modality was used in AGS human gastric adenocarcinoma cells and CT26 murine colon adenocarcinoma cells. It was observed that the immune system was stimulated and the cytotoxic activity of CD3+CD8+T-cells was enhanced (Table 1).
Sensitization to Radiotherapy
The concept of radiotherapy enhancement with suicide gene therapy was investigated by Sun X et al. [48] in a rat prostate model. The study design involved a bifunctional suicide gene therapy with cytosine deaminase (CD) and uracil phosphoribosyltransferase (UPRT) with 5-FC and radiotherapy. The suicide gene expression was designed to be activated by hypoxia with a hypoxia inducible promoter. The hypoxia-induced activation of cytosine deaminase and uracil phosphoribosyltransferase mDsRed was evaluated with different methods: western blotting, fluorescence microscopy and flow cytometry. The protein CDUPRTmDsRed was distributed efficiently throughout the tumor. The 5-FU is well known to be a sensitizer of cancer cells to radiotherapy and the results in this experiment verified previous publications. The hypoxia inducible promoter can probably be used in resistant cancer cell line. An additional, study investigated the enhancement of radiation with suicide gene therapy by Xiong J et Al. [82] in four different cancer cell lines (Table 1).
Suicide Gene Therapy to Prevent Iatrogenic Carcinogenesis in Post-cancer-therapy Organ Regeneration with Stem Cells
Surgery, radiotherapy, or chemotherapy of cancer may have serious iatrogenic impact upon the patients. For example, preventive or radical surgery of the advanced breast cancer requires mastectomy, which leaves women disfigured and psychologically distressed. Surgery of prostate cancer may lead to urinary incontinence and sexual impotence. Radiation therapy of cancer of the ovaries may lead to infertility or genomic mutations. Chemotherapy of cancer of the testes may lead to the mutations in the patients’ genomes resulting in congenital disorders in the offspring. These are only a few from many examples of iatrogenic injuries.
Trauma inflicted by cancer does not end, even at the time, when the therapy is successful and the patient is cancer free. Surely, the targeted therapies, including targeted suicide gene therapies, which are crafted as personalized therapy with the considerations for exact molecular profiles of a particular cancer and genomic analysis of an individual patient, may reduce the iatrogenic impact.
Nevertheless, the complete cure and full recovery can only be attained, if we as physicians are capable not only to eliminate the cancer itself, but also to restore the full vital functions of the patients. In this respect, it is worth considering rapid contributions of stem cell research to the clinical trials of regenerative medicine [129-130]. Along these lines, stem cell therapy, applied as the means for regenerating of the organs impaired or damaged by cancer and/or by radical therapies of cancer, carries the great potential.
Some of the main problems, preventing rapid streamlining of the successes in research on stem cells into regenerative medicine, included the ethical constraints for using pluripotent embryonic stem cells, rejection of foreign stem cells by the immune systems of the patients, incompatibility of the human pluripotency stem cells’ biomarkers with those of other species, controversies surrounding trans-differentiation. They have recently been alleviated by generation of the human, autologous pluripotent, induced stem cells (hapiSCs). Trauma associated with the surgical acquisition of the patients’ own bone marrow or fibroblasts is reduced by inducing pluripotency of the peripheral blood mononuclear cells. High viability and purity of the stem cells, which are essential for the attaining the intended goals of the therapy, have been developed based upon the labeling of the stem cell biomarkers with the antibodies for magnetic activated cell sorting and fluorescence activated cell sorting [104,105,138,139]. The major problem with attaining efficient levels of organ reconstruction in regenerative medicine is targeting and retention of the stem cells to the sites of the therapeutic interventions. These were greatly improved by using bispecific and bivalent antibodies or heterospecific, tetravalent nano-antibodies [140-142]. Some of the risks for iatrogenic triggering of cancerogenesis in the delivered stem cells were significantly reduced by episomal induction of pluripotency with non-integrating vectors, purified transcription factors, or miRNA. However, a risk of neoplasmic transformation of the pluripotent stem cells is still looming over the clinical trials involving pluripotent stem cells. Various strategies for inactivating or destroying pluripotency inducing vectors, as well as, introducing genes aimed at triggering suicide of remaining undifferentiated, pluripotent, stem cells, being at risk of neoplasmic transformation, are currently explored [143-147].
As the populations of human pluripotent stem cells, including human embryonic and induced pluripotent stem cells, are very heterogeneous, the first step, which would be desired prior to administering these cells in cell therapy clinical trials, should be an attempt of identifying and removing of the cells, which could potentially turn into neoplasms [148]. Identification of the stage-specific embryonic antigen (SSEA-5) on the neoplasms’ generating stem cells and development of monoclonal antibodies for this antigen facilitated depletion of the incompletely differentiated stem cells’ batches; thus to reduce the teratoma-forming potential [104,105,149]. Another biomarker of pluripotent stem cells was identified as claudin-6 [150]. Anti-claudin-6 antibodies, modified with toxins, were applied to eliminate residual, undifferentiated cells. Finally, after inducing the directed differentiation, the cells remaining undifferentiated and proliferating, thus creating a risk of neoplasmic transformation, were eliminated with the superparamagnetic nano-antibodies [140].
As a safeguarding strategy, human embryonic stem cells potentially destined for therapeutic administration, were genetically engineered to express thymidine kinase. Thus, they became sensitive to ganciclovir at the doses far lower than those toxic to the healthy cells [151,152]. With the mechanisms identical to those in suicide gene therapy of cancer outlined above, the undifferentiated cells were efficiently eliminated. Similarly, the human induced stem cells, as well as the human embryonic stem cells were genetically transduced by inserting truncated herpes simplex virus delta thymidine kinase (deltaTK) gene controlled by EF1α or Nanog promoter [153]. This insertion of deltaTK gene did not affect pluripotency of the cells. Nevertheless, expression of thymidine kinase rendered them sensitive to ganciclovir. This created an opportunity to eliminate residual, undifferentiated stem cells.
Finally, selective elimination of proliferating and undifferentiated stem cells, from among differentiating cells, was attained by inducible expression of transgenes for DNases, similarly to the strategy of suicide therapy for cancers of ovaries and testes [109].
Cancer Suicide Gene Therapy Applied to Different Cancers
Lung cancer
In the study by Michaelsen SR et al. [47] small cell lung cancer cell (SCLC) lines were treated by suicide gene therapy with the insulinoma-associated 1 (INSM1) promoter-driven (SG) with or without the combination of chemotherapeutic agents. The chemotherapeutic agents used were: a) Etoposide (VP-16), Cisplatin, (CP), Daunorubicin (DAU), carmustine (BCNU), 5-Fluorocytosine (5-FC) and 5-Fluoracil (5-FU). Two different systems were used to evaluate the sensitivity of the INSM1 promoter driven suicide gene therapy: 1) yeast cytosine deaminase-uracil phosphoribosil transferase (YCD-YUPRT) with the pro-drug 5-FC or 2) Escherichia Coli nitroreductase (NTR) with the pro-drug SN27686. The results indicate that this methodology was efficient against chemo-sensitive and chemo-resistant SCLC and chemotherapy plus the suicide gene therapy enhanced further the cytotoxicity of the treatment.
In an effort to enhance the intracellular trafficking of therapeutic DNA the group of Cramer F et al. [28] exploited the concept of utilizing the endogenous nuclear-shuttling activity by using the nuclear factor kappa B (NFκB). Several SCLC cancer cell lines such as: GLC16, DMS53 and NCI-H69 and non-small cell lung cancer: H1299 and A549 were used in the experiments. The DNA nuclear targeting sequence (DTS) was recognized by the nuclear factor kappa B and the plasmid nuclear delivery was augmented. This study was the first of its kind where intracellular DNA trafficking was increased with this method.
In the study by Mader RM et al. [8] chemo-resistant CCL227 colon cancer cell line (of low and intermediate chemo-resistance) was used in vitro to evaluate whether suicide gene therapy was efficient against chemo-resistant tumors. The conversion of the pro-drug 5-FC was 100% with the tumor associated promoters carcino-embryonic antigen (CEA) and H19 suicide systems. However; it should be noted that this colon cell line had low resistance to chemotherapy agents in comparison to other cancer cell lines such as SCLC (H69-VP, H69-CPR, H69-DAU, H69-BCNU) (Table 1).
Hepatocellular
In the study by Sia KC et al. [9] the HCC 26-1004 hepatocellular cancer cell line was used to evaluate a suicide gene model. The herpes simplex virus type 1 (HSV-1) amplicon viral vector was coupled with yCD 5-FC administration. The HCC 26-1004 tumor xenografts expressed high levels of yCD genes, as it was displayed with non-invasive imaging. The firefly luciferase reporter gene was not compatible in order to be used in clinical gene therapy trials, however; remained a valuable tool to monitor the activity and bio distribution of the therapeutic gene expression. Additionally, the authors proposed that a future protocol could include radiation as 5-FU is a known radiosensitizer agent [74] (Table 1).
Medulloblastomas
In the study by Li S et al. [30] the neural stem cell line C17.2 was obtained and used with herpes simplex virus thymidine kinase (HSVtk) and ganciclovir (GCV) in a suicide gene therapy model. The non-toxic Ganciclovir was converted to the toxic GCV triphosphate. The phosphorylated GCV molecules were known to move between gap junctions. The abnormal tumor structure was known for the impaired gap junction intercellular communication (GJIC), which played a major role in tumor evolution and drug blocking [75]. In order to enhance the bystander effect the all-trans retinoic acid (ATRA) was used to induce the expression of Cx43 in Daoy cells. The Daoy and C17.2-tk cells were cultured in the presence and absence of ATRA. The colorimetric assay for measuring the activity of cellular enzymes that reduce the tetrazolium dye (MTT) test was used to evaluate the cell viability. The results demonstrated efficient tumor growth control. However; brain tumor control with this method in human trials presented discouraging results [76]. The main reason is the failure of the HSV-tk Vector to efficiently disseminate throughout the tumor cells. This may resulted from the abnormal gap junctions in those tumors. Therefore the ATRA, a lipophilic molecule, was an excellent addition because of its property to cross the blood brain-barrier without any additional toxicity to the body. The ATRA enhanced the GJIC function and tumoricidal effect of the bystander effect (Table 1).
Glioblastomas
A lentiviral delivery of the herpes siplex virus thymidine kinasegene with gancyclovir treatment was found to have efficient bystander effects in glioblastoma cell lines [40]. This was due to the connexin 43 (Cx43), which can be found differently expressed in different glioblastoma cell lines. In the study by Cottin S. et. al. [40] the authors investigated the effect of positive Cx43 and whether the localization of Cx43 plays a role in the treatment efficacy. Although the aberrant localization should have a negative impact for the efficiency of the suicide gene therapy it did not in this case. These findings presented useful data for the design of chemotherapy drugs that rely on the gap junction-mediated bystander effect.
The non-viral vector poly (oligo-D-arginine) was chosen to deliver the HSV-tk/GCV suicide gene therapy in four different glioma cell lines (C6, U87, F98 and 9L). The specific non-viral vector has demonstrated low toxicity and efficient tumor growth suppression. The authors designed an excellent spinal cord cancer model and evaluated the locomotor activity in correlation to tumor control. The locomotor activity was maintained throughout the treatment. This study presented favorable data regarding the non-viral vectors utility in suicide gene therapy. However; there is still a long way until we have a safe non-viral vector [66,65]. The non-viral vectors have also different gene transfection efficiency depending upon the tissue they target (Table 1).
Neuroendocrine tumors
Neuroendocrine tumors are known to have reactivated the insulinoma-associated protein-1 (INSM1). Several neuroendocrine cancer cell lines were used by Akerstrom V et al. [31] with HSV-tk suicide gene therapy to generate the Ad-K5 virus. Improvements of the INSM1 promoter resulted in more selective adenovirus and therefore a more efficient treatment for high risk neuroendocrine tumors (Table 1).
Colon Cancer
In the study by Finzi et al. [25] the concept of enhancing gene transfer based on the synchronization of cell cycle was investigated with three different drugs: a) methotrexate (MTX), b) ara-C and aphidicolin. Two colon cell lines were used in this experiment the HT29 and DHDK12. Three drugs induced different cell cycle synchronization. The retroviral HSV-tk was used for suicide gene therapy. The cell cycle synchronization offered increased number of cells being accessible to drug targeting DNA and therefore improved the action of anti-proliferative chemotherapies. The S phase had been extensively investigated. The use of MTX for cell cycle synchronization was the most effective within 24hours of suicide gene therapy administration.
In an effort to augment the synergistic effect against human gastric cancer cell lines the group of Li J et al. [77] constructed a novel vector the pcDNA3.1(−)hTERT-shRNA/yCDglyTK. The interfering RNA (RNAi) targeted the human telomerase reverse transcriptase (hTERT). The plasmids were delivered to the cancer cells with phosphate nanoparticles (CPNPs). The new vector was efficient and using RNAi to target hTERT resulted in a synergistic effect with suicide gene therapy (Table 1).
Head and Neck
The mifepristone-dependent Gene Switch was used to regulate the expression of a deletion mutant Pseudomonas exotoxin A in a squamous head and neck cancer cell line. The Gene Switch system consists of two plasmids the pSwitch and the pGene/V5-His’s plasmid, from which the toxin mutant pGene/V5-His’s-ETA was, cloned [41]. Currently there is no receptor-mediated cancer therapy for head and neck cancer since there is no surface biomarker exclusively expressed in this type of carcinoma. The study by Schmidt M et al. revealed in vitro that the use of toxin genes in combination with Gene Switch System could be one day a treatment for head and neck cancer (Table 1).
Bladder Cancer
The group of Yin X et. al. [36] investigated the bifidobacterium infantis (BI)-mediated-HSV/GCV suicide gene therapy in a bladder cancer model. N-methyl-nitrosourea perfusion was used to create bladder cancer in mice and three groups were studied. The group A receiving normal saline, group B receiving the complex BI/PGEX-1λT and the group C receiving BI-TK. The group C presented effective apoptosis in extrinsic and intrinsic pathways with the expression levels of Fas, FasL, Cyt-C and caspase-9 being also significantly higher than group B. The BI can infect the tumor cells effectively, however the underlying mechanisms, in which /tumor cells/ and escape suicide gene therapy, still remain to be elicited (Table 1).
Breast Cancer
Two breast cancer cell lines the MCF7 and MDA-MB-231 were used to evaluate antitumor efficiency of the multisubstrate deoxyribonucleoside kinase of Drosophila melanogaster (Dm-dNK) due to the broad substrate specificity. The Dm-dNK was used as a combined suicide gene therapy with chemotherapy. The pro-drug/Dm-dNK when compared to HSV-1-tk was more efficient due tothe broad substrates’ specificity. They were both located in the nucleus when expressed in human cells [78]. A number of mutations observed in Dm-dNK confered to higher cytosolic localization and catalytic activity [79]. Low toxicity was observed in non-transduced cells and higher tumor inhibition was demonstrated in Dm-dNK/araC positive tumors (Table 1).
Discussion
One of the major challenges in suicide gene therapy is the activation of specific pro-drugs. Therefore, there is currently the urgent need for novel systems activating more pro-drugs. Along these lines, the thymidine-active mutant of dCK, dCK.DM.S74E was created which activates multiple pro-drugs such as; BVdU, LdUNAs and LdT. This system has the ability to sensitize and re-sensitize tumors to chemotherapeutic agents. Furthermore, it was observed that prevents multidrug resistance to be induced and finally it can simultaneously activate more than one pro-drug without any additional side effects [83].
In an effort to enhance the suicide gene therapy the local administration to the tumor site has been investigated without any remarkable histological adverse effects in lung cancer patients, and in glioma cancer cell lines [6,68]. However, loco-regional administration is not always possible. Therefore the “Trojan horse” approach has been investigated. The concept relies on the “stealth” ability that tumor-tropic neural stem cells (NSCs) have for a sustained release gene expression products. In the study by Zhao Y et al. (2012) the NSCs derived from HES1 human embryonic stem cell line had the ability to migrate from the injection site (vein systemic administration) or intracranial to the intracranial glioma xenografts. A baculovirus vector was used to insert the HSV-tk suicide gene into the cells. Additionally, ganciclovir was administered in order for an amount of concentration to be present locally for the suicide system to act. It was also observed that the transgene expression was present for three weeks. This study presented additional data regarding a sustain release system, which was much needed for suicide gene therapy and a future concept for this treatment modality [38]. The same concept has been also applied with MSC in a HCC model [84]. Additionally, Wang C et al. [37] investigated NSCs (F3) as dual suicide gene therapy with CD and TK creating the NSC-F3.CD-TK and the result was enhanced antitumor activity against lung cancer metastasis in comparison to single suicide gene therapy. The same technique was again used in lung cancer cell lines with a carcinoembryonic antigen (CEA) promoter with TK and CD constructing the pCEA-TK/CD [29]. Dual suicide gene therapy was also investigated with surviving promoter with Ad-survivin/GFP and Ad-survivin/CD/TK and higher efficiency was observed again with the dual in comparison to single suicide gene therapy [62].
Further to the previous concept of combination suicide gene therapy the administration of (VSV)-ΜΔ51-expressing (CD::UPRT)-5FC was investigated in four different cancer cell lines (Prostate PC3, Breast MCF7, TSA mammary, Adenocarcinoma, B-Lymphoma Karpas-422, and Melanoma B16-F10) and increased tumor oncolysis was observed [85]. The combination treatment was more effective against the TSA mammary adenocarcinoma. Although combination treatments might demonstrate efficiency in comparison to single gene therapy approach this was not applicable to all cancers.
A method to enhance the local treatment was investigated by Tang Q et al. [51], for a human hepatic cancer cell line model intratumoral administration of KDR-TK and AFP-TK with microbubble contrast agent prior to ultrasound treatment. The pro-drugs 5-FC and GCV were administered after the intratumoral therapy. The survival time did not differ between treatment groups, however; an increased tumor inhibition rate was observed.
It should be mentioned at this point that the time of the pro-drug administration plays a crucial role in the efficiency of suicide gene therapy, as the pro-drug has to be already diffused within the target tissue for the administered gene therapy to effectively kill the cancer cells. Diffusion of the plasmid is also crucial and several methods of local transport have been presented.
Nanomedicine has been used efficiently in many treatment modalities [4]. In a suicide gene therapy model the group of Duan X et al. [50] explored the gene transfection of the novel cationic self-assembled DOTAP and MPEG-PCL hybrid micelles (DMP). The new transport had less toxicity when compared to the polymer Polyethyleneimine (PEI) with 25kDa. The DMP delivered efficiently the urvivin-T34 gene (S-T34A) to treat C-26 colon cancer cell lines. Efficient metastasis and malignant ascites effusioncontrol was observed. CPNPs nanoparticles have been also used in previous studies [44,77].
Patient studies with suicide gene therapy are still limited and every effort towards this treatment modality is welcomed [6,63,64,86,87,76,88-92,60,93,94]. Recently the first clinical trial design for early prostate cancer was published [33,92].
Moreover, novel suicide genes such as the TK.007, have already been introduced and demonstrated efficiency in several cancer cell lines (G62 human glioblastoma cell line, A549 human lung carcinoma, SW620 human colorectal adenocarcinoma cell line and IPC298 human melanoma cell line) [95]. The TK.007 demonstrated higher gene transfection in comparison to HSV-tk with lower doses of GCV and especially to G62 glioblastoma cell line. Another method to enhance the specificity has been achieved with the spliceosome-mediated RNA trans-splicing technology. This technology has the ability to replace a tumor-specific transcript with one encoding a cell death-inducing peptide/toxin. The group of Gruber C et al. [96] investigated the efficiency of 3′ pre-trans-splicing molecules (PTM) and the results indicated that this method is efficient against highly malignant tumors. Another novel suicide gene therapy system was developed by Di Stasi et al. [97]. The new system targets the inducible caspase 9 (iCasp9) gene. It was applied to children who developed graft-vs-host disease (GVHD) by donor lymphocytes. That process was reversed with the novel suicide gene therapy.
There are currently available several delivery and effector systems at work for suicide gene therapy. Each of them demonstrating effectiveness against specific cancer cell lines. Combining different systems has demonstrated enhanced antitumor efficiency. Combination with interventional techniques has also demonstrated enhanced tumor control in less time than a single modality therapy in tumor models involving extensive tumor vascularization.
Regarding the non-viral vectors there are novel molecules that are being constructed in order to present low toxicity and high DNA loading capacity. Several new pathways as targeted suicide gene therapy are under investigation along with new promoters of over-expressed genes. Suicide gene therapy has presented data where although the drug formulation is injected to a distant site of the primary disease it can efficiently control the primary lesion. The same is observed for distant metastases control. The suicide gene therapy enhances the immune system not only locally, but also distally to the metastatic sites.
Finally, each system uses a different mechanism of diffusion. Nanomedicine has the potential to offer nanoparticles as a transporting mode which is much needed to bypass the increased interstitial fluid pressure of the tumor and to increase the EPR effect. Efficient suicide therapy depends not only on the characteristics of the targeted tissue, but the organ bearing the tumor. The study design should be based on both parameters.
Acknowledgments
Source of Funding
Marek Malecki, MD, PhD, was Principal Investigator on the projects supported by the funds from the National Institutes of Health [grant numbers: P41 RR000570 and P41 RR002301]; from the National Science Foundation [grant numbers: 9420056, 9522771, 9902020, and 0094016]; and from the Phoenix Biomolecular Engineering Foundation [grant number: 2006071401]. Lonny Yarmus, MD was Principal Investigator on the projects supported by the fund from the National Institutes of Health [grant number: R42CA141907-03].
Administrators of the funding institutions and managers of the facilities had no influence on the data presented.
References
- 1.Curigliano G, Cardinale D, Suter T, Plataniotis G, de Azambuja E, et al. Cardiovascular toxicity induced by chemotherapy, targeted agents and radiotherapy: ESMO Clinical Practice Guidelines. Ann Oncol. 2012;23(Suppl 7):vii155–166. doi: 10.1093/annonc/mds293. [DOI] [PubMed] [Google Scholar]
- 2.Kuwabara H, Fushimi K. The impact of a new payment system with case-mix measurement on hospital practices for breast cancer patients in Japan. Health Policy. 2009;92:65–72. doi: 10.1016/j.healthpol.2009.02.010. [DOI] [PubMed] [Google Scholar]
- 3.Lee MK, Dodson TB, Nalliah RP, Karimbux NY, Allareddy V. Nine-year trend analysis of hospitalizations attributed to oral and oropharyngeal cancers in the United States. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013 doi: 10.1016/j.oooo.2013.01.019. [DOI] [PubMed] [Google Scholar]
- 4.Zarogoulidis P, Chatzaki E, Porpodis K, Domvri K, Hohenforst-Schmidt W, et al. Inhaled chemotherapy in lung cancer: future concept of nanomedicine. Int J Nanomedicine. 2012;7:1551–1572. doi: 10.2147/IJN.S29997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zarogoulidis P, Eleftheriadou E, Sapardanis I, Zarogoulidou V, Lithoxopoulou H, et al. Feasibility and effectiveness of inhaled carboplatin in NSCLC patients. Invest New Drugs. 2012;30:1628–1640. doi: 10.1007/s10637-011-9714-5. [DOI] [PubMed] [Google Scholar]
- 6.Zarogoulidis P, Chatzaki E, Hohenforst-Schmidt W, Goldberg EP, Galaktidou G, et al. Management of malignant pleural effusion by suicide gene therapy in advanced stage lung cancer: a case series and literature review. Cancer Gene Ther. 2012;19:593–600. doi: 10.1038/cgt.2012.36. [DOI] [PubMed] [Google Scholar]
- 7.Darwiche K, Zarogoulidis P, Karamanos NK, Domvri K, Chatzaki E, et al. Efficacy versus safety concerns for aerosol chemotherapy in non-small-cell lung cancer: a future dilemma for micro-oncology. Future Oncol. 2013;9:505–525. doi: 10.2217/fon.12.205. [DOI] [PubMed] [Google Scholar]
- 8.Mader RM, Kalipciyan M, Ohana P, Hochberg A, Steger GG. Suicide activation in a 5-fluorouracil resistant colon cancer model in vitro. Int J Clin Pharmacol Ther. 2011;49:69–70. [PubMed] [Google Scholar]
- 9.Sia KC, Huynh H, Chinnasamy N, Hui KM, Lam PY. Suicidal gene therapy in the effective control of primary human hepatocellular carcinoma as monitored by noninvasive bioimaging. Gene Ther. 2012;19:532–542. doi: 10.1038/gt.2011.131. [DOI] [PubMed] [Google Scholar]
- 10.Celikoglu F, Celikoglu SI, Goldberg EP. Bronchoscopic intratumoral chemotherapy of lung cancer. Lung Cancer. 2008;61:1–12. doi: 10.1016/j.lungcan.2008.03.009. [DOI] [PubMed] [Google Scholar]
- 11.Zhibing W, Qinghua D, Shenglin M, Ke Z, Kan W, et al. Clinical study of cisplatin hyperthermic intraperitoneal perfusion chemotherapy in combination with docetaxel, 5-flourouracil and leucovorin intravenous chemotherapy for the treatment of advanced-stage gastric carcinoma. Hepatogastroenterology. 2013:60. doi: 10.5754/hge13038. [DOI] [PubMed] [Google Scholar]
- 12.Porpodis K, Karanikas M, Zarogoulidis P, Kontakiotis T, Mitrakas A, et al. A case of typical pulmonary carcinoid tumor treated with bronchoscopic therapy followed by lobectomy. J Multidiscip Healthc. 2012;5:47–51. doi: 10.2147/JMDH.S29709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Podolska K, Stachurska A, Hajdukiewicz K, Malecki M. Gene therapy prospects--intranasal delivery of therapeutic genes. Adv Clin Exp Med. 2012;21:525–534. [PubMed] [Google Scholar]
- 14.Domvri K, Zarogoulidis P, Porpodis K, Koffa M, Lambropoulou M, et al. Gene therapy in liver diseases: state-of-the-art and future perspectives. Curr Gene Ther. 2012;12:463–483. doi: 10.2174/156652312803519788. [DOI] [PubMed] [Google Scholar]
- 15.Weng L, Zhang L, Peng Y, Huang RS. Pharmacogenetics and pharmacogenomics: a bridge to individualized cancer therapy. Pharmacogenomics. 2013;14:315–324. doi: 10.2217/pgs.12.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cervino AR, Burei M, Mansi L, Evangelista L. Molecular pathways and molecular imaging in breast cancer: an update. Nucl Med Biol. 2013;40:581–591. doi: 10.1016/j.nucmedbio.2013.03.002. [DOI] [PubMed] [Google Scholar]
- 17.Matthaios D, Zarogoulidis P, Balgouranidou I, Chatzaki E, Kakolyris S. Molecular pathogenesis of pancreatic cancer and clinical perspectives. Oncology. 2011;81:259–272. doi: 10.1159/000334449. [DOI] [PubMed] [Google Scholar]
- 18.Machairiotis N, Kougioumtzi I, Zarogoulidis P, Stylianaki A, Tsimogiannis K, et al. Gastrointestinal stromal tumor mesenchymal neoplasms: the offspring that choose the wrong path. J Multidiscip Healthc. 2013;6:127–131. doi: 10.2147/JMDH.S43703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Trent RJ, Cheong PL, Chua EW, Kennedy MA. Progressing the utilisation of pharmacogenetics and pharmacogenomics into clinical care. Pathology. 2013;45:357–370. doi: 10.1097/PAT.0b013e328360b66e. [DOI] [PubMed] [Google Scholar]
- 20.Vachani A, Moon E, Wakeam E, Haas AR, Sterman DH, et al. Gene therapy for lung neoplasms. Clin Chest Med. 2011;32:865–885. doi: 10.1016/j.ccm.2011.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Duarte S, Carle G, Faneca H, de Lima MC, Pierrefite-Carle V. Suicide gene therapy in cancer: where do we stand now? Cancer Lett. 2012;324:160–170. doi: 10.1016/j.canlet.2012.05.023. [DOI] [PubMed] [Google Scholar]
- 22.Malecki M. Frontiers in Suicide Gene Therapy of Cancer. J Genet Syndr Gene Ther. 2012:2012. doi: 10.4172/2157-7412.1000e114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Morgan RA. Live and let die: a new suicide gene therapy moves to the clinic. Mol Ther. 2012;20:11–13. doi: 10.1038/mt.2011.273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ahn YH, Yi H, Shin JY, Lee KD, Shin SP, et al. STAT3 silencing enhances the efficacy of the HSV.tk suicide gene in gastrointestinal cancer therapy. Clin Exp Metastasis. 2012;29:359–369. doi: 10.1007/s10585-012-9458-4. [DOI] [PubMed] [Google Scholar]
- 25.Finzi L, Kraemer A, Capron C, Noullet S, Goere D, et al. Improved retroviral suicide gene transfer in colon cancer cell lines after cell synchronization with methotrexate. J Exp Clin Cancer Res. 2011;30:92. doi: 10.1186/1756-9966-30-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Niu HX, Du T, Xu ZF, Zhang XK, Wang RG. Role of wild type p53 and double suicide genes in interventional therapy of liver cancer in rabbits. Acta Cir Bras. 2012;27:522–528. doi: 10.1590/s0102-86502012000800002. [DOI] [PubMed] [Google Scholar]
- 27.Marukawa Y, Nakamoto Y, Kakinoki K, Tsuchiyama T, Iida N, et al. Membrane-bound form of monocyte chemoattractant protein-1 enhances antitumor effects of suicide gene therapy in a model of hepatocellular carcinoma. Cancer Gene Ther. 2012;19:312–319. doi: 10.1038/cgt.2012.3. [DOI] [PubMed] [Google Scholar]
- 28.Cramer F, Christensen CL, Poulsen TT, Badding MA, Dean DA, et al. Insertion of a nuclear factor kappa B DNA nuclear-targeting sequence potentiates suicide gene therapy efficacy in lung cancer cell lines. Cancer Gene Ther. 2012;19:675–683. doi: 10.1038/cgt.2012.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Qiu Y, Peng GL, Liu QC, Li FL, Zou XS, He JX. Selective killing of lung cancer cells using carcinoembryonic antigen promoter and double suicide genes, thymidine kinase and cytosine deaminase (pCEA-TK/CD) Cancer Lett. 2012;316:31–38. doi: 10.1016/j.canlet.2011.10.015. [DOI] [PubMed] [Google Scholar]
- 30.Li S, Gao Y, Pu K, Ma L, Song X, et al. All-trans retinoic acid enhances bystander effect of suicide-gene therapy against medulloblastomas. Neurosci Lett. 2011;503:115–119. doi: 10.1016/j.neulet.2011.08.019. [DOI] [PubMed] [Google Scholar]
- 31.Akerstrom V, Chen C, Lan MS, Breslin MB. Adenoviral insulinoma-associated protein 1 promoter-driven suicide gene therapy with enhanced selectivity for treatment of neuroendocrine cancers. Ochsner J. 2013;13:91–99. [PMC free article] [PubMed] [Google Scholar]
- 32.Won YW, Kim KM, An SS, Lee M, Ha Y, et al. Suicide gene therapy using reducible poly (oligo-D-arginine) for the treatment of spinal cord tumors. Biomaterials. 2011;32:9766–9775. doi: 10.1016/j.biomaterials.2011.08.089. [DOI] [PubMed] [Google Scholar]
- 33.Lu M, Freytag SO, Stricker H, Kim JH, Barton K, et al. Adaptive seamless design for an efficacy trial of replication-competent adenovirus-mediated suicide gene therapy and radiation in newly-diagnosed prostate cancer (ReCAP Trial) Contemp Clin Trials. 2011;32:453–460. doi: 10.1016/j.cct.2011.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ma S, Zhao L, Zhu Z, Liu Q, Xu H, et al. The multisubstrate deoxyribonucleoside kinase of Drosophila melanogaster as a therapeutic suicide gene of breast cancer cells. J Gene Med. 2011;13:305–311. doi: 10.1002/jgm.1573. [DOI] [PubMed] [Google Scholar]
- 35.Yi BR, Choi KJ, Kim SU, Choi KC. Therapeutic potential of stem cells expressing suicide genes that selectively target human breast cancer cells: evidence that they exert tumoricidal effects via tumor tropism (review) Int J Oncol. 2012;41:798–804. doi: 10.3892/ijo.2012.1523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yin X, Yu B, Tang Z, He B, Ren J, et al. Bifidobacterium infantis-mediated HSV-TK/GCV suicide gene therapy induces both extrinsic and intrinsic apoptosis in a rat model of bladder cancer. Cancer Gene Ther. 2013;20:77–81. doi: 10.1038/cgt.2012.86. [DOI] [PubMed] [Google Scholar]
- 37.Yang ZF, Zhan YQ, Chen RC, Zhou R, Wang YT, et al. A prospective comparison of the epidemiological and clinical characteristics of pandemic (H1N1) 2009 influenza A virus and seasonal influenza A viruses in Guangzhou, South China in 2009. Jpn J Infect Dis. 2012;65:208–214. doi: 10.7883/yoken.65.208. [DOI] [PubMed] [Google Scholar]
- 38.Zhao Y, Lam DH, Yang J, Lin J, Tham CK, et al. Targeted suicide gene therapy for glioma using human embryonic stem cell-derived neural stem cells genetically modified by baculoviral vectors. Gene Ther. 2012;19:189–200. doi: 10.1038/gt.2011.82. [DOI] [PubMed] [Google Scholar]
- 39.Kosaka H, Ichikawa T, Kurozumi K, Kambara H, Inoue S, et al. Therapeutic effect of suicide gene-transferred mesenchymal stem cells in a rat model of glioma. Cancer Gene Ther. 2012;19:572–578. doi: 10.1038/cgt.2012.35. [DOI] [PubMed] [Google Scholar]
- 40.Cottin S, Gould PV, Cantin L, Caruso M. Gap junctions in human glioblastomas: implications for suicide gene therapy. Cancer Gene Ther. 2011;18:674–681. doi: 10.1038/cgt.2011.38. [DOI] [PubMed] [Google Scholar]
- 41.Schmidt M, Gruensfelder P, Roller J, Hagen R. Suicide gene therapy in head and neck carcinoma cells: an in vitro study. Int J Mol Med. 2011;27:591–597. doi: 10.3892/ijmm.2011.610. [DOI] [PubMed] [Google Scholar]
- 42.Finocchiaro LM, Villaverde MS, Gil-Cardeza ML, Riveros MD, Glikin GC. Cytokine-enhanced vaccine and interferon-?2 plus suicide gene as combined therapy for spontaneous canine sarcomas. Res Vet Sci. 2011;91:230–234. doi: 10.1016/j.rvsc.2010.12.012. [DOI] [PubMed] [Google Scholar]
- 43.Leng A, Yang J, Liu T, Cui J, Li XH, et al. Nanoparticle-delivered VEGF-silencing cassette and suicide gene expression cassettes inhibit colon carcinoma growth in vitro and in vivo. Tumour Biol. 2011;32:1103–1111. doi: 10.1007/s13277-011-0210-5. [DOI] [PubMed] [Google Scholar]
- 44.Liu T, Ye L, He Y, Chen X, Peng J, et al. Combination gene therapy using VEGF-shRNA and fusion suicide gene yCDglyTK inhibits gastric carcinoma growth. Exp Mol Pathol. 2011;91:745–752. doi: 10.1016/j.yexmp.2011.07.007. [DOI] [PubMed] [Google Scholar]
- 45.Bondanza A, Hambach L, Aghai Z, Nijmeijer B, Kaneko S, et al. IL-7 receptor expression identifies suicide gene-modified allospecific CD8+ T cells capable of self-renewal and differentiation into antileukemia effectors. Blood. 2011;117:6469–6478. doi: 10.1182/blood-2010-11-320366. [DOI] [PubMed] [Google Scholar]
- 46.Xu Y, Hou J, Liu Z, Yu H, Sun W, et al. Gene therapy with tumor-specific promoter mediated suicide gene plus IL-12 gene enhanced tumor inhibition and prolonged host survival in a murine model of Lewis lung carcinoma. J Transl Med. 2011;9:39. doi: 10.1186/1479-5876-9-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Michaelsen SR, Christensen CL, Sehested M, Cramer F, Poulsen TT, et al. Single agent- and combination treatment with two targeted suicide gene therapy systems is effective in chemoresistant small cell lung cancer cells. J Gene Med. 2012;14:445–458. doi: 10.1002/jgm.2630. [DOI] [PubMed] [Google Scholar]
- 48.Sun X, Xing L, Deng X, Hsiao HT, Manami A, et al. Hypoxia targeted bifunctional suicide gene expression enhances radiotherapy in vitro and in vivo. Radiother Oncol. 2012;105:57–63. doi: 10.1016/j.radonc.2012.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kakinoki K, Nakamoto Y, Kagaya T, Tsuchiyama T, Sakai Y, et al. Prevention of intrahepatic metastasis of liver cancer by suicide gene therapy and chemokine ligand 2/monocyte chemoattractant protein-1 delivery in mice. J Gene Med. 2010;12:1002–1013. doi: 10.1002/jgm.1528. [DOI] [PubMed] [Google Scholar]
- 50.Duan X, Wang P, Men K, Gao X, Huang M, et al. Treating colon cancer with a suicide gene delivered by self-assembled cationic MPEG-PCL micelles. Nanoscale. 2012;4:2400–2407. doi: 10.1039/c2nr30079f. [DOI] [PubMed] [Google Scholar]
- 51.Tang Q, He X, Liao H, He L, Wang Y, et al. Ultrasound microbubble contrast agent-mediated suicide gene transfection in the treatment of hepatic cancer. Oncol Lett. 2012;4:970–972. doi: 10.3892/ol.2012.845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Floeth FW, Shand N, Bojar H, Prisack HB, Felsberg J, et al. Local inflammation and devascularization--in vivo mechanisms of the “bystander effect” in VPC-mediated HSV-Tk/GCV gene therapy for human malignant glioma. Cancer Gene Ther. 2001;8:843–851. doi: 10.1038/sj.cgt.7700382. [DOI] [PubMed] [Google Scholar]
- 53.Bi W, Kim YG, Feliciano ES, Pavelic L, Wilson KM, et al. An HSVtk-mediated local and distant antitumor bystander effect in tumors of head and neck origin in athymic mice. Cancer Gene Ther. 1997;4:246–252. [PubMed] [Google Scholar]
- 54.Dilber MS, Abedi MR, Bjorkstrand B, Christensson B, Gahrton G, et al. Suicide gene therapy for plasma cell tumors. Blood. 1996;88:2192–2200. [PubMed] [Google Scholar]
- 55.Tomicic MT, Thust R, Kaina B. Ganciclovir-induced apoptosis in HSV-1 thymidine kinase expressing cells: critical role of DNA breaks, Bcl-2 decline and caspase-9 activation. Oncogene. 2002;21:2141–2153. doi: 10.1038/sj.onc.1205280. [DOI] [PubMed] [Google Scholar]
- 56.Beltinger C, Fulda S, Kammertoens T, Meyer E, Uckert W, et al. Herpes simplex virus thymidine kinase/ganciclovir-induced apoptosis involves ligand-independent death receptor aggregation and activation of caspases. Proc Natl Acad Sci U S A. 1999;96:8699–8704. doi: 10.1073/pnas.96.15.8699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Abate-Daga D, Garcia-Rodriguez L, Sumoy L, Fillat C. Cell cycle control pathways act as conditioning factors for TK/GCV sensitivity in pancreatic cancer cells. Biochim Biophys Acta. 2010;1803:1175–1185. doi: 10.1016/j.bbamcr.2010.06.009. [DOI] [PubMed] [Google Scholar]
- 58.Fischer U, Steffens S, Frank S, Rainov NG, Schulze-Osthoff K, et al. Mechanisms of thymidine kinase/ganciclovir and cytosine deaminase/5-fluorocytosine suicide gene therapy-induced cell death in glioma cells. Oncogene. 2005;24:1231–1243. doi: 10.1038/sj.onc.1208290. [DOI] [PubMed] [Google Scholar]
- 59.Negroni L, Samson M, Guigonis JM, Rossi B, Pierrefite-Carle V, et al. Treatment of colon cancer cells using the cytosine deaminase/5-fluorocytosine suicide system induces apoptosis, modulation of the proteome, and Hsp90beta phosphorylation. Mol Cancer Ther. 2007;6:2747–2756. doi: 10.1158/1535-7163.MCT-07-0040. [DOI] [PubMed] [Google Scholar]
- 60.Pandha HS, Martin LA, Rigg A, Hurst HC, Stamp GW, et al. Genetic prodrug activation therapy for breast cancer: A phase I clinical trial of erbB-2-directed suicide gene expression. J Clin Oncol. 1999;17:2180–2189. doi: 10.1200/JCO.1999.17.7.2180. [DOI] [PubMed] [Google Scholar]
- 61.Kanai F, Kawakami T, Hamada H, Sadata A, Yoshida Y, et al. Adenovirus-mediated transduction of Escherichia coli uracil phosphoribosyltransferase gene sensitizes cancer cells to low concentrations of 5-fluorouracil. Cancer Res. 1998;58:1946–1951. [PubMed] [Google Scholar]
- 62.Luo XR, Li JS, Niu Y, Miao L. Adenovirus-mediated double suicide gene selectively kills gastric cancer cells. Asian Pac J Cancer Prev. 2012;13:781–784. doi: 10.7314/apjcp.2012.13.3.781. [DOI] [PubMed] [Google Scholar]
- 63.Sterman DH, Recio A, Carroll RG, Gillespie CT, Haas A, et al. A phase I clinical trial of single-dose intrapleural IFN-beta gene transfer for malignant pleural mesothelioma and metastatic pleural effusions: high rate of antitumor immune responses. Clin Cancer Res. 2007;13:4456–4466. doi: 10.1158/1078-0432.CCR-07-0403. [DOI] [PubMed] [Google Scholar]
- 64.Sterman DH, Recio A, Haas AR, Vachani A, Katz SI, et al. A phase I trial of repeated intrapleural adenoviral-mediated interferon-beta gene transfer for mesothelioma and metastatic pleural effusions. Mol Ther. 2010;18:852–860. doi: 10.1038/mt.2009.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zarogouldis P, Karamanos NK, Porpodis K, Domvri K, Huang H, et al. Vectors for inhaled gene therapy in lung cancer. Application for nano oncology and safety of bio nanotechnology. Int J Mol Sci. 2012;13:10828–10862. doi: 10.3390/ijms130910828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zarogoulidis P, Karamanos NK, Porpodis K, Domvri K, Huang H, et al. Zarogouldis p., et al. vectors for inhaled gene therapy in lung cancer. Application for nano oncology and safety of bio nanotechnology. Int. J. Mol. Sci. 2012;13:10828–10862. doi: 10.3390/ijms130910828. 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]; Int J Mol Sci. 13:17290–17291. [Google Scholar]
- 67.Zarogoulidis P, Kontakiotis T, Zarogoulidis K. Inhaled gene therapy in lung cancer: “as for the future, our task is not to foresee it, but to enable it”. Ther Deliv. 2012;3:919–921. doi: 10.4155/tde.12.71. [DOI] [PubMed] [Google Scholar]
- 68.Amano S, Gu C, Koizumi S, Tokuyama T, Namba H. Tumoricidal bystander effect in the suicide gene therapy using mesenchymal stem cells does not injure normal brain tissues. Cancer Lett. 2011;306:99–105. doi: 10.1016/j.canlet.2011.02.037. [DOI] [PubMed] [Google Scholar]
- 69.Iyer AK, Khaled G, Fang J, Maeda H. Exploiting the enhanced permeability and retention effect for tumor targeting. Drug Discov Today. 2006;11:812–818. doi: 10.1016/j.drudis.2006.07.005. [DOI] [PubMed] [Google Scholar]
- 70.Jacobs A, Voges J, Reszka R, Lercher M, Gossmann A, et al. Positron-emission tomography of vector-mediated gene expression in gene therapy for gliomas. Lancet. 2001;358:727–729. doi: 10.1016/s0140-6736(01)05904-9. [DOI] [PubMed] [Google Scholar]
- 71.Penuelas I, Mazzolini G, Boan JF, Sangro B, Marti-Climent J, et al. Positron emission tomography imaging of adenoviral-mediated transgene expression in liver cancer patients. Gastroenterology. 2005;128:1787–1795. doi: 10.1053/j.gastro.2005.03.024. [DOI] [PubMed] [Google Scholar]
- 72.Malecki M. HER2/neu gene expression products evaluated with superparamagnetic, genetically engineered antibodies. Proc S Dak Acad Sci. 2007;86:297–308. [PMC free article] [PubMed] [Google Scholar]
- 73.Stegman LD, Rehemtulla A, Beattie B, Kievit E, Lawrence TS, et al. Noninvasive quantitation of cytosine deaminase transgene expression in human tumor xenografts with in vivo magnetic resonance spectroscopy. Proc Natl Acad Sci U S A. 1999 Aug 17;96(17):9821–6. doi: 10.1073/pnas.96.17.9821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lawrence TS, Davis MA, Maybaum J. Dependence of 5-fluorouracil-mediated radiosensitization on DNA-directed effects. Int J Radiat Oncol Biol Phys. 1994;29:519–523. doi: 10.1016/0360-3016(94)90448-0. [DOI] [PubMed] [Google Scholar]
- 75.Jimenez T, Fox WP, Naus CC, Galipeau J, Belliveau DJ. Connexin over-expression differentially suppresses glioma growth and contributes to the bystander effect following HSV-thymidine kinase gene therapy. Cell Commun Adhes. 2006;13:79–92. doi: 10.1080/15419060600631771. [DOI] [PubMed] [Google Scholar]
- 76.Rainov NG. A phase III clinical evaluation of herpes simplex virus type 1 thymidine kinase and ganciclovir gene therapy as an adjuvant to surgical resection and radiation in adults with previously untreated glioblastoma multiforme. Hum Gene Ther. 2000;11:2389–2401. doi: 10.1089/104303400750038499. [DOI] [PubMed] [Google Scholar]
- 77.Li J, Zhang G, Liu T, Gu H, Yan L, et al. Construction of a novel vector expressing the fusion suicide gene yCDglyTK and hTERT-shRNA and its antitumor effects. Exp Ther Med. 2012;4:442–448. doi: 10.3892/etm.2012.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Degreve B, Johansson M, De Clercq E, Karlsson A, Balzarini J. Differential intracellular compartmentalization of herpetic thymidine kinases (TKs) in TK gene-transfected tumor cells: molecular characterization of the nuclear localization signal of herpes simplex virus type 1 TK. J Virol. 1998;72:9535–9543. doi: 10.1128/jvi.72.12.9535-9543.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Knecht W, Sandrini MP, Johansson K, Eklund H, Munch-Petersen B, et al. A few amino acid substitutions can convert deoxyribonucleoside kinase specificity from pyrimidines to purines. EMBO J. 2002;21:1873–1880. doi: 10.1093/emboj/21.7.1873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Finocchiaro LM, Fiszman GL, Karara AL, Glikin GC. Suicide gene and cytokines combined nonviral gene therapy for spontaneous canine melanoma. Cancer Gene Ther. 2008;15:165–172. doi: 10.1038/sj.cgt.7701096. [DOI] [PubMed] [Google Scholar]
- 81.Walther W, Petkov S, Kuvardina ON, Aumann J, Kobelt D, et al. Novel Clostridium perfringens enterotoxin suicide gene therapy for selective treatment of claudin-3- and -4-overexpressing tumors. Gene Ther. 2012;19:494–503. doi: 10.1038/gt.2011.136. [DOI] [PubMed] [Google Scholar]
- 82.Xiong J, Sun WJ, Wang WF, Liao ZK, Zhou FX, et al. Novel, chimeric, cancer-specific, and radiation-inducible gene promoters for suicide gene therapy of cancer. Cancer. 2012;118:536–548. doi: 10.1002/cncr.26289. [DOI] [PubMed] [Google Scholar]
- 83.Neschadim A, Wang JC, Lavie A, Medin JA. Bystander killing of malignant cells via the delivery of engineered thymidine-active deoxycytidine kinase for suicide gene therapy of cancer. Cancer Gene Ther. 2012;19:320–327. doi: 10.1038/cgt.2012.4. [DOI] [PubMed] [Google Scholar]
- 84.Niess H, Bao Q, Conrad C, Zischek C, Notohamiprodjo M, et al. Selective targeting of genetically engineered mesenchymal stem cells to tumor stroma microenvironments using tissue-specific suicide gene expression suppresses growth of hepatocellular carcinoma. Ann Surg. 2011;254:767–774. doi: 10.1097/SLA.0b013e3182368c4f. [DOI] [PubMed] [Google Scholar]
- 85.Leveille S, Samuel S, Goulet ML, Hiscott J. Enhancing VSV oncolytic activity with an improved cytosine deaminase suicide gene strategy. Cancer Gene Ther. 2011;18:435–443. doi: 10.1038/cgt.2011.14. [DOI] [PubMed] [Google Scholar]
- 86.Dong M, Li X, Hong LJ, Xie R, Zhao HL, et al. Advanced malignant pleural or peritoneal effusion in patients treated with recombinant adenovirus p53 injection plus cisplatin. J Int Med Res. 2008;36:1273–1278. doi: 10.1177/147323000803600614. [DOI] [PubMed] [Google Scholar]
- 87.Zhao WZ, Wang JK, Li W, Zhang XL. [Clinical research on recombinant human Ad-p53 injection combined with cisplatin in treatment of malignant pleural effusion induced by lung cancer] Ai Zheng. 2009;28:1324–1327. doi: 10.5732/cjc.009.10149. [DOI] [PubMed] [Google Scholar]
- 88.Voges J, Reszka R, Gossmann A, Dittmar C, Richter R, et al. Imaging-guided convection-enhanced delivery and gene therapy of glioblastoma. Ann Neurol. 2003;54:479–487. doi: 10.1002/ana.10688. [DOI] [PubMed] [Google Scholar]
- 89.Nasu Y, Saika T, Ebara S, Kusaka N, Kaku H, et al. Suicide gene therapy with adenoviral delivery of HSV-tK gene for patients with local recurrence of prostate cancer after hormonal therapy. Mol Ther. 2007;15:834–840. doi: 10.1038/sj.mt.6300096. [DOI] [PubMed] [Google Scholar]
- 90.Xu F, Li S, Li XL, Guo Y, Zou BY, et al. Phase I and biodistribution study of recombinant adenovirus vector-mediated herpes simplex virus thymidine kinase gene and ganciclovir administration in patients with head and neck cancer and other malignant tumors. Cancer Gene Ther. 2009;16:723–730. doi: 10.1038/cgt.2009.19. [DOI] [PubMed] [Google Scholar]
- 91.Li N, Zhou J, Weng D, Zhang C, Li L, et al. Adjuvant adenovirus-mediated delivery of herpes simplex virus thymidine kinase administration improves outcome of liver transplantation in patients with advanced hepatocellular carcinoma. Clin Cancer Res. 2007;13:5847–5854. doi: 10.1158/1078-0432.CCR-07-0499. [DOI] [PubMed] [Google Scholar]
- 92.Freytag SO, Movsas B, Aref I, Stricker H, Peabody J, et al. Phase I trial of replication-competent adenovirus-mediated suicide gene therapy combined with IMRT for prostate cancer. Mol Ther. 2007;15:1016–1023. doi: 10.1038/mt.sj.6300120. [DOI] [PubMed] [Google Scholar]
- 93.Freytag SO, Stricker H, Pegg J, Paielli D, Pradhan DG, et al. Phase I study of replication-competent adenovirus-mediated double-suicide gene therapy in combination with conventional-dose three-dimensional conformal radiation therapy for the treatment of newly diagnosed, intermediate- to high-risk prostate cancer. Cancer Res. 2003;63:7497–7506. [PubMed] [Google Scholar]
- 94.Nemunaitis J, Cunningham C, Senzer N, Kuhn J, Cramm J, et al. Pilot trial of genetically modified, attenuated Salmonella expressing the E. coli cytosine deaminase gene in refractory cancer patients. Cancer Gene Ther. 2003;10:737–744. doi: 10.1038/sj.cgt.7700634. [DOI] [PubMed] [Google Scholar]
- 95.Preuss E, Muik A, Weber K, Otte J, von Laer D, et al. Cancer suicide gene therapy with TK.007: superior killing efficiency and bystander effect. J Mol Med (Berl) 2011;89:1113–1124. doi: 10.1007/s00109-011-0777-8. [DOI] [PubMed] [Google Scholar]
- 96.Gruber C, Gratz IK, Murauer EM, Mayr E, Koller U, et al. Spliceosome-mediated RNA trans-splicing facilitates targeted delivery of suicide genes to cancer cells. Mol Cancer Ther. 2011;10:233–241. doi: 10.1158/1535-7163.MCT-10-0669. [DOI] [PubMed] [Google Scholar]
- 97.Di Stasi A, Tey SK, Dotti G, Fujita Y, Kennedy-Nasser A, et al. Inducible apoptosis as a safety switch for adoptive cell therapy. N Engl J Med. 2011;365:1673–1683. doi: 10.1056/NEJMoa1106152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Heimberger AB, Sampson JH. The PEPvIII-KLH (CDX-110) vaccine in glioblastoma multiforme patients. Expert Opin Biol Ther. 2009;9:1087–1098. doi: 10.1517/14712590903124346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Heider KH, Kuthan H, Stehle G, Munzert G. CD44v6: a target for antibody-based cancer therapy. Cancer Immunol Immunother. 2004;53:567–579. doi: 10.1007/s00262-003-0494-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zhang SS, Han ZP, Jing YY, Tao SF, Li TJ, et al. CD133(+)CXCR4(+) colon cancer cells exhibit metastatic potential and predict poor prognosis of patients. BMC Med. 2012;10:85. doi: 10.1186/1741-7015-10-85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Duarte S, Faneca H, de Lima MC. Non-covalent association of folate to lipoplexes: a promising strategy to improve gene delivery in the presence of serum. J Control Release. 2011;149:264–272. doi: 10.1016/j.jconrel.2010.10.032. [DOI] [PubMed] [Google Scholar]
- 102.Habashy HO, Powe DG, Staka CM, Rakha EA, Ball G, et al. Transferrin receptor (CD71) is a marker of poor prognosis in breast cancer and can predict response to tamoxifen. Breast Cancer Res Treat. 2010;119:283–293. doi: 10.1007/s10549-009-0345-x. [DOI] [PubMed] [Google Scholar]
- 103.Torres MP, Chakraborty S, Souchek J, Batra SK. Mucin-based targeted pancreatic cancer therapy. Curr Pharm Des. 2012;18:2472–2481. doi: 10.2174/13816128112092472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Malecki M, Anderson M, Beauchaine M, Seo S, Tombokan X. TRA-1-60(+), SSEA-4(+), Oct4A(+), Nanog(+) Clones of Pluripotent Stem Cells in the Embryonal Carcinomas of the Ovaries. J Stem Cell Res Ther. 2012 Nov 18;2(5):130. pii. [PMC free article] [PubMed] [Google Scholar]
- 105.Malecki M, Tombokan X, Anderson M, Malecki R, Beauchaine M. TRA-1-60+, SSEA-4+, POU5F1+, SOX2+, NANOG+ Clones of Pluripotent Stem Cells in the Embryonal Carcinomas of the Testes. J Stem Cell Res Ther. 2013;3:134. doi: 10.4172/2157-7633.1000134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Park HS, Cheon J, Cho HY, Ko YH, Bae JH, et al. In vivo characterization of a prostate-specific antigen promoter-based suicide gene therapy for the treatment of benign prostatic hyperplasia. Gene Ther. 2003;10:1129–1134. doi: 10.1038/sj.gt.3301972. [DOI] [PubMed] [Google Scholar]
- 107.Yamamoto M, Alemany R, Adachi Y, Grizzle WE, Curiel DT. Characterization of the cyclooxygenase-2 promoter in an adenoviral vector and its application for the mitigation of toxicity in suicide gene therapy of gastrointestinal cancers. Mol Ther. 2001;3:385–394. doi: 10.1006/mthe.2001.0275. [DOI] [PubMed] [Google Scholar]
- 108.Malecki M, Szybalski W. Isolation of single, intact chromosomes from single, selected ovarian cancer cells for in situ hybridization and sequencing. Gene. 2012;493:132–139. doi: 10.1016/j.gene.2011.11.044. [DOI] [PubMed] [Google Scholar]
- 109.Malecki M. Cancer suicide gene therapy: Apoptosis of the ovarian cancer cells induced by EGFRvIII targeted delivery and cell nucleus targeted expression of the DNase transgenes. J Genet Syndr Gene Ther. 2012;3:4. [Google Scholar]
- 110.Ishiwata N, Inase N, Fujie T, Tamaoka M, Yoshizawa Y. Suicide gene therapy using keratin 19 enhancer and promoter in malignant mesothelioma cells. Anticancer Res. 2003;23:1405–1409. [PubMed] [Google Scholar]
- 111.Song JS, Kim HP, Yoon WS, Lee KW, Kim MH, et al. Adenovirus-mediated suicide gene therapy using the human telomerase catalytic subunit (hTERT) gene promoter induced apoptosis of ovarian cancer cell line. Biosci Biotechnol Biochem. 2003;67:2344–2350. doi: 10.1271/bbb.67.2344. [DOI] [PubMed] [Google Scholar]
- 112.Malecki M, Hsu A, Truong L, Sanchez S. Molecular immunolabeling with recombinant single-chain variable fragment (scFv) antibodies designed with metal-binding domains. Proc Natl Acad Sci U S A. 2002;99:213–218. doi: 10.1073/pnas.261567298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Malecki M, Malecki R. Ovarian cancer suicide gene therapy with genetically engineered, transgenically expressed, intracellular scFv antibodies against anti- oxidative enzymes. Proc S Dak Acad Sci. 2008;87:249–260. [PMC free article] [PubMed] [Google Scholar]
- 114.Zhang Z, Lin J, Chu J, Ma Y, Zeng S, et al. Activation of caspase-3 noninvolved in the bystander effect of the herpes simplex virus thymidine kinase gene/ganciclovir (HSV-tk/GCV) system. J Biomed Opt. 2008;13:031209. doi: 10.1117/1.2937830. [DOI] [PubMed] [Google Scholar]
- 115.Beltinger C, Fulda S, Kammertoens T, Meyer E, Uckert W, et al. Herpes simplex virus thymidine kinase/ganciclovir-induced apoptosis involves ligand-independent death receptor aggregation and activation of caspases. Proc Natl Acad Sci U S A. 1999;96:8699–8704. doi: 10.1073/pnas.96.15.8699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Kim HA, Nam K, Lee M, Kim SW. Hypoxia/hepatoma dual specific suicide gene expression plasmid delivery using bio-reducible polymer for hepatocellular carcinoma therapy. J Control Release. 2013;171:1–10. doi: 10.1016/j.jconrel.2013.06.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Tang HL, Yuen KL, Tang HM, Fung MC. Reversibility of apoptosis in cancer cells. Br J Cancer. 2009;100:118–122. doi: 10.1038/sj.bjc.6604802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Hazai E, Hazai I, Ragueneau-Majlessi I, Chung SP, Bikadi Z, et al. Predicting substrates of the human breast cancer resistance protein using a support vector machine method. BMC Bioinformatics. 2013;14:130. doi: 10.1186/1471-2105-14-130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Deng W, Dai CL, Chen JJ, Kathawala RJ, Sun YL, et al. Tandutinib (MLN518) reverses multidrug resistance by inhibiting the efflux activity of the multidrug resistance protein 7 (ABCC10) Oncol Rep. 2013;29:2479–2485. doi: 10.3892/or.2013.2362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Sims JT, Ganguly SS, Bennett H, Friend JW, Tepe J, et al. Imatinib reverses doxorubicin resistance by affecting activation of STAT3-dependent NF-PB and HSP27/p38/AKT pathways and by inhibiting ABCB1. PLoS One. 2013;8:e55509. doi: 10.1371/journal.pone.0055509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Zhao WT, Wang YT, Huang ZW, Fang J. BRCA2 affects the efficiency of DNA double-strand break repair in response to N-nitroso compounds with differing carcinogenic potentials. Oncol Lett. 2013;5:1948–1954. doi: 10.3892/ol.2013.1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Ribeiro E, Ganzinelli M, Andreis D, Bertoni R, Giardini R, et al. Triple negative breast cancers have a reduced expression of DNA repair genes. PLoS One. 2013;8:e66243. doi: 10.1371/journal.pone.0066243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Park HH. Structural features of caspase-activating complexes. Int J Mol Sci. 2012;13:4807–4818. doi: 10.3390/ijms13044807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.MacKenzie SH, Clark AC. Targeting cell death in tumors by activating caspases. Curr Cancer Drug Targets. 2008;8:98–109. doi: 10.2174/156800908783769391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Carlotti F, Zaldumbide A, Martin P, Boulukos KE, Hoeben RC, et al. Development of an inducible suicide gene system based on human caspase 8. Cancer Gene Ther. 2005;12:627–639. doi: 10.1038/sj.cgt.7700825. [DOI] [PubMed] [Google Scholar]
- 126.Xie X, Zhao X, Liu Y, Zhang J, Matusik RJ, et al. Adenovirus-mediated tissue-targeted expression of a caspase-9-based artificial death switch for the treatment of prostate cancer. Cancer Res. 2001;61:6795–6804. [PubMed] [Google Scholar]
- 127.Shariat SF, Desai S, Song W, Khan T, Zhao J, et al. Adenovirus-mediated transfer of inducible caspases: a novel “death switch” gene therapeutic approach to prostate cancer. Cancer Res. 2001;61:2562–2571. [PubMed] [Google Scholar]
- 128.Putt KS, Chen GW, Pearson JM, Sandhorst JS, Hoagland MS, et al. Small-molecule activation of procaspase-3 to caspase-3 as a personalized anticancer strategy. Nat Chem Biol. 2006;2:543–550. doi: 10.1038/nchembio814. [DOI] [PubMed] [Google Scholar]
- 129.Takahashi K, Yamanaka S. Induced pluripotent stem cells in medicine and biology. Development. 2013;140:2457–2461. doi: 10.1242/dev.092551. [DOI] [PubMed] [Google Scholar]
- 130.Das AK, Pal R. Induced pluripotent stem cells (iPSCs): the emergence of a new champion in stem cell technology-driven biomedical applications. J Tissue Eng Regen Med. 2010;4:413–421. doi: 10.1002/term.258. [DOI] [PubMed] [Google Scholar]
- 131.Li M, Chen M, Han W, Fu X. How far are induced pluripotent stem cells from the clinic? Ageing Res Rev. 2010;9:257–264. doi: 10.1016/j.arr.2010.03.001. [DOI] [PubMed] [Google Scholar]
- 132.Gerecht-Nir S, Itskovitz-Eldor J. Human embryonic stem cells: a potential source for cellular therapy. Am J Transplant. 2004;4(Suppl 6):51–57. doi: 10.1111/j.1600-6135.2004.0345.x. [DOI] [PubMed] [Google Scholar]
- 133.Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 134.Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318:1917–1920. doi: 10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- 135.Park IH, Zhao R, West JA, Yabuuchi A, Huo H, et al. Reprogramming of human somatic cells to pluripotency with defined factors. Nature. 2008;451:141–146. doi: 10.1038/nature06534. [DOI] [PubMed] [Google Scholar]
- 136.Mali P, Ye Z, Hommond HH, Yu X, Lin J, et al. Improved efficiency and pace of generating induced pluripotent stem cells from human adult and fetal fibroblasts. Stem Cells. 2008;26:1998–2005. doi: 10.1634/stemcells.2008-0346. [DOI] [PubMed] [Google Scholar]
- 137.Lowry WE, Richter L, Yachechko R, Pyle AD, Tchieu J, et al. Generation of human induced pluripotent stem cells from dermal fibroblasts. Proc Natl Acad Sci U S A. 2008;105:2883–2888. doi: 10.1073/pnas.0711983105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Fong CY, Peh GS, Gauthaman K, Bongso A. Separation of SSEA-4 and TRA-1-60 labelled undifferentiated human embryonic stem cells from a heterogeneous cell population using magnetic-activated cell sorting (MACS) and fluorescence-activated cell sorting (FACS) Stem Cell Rev. 2009;5:72–80. doi: 10.1007/s12015-009-9054-4. [DOI] [PubMed] [Google Scholar]
- 139.Pruszak J, Sonntag KC, Aung MH, Sanchez-Pernaute R, Isacson O. Markers and methods for cell sorting of human embryonic stem cell-derived neural cell populations. Stem Cells. 2007;25:2257–2268. doi: 10.1634/stemcells.2006-0744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Malecki M. Improved Targeting and Enhanced Retention of the Human, Autologous, Fibroblast-Derived, Induced, Pluripotent Stem Cells to the Sarcomeres of the Infarcted Myocardium with the Aid of the Bioengineered, Heterospecific, Tetravalent Antibodies. J Stem Cell Res Ther. 2013;3:138. doi: 10.4172/2157-7633.1000138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Zhao TC, Tseng A, Yano N, Tseng Y, Davol PA, et al. Targeting human CD34+ hematopoietic stem cells with anti-CD45 × anti-myosin light-chain bispecific antibody preserves cardiac function in myocardial infarction. J Appl Physiol. 2008;104:1793–1800. doi: 10.1152/japplphysiol.01109.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Lee RJ, Fang Q, Davol PA, Gu Y, Sievers RE, et al. Antibody targeting of stem cells to infarcted myocardium. Stem Cells. 2007;25:712–717. doi: 10.1634/stemcells.2005-0602. [DOI] [PubMed] [Google Scholar]
- 143.Lowry WE, Quan WL. Roadblocks en route to the clinical application of induced pluripotent stem cells. J Cell Sci. 2010;123:643–651. doi: 10.1242/jcs.054304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Ben-David U, Benvenisty N. The tumorigenicity of human embryonic and induced pluripotent stem cells. Nat Rev Cancer. 2011;11:268–277. doi: 10.1038/nrc3034. [DOI] [PubMed] [Google Scholar]
- 145.Goldring CE, Duffy PA, Benvenisty N, Andrews PW, Ben-David U, et al. Assessing the safety of stem cell therapeutics. Cell Stem Cell. 2011;8:618–628. doi: 10.1016/j.stem.2011.05.012. [DOI] [PubMed] [Google Scholar]
- 146.Kooreman NG, Wu JC. Tumorigenicity of pluripotent stem cells: biological insights from molecular imaging. J R Soc Interface. 2010;7(Suppl 6):S753–763. doi: 10.1098/rsif.2010.0353.focus. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Knoepfler PS. Key anticipated regulatory issues for clinical use of human induced pluripotent stem cells. Regen Med. 2012;7:713–720. doi: 10.2217/rme.12.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Tonge PD, Shigeta M, Schroeder T, Andrews PW. Functionally defined substates within the human embryonic stem cell compartment. Stem Cell Res. 2011;7:145–153. doi: 10.1016/j.scr.2011.04.006. [DOI] [PubMed] [Google Scholar]
- 149.Tang C, Lee AS, Volkmer JP, Sahoo D, Nag D, et al. An antibody against SSEA-5 glycan on human pluripotent stem cells enables removal of teratoma-forming cells. Nat Biotechnol. 2011;29:829–834. doi: 10.1038/nbt.1947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Ben-David U, Nudel N, Benvenisty N. Immunologic and chemical targeting of the tight-junction protein Claudin-6 eliminates tumorigenic human pluripotent stem cells. Nat Commun. 2013;4:1992. doi: 10.1038/ncomms2992. [DOI] [PubMed] [Google Scholar]
- 151.Schuldiner M, Itskovitz-Eldor J, Benvenisty N. Selective ablation of human embryonic stem cells expressing a “suicide” gene. Stem Cells. 2003;21:257–265. doi: 10.1634/stemcells.21-3-257. [DOI] [PubMed] [Google Scholar]
- 152.Rong Z, Fu X, Wang M, Xu Y. A scalable approach to prevent teratoma formation of human embryonic stem cells. J Biol Chem. 2012;287:32338–32345. doi: 10.1074/jbc.M112.383810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Cheng F, Ke Q, Chen F, Cai B, Gao Y, et al. Protecting against wayward human induced pluripotent stem cells with a suicide gene. Biomaterials. 2012;33:3195–3204. doi: 10.1016/j.biomaterials.2012.01.023. [DOI] [PubMed] [Google Scholar]