Abstract
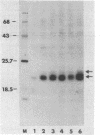
Two flat revertants have been isolated from mutagen-treated populations of Kirsten murine sarcoma virus (Ki-MuSV)-transformed NIH/3T3 cells. These revertants, which appear to be cellular variants resistant to transformation by the Ki-MuSV oncogene v-Ki-ras, contain Ki-MuSV-specific DNA, elevated levels of the v-Ki-ras gene product p21, and rescuable transforming virus. Cell hybridization studies indicated that the revertant phenotype is dominant in hybrids between revertant cells and cells transformed by Ki-MuSV or the closely related Harvey MuSV and BALB MuSV. Analysis of hybrid cells resulting from the fusion of these revertants to cell lines transformed by other retroviruses showed that the action of certain oncogenes structurally unrelated to v-Ki-ras also could be suppressed. Thus, there appear to be functional relationships and diversities among transforming genes (oncogenes) not readily apparent from their structural characteristics.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aaronson S. A., Barbacid M. Origin and biological properties of a new BALB/c mouse sarcoma virus. J Virol. 1978 Aug;27(2):366–373. doi: 10.1128/jvi.27.2.366-373.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aaronson S. A., Weaver C. A. Characterization of murine sarcoma virus (Kirsten) transformation of mouse and human cells. J Gen Virol. 1971 Nov;13(2):245–252. doi: 10.1099/0022-1317-13-2-245. [DOI] [PubMed] [Google Scholar]
- Anderson S. M., Scolnick E. M. Construction and isolation of a transforming murine retrovirus containing the src gene of Rous sarcoma virus. J Virol. 1983 May;46(2):594–605. doi: 10.1128/jvi.46.2.594-605.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bassin R. H., Tuttle N., Fischinger P. J. Isolation of murine sarcoma virus-transformed mouse cells which are negative for leukemia virus from agar suspension cultures. Int J Cancer. 1970 Jul 15;6(1):95–107. doi: 10.1002/ijc.2910060114. [DOI] [PubMed] [Google Scholar]
- Benjamin T. L., Burger M. M. Absence of a cell membrane alteration function in non-transforming mutants of polyoma virus. Proc Natl Acad Sci U S A. 1970 Oct;67(2):929–934. doi: 10.1073/pnas.67.2.929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boettiger D. Virogenic nontransformed cells isolated following infection of normal rat kidney cells with B77 strain Rous sarcoma virus. Cell. 1974 Sep;3(1):71–76. doi: 10.1016/0092-8674(74)90042-7. [DOI] [PubMed] [Google Scholar]
- Chang E. H., Furth M. E., Scolnick E. M., Lowy D. R. Tumorigenic transformation of mammalian cells induced by a normal human gene homologous to the oncogene of Harvey murine sarcoma virus. Nature. 1982 Jun 10;297(5866):479–483. doi: 10.1038/297479a0. [DOI] [PubMed] [Google Scholar]
- Coffin J. M., Varmus H. E., Bishop J. M., Essex M., Hardy W. D., Jr, Martin G. S., Rosenberg N. E., Scolnick E. M., Weinberg R. A., Vogt P. K. Proposal for naming host cell-derived inserts in retrovirus genomes. J Virol. 1981 Dec;40(3):953–957. doi: 10.1128/jvi.40.3.953-957.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper G. M. Cellular transforming genes. Science. 1982 Aug 27;217(4562):801–806. doi: 10.1126/science.6285471. [DOI] [PubMed] [Google Scholar]
- Davidson R. L., Gerald P. S. Improved techniques for the induction of mammalian cell hybridization by polyethylene glycol. Somatic Cell Genet. 1976 Mar;2(2):165–176. doi: 10.1007/BF01542629. [DOI] [PubMed] [Google Scholar]
- DeLeo A. B., Shiku H., Takahashi T., John M., Old L. J. Cell surface antigens of chemically induced sarcomas of the mouse. I. Murine leukemia virus-related antigens and alloantigens on cultured fibroblasts and sarcoma cells: description of a unique antigen on BALB/c Meth A sarcoma. J Exp Med. 1977 Sep 1;146(3):720–734. doi: 10.1084/jem.146.3.720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng C. T., Boettiger D., Macpherson I., Varmus H. E. The persistence and expression of virus-specific DNA in revertants of Rous sarcoma virus-transformed BHK-21 cells. Virology. 1974 Dec;62(2):512–521. doi: 10.1016/0042-6822(74)90411-5. [DOI] [PubMed] [Google Scholar]
- Dhar R., Ellis R. W., Shih T. Y., Oroszlan S., Shapiro B., Maizel J., Lowy D., Scolnick E. Nucleotide sequence of the p21 transforming protein of Harvey murine sarcoma virus. Science. 1982 Sep 3;217(4563):934–936. doi: 10.1126/science.6287572. [DOI] [PubMed] [Google Scholar]
- Duran-Troise G., Bassin R. H., Rein A., Gerwin B. I. Loss of Fv-1 restriction in Balb/3T3 cells following infection with a single N tropic murine leukemia virus particle. Cell. 1977 Mar;10(3):479–488. doi: 10.1016/0092-8674(77)90035-6. [DOI] [PubMed] [Google Scholar]
- Dyson P. J., Quade K., Wyke J. A. Expression of the ASV src gene in hybrids between normal and virally transformed cells: specific suppression occurs in some hybrids but not others. Cell. 1982 Sep;30(2):491–498. doi: 10.1016/0092-8674(82)90246-x. [DOI] [PubMed] [Google Scholar]
- Even J., Anderson S. J., Hampe A., Galibert F., Lowy D., Khoury G., Sherr C. J. Mutant feline sarcoma proviruses containing the viral oncogene (v-fes) and either feline or murine control elements. J Virol. 1983 Mar;45(3):1004–1016. doi: 10.1128/jvi.45.3.1004-1016.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischinger P. J., Nomura S., Peebles P. T., Haapala D. K., Bassin R. H. Reversion of murine sarcoma virus transformed mouse cells: variants without a rescuable sarcoma virus. Science. 1972 Jun 2;176(4038):1033–1035. doi: 10.1126/science.176.4038.1033. [DOI] [PubMed] [Google Scholar]
- Frankel A. S., Haapala D. K., Neubauer R. L., Fischinger P. J. Elimination of the sarcoma genome from murine sarcoma virus transformed cat cells. Science. 1976 Mar 26;191(4233):1264–1266. doi: 10.1126/science.1257745. [DOI] [PubMed] [Google Scholar]
- Furth M. E., Davis L. J., Fleurdelys B., Scolnick E. M. Monoclonal antibodies to the p21 products of the transforming gene of Harvey murine sarcoma virus and of the cellular ras gene family. J Virol. 1982 Jul;43(1):294–304. doi: 10.1128/jvi.43.1.294-304.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gazdar A. F., Stull H. B., Chopra H. C., Ikawa Y. Properties of flat variants of murine sarcoma virus transformed non-producer cells isolated after high-temperature passage. Int J Cancer. 1974 Feb 15;13(2):219–226. doi: 10.1002/ijc.2910130209. [DOI] [PubMed] [Google Scholar]
- Gisselbrecht S., Bassin R. H., Gerwin B. I., Rein A. Dual susceptibility of A 3T3 mouse cell line to infection by N- and B-tropic murine leukemia virus: apparent lack of expression of the FV-1 gene. Int J Cancer. 1974 Jul 15;14(1):106–113. doi: 10.1002/ijc.2910140113. [DOI] [PubMed] [Google Scholar]
- Greenberger J. S., Anderson G. R., Aaronson S. A. Transformation-defective virus mutants in a class of morphologic revertants of sarcoma virus transformed nonproducer cells. Cell. 1974 Aug;2(4):279–286. doi: 10.1016/0092-8674(74)90022-1. [DOI] [PubMed] [Google Scholar]
- Hatanaka M., Klein R., Toni R., Walker J., Gilden R. Mutants of nonproducer cell lines transformed by murine sarcoma viruses. I. Induction, isolation, particle production, and tumorigenicity. J Exp Med. 1973 Aug 1;138(2):356–363. doi: 10.1084/jem.138.2.356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue H., Yutsudo M., Hakura A. Rat mutant cells showing temperature sensitivity for transformation by wild-type Moloney murine sarcoma virus. Virology. 1983 Feb;125(1):242–245. doi: 10.1016/0042-6822(83)90078-8. [DOI] [PubMed] [Google Scholar]
- Krzyzek R. A., Lau A. F., Faras A. J., Spector D. H. Post-transcriptional control of avian oncornavirus transforming gene sequences in mammalian cells. Nature. 1977 Sep 8;269(5624):175–179. doi: 10.1038/269175a0. [DOI] [PubMed] [Google Scholar]
- LITTLEFIELD J. W. SELECTION OF HYBRIDS FROM MATINGS OF FIBROBLASTS IN VITRO AND THEIR PRESUMED RECOMBINANTS. Science. 1964 Aug 14;145(3633):709–710. doi: 10.1126/science.145.3633.709. [DOI] [PubMed] [Google Scholar]
- MACPHERSON I., MONTAGNIER L. AGAR SUSPENSION CULTURE FOR THE SELECTIVE ASSAY OF CELLS TRANSFORMED BY POLYOMA VIRUS. Virology. 1964 Jun;23:291–294. doi: 10.1016/0042-6822(64)90301-0. [DOI] [PubMed] [Google Scholar]
- MOORHEAD P. S., NOWELL P. C., MELLMAN W. J., BATTIPS D. M., HUNGERFORD D. A. Chromosome preparations of leukocytes cultured from human peripheral blood. Exp Cell Res. 1960 Sep;20:613–616. doi: 10.1016/0014-4827(60)90138-5. [DOI] [PubMed] [Google Scholar]
- Marshall C. J. Suppression of the transformed phenotype with retention of the viral 'src' gene in cell hybrids between Rous sarcoma virus-transformed rat cells and untransformed mouse cells. Exp Cell Res. 1980 Jun;127(2):373–384. doi: 10.1016/0014-4827(80)90442-5. [DOI] [PubMed] [Google Scholar]
- Ozer H. L., Jha K. K. Malignancy and transformation: expression in somatic cell hybrids and variants. Adv Cancer Res. 1977;25:53–93. doi: 10.1016/s0065-230x(08)60632-6. [DOI] [PubMed] [Google Scholar]
- Reddy E. P., Reynolds R. K., Santos E., Barbacid M. A point mutation is responsible for the acquisition of transforming properties by the T24 human bladder carcinoma oncogene. Nature. 1982 Nov 11;300(5888):149–152. doi: 10.1038/300149a0. [DOI] [PubMed] [Google Scholar]
- Shih T. Y., Papageorge A. G., Stokes P. E., Weeks M. O., Scolnick E. M. Guanine nucleotide-binding and autophosphorylating activities associated with the p21src protein of Harvey murine sarcoma virus. Nature. 1980 Oct 23;287(5784):686–691. doi: 10.1038/287686a0. [DOI] [PubMed] [Google Scholar]
- Shih T. Y., Weeks M. O., Young H. A., Scholnick E. M. Identification of a sarcoma virus-coded phosphoprotein in nonproducer cells transformed by Kirsten or Harvey murine sarcoma virus. Virology. 1979 Jul 15;96(1):64–79. doi: 10.1016/0042-6822(79)90173-9. [DOI] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Stehelin D., Varmus H. E., Bishop J. M., Vogt P. K. DNA related to the transforming gene(s) of avian sarcoma viruses is present in normal avian DNA. Nature. 1976 Mar 11;260(5547):170–173. doi: 10.1038/260170a0. [DOI] [PubMed] [Google Scholar]
- Stephenson J. R., Reynolds R. K., Aaronson S. A. Characterization of morphologic revertants of murine and avian sarcoma virus-transformed cells. J Virol. 1973 Feb;11(2):218–222. doi: 10.1128/jvi.11.2.218-222.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabin C. J., Bradley S. M., Bargmann C. I., Weinberg R. A., Papageorge A. G., Scolnick E. M., Dhar R., Lowy D. R., Chang E. H. Mechanism of activation of a human oncogene. Nature. 1982 Nov 11;300(5888):143–149. doi: 10.1038/300143a0. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuchida N., Ryder T., Ohtsubo E. Nucleotide sequence of the oncogene encoding the p21 transforming protein of Kirsten murine sarcoma virus. Science. 1982 Sep 3;217(4563):937–939. doi: 10.1126/science.6287573. [DOI] [PubMed] [Google Scholar]
- Varmus H. E., Quintrell N., Ortiz S. Retroviruses as mutagens: insertion and excision of a nontransforming provirus alter expression of a resident transforming provirus. Cell. 1981 Jul;25(1):23–36. doi: 10.1016/0092-8674(81)90228-2. [DOI] [PubMed] [Google Scholar]
- Varmus H. E., Quintrell N., Wyke J. Revertants of an ASV-transformed rat cell line have lost the complete provius or sustained mutations in src. Virology. 1981 Jan 15;108(1):28–46. doi: 10.1016/0042-6822(81)90525-0. [DOI] [PubMed] [Google Scholar]
- Vogel A., Pollack R. Methods for obtaining revertants of transformed cells. Methods Cell Biol. 1974;8(0):75–92. doi: 10.1016/s0091-679x(08)60444-6. [DOI] [PubMed] [Google Scholar]
- Weinberg R. A. Fewer and fewer oncogenes. Cell. 1982 Aug;30(1):3–4. doi: 10.1016/0092-8674(82)90003-4. [DOI] [PubMed] [Google Scholar]
- Wyke J. A method of isolating cells incapable of multiplication in suspension culture. Exp Cell Res. 1971 May;66(1):203–208. doi: 10.1016/s0014-4827(71)80030-7. [DOI] [PubMed] [Google Scholar]