Abstract
Doxapram is a respiratory stimulant used to treat hypoventilation. Here we investigated whether doxapram could also trigger respiratory neuroplasticity. Specifically, we hypothesized that intermittent delivery of doxapram at low doses would lead to long-lasting increases (i.e., facilitation) of phrenic motor output in anesthetized, vagotomized, and mechanically-ventilated rats. Doxapram was delivered intravenously in a single bolus (2 or 6 mg/kg) or as a series of 3 injections (2 mg/kg) at 5 min intervals. Control groups received pH-matched saline injections (vehicle) or no treatment (anesthesia time control). Doxapram evoked an immediate increase in phrenic output in all groups, but a persistent increase in burst amplitude only occurred after repeated dosing with 2 mg/kg. At 60 min following the last injection, phrenic burst amplitude was 168±24% of baseline (%BL) in the group receiving 3 injections (P < 0.05 vs. controls), but was 103±8%BL and 112±4%BL in the groups receiving a single dose of 2 or 6 mg/kg, respectively. Following bilateral section of the carotid sinus nerves, the acute phrenic response to doxapram (2 mg/kg) was reduced by 68% suggesting that at low doses the drug was acting primarily via the carotid chemoreceptors. We conclude that intermittent application of doxapram can trigger phrenic neuroplasticity, and this approach might be of use in the context of respiratory rehabilitation following neurologic injury.
Keywords: phrenic motor facilitation, doxapram, respiratory neuroplasticity
Introduction
Targeted induction of neuroplasticity in respiratory neurons and/or networks may be therapeutically beneficial in clinical conditions associated with blunted respiratory motor output (Mitchell, 2007). The majority of prior studies of respiratory neuroplasticity in animal models (MacFarlane, et al., 2009) and humans (Mateika and Sandhu, 2011) have used periodic reductions in inspired oxygen (i.e., intermittent hypoxia) as the stimulus for inducing plasticity. In this regard, hypoxia has great value as both an experimental tool as well as an adjunct to conventional clinical rehabilitation strategies (Trumbower, et al., 2011). There has been comparatively less work, however, on the use of pharmacologic agents for targeted induction of respiratory neuroplasticity. Since increases in respiratory motor activity have the potential to initiate neuroplasticity, pharmacologic agents which stimulate breathing could provide a useful tool in the contexts of neurorehabilitation. For example, Nantwi and colleagues found that the respiratory stimulant theophylline triggers persistent increases in phrenic motor activity following spinal cord injury (Nantwi, et al., 2003). Phrenic motor recovery was observed well beyond the theophylline application thereby confirming that the drug had triggered neuroplastic processes. Doxapram is another respiratory stimulant which might be of value for targeted induction of respiratory neuroplasticity. This FDA approved drug is used to stimulate breathing during acute respiratory insufficiency (Burki, 1984, Moser, et al., 1973) such as occurs during opioid-induced respiratory depression (Bowdle, 1988). Although doxapram is classified as an analeptic agent (i.e., a neural stimulant), it has a large margin of safety with a therapeutic ratio of about 20–30 compared with 2–4 for most analeptic drugs (Funderburk, et al., 1966, Pleuvry, 1978). In other words, doses that are considerably higher than the therapeutic range are well-tolerated and appear to be clinically safe.
Most evidence indicates low doses of doxapram increases breathing via stimulation of the carotid bodies (Yost, 2006). In turn, this suggests that doxapram may be used to trigger respiratory neuroplasticity since appropriate patterns of carotid chemoafferent stimulation can lead to prolonged increases in respiratory motor output (Hayashi, et al., 1993, Millhorn, et al., 1980, Peng and Prabhakar, 2003). For example, intermittent electrical stimulation of the carotid sinus nerve increases phrenic nerve activity well beyond the period of stimulation (Millhorn, et al., 1980). In addition, a short period of intermittent hypoxia triggers a prolonged (>60 min) increase in phrenic motor output (i.e., long term facilitation, LTF) (Bach and Mitchell, 1996) that is significantly attenuated following carotid chemoreceptor denervation (Bavis and Mitchell, 2003). The pattern of carotid chemoafferent stimulation is a particularly important consideration, since facilitation of phrenic activity is more effectively triggered by intermittent than continuous stimulation (Baker and Mitchell, 2000). Based on these considerations, the current experiments were designed to test the hypothesis that repeated application of doxapram can induce long-lasting phrenic motor facilitation.
Methods
Animals
All procedures were approved by the University of Florida Institutional Animal Care and Use Committee. To determine the impact of doxapram on phrenic motor facilitation (Dale-Nagle, et al., 2010), adult male Sprague Dawley rats (Harlan Inc., Indianapolis, IN, USA) were randomly divided into the following experimental groups: Intermittent delivery of low-dose doxapram (i.e., 3 × 2 mg/kg, n=5), single bolus delivery of doxapram at a low- (2 mg/kg, n=6) or high-dose (6 mg/kg, n=6), pH matched saline (i.e., 3 × vehicle injection, n=5), and finally an anesthesia time control group which received no treatment (n=5). Body weight (g) was similar between the experimental groups: intermittent doxapram = 395 ± 20 g, single doxapram = 397 ± 17g (2 mg/kg) and 376 ± 16g (6 mg/kg), vehicle = 378 ± 20g and time control = 378 ± 16g. In additional experiments, the carotid sinus nerves were sectioned before giving a range of doxapram doses as a single bolus (2, 4, 8, and 16 mg/kg, N=5). These latter experiments were conducted to confirm that the phrenic motor response to the doxapram doses used in this study were mediated primarily via the carotid bodies.
Experimental Preparation
Our experimental preparation for nerve recordings has been described previously (Sandhu, et al., 2010). Rats were initially anesthetized with 5% isoflurane (balance O2) in a small chamber and were then transferred to a nose cone where they breathed a mixture of 2–3% isoflurane in 50% O2 (balance N2). The trachea was cannulated with PE-240 tubing and rats were mechanically ventilated for the remainder of the experiment. To minimize atelectasis, the lungs were briefly hyperinflated by occluding the expired line once per hour. A PE-50 femoral catheter was placed and used to induce urethane anesthesia (1.6 g/kg, i.v.; 0.12 g/ml distilled water). The urethane solution was gradually administered over a period of 10 min, and isoflurane was gradually titrated down during this time. A femoral arterial catheter was also used to measure blood pressure (Statham P-10EZ pressure transducer, CP122 AC/DC strain gage amplifier, Grass Instruments, West Warwick, RI, USA) and to periodically withdraw blood samples.
Rats were bilaterally vagotomized to prevent entrainment of phrenic motor output with the ventilator and a nicotinic cholinergic antagonist was used to block neuromuscular transmission (pancuronium bromide, 2.5 mg/kg, i.v.) and prevent spontaneous breathing efforts. Following paralysis, the adequacy of anesthesia was monitored by observing blood pressure and phrenic nerve activity during toe pinch; supplemental urethane was given if indicated (0.3 g/kg, i.v.). An infusion of lactated Ringer’s solution and sodium bicarbonate (3:1, 1.5 ml/h) was maintained to promote acid-base balance. Arterial partial pressures of O2 (PaO2) and CO2 (PaCO2) as well as pH were determined from 0.2 ml arterial blood samples using an i-Stat blood gas analyzer (Heska, Fort Collins, CO, USA). Blood gas and pH values were corrected to the rectal temperature measured at the time of the blood sample. The end-tidal CO2 partial pressure (PETCO2) was continuously sampled using a mainstream CO2 analyzer (Capnogard CO2 monitor, Novametrix Medical Systems, Wallingford, CT, USA). The sensor was placed a few cm from the tracheostomy tube on the expired line of the ventilator circuit. Rectal temperature was maintained within 37±1 °C using a rectal thermistor coupled with a servo-controlled heating pad (model TC-1000, CWE Inc., Ardmore, PA, USA).
The phrenic nerves were isolated medial to the brachial plexus using a ventral surgical approach. The electrical activity in the phrenic nerves was recorded using silver wire electrodes with a monopolar configuration. The signal was amplified (1000x) and band pass filtered (300–10,000 Hz, notch=60 Hz) using a differential A/C amplifier (Model 1700, A-M Systems, Carlsborg, WA, USA). The amplified signal was full-wave rectified and moving averaged (time constant 100 ms; model MA-1000; CWE Inc., Ardmore, PA, USA). Data were digitized using a CED Power 1401 data acquisition interface and recorded on a PC (Dell Precision T1600) using Spike2 v. 7.10 software (Cambridge Electronic Design Limited, Cambridge, England).
Doxapram injections
After an adequate plane of anesthesia was established, the PETCO2 was maintained at 40±2 mmHg for 5–10 min with inspired O2 fraction (FIO2) held at 0.5. The end-tidal CO2 apneic threshold for inspiratory phrenic activity was then determined by gradually increasing the ventilator pump rate until phrenic inspiratory bursting ceased. Apnea was maintained for one min, and the ventilator rate was then gradually decreased until inspiratory bursting resumed. The PETCO2 associated with the onset of inspiratory bursting was designated as the “CO2 recruitment threshold”. PETCO2 was monitored for the duration of the experiment and the ventilator rate was adjusted as necessary to maintain PETCO2 at 2 mmHg above the CO2 recruitment threshold for the duration of the experiment to maintain isocapnia. After a 10–20 min baseline period, a 0.2 ml arterial blood sample was drawn for blood gas analysis, and the protocol was initiated. Doxapram was prepared in 0.9% saline at a concentration of 2 mg/ml and delivered through the femoral venous catheter. Rats in the intermittent doxapram group received three injections of 2 mg/kg (for a total dose of 6 mg/kg) at 5 min intervals. The single injection groups were given a bolus injection with either the same total dose as the intermittent group (6 mg/kg) or a lower dose (2 mg/kg). Animals in the anesthesia “time control” group did not receive any injections, but all other aspects of the protocol were maintained. The doxapram solution had a pH of 5.5, and accordingly a vehicle injection procedure was needed. Saline was pH-matched to 5.5 by adding hydrochloric acid, and the vehicle group received three injections of the saline solution. Arterial blood samples were obtained at 30 and 60 min post-doxapram delivery, or at equivalent time-points during the vehicle injections and time control.
Carotid sinus nerve section (CSNX)
In a separate group of rats, the carotid sinus nerve was isolated using a ventral surgical approach and sectioned bilaterally prior to delivering a series of doxapram injections (2 – 16 mg/Kg). Blunt dissection was initially used to visualize the bifurcation of the common carotid artery. The carotid sinus nerve was identified at its junction with the glossopharyngeal nerve and sectioned near the bifurcation. Phrenic nerve recordings were conducted as described above. Once a stable baseline had been obtained, a 2 mg/kg injection of doxapram was given as described above. Subsequent doses of 4, 8 and 16 mg/kg were given after phrenic output had returned to baseline levels.
Data analyses
Phrenic neurograms were analyzed as recently described (Lee, et al., 2010). For assessment of phrenic motor facilitation, peak integrated phrenic amplitude (∫Phr; expressed in mV) and burst frequency (bursts/min) were averaged over 1 min bins during baseline and at both 30 and 60 min after doxapram injections. To assess the acute doxapram response, phrenic amplitude and frequency were averaged over a 30 sec period immediately following doxapram injection. Burst amplitude and frequency were expressed as a percentage of baseline values. Statistical tests were conducted using SigmaPlot v. 12 software. Mean arterial pressure (MAP) was calculated by using the equation: MAP = [(diastolic pressure × 2) + systolic pressure]/3. Statistical differences were determined using a two-way repeated measures analysis of variance (RM ANOVA) with factor 1 as time point (i.e. injection 1, 2 and 3 or 30 and 60 min post-injection) and factor 2 as treatment (i.e. intermittent doxapram, 2mg/kg or 6 mg/kg single doxapram, pH-matched saline (i.e., vehicle injection) and time control). When applicable, the Student–Neuman–Keuls test was used for post hoc analyses. Body weight and the effect of doxapram after carotid sinus nerve sectioning were compared between groups using one-way ANOVA. Differences were considered statistically significant when the P value ≤0.05. All dataare presented as mean ± SEM.
Results
Intravenous doxapram delivery resulted in an increase of MAP (P < 0.001 vs. baseline, Table 1). However, changes in MAP were transient, and values were not significantly different from baseline in any of the experimental groups when assessed at 30 or 60 min post-doxapram delivery (all P > 0.35, Table 2). Arterial blood gases measured before doxapram administration (baseline) and 60 min after the last injection were not significantly different (all P > 0.21, Table 2). Blood gas values were also similar between the experimental groups (Table 2).
Table 1.
Mean arterial blood pressure (MAP) during baseline, first doxapram injection, and at 30 and 60 min post-doxapram or corresponding time points in control groups.
| Baseline | Injection 1 | 30 min | 60 min | |
|---|---|---|---|---|
| Intermittent doxapram | 94 ± 8 | 129 ± 11* | 96 ± 11 | 87 ± 11 |
| Single doxapram 2mg/kg | 106 ± 16 | 138 ± 19* | 109 ± 17 | 99 ± 14 |
| Single doxapram 6mg/kg | 101 ± 13 | 151 ± 7* | 99 ± 17 | 103 ± 11 |
| Vehicle injections | 112 ± 20 | 124 ± 21 | 108 ± 21 | 101 ± 21 |
| Time control | 102 ± 13 | 102 ± 11 | 95 ± 11 | 95 ± 12 |
different from baseline, 30 min and 60 min post-injection.
Table 2.
Mean partial pressure of arterial oxygen (PaO2), carbon dioxide (PaCO2), and pH during baseline and 60 min post-doxapram administration.
| Baseline | 60 min post-injection | |
|---|---|---|
| PaO2 (mmHg) | ||
| Intermittent doxapram | 133 ± 4 | 118 ± 10 |
| Single doxapram 2mg/kg | 142 ± 9 | 138 ± 7 |
| Single doxapram 6mg/kg | 134 ± 10 | 126 ± 9 |
| Vehicle injections | 141 ± 6 | 132 ± 3 |
| Time control | 145 ± 16 | 139 ± 13 |
| PaCO2 (mmHg) | ||
| Intermittent doxapram | 39 ± 2 | 38 ± 2 |
| Single doxapram 2mg/kg | 39 ± 3 | 39 ± 3 |
| Single doxapram 6mg/kg | 41 ± 1 | 42 ± 1 |
| Vehicle injections | 43 ± 3 | 45 ± 3 |
| Time control | 34 ± 4 | 36 ± 4 |
| pH | ||
| Intermittent doxapram | 7.30 ± 0.01 | 7.29 ± 0.01 |
| Single doxapram 2mg/kg | 7.32 ± 0.02 | 7.32 ± 0.02 |
| Single doxapram 6mg/kg | 7.31 ± 0.01 | 7.30 ± 0.02 |
| Vehicle injections | 7.30 ± 0.02 | 7.29 ± 0.02 |
| Time control | 7.34 ± 0.03 | 7.33 ± 0.01 |
Immediate impact of doxapram on phrenic activity
An example neurogram depicting a typical phrenic response to doxapram is provided in Fig. 1. Doxapram evoked an immediate and significant increase in ∫Phr burst amplitude (P < 0.001, Fig. 2) that was sustained for approximately 5 min. Subsequent doxapram injections evoked a progressive augmentation that was characterized by a gradual increase in ∫Phr burst amplitude from the initial to the final injection. Thus, in the intermittent doxapram group, the mean burst amplitude following injections 2 and 3 was significantly greater than following the first injection (Fig. 2, P < 0.005). In animals that received a single dose of doxapram (both 2 mg/kg and 6 mg/kg), ∫Phr amplitude returned to baseline within a few min, and was not different from the time control or vehicle injection groups by 5 min post injection (P > 0.15). Phrenic burst frequency increased in all animals that received doxapram (all P < 0.042 vs. time control), but this response was not different from vehicle injections of low pH saline (P > 0.16). The frequency response was not persistent and values were not different than baseline by 5 min post doxapram or vehicle injection (Fig. 2). In contrast to burst amplitude, the frequency response did not show a progressive augmentation in the intermittent doxapram group (Fig. 2).
Figure 1.
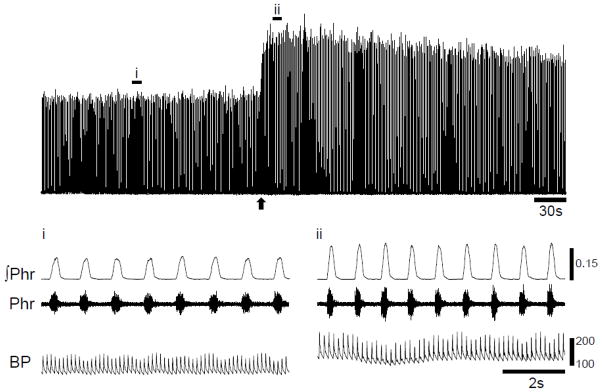
Representative example illustrating the acute impact of intravenously delivered doxapram (2 mg/kg) on phrenic nerve activity. The top panel shows the moving averaged or “integrated” phrenic signal (∫Phr, mV). The bottom panel shows the expanded traces for the time points indicated by (i) and (ii) as wells as the unprocessed or “raw” signal (Phr) and corresponding blood pressure trace (mmHg). Delivery of doxapram (indicated by an arrow) caused an immediate increase in phrenic burst amplitude, frequency and MAP lasting several minutes.
Figure 2.
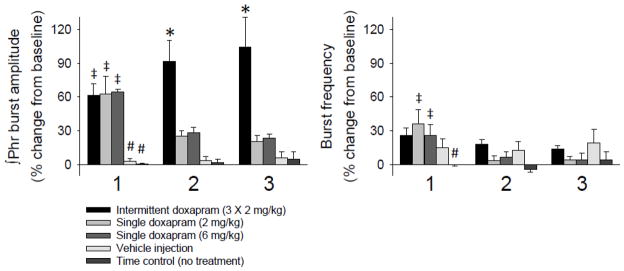
Mean ∫Phr amplitude and burst frequency immediately following doxapram injection. Data are presented from five experimental groups: intermittent doxapram (2 mg/kg), single doxapram (2 or 6 mg/kg), vehicle injection and time control. The plots are labeled “1, 2 or 3” to correspond to the intermittent doxapram injections. In the four groups that did not receive intermittent injections, measures of phrenic activity were taken at a comparable time point. All of the doxapram treated groups showed a similar increase in phrenic output during period 1. A progressive augmentation of ∫Phr amplitude occurred in rats receiving intermittent doxapram injections. *, significantly higher vs. single doxapram (2mg/kg and 6mg/kg), vehicle injections and time control; #, significantly different vs. doxapram injections; ‡, significantly different than injections 2 and 3, or comparable time points.
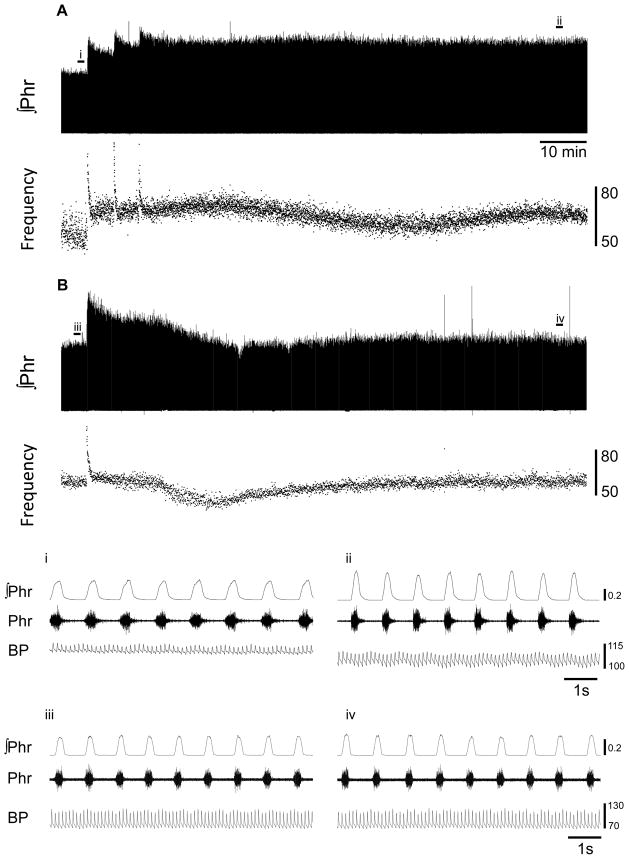
Phrenic motor facilitation following repeated doxapram injections
As shown in Fig. 3, three successive injections of doxapram resulted in a persistent increase in phrenic burst amplitude. The presence of phrenic motor facilitation was determined by comparing ∫Phr burst amplitude (% baseline) in the doxapram groups to the corresponding values obtained in the time control and pH-matched saline groups. An increase in ∫Phr burst amplitude was evident immediately following the final injection of doxapram (e.g., Fig. 3). Thus, during the post-injection period, ∫Phr amplitude in the intermittent doxapram group was significantly elevated in comparison with each of other 4 groups (all P < 0.02, Fig. 4). Neither the two control groups (vehicle and time control) nor the single doxapram injection groups (2 and 6 mg/kg) showed evidence for phrenic motor facilitation (Figs. 3 and 4). In addition, ∫Phr amplitude at the 30 and 60 min time points was similar across the control and single doxapram injection groups (all P > 0.56; Fig. 4). There was no indication of a persistent change in inspiratory phrenic burst frequency (burst*min−1) in any of the doxapram or control groups (Fig. 4).
Figure 3.
Representative examples showing the phrenic motor response to three successive injections of doxapram at 2 mg/kg (A) or a single bolus of doxapram at 6 mg/kg (B). The panels depict the moving averaged or “integrated” phrenic signal (∫Phr, mV) and the breath-by-breath inspiratory burst frequency (breaths per minute). Data are shown during the baseline period, doxapram injections and an approximately 1 hour period following the final injection. The bottom panels show expanded traces for the time points indicated by i - iv. The calibration bars for blood pressure (BP) represent mmHg.
Figure 4.
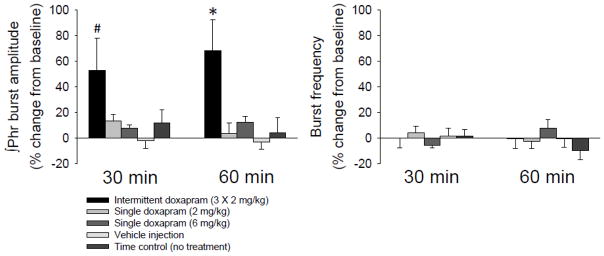
Mean phrenic nerve activity at 30 and 60 min following doxapram. Data are presented from five experimental groups: intermittent doxapram (2 mg/kg), single doxapram (2 or 6 mg/kg), vehicle injection and time control. Mean ∫Phr amplitude and burst frequency are shown relative to baseline (% baseline). #, significantly higher than vehicle injection and time control groups; *, significantly higher than all other groups.
Phrenic responses to doxapram after CSNX
The phrenic response to a range of doxapram doses (2, 4, 8 and 16 mg/kg) was examined in CSNX rats and compared to the response evoked by 2 mg/kg doxapram injections in carotid intact rats (Fig. 5). Following CSNX, the increase in ∫Phr burst amplitude evoked by doxapram at 2 or 4 mg/kg was attenuated by approximately 68% (both P < 0.03 vs. carotid-intact group). The increase in ∫Phr bursting following the 8 and 16 mg/kg doses tended to be reduced, but these responses were not statistically different from the carotid-intact data (Fig. 5; P = 0.06 and 0.11, respectively). The impact of doxapram on phrenic burst frequency was also blunted after CSNX. Thus, the burst frequency (% baseline) following each of the doxapram doses was significantly reduced compared to the carotid intact response (all P < 0.02, Fig. 5).
Figure 5.
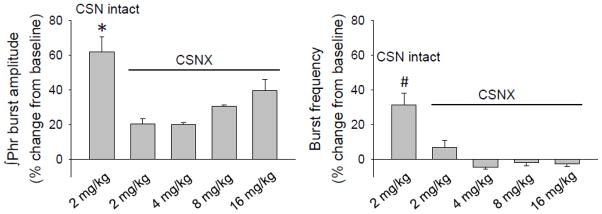
Mean phrenic response to doxapram in animals that received bilateral carotid sinus nerve sectioning (CSNX). ∫Phr amplitude and burst frequency are shown relative to baseline (% baseline). *, significantly higher vs. 2mg/kg and 4mg/kg doxapram doses; #, significantly different vs. all groups with CSNX.
Discussion
Our primary finding is that systemic administration of a low dose of doxapram can induce phrenic motor facilitation. Induction of phrenic neuroplasticity required intermittent doxapram delivery since a single injection only transiently increased phrenic activity while repeated dosing resulted in a long-lasting increase in phrenic burst amplitude. At the doses used to induce facilitation, doxapram appears to act primarily via the carotid chemoreceptors since cutting the carotid sinus nerves almost eliminated phrenic responses. This is the first report that respiratory neuroplasticity can be triggered by this clinically available drug, and these results support further investigation of doxapram as a compliment to respiratory rehabilitation approaches after neurological injury.
Mechanisms by which doxapram stimulates breathing
While the principal site of action of doxapram is not definitively established (Yost, 2006), the majority of published work suggests that carotid body stimulation is a primary mechanism (Hirsh and Wang, 1974, Kato and Buckley, 1964). For example, the phrenic motor response to low doses of doxapram (e.g., 1–2 mg/kg) is quantitatively indistinguishable from the response to mild hypoxia (e.g., PaO2 = 35–40 mmHg) (Mitchell and Herbert, 1975). In addition, doxapram doses of up to 6 mg/kg fail to stimulate ventilation after carotid body denervation (Mitchell and Herbert, 1975, Nishino, et al., 1982) (also see Fig. 5). The mechanisms by which doxapram stimulates the carotid body appear to be similar to the more extensively investigated hypoxic response (Cotten, 2013, Knill and Gelb, 1978). The generally accepted model of acute O2 sensing in the carotid bodies is that glomular cell depolarization triggers an increase in intracellular calcium leading to release of one or more neurotransmitters (Iturriaga, et al., 2007). This depolarization is initiated by hypoxic inhibition of TASK-1 and TASK-3 potassium channel subtypes (Buckler, 1999). Doxapram also inhibits TASK-1 and TASK-3 potassium channel function (Cotten, 2013, Cotten, et al., 2006), and this supports the hypothesis that pharmacological stimulation of the carotid body with doxapram is physiologically similar to stimulation with hypoxia.
The mechanisms by which doxapram stimulates breathing are likely to be dose-dependent. High doses of doxapram, for example, can stimulate breathing via a direct impact on the central nervous system (Hirsh and Wang, 1974, Kato and Buckley, 1964). Hirsh and Wang (1974) demonstrated that the ventilatory response to vertebral artery injections of 0.25 mg/kg doxapram in decerebrate cats was abolished after carotid sinus denervation, but higher doses resulted in increased ventilation despite the carotid denervation. Administration of higher doses (8 and 16 mg/kg) of doxapram in our study also evoked a phrenic motor response in carotid denervated rats. High doses of doxapram (e.g., 10–40 mg/kg) can also activate non-respiratory medullary neurons and/or result in convulsions (Hirsh and Wang, 1974) indicating nonselective CNS excitation.
Although doxapram has not previously been studied in the context of neuroplasticity, prior work has established that stimulation of carotid chemoafferent neurons can initiate respiratory neuroplasticity (Millhorn, et al., 1980). Initial studies from Millhorn and colleagues showed that electrical stimulation of the carotid sinus nerve increases inspiratory phrenic nerve activity for periods extending well beyond the stimulation (Millhorn, et al., 1980). A similar experimental protocol can evoke facilitation of inspiratory intercostal motor activity (Fregosi and Mitchell, 1994). Subsequent work has primarily focused on intermittent hypoxia as a physiological stimulus for eliciting phrenic motor facilitation (Mitchell, et al., 2001), with particular emphasis on central neural mechanisms initiated during hypoxia (Devinney, et al., 2013, Fuller, et al., 2001). However, repetitive hypoxia can also induce plasticity within the carotid bodies. Prabhakar’s group has shown that long term facilitation of carotid chemoafferent discharge can be observed when rats chronically exposed to intermittent hypoxia are exposed to subsequent bouts of hypoxia (Peng, et al., 2003). It should be noted that the carotid bodies are not essential for expression of phrenic LTF since carotid denervated rats also exhibit facilitation after intermittent hypoxia (Bavis and Mitchell, 2003, Sibigtroth and Mitchell, 2011). However, the magnitude of LTF is attenuated after CSNX in rats. Indeed, Bavis and Mitchell reported that LTF expression was blunted by approximately 50% after CSNX.
Based on the available literature and the current results, we hypothesize that doxapram-induced phrenic facilitation is initiated by activation of carotid chemoafferent neurons. Comparison of the doxapram response to the well-established hypoxia-induced LTF model may provide insight regarding the underlying mechanisms. In the case of LTF, the pattern of hypoxia exposure (e.g., continuous vs. intermittent) is a critical component, and an intermittent pattern of stimulation is a requirement (Baker and Mitchell, 2000). In the current work, we found that delivery of 6 mg/kg doxapram as single bolus evoked only a transient increase in phrenic output whereas application as three successive doses at 2 mg/kg produced phrenic motor facilitation. Thus, similar to LTF, the mechanisms underlying doxapram-induced facilitation are sensitive to pattern of respiratory stimulation. However, differences in the typical onset pattern of intermittent hypoxia-induced LTF versus doxapram-induced facilitation suggest that the underlying molecular mechanisms may be different. When studied in anesthetized rats, hypoxia-induced LTF typically proceeds in a ramp like fashion with progressive increases in phrenic activity occurring after intermittent hypoxia (Fuller, et al., 2001). In contrast, doxapram-induced phrenic facilitation was not typically characterized by progressive increases in motor output, but rather by an immediate and persistent response (e.g., Figs. 3 and 4). Alternatively, it is possible that intermittent hypoxia activates inhibitory mechanisms that prevent phrenic LTF from occurring immediately post-hypoxia (Baker, et al., 2001), and that doxapram does not exert similar inhibitory influences. Interestingly, a pattern of phrenic motor facilitation that is similar to the doxapram-induced response (e.g., Fig. 3) was previously reported by Morris, et al. (2001). These authors showed that repeated chemoreceptor stimulation by arterial injections of CO2-saturated saline induced an immediate increase in phrenic motor output that was sustained for at least 15 min.
The LTF model proposed by Mitchell and colleagues postulates that hypoxia leads to activation of raphe-spinal neurons which release serotonin on or near spinal phrenic motoneurons (Devinney, et al., 2013). In turn, activation of spinal serotonin receptors during, but not following hypoxia leads to synthesis of BDNF and increased phrenic motor output (Fuller, et al., 2001). Whether doxapram can initiate similar mechanisms is not clear, but we suggest that de novo synthesis of proteins (e.g., BDNF) is not likely to be a prominent component since the onset occurs more quickly than LTF. Doxapram has an alpha half-life (i.e., plasma distribution) of 6 min and a beta half-life (i.e., tissue elimination) of 62 min, and the respiratory response to a single dose of intravenous doxapram at 1.5 mg/kg lasts 8–10 min in humans (Clements, et al., 1979, Stephen and Talton, 1964). In the current studies, the acute phrenic motor response to doxapram had a similar time course (e.g., Figs. 3 and 4). While we cannot rule out the possibility that the persistent increase in phrenic output (e.g., >30 min following intermittent doxapram) reflected on ongoing doxapram-induced stimulation of respiratory neurons, we do not favor this hypothesis since a single high dose of doxapram with the same cumulative dose as the intermittent paradigm did not trigger phrenic motor facilitation. Rather, we favor the hypothesis that the observed phrenic motor facilitation resulted from neuroplastic changes in the respiratory motor circuit and not a cumulative or ongoing impact of doxapram. Future work is needed to confirm this hypothesis, and to determine the most relevant site of plasticity (e.g., peripheral chemoreceptors, brainstem, spinal cord, etc.) as well as the underlying molecular mechanisms by which doxapram can lead to long-lasting phrenic motor facilitation.
Potential clinical significance
Targeted induction of neuroplasticity in central respiratory circuitry may be beneficial for treating respiratory insufficiency after neurologic injury or in cases of neurodegeneration (Dale-Nagle, et al., 2010, Nichols, et al., 2013). Intermittent-hypoxia induced phrenic LTF has been the premier experimental model in this regard (Vinit, et al., 2009). For example, intermittent-hypoxia can induce persistent increases in phrenic motor output when applied to rats with chronic spinal cord injury (Doperalski and Fuller, 2006, Golder and Mitchell, 2005) and also in rats in advanced stages of phrenic neurodegeneration (Nichols, et al., 2013). Improvements in somatic motor function have also been reported following daily acute intermittent-hypoxia in spinal cord injured rats (Lovett-Barr, et al., 2012). The success of intermittent hypoxia “therapy” in rodent models has lead several groups to begin translating this approach to human clinical applications. Mateika and colleagues have established that ventilatory LTF can be triggered by intermittent hypoxia in awake human subjects (Harris, et al., 2006). In addition, our group recently built upon this work by showing that LTF of ventilation can be induced by repeated bouts of hypoxia in humans with chronic cervical spinal cord injury (Tester, et al., 2011). Finally, intermittent hypoxia can also enhance somatic motor output in persons with chronic spinal cord injuries, highlighting the role of hypoxia in non-respiratory motor plasticity (Trumbower, 2009). Thus, hypoxia clearly represents a useful tool for understanding the mechanisms associated with respiratory motor plasticity, and also is likely to have clinical applications in the context of rehabilitation (Dale-Nagle, et al., 2010). However, hypoxia is not likely to be indicated in all clinical cases, and pharmacological treatments that can induce respiratory neuroplasticity may prove useful either alone or in conjunction with conventional respiratory therapy. To date, comparatively few studies have investigated the use of pharmacological methods for inducing respiratory plasticity (Nantwi, 2009, Nelson, et al., 2011). Goshgarian and colleagues showed that theophylline can enhance phrenic output in a rodent model of spinal cord injury (Nantwi and Goshgarian, 2005), but reported mixed results when the drug was applied to spinally injured humans (Bascom, et al., 2005). In addition, recent work by Dale and colleagues showed that cervical spinal delivery of vascular endothelial growth factor and erythropoietin can also elicit long lasting phrenic motor plasticity (Dale-Nagle, et al., 2011, Dale, et al., 2012). Our study establishes a proof-of-principle that low-dose intravenous doxapram is capable of inducing a substantial respiratory facilitation when applied in an appropriate pattern. Doxapram is an attractive drug in this regard as it is already FDA approved for clinical use in conditions such as acute respiratory failure, post-anesthetic respiratory depression and premature apnea (Moser, et al., 1973, Rappolt, et al., 1980, Winnie and Collins, 1966). The robust respiratory facilitation that was triggered by repeated administration of low-dose doxapram in our study suggests that clinicians could potentially achieve longer-lasting respiratory effects by using a lower drug dose that is administered in an intermittent pattern. However, further work is required to establish the appropriate dosage, and delivery protocols in awake, spontaneously breathing animals and ultimately humans with ventilatory compromise.
Highlights.
A single doxapram bolus transiently increased phrenic nerve activity
Repeated intravenous doxapram persistently increased phrenic nerve activity
Phrenic responses to doxapram were attenuated after carotid denervation
Intermittent application of doxapram can trigger phrenic neuroplasticity
Acknowledgments
This work was supported by the Neilsen foundation grant #220521 (MSS) and a National Institutes of Health Grant to DDF (1R01NS080180-01A1)
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Bach KB, Mitchell GS. Hypoxia-induced long-term facilitation of respiratory activity is serotonin dependent. Respir Physiol. 1996;104:251–260. doi: 10.1016/0034-5687(96)00017-5. [DOI] [PubMed] [Google Scholar]
- 2.Baker TL, Fuller DD, Zabka AG, Mitchell GS. Respiratory plasticity: differential actions of continuous and episodic hypoxia and hypercapnia. Respir Physiol. 2001;129:25–35. doi: 10.1016/s0034-5687(01)00280-8. [DOI] [PubMed] [Google Scholar]
- 3.Baker TL, Mitchell GS. Episodic but not continuous hypoxia elicits long-term facilitation of phrenic motor output in rats. J Physiol. 2000;529(Pt 1):215–219. doi: 10.1111/j.1469-7793.2000.00215.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bascom AT, Lattin CD, Aboussouan LS, Goshgarian HG. Effect of acute aminophylline administration on diaphragm function in high cervical tetraplegia: a case report. Chest. 2005;127:658–661. doi: 10.1378/chest.127.2.658. [DOI] [PubMed] [Google Scholar]
- 5.Bavis RW, Mitchell GS. Intermittent hypoxia induces phrenic long-term facilitation in carotid-denervated rats. J Appl Physiol. 2003;94:399–409. doi: 10.1152/japplphysiol.00374.2002. [DOI] [PubMed] [Google Scholar]
- 6.Bowdle TA. Clinical pharmacology of antagonists of narcotic-induced respiratory depression. A brief review. Acute Care. 1988;12(Suppl 1):70–76. [PubMed] [Google Scholar]
- 7.Buckler KJ. Background leak K+-currents and oxygen sensing in carotid body type 1 cells. Respir Physiol. 1999;115:179–187. doi: 10.1016/s0034-5687(99)00015-8. [DOI] [PubMed] [Google Scholar]
- 8.Burki NK. Ventilatory effects of doxapram in conscious human subjects. Chest. 1984;85:600–604. doi: 10.1378/chest.85.5.600. [DOI] [PubMed] [Google Scholar]
- 9.Clements JA, Robson RH, Prescott LF. The disposition of intravenous doxapram in man. Eur J Clin Pharmacol. 1979;16:411–416. doi: 10.1007/BF00568202. [DOI] [PubMed] [Google Scholar]
- 10.Cotten JF. TASK-1 (KCNK3) and TASK-3 (KCNK9) Tandem Pore Potassium Channel Antagonists Stimulate Breathing in Isoflurane-Anesthetized Rats. Anesth Analg. 2013;116:810–816. doi: 10.1213/ANE.0b013e318284469d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cotten JF, Keshavaprasad B, Laster MJ, Eger EI, 2nd, Yost CS. The ventilatory stimulant doxapram inhibits TASK tandem pore (K2P) potassium channel function but does not affect minimum alveolar anesthetic concentration. Anesth Analg. 2006;102:779–785. doi: 10.1213/01.ane.0000194289.34345.63. [DOI] [PubMed] [Google Scholar]
- 12.Dale-Nagle EA, Hoffman MS, MacFarlane PM, Mitchell GS. Multiple pathways to long-lasting phrenic motor facilitation. Adv Exp Med Biol. 2010;669:225–230. doi: 10.1007/978-1-4419-5692-7_45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dale-Nagle EA, Hoffman MS, MacFarlane PM, Satriotomo I, Lovett-Barr MR, Vinit S, Mitchell GS. Spinal plasticity following intermittent hypoxia: implications for spinal injury. Ann N Y Acad Sci. 2010;1198:252–259. doi: 10.1111/j.1749-6632.2010.05499.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dale-Nagle EA, Satriotomo I, Mitchell GS. Spinal vascular endothelial growth factor induces phrenic motor facilitation via extracellular signal-regulated kinase and Akt signaling. J Neurosci. 2011;31:7682–7690. doi: 10.1523/JNEUROSCI.0239-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dale EA, Satriotomo I, Mitchell GS. Cervical spinal erythropoietin induces phrenic motor facilitation via extracellular signal-regulated protein kinase and Akt signaling. J Neurosci. 2012;32:5973–5983. doi: 10.1523/JNEUROSCI.3873-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Devinney MJ, Huxtable AG, Nichols NL, Mitchell GS. Hypoxia-induced phrenic long-term facilitation: emergent properties. Ann N Y Acad Sci. 2013;1279:143–153. doi: 10.1111/nyas.12085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Doperalski NJ, Fuller DD. Long-term facilitation of ipsilateral but not contralateral phrenic output after cervical spinal cord hemisection. Exp Neurol. 2006;200:74–81. doi: 10.1016/j.expneurol.2006.01.035. [DOI] [PubMed] [Google Scholar]
- 18.Fregosi RF, Mitchell GS. Long-term facilitation of inspiratory intercostal nerve activity following carotid sinus nerve stimulation in cats. J Physiol. 1994;477 (Pt 3):469–479. doi: 10.1113/jphysiol.1994.sp020208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fuller DD, Zabka AG, Baker TL, Mitchell GS. Phrenic long-term facilitation requires 5-HT receptor activation during but not following episodic hypoxia. J Appl Physiol. 2001;90:2001–2006. doi: 10.1152/jappl.2001.90.5.2001. discussion 2000. [DOI] [PubMed] [Google Scholar]
- 20.Funderburk WH, Oliver KL, Ward JW. Electrophysiologic analysis of the site of action of doxapram hydrochloride. J Pharmacol Exp Ther. 1966;151:360–368. [PubMed] [Google Scholar]
- 21.Golder FJ, Mitchell GS. Spinal synaptic enhancement with acute intermittent hypoxia improves respiratory function after chronic cervical spinal cord injury. J Neurosci. 2005;25:2925–2932. doi: 10.1523/JNEUROSCI.0148-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Harris DP, Balasubramaniam A, Badr MS, Mateika JH. Long-term facilitation of ventilation and genioglossus muscle activity is evident in the presence of elevated levels of carbon dioxide in awake humans. Am J Physiol Regul Integr Comp Physiol. 2006;291:R1111–1119. doi: 10.1152/ajpregu.00896.2005. [DOI] [PubMed] [Google Scholar]
- 23.Hayashi F, Coles SK, Bach KB, Mitchell GS, McCrimmon DR. Time-dependent phrenic nerve responses to carotid afferent activation: intact vs. decerebellate rats. Am J Physiol. 1993;265:R811–819. doi: 10.1152/ajpregu.1993.265.4.R811. [DOI] [PubMed] [Google Scholar]
- 24.Hirsh K, Wang SC. Selective respiratory stimulating action of doxapram compared to pentylenetetrazaol. J Pharmacol Exp Ther. 1974;189:1–11. [PubMed] [Google Scholar]
- 25.Iturriaga R, Varas R, Alcayaga J. Electrical and pharmacological properties of petrosal ganglion neurons that innervate the carotid body. Respir Physiol Neurobiol. 2007;157:130–139. doi: 10.1016/j.resp.2006.12.006. [DOI] [PubMed] [Google Scholar]
- 26.Kato H, Buckley JP. Possible Sites of Action of the Respiratory Stimulant Effect of Doxapram Hydrochloride. J Pharmacol Exp Ther. 1964;144:260–264. [PubMed] [Google Scholar]
- 27.Knill RL, Gelb AW. Ventilatory responses to hypoxia and hypercapnia during halothane sedation and anesthesia in man. Anesthesiology. 1978;49:244–251. doi: 10.1097/00000542-197810000-00004. [DOI] [PubMed] [Google Scholar]
- 28.Lee KZ, Sandhu MS, Dougherty BJ, Reier PJ, Fuller DD. Influence of vagal afferents on supraspinal and spinal respiratory activity following cervical spinal cord injury in rats. J Appl Physiol. 2010 doi: 10.1152/japplphysiol.01429.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lovett-Barr MR, Satriotomo I, Muir GD, Wilkerson JE, Hoffman MS, Vinit S, Mitchell GS. Repetitive intermittent hypoxia induces respiratory and somatic motor recovery after chronic cervical spinal injury. J Neurosci. 2012;32:3591–3600. doi: 10.1523/JNEUROSCI.2908-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.MacFarlane PM, Satriotomo I, Windelborn JA, Mitchell GS. NADPH oxidase activity is necessary for acute intermittent hypoxia-induced phrenic long-term facilitation. J Physiol. 2009;587:1931–1942. doi: 10.1113/jphysiol.2008.165597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mateika JH, Sandhu KS. Experimental protocols and preparations to study respiratory long term facilitation. Respir Physiol Neurobiol. 2011;176:1–11. doi: 10.1016/j.resp.2011.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Millhorn DE, Eldridge FL, Waldrop TG. Prolonged stimulation of respiration by a new central neural mechanism. Respir Physiol. 1980;41:87–103. doi: 10.1016/0034-5687(80)90025-0. [DOI] [PubMed] [Google Scholar]
- 33.Mitchell GS. Respiratory plasticity following intermittent hypoxia: a guide for novel therapeutic approaches to ventilatory control disorders. In: Gaultier C, editor. Genetic Basis for Respiratory Control Disorders. Springer Publishing Company; New York: 2007. [Google Scholar]
- 34.Mitchell GS, Baker TL, Nanda SA, Fuller DD, Zabka AG, Hodgeman BA, Bavis RW, Mack KJ, Olson EB., Jr Invited review: Intermittent hypoxia and respiratory plasticity. J Appl Physiol. 2001;90:2466–2475. doi: 10.1152/jappl.2001.90.6.2466. [DOI] [PubMed] [Google Scholar]
- 35.Mitchell RA, Herbert DA. Potencies of doxapram and hypoxia in stimulating carotid-body chemoreceptors and ventilation in anesthetized cats. Anesthesiology. 1975;42:559–566. doi: 10.1097/00000542-197505000-00009. [DOI] [PubMed] [Google Scholar]
- 36.Morris KF, Shannon R, Lindsey BG. Changes in cat medullary neurone firing rates and synchrony following induction of respiratory long-term facilitation. J Physiol. 2001;532:483–497. doi: 10.1111/j.1469-7793.2001.0483f.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Moser KM, Luchsinger PC, Adamson JS, McMahon SM, Schlueter DP, Spivack M, Weg JG. Respiratory stimulation with intravenous doxapram in respiratory failure. A double-blind co-operative study. N Engl J Med. 1973;288:427–431. doi: 10.1056/NEJM197303012880901. [DOI] [PubMed] [Google Scholar]
- 38.Nantwi KD. Recovery of respiratory activity after C2 hemisection (C2HS): involvement of adenosinergic mechanisms. Respir Physiol Neurobiol. 2009;169:102–114. doi: 10.1016/j.resp.2009.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nantwi KD, Basura GJ, Goshgarian HG. Effects of long-term theophylline exposure on recovery of respiratory function and expression of adenosine A1 mRNA in cervical spinal cord hemisected adult rats. Exp Neurol. 2003;182:232–239. doi: 10.1016/s0014-4886(03)00109-2. [DOI] [PubMed] [Google Scholar]
- 40.Nantwi KD, Goshgarian HG. Adenosinergic mechanisms underlying recovery of diaphragm motor function following upper cervical spinal cord injury: potential therapeutic implications. Neurol Res. 2005;27:195–205. doi: 10.1179/016164105X21977. [DOI] [PubMed] [Google Scholar]
- 41.Nelson NR, Bird IM, Behan M. Testosterone restores respiratory long term facilitation in old male rats by an aromatase-dependent mechanism. J Physiol. 2011;589:409–421. doi: 10.1113/jphysiol.2010.198200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Nichols NL, Gowing G, Satriotomo I, Nashold LJ, Dale EA, Suzuki M, Avalos P, Mulcrone PL, McHugh J, Svendsen CN, Mitchell GS. Intermittent hypoxia and stem cell implants preserve breathing capacity in a rodent model of amyotrophic lateral sclerosis. Am J Respir Crit Care Med. 2013;187:535–542. doi: 10.1164/rccm.201206-1072OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nishino T, Mokashi A, Lahiri S. Stimulation of carotid chemoreceptors and ventilation by doxapram in the cat. J Appl Physiol. 1982;52:1261–1265. doi: 10.1152/jappl.1982.52.5.1261. [DOI] [PubMed] [Google Scholar]
- 44.Peng YJ, Overholt JL, Kline D, Kumar GK, Prabhakar NR. Induction of sensory long-term facilitation in the carotid body by intermittent hypoxia: implications for recurrent apneas. Proc Natl Acad Sci U S A. 2003;100:10073–10078. doi: 10.1073/pnas.1734109100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Peng YJ, Prabhakar NR. Reactive oxygen species in the plasticity of respiratory behavior elicited by chronic intermittent hypoxia. J Appl Physiol. 2003;94:2342–2349. doi: 10.1152/japplphysiol.00613.2002. [DOI] [PubMed] [Google Scholar]
- 46.Pleuvry BJ. A study of the enhanced toxicity of doxapram in rodents treated with narcotic analgesics. Br J Anaesth. 1978;50:451–458. doi: 10.1093/bja/50.5.451. [DOI] [PubMed] [Google Scholar]
- 47.Rappolt RT, Sr, Gay GR, Decker WJ, Inaba DS. NAGD regimen for the coma of drug-related overdose. Ann Emerg Med. 1980;9:357–363. doi: 10.1016/s0196-0644(80)80112-0. [DOI] [PubMed] [Google Scholar]
- 48.Sandhu MS, Lee KZ, Fregosi RF, Fuller DD. Phrenicotomy alters phrenic long term facilitation (LTF) following intermittent hypoxia in anesthetized rats. J Appl Physiol. 2010 doi: 10.1152/japplphysiol.01422.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sibigtroth CM, Mitchell GS. Carotid chemoafferent activity is not necessary for all phrenic long-term facilitation following acute intermittent hypoxia. Respir Physiol Neurobiol. 2011;176:73–79. doi: 10.1016/j.resp.2010.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Stephen CR, Talton I. Investigation of Doxapram as a Postanesthetic Respiratory Stimulant. Anesth Analg. 1964;43:628–640. [PubMed] [Google Scholar]
- 51.Tester NJ, Fuller DD, Behrman AL, Burtis DB, Fromm JS, Mateika JH. Acute intermittent hypoxia induces respiratory long term facilitation post-motor incomplete cervical spinal cord injury in the human. Program No. 808.09. 2011 Neuroscience Meeting Planner; Washington, DC: Society for Neuroscience; 2011. 2011. Online. [Google Scholar]
- 52.Trumbower RD, Jayaraman A, Mitchell GS, Rymer WZ. Exposure to Acute Intermittent Hypoxia Augments Somatic Motor Function in Humans With Incomplete Spinal Cord Injury. Neurorehabil Neural Repair. 2011 doi: 10.1177/1545968311412055. [DOI] [PubMed] [Google Scholar]
- 53.Trumbower RD, Jayaraman A, Schmit BD, Hornby TG, Mitchell GS, Rymer WZ. 2009 Neuroscience Meeting Planner. Chicago, IL: Society for Neuroscience; 2009. Effects of repetitive acute intermittent hypoxia on lumbosacral motor function in human SCI. 2009. Online. [Google Scholar]
- 54.Vinit S, Lovett-Barr MR, Mitchell GS. Intermittent hypoxia induces functional recovery following cervical spinal injury. Respir Physiol Neurobiol. 2009;169:210–217. doi: 10.1016/j.resp.2009.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Winnie AP, Collins VJ. The search for a pharmacologic ventilator. Acta Anaesthesiol Scand Suppl. 1966;23:63–71. doi: 10.1111/j.1399-6576.1966.tb00993.x. [DOI] [PubMed] [Google Scholar]
- 56.Yost CS. A new look at the respiratory stimulant doxapram. CNS Drug Rev. 2006;12:236–249. doi: 10.1111/j.1527-3458.2006.00236.x. [DOI] [PMC free article] [PubMed] [Google Scholar]