Abstract
Objective
To assess the association between frailty and risk for heart failure (HF) in older adults.
Background
Frailty is common in the elderly and is associated with adverse health outcomes. Impact of frailty on HF risk is not known.
Methods
We assessed the association between frailty, using the Health ABC Short Physical Performance Battery (HABC battery) and the Gill index, and incident HF in 2825 participants aged 70-79 years.
Results
Mean age of participants was 74±3years; 48% were men and 59% were white. During a median follow up of 11.4 (7.1-11.7) years, 466 participants developed HF. Compared to non-frail participants, moderate (hazard ratio HR 1.36, 95%CI 1.08-1.71) and severe frailty (HR 1.88, 95%CI 1.02-3.47) by Gill index was associated with a higher risk for HF. HABC battery score was linearly associated with HF risk after adjusting for the Health ABC HF Model (HR 1.24, 95%CI 1.13, 1.36 per SD decrease in score), and remained significant when controlled for death as a competing risk (HR 1.30; 95%CI 1.00-1.55). Results were comparable across age, sex, and race, and in sub-groups based on diabetes mellitus or cardiovascular disease at baseline. Addition of HABC battery scores to the Health ABC HF Risk Model improved discrimination (change in C-index, 0.014; 95%CI 0.018-0.010) and appropriately reclassified 13.4% (NRI 0.073, 95%CI 0.021-0.125; P=0.006) of participants (8.3% who developed HF and 5.1% who did not).
Conclusions
Frailty is independently associated with risk of HF in older adults.
Keywords: Frailty, Heart Failure, Elderly
Frailty in older adults is associated with poor physical functioning and outcomes (1,2). Several studies have suggested an association between frailty and cardiovascular disease (CVD), with frailty identified in 25-50% of all patients with CVD (3,4). The population prevalence of frailty in the community is 3-7% among individuals aged 65-75 years, 20-26% in octogenarians, and a third in nonagenarians(5). Observational studies have shown a strong association between frailty with incident CVD and mortality in elderly (6-8). The importance to better understand frailty in context of CVD in elderly was recently emphasized (9). Heart failure (HF) prevalence is highest in the elderly but there are limited data on frailty and HF risk. Several mechanistic pathways including inflammation and metabolic disturbances underlie frailty and may be related to its association with HF; however limited data exist in this regard (2,10-11).
The study of frailty is complicated by the lack of a consensus definition and the multiplicity of scales used to define frailty. Physical performance measures have been used to assess functional status.(12) Gill index categorizes individuals into non-frail, or moderately or severely frail, based on a combination of chair-stand and walking speed tests(13). Studies have shown that objective measures of lower extremity function are highly predictive of subsequent disability and hospitalizations(14-16). The Health Aging and Body Composition (Health ABC) Short Physical Performance Battery (HABC Battery) evaluates physical performance via a composite of gait speed, repeated chair stands, and tandem balance tests(17) and helps to identify individuals with preclinical disability. The objective of this study is to assess the association of frailty with incident HF in the Health ABC Study using these frailty measures.
METHODS
Study Population
The Health ABC Study enrolled 3075 well-functioning, community dwelling individuals aged 70 to 79 years from 1997-1998. Participants were identified from a random sample of white Medicare beneficiaries and all age eligible black community residents from Pittsburgh and Memphis. Participants with manifest or missing data on HF at baseline (n=140) were excluded. Of the 2935 such participants, 2825 (96%) had data for the Gill criteria and HABC battery, and were included in this analysis.
Study Definitions
Diabetes was defined based on positive history or use of anti-hyperglycemic medication. Smoking was defined as current, past (≥100 lifetime cigarettes), or never. Physical activity was ascertained using a standardized questionnaire designed by the Health ABC study. Kilocalories per week (kcal/week) expended in common exercise (e.g., walking for exercise, exercise classes, weightlifting) and lifestyle activities (e.g., gardening, housework, yard work, non-exercise walking) were collected. From the reported duration, intensity, and estimated metabolic cost (18), a summary variable of kcal/week was calculated for each activity performed. Left ventricular hypertrophy was defined based on electrocardiogram using the Minnesota voltage criteria. Hypertension was defined as history of physician diagnosis accompanied by use of antihypertensive drugs. Coronary heart disease (CHD) was defined as history of revascularization or electrocardiographic evidence of myocardial infarction or self-reported history of myocardial infarction or angina accompanied by use of anti-anginal medications. Cerebrovascular disease was defined as self-reported history of stroke, transient ischemic attack, or carotid endarterectomy. Incident CHD events were defined as hospitalization for myocardial infarction, angina pectoris, or elective coronary revascularization, either surgical or percutaneous.
Frailty Assessment
The Gill index is based on a composite of chair-stand and walking speed tests (13). Severe frailty is defined when the subject is unable to stand-up from the chair without the use of the arms and showed a walking speed lower than 0.6 m/s; moderate frailty was defined as only one of the two tests being abnormal; and non-frailty if neither were present. Frailty was also assessed by the Health ABC physical performance battery (17). Briefly, the scale is an extension of the lower extremity performance tests used in the Epidemiologic Studies of the Elderly (19) and includes , five repeated chair stands, standing balance (semi-and full-tandem stands and a single leg stand for 30 second), a 6-m walk to assess usual gait speed, and a narrow 6-m walk between lines 20 cm apart.. A ratio score from 0 to 1 was created for each test and the four tests were added to provide a continuous scale from 0 to 4. The scoring system for the HABC battery was developed to minimize ceiling effects (17). Lower levels of Health ABC battery score reflect more severe frailty.
Serum Biomarkers
Blood samples were obtained after overnight fasting. Cytokines were measured in duplicate by ELISA. The detectable limit for interleukin-6 (IL-6) was 0.10 pg/mL and for tumor necrosis factor-alpha (TNF-α) 0.18 pg/mL. C reactive protein (CRP) was measured in by ELISA with a sensitivity of 0.08 mg/L. Blind duplicate analyses (n=150) for IL-6, CRP, and TNF-α showed inter-assay coefficients of variation of 10.3%, 8.0%, and 15.8%, respectively. Serum resistin was measured using sandwich ELISA; intra- and inter-assay coefficients of variation for this assay are 4.5% and 7.4%, respectively.14
Study Outcome
All participants were asked to report any hospitalizations. Every 6 months, they were asked direct questions about interim events. Medical records for hospitalizations were reviewed. All first admissions confirmed to be related to HF were classified as incident HF. The Events Committee adjudicated all HF diagnoses. The adjudicators reviewed all pertinent data, including history, physical examination, chest radiography, and medication use. HF event was confirmed if in addition to a physician diagnosis, there was (1) documentation of HF symptoms and signs; (2) supporting findings (e.g. pulmonary edema on X-ray); or (3) HF treatment, including diuretics, digitalis, ACE inhibitors, or β-blockers were given (20-21).
Statistical Analysis
Cross-sectional associations of frailty with various risk factors were assessed using linear regression models adjusted for age and gender. Time-to-event analyses were conducted using Cox proportional hazard models, stratified by baseline CHD. The proportional hazards assumption was evaluated by the Schoenfeld residuals. The shape of association with HF risk was assessed by plotting hazard ratios (HR) calculated within quintile of baseline HABC battery scores against the mean score within each quintile. Floating variances were used to calculate 95% confidence intervals (CI) for the HR in each group, including the reference group, to allow for comparisons across the groups irrespective of the arbitrarily chosen reference category, i.e. bottom fifth (22). As the association showed an approximately linear shape, HRs were calculated per SD change in HABC battery, progressively adjusted for independent clinical predictors of incident HF as identified in the Health ABC HF Risk Score (including age, smoking, systolic blood pressure, heart rate, fasting glucose, left ventricular hypertrophy, serum albumin and creatinine) (23). The HR was further adjusted for physical activity, inflammatory biomarkers, and time-varying incident CHD evaluated at 1-year intervals in nested models. These analyses were repeated in subgroups defined by CVD status at baseline. Finally, analyses were repeated with death as a competing event, using the competing risks model (proportional sub-hazards model) proposed by Fine and Gray(24). Appropriate interaction terms were introduced to examine for differences in HRs across levels of pre-specified characteristics (age, sex, race, history of diabetes, CHD, and presence of left ventricular hypertrophy). In parallel analyses we also assessed the risk of incident HF across categories of frailty as defined by the Gill index. The discriminative properties of the HABC battery score versus Gill index for HF prediction were compared using Harell’s C-index. To assess the incremental value of frailty in assessment of HF risk prediction, we calculated C-index for two risk prediction models, one with the conventional risk predictors using the Health ABC Risk Score as a base model, and the second with this model plus HABC battery score.
For reclassification, 5-year risk prediction was considered relevant. Individuals whose HF events occurred after 5 years were categorized as non-cases. The reference risk prediction model included HABC HF Risk Model, which was then extended to additionally include HABC battery score as the alternative model. Individuals were classified into <5%, 5-10%, and >10% 5-year risk categories. Reclassification was summarized using net-reclassification-improvement [NRI] (25). In addition, we also employed cross-validation approaches to these analyses (e.g. performing model derivation and validation in separate subsets of participants) to investigate optimism in estimates of predictive ability. For multivariable models, missing values of covariates were imputed using multiple imputation by chained equations as introduced by van Buuren et al(26),(27). Parameter estimates and confidence intervals (CI) were obtained by combining 5 imputed datasets using the method described by Barnard and Rubin to account for possible error in missing value analysis(28). A 2-sided P<0.05 was accepted as statistically significant for all analyses. All analyses were performed using Stata release 11 (Stata Corp LP, College Station, Texas).
The Health ABC study was supported in part by the Intramural Research Program of the National Institutes of Health, National Institute on Aging (NIA), and contracts, N 01-AG-6-2101; N01-AG-6-2103; N01-AG-6-2106; NIA grant: R01-AG028050, and NINR grant R01-NR012459. The authors are solely responsible for the design and conduct of this study, all study analyses, the drafting and editing of the paper and its final contents
RESULTS
Baseline Participant Characteristics
The mean age of participants was 74±3 year; 48% were men and 59% were white. Median HABC battery score was 2.3 (interquartile range [IQR] 1.9, 2.6). Table 1 presents the baseline characteristics. Frailty was present in 17.5% (moderate 16.2% and severe 1.3%) of participants by Gill criteria. Frail participants were older (74±3 vs. 73±3 years, P<0.001), more frequently women (60.1% vs. 50.4%, P<0.001), and black (53% vs. 38%, P<0.001). Frailty status by the two indexes was concordant in 1882 (66.7%) participants. Age, measures of adiposity (body mass index and waist to thigh ratio) and inflammation (tumor necrosis factor-α, interleukin-6), systolic blood pressure, heart rate, baseline diabetes mellitus and CVD, and fasting glucose, serum resistin and creatinine levels, all correlated with HABC battery score, Table 2.
Table 1. Baseline Characteristics of Health ABC Participants.
| HABC Battery score |
||||
|---|---|---|---|---|
| Characteristic | Overall N=2825 |
Non Frail N=1400 |
Frail N=1425 |
P value |
| Age (years) | 73.6 (2.9) | 74.0(2.9) | 73.2(2.8) | <0.001 |
| Female (%) | 52.1 | 64.2 | 39.8 | <0.001 |
| Black race, % | 40.6 | 52.4 | 28.7 | <0.001 |
| Body mass index, kg/m2 | 27.3(4.7) | 28.1(5.2) | 26.4(4.0) | <0.001 |
| Waist/thigh circumference | 1.9(0.2) | 1.9(0.2) | 1.9(0.2) | 0.811 |
| Smoking, % | <0.001 | |||
| Current | 45.3 | 44.7 | 48.6 | |
| Past | 10.4 | 13.4 | 7.4 | |
| Never | 44.3 | 42.0 | 44.0 | |
| Alcohol consumption, % | <0.001 | |||
| Never | 49.9 | 56.9 | 42.8 | |
| <1 drink/week | 20.8 | 19.0 | 22.7 | |
| 1–7 drinks/week | 21.8 | 18.6 | 25.0 | |
| >7 drinks/week | 7.5 | 5.5 | 9.5 | |
| Diabetes mellitus, % | 14.3 | 17.2 | 11.4 | <0.001 |
| Hypertension, % | 50.0 | 56.5 | 43.4 | <0.001 |
| Coronary heart disease, % | 19.5 | 20.2 | 18.9 | 0.318 |
| Peripheral arterial disease, % | 4.7 | 5.5 | 3.9 | 0.041 |
| Left ventricular hypertrophy, % | 11.5 | 13.5 | 9.5 | 0.001 |
| Physical activity, kcal/kg/week | 83.9(69.2) | 76.1(66.9) | 91.9(70.5) | <0.001 |
| Systolic blood pressure, mm Hg | 135.9(20.7) | 136.9(21.6) | 134.8(19.7) | 0.007 |
| Diastolic blood pressure, mm Hg | 71.4(11.7) | 71.0(12.0) | 71.8(11.4) | 0.047 |
| Heart rate, beats/min | 65.2(11.0) | 66.5(11.3)) | 91.9(70.5) | <0.001 |
| Fasting glucose, mg/dl* | 94.0(87.0,105.0) | 95.0(87.0,107.0) | 93.0(87.0,103.0) | 0.001 |
| Albumin, g/dl | 4.0(0.3) | 4.0(0.3) | 71.8(11.4) | <0.001 |
| Creatinine, mg/dl* | 1.1(0.4) | 1.1(0.5) | 1.0(0.2) | 0.087 |
| Total cholesterol, mg/dl | 203.3(38.3) | 206.0(39.6) | 200.5(36.7) | <0.001 |
| Low-density lipoprotein, mg/dl | 121.9(34.6) | 122.7(36.2) | 121.2(33.0) | 0.258 |
| High-density lipoprotein, mg/dl | 54.3(16.9) | 56.3(17.7) | 52.2(15.9) | <0.001 |
| Triglycerides, mg/dl* | 118.0(89.0,163.0)) | 117.0(89.0,163.0) | 119.0(88.0,163.0) | 0.615 |
| Resistin, mg/ml | 20.4(10.9) | 21.4(11.6) | 19.3(10.1) | <0.001 |
| TNF alpha, pg/ml* | 3.5(1.7) | 3.6(1.8) | 3.4(1.6) | 0.002 |
| Interleukin-6, pg/ml* | 2.3(1.9) | 2.5(2.0) | 2.2(1.8) | <0.001 |
| Ankle arm index | 1.1(0.2) | 1.0(0.2) | 1.1(0.2) | <0.001 |
HABC battery score, median = 2.3, was used to categorize participants as being frail (<2.3) and non-frail (≥23).
Table 2. Correlates of HABC battery score.
| Characteristic | r (95% CI)tt | P-value |
|---|---|---|
| Categorical variable | Mean difference (95% CI) compared to ref ‡ | |
|
| ||
| Male | ref | |
| Female | −0.29 (−0.33 to −0.26) | <0.001 |
| White race | ref | |
| Black race | −0.28 (−0.32 to −0.25) | <0.001 |
| Smoking | ||
| No | ref | |
| Current | −0.25 (−0.32 to −0.19) | <0.001 |
| Past | −0.07 (−0.11 to −0.03) | <0.001 |
| Alcohol consumption | ||
| No | ref | |
| <1 drink/week | 0.12 (0.07 to 0.17) | <0.001 |
| 1–7 drinks/week | 0.15 (0.11 to 0.20) | <0.001 |
| >7 drinks/week | 0.11 (0.04 to 0.19) | <0.001 |
| No history of Diabetes | ref | |
| History of Diabetes | −0.22 (−0.27 to −0.17) | <0.001 |
| No history of Hypertension | ref | |
| History of Hypertension | −0.15 (−0.18 to −0.11) | <0.001 |
| No history of Coronary heart disease | ref | |
| History of Coronary heart disease | −0.08 (−0.13 to −0.03) | <0.001 |
| No history of Peripheral arterial disease | ref | |
| History of Peripheral arterial disease | −0.17 (−0.26 to −0.08) | <0.001 |
| No history of Left ventricular hypertrophy | ref | |
| Left ventricular hypertrophy | −0.12 (−0.18 to −0.06) | <0.001 |
|
| ||
| Continuous variables | Pearson correlation (95% CI) ‡ | |
|
| ||
| Age | −0.21 (−0.25 to −0.18) | <0.001 |
| Body mass index | −0.23 (−0.27 to −0.20) | <0.001 |
| Waist/thigh circumference ratio | −0.08 (−0.11 to −0.04) | <0.001 |
| Physical activity, kcal/kg/week | 0.15 (0.11 to 0.18) | <0.001 |
| Systolic blood pressure | −0.05 (−0.09 to −0.01) | 0.008 |
| Diastolic blood pressure | −0.02 (−0.06 to 0.02) | 0.27 |
| Heart rate | −0.14 (−0.17 to −0.10) | <0.001 |
| Fasting glucose | −0.15 (−0.19 to −0.11) | <0.001 |
| Albumin | 0.07 (0.04 to 0.11) | <0.001 |
| Creatinine | −0.15 (−0.18 to −0.11) | <0.001 |
| Total cholesterol | 0.00 (−0.03 to 0.04) | 0.83 |
| Low-density lipoprotein | 0.01 (−0.03 to 0.05) | 0.6 |
| High-density lipoprotein | −0.02 (−0.06 to 0.01) | 0.24 |
| Triglycerides | 0.02 (−0.02 to 0.06) | 0.33 |
| Resistin | −0.12(−0.15 to −0.08) | <0.001 |
| TNF alpha | −0.09 (−0.12 to −0.05) | <0.001 |
| Interleukin-6 | −0.12 (−0.16 to −0.09) | <0.001 |
| Ankle Arm Index | 0.14 (0.10 to 0.18) | <0.001 |
Adjusted for age, sex
Note: lower scores on the HABC battery indicate more severe frailty.
Frailty and Incident Heart Failure
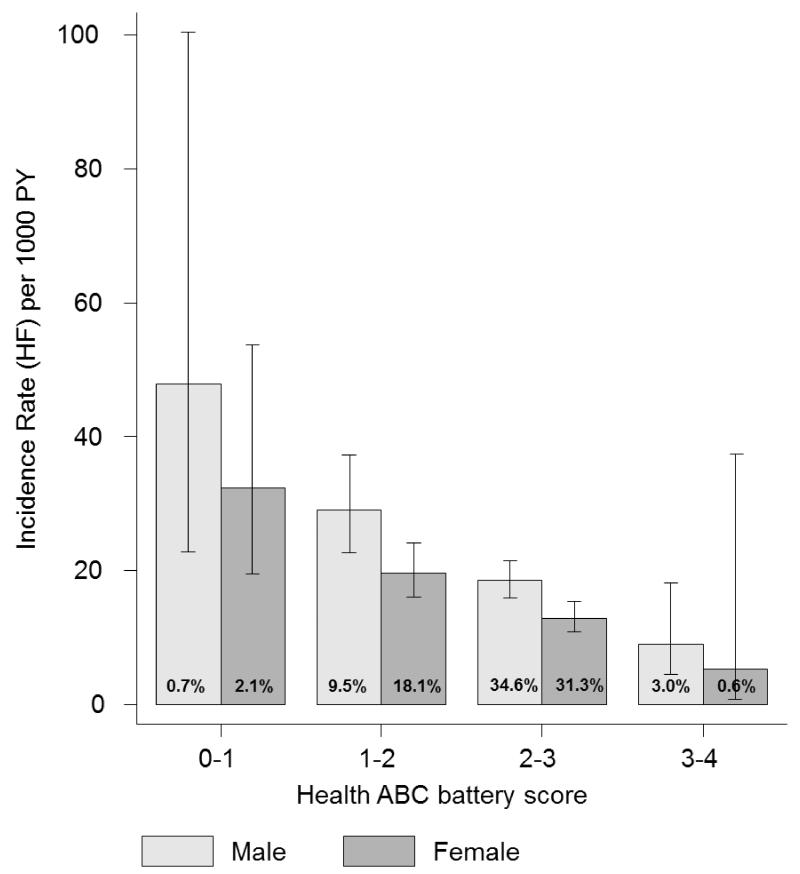
During a median follow-up of 11.4 (7.1-11.7) years, 466 (15.9%) participants developed HF (17.7 per 1000 person-years), Figure 1. Compared to non-frail participants, those classified frail by the Gill index were at higher risk for HF (HR 1.36, 95%CI 1.08-1.71 and HR 1.88, 95%CI 1.02-3.47, for moderate and severe frailty respectively). HABC battery score was linearly associated with HF risk, Figure 2. In univariate analysis, HABC battery score was associated with HF risk (HR 1.40, 95%CI 1.28-1.53 per SD decrease in score); this association persisted after adjustment for HABC HF Risk Score (HR 1.24, 95%CI 1.13-1.36). HR did not vary importantly in analyses that excluded HF events recorded in the first year of follow-up (data not shown). Progressive adjustments for serum lipids, physical activity, serum resistin, TNF-α, IL-6, history of arrhythmias and respiratory function (FEV1/FVC) did not attenuate the association, Table 3. Frailty remained a significant predictor of HF after controlling for incident CHD as a time varying covariate and death as a competing event.
Figure 1. HABC battery score and incident heart failure.
Lower baseline HABC battery score was associated with a higher risk for heart failure. Rates are shown per 1000 person-years.
Figure 2. HABC battery score and heart failure risk.
Hazard ratio and 95% “floating absolute” confidence intervals per quintile Health ABC battery are shown. The size of the box is proportional to the inverse of the variance of hazard ratio. Model adjusted for age and gender. Note: The fifth category is the reference
Table 3. Heart failure risk per standard deviation decrease in HABC battery score.
| Factor progressively adjusted for | All Participants (n=2,825) |
|---|---|
| HR (95% CI) | |
| Unadjusted | 1.40 (1.28-1.53) |
| Age and gender | 1.36 (1.24-1.48) |
| plus HABC Score | 1.24 (1.13-1.36) |
| plus high- and low-density lipoprotein, log Triglycerides | 1.24 (1.13-1.37) |
| plus physical activity | 1.25 (1.14-1.38) |
| plus resistin, TNF alpha, interleukin-6 | 1.23 (1.12-1.36) |
| plus history of arrhythmias, FEV1/FVC | 1.23 (1.12-1.36) |
| plus time varying incident CHD | 1.24 (1.13-1.36) |
FEV1/FVC. Ratio of Forced expiratory volume at 1 second to Forced Vital Capacity.
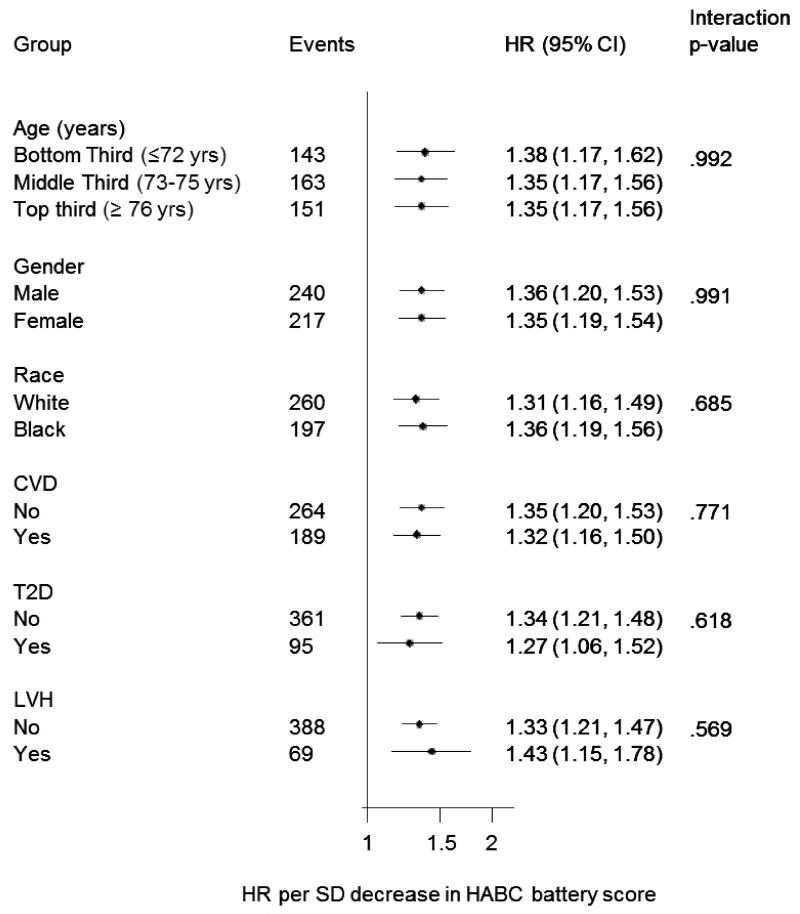
The association between HABC battery score and incident HF was similar across subgroups defined by age, gender, race, prevalent CHD, diabetes mellitus and left ventricular hypertrophy, Figure 3. HABC battery score had better discrimination than Gill index for HF prediction (C 0.527 vs. 0.586 for Gill index and SBBP score, respectively), Figure 4. The Health ABC HF Risk Score yielded a C-index of 0.657 (95%CI 0.646-0.668); addition of HABC battery score improved the model (change in C index, 0.014; 95%CI 0.018-0.010). The addition of the HABC battery score to the Health ABC risk predictors appropriately reclassified 8.3% of participants who developed HF over a 5 year follow-up period, and 5.15 of participants who did not (NRI 0.073, 95%CI 0.021-0.125; P=0.006), Table 4.
Figure 3. HABC battery scores and heart failure risk in subgroups.
Frailty was associated with higher heart failure risk in multiple subgroups studied. Note: The model is adjusted for age and gender and stratified for prevalent coronary heart disease. CVD - Cardiovascular disease; LVH - left ventricular hypertrophy; T2D - type 2 diabetes mellitus
Figure 4. Comparison of discrimination of HABC battery scores and Gill index.
Table 4. Risk discrimination and reclassification with HABC battery score added to HABC Heart Failure Risk Model.
| Discrimination | |
|---|---|
| C-index (95% CI): HABC score | 0.657(0.646,0.668) |
| C-index (95% CI): HABC score plus HABC battery | 0.671(0.661,0.682) |
| C-index change (95% CI) | 0.014(0.018,0.010) |
| P-value | <0.001 |
|
| |
| Reclassification | |
|
| |
| Participants who did not develop HF at 5 years | |
| Appropriately reclassified | 166(5.18%) |
| Inappropriately reclassified | 137(6.28%) |
| Participants who developed HF at 5 years | |
| Appropriately reclassified | 15(8.25%) |
| Inappropriately reclassified | 4(2.03%) |
| Net reclassification index (95% CI) | 0.073(0.021,0.125) |
| P-value | 0.006 |
| Integrated discrimination index (95% CI) | 0.003(−0.046,0.052) |
| P-value | 0.89 |
Frailty, Incident CHD, and Heart Failure
In participants without CHD at baseline (n=2229), the unadjusted association of frailty using the HABC battery scores with incident HF risk was similar in those who developed CHD during follow-up (n=373; HR 1.36, 95%CI 1.16-1.60) and in those who remained free of CHD (n=1856; HR 1.41, 95%CI 1.21-1.63; P=0.79 for interaction).
DISCUSSION
In this cohort of community-based elderly, frailty was a significant predictor of incident HF. A unit decrease in HABC battery score, signifying worsening frailty, was associated with 30% (10-55%) increase in HF risk. These findings were consistent in both sexes, and in blacks and whites, and were independent of clinical risk factors, inflammatory markers, ankle arm index, incident CHD, and death as a competing event. These results highlight that frailty raises HF risk independent of other known risk factors.
Poor lower-extremity performance has been previously reported to be predictive of adverse outcomes (14,29). Simple measures of physical performance such as gait speed and short walk time capture many aspects of chronic conditions and overall functional status, and may be combined together as a battery of tests for assessing multi-dimensional syndromes like frailty. However simple measures like the Gill criteria or the 6 minute walk speeds alone may be insensitive to capture the early stages of risk typically showing ceiling effects (17). The HABC battery test based on composite of physical assessments was modeled to address this limitation while recognizing the multi systemic nature of the frailty syndrome. In our study, both scales were associated with a higher HF risk.
Frailty has been described as a multidimensional syndrome resulting from the interplay of genetic, biological, physical, psychological, social and environmental factors (10). Studies have shown that impairment of inflammatory(10-11) and metabolic pathways(10, 30) may be the underlying biological process involved. Consistent with such suggestions, we observed that frailty was significantly correlated with measures of adiposity, glycemia, and inflammation. Progressive adjustment for these correlates only mildly attenuated the association of frailty with HF, suggesting that there are as yet unexplored mediators of the association between frailty and HF that need to be identified.
Despite of the poor understanding of the biological basis, exercise has been identified as an important frailty “therapy” shown to improve outcomes and quality of life for these patients (31). Restoration of physical function through resistance training has shown benefit in frail elderly (32-34). Physical activity has been seen to have a positive effect on several modulatory pathways. A decrease in circulating levels of inflammatory markers (35), production of free radical scavengers (36), and improvement of insulin resistance (37) are all intrinsic pathways through which physical training helps prevent debility. Recognition of frailty can alert physicians that the patient may benefit from geriatric care and or an exercise program.
The Health ABC Study cohort was selected on the basis of absence of disability and mobility impairment. Thus the distribution of frailty scores may not be representative of the general population. Echocardiography was not performed at baseline in the Health ABC Study. Thus, the association of frailty with sub-clinical left ventricular dysfunction could not be assessed. However, we intended to assess the association between frailty and clinical HF, and not asymptomatic left ventricular dysfunction. Also, because HF is unlikely to remain undiagnosed for several years, we contend that the observed association cannot be ascribed merely to undetected HF at baseline. From prevention perspective, clinically manifest disease is more relevant, e.g. in coronary disease prevention strategies, participants do not undergo coronary angiography to rule out sub-clinical coronary disease. Finally reclassification metrics are sensitive to choice of follow-up interval and clinical risk categories (32).
In conclusion, we demonstrate a significant association between frailty and risk of incident HF. in older individuals. These findings were consistent across gender and race, and persisted after controlling for other HF risk factors. Practical implications of our findings include screening elderly for frailty with the aim to identify individuals at increased risk for HF who may benefit from interventions aimed at improving their physical functioning and quality of life.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflicts of Interest: None
REFERENCES
- 1.Lupón J, González B, Santaeugenia S, et al. Prognostic implication of frailty and depressive symptoms in an outpatient population with heart failure. Rev Esp Cardiol. 2008;61(8):835–42. [PubMed] [Google Scholar]
- 2.Murad K, Kitzman DW. Frailty and multiple comorbidities in the elderly patient with heart failure: implications for management. Heart Fail Rev. 2012;17(4-5):581–8. doi: 10.1007/s10741-011-9258-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Purser JL, Kuchibhatla MN, Fillenbaum GG, et al. Identifying frailty in hospitalized older adults with significant coronary artery disease. J Am Geriatr Soc. 2006;54(11):1674–81. doi: 10.1111/j.1532-5415.2006.00914.x. [DOI] [PubMed] [Google Scholar]
- 4.Afilalo J. Frailty in Patients with Cardiovascular Disease: Why, When, and How to Measure. Curr Cardiovasc Risk Rep. 2011;5(5):467–72. doi: 10.1007/s12170-011-0186-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pel-Littel RE, Schuurmans MJ, Emmelot-Vonk MH, et al. Frailty: defining and measuring of a concept. J Nutr Health Aging. 2009;13(4):390–4. doi: 10.1007/s12603-009-0051-8. [DOI] [PubMed] [Google Scholar]
- 6.Newman AB, Simonsick EM, Naydeck BL, et al. Association of long-distance corridor walk performance with mortality, cardiovascular disease, mobility limitation, and disability. JAMA. 2006;295(17):2018–26. doi: 10.1001/jama.295.17.2018. [DOI] [PubMed] [Google Scholar]
- 7.Newman AB, Gottdiener JS, Mcburnie MA, et al. Associations of subclinical cardiovascular disease with frailty. J. Gerontol. A Biol. Sci. Med. Sci. 2001;56(3):M158–166. doi: 10.1093/gerona/56.3.m158. [DOI] [PubMed] [Google Scholar]
- 8.Cacciatore F, Abete P, Mazzella F, et al. Frailty predicts long-term mortality in elderly subjects with chronic heart failure. Eur. J. Clin. Invest. 2005;35(12):723–30. doi: 10.1111/j.1365-2362.2005.01572.x. [DOI] [PubMed] [Google Scholar]
- 9.Alexander KP, Newby LK, Cannon CP. Acute coronary care in the elderly, part I: Non-ST-segment-elevation acute coronary syndromes: a scientific statement for healthcare professionals from the American Heart Association Council on Clinical Cardiology: in collaboration with the Society of Geriatric Cardiology. Circulation. 2007;115(19):2549–69. doi: 10.1161/CIRCULATIONAHA.107.182615. Aet al. [DOI] [PubMed] [Google Scholar]
- 10.Walston J, Hadley EC, Ferrucci L, et al. Research agenda for frailty in older adults: toward a better understanding of physiology and etiology: summary from the American Geriatrics Society/National Institute on Aging Research Conference on Frailty in Older Adults. J Am Geriatr Soc. 2006;54(6):991–1001. doi: 10.1111/j.1532-5415.2006.00745.x. [DOI] [PubMed] [Google Scholar]
- 11.Ferrucci L, Harris TB, Guralnik JM, et al. Serum IL-6 level and the development of disability in older persons. J Am Geriatr Soc. 1999;47(6):639–46. doi: 10.1111/j.1532-5415.1999.tb01583.x. [DOI] [PubMed] [Google Scholar]
- 12.Abellan van Kan G, Rolland Y, Bergman H, et al. The I.A.N.A Task Force on frailty assessment of older people in clinical practice. J Nutr Health Aging. 2008;12(1):29–37. doi: 10.1007/BF02982161. [DOI] [PubMed] [Google Scholar]
- 13.Gill TM, Baker DI, Gottschalk M, et al. A program to prevent functional decline in physically frail, elderly persons who live at home. N. Engl. J. Med. 2002;347(14):1068–74. doi: 10.1056/NEJMoa020423. [DOI] [PubMed] [Google Scholar]
- 14.Penninx BW, Ferrucci L, Leveille SG, et al. Lower extremity performance in nondisabled older persons as a predictor of subsequent hospitalization. J. Gerontol. A Biol. Sci. Med. Sci. 2000;55(11):M691–697. doi: 10.1093/gerona/55.11.m691. [DOI] [PubMed] [Google Scholar]
- 15.Chiarantini D, Volpato S, Sioulis F, et al. Lower extremity performance measures predict long-term prognosis in older patients hospitalized for heart failure. J. Card. Fail. 2010;16(5):390–5. doi: 10.1016/j.cardfail.2010.01.004. [DOI] [PubMed] [Google Scholar]
- 16.Volpato S, Cavalieri M, Sioulis F, et al. Predictive value of the Short Physical Performance Battery following hospitalization in older patients. J. Gerontol. A Biol. Sci. Med. Sci. 2011;66(1):89–96. doi: 10.1093/gerona/glq167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Simonsick EM, Newman AB, Nevitt MC, et al. Measuring higher level physical function in well-functioning older adults: expanding familiar approaches in the Health ABC study. J. Gerontol. A Biol. Sci. Med. Sci. 2001;56(10):M644–649. doi: 10.1093/gerona/56.10.m644. [DOI] [PubMed] [Google Scholar]
- 18.Ainsworth BE, Haskell WL, Whitt MC, et al. Compendium of physical activities: an update of activity codes and MET intensities. Med Sci Sports Exerc. 2000;32(9 Suppl):S498–504. doi: 10.1097/00005768-200009001-00009. [DOI] [PubMed] [Google Scholar]
- 19.Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49(2):M85–94. doi: 10.1093/geronj/49.2.m85. [DOI] [PubMed] [Google Scholar]
- 20.Fried LP, Borhani NO, Enright P, et al. The Cardiovascular Health Study: design and rationale. Ann Epidemiol. 1991;1(3):263–76. doi: 10.1016/1047-2797(91)90005-w. [DOI] [PubMed] [Google Scholar]
- 21.Rodondi N, Newman AB, Vittinghoff E, et al. Subclinical hypothyroidism and the risk of heart failure, other cardiovascular events, and death. Arch. Intern. Med. 2005;165(21):2460–6. doi: 10.1001/archinte.165.21.2460. [DOI] [PubMed] [Google Scholar]
- 22.Easton DF, Peto J, Babiker AG. Floating absolute risk: an alternative to relative risk in survival and case-control analysis avoiding an arbitrary reference group. Stat Med. 1991;10(7):1025–35. doi: 10.1002/sim.4780100703. [DOI] [PubMed] [Google Scholar]
- 23.Butler J, Kalogeropoulos A, Georgiopoulou V, et al. Incident heart failure prediction in the elderly: the health ABC heart failure score. Circ Heart Fail. 2008;1(2):125–33. doi: 10.1161/CIRCHEARTFAILURE.108.768457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509. [Google Scholar]
- 25.Pencina MJ, D’Agostino RB, Sr, D’Agostino RB, Jr, et al. Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat Med. 2008;27(2):157–172. doi: 10.1002/sim.2929. discussion 207-212. [DOI] [PubMed] [Google Scholar]
- 26.Van Buuren S, Boshuizen HC, Knook DL. Multiple imputation of missing blood pressure covariates in survival analysis. Stat Med. 1999;18(6):681–94. doi: 10.1002/(sici)1097-0258(19990330)18:6<681::aid-sim71>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- 27.Ambler G, Omar RZ, Royston P. A comparison of imputation techniques for handling missing predictor values in a risk model with a binary outcome. Stat Methods Med Res. 2007;16(3):277–98. doi: 10.1177/0962280206074466. [DOI] [PubMed] [Google Scholar]
- 28.Barnard J, Rubin D. Small-sample degrees of freedom with multiple imputation. Biometrika. 1999;86:948–55. [Google Scholar]
- 29.Bittner V, Weiner DH, Yusuf S, et al. Prediction of mortality and morbidity with a 6-minute walk test in patients with left ventricular dysfunction. SOLVD Investigators. JAMA. 1993;270(14):1702–7. [PubMed] [Google Scholar]
- 30.Schalk BWM, Visser M, Deeg DJH, et al. Lower levels of serum albumin and total cholesterol and future decline in functional performance in older persons: the Longitudinal Aging Study Amsterdam. Age Ageing. 2004;33(3):266–72. doi: 10.1093/ageing/afh073. [DOI] [PubMed] [Google Scholar]
- 31.Binder EF, Schechtman KB, Ehsani AA, et al. Effects of exercise training on frailty in community-dwelling older adults: results of a randomized, controlled trial. J Am Geriatr Soc. 2002;50(12):1921–8. doi: 10.1046/j.1532-5415.2002.50601.x. [DOI] [PubMed] [Google Scholar]
- 32.Brown M, Sinacore DR, Ehsani AA, et al. Low-intensity exercise as a modifier of physical frailty in older adults. Arch Phys Med Rehabil. 2000;81(7):960–5. doi: 10.1053/apmr.2000.4425. [DOI] [PubMed] [Google Scholar]
- 33.Fiatarone MA, O’Neill EF, Ryan ND, et al. Exercise training and nutritional supplementation for physical frailty in very elderly people. N. Engl. J. Med. 1994;330(25):1769–75. doi: 10.1056/NEJM199406233302501. [DOI] [PubMed] [Google Scholar]
- 34.Nelson ME, Fiatarone MA, Morganti CM, et al. Effects of high-intensity strength training on multiple risk factors for osteoporotic fractures. A randomized controlled trial. JAMA. 1994;272(24):1909–14. doi: 10.1001/jama.1994.03520240037038. [DOI] [PubMed] [Google Scholar]
- 35.Adamopoulos S, Parissis J, Karatzas D, et al. Physical training modulates proinflammatory cytokines and the soluble Fas/soluble Fas ligand system in patients with chronic heart failure. J. Am. Coll. Cardiol. 2002;39(4):653–63. doi: 10.1016/s0735-1097(01)01795-8. [DOI] [PubMed] [Google Scholar]
- 36.Meijer EP, Goris AHC, van Dongen JLJ, et al. Exercise-induced oxidative stress in older adults as a function of habitual activity level. J Am Geriatr Soc. 2002;50(2):349–53. doi: 10.1046/j.1532-5415.2002.50069.x. [DOI] [PubMed] [Google Scholar]
- 37.Kirwan JP, Kohrt WM, Wojta DM, et al. Endurance exercise training reduces glucose-stimulated insulin levels in 60- to 70-year-old men and women. J Gerontol. 1993;48(3):M84–90. doi: 10.1093/geronj/48.3.m84. [DOI] [PubMed] [Google Scholar]
- 38.Pencina MJ, AgostinoD’ RB, Sr, Steyerberg EW. Extensions of net reclassification improvement calculations to measure usefulness of new biomarkers. Stat Med. 2011;30(1):11–21. doi: 10.1002/sim.4085. [DOI] [PMC free article] [PubMed] [Google Scholar]