Abstract
Purpose: Monoclonal antibodies or specific antibodies are now an essential tool of biomedical research and are of great commercial and medical value. The purpose of this study was to produce large scale of monoclonal antibody against CD34 in order to diagnostic application in leukemia and purification of human hematopoietic stem/progenitor cells. Methods: For large scale production of monoclonal antibody, hybridoma cells that produce monoclonal antibody against human CD34 were injected into the peritoneum of the Balb/c mice which have previously been primed with 0.5 ml Pristane. 5 ml ascitic fluid was harvested from each mouse in two times. Evaluation of mAb titration was assessed by ELISA method. The ascitic fluid was examined for class and subclasses by ELISA mouse mAb isotyping Kit. mAb was purified from ascitic fluid by affinity chromatography on Protein A-Sepharose. Purity of monoclonal antibody was monitored by SDS -PAGE and the purified monoclonal antibody was conjugated with FITC. Results: Monoclonal antibodies with high specificity and sensitivity against human CD34 by hybridoma technology were prepared. The subclass of antibody was IgG1 and its light chain was kappa. Conclusion: The conjugated monoclonal antibody could be a useful tool for isolation, purification and characterization of human hematopoietic stem cells.
Keywords: Monoclonal antibody, Large Scale generation, Ascetic fluid, Human CD34
Introduction
Hybridomas are cells that have been engineered to produce a desired monoclonal antibody in large amounts.1,2 Hybridoma technology is a well-known technique introduced to produce monoclonal antibodies in specialized cells.3
The CD34 antigen is a glycoprotein, expressed on all measurable hematopoietic stem cells and progenitor cells. The surface molecule CD34 is frequently used as a marker to identify hematopoietic progenitor cells with a molecular weight about 110 kDa.4,5 CD34 has a heavily glycosylated type I transmembrane protein. There is a wide range of kinases such as Protein kinase C and Tyrosine kinases could be used to phosphorylate this transmembrane protein.6,7
The CD34 mAbs recognize different epitopes on the CD34 antigen. The classification of epitopes detected by different CD34 mAbs has aided the selection of appropriate antibodies for use in specific clinical and research laboratory settings.8
For mass- production of the monoclonal antibody, hybridoma cells must be grown by one of the following methods: in vivo method; Injection of requested clone into the abdominal cavity of a suitably prepared mouse or in vitro method; Culture of the cells in tissue culture flasks.9
Further processing of the mouse ascitic fluid and of the tissue culture supernatant are required to obtain mAb with the required purity and concentration. The mouse method is generally familiar, well understood, and widely available in many laboratories. The tissue- culture methods have been expensive and time-consuming and often failed to produce the required amount of antibody without considerable skilled manipulation.9-12
The aim of this study was to produce large scale of monoclonal antibody against CD34 in order to diagnostic application in leukemia and purification of human hematopoietic stem/progenitor cells.
Materials and Methods
Production of ascitic fluid in peritoneum of mouse
Balb/c female mice (4-6 weeks old) were provided from Pasteur institute of Iran. 0.5 ml Pristane (2, 6, 10, 14 tetra methyl pentadecane, Sigma) was injected intraperitoneally into each mouse. Ten days after priming with Pristane, the cells of a suitable mono clone in density of 1–2×106 cells/ 0.5 ml PBS were injected intraperitoneally into each mouse. The mice were surveyed daily for production of ascitic fluid after the injection of hybridoma cells. About ten days after the injection of cells, abdomen of the mice were completely enlarged and their skins were extended. Using 19 gage needles, their ascitic fluids were harvested.After 4 days, ascitic fluid of the mice were harvested again and centrifuged and the related supernatants were collected for characterization.13
Titration of antibody
The titer of monoclonal antibody was assessed by ELISA method. Wells of ELISA plate (Nunc, Germany) were coated with 100 µl of BSA-conjugated peptide (20 μg/ml in PBS) overnight at 4 °C. Next day the plate was washed 3 times with PBS containing 0.05% Tween 20 (PBS-T) for 5 min. Non-specific sites of the plate were blocked with 2% BSA and incubated at 37ºC for 90 minutes. Wells were then washed 3 times as above and ascitic fluid were added to the wells in two fold serial dilutions starting from 1:1000. The plate was incubated at 37 °C for 1.5 hr and washed again with PBS-T. At the next step, 100 μl of 1:4000 dilution of HRP-conjugated rabbit anti-mouse Ig (Sigma-Aldrich Co. Louis, USA) was added to the wells and incubation was continued for 1.5 hr at 37 °C. After washing, 100 ul of Tetramethylbenzidine (TMB) substrate was added to each well and the plate was incubated at room temperature in a dark place. After 20 min, the reaction was stopped by adding 100μ1 of stopping solution (0.16 M H2SO4) to each well. The Optical Density (OD) of the reactions was measured at 450 nm by an ELISA reader (STAT FAX 303+).14
Determination of mAb isotype
ELISA mouse mAb isotyping Kit (Thermo, USA) was used for determination the class and subclass of the mAbs. In this assay was used ELISA strip-well plates with individual wells pre-coated with anti-mouse heavy-chain capture antibody (anti-IgG1, IgG2a, IgG2b, IgG3, IgA and IgM) or anti-mouse light-chain capture antibody (kappa or lambda). First, Tris buffer saline (TBS) was used for 1/50000 dilution of the ascitic fluid and 50 µl of diluted antibody added to each well of the 8-well strip. Then 50 µl of the anti-mouse IgG + IgA+ IgM + HRP conjugated was added to each well of the 8-well strip and incubated for an hour at room temperature. After 3 times washing, 75 µl of TMB substrate was added to each well and the plate was incubated at room temperature in a dark place for10 min. At the next step, the reaction was stopped by 75μl of 5% solution of Sulfuric Acid. The absorbance of each well was read by ELISA Reader (STAT FAX 303+) at 450nm.
Antibody purification
The ascitic fluids were diluted two times with PBS and fractionated with 40% saturated ammonium sulfate. After several times of washing with 40% ammonium sulfate, the fraction was centrifuged for 15 minutes in 5000g. The precipitated fraction was dialyzed against 10 mM PBS, pH 7.4 and purified using Sepharose beads conjugated with Protein A column affinity chromatography equilibrated with 5-10 column volumes with the same buffer.
Mouse IgG1 was eluted with 0.1 M sodium phosphate buffer in pH 6.0. Confirmation of the purified fractions was monitored by SDS polyacrylamide gel electrophoresis. Finally, the purified fractions were kept for conjugation with fluorescein isothiocyanate.15
Confirmation of the mAb purity by SDS-PAGE
Purity of the monoclonal antibody was checked by SDS polyacrylamide gel electrophoresis in non-reducing condition and reducing form. 10 µg of purified mAb was mixed with 10 µl of sample buffer. The samples were boiled for 10 min at 100 °C .Electrophoresis was done in a 12.5% SDS-PAGE gel with a mini- PROTEAN electrophoresis instrument (Bio- Rad Laboratories, Hercules, CA, USA) 100 V for 1 hr. The gel was stained with Coomassie Brilliant Blue R-250 (Sigma).13
Conjugation of monoclonal antibody with fluorescein isothiocyanate (FITC)
For conjugation, 200 μl mAb (5mg/ml) was added in 800 μl Reaction Buffer (500 Mm Carbonate, pH=9.2) and dialyzed against PBS buffer in 24 hours. The antibody concentration was measured after buffer equilibration in 280 nm. 10 mg of FITC was dissolved in 1 mL anhydrous DMSO immediately before use. FITC (SIGMA, Germany) was added to give a ratio of 80 µg per mg of antibody and mixed immediately. The tubes were wrapping in foil then incubated and rotated at room temperature for 1 hour. The unreacted FITC was removed and exchanged the antibody into Storage Buffer (10mM Tris, 150mM Nacl, 0.1% NaH3, pH=8.2) by dialysis during overnight.16
Direct Immunofluorescence Staining
This technique was used for confirming the result of conjugation method and reactivity of fluorochrome-conjugated monoclonal antibody. First, human hematopoietic stem / progenitor cells as a positive control (CD34+) and SP2/0 as a negative control (CD34-) were prepared. Then 1/1000 dilution of FITC-conjugated monoclonal antibody was added. The mixture was incubated for 45 minutes at RT. Cells were washed two times with PBS buffer for 5 minutes. Cells were suspend in 200 μl of 3.7% formaldehyde solution for fixation of cells in 10 min at Room temperature. The washing was repeated then stained cells were examined by florescent microscope.17
Results
After priming of the mouse peritoneal with pristane, 1–2 millions cells related to the suitable mono clone (Figure 1) were suspended in 0.5 ml of sterile PBS and injected to each mouse. Approximately ten days later, 5 ml ascitic fluid collected from each mouse in two times (About 3.5 ml ascitic fluid was harvested from each mouse after ten day and about 1.5 ml ascitic fluid was harvested from their peritoneum for a second time, after 4 days ).
Figure 1 .
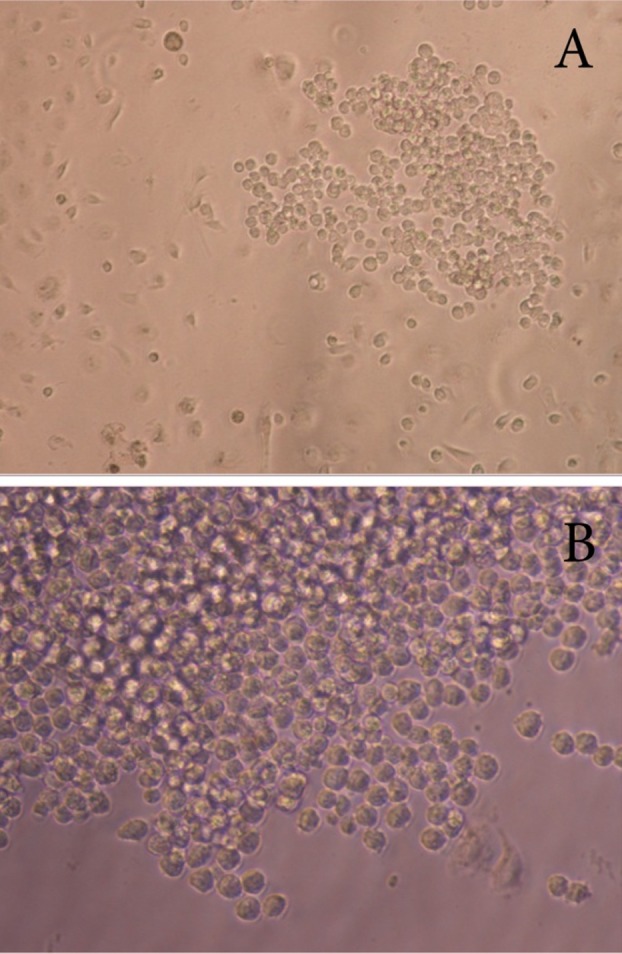
Proliferated suitable mono clone (Mag.10X) selected for injection into the peritoneum of mice. Monoclone in the growing form (A), Monoclone in the highly proliferated form (B)
The titer of monoclonal antibody in ascitic fluid was assessed by ELISA method. The mean absorbance of non-immune mouse serum, Immune mouse serum, and ascitic fluid was compared in Table 1 at 450 nm. The results showed that its 1/32000 dilution has high absorbance with CD34 antigen (above 1).
Table 1. Comparison of the mean absorbance of ascetic fluid at 450 nm .
| NC (SP/0) | NC* (Non-Immune mouse serum) | PC** (Immune mouse serum) | Ascetic fluid (1/32000 dilution) |
| 0.09 | 0.13 | 1.18 | 1.03 |
| * Negative Control with 1/8000 dilution | |||
| ** Positive Control with 1/8000 dilution | |||
Further characterization of this antibody showed that it is an IgG1 isotope with a kappa light chain. The product was precipitated by saturated ammonium sulfate and dialyzed against PBS. Concentration of the dialyzed product in assay with UV at 280 nm was about 35 mg. Purification by Protein-A-Sepharose column affinity chromatography yielded about 5.5 mg of monoclonal antibody.
The result of purification was confirmed with non-reducing SDS-PAGE and reducing SDS-PAGE. In reducing SDS-PAGE, two bands of 50 KD and 25 KD were appeared that demonstrator heavy and light chains. In non-reducing SDS-PAGE, only one 150 KD band was appeared that demonstrator of purified antibody (Figure 2).
Figure 2.
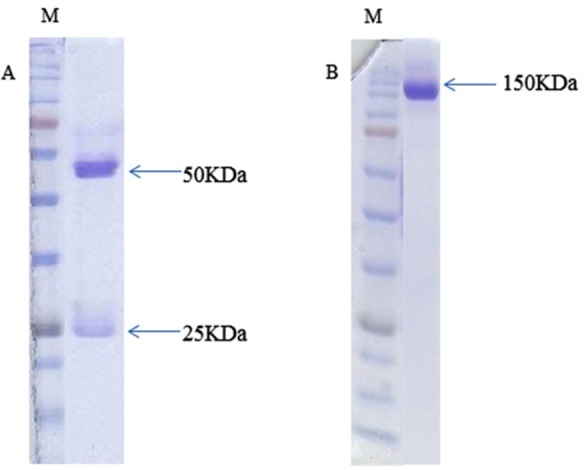
SDS-PAGE analysis of fraction from Protein-A affinity purification. Reducing SDS-PAGE (A) and non-reducing SDS-PAGE (B) of produced monoclonal antibody. In reduced form, two bands were seen in 50 & 25 kDa but in non-reducing SDS-PAGE condition, only one band was seen in about 150 kDa.
The purified monoclonal antibody was conjugated with fluorescein isothiocyanate (FITC) and used for direct staining of cell lines. To examine the specific attachment of purified mAb with CD34 antigen, cell lines were subjected to immunofluorescent staining (Figure 3).
Figure 3 .
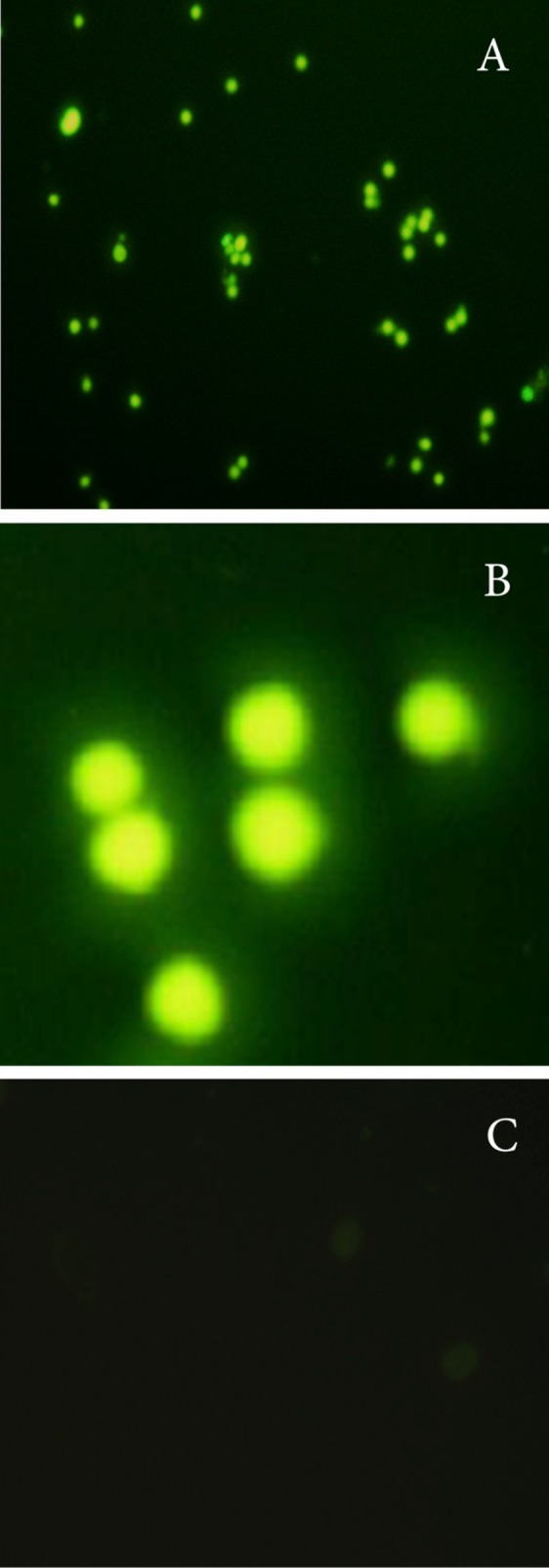
Direct Immunofluorescence Staining was used for confirming specific binding of purified mAb with CD34 antigen in the surface of Human hematopoietic stem / progenitor cells as a positive control (A&B Mag.10X, 40X respectively) and SP2/0 as a negative control (C).
Discussion
Monoclonal antibodies or specific antibodies are now an essential tool of biomedical research and are of great commercial and medical value. In recent century, after the first antibody-based therapy introduced, many researchers tried to use these molecules for diagnosis and treatment of several diseases.18-20 Koehler and Milstein developed the basic methods of producing monoclonal antibodies in hybridoma cell of the mouse in 1975 which has changed slightly to date.21
The production of monoclonal antibody in the ascitic fluid is commercially useful for mass production. In this study, ascitic fluids were collected from the peritoneal cavity and the titer of monoclonal antibody was assessed by ELISA method. The results showed that 1/32000 dilution has high absorbance with CD34 antigen (above 1). Choice of procedure for antibody purification depends on the intended use of the antibodies, isotyping of antibodies and on the available resources.22
The subclass of monoclonal antibody was IgG1 and its light chain was “kappa” type. The affinity chromatography method appears to be a simple, inexpensive, one-step and time-efficient approach in comparison with the other chromatography methods such as ion-exchange chromatography. Accordingly, we utilized this technique for purification of mAb produced.
The method of choice for determining purity is SDS-PAGE. Therefore, purity was evaluated by SDS-PAGE in non-reducing form and by SDS-PAGE in reducing condition. SDS-PAGE analysis showed that we obtained a protein with suitable purity after purification step.
The purified monoclonal antibody was conjugated with fluorescein isothiocyanate (FITC). Specific binding of purified mAb with CD34 antigen was monitored using immunofloresance techniques in the surface of human hematopoietic stem / progenitor cells. The results clearly showed that our antibody strongly reacts with CD34 antigen (shown in Figure 3).
Currently, we know two ways for production of the desired antibody: the mouse ascites method and the tissue-culture method which could be performed in vivo and in vitro.23
In vitro cell-culture method requires some expertise, requires special media, and can be expensive and time-consuming. Moreover, in this technique, unsuitable glycosylation may lead antibody to be unusable in in vivo experiments resulting from changes in immunogenicity, binding affinity, biologic functions, or clearance in vivo.10,12
In in vivo production method, at first, a primer such as pristane or Freud’s incomplete adjuvant is injected to suppress the immune systems and then the multiplied hybridoma cells form antibody-rich ascitic fluid in the peritoneal cavity. The ascites technique has some advantages, mainly, high levels of antibody production ranges from 1 to 20 mg/ml. In addition, this technique is not excessively labour- intensive.9 Briefly, ascitic fluid production enriched with mAb in mice is a rapid and economic method.
The amount of the injected pristane and the interval of priming with hybridoma cells are very important factors in ascitic fluid production. In addition it is important that side effects of tumor growth can be more severe due to incorrect i.p. injection of hybridoma cells as a result of insemination of hybridoma cells in abdominal organs, such as urinary bladder or intestines.24 On the other hand the number of the cells injected to the peritoneum of mouse is highly effective on the acceleration of ascitic fluid production.25
During ascites development, animals should be observed at least three times per week for the first week and daily thereafter to monitor the degree of abdominal distention and signs of illness.26 Peterson evaluated the effects on well-being of pristane injection and ascites production using factors such as wheel-running activity, food and water consumption, clinical observation, and plasma corticosterone concentration. No significant evidence of distress was obtained in the animals studied.27 But, Mauch et al reported that elevation of the diaphragm due to ascites is associated with dyspnea, orthopnea, or tachypnea. It therefore seems reasonable to assume that mice with large accumulations of ascitic fluid experience discomfort and distress.28
In similar previous study, Baradaran et al used in vivo method for mass production of monoclonal antibody against EGFR in ascitic fluid efficiently and 10.4 mg antibody was purified with Ion exchange chromatography (IEC).29
In other study Brian Scott Hafley used in vivo method for development of monoclonal antibodies.30 Moreover Mittal et al used the same in production of murine monoclonal antibodies against Haemophilus parasuis .31 Furthermore Galen et al used in vivo method for mass production of monoclonal antibody against human rennin in ascitic fluid then the mAb with high purity was obtained by affinity chromatography. 32
In all these studies, the mouse ascites methods were preferred for its economical, efficient and high concentrations of mAbs produced.
On the other hand, Shu-Fen Chou et al used in the tissue- culture in flasks method for scale-up of anti-AFP mAbs. Then monoclonal antibodies were purified by affinity chromatography on protein A Sepharose.33
Based on documented evidence, analysis of mAb produced in tissue culture reveals that a desired antibody function is diminished or lost. Furthermore tissue culture might be maintained for long periods, and some mAb were denatured during concentration or purification.
Nevertheless, for several parameters, in vivo method has fallen into disfavor. The most important reasons relate to the following: significant pain and distress in mice; High-quality in vitro production systems are progressing; and contamination mAb with infectious agents, such as viruses and other microorganisms.25 Although, based on various reports there is now consensus that ascites production should be the exception, requiring rigorous and well-documented justification. Special circumstances that might justify the use of ascites production include the following: emerging therapeutic applications; downstream concentration of mAb from in vitro; denaturation and decreased antibody activity in the tissue culture; and poor growth of hybridoma cells in vitro system.9,12
Based on the reasons described above, it is reasonable to conclude that new developments in in vitro mAb production gradually will limit the use of animals for this purpose.
In general, ascitic fluid production method seems to be a very useful, inexpensive and economic. Finally, we recommend that mAb generation by the mouse ascites method be permitted if scientifically justified and approved by the relevant Institutional Animal Care and Use Committee (IACUC).
Acknowledgements
We would like to thank for Immunology Research Center (IRC) and Tabriz International University of Medical Sciences (Aras) for kind assistance, respectively. This work was supported by a grant from Faculty of Medicine, Tabriz University of Medical Sciences.
Conflict of Interest
The authors report no conflicts of interest in this work.
References
- 1.Pandey SH. Hybridoma technology for production of monoclonal antibodies. Int J Pharm Sci Rev Res . 2010;1(2):88–94. [Google Scholar]
- 2.Hybridoma technology. [Online encyclopedia] USA: Wikipedia; [cited 2013 January]; Available from: http://en.wikipedia.org/wiki/Hybridoma_technology.
- 3.Kohler G, Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 1975;256(5517):495–7. doi: 10.1038/256495a0. [DOI] [PubMed] [Google Scholar]
- 4.Qian W, Wang L, Li B, Wang H, Hou S, Hong X. et al. Development of new versions of anti-human CD34 monoclonal antibodies with potentially reduced immunogenicity. Biochem Biophys Res Commun. 2008;367(2):497–502. doi: 10.1016/j.bbrc.2007.12.184. [DOI] [PubMed] [Google Scholar]
- 5.Gunsilius E, Gastl G, Petzer AL. Hematopoietic stem cells. Biomed Pharmacother. 2001;55(4):186–94. doi: 10.1016/s0753-3322(01)00051-8. [DOI] [PubMed] [Google Scholar]
- 6.Deterding LJ, Williams JG, Humble MM, Petrovich RM, Wei SJ, Trempus CS. et al. CD34 antigen: Determination of specific sites of phosphorylation in vitro and in vivo. Int J Mass spectrom. 2011;301(1-3):12–21. doi: 10.1016/j.ijms.2010.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Krause DS, Fackler MJ, Civin CI, May WS. CD34: structure, biology, and clinical utility. Blood. 1996;87(1):1–13. [PubMed] [Google Scholar]
- 8.Lanza F, Healy L, Sutherland DR. Structural and functional features of the CD34 antigen: an update. J Biol Regul Homeost Agents. 2001;15(1):1–13. [PubMed] [Google Scholar]
- 9.Jackson LR, Trudel LJ, Fox JG, Lipman NS. Monoclonal antibody production in murine ascites. I. Clinical and pathologic features. Lab Anim Sci. 1999;49(1):70–80. [PubMed] [Google Scholar]
- 10.Mc Ardle J. Alternatives to ascites production of monoclonal antibodies. Ani Welf Inform Cent Newslett. 1998;8:3–4. [Google Scholar]
- 11.Lang AB, Schuerch U, Cryz SJ Jr. Optimization of growth and secretion of human monoclonal antibodies by hybridomas cultured in serum-free media. Hybridoma. 1991;10(3):401–9. doi: 10.1089/hyb.1991.10.401. [DOI] [PubMed] [Google Scholar]
- 12.Jackson LR, Trudel LJ, Fox JG, Lipman NS. Monoclonal antibody production in murine ascites. II. Production characteristics. Lab Anim Sci. 1999;49(1):81–6. [PubMed] [Google Scholar]
- 13.Baradaran B, Majidi J, Hassan ZM, Abdolalizadeh J. Large scale production and characterization of anti- human IgG monoclonal antibody in peritoneum of Balb/c mice. Am J Biochem Biotechnol. 2006;1(4):190–3. [Google Scholar]
- 14.Hadavi R, Zarnani AH, Ahmadvand N, Mahmoudi AR, Bayat AA, Mahmoudian J. et al. Production of Monoclonal Antibody against Human Nestin. Avicenna J Med Biotech. 2010;2(2):69–76. [PMC free article] [PubMed] [Google Scholar]
- 15.Thurston CF, Henle LF. New Protein Techniques. In: Walker JM, editor. Methods in Molecular Biology. Clifton NJ: The Humana Press Inc.; 1988. P. 149-58.
- 16.Harlow E, Lane D. Labeling antibodies with fluorochromes. CSH protocols 2006;2006(2). [DOI] [PubMed]
- 17.Tabatabaei-Panah AS, Zarnani AH, Montaser-Kouhsar Sh, Chamankhah M, Ghods R, Bayat AA. et al. Production and Characterization of Anti-Her2 Monoclonal Antibodies. Yakhteh Med J. 2008;10(2):109–20. [Google Scholar]
- 18.Robinson M, Weiner L, Adams G. Improving Monoclonal Antibodies for Cancer Therapy. Drug Dev Res. 2004;61:172–87. [Google Scholar]
- 19.Schrama D, Reisfeld R, Becker J. Antibody targeted drugs as cancer therapeutics. Annu Rev Med. 2006;5:174–90. doi: 10.1038/nrd1957. [DOI] [PubMed] [Google Scholar]
- 20.Enever C, Batuwangala T, Plummer C, Sepp A. Next generation immunotherapeutics--honing the magic bullet. Curr Opin Biotechnol. 2009;20(4):405–11. doi: 10.1016/j.copbio.2009.07.002. [DOI] [PubMed] [Google Scholar]
- 21.Modjtahedi H. Monoclonal Antibodies as Therapeutic Agents: Advances and Challenges. Iran J Immunol. 2005;2(1):3–21. [Google Scholar]
- 22.Fitzgerald J, Leonard P, Darcy E, O'Kennedy R. Immunoaffinity chromatography. Methods Mol Biol. 2011;681:35–59. doi: 10.1007/978-1-60761-913-0_3. [DOI] [PubMed] [Google Scholar]
- 23.Peterson NC, Peavey JE. Comparison of in vitro monoclonal antibody production methods with an in vivo ascites production technique. Contemp Top Lab Anim Sci. 1998;37(5):61–6. [PubMed] [Google Scholar]
- 24.Walvoort NC. Assessment of distress through pathological examination. In: Hendriksen CFM, Köeter HBWM, editors. Replacement, Reduction and Refinement: Present Possibilities and Future Prospects. Amsterdam: Elsevier; 1991. P. 265-73.
- 25.Leenaars M, Hendriksen CF. Critical steps in the production of polyclonal and monoclonal antibodies: evaluation and recommendations. ILAR J. 2005;46(3):269–79. doi: 10.1093/ilar.46.3.269. [DOI] [PubMed] [Google Scholar]
- 26.Peterson NC. Advances in monoclonal antibody technology: genetic engineering of mice, cells, and immunoglobulins. ILAR J. 2005;46(3):314–9. doi: 10.1093/ilar.46.3.314. [DOI] [PubMed] [Google Scholar]
- 27.Peterson NC. Behavioral, clinical, and physiologic analysis of mice used for ascites monoclonal antibody production. Comp Med. 2000;50(5):516–26. [PubMed] [Google Scholar]
- 28.Mauch P,Ultmann. Treatment of malignant ascites. In: DeVita VT, Hellman S, Rosenberg SA, editors. Cancer: Principles and Practice of Oncology. Philadelphia: Lippincott; 1985. P. 2150-3.
- 29.Baradaran B, Hosseini AZ, Majidi J, Farajnia S, Barar J, Saraf ZH. et al. Development and characterization of monoclonal antibodies against human epidermal growth factor receptor in Balb/c mice. Hum Antibodies. 2009;18(1-2):11–6. doi: 10.3233/HAB-2009-0195. [DOI] [PubMed] [Google Scholar]
- 30.Hafley BS. Development of Monoclonal Antibodies for a Multiple Antigen ELISA to Verify Safe Cooking End-Point Temperatures in Beef and Pork [PhD Dissertation]. USA: Texas A&M University; 2005.
- 31.Tadjine M, Mittal KR, Bourdon S, Gottschalk M. Production and characterization of murine monoclonal antibodies against Haemophilus parasuis and study of their protective role in mice. Microbiology. 2004;150(Pt 12):3935–45. doi: 10.1099/mic.0.27443-0. [DOI] [PubMed] [Google Scholar]
- 32.Galen FX, Devaux C, Atlas S, Guyenne T, Menard J, Corvol P. et al. New monoclonal antibodies directed against human renin. Powerful tools for the investigation of the renin system. J Clin Invest. 1984;74(3):723–35. doi: 10.1172/JCI111488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chou SF, Hsu WL, Hwang JM, Chen CY. Production of monoclonal and polyclonal antibodies against human alphafetoprotein, a hepatocellular tumor marker. Hybrid Hybridomics. 2002;21(4):301–5. doi: 10.1089/153685902760213921. [DOI] [PubMed] [Google Scholar]


