Abstract
Previous studies have demonstrated the ability of an eicosapentaenoic acid (EPA)-derived endogenous cyclopentenone prostaglandin (CyPG) metabolite, Δ12-PGJ3, to selectively target leukemic stem cells, but not the normal hematopoietic stems cells, in in vitro and in vivo models of chronic myelogenous leukemia (CML). Here we evaluated the stability, bioavailability, and hypersensitivity of Δ12-PGJ3. The stability of Δ12-PGJ3 was evaluated under simulated conditions using artificial gastric and intestinal juice. The bioavailability of Δ12-PGJ3 in systemic circulation was demonstrated upon intraperitoneal injection into mice by LC-MS/MS. Δ12-PGJ3 being a downstream metabolite of PGD3 was tested in vitro using primary mouse bone marrow-derived mast cells (BMMCs) and in vivo mouse models for airway hypersensitivity. ZK118182, a synthetic PG analog with potent PGD2 receptor (DP)-agonist activity and a drug candidate in current clinical trials, was used for toxicological comparison. Δ12-PGJ3 was relatively more stable in simulated gastric juice than in simulated intestinal juice that followed first-order kinetics of degradation. Intraperitoneal injection into mice revealed that Δ12-PGJ3 was bioavailable and well absorbed into systemic circulation with a Cmax of 263 µg/L at 12 h. Treatment of BMMCs with ZK118182 for 12 h resulted in increased production of histamine, while Δ12-PGJ3 did not induce degranulation in BMMCs nor increase histamine. In addition, in vivo testing for hypersensitivity in mice showed that ZK118182 induces higher airways hyperresponsiveness when compared Δ12-PGJ3 and/or PBS control. Based on the stability studies, our data indicates that intraperitoneal route of administration of Δ12-PGJ3 was favorable than oral administration to achieve effective pharmacological levels in the plasma against leukemia. Δ12-PGJ3 failed to increase histamine and IL-4 in BMMCs, which is in agreement with reduced airway hyperresponsiveness in mice. In summary, our studies suggest Δ12-PGJ3 to be a promising bioactive metabolite for further evaluation as a potential drug candidate for treating CML.
Introduction
Prostaglandins (PGs) are cyclooxygenase-derived products of long chain polyunsaturated fatty acids (PUFA), which exhibit diverse biological functions depending on their structure, location, and concentration [1]. PGE1, PGI2 and PGF2α possess therapeutic potential with limited stability at various physiological and non-physiological conditions [2]–[4]. Younger et al, reported that saline solution of PGE1, a vasodilator and smooth muscle relaxant widely used for treatment of erectile dysfunction, was found to be less stable at 37°C, pH 7.4, but the stability was significantly improved at 4°C [2]. Pifer et al demonstrated that PGI2 was more stable in the presence of human plasma than in a buffer devoid of plasma protein at pH 7.55, where binding of PGI2 to albumin stabilized the molecule from degradation [4].
Cyclopentenone prostaglandins (CyPGs) are metabolites of PGs that possess an alkylidenecyclopentenone ring and are produced by non-enzymatic dehydration and isomerization of PGD2 and PGD3 from arachidonic acid (ARA) and eicosapentaenoic acid (EPA), respectively [5], [6]. Of these, ARA-derived Δ12-prostaglandin J2 (Δ12-PGJ2) and 15-deoxy-Δ12,14-prostaglandin J2 (15d-PGJ2) have been characterized for their antineoplastic, antiviral, and anti-inflammatory activities [5]. Recently, we described the role of a CyPG metabolite, Δ12-prostaglandin J3 (Δ12-PGJ3) (Fig. 1B inset) derived from EPA, a marine omega-3 (n-3) PUFA, to alleviate the progression of leukemia in a murine model of chronic myelogenous leukemia (CML) [7]. It was found that Δ12-PGJ3 selectively targeted leukemia stem cells (LSC) for apoptosis in the spleen and bone marrow, but spared the normal hematopoietic stem cells (HSC) [7]. Although the mechanisms of apoptosis are currently being investigated, it is known that CyPGs primarily act via two different mechanisms that includes binding to two G protein-coupled receptors, DP1 and CRTH2 (DP2) [8], [9], and/or via covalent modification of specific cysteine residues in a subset of cellular proteins [10]. Synthetic agonists of the DP receptor such as ZK118182 have been used to treat high intraocular pressure in glaucoma [11]. On the other hand, activation of DP receptor(s) have been associated with increasing risk of airway hypersensitive responses mediated through CRTH2 in the degranulation of mast cells and eosinophils [11], [12]. Therefore, considering the evolving pharmacological role of Δ12-PGJ3, we deemed it essential to further assess the stability under physiological conditions and possible toxicities in terms of hypersensitivity that essentially dictates its efficacy as a potential therapeutic for CML.
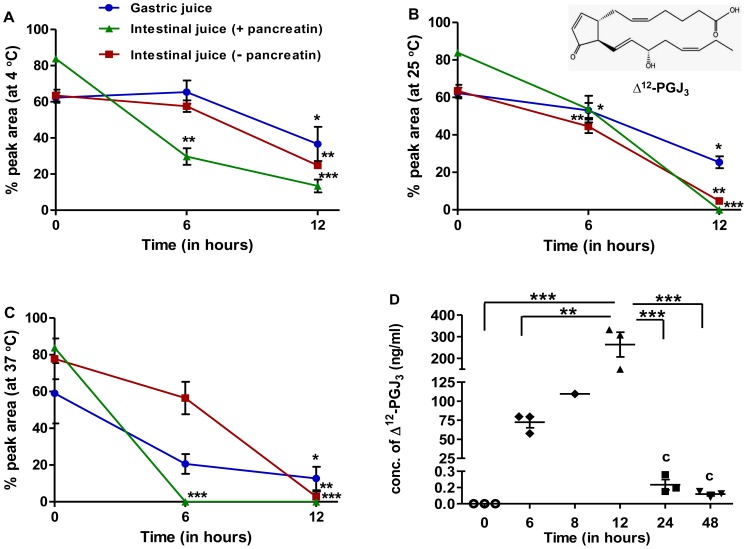
Figure 1. Study on stability and bioavailability of Δ12-PGJ3.
(A–C) Δ12-PGJ3 (0.5 µg/mL) was incubated in artificial gastric and intestinal juice (with or without pancreatin) for various time intervals and temperatures (4°C (A), 25°C (B), 37°C (C) and estimated for % of Δ12-PGJ3 remaining by HPLC. Significant differences between concentrations Δ12-PGJ3 at different time points as compared to t = 0 are indicated by *p≤0.05; **p≤0.01; ***p≤0.001 respectively. Data shown are the mean ± SEM (n = 3). (D) Time course analysis for bioavailability of Δ12-PGJ3 in mouse plasma. Δ12-PGJ3 (0.025 mg/kg body weight) was injected intraperitoneally into C57BL/6 mice. The concentration of Δ12-PGJ3 in plasma was measured at indicated times post injection by LC-MS/MS. The chemical structure of Δ12-PGJ3 is shown in the inset of panel B. All data shown are mean ± SEM (n = 3) and statistical significance represented as *- p≤0.05; **- p≤0.01; ***- p≤0.001 respectively.
Here we have examined the thermal stability and pH stability of Δ12-PGJ3 under simulated conditions followed by its bioavailability in vivo and the likelihood of any potential risk of hypersensitivity of Δ12-PGJ3 to cause degranulation of mast cells. Our studies indicate that Δ12-PGJ3 was stable and bioavailable upon intraperitoneal administration in mice. Furthermore, in vitro and in vivo experiments clearly show that Δ12-PGJ3 differed from the synthetic agonist of DP1 receptor, ZK118182, in terms of its ability to induce airways hyperresponsiveness, release of histamine, and IL-4 expression, which are all associated with hypersensitivity. In summary, these studies should pave way for in-depth immunotoxicological studies of Δ12-PGJ3 for use as a therapeutic agent in CML.
Materials and Methods
Materials
Artificial gastric juice (hydrochloric acid 1%, pepsin (1∶3000) <1%, water 98%), artificial intestinal juice (potassium phosphate, monobasic <1%, sodium hydroxide 50%, <1%, water 99%) and pancreatin (a crude mixture of digestive enzymes composed of amylases, lipases, and proteases) were purchased from ScholAR® chemistry (Rochester, NY). ZK118182 was purchased from Cayman Chemicals (Ann Arbor, MI) and used without any further purification. Δ12 PGJ3 was prepared as described earlier from our laboratory [7]. The purity of Δ12-PGJ3 was confirmed by LC-MS/MS to be ≥99%. Toluidine blue, ionomycin, and all HPLC grade-solvents used were from Sigma-Aldrich (St. Louis, MO). Histamine EIA kit was purchased from Cayman Chemical (Ann Arbor, MI).
Methods
Measurement of the stability of Δ12-PGJ3 in simulated gastric and intestinal juices
Aliquots of Δ12-PGJ3 (0.5 µg) in ethyl acetate were evaporated to dryness under a stream of N2. The dry residue was dissolved in 1 mL of simulated gastric or intestinal juice (with or without pancreatin) and incubated at 4°C, 25°C and 37°C for different time durations (0, 6 and 12 h). Following incubation, the solutions were extracted with two volumes of hexane-diethyl ether (1∶1) mixture twice after acidification with HCl (6 N) to ∼pH 3.0. The extracts were pooled and evaporated under a stream of N2, re-constituted with mobile phase (acetonitrile: water = 30∶70 v/v, 0.1% v/v acetic acid, 0.01% triethylamine), and analyzed by HPLC.
Analysis by liquid chromatography
Samples were chromatographically analyzed using Dynamax C18 column (10×250 mm; pore size = 300 Å) on a Beckman system Gold® HPLC. The detector was set at 244 nm. A gradient solvent system was applied at a flow rate of 1 mL/min using mobile phase consisting of solvent-A (acetonitrile/water (30∶70 v/v), 0.1% acetic acid, and 0.01% TEA) and solvent-B (acetonitrile (100%), 0.1% acetic acid, and 0.01% TEA). At injection and 10 min thereafter, the mobile phase was at 100% solvent-A. A linear gradient was achieved over a period of 40 min to 60% solvent-B and further continued at that composition for another 10 min. The peak area was calculated and stability of PG’s at each designated time interval was expressed as the percentage of its original concentration (% remaining). All studies were performed in triplicate.
Determination of the half-life of Δ12-PGJ3 in simulated gastric and intestinal juices
The rate constant and half-life for Δ12-PGJ3 was determined using Arrhenius equation (first order kinetics) as given below:
| (1) |
Where, [Ao] and [A] are the initial and final concentration of the compound, ê(−kt) is the exponential product of rate constant (k) and time (t).
| (2) |
Where, ‘t1/2’ is the amount of time needed for a reactant concentration to decrease by half compared to its initial concentration at any given condition and ‘k’ is the reaction rate constant.
Ethics Statement
All animal work reported here has been conducted according to United States Animal Welfare Act (Public Law 99–198). These studies were pre-approved by IACUC (#40679 and #2013–0014) at The Pennsylvania State University and Cornell University, respectively. Accordingly the authors have taken all steps to ameliorate suffering.
Bioavailability of Δ12-PGJ3 in Mice
Δ12-PGJ3 (0.025 mg/kg body weight) was intraperitoneally injected into C57BL/6 mice (6–8 month old, Charles River Laboratories International, Inc., USA). Post 6, 12, 24 and 48 h of injection, blood (500 µL) was collected in an EDTA coated collection vial (CAPIJECT®, Terumo Medical Corp, USA) by retro-orbital venous puncture. A group comprising of PBS injected mice were designated as vehicle control. The plasma was separated by centrifugation at 1200 xg for 15 min and Δ12-PGJ3 was extracted by using a Sep-Pak® C18 classic cartridge (Waters corporation, USA). Briefly, plasma (100 µL) was diluted to 2 mL using PBS and acidified with 6 N HCl to pH 3. The C18 cartridge was cleaned with methanol (5 mL) followed by 5 mL of PBS and then plasma was passed through twice. The column was washed with PBS and hexane (5 mL each) and the bound PGs were eluted with methanol (5 mL). This fraction was evaporated to dryness under a stream of N2 and re-dissolved with 100 µL solvent-A and analyzed by LC-MS/MS as described below.
LC-MS/MS-MRM Analysis
The HPLC system consisted of LC-20AD UFLC pumps with a SIL-20AC autosampler (Shimadzu Corporation, Columbia, MD). A Luna (Phenomenex, Torrance, CA) phenyl-hexyl analytical column (2×150 mm, 3 µm) developed with a 30 min isocratic elution with methanol/water (70∶30 v/v) containing 0.1% acetic acid at a flow rate of 150 µL/min was used for the quantitation of Δ12-PGJ3. The injection volume was 50 µL. Negative ion electrospray tandem mass spectrometric analysis was carried out using API 2000 triple quadruple mass spectrometer (AB Sciex, Foster city, CA) at unit resolution with multiple reaction-monitoring mode (MRM). The source temperature was at 450°C, electrospray voltage was −4500 V and the declustering potential was set at −16 V. Nitrogen was used as collision gas at −20 eV and the dwell time was 150 ms/ion. During MRM, Δ12-PGJ3 was measured by recording the signal for the transition of the deprotonated molecule of m/z 331 to the most abundant fragment ion with m/z 269. Data were acquired and analyzed using Analyst software program version 1.5 (AB Sciex, Foster city, CA).
In vitro Testing for Drug Hypersensitivity using Murine Bone Marrow-derived Mast Cells (BMMCs)
a) Culturing murine BMMCs
BMMCs were cultured as described previously with some modifications [13]. Briefly, femoral marrow was extracted from C57BL/6 mice (∼4 month old) and cultured in DMEM supplemented with 10% FBS (HyClone, Logan, UT), 100 U/mL penicillin, 100 µg/mL streptomycin, 100 µM non-essential amino acids, 1 mM sodium pyruvate, 2 mM glutamine (Invitrogen, Grand Island, NY), 50 µM 2-mercaptoethanol (Sigma-Aldrich, St. Louis, MO) and recombinant murine IL-3 (rmIL-3; 20 ng/mL; Peprotech, Rocky Hill, NJ). Cells were passaged every three days in fresh medium and used for experiments after 5–6 weeks. The mature BMMCs were stained with toluidine blue and assessed histochemically for the maturity of mast cells and found to be greater that 95%. These cells were utilized for the experiments described below.
b) Cytological analysis
BMMCs (1×106 cells/mL) cultured in growth media were treated for 6 h and 12 h with PBS, Δ12-PGJ3 (0.1 µM), or ZK118182 (0.1 µM). As a positive control, ionomycin (1 µM) was added to the cells for 1 h. Following incubation, the cells were fixed on glass slides using methanol for 30 min followed by staining with toluidine blue for 5 min, rinsed with Milli-Q water, and examined microscopically for any signs of degranulation.
c) Histamine release assay
The supernatant and cell lysates from BMMCs subjected to various treatments (as described above) were used in the quantitation of histamine using the histamine EIA kit. The amount of histamine released was expressed as the ratio of percent of histamine released in the supernatant compared to the intracellular histamine content.
d) Analysis for inflammatory markers by real-time PCR
Total RNA from treated BMMCs (as described above) was isolated using Trizol reagent according to manufacturer’s instructions (Life Technologies, Grand Island, NY). Reverse transcription-PCR was performed using 1 µg of total RNA with random primers using the High Capacity cDNA Reverse Transcription Kit (ABI- Life technologies, Grand Island, NY) in a PTC-100 thermal cycler (MJ Research Inc, Waltham, MA) as per the manufacturer’s protocol. Real-time PCR was performed using Taqman probes for interferon-γ (IFN-γ), TNF-α, IL-13, and IL-4 (ABI-Life Technologies, Grand Island, NY). Real-time PCR was performed according to manufacturer’s instructions using PerfeCTa® qPCR kit (Quanta biosciences, Gaithersburg, MD) on ABI 7300 real-time PCR (ABI- Life Technologies, Grand Island, NY). Results were expressed as 2−▵▵CT that is the expression of target gene relative to the house-keeping gene (GAPDH) and normalized to the negative control (PBS treated cells). All studies were performed in triplicate.
In vitro Testing for Drug Hypersensitivity in Mice
a) Acute and chronic drug toxicity test
Mice (C57BL/6, 6–8 months old) were injected intraperitoneally with Δ12-PGJ3 (0.025 mg/kg body weight) or sterile PBS (300 µL) and euthanized after 6 h or 12 h of injection. The lung tissues were extracted, fixed, sectioned, and stained with hematoxylin and eosin (H&E) for histological examination. Furthermore, inflammatory gene expression in alveolar tissue was also examined using RT-PCR as described earlier and histamine levels in the serum measured using histamine EIA kit as described earlier.
In a separate experiment, mice were treated with Δ12-PGJ3 and ZK118182 at two doses (0.025 mg/kg body weight/day and 0.050 mg/kg body weight/day) intraperitoneally for two weeks, following which drug hypersensitivity response was determined by evaluating the histamine levels in the plasma. In addition, a complete blood count (CBC) and tests for blood urea nitrogen (BUN), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) were carried out at the Centralized Biological Laboratory, The Pennsylvania State University, University Park, PA. Cytokine analysis was carried-out with the plasma using a mouse Th1/Th2 9-plex ultra-sensitive multi-spot cytokine panel (Meso Scale Discovery, Gaithersburg, MD). Lung tissues from the treated mice were extracted, fixed, sectioned, and stained with H&E for histological examination.
b) Measurement of Airways Hyperresponsiveness (AHR)
Mice (C57BL/6, 6–8 weeks old) were injected intraperitoneally with Δ12-PGJ3; ZK118182 at two doses (0.025 mg/kg body weight/day and 0.050 mg/kg body weight/day) or sterile PBS (300 µL) for seven days. After twenty-four hours of final challenge, AHR was analyzed using FlexiVent apparatus (SCIREQ USA Inc., Tempe, AZ) as described [14], [15]. Briefly, airway hyperresponsiveness was measured using a mechanical ventilator apparatus in response to PBS or increasing doses of aerosolized sympathomimetic methacholine from 1 mg/mL to 100 mg/mL. The results were plotted as function of respiratory system resistance (Rrs) values (cm H2O/ml/sec) versus methacholine concentration. Subsequent to AHR measurements, real-time PCR was carried-out on the alveolar tissue to analyze the expression of inflammatory genes.
Statistics
All results are expressed as mean ± SEM. An un-paired two-tailed t-test was used to compare the mean for each treatment group with the mean of the control group and one-way ANOVA (Tukey multiple comparison method) or two-way ANOVA was performed in order to compare various treatment groups within in vivo studies using GraphPad Prism 5.0 program (GraphPad software Inc., San Diego, CA). p values ≤0.05 were considered as statistically significant (*p≤0.05; **p≤0.01; ***p≤0.001).
Results
Effect of Temperature and pH on the Stability of Δ12-PGJ3 under Simulated Conditions
Analysis of extraction efficiency of Δ12-PGJ3 from gastric and intestinal juice (with or without pancreatin) indicated the recovery in the range of 60–80% at t = 0. A time-dependent reduction in Δ12-PGJ3 levels was seen in both simulated gastric and intestinal juice (Fig. 1 A–C) upon incubation at 4°C, 25°C, and 37°C.
After 12 h of incubation at 4°C, Δ12-PGJ3 levels were reduced to ∼50% in the presence of gastric and intestinal juice (−pancreatin). However, addition of pancreatin led to a further decrease in the extractable Δ12-PGJ3 with only a <25% final recovery. At 25°C, ∼50% of Δ12-PGJ3 was recovered after 12 h incubation in gastric juice, but only <5% was recovered upon incubation with intestinal juice (+/− pancreatin). Incubation at 37°C resulted in ∼30% recovery of Δ12-PGJ3 after 12 h incubation in gastric juice and <5% recovery in intestinal juice (−pancreatin). However, after 6 h of incubation, >50% loss was observed in gastric juice, which further reduced to ∼10% at the end of 12 h. In the case of Δ12-PGJ3 incubated in intestinal juice (without pancreatin) at t = 6 h, there was <25% loss in concentration and a significant reduction was observed at the end of 12 h with only a <5% recovery. Addition of pancreatin to the intestinal juice had a significant impact on the stability of Δ12-PGJ3 with no extractable free Δ12-PGJ3 after 6–12 h of incubation at 37°C. Thus, our results suggest that the oral route of administration may be least effective for Δ12-PGJ3.
Based on stability studies of Δ12-PGJ3, it was found that under given conditions the rate constant and half-life did not change with time and hence they were considered to follow first-order kinetics of degradation (data not shown). The half life of Δ12-PGJ3 was compared in simulated gastric and intestinal juice (in the presence or absence of pancreatin) at three separate temperatures, 4°C, 25°C, and 37°C. The kinetics of degradation of Δ12-PGJ3 was faster at higher temperatures and under increasing pH (gastric to intestinal). At 37°C, the t0.5 in gastric juice, intestinal juice (−pancreatin), and intestinal juice (+pancreatin) were 5.5, 3.3, and 1 h, respectively (Table 1). On the other hand, Δ12-PGJ3 was relatively more stable at 4°C. Furthermore, a change in pH towards basic conditions increased the degradation of Δ12-PGJ3 indicating poor stability in the presence of pancreatic enzymes (Table 1).
Table 1. Calculated half-life (t1/2) and rate constant (k) for Δ12-PGJ3 #.
| Incubated temperatures | |||
| Samples | 4°C | 25°C | 37°C |
| Gastric juice | 13.1 h [1.47E-05] | 7 h [2.76E-05] | 5.5 h [3.52E-05] |
| Intestinal (−Pancreatin) | 9.7 h [1.97E-05] | 3.5 h [5.45E-05] | 3.3 h [5.75E-05] |
| Intestinal (+Pancreatin) | 5.9 h [3.26E-05] | 1.2 h [8.56E-05] | 1 h [8.9E-05] |
Half-life (t1/2) and Reaction rate constant (k) (indicated in square brackets) were calculated based on Arrhenius equation-first order kinetics for Δ12-PGJ3 at various incubated temperatures and simulated physiological conditions.
Bioavailability of Δ12-PGJ3 upon Intra-peritoneal Administration
Bioavailability is described as the amount of “unmodified free drug” that is present in the systemic circulation when introduced through non-systemic routes, including intraperitoneal route of administration. In the present study, we used a dose of 0.025 mg/kg/day of Δ12-PGJ3 that was previously reported to be effective in targeting LSCs in two in vivo models of leukemia [7]. Here, we examined the systemic levels of Δ12-PGJ3 for up to 48 h following a single intra-peritoneal administration. Assuming that the Δ12-PGJ3 was uniformly absorbed by mice (that were strain, sex, and age-matched) and that average total volume of blood in each mouse was about ∼8% of its body weight [16], we determined the extractable amount of Δ12-PGJ3 in the plasma in mice as a function of time. LC-MS/MS-MRM analysis indicated that upon 12 h of injection of 0.025 mg/kg, only ∼45% of the total Δ12-PGJ3 was found in systemic circulation with a Cmax of 263 µg/L that decreased to baseline levels by 48 h (Fig. 1D). These results indicate that Δ12-PGJ3 was stable and well absorbed into systemic circulation when injected intra-peritoneally. The peak plasma concentrations of Δ12-PGJ3 were significantly higher than the IC50 of 7–10 nM for LSC apoptosis [7].
Treatment of BMMCs with Δ12-PGJ3 does not Increase Histamine Production
a) Cytological examination of BMMCs upon treatment with Δ12-PGJ3
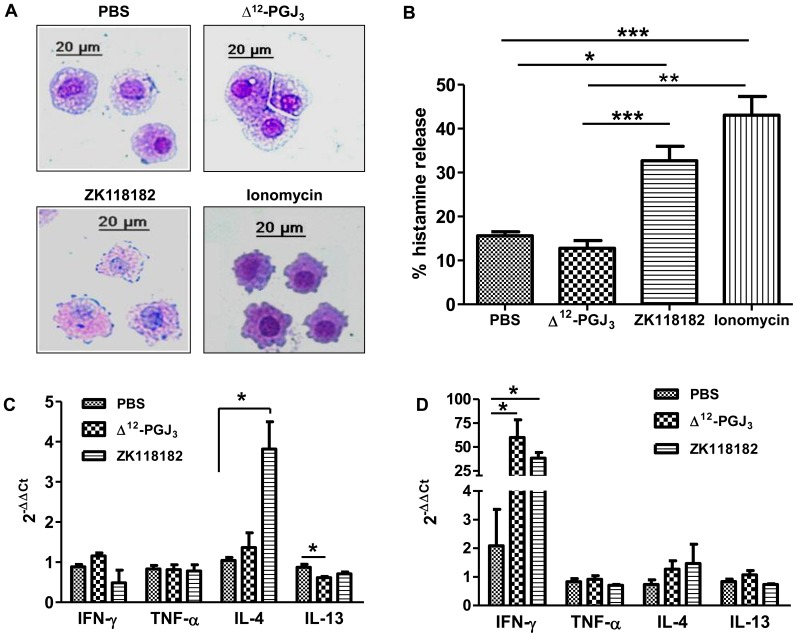
Primary BMMCs treated with PBS or Δ12-PGJ3 (0.1 µM) for 6 h and 12 h showed uniform presence of metachromatic granules when stained with toluidine blue with no morphological changes. However, BMMCs treated with ZK118182 showed prominent loss in cellular integrity with degranulation upon 6 h and 12 h of treatment (Fig. 2A). As a control, ionomycin treatment for 1 h led to extensive degranulation and loss in cellular integrity.
Figure 2. In vitro effect of Δ12-PGJ3 and ZK118182 on BMMCs.
(A) Cytological evaluation for drug hypersensitivity upon treatment of BMMCs with Δ12-PGJ3 and ZK118182. Cultured BMMCs (1×106 cells/mL) were treated with Δ12-PGJ3 and ZK118182 at 0.1 µM for 12 h, followed by fixation and staining with toluidine blue. BMMCs treated with ionomycin (1 µM) for 1 h was used as a positive control. All treatments were carried out in triplicate and a representative image is shown (magnification: 32X). (B) The cells and media treated as mentioned above were used for the estimation of histamine (total and released) using EIA method. (C–D) Real time PCR analysis was carried-out for pro-inflammatory gene markers upon treatment of BMMCs with PBS, ΔΔ12-PGJ3 and ZK118182 as mentioned above (C-6 h; D-12 h). The data shown are mean ± SEM (n = 3) and statistical significance are represented as *- p≤0.05; **- p≤0.01; ***- p≤0.001 respectively.
b) Histamine release assay
Histamine release assay was carried out on the total BMMC lysates as well as in the corresponding culture media supernatants in the above treatments (Fig. 2B). The results were calculated as a percent of histamine released into the culture media, taking into account the intracellular histamine content. Treatment of BMMCs with Δ12-PGJ3 and ZK118182 for 6 h failed to show any significant increase in histamine release when compared to PBS-treated group. However, incubation of BMMCs with ZK118182 for 12 h induced a ∼3.5 fold increase in histamine release into the supernatant, while Δ12-PGJ3 and PBS treatments failed to induce any response. Ionomycin treatment led to ∼4.5 fold increase in histamine release compared to PBS-treated cells.
c) Real-time PCR analysis
Real-time PCR was carried out to analyze expression of IFN-γ, TNF-α, IL-4 and IL-13 upon treatment with PBS, Δ12-PGJ3 (0.1 µM), or ZK118182 (0.1 µM). Treatment with ZK118182 for 6 h increased IL-4 by ∼4 fold (Fig. 2C). However, no significant changes were seen in the expression of IFN-γ, TNF-α, and IL-13 by Δ12-PGJ3 or ZK118182 when compared to PBS control. Interestingly, upon 12 h of treatment, a significant increase in IFN-γ expression was noted in Δ12-PGJ3 (∼60 fold) and ZK118182 (∼40 fold) treated cells (Fig. 2D).
Toxicological Evaluation of Δ12-PGJ3 in vivo
a) Acute and chronic drug toxicity tests in mice
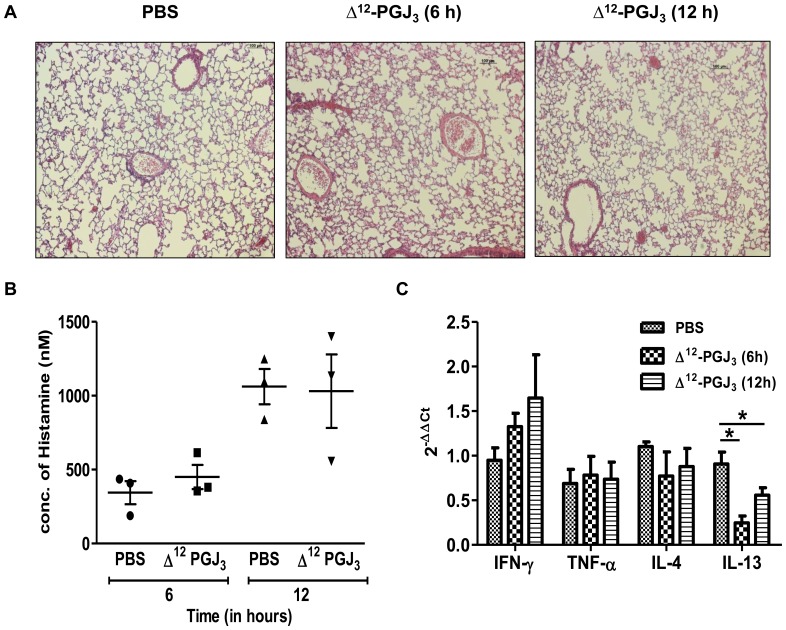
Histological examination was conducted on the alveolar tissue of mice treated with Δ12- PGJ3 for 6 h and 12 h (Fig. 3A). Blinded H&E sections were scored based on the infiltration of PMNs and macrophages, presence of peribronchiolar and perivascular lymphoid aggregates, airway hyperplasia, and necrosis. Based on these criteria, examination of tissues indicated no significant changes in the gross pathology upon treatment with Δ12-PGJ3 (data not shown). In addition, no significant changes in the histamine content in the plasma of Δ12-PGJ3 treated animals were seen when compared to the PBS-treated control mice (Fig. 3B). Furthermore, real-time PCR-based expression analysis of the alveolar tissue indicated a time-dependent increase in IFN-γ, while IL-4 and IL-13 were decreased and TNF- α was not affected upon treatment with Δ12-PGJ3 (Fig. 3C).
Figure 3. In vivo evaluation for hypersensitivity by acute toxicity test.
(A) Δ12-PGJ3 (0.025 mg/kg body weight) was injected intraperitoneally into C57BL/6 mice. Post 6 h and 12 h the mice were sacrificed and the lungs extracted, fixed and stained by H&E. Mice treated with PBS for 12 h was used as a placebo control. All treatments were carried out in triplicates and a representative image has been shown (magnification: 10X). (B) Histamine release assay was carried out on the plasma from the mice treated as mentioned above by EIA method. (C) A real-time PCR analysis was carried out for pro-inflammatory gene markers upon treatment of mice with PBS and Δ12-PGJ3 as mentioned above. The data shown are mean ± SEM (n = 3) and statistical significance are represented as *- p≤0.05.
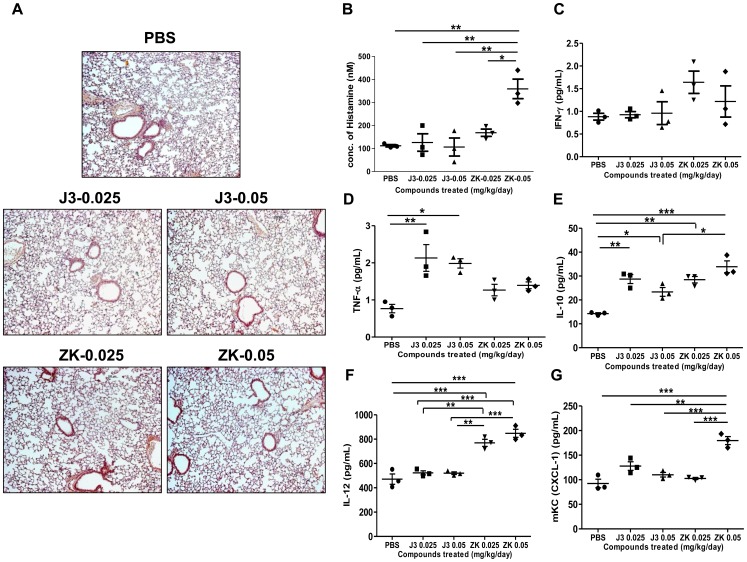
In the second set of mice that were treated with multiple doses (long-term treatment for 14 days) of Δ12-PGJ3, CBC analysis revealed no significant changes in the levels of WBCs and RBCs as compared to initial levels, where ZK118182 was used for comparison (Fig. S1a–d). However, a significant increase in platelet count was observed in Δ12-PGJ3 treated mice (Fig. S1e–f). A single slide chemical analysis of the serum derived from the mice was performed as an indicator to gauge the performance of vital organs. The results revealed no significant deviation in BUN levels between the control and test groups and were found to be within the average range of 18–22 mg/dL (Fig. S1g). In addition, liver function tests for AST activity showed no significant differences between the test groups and the vehicle control; however Δ12-PGJ3 and ZK118182 treatment led to decreased levels of ALT activity in the serum suggesting lack of any overt hepatotoxicity (Fig. S1 h–i). Furthermore, tests were performed to evaluate the histamine levels in the plasma (Fig. 4B), which indicated that the control group and 12-PGJ3 (0.025 and 0.05 mg/kg/day) treated mice produced ∼100 nM of histamine post 24 h of final injection. However, mice treated with ZK118182 (0.025 mg/kg/day) produced ∼175 nM of histamine. Furthermore, mice treated with 0.05 mg/kg/day of ZK118182 nearly doubled the level of histamine (∼375 nM). Plasma cytokine analysis using a 9-plex multi-spot array kit showed a significant increase in IL-10, IL-12, and mKC (CXCL-1) upon treatment of mice with ZK118182 (at 0.025 mg/kg/day and 0.05 mg/kg/day) (Fig. 4E–G); however, treatment with Δ12-PGJ3 (at doses 0.025 mg/kg/day and 0.05 mg/kg/day) slightly elevated the levels of TNF-α, but a significant increase in IL-10 when compared to PBS treated mice was noticed (Fig. 4D–E). There was no change in the levels of IFN-γ as compared to the PBS treated mice (Fig. 4C). The histological examination of the alveolar tissues of treated mice showed no significant changes in the score (data not shown) when compared to the PBS control (Fig. 4A).
Figure 4. In vivo evaluation for hypersensitivity by chronic toxicity test.
(A) Δ12-PGJ3 (0.025 and 0.05 mg/kg body weight) and ZK118182 (0.025 and 0.05 mg/kg body weight) were injected intraperitoneally into C57BL/6 mice. Post two weeks of treatment, the alveolar tissue was extracted, fixed, and stained with H&E. Mice treated with PBS were used as placebo control. All treatments were carried out in triplicates and a representative image has been shown (magnification: 10X). (B) Histamine release assay was carried out on the plasma from the mice treated as mentioned above by EIA method. (C–G) Multi-array Th1/Th2 cytokine analysis was carried-out using the plasma, upon treatment of mice as mentioned above. The figures shown are for: (C) IFN-γ, (D) TNF-α, (E) IL-10, (F) IL-12 and (G) mKC (CXCL-1). The data shown are mean ± SEM (n = 3) and statistical significance are represented as *- p≤0.05; **- p≤0.01; ***- p≤0.001 respectively.
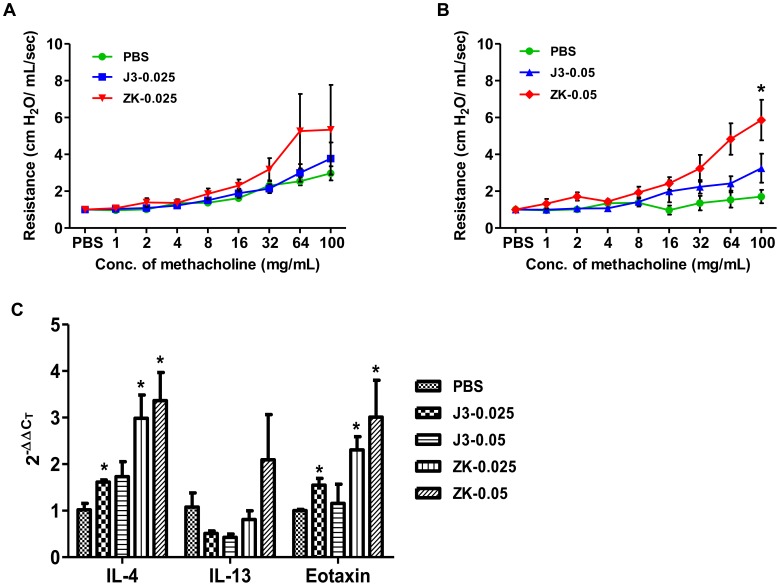
b) Analysis of Airways Hyperresponsiveness (AHR) in mice
An AHR experiment was conducted to examine any physiological changes in the respiratory function upon treatment of mice with Δ12-PGJ3 and ZK118182 (both at 0.025 mg/kg/day and 0.05 mg/kg/day). After seven days of treatment with the two compounds and placebo control (PBS), mice were examined for airway sensitivity by methacholine challenge. Mice treated with ZK118182 at 0.025 mg/kg/day and 0.05 mg/kg/day showed higher AHR when compared to PBS and Δ12-PGJ3 treated mice (Fig. 5A–B). Surprisingly, the AHR response was nearly similar in the groups treated with 0.025 mg/kg/day and 0.05 mg/kg/day of ZK118182. Real-time PCR analysis of the alveolar tissue from the above experiment for the expression of Th2 genes indicated that upon seven days of treatment with ZK118182 (0.025 mg/kg/day and 0.05 mg/kg/day), a ∼3–4 fold increase in levels of IL-4 and eotaxin was seen; while a two-fold increase in IL-13 (with 0.05 mg/kg/day) was found as compared to the PBS control (Fig. 5C). However, administration for Δ12-PGJ3 for seven days led to significant reduction in the levels of IL-13 by 2-fold (with 0.025 mg/kg/day and 0.05 mg/kg/day) with a ∼1.5 fold increase in IL-4 (with 0.025 mg/kg/day and 0.05 mg/kg/day) and eotaxin (with 0.025 mg/kg/day) as compared to PBS control.
Figure 5. Airways hyperresponsiveness test.
Δ12-PGJ3 and ZK118182 was injected intraperitoneally into C57BL/6 mice at (A) 0.025 mg/kg body weight/day and (B) 0.050 mg/kg body weight/day. Post seven days of administration, the AHR response was determined upon methacholine challenge. (C) Real-time PCR analysis was carried-out for expression of Th2 cytokines in the alveolar tissue of mice that were subjected to AHR analysis. The data shown here are mean ± SEM (n = 3) and statistical significance were calculated in comparison to (PBS) control and represented as *(p≤0.05).
Discussion
Recent studies from our laboratory have demonstrated the ability of EPA-derived Δ12-PGJ3, to specifically eradicate LSCs in two models of leukemia [7]. To further expand on these results and explore the therapeutic potential of Δ12-PGJ3 as an anti-leukemic drug, we evaluated the stability, bioavailability, and hypersensitivity of this novel EPA-derived CyPG. The stability of Δ12-PGJ3 was evaluated under simulated conditions using artificial gastric juice and also in intestinal juice in the presence or absence of pancreatin. Our study based on HPLC profiling and half-life calculations indicated that the drug was stable under acidic conditions (gastric juice) and lower temperatures (4°C) for at least 6 h. However, prolonged incubation in intestinal juice (basic conditions) significantly decreased the stability of Δ12-PGJ3. Addition of pancreatin mixture in the intestinal juice further enhanced the reduction in the stability of Δ12-PGJ3. Our studies indicated that Δ12-PGJ3 followed first-order kinetics of degradation, where the rate of reaction was independent of initial concentration of the drug with an exponential reduction in concentration with time. Thus, from a therapeutic dosing point of view, oral administration of Δ12-PGJ3 appears to be the least favorable route of administration suggesting chemical modifications and/or formulation in a suitable system may likely mitigate this issue.
To evaluate bioavailability of Δ12-PGJ3, intra-peritoneal mode of administration was preferred given its efficacious ablation of LSCs in the in vivo models at a dose of 0.025 mg/kg/day [7]. The rate of absorption of drug upon intraperitoneal injection was found to be slow but steady with a plasma peak at 12 h post administration that accounted for ∼45% of the injected drug in systemic circulation. This result not only shows that Δ12-PGJ3 is well absorbed into systemic circulation, but also indicates its stability in vivo. A serum Cmax of 263 ng/mL was achieved at 12 h that was high enough to cause apoptosis of LSCs despite the likelihood of being metabolized by reduction or conjugation as reported for 15-deoxy-Δ12,14-PGJ2 [7], [17].
ARA-derived PGD2 has been shown to play an important role in the pathogenesis of asthma and allergy via the activation of DP1 and CRTH2 (DP2) receptors [18], [19], and the fact that Δ12-PGJ3 shares structural similarity with Δ12-PGJ2, a product of PGD2 that is also known to bind to DP receptors [8], we evaluated the drug-induced hypersensitivity of Δ12-PGJ3 in various in vitro and in vivo models. Although we do not have any data to demonstrate Δ12-PGJ3 to act through the DP receptor per se, we predict that Δ12-PGJ3 could also activate the DP receptors. Therefore, we compared Δ12-PGJ3 with a potent DP-agonist ZK118182 for their ability to cause degranulation of primary BMMCs. Unlike ZK118182, Δ12-PGJ3 failed to induce any adverse reaction that corroborated with the lack of membrane blebbing or metachromatic changes as in the PBS control. Furthermore, treatment of BMMCs for 12 h with ZK118152, but not Δ12-PGJ3, significantly increased histamine production, which is consistent with the cytological observations. The reason for such a differential effect is not clear and is currently being investigated in our laboratory.
The lack of cellular response to Δ12-PGJ3 in BMMCs was further corroborated by in vivo studies that examined drug-induced hypersensitivity with a single dose or a two-week treatment (acute or chronic drug toxicity test). Along the same lines, histological, hematological, kidney- and liver-function tests suggested that Δ12-PGJ3 behaved differently from ZK118182. There were no significant changes in the levels of RBC and WBC upon long-term treatment (14 days) with Δ12-PGJ3 or ZK118182 when compared to untreated control. However, treatment with Δ12-PGJ3 led to a significant increase in levels of platelets without any apparent cardiotoxicity. Though the mechanism is unknown, a previous report indicated that the arachidonic acid derived 15d-PGJ2 increased platelets during megakaryopoesis at concentrations two-orders of magnitude higher than that is used here, and it is very likely that increasing platelet counts may be beneficial in treating thrombocytopenic patients [20]. The serum BUN and AST levels in the Δ12-PGJ3 or ZK118182 were found to be similar to that of vehicle control. However, decreased ALT activity was seen in Δ12-PGJ3 and ZK118182 groups compared to the PBS control. Since increased serum ALT activity is marker for liver injury, lack or decrease in ALT activity may be interpreted as lack of any overt hepatotoxicity. Multiplex cytokine analysis of the plasma of mice treated with ZK118182 for two weeks showed significant increase in production of IL-10, IL-12 (total) and mKC (CXCL-1). This may indicate a regulatory mechanism that could be controlling the pro- and anti-inflammatory cytokine production resulting in a balance. In such a scenario, any imbalance of such a “homeostasis” could result in side effects that have been reported earlier [11], [21]. Δ12-PGJ3, however, did not show any significant increase in any of the Th1/Th2 cytokine levels except for a 2-fold increase in TNF-α without any apparent histological lesions indicative of inflammation. Real-time PCR analysis (after 6 and 12 h treatment) further indicated decreased expression of IL-4 and IL-13 in the alveolar tissue of mice treated with Δ12-PGJ3. Although the role of IFN-γ in asthma and allergies is controversial [22], the fact that Δ12-PGJ3 treatment had no effect on IFN-γ (cytokine) production in mice, further suggests that this EPA metabolite may lack adverse responses. Along these lines, a recent report has shown that intake of fish oil can attenuate the classical allergen-induced airway inflammation and hyperreactivity in mice, which further supports our observation [23].
In vitro assays with BMMCs treated with ZK118182 showed increased degranulation leading to high histamine production, which corroborated well with the increased airway hyperresponsiveness of ZK118182 and increased expression of IL-4 (at 0.05 mg/kg/day), eotaxin, and IL-13 (both at 0.025 mg/kg/day and 0.05 mg/kg/day). Surprisingly, ZK118182 treatment failed to show any histological lesions in the alveolar tissue sections suggesting functional dysregulation despite active resolution.
In summary, our studies indicate the intraperitoneal route of administration of Δ12-PGJ3 is preferred over oral administration given the poor stability under basic conditions to reach a high enough Cmax in the serum that has been shown to be effective in targeting LSCs in vivo. Δ12-PGJ3 did not induce histamine and IL-4 in primary murine BMMCs, which is in agreement with reduced airways hyperresponsiveness in mice. Studies are currently underway to improve the oral bioavailability and stability of Δ12-PGJ3 for future clinical trials in CML.
Supporting Information
Complete blood count analysis and organ-function tests. PBS (A), Δ12-PGJ3 at (0.025 (B) and 0.050 (C) mg/kg body weight/day) and ZK118182 (0.025 (D) and 0.050 (E) mg/kg body weight/day) were injected intraperitoneally into C57BL/6 mice. Post 0 and 14 days of administration, the blood was collected and analyzed. Panels a, c and e indicate levels of WBC, RBC and platelets at day = 0, respectively. Panels b, d and f indicate levels of WBC, RBC and platelets at day 14, respectively. Panels g-i indicate single slide chemical analyses were carried-out after 14 days of treatments. The blood was collected and plasma analyzed for BUN (g), ALT (h) and AST (i). The data shown here are mean ± SEM (n = 3) and statistical significance represented as *(p≤0.05).
(TIF)
Acknowledgments
We thank Dr. Ramesh Ramachandran for generously permitting us to use the inverted microscope in his laboratory and Animal Diagnostic Laboratory for preparing histology slides.
Funding Statement
The studies were funded by a Public Health Service grant from the National Institute of Health CA175576-01. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Smith WL (2005) Cyclooxygenases, peroxide tone and the allure of fish oil. Curr Opin Cell Biol 17: 174–182. [DOI] [PubMed] [Google Scholar]
- 2. Younger EW, Szabo RM (1986) The stability of prostaglandin E1 in dilute physiological solutions at 37 degrees C. Prostaglandins. 31: 923–927. [DOI] [PubMed] [Google Scholar]
- 3. Bedrick AD, Britton JR, Johnson S, Koldovsky O (1989) Prostaglandin stability in human milk and infant gastric fluid. Biol Neonate 56: 192–197. [DOI] [PubMed] [Google Scholar]
- 4. Pifer DD, Cagen LM, Chesney CM (1981) Stability of prostaglandin I2 in human blood. Prostaglandins 21: 165–175. [DOI] [PubMed] [Google Scholar]
- 5. Straus DS, Glass CK (2001) Cyclopentenone prostaglandins: new insights on biological activities and cellular targets. Med Res Rev 21: 185–210. [DOI] [PubMed] [Google Scholar]
- 6. Fitzpatrick FA, Wynalda MA (1983) Albumin-catalyzed metabolism of prostaglandin D2. Identification of products formed in vitro. J Biol Chem 258: 11713–11718. [PubMed] [Google Scholar]
- 7. Hegde S, Kaushal N, Ravindra KC, Chiaro C, Hafer KT, et al. (2011) Δ12-prostaglandin J3, an omega-3 fatty acid-derived metabolite, selectively ablates leukemia stem cells in mice. Blood 118: 6909–6919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Sawyer N, Cauchon E, Chateauneuf A, Cruz RP, Nicholson DW, et al. (2002) Molecular pharmacology of the human prostaglandin D2 receptor, CRTH2. Br J Pharmacol 137: 1163–1172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Almishri W, Cossette C, Rokach J, Martin JG, Hamid Q, et al. (2005) Effects of prostaglandin D2, 15-deoxy-Δ12,14-prostaglandin J2, and selective DP1 and DP2 receptor agonists on pulmonary infiltration of eosinophils in Brown Norway rats. J Pharmacol Exp Ther 313: 64–69. [DOI] [PubMed] [Google Scholar]
- 10. Ravindra KC, Narayan V, Lushington GH, Peterson BR, Prabhu KS (2012) Targeting of histone acetyltransferase p300 by cyclopentenone prostaglandin Δ12-PGJ2 through covalent binding to Cys(1438). Chem Res Toxicol 25: 337–347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hellberg MR, Conrow RE, Sharif NA, McLaughlin MA, Bishop JE, et al. (2002) 3-Oxa-15-cyclohexyl prostaglandin DP receptor agonists as topical antiglaucoma agents. Bioorg Med Chem 10: 2031–2049. [DOI] [PubMed] [Google Scholar]
- 12. Pettipher R (2008) The roles of the prostaglandin D(2) receptors DP1 and CRTH2 in promoting allergic responses. Br J Pharmacol 153 Suppl 1S191–199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jensen BM, Swindle EJ, Iwaki S, Gilfillan AM (2006) Generation, isolation, and maintenance of rodent mast cells and mast cell lines. Curr Protoc Immunol Chapter 3: Unit 3 23. [DOI] [PubMed]
- 14. Sahu N, Morales JL, Fowell D, August A (2010) Modeling susceptibility versus resistance in allergic airway disease reveals regulation by Tec kinase Itk. PLoS One 5: e11348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ewart S, Levitt R, Mitzner W (1995) Respiratory system mechanics in mice measured by end-inflation occlusion. J Appl Physiol 79: 560–566. [DOI] [PubMed] [Google Scholar]
- 16. Hoff J (2000) Methods of Blood Collection in the Mouse. Lab Animal 29: 47–53. [Google Scholar]
- 17. Brunoldi EM, Zanoni G, Vidari G, Sasi S, Freeman ML, et al. (2007) Cyclopentenone prostaglandin, 15-deoxy-Δ12,14-PGJ2, is metabolized by HepG2 cells via conjugation with glutathione. Chem Res Toxicol 20: 1528–1535. [DOI] [PubMed] [Google Scholar]
- 18. Arima M, Fukuda T (2011) Prostaglandin D2 and Th2 inflammation in the pathogenesis of bronchial asthma. Korean J Intern Med 26: 8–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Luster AD, Tager AM (2004) T-cell trafficking in asthma: lipid mediators grease the way. Nat Rev Immunol 4: 711–724. [DOI] [PubMed] [Google Scholar]
- 20. O’Brien JJ, Spinelli SL, Tober J, Blumberg N, Francis CW, et al. (2008) 15-deoxy-Δ12,14-PGJ2 enhances platelet production from megakaryocytes. Blood 112: 4051–4060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Schulz BG, Beckmann R, Muller B, Schroder G, Maass B, et al. (1991) Cardio- and hemodynamic profile of selective PGD2-analogues. Adv Prostaglandin Thromboxane Leukot Res 21B: 591–594. [PubMed] [Google Scholar]
- 22. Yu M, Eckart MR, Morgan AA, Mukai K, Butte AJ, et al. (2011) Identification of an IFN-gamma/mast cell axis in a mouse model of chronic asthma. J Clin Invest 121: 3133–3143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Bargut TCL, Ferreira TPT, Daleprane JB, Martins MA, Silva PMR, et al. (2013) Fish oil has beneficial effects on allergen-induced airway inflammation and hyperreactivity in mice. PLoS ONE 8: e75059. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Complete blood count analysis and organ-function tests. PBS (A), Δ12-PGJ3 at (0.025 (B) and 0.050 (C) mg/kg body weight/day) and ZK118182 (0.025 (D) and 0.050 (E) mg/kg body weight/day) were injected intraperitoneally into C57BL/6 mice. Post 0 and 14 days of administration, the blood was collected and analyzed. Panels a, c and e indicate levels of WBC, RBC and platelets at day = 0, respectively. Panels b, d and f indicate levels of WBC, RBC and platelets at day 14, respectively. Panels g-i indicate single slide chemical analyses were carried-out after 14 days of treatments. The blood was collected and plasma analyzed for BUN (g), ALT (h) and AST (i). The data shown here are mean ± SEM (n = 3) and statistical significance represented as *(p≤0.05).
(TIF)