Abstract
Surface perception is fundamental to human vision, yet most studies of visual cortex have focused on the processing of borders. We therefore investigated the responses of human visual cortex to parametric changes in the luminance of uniform surfaces by using functional MRI. Early visual areas V1 and V2/V3 showed strong and reliable increases in signal for both increments and decrements in surface luminance. Responses were significantly larger for decrements than for increments, which was fully accounted for by differences in retinal illumination arising from asymmetric pupil dynamics. Responses to both sustained and transient changes of illumination were transient. Signals in early visual cortex scaled linearly with the magnitude of change in retinal illumination, as did subjects' subjective ratings of the perceived brightness of the stimuli. Our findings show that early visual cortex responds strongly to surfaces and that perception of surface brightness is compatible with brain responses at the earliest cortical stages of processing.
The perception of surfaces is fundamental to visual behavior, yet little is known about how they are processed in visual cortex. Early studies suggested that neurons in visual cortex respond strongly to edges but only weakly or not at all to uniform illumination (1). Subsequent work has therefore focused almost exclusively on neural mechanisms of contour processing. Indeed, in most contemporary computational models, visual cortical cells are characterized as filters that are unresponsive to uniform illumination (2, 3). The presence of strong edge responses and weak surface responses in early visual cortex is contrary to the intuition that perception of surface brightness should be mediated by neurons responding strongly to the entire spatial extent of a surface. Theoretical accounts of surface perception have therefore often postulated a processing of “filling in” that mediates creation of surface representations at some level in the visual system (4, 5). However, more recent reports suggest that some cells in primary visual cortex do indeed respond to the luminance of uniform surfaces (6–10). Furthermore, in humans there is a close relationship between perceived brightness contrast and responses in primary visual cortex (11–15), suggesting that other sensations of brightness may also be encoded in primary visual cortex. However, the cortical response function for surfaces of uniform luminance in early visual areas, and its relationship to perceived brightness, has remained uninvestigated.
We therefore sought to address this question by using functional MRI (fMRI) to characterize the relationship between parametric variations in surface luminance, cortical responses, and perceived brightness. Understanding such a relationship is a prerequisite for a complete understanding of the general mechanisms and principles that underlie perception of brightness.
When studying responses to uniform surfaces, it is critical to avoid contamination by contour-related processes. With one exception (10), previous fMRI studies of vision in humans have not disentangled the contribution of contours and surfaces. A surface area and its boundary are mapped to separate cortical locations because the visual field is mapped topographically onto visual cortex (16). However, single cells in visual cortex are known to integrate information over an extended region of the visual field (17); thus, there could be important interactions between regions representing the surface and those representing the border. To ensure that the responses we measured were caused by the local surface alone rather than remote contours, we studied the representation of parts of a surface that were separated from the closest contour by at least 5°. This distance is beyond the influence of boundary processing that has been measured in human V1, V2, and V3 (10).
Methods
We measured brain activity with fMRI in two experiments. A total of nine human volunteers with normal or corrected-to-normal vision (five males, four females, ages between 30 and 36 years) were studied. Subjects viewed a large circular projection screen at the rear of the scanner bore with a mirror positioned within the head-coil. The screen extended from fixation to 13° eccentricity in each direction. With the exception of a small central fixation spot, the display was of constant luminance. The area surrounding the screen was covered with black cardboard, leading to the perception of a large circular (26° diameter) homogenously gray surface on a dark background. The baseline luminance of the surface was 100 cd/m2. Two experiments were performed in which the surface luminance was changed from baseline level for different durations and various amplitudes. When comparing responses of visual cortex to increments and decrements of varying amplitudes, we ensured that the level of retinal adaptation and pupil size were identical by presenting a 15-s adaptation screen of constant luminance before each stimulus.
Experiment 1: Cortical Responses to Increases and Decreases in Surface Luminance. We recorded cortical responses of five subjects to sustained luminance changes. Luminance of the entire visual display was either increased or decreased by 90 cd/m2 for 15 s, after which it returned to baseline luminance. Between repeated increases and decreases, the stimulus was maintained at baseline luminance for 15 s to ensure that responses to increases and decreases were measured relative to identical baselines. Stimulation alternated between two pseudorandom sequences on successive runs. Attentional state was controlled by requiring subjects to detect changes to the small central fixation spot occurring at a mean rate of 0.5 Hz. Subjects responded with a key press. Stimuli were presented by using matlab and cogent 2000 toolbox (www.vislab.ucl.ac.uk/cogent/index.html) running on a computer connected to a NEC LT158 LCD projector. Luminance calibration used a Minolta LS-100 spot photometer. Pupil responses were recorded by using an infrared eye tracker (Applied Science Laboratories, Waltham, MA) with remote optics (model 504, sampling rate 60 Hz) custom-adapted for use in the scanner.
To study surface responses without contamination by contour-related responses, we restricted our analysis to cortical regions representing parts of the visual field that were at least 5° from both the fixation spot and the border of the screen. These small regions of interest (ROIs) were identified in separate localizer runs by using two high-contrast checkerboard patches of 2.5° × 2.5° presented at an eccentricity of 5° left and right from fixation on the horizontal meridian. ROIs on the horizontal meridian were selected to maximally separate the representations in primary and extrastriate visual cortex. To independently identify primary visual cortex (V1) and extrastriate areas, we mapped the horizontal and vertical meridians of the visual field according to standard procedures (18).
A 3T Siemens Allegra system was used to acquire blood oxygenation level-dependent (BOLD) contrast image volumes. Volumes were acquired continuously every 2.08 s, each comprising 32 contiguous 3-mm-thick slices to give coverage of the entire occipital lobes and neighboring temporoparietal cortex with an in-plane resolution of 3 × 3 mm. Functional imaging was performed in four to nine scanning runs, each comprising 136 image volumes. In each scanning run, five image volumes preceding presentation of the experimental conditions were discarded to allow for T1 equilibration effects. Two separate scanning runs were also performed for the localizer and meridian mapping. A localizer checkerboard was contrast inverted at 4 Hz for 6.5 s followed by an 11.5-s rest period, repeating eight times per run. In the meridian mapping runs, checkerboard patterns covering either the horizontal or vertical meridian were alternated with rest periods by using the same timing parameters as in the localizer.
Experiment 2: Cortical Responses to Parametric Changes in Surface Luminance. Next, we recorded brain responses of six subjects to transient luminance changes. The protocol was similar to experiment 1 with differences in timing and amplitude of stimuli. Luminance of the entire screen was increased or decreased for 1,300 ms, after which the luminance returned to baseline for 15 s. The amplitude was varied between 30, 60, and 90 cd/m2. In a single run, a pseudorandomized sequence of all three increments and decrements was presented and then repeated in reverse order. A central fixation task was used to control attentional state, and meridian mapping of visual cortical areas was performed as described above. The same MRI scanner and sequence were used to acquire between 6 and 12 scanning runs per subject, each comprising 109 volumes. The pupil time courses during the entire duration of each stimulus type (1,300 ms) were used to convert image luminance to retinal illumination according to the following formula:
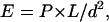 |
where E is retinal illumination, P is pupil area [m2], L is the luminance of the image [lm/m2/sr], and d is the posterior nodal distance (19).
Control Experiments. To rule out the possibility that signals recorded in our ROI were contaminated by signals from the border of the illuminated region, we performed additional control experiments in two subjects. In the first experiment (on/off), a black and white contour (a white ring of 0.5° width and 12.5° radius immediately surrounded by a black ring of 0.5° width and 13° radius) was presented on a background luminance of 100 cd/m2 for 1,300 ms, followed by an interstimulus interval of 15 s. A central fixation task was used to control attentional state, and 288 BOLD contrast volumes were acquired from a single subject. A second control experiment (counterphasing) used a modified version of this stimulus that contrast-inverted during each trial for 3,000 ms at 4 Hz to make the stimulation stronger. In other respects, the display and stimulus geometry and fixation task were identical. For this control experiment, 259 image volumes were acquired from a second subject.
Experiment 3: Psychophysics of Perceived Brightness. To understand how responses in V1 and V2/V3 were related to perceived brightness, we conducted a third, exclusively psychophysical, experiment. Typically, the relationship between perceived brightness and surface luminance can be approximated by a power function, but the exponent of the function can vary strongly and depends critically on stimulus parameters (20). We therefore used exactly the same stimuli as in experiment 2 together with a magnitude estimation procedure. Six subjects with normal vision were presented with either increments or decrements of different luminance. After each stimulus, the subjects were presented with a rating scale comprising 80 small subdivisions on the computer screen. The subjects were instructed to rate the perceived magnitude of the brightness changes by using a mouse and to ensure that equal differences in perceived brightness corresponded to equal distances on the scale. Two repetitions of the entire stimulus set were used for training, during which subjects were made familiar with the stimulus range to avoid floor or ceiling effects. After training, 20 repetitions of the entire set of stimuli were presented in pseudorandomized order. Stimulus duration was 1,300 ms. Increments and decrements were tested in separate sessions. To characterize the relationship between perceived brightness and surface luminance, a power function, was fitted to each subject's brightness ratings, where PB is the perceived brightness rating, RI is the retinal illumination, and a, b, and c are constants.
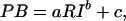 |
Data Analysis. Imaging data were analyzed by using spm2 (www.fil.ion.ucl.ac.uk/spm). For both experiments, each imaging time series was realigned and coregistered to each subject's structural scan by using the mean image of the time series. High-pass filtering removed low-frequency drifts in the signal. Voxels activated by the localizer and meridian stimuli were identified by using a linear model in which the regressors were boxcar waveforms modeling each stimulus category, convolved with a canonical hemodynamic response function. Voxels responding to the localizer stimulus were identified by using a T statistic with a threshold of P < 0.05 (corrected). The V1 ROI was identified as the cluster of activated voxels in the calcarine sulcus of each hemisphere between the upper and lower representation of the vertical meridian. The V2/V3 ROI was identified as the clusters immediately ventral and dorsal to the representation of the vertical meridian. The human homologue of monkey middle temporal area (MT) ROI was identified on anatomical landmarks as the cluster of voxels in the posterior inferior temporal sulcus (21). Each stimulus type was separately modeled by using a series of delta functions convolved with a hemodynamic response function (four regressors in experiment 1 and six regressors in experiment 2). The resulting regression parameters were then extracted for each ROI to yield the plots in Figs. 1B, 2B, and 3.
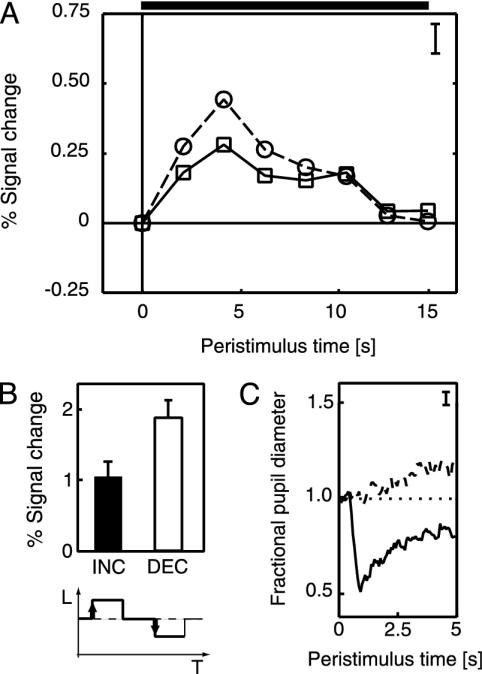
Fig. 1.
Responses in primary visual cortex to sustained (15 s) changes in surface luminance. (A) Time course of fMRI signal in a V1 ROI after sustained increments (solid line, □) and decrements (dashed line, ○) of 90 cd/m2 (the horizontal solid bar indicates the duration of the stimulus; the error bar shows ± 1 SE). The response to the decrement is stronger than the response to the increment. (These and all following fMRI signal changes are plotted as percentage of the mean signal in the voxel of interest.) (B) Percent signal change modeled by a general linear model assuming a canonical hemodynamic response after each luminance change (black, increment; white, decrement; error bars, 1 SE). The response to decrements is again stronger than that to increments. The values for the decrease from 190 cd/m2 to 100 cd/m2 and the increase from 10 cd/m2 to 100 cd/m2 are not directly comparable because they are recorded at different baseline luminances and different states of retinal adaptation. The values are 1.95 (increment 10 cd/m2 to 100 cd/m2) and 0.41 (decrement 190 cd/m2 to 100 cd/m2). (C) Pupil responses. Luminance increments lead to a strong pupil constriction (solid line), whereas decrements have only a weak dilating effect (dashed line). Responses are expressed as a fraction of the mean pupil diameter during a 65-ms prestimulus baseline (error bar indicates ±1 SE).
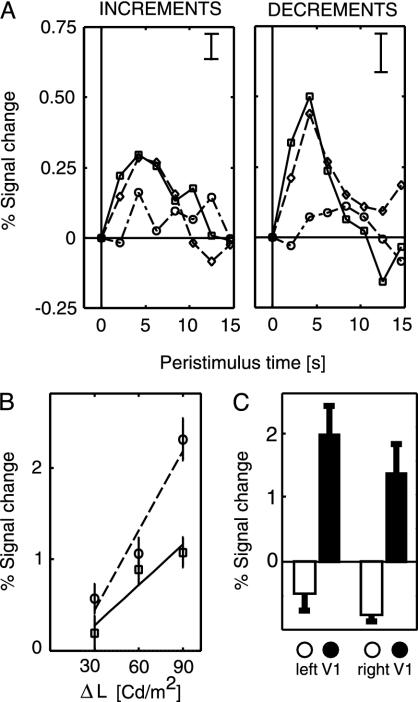
Fig. 2.
Responses in V1 ROI to transient changes in surface luminance of various amplitudes. (A Left) Time course of fMRI signal after luminance increments of +30 cd/m2 (dash-dotted line), +60 cd/m2 (dashed line), and +90 cd/m2 (solid line). (Right) Responses for decrements of the same amplitudes (error bar, ±1 SE). Responses are stronger for increasing amplitudes of the transient changes, and responses to decrements are stronger than those to increments. (B) Percent signal change modeled by a general linear model assuming a canonical hemodynamic response after each luminance change (error bars, ±1 SE). Responses to both increments (□, solid line) and decrements (○, dashed line) show a monotonic increase with the amplitude of the luminance step, the responses to decrements being stronger. The lines show the best fitting linear regression function. The quadratic term of the polynomial expansion was not significant (22). (C) Control experiment with high-contrast contour stimulus contrast reversing at 4 Hz. The filled bars show the mean responses in this subject to a surface stimulus in the left and right V1 ROI at 5° eccentricity. The open bars show the responses to a contour-only stimulus for the same voxels. In comparison to the surface stimulus, the contour stimulus leads to a weak inhibitory response in the ROI.
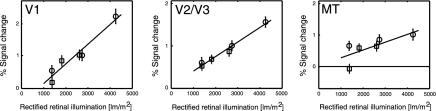
Fig. 3.
(Left) V1 responses from Fig. 2B plotted as a function of retinal illumination. Retinal illumination was computed by taking the measured pupil time course for each stimulus type into account (see Methods). The distinct functions of Fig. 2B collapse into a single linear function relating signal change to the rectified retinal illumination. (Center and Right) Responses in ROI in V2/V3 and MT, quantified by using the same methodology as for V1. In V2, the responses show a linear dependency on retinal illumination, whereas in MT the response is not so well fit with a linear function. The lines show the best fitting linear regression functions (□, increments; ○, decrements).
Results
Primary visual cortex responded reliably to sustained increases and decreases of surface luminance (experiment 1; see Fig. 1 A). Responses were transient and fell to baseline before the end of the sustained phase of luminance change (15 s after onset). The responses were rectified, with a positive BOLD response to both increments and decrements in surface luminance. Surprisingly, the response to increased luminance (from 100 to 190 cd/m2) was weaker compared to the response to a decrease of equal magnitude (from 100 to 10 cd/m2). This difference observed in the raw data (Fig. 1 A) is formally reflected in the modeled response to luminance increments and decrements by using a canonical hemodynamic response function (Fig. 1B). This difference may be partly explained by the asymmetry of the pupil responses (Fig. 1C). The pupil responded to the luminance increases with a strong and rapid constriction, thus counteracting the luminance change. Luminance decrements, on the other hand, led to relatively weak and slow pupil dilation. Qualitatively, the effective change in retinal illumination is much higher in response to a luminance decrease compared to a luminance increase of equal amplitude.
Fig. 2 A shows the response of human V1 to transient increases and decreases of luminance with varying amplitudes of change (experiment 2). This replicates the findings of experiment 1 that responses to both increments and decrements are positive and the responses to decrements are stronger. The time course of the event-related response is very similar to that recorded for sustained luminance changes. Fig. 2B shows the same result as modeled by a canonical hemodynamic response function. The response amplitude increased monotonically with increased luminance changes. Qualitatively, the data show an approximately linear dependency of BOLD signal amplitude on the degree of luminance change. To test whether the slight deviations from linearity apparent in the raw data were significant, we fitted a parametric model by using a polynomial expansion of the luminance steps with a constant, linear, and quadratic component (22). The fit of both constant and linear regressors was highly consistent across subjects with regression coefficients significantly more than 0. However, the quadratic component was fitted inconsistently and with coefficients that were never significantly more than 0. This finding suggests that the response of human V1 to both increments and decrements of luminance is linear. Voxels further anterior in the calcarine sulcus (representing areas outside the surface) showed no significant responses (0.02 ± 0.31 SE) to the surface stimulus, indicating that responses of V1 to changes in surface luminance were restricted to the retinotopic representation of that surface in V1.
Two control experiments assessed responses in our ROI to contours placed at the border of the illuminated region. In the first control experiment (on/off), responses to the contour-only stimulus in our ROI did not significantly differ from zero (left hemisphere, 0.31 ± 0.84 SE; right hemisphere, 0.35 ± 0.81 SE). However, the overall signal-to-noise level was low during this experiment, perhaps because the contour was only flashed once per trial. To rule out the possibility that ROI responses might be seen with a stronger contour stimulus, we conducted a second control experiment (counterphasing) by using a stronger contrast-reversing contour-only stimulus. A weak inhibitory response was observed in the ROI (Fig. 2C). This finding contrasts with the strongly positive response evoked by luminance changes in the surface stimulus. Such observations are compatible with previous demonstrations of negative BOLD responses in cortical regions that represent the surround of a high-contrast contour (23). More importantly, both control experiments indicate that the responses to surface luminance changes we recorded in our ROI cannot be explained by remote effects of the border of the illuminated surface.
Experiment 1 raised the possibility that the observed difference between responses to luminance increments and decrements might be explained by differential pupil responses. We therefore computed the retinal illumination for each stimulus from the pupil responses recorded for each luminance step (19). The cortical response was then replotted as a function of rectified retinal illumination (Fig. 3 Left). It was immediately apparent that this entirely eliminated the previously observed strong differences between increments and decrements in V1. Instead, the cortical response showed an almost identical monotonic linear dependency on the retinal illumination caused by the stimulus for both increments and decrements.
We also characterized responses of extrastriate visual cortex to transient luminance changes. Our visual ROI lay on the horizontal meridian, and so its cortical representation lay on the border between the areas V2 and V3. We therefore identified the responses of this region as V2/V3 (Fig. 3 Center). Responses in V2/V3 showed an almost perfect linear dependency on retinal illumination. Responses in MT also showed a monotonic dependence on the amplitude of retinal illumination with a significant linear component (Fig. 3 Right). However, these data were not fit well with a linear function.
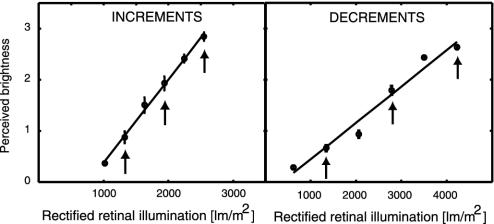
In separate psychophysical sessions, we asked six subjects to rate the perceived brightness of our increment and decrement stimuli. Perceived magnitude judgments for increments showed an almost perfect linear dependency on retinal illumination (Fig. 4). Perceptual judgments for luminance decrements also revealed a linear dependency, particularly for the stimuli used in the scanning session (see arrows in Fig. 4). A power function was fitted to the behavioral data as described above. The average exponent was 1.05 (0.11 SE) for decrements and 1.01 (0.16 SE) for increments. The relationship between surface luminance changes and phenomenal brightness perception was therefore strongly linear, and in this respect it closely resembled the dependence of BOLD contrast responses in V1 and V2/V3 on surface luminance (Fig. 3).
Fig. 4.
Psychophysical perceived magnitude ratings for luminance increments (Left) and decrements (Right). Perceived magnitude of increments showed a linear dependency on retinal illumination, whereas decrements showed only minor deviations from linearity, especially when only the stimuli that were used in the fMRI study (indicated by the arrows) are considered.
Discussion
We found that human visual cortex responded reliably to changes in the luminance of uniform surfaces, when contamination of the result by border processing was excluded. Our two experiments independently demonstrated that the BOLD response to both surface luminance increments and decrements is positive and scales with the magnitude of luminance change. The time courses of responses to both transient and sustained changes in illumination are highly similar and suggest that the responses are determined mainly by a transient stage of processing. Surprisingly, we found that responses to luminance decrements were larger than those to increments. However, we also observed that pupil responses differed in response to luminance increments and decrements. When pupil diameter was taken into account, increments and decrements showed the same linear dependency on retinal illumination. Moreover, this linear dependence of cortical responses on illumination was paralleled by a linear dependence of the phenomenal property of perceived brightness on retinal illumination.
Apart from the present findings, there is a small amount of data concerning the representation of uniform surfaces in human cortex. A study of the spatial summation properties of visual cortex revealed modest responses to large uniform stimuli, consistent with the present findings (10). In another study, luminance-defined rings used as retinotopic mapping stimuli did not evoke modulation of cortical responses at their fundamental frequency (16). However, neither of these studies intended to directly address the question of how surface brightness is represented in visual cortex. Finally, one recent study (reported only in abstract form) suggests that a uniform flickering stimulus set within a surround of different luminance evokes activity in visual cortex only at the stimulus borders and not the center.∥ The authors of that study conclude that primary visual cortex does not encode surface brightness. However, the use of a flickering stimulus may have inadvertently minimized surface-related activity in visual cortex. Regions of visual cortex that represent areas of the visual field that are unstimulated but are in the immediate surround of high-contrast pattern stimuli are inhibited (23), presumably because of lateral propagation of activity along horizontal connections. This process is very slow compared to rapid feed-forward connections (24). We suggest that extended flickering may enable a build-up of lateral inhibitory activity originating from regions processing the surface boundary that opposes the weaker activation caused by surface processing. In contrast, the present study measured cortical responses to single sustained or transient luminance changes, which may therefore be more effective in eliciting surface responses.
We found a linear relationship between fMRI responses and retinal illumination. There is very little information available on how response amplitude scales with luminance in populations of single cells in monkeys and none in humans. The few published examples of individual neurons seem to show a compressive dependency of discharge rate on luminance, with responses saturating for increasingly higher increments (6, 7), which represents a linear dependency on the logarithm of luminance and is in contrast to the present data. However, the precise shape of this function within visual cortex has not been systematically studied, unlike that for contrast stimuli (11, 25). Moreover, the population responses measured with functional imaging represent the average response of a large number of cells, each with slightly different response functions. Our data represent the characterization of how human visual cortical responses change with surface luminance and suggest that the overall population response in both primary visual cortex and higher visual areas is linear.
For stimuli varying in brightness contrast, there is a close correlation between fMRI or magnetoencephalography signals in V1 and the perceived contrast of stimuli (12, 13, 15). Our data show that for the stimuli used in this experiment, responses in V1 and V2/V3 are scaled in a similar way to the perceived brightness function. These cortical areas are therefore possible candidates for representing the dimension of perceived brightness. In contrast, the responses we recorded in MT are incompatible with brightness perception. However, in the present paradigm, perceived brightness and luminance (or retinal illumination) were confounded. To determine with certainty the cortical substrate of perceived brightness perception, it will be necessary to independently manipulate retinal illumination and perceived brightness. The perceived brightness of a surface depends not only on surface luminance but also on the luminance distribution in the surrounding visual field (26, ∥). It has already been demonstrated in cats and monkeys that single neurons in primary visual cortex exhibit a surround modulation that matches contextual effects in human brightness perception (6, 9, 27, 28). By establishing the dynamics of surface-related responses in early visual cortex, our findings therefore provide the fundamental information necessary for the interpretation of future neuroimaging studies of contextual effects in human subjects.
An intriguing aspect of our data is that the BOLD contrast response was positive for both luminance increments and decrements. Such a finding places important constraints on possible physiological mechanisms for signaling the brightness of uniform surfaces. In monkey, the majority of neurons signaling surface luminance respond with either an increase or a decrease in firing to changes in luminance (photergic or scotergic, respectively) (6, 7). The responses in human visual cortex we observed may therefore reflect the activation of two neuronal subpopulations homologous to those observed in monkey, signaling dark and light, respectively. For a net positive population response to luminance increments, the response (or number) of bright-signaling neurons must exceed the response (or number) of dark-signaling neurons, which is the case in monkey primary visual cortex where the ratio of photergic to scotergic neurons is 4:1 (7). However, such a population imbalance in favor of bright-signaling neurons cannot additionally account for our observation of a positive response to luminance decrements. A second, more speculative, possibility is therefore that the rectification we observed reflects the relationship between single cell activity and the BOLD signal. Under some situations, single-cell spiking activity and BOLD responses can be dissociated (29). It has been proposed that this may arise because the BOLD signal largely reflects dendritic processing (local field potentials) rather than axonal spiking. If this is the case, then increases in either excitatory or inhibitory dendritic activity (perhaps reflecting luminance increments and decrements, respectively) might result in increases in BOLD signal, even though the consequences of this activity for axonal spiking would be opposite to each other. However, such an explanation is unlikely, as previous studies have suggested that a decrease in neuronal activity is accompanied by a negative fMRI response (23), which was not observed in our data.
A final possibility is that rectified luminance responses reflect predominantly transient responses of human surface processing cells. In monkey, transient and sustained phases of processing exhibit different response profiles. During the sustained phase, cells almost exclusively show the photergic response profile. However, in the transient phase a large proportion of cells (40%) exhibit biphasic responses that signal the amplitude of both positive and negative deviations from a luminance baseline with increases in response rate (6). This biphasic response profile is rectified in a similar way as our responses to luminance decrements. The rectification we observed in BOLD contrast responses to luminance increments and decrements may therefore directly reflect the biphasic responses of single cells during the transient phase, rather than the responses of different subpopulations of cells specialized for signaling either light or dark. A predominance of transient responses is also supported by the observation that sustained changes in luminance in experiment 1 do not lead to more sustained fMRI responses than the transient changes in experiment 2. We therefore suggest that the responses we observed in human visual cortex to changes in surface luminance reflect the transient responses of brightness-signaling cells homologous to those observed in monkey visual cortex. It would be interesting to further investigate the nature of the transient response in a future study. Slowly ramping the stimulus on and off could reveal whether the high rate of change of illumination at stimulus onset is responsible for the transient response.
Finally, in addition to the neurobiological significance of our findings, our experiments reveal an important methodological issue that may have wider implications for fMRI studies using visual stimuli. We observed that pupillary responses to luminance increments and decrements were highly asymmetric (Fig. 1C), which resulted in physical manipulations of the stimulus that were of equal magnitude having unequal effects on retinal illumination. Specifically, weak pupillary dilatation to luminance decrements resulted in stronger overall retinal illumination changes than for equally large luminance increments. As a result, the BOLD signal for physical changes of equal magnitude was almost doubled because of the differential retinal illumination (see Figs. 1B and 2B). This finding suggests that experiments measuring BOLD signals to visual stimuli varying in luminance should consider measuring pupil size to eliminate the possibility of stimulus-driven confounds arising from asymmetric pupillary responses.
Conclusions
Human primary visual cortex and early extrastriate areas showed responses to uniform surfaces that systematically scaled with the magnitude of surface luminance change. These population responses may reflect transient biphasic neuronal responses homologous to those observed in monkey. Moreover, the linear cortical response to surface luminance was closely reflected in psychophysical measurements of perceived brightness.
Acknowledgments
This work was funded by the Wellcome Trust and facilitated by the Medical Research Council Cooperative for the Analysis of Cognitive Impairment and Imaging of Cognition at University College London.
This paper was submitted directly (Track II) to the PNAS office.
Abbreviations: fMRI, functional MRI; ROI, region of interest; BOLD, blood oxygenation level-dependent; MT, human homologue of monkey middle temporal area.
Footnotes
Cornelissen, F., Wade, A. R., Dougherty, R. F. & Wandell, B. A. (2003) J. Vision 3, 57a (abstr.).
References
- 1.Hubel, D. H. & Wiesel, T. N. (1968) J. Physiol. (London) 195, 215-243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Daugman, J. G. (1985) J. Opt. Soc. Am. A 2, 1160-1169. [DOI] [PubMed] [Google Scholar]
- 3.Itti, L., Koch, C. & Braun, J. (2000) J. Opt. Soc. Am. A 17, 1899-1917. [DOI] [PubMed] [Google Scholar]
- 4.Cohen, M. A. & Grossberg, S. (1984) Percept. Psychophys. 36, 428-456. [DOI] [PubMed] [Google Scholar]
- 5.Pessoa, L. & De Weerd, P. (2003) Filling-In: From Perceptual Completion to Cortical Reorganization (Oxford Univ. Press, Oxford).
- 6.Kinoshita, M. & Komatsu, H. (2001) J. Neurophysiol. 86, 2559-2570. [DOI] [PubMed] [Google Scholar]
- 7.Kayama, Y., Riso, R. R., Bartlett, J. R. & Doty, R. W. (1979) J. Neurophysiol. 42, 1495-1517. [DOI] [PubMed] [Google Scholar]
- 8.MacEvoy, S. P., Kim, W. & Paradiso, M. A. (1998) Nat. Neurosci. 1, 616-620. [DOI] [PubMed] [Google Scholar]
- 9.Rossi, A. F., Rittenhouse, C. D. & Paradiso, M. A. (1996) Science 273, 1104-1107. [DOI] [PubMed] [Google Scholar]
- 10.Press, W. A., Brewer, A. A., Dougherty, R. F., Wade, A. R. & Wandell, B. A. (2001) Vision Res. 41, 1321-1332. [DOI] [PubMed] [Google Scholar]
- 11.Boynton, G. M., Demb, J. B., Glover, G. H. & Heeger, D. J. (1999) Vision Res. 39, 257-269. [DOI] [PubMed] [Google Scholar]
- 12.Goodyear, B. G., Nicolle, D. A., Humphrey, G. K. & Menon, R. S. (2000) J. Neurophysiol. 84, 1907-1913. [DOI] [PubMed] [Google Scholar]
- 13.Haynes, J. D., Roth, G., Stadler, M. & Heinze, H. J. (2003) J. Neurophysiol. 89, 2655-2666. [DOI] [PubMed] [Google Scholar]
- 14.Zenger-Landolt, B. & Heeger, D. J. (2003) J. Neurosci. 23, 6884-6893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Williams, A. L., Singh, K. D. & Smith, A. T. (2003) J. Neurophysiol. 89, 525-533. [DOI] [PubMed] [Google Scholar]
- 16.Engel, S. A., Glover, G. H. & Wandell, B. A. (1997) Cereb. Cortex 7, 181-192. [DOI] [PubMed] [Google Scholar]
- 17.Fitzpatrick, D. (2000) Curr. Opin. Neurobiol. 10, 438-443. [DOI] [PubMed] [Google Scholar]
- 18.Kastner, S., De Weerd, P., Desimone, R. & Ungerleider, L. G. (1998) Science 282, 108-111. [DOI] [PubMed] [Google Scholar]
- 19.Rodieck, R. W. (1998) The First Steps in Seeing (Sinauer, Sunderland, MA).
- 20.Nundy, S. & Purves, D. (2002) Proc. Natl. Acad. Sci. USA 99, 14482-14487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dumoulin, S. O., Bittar, R. G., Kabani, N. J., Baker, C. L., Jr., Le Goualher, G., Bruce, P. G. & Evans, A. C. (2000) Cereb. Cortex 10, 454-463. [DOI] [PubMed] [Google Scholar]
- 22.Rees, G., Friston, K. & Koch, C. (2000) Nat. Neurosci. 3, 716-723. [DOI] [PubMed] [Google Scholar]
- 23.Shmuel, A., Yacoub, E., Pfeuffer, J., Van de Moortele, P. F., Adriany, G., Hu, X. & Ugurbil, K. (2002) Neuron 36, 1195-1210. [DOI] [PubMed] [Google Scholar]
- 24.Bullier, J. (2001) Brain Res. Rev. 36, 96-107. [DOI] [PubMed] [Google Scholar]
- 25.Albrecht, D. G. & Hamilton, D. B. (1982) J. Neurophysiol. 48, 217-237. [DOI] [PubMed] [Google Scholar]
- 26.Lotto, R. B. & Purves, D. (2002) Trends Neurosci. 25, 84-88. [DOI] [PubMed] [Google Scholar]
- 27.MacEvoy, S. P. & Paradiso, M. A. (2001) Proc. Natl. Acad. Sci. USA 98, 8827-8831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hung, C. P., Ramsden, B. M., Chen, L. M. & Roe, A. W. (2001) Vision Res. 41, 1389-1407. [DOI] [PubMed] [Google Scholar]
- 29.Logothetis, N. K. (2003) J. Neurosci. 23, 3963-3971. [DOI] [PMC free article] [PubMed] [Google Scholar]