Summary
Background and objectives
Greater dietary acid has been associated with lower serum bicarbonate levels in patients with CKD. Whether this association extends to the general population and if it is modified by age are unknown.
Design, setting, participants, & measurements
This study examined the association of the dietary acid load, estimated by net endogenous acid production, with serum bicarbonate levels in adult participants in the National Health and Nutrition Examination Survey 1999–2004.
Results
The mean serum bicarbonate was 24.9 mEq/L (SEM=0.1), and the mean estimated net endogenous acid production was 57.4 mEq/d (SEM=0.4). Serum bicarbonate was linearly associated with age, such that the oldest participants had the highest serum bicarbonate levels. After multivariable adjustment, participants in the highest quartile of net endogenous acid production had 0.40 mEq/L (95% confidence interval, −0.55 to −0.26) lower serum bicarbonate and a 33% (95% confidence interval, 3 to 72) higher likelihood of acidosis compared with those participants in the lowest quartile. There was a significant interaction by age of the association of net endogenous acid production with serum bicarbonate (P=0.005). Among participants 20–39, 40–59, and ≥60 years old, those participants in the highest net endogenous acid production quartile had 0.26 (95% confidence interval, −0.49 to −0.03), 0.60 (95% confidence interval, −0.92 to −0.29), and 0.49 (95% confidence interval, −0.84 to −0.14) mEq/L lower serum bicarbonate, respectively, compared with participants in the lowest quartile.
Conclusion
Greater dietary acid is associated with lower serum bicarbonate in the general US population, and the magnitude of this association is greater among middle-aged and elderly persons than younger adults.
Introduction
Acid–base homeostasis is tightly controlled in normal humans, and even minimal disturbances can have deleterious effects. Chronic metabolic acidosis as a manifestation of CKD is believed to have adverse affects on bone metabolism and skeletal muscle breakdown (1). In addition, reduced albumin synthesis, insulin resistance, and progression of kidney disease have all been linked with acidosis (2,3).
A low-level metabolic acidosis may exist even in individuals without kidney disease (4). This subclinical acidosis, mediated in part by the effects of aging and the Western diet, may have important sequelae on bone, skeletal muscle, and insulin resistance (5–7). Oral alkali administered to postmenopausal women has reduced bone resorption, improved bone mineral density, and decreased urinary nitrogen excretion, suggesting that acidosis has clinical sequelae even in this population (8–10).
In normal subjects, endogenous acid production, which varies according to the composition of the diet, affects steady state acid–base balance in metabolic studies (11). The magnitude of the net acid load is determined by the balance of acid precursors and base precursors in the diet. This dietary stimulus for acid production can vary as much as 10-fold between individuals consuming diets of differing compositions (11). Net endogenous acid production (NEAP) may be estimated using the dietary protein and potassium contents as indices of acid and base precursors, respectively. In CKD patients, higher NEAP has been associated with lower serum bicarbonate (12). However, the effect of the dietary acid load on acid–base status has not been examined in the general population. Because acidosis is associated with adverse outcomes on a population-wide level, even in people without kidney disease (6,7,13–15), determining the extent to which dietary acid may be a modifiable contributor to low-grade acidosis could have important public health implications. In addition, because older persons have lower capacity to excrete an acid load (16), this effect might be modified by age.
We hypothesized that dietary acid would be associated with acid–base status in the general population and that this effect would be greater in older than younger persons. We tested these hypotheses in adult participants ages 20 years and older with data on serum bicarbonate and dietary intake in the National Health and Nutrition Examination Survey (NHANES) 1999–2004.
Materials and Methods
Study Population
NHANES 1999–2004 was a nationally representative survey of the noninstitutionalized civilian population in the United States (17). A stratified, multistage, probability sampling design was used to select participants. The NHANES protocol was approved by the National Center for Health Statistics ethics review board, and written informed consent was obtained from all participants. Overall, 13,274 adults≥20 years of age completed the interview and examination components and had available serum bicarbonate data. We excluded participants who had a history of emphysema or chronic bronchitis by self-report (n=969), were pregnant at the time of examination (n=680), had an estimated GFR (eGFR)<15 ml/min per 1.73 m2 (n=36), had reported use of carbonic anhydrase inhibitors (n=4), had invalid dietary data (n=531), or were missing covariate data (n=1273). Thus, 9781 participants were available for analysis.
Data Collection
Information on household income, education, smoking, comorbidities, and medication use in the previous month was obtained by self-report. Race/ethnicity was self-identified. Smoking was classified as never, former, or current smoker. Low socioeconomic status was defined as less than 100% of the poverty index. Data on dietary intake were obtained from a 24-hour dietary recall questionnaire. The diet-dependent net acid load was estimated as NEAP (mEq/d)=[54.5×protein (g/d)/potassium (mEq/d)]−10.2 (18). Hypertension was defined as systolic BP≥140 mmHg, diastolic BP≥90 mmHg, physician diagnosis, and/or antihypertensive medication use (19). Diabetes mellitus was defined as a physician diagnosis while not pregnant or the current use of insulin or oral hypoglycemic medications. Cardiovascular disease (CVD) was defined by self-report of a physician diagnosis of congestive heart failure, coronary heart disease, angina, myocardial infarction, or stroke.
Serum chemistry values were measured using the Hitachi 917 Multichannel Analyzer (Roche Diagnostics, Indianapolis, IN) in 1999–2001 and the Beckman Synchron LX20 (Beckman Coulter Inc., Brea, CA) in 2002–2004. Serum albumin was measured by the bromocresol purple method. Serum creatinine was measured by a modified kinetic Jaffé reaction. Values from 1999 to 2000 were calibrated to the Cleveland Clinic laboratory standard by multiplying by 1.013 and then adding 0.147. Correction of values from 2001 to 2004 was not necessary. eGFR was calculated using the CKD Epidemiology Collaboration Equation (20).
Outcome Variables
Serum bicarbonate was measured in two laboratories by the phosphoenolpyruvate carboxylase method from 1999 to 2001 and with a pH-sensitive electrode from 2002 to 2004. Because the mean serum bicarbonate was 1.105±0.178 mEq/L higher (P<0.001) among all NHANES participants in 2003–2004 compared with 1999–2002, serum bicarbonate levels in 1999–2002 were adjusted by adding 1.105 mEq/L, which has been done previously (13). Acidosis was defined as serum bicarbonate<23 mEq/L. Because Kidney Disease Outcomes Quality Initiative recommends maintaining the serum bicarbonate≥22 mEq/L in patients with CKD, we also repeated our analyses when defining acidosis as serum bicarbonate<22 mEq/L.
Statistical Analyses
All analyses used NHANES-appropriate sampling weights and accounted for the complex multistage cluster design using the survey command in Stata 11.1 (Stata Corporation, College Station, TX). The distributions of participant characteristics were examined by quartiles of NEAP. Linear and logistic regression models were created to examine the associations of NEAP with serum bicarbonate and acidosis, respectively. NEAP was analyzed as a continuous variable and within quartiles to examine nonlinear associations with either outcome. Models were created examining associations in the overall cohort without adjustment for additional covariates and then including age, sex, and race/ethnicity as covariates. Multivariable models were then created to include other potential confounders of the association of NEAP with acid–base status as covariates, including poverty and education (as markers of socioeconomic status), body mass index categories, smoking status, diagnosis of diabetes mellitus, hypertension, CVD, diuretic use, eGFR categories, log-transformed urine albumin–creatinine ratio, serum albumin, and log-transformed C-reactive protein. To determine whether participants with CKD drove our results, we repeated these analyses after excluding those participants with eGFR<60 ml/min per 1.73 m2. Effect modification by age was tested by including a multiplicative interaction term in the models. Age-stratified analyses were then performed to separately examine associations of NEAP with serum bicarbonate and acidosis within age categories. A P value<0.05 was considered statistically significant.
Sensitivity Analyses
Because NEAP does not account for variations in the sulfur content of animal versus vegetable protein, we repeated our analyses using animal protein-derived endogenous acid production (ADEAP), which we computed by replacing total dietary protein in the NEAP calculation with protein derived from meat, poultry, or fish consumption defined using US Department of Agriculture Food Codes 20000000–28500000. We also examined the associations of dietary protein and potassium separately with serum bicarbonate using linear regression models that included dietary energy as a covariate (21). To determine if residual confounding caused by imprecise ascertainment of obesity affected our results, we repeated our analyses and included percent total body fat as a covariate among the subgroup of 9499 participants who underwent whole-body dual-energy x-ray absorptiometry. Details of the dual-energy x-ray absorptiometry protocol and data validation are available (http://www.cdc.gov/nchs/nhanes/dxx/dxa.htm) (22–24).
Results
Participant Characteristics
The mean age was 45.7 years (SEM=0.3), and the mean serum bicarbonate was 24.9 mEq/L (SEM=0.1). The mean NEAP was 57.4 mEq/d (SEM=0.4). The median dietary protein and potassium intakes were 73 g/d (interquartile range [IQR]=52–100) and 63 mEq/d (IQR=45–86), respectively. Median estimated protein intake per kilogram body weight was 0.93 g/kg per day (IQR=0.66–1.30).
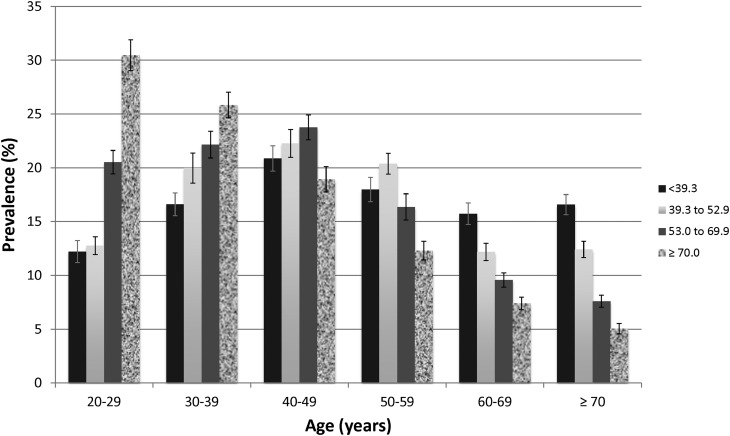
Participants with higher NEAP were younger, were more likely to be men, were more likely to be Mexican Americans or non-Hispanic blacks, were less likely to have hypertension, diabetes, or CVD, were less likely to use diuretics, and had higher eGFR (Table 1). They were more likely to be obese and current smokers, had lower dietary potassium and higher dietary protein intake, and had higher C-reactive protein levels. There was a progressive trend of lower NEAP among older participants (Figure 1).
Table 1.
Participant characteristics by quartiles of net endogenous acid production
| Characteristic | Net Endogenous Acid Production (mEq/d) | P | |||
|---|---|---|---|---|---|
| <39.3 | 39.3–52.9 | 53.0–69.9 | ≥70.0 | ||
| Number | 2477 | 2403 | 2411 | 2490 | |
| Age (yr) | 50.7 (0.45) | 48.4 (0.46) | 44.0 (0.42) | 39.8 (0.41) | <0.001 |
| Women (%) | 58.6 (1.1) | 49.0 (1.3) | 45.9 (1.5) | 43.6 (1.2) | <0.001 |
| Race/ethnicity (%) | <0.001 | ||||
| Non-Hispanic white | 78.1 (1.7) | 76.8 (1.5) | 71.9 (2.0) | 64.1 (2.3) | |
| Mexican American | 5.4 (0.7) | 7.9 (1.1) | 7.8 (1.0) | 8.7 (1.4) | |
| Non-Hispanic black | 7.3 (0.9) | 7.0 (0.7) | 9.4 (1.0) | 17.2 (1.7) | |
| Other | 9.2 (1.4) | 8.3 (0.9) | 10.8 (1.5) | 10.0 (1.3) | |
| Body mass index (kg/m2; %) | <0.001 | ||||
| <18.5 (underweight) | 2.2 (0.4) | 1.4 (0.3) | 0.9 (0.2) | 1.5 (0.3) | |
| 18.5–24.9 (normal) | 37.5 (1.3) | 33.4 (1.4) | 30.5 (1.5) | 29.3 (1.1) | |
| 25–29.9 (overweight) | 35.3 (1.2) | 37.3 (1.3) | 35.5 (1.1) | 33.1 (1.4) | |
| ≥30 (obese) | 24.8 (1.1) | 27.7 (1.3) | 33.0 (1.8) | 35.9 (1.0) | |
| Poverty (<100% poverty index; %) | 11.2 (0.9) | 11.5 (1.0) | 11.1 (0.9) | 16.5 (1.1) | <0.001 |
| Less than high school diploma (%) | 18.4 (1.3) | 17.5 (1.2) | 16.7 (0.9) | 21.6 (1.3) | 0.11 |
| Smoking (%) | <0.001 | ||||
| Never | 50.7 (1.5) | 50.7 (1.8) | 51.5 (1.5) | 50.9 (1.9) | |
| Former | 28.7 (1.3) | 27.8 (1.5) | 23.4 (1.3) | 19.9 (1.1) | |
| Current | 20.6 (1.2) | 21.5 (1.2) | 25.1 (1.4) | 29.2 (1.7) | |
| Dietary protein (g/d) | 60.6 (0.9) | 81.3 (1.2) | 92.7 (1.2) | 99.3 (1.1) | <0.001 |
| Dietary potassium (mEq/d) | 82.0 (1.2) | 78.4 (1.2) | 71.1 (1.0) | 54.6 (0.7) | <0.001 |
| Diuretic use (%) | 11.5 (0.8) | 10.5 (1.0) | 8.7 (0.8) | 8.7 (0.8) | 0.001 |
| Hypertension (%) | 43.5 (1.7) | 42.1 (1.7) | 36.1 (1.5) | 34.9 (1.3) | <0.001 |
| Cardiovascular disease (%) | 10.6 (0.8) | 7.9 (0.9) | 7.6 (0.7) | 6.0 (0.6) | <0.001 |
| Diabetes mellitus (%) | 7.7 (0.5) | 6.7 (0.7) | 6.8 (0.7) | 6.2 (0.6) | 0.03 |
| Estimated GFR (ml/min per 1.73 m2; %) | <0.001 | ||||
| ≥120 | 6.8 (0.6) | 8.1 (0.7) | 10.1 (0.7) | 16.6 (1.2) | |
| 90–119 | 43.5 (1.7) | 46.7 (1.3) | 52.4 (1.3) | 50.9 (1.6) | |
| 60–89 | 40.5 (1.5) | 38.9 (1.5) | 33.4 (1.4) | 28.6 (1.6) | |
| 45–59 | 6.7 (0.6) | 4.5 (0.5) | 2.8 (0.3) | 2.8 (0.3) | |
| 30–44 | 1.9 (0.3) | 1.4 (0.2) | 0.9 (0.2) | 1.0 (0.2) | |
| 15–29 | 0.6 (0.1) | 0.3 (0.1) | 0.3 (0.1) | 0.07 (0.04) | |
| Urine albumin–creatinine ratio (mg/g; %) | 0.36 | ||||
| <30 | 90.8 (0.8) | 90.5 (0.9) | 92.9 (0.7) | 91.1 (0.8) | |
| 30–300 | 8.3 (0.8) | 8.3 (0.9) | 6.1 (0.6) | 7.6 (0.7) | |
| >300 | 1.0 (0.2) | 1.2 (0.3) | 1.0 (0.2) | 1.3 (0.3) | |
| Serum albumin (g/dl) | 4.36 (1.2) | 4.38 (1.0) | 4.37 (1.2) | 4.37 (1.1) | 0.48 |
| C-reactive protein (mg/dl; %) | 0.02 | ||||
| <0.36 | 71.1 (1.1) | 71.2 (1.2) | 69.7 (1.3) | 66.8 (1.2) | |
| 0.36–0.99 | 19.7 (0.8) | 20.4 (1.1) | 20.8 (1.0) | 24.0 (1.1) | |
| ≥1 | 9.2 (0.8) | 8.4 (0.7) | 9.5 (0.9) | 9.2 (0.7) | |
Data are expressed as mean (SEM) or percent (SEM).
Figure 1.
Association of estimated net endogenous acid production (NEAP; mEq/d) with age in 9781 participants of the National Health and Nutrition Examination Survey 1999–2004. Each bar represents an NEAP quartile. Error bars represent SEMs.
Association of NEAP with Serum Bicarbonate
In unadjusted analysis, a 1 SD higher NEAP was associated with 0.22 mEq/L (95% confidence interval [95% CI], −0.28 to −0.16) lower serum bicarbonate (Table 2). Multivariable adjustment somewhat attenuated this association (−0.15 mEq/L [95% CI, −0.20 to −0.10] per 1 SD higher NEAP). Compared with participants in the lowest quartile of NEAP, participants in the highest quartile had 0.59 mEq/L (95% CI, −0.76 to −0.43) and 0.40 mEq/L (95% CI, −0.55 to −0.26) lower serum bicarbonate in unadjusted and multivariable-adjusted analyses, respectively.
Table 2.
Association of net endogenous acid production with serum bicarbonate in 9781 participants of the National Health and Nutrition Examination Survey 1999–2004
| Net Endogenous Acid Production | Coefficient (95% Confidence Interval) | ||
|---|---|---|---|
| Model 1 | Model 2 | Model 3 | |
| Continuousa | −0.22 (−0.28 to −0.16)b | −0.20 (−0.26 to −0.14)b | −0.15 (−0.20 to −0.10)b |
| <39.3 mEq/d | Reference | Reference | Reference |
| 39.3–52.9 mEq/d | −0.12 (−0.27 to −0.04) | −0.13 (−0.28 to −0.01) | −0.11 (−0.26 to −0.03)b |
| 53.0–69.9 mEq/d | −0.35 (−0.55 to −0.16)b | −0.33 (−0.52 to −0.14)b | −0.24 (−0.42 to −0.07)b |
| ≥70.0 mEq/d | −0.59 (−0.76 to −0.43)b | −0.53 (−0.70 to −0.36)b | −0.40 (−0.55 to −0.26)b |
| P for trend | <0.001 | <0.001 | <0.001 |
Model 1: unadjusted. Model 2: adjusted for age, sex, and race/ethnicity. Model 3: model 2 adjusted for body mass index, poverty, education, smoking status, diuretic use, diagnosis of diabetes mellitus, hypertension, cardiovascular disease, estimated GFR, log-transformed urine albumin–creatinine ratio, serum albumin, and log-transformed C-reactive protein.
Per SD higher net endogenous acid production (SD=26.4 mEq/d).
P<0.05.
We defined acidosis as a serum bicarbonate<23 mEq/L. A 1 SD higher NEAP was associated with a 1.09 (95% CI, 1.00 to 1.21) greater likelihood of acidosis after multivariate adjustment (Table 3). Participants in the highest NEAP quartile, compared with the lowest NEAP quartile, had a multivariable-adjusted odds ratio for acidosis of 1.33 (95% CI, 1.03 to 1.72). Our results were unchanged after excluding participants with eGFR<60 ml/min per 1.73 m2 (Supplemental Tables 1 and 2). Similar results were found defining acidosis as serum bicarbonate<22 mEq/L (Supplemental Table 3).
Table 3.
Odds ratio of acidosis by net endogenous acid production in 9781 participants of the National Health and Nutrition Examination Survey 1999–2004
| Net Endogenous Acid Production | Odds Ratio (95% Confidence Interval) | ||
|---|---|---|---|
| Model 1 | Model 2 | Model 3 | |
| Continuousa | 1.17 (1.08 to 1.28)b | 1.15 (1.05 to 1.26)b | 1.09 (1.00 to 1.21) |
| <39.3 mEq/d | Reference | Reference | Reference |
| 39.3–52.9 mEq/d | 1.11 (0.90 to 1.37) | 1.12 (0.92 to 1.38) | 1.11 (0.90 to 1.38) |
| 53.0–69.9 mEq/d | 1.38 (1.08 to 1.78)b | 1.34 (1.05 to 1.73)b | 1.25 (0.97 to 1.60) |
| ≥70.0 mEq/d | 1.61 (1.26 to 2.05)b | 1.51 (1.16 to 1.95)b | 1.33 (1.03 to 1.72)b |
| P for trend | <0.001 | 0.002 | 0.03 |
Acidosis defined as serum bicarbonate<23 mEq/L. Model 1: unadjusted. Model 2: adjusted for age, sex, and race/ethnicity. Model 3: model 2 adjusted for body mass index, poverty, education, smoking status, diuretic use, diagnosis of diabetes mellitus, hypertension, cardiovascular disease, estimated GFR, log-transformed urine albumin–creatinine ratio, serum albumin, and log-transformed C-reactive protein.
Per SD higher net endogenous acid production (SD=26.4 mEq/d).
P<0.05.
Interaction of Age with NEAP and Serum Bicarbonate
In the fully adjusted model, a 10 year greater age was associated with a 0.24 mEq/L (95% CI, 0.18 to 0.31) higher serum bicarbonate. We next examined the distribution of serum bicarbonate across age groups (Figure 2). Serum bicarbonate was linearly associated with age, such that bicarbonate levels were highest among the oldest participants. Additional examination stratified by NEAP quartiles showed that the magnitude of this association was dependent on NEAP. The largest increase in serum bicarbonate across age groups was in the lowest NEAP quartile, whereas the smallest difference was seen in the highest quartile.
Figure 2.
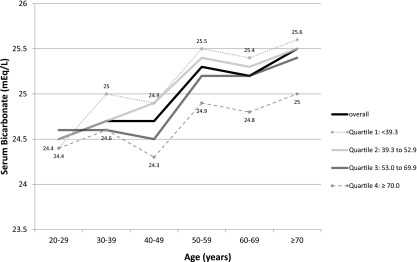
Mean serum bicarbonate levels across age categories, overall, and within quartiles of estimated net endogenous acid production (NEAP; mEq/d) in 9781 participants of the National Health and Nutrition Examination Survey 1999–2004. Individual lines represent the overall population and individual NEAP quartiles. Within each age category, the mean serum bicarbonate is listed for the highest and lowest quartile of NEAP.
We found a significant interaction by age of the association of NEAP with serum bicarbonate (P=0.005). Participants were then stratified by age into three categories: 20–39, 40–59, and ≥60 years old (Table 4). Among participants in the youngest age group, NEAP as a continuous variable was not significantly associated with lower serum bicarbonate after multivariable adjustment. Compared with participants in the lowest NEAP quartile, participants in the highest NEAP quartile had 0.26 mEq/L (95% CI, −0.49 to −0.03) lower serum bicarbonate. Among participants 40–59 and ≥60 years old, NEAP was significantly associated with serum bicarbonate as both a continuous variable and within quartiles. In these age groups, after multivariable adjustment, participants in the highest NEAP quartile had 0.60 (95% CI, −0.92 to −0.29) and 0.49 (95% CI, −0.84 to −0.14) mEq/L lower serum bicarbonate, respectively, compared with those participants in the lowest quartile.
Table 4.
Association of net endogenous acid production with serum bicarbonate within age categories
| Net Endogenous Acid Production | Coefficient (95% Confidence Interval) | ||
|---|---|---|---|
| Model 1 | Model 2 | Model 3 | |
| Age 20–39 yr (n=3299) | |||
| Continuousa | −0.05 (−0.14 to 0.03) | −0.12 (−0.20 to −0.03)b | −0.07 (−0.16 to 0.01) |
| <39.3 mEq/d | Reference | Reference | Reference |
| 39.3–52.9 mEq/d | −0.10 (−0.42 to 0.21) | −0.17 (−0.49 to 0.14) | −0.12 (−0.42 to 0.18) |
| 53.0–69.9 mEq/d | −0.14 (−0.48 to 0.20) | −0.27 (−0.56 to 0.02) | −0.15 (−0.40 to 0.11) |
| ≥70.0 mEq/d | −0.25 (−0.51 to 0.02) | −0.40 (−0.64 to −0.17)b | −0.26 (−0.49 to −0.03)b |
| P for trend | 0.05 | 0.001 | 0.03 |
| Age 40–59 yr (n=3139) | |||
| Continuousa | −0.29 (−0.39 to −0.19)b | −0.33 (−0.43 to −0.23)b | −0.26 (−0.37 to −0.16)b |
| <39.3 mEq/d | Reference | Reference | Reference |
| 39.3–52.9 mEq/d | −0.03 (−0.34 to 0.28) | −0.13 (-0.43 to 0.17) | −0.11 (−0.40 to 0.18) |
| 53.0–69.9 mEq/d | −0.36 (−0.65 to −0.08)b | −0.42 (−0.69 to −0.14)b | −0.35 (−0.63 to −0.07)b |
| ≥70.0 mEq/d | −0.64 (−0.96 to −0.32)b | −0.76 (−1.08 to −0.43)b | −0.60 (−0.92 to −0.29)b |
| P for trend | <0.001 | <0.001 | <0.001 |
| Age≥60 yr (n=3343) | |||
| Continuousa | −0.21 (−0.35 to −0.07)b | −0.20 (−0.34 to −0.06)b | −0.17 (−0.30 to −0.04)b |
| <39.3 mEq/d | Reference | Reference | Reference |
| 39.3–52.9 mEq/d | −0.11 (−0.38 to 0.16) | −0.10 (−0.37 to 0.18) | −0.11 (−0.39 to 0.17) |
| 53.0–69.9 mEq/d | −0.22 (−0.50 to 0.06) | −0.18 (−0.45 to 0.09) | −0.15 (−0.40 to 0.09) |
| ≥70.0 mEq/d | −0.58 (−0.93 to −0.23)b | −0.55 (−0.90 to −0.19)b | −0.49 (−0.84 to −0.14)b |
| P for trend | 0.002 | 0.004 | 0.008 |
Model 1: unadjusted. Model 2: adjusted for age, sex, and race/ethnicity. Model 3: model 2 adjusted for body mass index, poverty, education, smoking status, diuretic use, diagnosis of diabetes mellitus, hypertension, cardiovascular disease, estimated GFR, log-transformed urine albumin–creatinine ratio, serum albumin, and log-transformed C-reactive protein. P for interaction by age=0.005.
Per SD higher net endogenous acid production (SD=26.4 mEq/d).
P<0.05.
We similarly found significant effect modification by age using acidosis as the outcome (P=0.02). There was no statistically significant association of NEAP with acidosis in the youngest age group (Table 5). There was a significant association of higher NEAP with greater likelihood of acidosis among participants 40–59 years old; there was a similar trend among those participants≥60 years of age, but it did not reach statistical significance.
Table 5.
Odds ratio of acidosis by net endogenous acid production within age categories
| Net Endogenous Acid Production | Odds Ratio (95% Confidence Interval) | ||
|---|---|---|---|
| Model 1 | Model 2 | Model 3 | |
| Age 20–39 yr (n=3299) | |||
| Continuousa | 1.02 (0.92 to 1.13) | 1.06 (0.96 to 1.19) | 1.03 (0.92 to 1.15) |
| <39.3 mEq/d | Reference | Reference | Reference |
| 39.3–52.9 mEq/d | 0.97 (0.63 to 1.50) | 1.02 (0.66 to 1.58) | 0.96 (0.61 to 1.52) |
| 53.0–69.9 mEq/d | 1.15 (0.77 to 1.72) | 1.28 (0.87 to 1.87) | 1.14 (0.80 to 1.62) |
| ≥70.0 mEq/d | 1.16 (0.83 to 1.61) | 1.31 (0.96 to 1.79) | 1.04 (0.84 to 1.56) |
| P for trend | 0.25 | 0.08 | 0.28 |
| Age 40–59 yr (n=3139) | |||
| Continuousa | 1.27 (1.10 to 1.48)b | 1.31 (1.13 to 1.52)b | 1.22 (1.03 to 1.45)b |
| <39.3 mEq/d | Reference | Reference | Reference |
| 39.3–52.9 mEq/d | 1.19 (0.81 to 1.73) | 1.25 (0.86 to 1.80) | 1.27 (0.88 to 1.82) |
| 53.0–69.9 mEq/d | 1.39 (0.98 to 1.95) | 1.42 (1.01 to 2.01)b | 1.38 (0.94 to 2.04) |
| ≥70.0 mEq/d | 1.78 (1.07 to 2.94)b | 1.91 (1.18 to 3.08)b | 1.65 (0.98 to 2.78) |
| P for trend | 0.02 | 0.01 | 0.06 |
| Age≥60 yr (n=3343) | |||
| Continuousa | 1.13 (0.96 to 1.34) | 1.12 (0.94 to 1.34) | 1.11 (0.93 to 1.33) |
| <39.3 mEq/d | Reference | Reference | Reference |
| 39.3–52.9 mEq/d | 1.08 (0.71 to 1.63) | 1.06 (0.69 to 1.63) | 1.09 (0.69 to 1.72) |
| 53.0–69.9 mEq/d | 1.27 (0.84 to 1.91) | 1.24 (0.81 to 1.88) | 1.22 (0.81 to 1.84) |
| ≥70.0 mEq/d | 1.47 (0.97 to 2.25) | 1.46 (0.93 to 2.28) | 1.44 (0.91 to 2.29) |
| P for trend | 0.05 | 0.08 | 0.09 |
Model 1: unadjusted. Model 2: adjusted for age, sex, and race/ethnicity. Model 3: model 2 adjusted for body mass index, poverty, education, smoking status, diuretic use, diagnosis of diabetes mellitus, hypertension, cardiovascular disease, estimated GFR, log-transformed urine albumin–creatinine ratio, serum albumin, and log-transformed C-reactive protein. P for interaction by age=0.02.
Per SD higher net endogenous acid production (SD=26.4 mEq/d).
P<0.05.
Sensitivity Analyses
The distribution of ADEAP quartiles across age categories was similar to the distribution of NEAP quartiles (Supplemental Figure 1). Higher ADEAP was associated with lower serum bicarbonate (Supplemental Table 4), and there was a significant interaction by age of the association of ADEAP with serum bicarbonate (P for interaction=0.01) (Supplemental Table 3). Higher dietary protein (Supplemental Table 6) and lower dietary potassium (Supplemental Table 7) were each associated with lower serum bicarbonate, although the associations seemed more robust for potassium intake. Adjustment for percent total body fat did not affect our results (Supplemental Tables 8 and 9).
Discussion
The principle objective of this study was to investigate the association of dietary acid with metabolic acidosis in the general population and examine the possibility of effect modification by age of this association. We found that a higher dietary acid load, which was measured by NEAP, was associated with lower serum bicarbonate and a higher likelihood of acidosis in a nationally representative sample of Americans. In addition, the magnitude of these associations was greater in middle-aged and elderly participants than younger participants. This result suggests that the deleterious effects of dietary acid may be most prominent among older individuals.
Previous physiologic studies in normal adults have shown that differences in dietary acid affect extracellular pH and serum bicarbonate (8,11). Despite these variations in diet, renal acid excretion maintains acid–base homeostasis in normal persons, but this capacity declines with age (25). Our results extend these findings to indicate that dietary acid plays a role in determining acid–base status on a population-wide level and that this effect is most pronounced among older persons.
This finding could have important clinical implications. Even among individuals without kidney disease, a low-grade acidosis contributes to bone breakdown and possibly loss of muscle mass (9,10), and low-normal serum bicarbonate levels have been associated with lesser muscle strength and cardiovascular fitness, greater insulin resistance, and faster progression of kidney disease (6,7,13,26). Interventions to treat acidosis, including dietary modifications (27,28), may be most efficacious among older persons, in whom the capacity to excrete the daily acid load from modern Western diets is diminished.
We also observed an unexpected association between serum bicarbonate and age. Contrary to previous literature suggesting that older people have lower serum bicarbonate because of an age-related decline in renal acid excretion (29,30), we found that older age was associated with higher serum bicarbonate. This finding seems partly caused by lower dietary acid intake among older persons, suggesting that NEAP may be equally important as the age-related decline in acid excretion as a determinant of serum bicarbonate. However, even when comparing older and younger Americans within the same dietary acid quartile, bicarbonate levels were higher among the former group. This association persisted after accounting for possible explanatory factors, including diuretic use. One possible explanation is that of a secular trend, in which additional factors that lower serum bicarbonate exist among younger but not older persons. We speculate that such factors could relate to diet or the worsening epidemic of obesity. Over time, these individuals would develop lower serum bicarbonate and worsening low-grade metabolic acidosis if our understanding of the physiology relating aging and renal acid excretory capacity remains correct. Compared with the current cohort of older Americans, however, these younger individuals have lower bicarbonate levels. Therefore, the association of older age with higher serum bicarbonate would not be causal but rather an artifact of the cross-sectional nature of our analyses. It should be noted, however, that the previous reports of declining bicarbonate levels with age also resulted from cross-sectional studies (29,30). In contrast to those data, which come from participants of small clinical and metabolic studies, our findings are from a large, nationally representative dataset.
It remains possible that a more precise measurement of dietary acid intake might account for all of the increase in serum bicarbonate that was seen across age categories. There may also be unaccounted factors related to aging that result in higher bicarbonate levels. Tubular sodium reabsorption is impaired in the elderly (31); resultant mild volume depletion, if present, would increase bicarbonate reabsorption. Subclinical changes in respiratory status, such as mild CO2 retention, could also occur with aging. If such factors were more common among elderly than middle-aged participants, they could also mask the effect of dietary acid to a greater extent in the elderly. This result could explain why the associations of NEAP with serum bicarbonate and acidosis seemed somewhat stronger among middle-aged adults. Longitudinal population-based data are needed to address this question.
Serum bicarbonate levels among 20–29 year olds did not differ by dietary acid level. This finding suggests that young adults handle even large acid loads within the range of usual dietary intake without perturbing acid–base status. Our data indicate that this capacity already begins to diminish by the fourth decade of life, which is consistent with previous reports (25,30). It should be noted, however, that acid retention may occur even without apparent perturbation of the serum bicarbonate or extracellular pH. A study of patients with mildly reduced eGFR and normal serum bicarbonate found that changes in urine net acid excretion may be seen even without a change in blood acid–base parameters (32). Therefore, high dietary acid could produce the sequelae of a subclinical acidosis in these younger adults even without observable differences in plasma acid–base parameters.
Intracellular buffering would blunt the effect of NEAP on extracellular buffers, and buffering capacity likely varies directly with the serum bicarbonate (33). Thus, the change in bicarbonate might particularly underestimate the effects of NEAP among older persons, who had the highest bicarbonate levels in our sample. Conversely, people with CKD may have reduced buffering capacity and would experience a relatively greater decrease in serum bicarbonate for any level of NEAP (32).
Several limitations of our analyses should be noted. Calculation of NEAP may have been affected by the limitations of dietary assessment using a single 24-hour recall period. Dietary acid load was not directly measured, and data on renal net acid excretion were not available. However, we used a validated formula to estimate NEAP that was derived using measurements of renal net acid excretion. Although this calculation does not account for differences in the sulfur content of protein or base precursors not associated with potassium, our results were substantively unchanged after replacing total protein with animal-derived sources of protein, which could be a more specific measure of dietary acid precursors. Participants may have underreported dietary protein intake; alternatively, even modestly elevated protein intake may be associated with observable differences in serum bicarbonate levels. We speculated regarding effects of dietary acid on outcomes, such as muscle catabolism and bone disease, based on differences in serum bicarbonate. We believed such speculation was reasonable and should be tested in future studies. We had only a single value of serum bicarbonate available and did not have measurements of serum pH or pCO2. Sample handling, including processing delays and exposure of serum to air, may affect bicarbonate levels (34–36). We could not account for this possibility, which would likely introduce nondifferential misclassification and bias our results to the null. Finally, we cannot rule out residual confounding caused by undetected kidney disease among older individuals, because eGFR is an imprecise measurement of kidney function. We addressed this possibility by restricting our analyses to participants with eGFR≥60 ml/min per 1.73 m2, which did not change the results. In addition, if older age was simply a marker for reduced kidney function, our findings would still be clinically relevant, because older persons constitute an expanding proportion of the population with kidney disease.
In summary, greater dietary acid is associated with lower serum bicarbonate and a higher prevalence of acidosis in the general US population, and this association is greater in older than younger Americans. In contrast to previous reports, serum bicarbonate levels are highest among older Americans. Longitudinal studies are needed to prospectively determine the association of the dietary acid load with acid–base status in free-living individuals. In the interim, clinicians should be aware that older persons may be most susceptible to the potential deleterious effects of diets high in acid-forming potential.
Disclosures
None.
Supplementary Material
Acknowledgments
This research was supported by Clinical and Translational Science Award (CTSA) Grants UL1RR025750, KL2RR025749, and TL1RR025748 from the National Center for Research Resources, a component of the National Institutes of Health.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
This article contains supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.03600413/-/DCSupplemental.
References
- 1.Kraut JA, Madias NE: Consequences and therapy of the metabolic acidosis of chronic kidney disease. Pediatr Nephrol 26: 19–28, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.de Brito-Ashurst I, Varagunam M, Raftery MJ, Yaqoob MM: Bicarbonate supplementation slows progression of CKD and improves nutritional status. J Am Soc Nephrol 20: 2075–2084, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ballmer PE, McNurlan MA, Hulter HN, Anderson SE, Garlick PJ, Krapf R: Chronic metabolic acidosis decreases albumin synthesis and induces negative nitrogen balance in humans. J Clin Invest 95: 39–45, 1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Frassetto L, Morris RC, Jr, Sellmeyer DE, Todd K, Sebastian A: Diet, evolution and aging—the pathophysiologic effects of the post-agricultural inversion of the potassium-to-sodium and base-to-chloride ratios in the human diet. Eur J Nutr 40: 200–213, 2001 [DOI] [PubMed] [Google Scholar]
- 5.Macdonald HM, New SA, Fraser WD, Campbell MK, Reid DM: Low dietary potassium intakes and high dietary estimates of net endogenous acid production are associated with low bone mineral density in premenopausal women and increased markers of bone resorption in postmenopausal women. Am J Clin Nutr 81: 923–933, 2005 [DOI] [PubMed] [Google Scholar]
- 6.Abramowitz MK, Hostetter TH, Melamed ML: Association of serum bicarbonate levels with gait speed and quadriceps strength in older adults. Am J Kidney Dis 58: 29–38, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Farwell WR, Taylor EN: Serum bicarbonate, anion gap and insulin resistance in the National Health and Nutrition Examination Survey. Diabet Med 25: 798–804, 2008 [DOI] [PubMed] [Google Scholar]
- 8.Sebastian A, Harris ST, Ottaway JH, Todd KM, Morris RC, Jr: Improved mineral balance and skeletal metabolism in postmenopausal women treated with potassium bicarbonate. N Engl J Med 330: 1776–1781, 1994 [DOI] [PubMed] [Google Scholar]
- 9.Jehle S, Zanetti A, Muser J, Hulter HN, Krapf R: Partial neutralization of the acidogenic Western diet with potassium citrate increases bone mass in postmenopausal women with osteopenia. J Am Soc Nephrol 17: 3213–3222, 2006 [DOI] [PubMed] [Google Scholar]
- 10.Frassetto L, Morris RC, Jr, Sebastian A: Potassium bicarbonate reduces urinary nitrogen excretion in postmenopausal women. J Clin Endocrinol Metab 82: 254–259, 1997 [DOI] [PubMed] [Google Scholar]
- 11.Kurtz I, Maher T, Hulter HN, Schambelan M, Sebastian A: Effect of diet on plasma acid-base composition in normal humans. Kidney Int 24: 670–680, 1983 [DOI] [PubMed] [Google Scholar]
- 12.Scialla JJ, Appel LJ, Astor BC, Miller ER, 3rd, Beddhu S, Woodward M, Parekh RS, Anderson CA: Estimated net endogenous acid production and serum bicarbonate in African Americans with chronic kidney disease. Clin J Am Soc Nephrol 6: 1526–1532, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Abramowitz MK, Hostetter TH, Melamed ML: Lower serum bicarbonate and a higher anion gap are associated with lower cardiorespiratory fitness in young adults. Kidney Int 81: 1033–1042, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Frassetto LA, Todd KM, Morris RC, Jr, Sebastian A: Worldwide incidence of hip fracture in elderly women: Relation to consumption of animal and vegetable foods. J Gerontol A Biol Sci Med Sci 55: M585–M592, 2000 [DOI] [PubMed] [Google Scholar]
- 15.Zhang L, Curhan GC, Forman JP: Diet-dependent net acid load and risk of incident hypertension in United States women. Hypertension 54: 751–755, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hilton JG, Goodbody MF, Jr, Kruesi OR: The effect of prolonged administration of ammonium chloride on the blood acid-base equilibrium of geriatric subjects. J Am Geriatr Soc 3: 697–703, 1955 [DOI] [PubMed] [Google Scholar]
- 17.Centers for Disease Control and Prevention (CDC), National Center for Health Statistics (NCHS) : About the National Health and Nutrition Examination Survey. Available at: http://www.cdc.gov/nchs/nhanes/about_nhanes.htm Accessed December 21, 2010
- 18.Frassetto LA, Todd KM, Morris RC, Jr, Sebastian A: Estimation of net endogenous noncarbonic acid production in humans from diet potassium and protein contents. Am J Clin Nutr 68: 576–583, 1998 [DOI] [PubMed] [Google Scholar]
- 19.Muntner P, Woodward M, Mann DM, Shimbo D, Michos ED, Blumenthal RS, Carson AP, Chen H, Arnett DK: Comparison of the Framingham Heart Study hypertension model with blood pressure alone in the prediction of risk of hypertension: The Multi-Ethnic Study of Atherosclerosis. Hypertension 55: 1339–1345, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J, CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) : A new equation to estimate glomerular filtration rate. Ann Intern Med 150: 604–612, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Willett WC, Howe GR, Kushi LH: Adjustment for total energy intake in epidemiologic studies. Am J Clin Nutr 65[4 Suppl]: 1220S–1231S, 1997 [DOI] [PubMed] [Google Scholar]
- 22.Heymsfield SB, Heo M, Thomas D, Pietrobelli A: Scaling of body composition to height: Relevance to height-normalized indexes. Am J Clin Nutr 93: 736–740, 2011 [DOI] [PubMed] [Google Scholar]
- 23.Li C, Ford ES, Zhao G, Balluz LS, Giles WH: Estimates of body composition with dual-energy X-ray absorptiometry in adults. Am J Clin Nutr 90: 1457–1465, 2009 [DOI] [PubMed] [Google Scholar]
- 24.Schenker N, Borrud LG, Burt VL, Curtin LR, Flegal KM, Hughes J, Johnson CL, Looker AC, Mirel L: Multiple imputation of missing dual-energy X-ray absorptiometry data in the National Health and Nutrition Examination Survey. Stat Med 30: 260–276, 2011 [DOI] [PubMed] [Google Scholar]
- 25.Berkemeyer S, Vormann J, Günther AL, Rylander R, Frassetto LA, Remer T: Renal net acid excretion capacity is comparable in prepubescence, adolescence, and young adulthood but falls with aging. J Am Geriatr Soc 56: 1442–1448, 2008 [DOI] [PubMed] [Google Scholar]
- 26.Shah SN, Abramowitz M, Hostetter TH, Melamed ML: Serum bicarbonate levels and the progression of kidney disease: A cohort study. Am J Kidney Dis 54: 270–277, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gennari FJ, Hood VL, Greene T, Wang X, Levey AS: Effect of dietary protein intake on serum total CO2 concentration in chronic kidney disease: Modification of Diet in Renal Disease study findings. Clin J Am Soc Nephrol 1: 52–57, 2006 [DOI] [PubMed] [Google Scholar]
- 28.Goraya N, Simoni J, Jo CH, Wesson DE: A comparison of treating metabolic acidosis in CKD stage 4 hypertensive kidney disease with fruits and vegetables or sodium bicarbonate. Clin J Am Soc Nephrol 8: 371–381, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Frassetto LA, Morris RC, Jr, Sebastian A: Effect of age on blood acid-base composition in adult humans: Role of age-related renal functional decline. Am J Physiol 271: F1114–F1122, 1996 [DOI] [PubMed] [Google Scholar]
- 30.Frassetto L, Sebastian A: Age and systemic acid-base equilibrium: Analysis of published data. J Gerontol A Biol Sci Med Sci 51: B91–B99, 1996 [DOI] [PubMed] [Google Scholar]
- 31.Musso CG, Oreopoulos DG: Aging and physiological changes of the kidneys including changes in glomerular filtration rate. Nephron, Physiol 119[Suppl 1]: 1–5, 2011 [DOI] [PubMed] [Google Scholar]
- 32.Wesson DE, Simoni J, Broglio K, Sheather S: Acid retention accompanies reduced GFR in humans and increases plasma levels of endothelin and aldosterone. Am J Physiol Renal Physiol 300: F830–F837, 2011 [DOI] [PubMed] [Google Scholar]
- 33.Adrogué HJ, Brensilver J, Cohen JJ, Madias NE: Influence of steady-state alterations in acid-base equilibrium on the fate of administered bicarbonate in the dog. J Clin Invest 71: 867–883, 1983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kirschbaum B: Spurious metabolic acidosis in hemodialysis patients. Am J Kidney Dis 35: 1068–1071, 2000 [DOI] [PubMed] [Google Scholar]
- 35.Laski ME: Penny wise and bicarbonate foolish. Am J Kidney Dis 35: 1224–1225, 2000 [DOI] [PubMed] [Google Scholar]
- 36.Bray SH, Tung RL, Jones ER: The magnitude of metabolic acidosis is dependent on differences in bicarbonate assays. Am J Kidney Dis 28: 700–703, 1996 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.