Abstract
Background
Next-generation sequencing technology is an important tool for the rapid, genome-wide identification of genetic variations. However, it is difficult to resolve the ‘signal’ of variations of interest and the ‘noise’ of stochastic sequencing and bioinformatic errors in the large datasets that are generated. We report a simple approach to identify regional linkage to a trait that requires only two pools of DNA to be sequenced from progeny of a defined genetic cross (i.e. bulk segregant analysis) at low coverage (<10×) and without parentage assignment of individual SNPs. The analysis relies on regional averaging of pooled SNP frequencies to rapidly scan polymorphisms across the genome for differential regional homozygosity, which is then displayed graphically.
Results
Progeny from defined genetic crosses of Tribolium castaneum (F4 and F19) segregating for the phosphine resistance trait were exposed to phosphine to select for the resistance trait while the remainders were left unexposed. Next generation sequencing was then carried out on the genomic DNA from each pool of selected and unselected insects from each generation. The reads were mapped against the annotated T. castaneum genome from NCBI (v3.0) and analysed for SNP variations. Since it is difficult to accurately call individual SNP frequencies when the depth of sequence coverage is low, variant frequencies were averaged across larger regions. Results from regional SNP frequency averaging identified two loci, tc_rph1 on chromosome 8 and tc_rph2 on chromosome 9, which together are responsible for high level resistance. Identification of the two loci was possible with only 5-7× average coverage of the genome per dataset. These loci were subsequently confirmed by direct SNP marker analysis and fine-scale mapping. Individually, homozygosity of tc_rph1 or tc_rph2 results in only weak resistance to phosphine (estimated at up to 1.5-2.5× and 3-5× respectively), whereas in combination they interact synergistically to provide a high-level resistance >200×. The tc_rph2 resistance allele resulted in a significant fitness cost relative to the wild type allele in unselected beetles over eighteen generations.
Conclusion
We have validated the technique of linkage mapping by low-coverage sequencing of progeny from a simple genetic cross. The approach relied on regional averaging of SNP frequencies and was used to successfully identify candidate gene loci for phosphine resistance in T. castaneum. This is a relatively simple and rapid approach to identifying genomic regions associated with traits in defined genetic crosses that does not require any specialised statistical analysis.
Keywords: Insecticide resistance, Rhyzopertha dominica, Linkage disequilibrium, Fitness cost, Gene interactions
Background
High-throughput “next-generation” sequencing is now a cost effective tool for most genetics laboratories, making new approaches for genetic analysis a possibility. One of the promising aspects of next-generation sequencing data is that it can be used to rapidly define genomic regions of interest in linkage and quantitative genetics experiments. There are myriad ways that this can be accomplished but no straightforward statistical and data processing approach has been developed to distinguish the ‘signal’ from the ‘noise’. Some researchers have simplified the analysis by establishing specially prepared genetic strains, as in linkage by introgression [1]. In other systems sequencing of the parental genomes has been used to allow parentage to be assigned to single nucleotide polymorphisms (SNPs), which is an alternative strategy to facilitate identification of linkage to a trait or traits. It is challenging, however, to confidently determine the parentage assignment of SNPs in genome-wide datasets that have a relatively low average coverage (<10×), such as might be encountered in organisms with large genomes.
In this study we developed a relatively simple and robust method that does not require the parental genomes to be known or sequenced. The method involves bulk-segregant sequence analysis and averaging frequencies of SNPs attaining differential homozygosity to display genomic regions containing genes for a specific trait. We used this method to identify genes conferring resistance to an insecticidal fumigant, phosphine (PH3) in a cosmopolitan grain insect pest, Tribolium castaneum. Phosphine is an effective fumigant, used worldwide to protect grain harvest from insect pests [2]. It has been in use for nearly five decades owing to its broad spectrum of activity and residue-free characteristics [3,4]. Heavy reliance on phosphine has given rise to high-level heritable resistance in several grain insect pests, threatening the efficacy of phosphine [5-10].
Classical genetic studies on resistance to phosphine in two major grain insect pests, the rust red flour beetle, T. castaneum[11] and the lesser grain borer, Rhyzopertha dominica[12] has confirmed that strong resistance is primarily conferred by two major genes, rph1 and rph2. Recently, a gene encoding a core metabolic enzyme, dihydrolipoamide dehydrogenase (dld) has been identified as rph2[13] in both R. dominica and T. castaneum and a free soil living nematode Caenorhabditis elegans, indicating that resistance mechanisms involve altered oxidative metabolism with key respiratory enzymes. T. castaneum is a model organism for both genetic and developmental studies and the wealth of background genetic information with a recently published draft genome [14], makes this insect an ideal system in which to test novel applications of next-generation sequencing to gene mapping. The present study aimed to (i) apply next generation sequencing methods to linkage mapping analyses by identifying the genomic location of genes conferring resistance to phosphine and (ii) develop SNP markers for understanding the biological and genetic aspects (gene interactions and fitness) of this trait on grain insect pests.
Results
Sequence analyses and SNP discovery
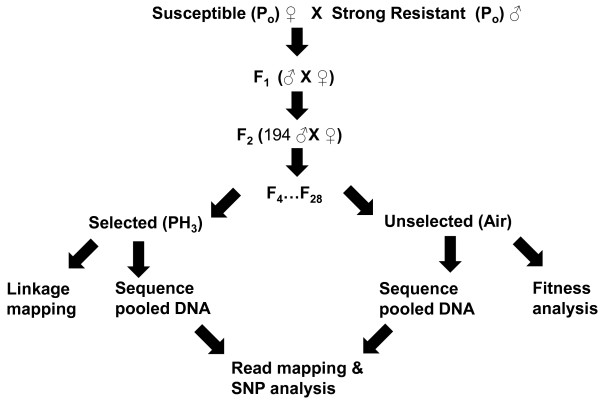
Two similar single pair inter-strain crosses (SIC), SICS/SRA and SICS/SRB (both S ♀ X Strong-R ♂) were established (Figure 1) and selected at different generational cohorts (F4 and F19 for the different crosses) for strong resistance. Selected (strongly resistant) and unselected progeny of these crosses were sequenced using the Illumina GAIIx platform and reads were mapped back to the T. castaneum genome assembly (NCBI version 3.0). The outline of the crossing schemes, selections, read mapping and SNP calling criteria are described in more detail in Materials and Methods section. The Illumina sequencing results for the generational selected and unselected pools are outlined in Table 1.
Figure 1.
Outline of the crossing scheme and sequencing strategy.
Table 1.
Results summary of sequencing and read mapping
| Cross | Cohort/pool | Read length(bp) | No. reads | Matched reads | Total bases matched | Approx. coverage | No. SNPs called |
|---|---|---|---|---|---|---|---|
| SICS/SRB |
F4 Unselected |
36 (paired) |
49,636,554 |
28,119,799 |
1,012,312,764 |
5× |
556,683 |
| SICS/SRB |
F4 Selected |
36 (paired) |
52,622,706 |
28,155,197 |
1,013,587,092 |
5× |
297,483 |
| SICS/SRA |
F19 Unselected |
75 (unpaired) |
7,944,144 |
6,443,622 |
483,271,650 |
2.4× |
310,852 |
| SICS/SRA | F19 Selected | 75 (paired) | 15,643,072 | 12,246,096 | 918,457,200 | 4.5× | 602,068 |
Identification of candidate regions
In order to visualise regions linked to resistance, i.e. selected for homozygosity in phosphine exposed animals, we used a simple spreadsheet analysis of SNP output data from CLC Genomics Workbench. CLC Genomics Workbench outputs each SNP position, the primary variant for each position and the frequency of the primary variant, i.e. the ratio of the number of reads of the variant/total number of matching reads expressed as a percentage. For each generation’s (F4 and F19) datasets, we combined the two SNP output tables (selected and unselected) for each chromosome into the same spreadsheet and put the primary variant frequencies in separate columns for each dataset. The data tables were then sorted by SNP position, so that the two datasets were aligned in order along the chromosome/scaffold and then the primary variant frequencies were averaged over 300 cells in the spreadsheet for the selected and the unselected columns (Additional file 1: Table S1). The window size does not need to be fixed at 300, but 300 cells gave us the best resolution on these datasets, with the least amount of artefacts caused by low coverage. The averages were then plotted graphically (Figure 2). For the smaller, unassigned scaffolds, we averaged 50 cells or less, depending on how many SNPs were called on each scaffold.
Figure 2.
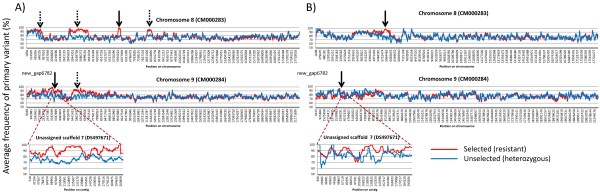
Average SNP frequency across Chr8 Chr9 and Uns-7, from A) F4 and B) F19 datasets (Selected vs Unselected). The window size used was an average of 300 cells (or 50 cells for Uns-7) in the spreadsheet analysis. The x-axis is a non-linear scale determined by the number of SNPs in the dataset. The regions showing linkage to resistance are highlighted by a dashed arrow; solid arrow indicates regions where the resistance loci were confirmed by fine-scale linkage mapping.
In this way, each plotted data point is an average of a virtual ‘dynamic window’, which changes in size on the corresponding chromosome sequence depending on the number of SNPs called. However the window at any point always covers the same region for both selected and unselected sets and moves down by one SNP each time. The actual physical distance which the dynamic window covers is determined by the span encompassed by the 300 contiguous SNPs in the window. This averaging causes artefacts due to very low SNP density, such as gaps in the scaffold sequence, or high SNP density such as SNP ‘clusters’, to be smoothed out by the analysis. The ideal window size cannot be calculated a priori, and is chosen empirically to reveal candidate regions which should be validated by detailed genetic analysis. The window size does not necessarily change with number of SNPs called, genome size or variant frequency. However, the length of the contig in fragmented genomes will affect the total number of SNPs called for a given contig, so smaller window sizes are preferable for shorter sequences. One consideration though, is that smaller window sizes are affected by the depth of coverage and can show up artefact differences between datasets that are caused by relatively low coverage. The depth of coverage can have large effects on individual SNP frequencies and sometimes regions can have very low coverage which can produce an average of ‘zero’ or be appear to be homozygous in smaller windows. Larger window sizes tend to ‘smooth out’ the analysis and account for SNPs that are missed. In general, there is an inverse relationship with the depth of sequencing coverage and the effective window size that will give reasonable resolution, i.e. the higher the depth of coverage, the better the estimation of SNP frequency will be for any particular SNP, and so the smaller the allowable window size.
The simple averaging method for genome wide analysis of SNP allele frequencies shows major regions linked to phosphine resistance displayed as differential SNP frequency caused by local regions of increased homozygosity in the selected dataset. The method does this without the aid of parentage assignment or other complex statistical calculation of association to the trait. The validity of the method is demonstrated by the fact that the regions found linked to resistance were confirmed in independent crosses from the same parental strains.
In the F4 data, four regions were found on Chr8, (region I: 274 to 896 Kbp, region II: 2.7 to 3.8 Mbp, region III: 5.83 to 6.01 Mbp and region IV: 7.96 to 8.54 Mbp), two regions in Chr9 (region I: 2.2 to 4.16 Mbp, region II: 5.07 to 7.2 Mbp) and one region in unassigned genomic scaffold 7 (DS497671), hereafter referred to as Uns-7, (86 to 200 Kbp), that constituted candidate regions associated with phosphine resistance in the T. castaneum genome (Figure 2). Analysis of the F19 data revealed that on Chr8, only one of the four previously identified regions was nearly homozygous and therefore confirmed to be linked to resistance (Figure 2). In the case of Chr9, neither candidate region previously identified in the F4 data was confirmed in the F19 analysis, i.e. no increase in the frequency of homozygosity of SNPs was observed in either genomic region in animals that survived exposure to phosphine (Figure 2). However, a region of the previously identified contig Uns-7 was retained as a candidate resistance locus (Figure 2). As a result, subsequent analysis was focused on region III in Chr8 and the candidate region in Uns-7. No other region of the genome showed an appreciable skew toward homozygosity of SNPs in selected versus unselected datasets of the F19 generation, indicating that no other resistance loci were present other than the locus on Chr8 and the locus on Uns-7 (Additional file 2: Figure S1).
Fine scale mapping
A subset of identified SNPs from Chr8 and Uns-7 that were homozygous in selected and heterozygous in unselected F19 datasets was chosen for further analysis (Additional file 3: Table S2). In addition, some Chr9 SNPs were included to determine whether the Uns-7 contig mapped to the candidate region of Chr9 that had been identified previously in the F4 generation. These SNPs were selected based on the availability of restriction enzymes that could digest the sequence of one allele, but not the other. The local regions around these SNPs were amplified and tested for polymorphism using the appropriate restriction enzymes (i.e. CAPS analysis). In one case there was an informative size polymorphism found after amplification and so was included in the analysis. These markers were analysed in the P0 parents of the SICS/SRA cross, the F1 pair used to establish the genetically segregating line, 96 individuals from the F19 generation that survived exposure to 0.8 mg L-1 phosphine and 160 individuals from the F28 generation that survived exposure to 0.5 mg L-1 phosphine. The naming convention used in this study describes tc as an acronym for T. castaneum, followed by the type of marker used (p) for a PCR-based size polymorphism or c for CAPS followed by their approximate genomic location in kilo (k) or mega (m) bases.
For Chr8, we also tested the linkage to resistance of three of the four identified regions, in 48 selected strongly resistant F4 individuals of the cross SICS/SRB that had survived at a high dose of 0.8 mg L-1. Linkage analysis of markers tcp8-777 k (Region I), tcc8-5.975 m (region III) and tcp8-8.11 m (region IV) on selected strongly resistant F4 individuals showed that they exhibited >75% linkage to resistance. The marker tcp8-777 k had 9 recombinants, while the marker tcp8-8.11 m had 7 recombinants, corresponding to estimated genetic distances of 3.1 cM and 2.5 cM from the resistance locus respectively. However, only one locus, tcc8-5.975 m, was absolutely linked to resistance in the F4 SICS/SRB cross and was the only one of the four regions that was identified in the F19 sequencing and confirmed further in fine scale mapping. This led us to conclude that there was only one locus on Chr8, in region III, and that we had not identified multiple resistance loci on Chr8 that were subsequently lost from the population between F4 and F19, either due to drift or pleiotropic effects on fitness. It also shows that the low coverage of sequencing identifies linked regions across several cM in the F4.
For mapping across the tightly linked Chr8 candidate region identified in the F19, SNP markers were designed across the entire candidate region, (Additional file 3: Table S2) and tested in 160 strongly resistant F28 individuals. Individuals that were non-recombinant for these SNP markers across the tc_rph1 candidate region were excluded from the analysis. The results of fine scale mapping defined a candidate region for tc_rph1 from tcc8-5.975 m to tcc8-6.04 m (Figure 3). Similarly, the marker tccU7-138.2 k with no recombinants in 83 strongly resistant selected F19 individuals confirmed the close linkage to second resistance locus, tc_rph2, on Uns-7 (Figure 4).
Figure 3.
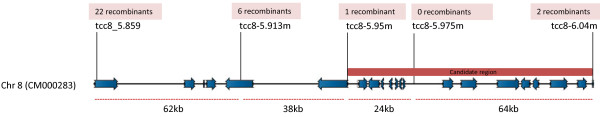
Fine-scale mapping of Chr8 defining the candidate region tc_rph1. The numbers of recombinants for each marker across the region are highlighted in pink, with the defined region containing resistance locus tc_rph1 in red.
Figure 4.
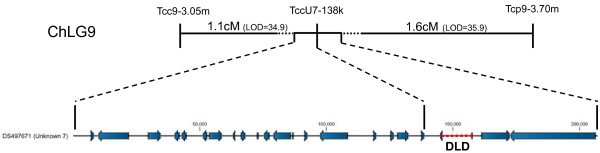
Linkage mapping of unassigned scaffold (Uns) - 7 postioning it on chromosome 9. The region highlighted in red on Uns-7 contains the known DLD resistance locus (tc_rph2).
Positioning the unassigned scaffold (Uns) - 7 into chromosome 9
The loss of homozygosity seen on Chr9 in the F19 dataset, compared to the observed homozygous peaks in F4 datasets, suggested that an error in genome sequence assembly had resulted in the omission of a segment of DNA sequence from Chr9 and this indicated that a resistance locus did indeed map to Chr9. In such a situation, the finer scale of the F19 genetic map would result in the loss of apparent linkage to Chr9. The fact that Uns-7 did contain a resistance locus and was not assigned to any particular site in the genome indicated that it could actually map to the region of Chr9 originally identified as a resistance locus in Chr9. To test this hypothesis, we mapped the relative genetic positions of three SNP markers: tcc9-3.05 m, tcp9-3.70 m (both from region I of Chr9) and tccU7-138.2 k, the marker for tc_rph2 locus (from Uns-7) in 92 individuals of an F2 population of the SICS/SRA cross (Figure 1). We found that marker tccU7-138.2 k is flanked by tcc9-3.05 m and tcp9-3.70 m, with map distances of 1.1 cM (LOD = 34.9) and 1.6 cM (LOD = 35.9), respectively, confirming our hypothesis that Uns-7 is a part of Chr9 (Figure 4), and most likely positioned in a gap of unknown size between two scaffolds (NW_001093536.1 and NW_001093382.1), designated ‘new_gap6782’ (Figure 2).
Candidate genes for phosphine resistance
The technique identified a target locus on the Uns-7 scaffold, even though it was not assembled into the larger genome and was itself incomplete and had many gaps in the sequence. This scaffold contains 19 predicted genes, of which two (TC006822 and TC006823) are actually the first and last exons of the dihydrolipoamide dehydrogenase (dld) gene (Table 2 and Figure 4). We used our paired end sequencing data to de novo assemble the region containing DLD, so that all the exons were represented in the genomic sequence. This assembled contig has been deposited into Genbank (KF032715). Comparative genetic analysis has confirmed this gene as the phosphine resistance locus rph2 in T. castaneum, R. dominica and the nematode, Caenorhabditis elegans[13]. This was taken as a validation of the SNP averaging technique.
Table 2.
List of candidate genes for phosphine resistance in Chr8 and Chr9 (Uns-7)
| Gene ID | Protein accession | Homology in Drosophila | E-value | Predicted biological function/identity |
|---|---|---|---|---|
|
Chr8 | ||||
| TC005925 |
EFA09201 |
CG5255 |
7.00E-28 |
Trypsin like serine protease H65 |
| TC005926 |
EFA08291 |
NA |
NA |
Unknown function |
| TC005927 |
EFA08292 |
CG13804 |
3.00E-150 |
Yellow-g2 |
| TC005928 |
EFA08293 |
CG6543 |
3.00E-158 |
Cyclohex-1-ene-1-carboxyl-CoA hydratase; enoyl CoA hydratase |
| TC005929 |
EFA08294 |
CG2663 |
3.00E-36 |
Sec14p-like lipid-binding domain; Alpha tocopherol transfer protein |
| TC005930 |
EFA08295 |
CG13096 |
1.00E-28 |
Ribosomal protein L1 |
| TC005931 |
EFA08296 |
NA |
NA |
Unknown function |
| TC005932 |
EFA08297 |
CG5670 |
0 |
Na(+)/K(+) ATPase alpha subunit |
| TC006224 |
EFA08569 |
CG12880 |
5.00E-29 |
Unknown function |
| TC006225 |
EFA08570 |
CG7463 |
1.00E-06 |
Yellow-k |
| TC006226 |
EFA08571 |
CG5717 |
3.00E-160 |
Yellow-g |
| TC006227 |
EFA08572 |
CG9792 |
1.00E-116 |
Yellow-e |
| TC006228 |
EFA08573 |
NA |
|
Unknown function |
| TC006229 |
EFA08574 |
CG9891 |
9.00E-89 |
Yellow-e3 |
| TC006230 |
EFA08575 |
CG1629 |
2.00E-132 |
Yellow-h |
| TC006231 |
EFA08576 |
CG13279 |
9.00E-104 |
Cytochrome b5 related; Fatty acid desaturase |
| TC006232 |
EFA08577 |
CG13279 |
1.00E-117 |
Cytochrome b5 related; Fatty acid desaturase |
|
Chr9 (Uns-7) | ||||
| TC006821 |
EFA13109 |
CG17514 |
0 |
Translational activator gcn-1, partial; Translator activator activity |
| TC006822 |
EFA13110 |
CG7430 |
1.00E-48 |
Dihydrolipoamide dehydrogenase E3 subunit |
| TC006823 |
EFA13111 |
CG7430 |
1.00E-82 |
Dihydrolipoamide dehydrogenase E3 subunit |
| TC006824 |
EFA13130 |
CG42698 |
7.00E-111 |
Pou domain motif 3 |
| TC006825 |
EFA13112 |
CG6551 |
3.00E-104 |
Serine/threonine protein kinase |
| TC006826 |
EFA13113 |
CG6551 |
8.00E-38 |
Serine/threonine protein kinase |
| TC006827 |
EFA13114 |
CG6551 |
6.00E-38 |
Serine/threonine protein kinase |
| TC006828 |
EFA13115 |
CG6178 |
5.00E-87 |
Fatty acetyl coA synthese activity |
| TC006829 |
NA |
CG11715 |
4.37E-08 |
Cytochrome P450; Cyp4g15 |
| TC006830 |
EFA13116 |
CG3800 |
1.00E-03 |
Polyprotein; Nucleic acid binding protein |
| TC006831 |
EFA13117 |
CG14660 |
3.90E-02 |
Putative serine protease; Labial associated factor |
| TC006832 |
EFA13118 |
CG3104 |
1.00E-18 |
Dopamine transporter; Ankyrin repeats involving in protein-protein interactions |
| TC006833 |
EFA13119 |
CG6178 |
5.00E-102 |
AMP binding enzyme; Fatty aceyl coA synthese activity |
| TC006834 |
EFA13120 |
CG6551 |
6.00E-17 |
Serine/threonine protein kinase; ATP Binding |
| TC006835 |
EFA13121 |
CG3104 |
2.00E-18 |
Dopamine transporter; Ankyrin repeats involving in protein-protein interactions |
| TC006836 |
EFA13122 |
NA |
NA |
Unknown function |
| TC006837 |
EFA13123 |
CG42231 |
3.00E-10 |
Unknown function; Uncharacterized conserved protein |
| TC006838 |
EFA13124 |
CG5245 |
6.25E-49 |
Nucleic acid/Zinc ion binding |
| TC006840 | EFA13125 | CG14938 | 3.00E-56 | Nucleic acid/Zinc ion binding |
The candidate region defined by markers tcc8-5.95 m and tcc8-6.04 m on Chr8 contains 17 gene predictions (Table 2 and Figure 3). While some of these genes have strong homologies to genes of known function, the functions of most of them are either ill-defined or unknown. Therefore further work is required to identify the resistance gene within this region.
Epistatic interactions between the two resistance loci
The relative contribution to the resistance phenotype of the two loci tc_rph1 and tc_rph2 was estimated by genotyping surviving F4 individuals of the cross SICS/SRA (Figure 1) exposed to a range of phosphine concentrations. Robust quantification of the relative strengths is difficult, but an estimate of the relative strengths of each genotype can be gleaned from Table 3 using the range of values that the LC99.9 is likely to fall between. The relative strength of the phenotype for each genotypic combination was SS (tc_rph1ss/tc_rph2ss, 0.02-0.03 mg L-1) < SR (tc_rph1rr/tc_rph2ss, 0.03-0.05 mg L-1) < RS (tc_rph1ss/tc_rph2rr, 0.06-0.1 mg L-1) < RR (tc_rph1rr /tc_rph2rr, >4.0 mg L-1). The LC99.9 value for the fully sensitive (QTC4) parental strain as calculated previously is 0.02 mg L-1[11]. Using this as a reference value we can estimate the individual contribution of tc_rph1 and tc_rph2, as approximately 1.5-2.5× and 3.0-5.0×, respectively. When homozygous for both resistance loci, the relative resistance ratio is >200×, demonstrating a strong synergistic interaction between the two loci.
Table 3.
Relative contribution of resistance loci, tc_rph1 and tc_rph2
| Genotypes | Dose of phosphine (mg L-1) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| tc_rph1 |
tc_rph2 |
US* |
0.008 |
0.01 |
0.02 |
0.03 |
0.05 |
0.06 |
0.1 |
0.2 |
0.5 |
0.8 |
1 |
2 |
3 |
4 |
|
ss |
ss |
6 |
1 |
0 |
1 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
sr |
ss |
16 |
11 |
6 |
4 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
rr |
ss |
15 |
8 |
6 |
15 |
9 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
ss |
sr |
8 |
4 |
2 |
2 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
sr |
sr |
25 |
21 |
29 |
5 |
1 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
rr |
sr |
18 |
16 |
19 |
31 |
19 |
5 |
12 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
ss |
rr |
3 |
0 |
2 |
1 |
1 |
7 |
2 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
|
sr |
rr |
2 |
3 |
3 |
2 |
10 |
9 |
6 |
8 |
1 |
3 |
0 |
0 |
0 |
0 |
0 |
|
rr |
rr |
0 |
1 |
4 |
7 |
4 |
11 |
3 |
5 |
7 |
7 |
4 |
3 |
3 |
3 |
1 |
|
No. analysed |
93 |
65 |
71 |
68 |
44 |
32 |
24 |
13 |
8 |
10 |
4 |
3 |
4 |
3 |
1 |
|
| PCR amplified |
96 |
90 |
71 |
68 |
44 |
32 |
28 |
14 |
10 |
10 |
4 |
4 |
4 |
3 |
2 |
|
| Survivors |
202 |
107 |
155 |
90 |
75 |
40 |
30 |
14 |
11 |
11 |
4 |
6 |
4 |
3 |
3 |
|
| Total no.of insects bioassayed | 202 | 130 | 201 | 201 | 200 | 313 | 204 | 295 | 312 | 300 | 320 | 302 | 310 | 300 | 320 | |
*US = Unselected beetles.
Relative fitness of tc_rph1 and tc_rph2
We estimated the change in frequency of resistance alleles in the absence of phosphine selection over 18 generations, from F2 to F20, using the markers tcc8-5.975 m (tc_rph1) and tccU7-138.2 k (tc_rph2) and analysed deviation from Hardy-Weinberg equilibrium using chi-square test (Table 4). The results showed a steady increase in the homozygous-resistant genotype tc_rph1rr in each generation from F5 to F20 (χ2 = 18.8; P < 0.001, df = 1), indicating that tc_rph1 may have a selective fitness advantage associated with phosphine resistance. In contrast, tc_rph2 showed a significant decrease in resistant homozygotes (tc_rph2rr) coupled with a steady increase in sensitive homozygotes (tc_rph2ss) over 18 generations (χ2 = 75.2, P < 0.001, df = 1) indicating a strong fitness cost associated with this resistance allele in the absence of selection.
Table 4.
Fitness analysis of resistance loci, tc_rph1 and tc_rph2 over 18 generations in the absence of phosphine selection
| Generations | Insects tested |
Genotypes |
Allelic frequency |
Chi-square analysis on genotypes† |
||||
|---|---|---|---|---|---|---|---|---|
| rr | rs | ss | p allele | q allele | χ2(df = 1) | P value | ||
|
tc_rph1 | ||||||||
| F2 |
94 |
28 |
41 |
27 |
0.51 |
0.49 |
2.15 |
0.143 |
| F5 |
94 |
34 |
43 |
17 |
0.59 |
0.41 |
6.83* |
0.009 |
| F10 |
96 |
38 |
45 |
13 |
0.63 |
0.37 |
13.4** |
<0.001 |
| F15 |
95 |
35 |
46 |
14 |
0.61 |
0.39 |
9.38* |
0.002 |
| F20 |
96 |
38 |
48 |
8 |
0.65 |
0.34 |
18.8** |
<0.001 |
|
tc_rph2 | ||||||||
| F2 |
94 |
20 |
52 |
22 |
0.49 |
0.51 |
1.15 |
0.283 |
| F5 |
96 |
5 |
54 |
37 |
0.33 |
0.67 |
22.8** |
<0.001 |
| F10 |
92 |
7 |
38 |
45 |
0.29 |
0.71 |
33.6** |
<0.001 |
| F15 |
96 |
0 |
28 |
64 |
0.15 |
0.85 |
99.0** |
<0.001 |
| F20 | 96 | 0 | 36 | 58 | 0.19 | 0.81 | 75.2** | <0.001 |
* Significant (P < 0.01); ** Significant (P < 0.001).
† Significance values are calculated from deviation from Hardy-Weinberg equilibrium compared to expected F2 (1:2:1) ratio.
Discussion
Finding single nucleotide changes that cause large phenotype changes in highly polymorphic genetic backgrounds is a formidable task. In the present study we have taken advantage of next-generation sequencing technology and the published reference genome sequences of T. castaneum to rapidly identify the genomic locations of phosphine resistance loci in T. castaneum. Our results demonstrate the power of our simple averaging method to scan for local regions of loss of heterozygosity, which is preferable to standard linkage mapping for identifying gene regions responsible for well-defined traits in advanced intercrosses. The method does not rely on a high quality reference genome, high depth of sequence coverage, use of inbred strains or parentage assignment of SNPs. The whole genome including multiple unassigned scaffolds can be easily scanned to identify linked regions.
The simple averaging method also has advantages in that it can reduce artefacts caused by gaps in reference genome assemblies. Large gaps that are included in the assembled sequence, as is the case for T. castaneum, can cause major problems for analyses that rely on defined physical distances, as there would appear to be no variants or genes within these gaps. Variable SNP density can also cause problems for analyses that rely on defined physical distances. Our technique eliminates this problem by allowing the SNPs in the datasets to define the region to be averaged and how far the analysis window is shifted along the chromosome for each iteration, rather than a user-defined physical distance. This is easily visualised with spreadsheet software, such as Excel™.
Our analysis defined two genomic regions on Chr8 and Chr9 (Uns-7) containing loci responsible for high-level resistance, tc_rph1 and tc_rph2. The results of F4 SNP analysis showed discontinuous SNP frequency signals in the candidate regions of both Chr8 and Chr9, represented as separate peaks in the F4 SNP frequency graphs (Figure 2) and suggested the possible disorientation or erroneous order of chromosomal scaffolds in the reference genome, either due to lack of genetic markers or low recombination in these regions in the crosses used to create the linkage map [15]. Our inheritance analysis of selected SNP markers from Chr8, Chr9 and Uns-7 on a F2 mapping population confirmed this and identified the location of Uns-7 on Chr9 (new_gap6782). Similar chromosomal sequence discrepancies on the T. castaneum reference genome especially on Chr9 were observed by Wang et al.[16] and the respective sequences were later rearranged to their proper location.
Candidate genes
Our next-generation sequencing and mapping results show that the major candidate resistance gene for tc_rph2 is on Uns-7. This scaffold contains the dihydrolipoamide dehydrogenase (dld) gene, which was confirmed as a resistance locus using more traditional methods that required much greater effort and expense [13]. This demonstrates the validity, power and efficiency of the simple averaging method. All sequence scaffolds, including those not assigned to chromosomes can be scanned independently and rapidly for linkage to a trait. Since more than 22 Mb (10%) of the T. castaneum genome sequence is still unassigned to genomic locations, this represents a powerful way of detecting linkage, even across smaller regions in fragmented genome assemblies.
At the tc_rph1 locus, the candidate gene list contains 17 predicted genes. However, none of them are associated with the same pathways as dld, consistent with the hypothesis of strongly resistant insects having at least two different mechanisms of resistance to phosphine [2,17-20]. Although it is difficult to discuss the potential roles of each of the candidates, genes in the mapped interval are involved in lipid metabolism, chitin metabolism, membrane permeability and maintaining ion gradients. A resistance gene in any of these pathways would be consistent with previous observations on phosphine mode of action through oxidative stress and lipid peroxidation and resistance through reduced uptake [13,19-24].
Epistatic interactions between the two resistance loci
We used markers tightly linked to resistance loci tc_rph1 and tc_rph2 to determine their relative phenotypic effects and genetic interactions in their homozygous and heterozygous states. In the genotype analysis, we showed that the resistance locus tc_rph2 exhibited a stronger phenotypic effect compared to tc_rph1. Both resistance loci are incompletely recessive but when both in a homozygous state they interact synergistically with resistance levels greater than 200×. This is a conservative estimate based on survival of the rph1rr/rph2rr genotype at doses up to 4.0 mg L-1, the highest dose tested. These results are in accordance with previous results showing synergistic interactions between resistance genes with resistance levels up to 431×, estimated using LC50 response values [11]. The synergism observed between the resistance alleles of T. castaneum resembles the synergistic interaction of rph1 and rph2 in R. dominica[6]. These results are also consistent with the two gene model of high-level phosphine resistance seen in R. dominica[6,12,25].
Pleiotropic effects on fitness
Previous analysis [11] on the relative change in the resistance phenotype in the absence of phosphine selection showed the existence of a possible fitness disadvantage in a T. castaneum population segregating for strong resistance alleles. In the present study, we observed a significant decrease in the frequency of the tc_rph2rr homozygous resistant genotype over 18 generations with a corresponding increase in heterozygote (tc_rph2rs) and susceptible genotypes (tc_rph2ss). This directly shows the presence of significant selective fitness disadvantage for homozygotes at the tc_rph2 locus. Field population based fitness studies [26-29] on phosphine resistant insect strains of R. dominca, T. castaneum, Sitophilus oryzae, Cryptolestes ferrugineus and Oryzaephilus surinamensis reported the existence of biological fitness cost associated with phosphine resistance.
In contrast to tc_rph2, we observed a significant increase in the frequency of the homozygous resistant allele of tc_rph1, suggesting a selective fitness advantage of weakly resistant homozygotes (tc_rph1rr) compared to susceptible genotypes. These results are consistent with previous fitness estimates on weakly resistant strains of T. castaneum, R. dominica and S. oryzae[30] and shows that there are fundamental differences between the genes that are associated with each resistance locus.
Conclusion
The results of the present study used a whole genome resequencing approach and we validated our method of processing the sequences for rapid scanning of polymorphisms and identifying the candidate genes associated with phosphine resistance trait in T. castaneum by linkage mapping. Although we have only validated the technique for a strongly selected and previously characterized trait, theoretically it is transferrable to nearly any species that has some genomic or transcriptomic reference data against which reads may be aligned, whose breeding may be manipulated for the generation of specific populations for linkage mapping. The detailed genotype analysis using identified SNP markers demonstrated that high-level resistance in T. castaneum is a result of two synergistic genes, tc_rph1 and tc_rph2 with the latter having significant fitness cost. The fine-scale mapping analysis also defined the genomic regions containing these loci. The results show the validity of the technique and that it can be applied in cases where parental genotypes are unknown and in circumstances where the genome coverage conferred by sequencing is relatively low, which would often be the case for organisms with large genomes.
Methods
Beetle strains
Two strains of T. castaneum were used, a phosphine susceptible strain, QTC4 (‘S’ strain) and a strongly resistant strain, QTC931 (Strong-R). The ‘S’ strain was derived from adults collected from a storage facility in Brisbane, southeast Queensland, Australia in 1965 [31] and since then, the population of this strain has been maintained in the laboratory without exposure to phosphine and is fully susceptible to phosphine. The Strong-R strain was derived from adults collected from central storage at Natcha, southeast Queensland, and Australia in 2000 [11]. Homozygosity for resistance was selected within the ‘Strong-R’ strain by phosphine selection for at least five generations. In addition, both S strain and Strong-R exhibit a linear response to phosphine in a probit mortality response analysis, indicating their respective homozygosity for the susceptibility/resistance trait [11]. The insects were cultured in whole wheat flour and yeast 20:1 and maintained at 30°C and 55% relative humidity (RH).
Genetic crosses
Two similar single pair inter-strain crosses (SIC), SICS/SRA and SICS/SRB (both S ♀ X Strong-R ♂) were established (Figure 1) between these strains as described previously [11]. For each cross, single virgin adult male and female siblings of the F1 progeny were mated to derive 194 F2 individuals, which were allowed to mate freely for several weeks to produce the F3 generation. After producing the F3 generation, 94 F2 individuals were retained and used for linkage mapping and SNP validation. The F3 and subsequent progeny were kept as discrete generational cohorts up to the F28 (Figure 1), and each further generational cohort consisted of approximately 500 to 1000 beetles. The SICS/SRA cross was established independently 20 months before the SICS/SRB cross and aimed to establish genetic map for T. castaneum. The latter (SICS/SRB) cross was established for the express purpose of genome resequencing to identify larger candidate regions that could then be fine-scale mapped in the later generational cohorts (F19) of the SICS/SRA cross without having to wait another 16 months for the generations to progress. As parental strains, S and Strong-R of these two crosses were highly homozygous in responding to the resistance trait, these two crosses were considered as a replicates.
Fumigation
Phosphine gas generation and all fumigation bioassays were performed for 20 hours at 25°C in sealed airtight desiccators as described previously by Daglish et al.[32] and Jagadeesan et al.[11].
DNA extraction and sample preparation for sequencing
A high discriminatory dose (0.8 mg L-1) selection was performed to isolate individuals that were homozygous for resistance trait by exposing more than 10,000 F4 progeny of the cross SICS/SRB and 20,000 F19 progeny of the cross SICS/SRA (Figure 1). For each cross, total genomic DNA was extracted from 100 survivors of each selection and 100 unselected beetles. The DNA was extracted using a QIAGEN DNeasy extraction kit according to the manufacturer’s protocol. The F4 samples were sequenced using the Illumina GAIIx platform with paired-end reads of 35 bp and the F19 was subsequently sequenced with 75 bp (paired end for selected, unpaired for unselected) (Figure 1).
DNA extraction and sample preparation for SNP genotyping and mapping
A modified high-salt DNA extraction procedure [33] was adopted for genotyping individual insects. Genomic DNA was extracted from the P0 parents, F1 hybrids, F2, F4, F19 and F28 individuals of the SICS/SRA intercross for fine-scale mapping of the resistance locus. Briefly, individual insects were placed in a 96-well PCR microtitre plate, snap frozen in liquid nitrogen and homogenized in 100 μL lysis buffer (0.4 M NaCl, 10 mM Tris–HCl pH 8.0 and 2 mM EDTA pH 8.0) using a multi pin header, for 10-15 s. Then 4 μL of 10% SDS (4% final concentration) and 1 μL of 10 mg ml-1 proteinase K (100 μg ml-1 final concentration) were added to each sample and mixed well. The samples were incubated at 37°C for overnight, after which 25 μL of 6 M NaCl (NaCl saturated H2O) was added to each. Samples were vortexed for 30 s at maximum speed, and tubes centrifuged for 45 min at 1500 g. The supernatant was transferred to fresh tubes. To each sample, an equal volume of ice cold 100% ethanol was added, mixed well, and then centrifuged for 45 min, at 1500 g. The pellet was washed with 70% ethanol, air dried and resuspended in 25 μL sterile distilled H2O. Finally, the samples were kept at 65°C for 10 min to denature any residual DNAses. The genomic DNA template was subsequently quantified and diluted to 2 ng μL-1 before inclusion in PCR.
Mapping short reads to the T. castaneum reference genome
The selected and unselected re-sequenced data sets from F4 and F19 progeny of the crosses SICS/SRB and SICS/SRA (Figure 1) were mapped separately to the reference T. castaneum genome (version Tcas_3.0, from NCBI) using CLCBio Genomics Workbench v4.9 software [34]. The F4 and F19 sequence data sets were mapped to the reference under the read mapping algorithm. Read mapping was performed with high stringency, requiring a perfect match and no unaligned ends with the following default parameters, Match +1 Mismatch − 2; Deletion − 3 and Insertion − 3.
SNP detection
Quality-based SNP detection in CLC Genomics Workbench is based on the Neighbourhood Quality Standard (NQS) algorithm described by [35,36]. For a SNP to be called, the default quality specifications such as window length (11), maximum number of gaps and mismatches (2), minimum average quality of surrounding bases (15) and minimum quality of the central base (20) were chosen together with a minimum coverage of six and minimum variant frequency of 35% within a pool.
The SNP detection output tables for selected and unselected F4 and F19 datasets obtained were directly exported to a separate Excel™ spreadsheet. SNP datasets for selected (resistant) and unselected (segregating) progeny were compared by separating the data by generation and chromosome, and sorted according to the reference base position and with the frequency of the primary variant, i.e. the most common allele for a SNP regardless of reference base identity, for the selected and unselected datasets being in separate columns. The frequency of the primary variant was then averaged over 300 cells in the spreadsheet for each dataset for chromosomes and 40 cells for unassigned scaffolds, given that the number of SNPs/scaffold is greatly reduced due to their smaller sizes.
SNP validation and analysis
A subset of SNPs were selected from identified candidate gene regions for further investigation from regions where the averaged SNP allelic frequency attained near homozygosity (90-100%) in selected datasets but remained heterozygous (~50-80%) in the unselected datasets (Additional file 3: Table S2 and Figure 1). SNPs of interest that were identified to be on restriction enzyme (RE) recognition sites, and thus amenable to CAPS analysis, were amplified from individual insects by PCR. For each insect, 4 ng of template DNA was amplified in a reaction volume of 25 μL containing: 1× PCR buffer, 4 mM MgCl2, 0.8 mM dNTPs (New England Biolabs), 1.25 U μl-1 Taq DNA polymerase (Bioline), 0.4 μM specific forward and reverse primers. The PCR conditions were: denaturation for 2 min at 95°C; then 40 cycles of 95°C for 45 s, 52°C for 45 s, 72°C for 45 s with a final extension at 72°C for 2 min. Restriction digestions were carried out in 15 μL final volume containing 5 μL of the PCR product, 1× concentrated restriction enzyme buffer with optional 1× Bovine Serum Albumin (BSA), and 1-3 U of RE for at least one hour to overnight at the approriate temperature. Products were analysed by 2.5% agarose gel electrophoresis (1× TAE, 200 V for 30 min).
Mapping of candidate regions
The CAPS markers from candidate regions in Chr8 (Genbank accession CM000283), Chr9 (CM000284) and Uns-7 (DS497671) were genotyped in parents and F1 for polymorphism and tested for Mendalian segregation in the segregating F2. The F2 genotypes were analysed by Map Manager QTXb20 and tested for 1:2:1 segregation of parental alleles using G test at 95% confidence interval [37,38]. Marker order was determined with a LOD score threshold of 3.0 and map distances were estimated by the Kosambi function [39].
The fine scale mapping was carried out on markers linked to resistance loci tc_rph1 and tc_rph2 to estimate the distance between each marker and their respective target genes. This was achieved by genotyping the segregation profile of markers linked to tc_rph1 and tc_rph2 in survivors of F4 (SICS/SRB), F19 (SICS/SRA) selected at high discriminating dose of 0.8 mg L-1 and F28 (SICS/SRA) progeny at 0.5 mg L-1, respectively (Figure 1). We reduced the discriminatory dose from 0.8 to 0.5 mg L-1 for the selection at F28 progenies, owing to the relatively small size of population of 4700 beetles produced in this generation and moreover, this will not affect selecting strongly resistant homozygous insects [11]. Surviving individuals, not homozygous for the resistance alleles (tc_rph1 and tc_rph2) were scored as recombinant at that locus. Individuals who were heterozygous across the candidate region were excluded from the analysis.
Determination of relative contribution and fitness cost of tc_rph1 and tc_rph2
To identify the relative contributions of resistance loci, tc_rph1 and tc_rph2, a selection at series of concentrations of phosphine from 0.008, 0.01, 0.02, 0.03, 0.05, 0.06, 0.1, 0.2, 0.5, 0.8, 1.0, 2.0, 3.0 and 4.0 mg L-1, each with approximately 150–200 insects of F4 progeny of the cross SICS/SRA was performed (Figure 1) and the surviving individuals at each concentration were genotyped using the respectively linked SNP markers, tcc8-5.975 m and tccU7-138.2 k. The effect of resistance phenotypes for different allelic combinations was measured by comparing the genotype pattern of these markers over increasing concentrations of phosphine. The same markers were also used to investigate changes in allele frequency associated with resistant (rr) and sensitive (ss) alleles of both the loci tc_rph1 and tc_rph2 in the absence of selection. The beetle populations not exposed to phosphine (unselected) were sampled randomly (~96) at discrete generations, F5, F10, F15 and F20 and genotyped for both the resistance loci to determine any observable fitness/heritability effects associated with resistance loci in the absence of selection using a chi-square test.
Abbreviations
Bp: Base pairs; CAPS: Cleaved Amplified Polymorphic Sequences; Chr: Chromosome; dNTPs: Deoxy Nucleotide Triphosphates; Gbp: Gig base pairs; Kbp: Kilo base pairs; LOD: Logarithmic odd score; Mbp: Mega base pairs; NQS: Neighbourhood Quality Score; PCR: Polymerised Chain Reaction; PH3: Phosphine; RE: Restriction Enzyme; S/SRA: Cross A: Susceptible X Strong-R (A); S/SRB: Cross B: Susceptible X Strong-R (B); SIC: Single pair Inter-strain Cross; SNPs: Single Nucleotide Polymorphisms; TAE: Tris-acetate-EDTA [Ethylenediaminetetraacetic acid]; tc_rph1: Gene1 conferring resistance to phosphine in Tribolium castaneum; tc_rph2: Gene2 conferring resistance to phosphine in Tribolium castaneum; U: Units; Uns: Unassigned genomic scaffold; V: Volts.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
RJ, DS and PE designed the study. Bioinformatic analyses were performed by DS with input from RJ. RJ performed the genetic crosses, genome sequencing and linkage mapping experiments and AF contributed to the fine scale mapping. RJ and DS drafted the manuscript; and PE revised the manuscript and provided critical comments. All authors read and approved the final manuscript.
Supplementary Material
An example table for averaging variant (SNP) frequency for chromosome 8 for the F4 generation Selected vs Unselected datasets in an Excel™ spreadsheet.
Illustrating the average SNP frequency across all chromosomes from F4 and F19 datasets (Selected vs Unselected).
Listing markers used for mapping resistance loci on Chr8 and Chr9 (Uns-7).
Contributor Information
Rajeswaran Jagadeesan, Email: Raj.Jagadeesan@daff.qld.gov.au.
Amelia Fotheringham, Email: meliebites@gmail.com.
Paul R Ebert, Email: p.ebert@uq.edu.au.
David I Schlipalius, Email: david.schlipalius@daff.qld.gov.au.
Acknowledgements
The authors would like to acknowledge the support of the Australian Government’s Cooperative Research Centres programme. RJ received an International Postgraduate Research Scholarship (IPRS) from the University of Queensland. We also thank all the staff members of the Postharvest Grain Protection Team at the Department of Agriculture, Fisheries and Forestry (DAFF) Queensland for providing insect strains and technical support.
References
- Earley EJ, Jones CD. Next-generation mapping of complex traits with phenotype-based selection and introgression. Genetics. 2011;189:1203-+. doi: 10.1534/genetics.111.129445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaudhry MQ. A review of the mechanisms involved in the action of phosphine as an insecticide and phosphine resistance in stored-product insects. Pestic Sci. 1997;49:213–228. doi: 10.1002/(SICI)1096-9063(199703)49:3<213::AID-PS516>3.0.CO;2-#. [DOI] [Google Scholar]
- Bell CH. Fumigation in the 21st century. Crop Prot. 2000;19:563–569. doi: 10.1016/S0261-2194(00)00073-9. [DOI] [Google Scholar]
- Fields PG, White NDG. Alternatives to methyl bromide treatments for stored-product and quarantine insects. Annu Rev Entomol. 2002;47:331–359. doi: 10.1146/annurev.ento.47.091201.145217. [DOI] [PubMed] [Google Scholar]
- Nayak OK, Holloway JC, Emery RN, Pavic H, Bartlet J, Collins PJ. Strong resistance to phosphine in the rusty grain beetle, Cryptolestes ferrugineus (Stephens) (Coleoptera: Laemophloeidae): its characterisation, a rapid assay for diagnosis and its distribution in Australia. Pest Manag Sci. 2012;69:48–53. doi: 10.1002/ps.3360. [DOI] [PubMed] [Google Scholar]
- Schlipalius DI, Chen W, Collins PJ, Nguyen T, Reilly P, Ebert PR. Gene interactions constrain the course of evolution of phosphine resistance in the lesser grain borer, Rhyzopertha dominica. Heredity. 2008;100:506–516. doi: 10.1038/hdy.2008.4. [DOI] [PubMed] [Google Scholar]
- Rajendran S, Parveen H, Begum K, Chethana R. Influence of phosphine on hatching of Cryptolestes ferrugineus (Coleoptera: Cucujidae), Lasioderma serricorne (Coleoptera: Anobiidae) and Oryzaephilus surinamensis (Coleoptera: Silvanidae) Pest Manag Sci. 2004;60:1114–1118. doi: 10.1002/ps.931. [DOI] [PubMed] [Google Scholar]
- Lorini I, Collins PJ, Daglish GJ, Nayak MK, Pavic H. Detection and characterisation of strong resistance to phosphine in Brazilian Rhyzopertha dominica (F.) (Coleoptera: Bostrychidae) Pest Manag Sci. 2007;63:358–364. doi: 10.1002/ps.1344. [DOI] [PubMed] [Google Scholar]
- Wang DX, Collins PJ, Gao XW. Optimising indoor phosphine fumigation of paddy rice bag-stacks under sheeting for control of resistant insects. J Stored Prod Res. 2006;42:207–217. doi: 10.1016/j.jspr.2005.02.001. [DOI] [Google Scholar]
- Daglish GJ, Kopittke RA, Cameron MC, Pavic H. Predicting mortality of phosphine-resistant adults of Sitophilus oryzae (L) (Coleoptera: Curculionidae) in relation to changing phosphine concentration. Pest Manag Sci. 2004;60:655–659. doi: 10.1002/ps.800. [DOI] [PubMed] [Google Scholar]
- Jagadeesan R, Collins PJ, Daglish GJ, Ebert P, Schlipalius DI. Phosphine resistance in the rust red flour beetle, Tribolium castaneum (Coleoptera: Tenebrionidae): inheritance, gene interactions and fitness costs. PLos one. 2012;7:e3152. doi: 10.1371/journal.pone.0031582. 3151-3112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins PJ, Daglish GJ, Bengston M, Lambkin TM, Pavic H. Genetics of resistance to phosphine in Rhyzopertha dominica (Coleoptera: Bostrichidae) J Econ Entomol. 2002;95:862–869. doi: 10.1603/0022-0493-95.4.862. [DOI] [PubMed] [Google Scholar]
- Schlipalius DI, Valmas N, Tuck AG, Jagadeesan R, Ma L, Kaur R, Goldinger A, Anderson C, Kuang J, Zuryn S. et al. A core metabolic enzyme mediates resistance to phosphine gas. Science. 2012;338:807–810. doi: 10.1126/science.1224951. [DOI] [PubMed] [Google Scholar]
- Richards S. The genome of the model beetle and pest Tribolium castaneum. Nature. 2008;452:949–955. doi: 10.1038/nature06784. [DOI] [PubMed] [Google Scholar]
- Lorenzen MD, Doyungan Z, Savard J, Snow K, Crumly LR, Shippy TD, Stuart JJ, Brown SJ, Beeman RW. Genetic linkage maps of the red flour beetle, Tribolium castaneum, based on bacterial artificial chromosomes and expressed sequence tags. Genetics. 2005;170:741–747. doi: 10.1534/genetics.104.032227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S, Lorenzen MD, Beeman RW, Brown JS. Analysis of repetitive DNA distribution patterns in Tribolium castaneum genome. Genome Biol. 2008;9:1–14. doi: 10.1186/gb-2008-9-3-r61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlipalius DI, Collins PJ, Mau Y, Ebert PR. New tools for management of phosphine resistance. Outlooks Pest Manag. 2006;17:52–56. doi: 10.1564/16apr02. [DOI] [Google Scholar]
- Benhalima H, Chaudhry MQ, Mills KA, Price NR. Phosphine resistance in stored-product insects collected from various grain storage facilities in Morocco. J Stored Prod Res. 2004;40:241–249. doi: 10.1016/S0022-474X(03)00012-2. [DOI] [Google Scholar]
- Hsu CH, Chi BC, Liu MY, Li JH, Chen CJ, Chen RY. Phosphine-induced oxidative damage in rats: role of glutathione. Toxicology. 2002;179:1–8. doi: 10.1016/S0300-483X(02)00246-9. [DOI] [PubMed] [Google Scholar]
- Hsu CH, Han BC, Liu MY, Yeh CY, Casida JE. Phosphine-induced oxidative damage in rats: attenuation by melatonin. Free Radic Biol Med. 2000;28:636–642. doi: 10.1016/S0891-5849(99)00277-4. [DOI] [PubMed] [Google Scholar]
- Chaudhry MQ, Price NR. Biochemistry of phosphine uptake in susceptible and resistant strains of two species of stored product beetles. Comp B Physiol C Pharmacol Toxicol Endocrinol. 1989;94:425–430. doi: 10.1016/0742-8413(89)90092-3. [DOI] [Google Scholar]
- Chaudhry MQ, Price NR. Comparison of the oxidant damage induced by phosphine and the uptake and tracheal exchange of phosphorus32radiolabelled phosphine in the susceptible and resistant strains of Rhyzopertha dominica F. (Coleoptera: Bostrychidae) Pestic Biochem Physiol. 1992;42:167–179. doi: 10.1016/0048-3575(92)90063-6. [DOI] [Google Scholar]
- Hsu CH, Chi BC, Casida JE. Melatonin reduces phosphine-induced lipid and DNA oxidation in vitro and in vivo in rat brain. J Pineal Res. 2002;32:53–58. doi: 10.1034/j.1600-079x.2002.10809.x. [DOI] [PubMed] [Google Scholar]
- Hsu CH, Quistad GB, Casida JE. Phosphine-induced oxidative stress in Hepa 1c1c7 cells. Toxicol Sci. 1998;46:204–210. doi: 10.1093/toxsci/46.1.204. [DOI] [PubMed] [Google Scholar]
- Schlipalius DI, Cheng Q, Reilly PEB, Collins PJ, Ebert PR. Genetic linkage analysis of the lesser grain borer, Rhyzopertha dominica identifies two loci that confer high-level resistance to the fumigant phosphine. Genetics. 2002;161:773–782. doi: 10.1093/genetics/161.2.773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pimentel MAG, Faroni LRD, Totola MR, Guedes RNC. Phosphine resistance, respiration rate and fitness consequences in stored-product insects. Pest Manag Sci. 2007;63:876–881. doi: 10.1002/ps.1416. [DOI] [PubMed] [Google Scholar]
- Pimentel MAG, Faroni LRD, Guedes RNC, Sousa AH, Totola MR. Phosphine resistance in Brazilian populations of Sitophilus zeamais Motschulsky (Coleoptera: Curculionidae) J Stored Prod Res. 2009;45:71–74. doi: 10.1016/j.jspr.2008.09.001. [DOI] [Google Scholar]
- Sousa AH, Faroni LRD, Pimentel MAG, Guedes RNC. Developmental and population growth rates of phosphine-resistant and susceptible populations of stored-product insect pests. J Stored Prod Res. 2009;45:241–246. doi: 10.1016/j.jspr.2009.04.003. [DOI] [Google Scholar]
- Pimentel MAG, Faroni LR, Silva FH, Batista MD, Guedes RN. Spread of phosphine resistance among brazilian populations of three species of stored product insects. Neotrop Entomol. 2010;39:1–7. doi: 10.1590/S1519-566X2010000100001. [DOI] [PubMed] [Google Scholar]
- Collins PJ, Daglish GJ, Nayak MK, Ebert PR, Schlipalius DI, Chen W. In: International conference on controlled atmoshphere and fumigation in stored products. Donahaye EJ, Navarro S, Leesch JG, editor. Fresno, CA: Executive Printing Services; 2001. Combating resistance to phosphine in Australia; pp. 593–607. [Google Scholar]
- Bengston M, Collins PJ, Daglish GJ, Hallman VL, Kopittke R, Pavic H. Inheritance of phosphine resistance in Tribolium castaneum (Coleoptera: Tenebrionidae) J Econ Entomol. 1999;92:17–20. [Google Scholar]
- Daglish GJ, Collins PJ, Pavic H, Kopittke RA. Effects of time and concentration on mortality of phosphine-resistant Sitophilus oryzae (L) fumigated with phosphine. Pest Manag Sci. 2002;58:1015–1021. doi: 10.1002/ps.532. [DOI] [PubMed] [Google Scholar]
- Miller SA, Dykes DD, Polesky HF. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acid Res. 1988;16:1215. doi: 10.1093/nar/16.3.1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CLC genomics workbench. http://www.clcbio.com/
- Atshuler D, Pollara VJ, Cowles CR, Van Etten EJ, Baldwin J, Linton L, Lander ES. An SNP map of the human genome generated by reduced representation shotgun sequencing. Nature. 2000;407:513–516. doi: 10.1038/35035083. [DOI] [PubMed] [Google Scholar]
- Brockman W, Alvarez P, Young S, Garber M, Giannoukos G, Lee WL, Russ C, Lander ES, Nusbaum C, Jaffee DB. Quality scores and SNP detection in sequencing by synthesis systems. Genome Res. 2008;18:763–770. doi: 10.1101/gr.070227.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manly KF. Overview of QTL mapping software and introduction to map manger QT. Mamm Genome. 1999;10:327–334. doi: 10.1007/s003359900997. [DOI] [PubMed] [Google Scholar]
- Manly KF. Map manager QTX, cross-platform software for genetic mapping. Mamm Genome. 2001;12:930–932. doi: 10.1007/s00335-001-1016-3. [DOI] [PubMed] [Google Scholar]
- Kosambi DD. The estimation of map distance from recombination value. Ann Eugenics. 1944;12:172–175. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
An example table for averaging variant (SNP) frequency for chromosome 8 for the F4 generation Selected vs Unselected datasets in an Excel™ spreadsheet.
Illustrating the average SNP frequency across all chromosomes from F4 and F19 datasets (Selected vs Unselected).
Listing markers used for mapping resistance loci on Chr8 and Chr9 (Uns-7).