Abstract
As the theory of stem cell plasticity was first proposed, we have explored an alternative hypothesis for this phenomenon: namely that adult bone marrow (BM) and umbilical cord blood (UCB) contain more developmentally primitive cells than hematopoietic stem cells (HSCs). In support of this notion, using multiparameter sorting we were able to isolate small Sca1+Lin−CD45− cells and CD133+Lin−CD45− cells from murine BM and human UCB, respectively, which were further enriched for the detection of various early developmental markers such as the SSEA antigen on the surface and the Oct4 and Nanog transcription factors in the nucleus. Similar populations of cells have been found in various organs by our team and others, including the heart, brain and gonads. Owing to their primitive cellular features, such as the high nuclear/cytoplasm ratio and the presence of euchromatin, they are called very small embryonic-like stem cells (VSELs). In the appropriate in vivo models, VSELs differentiate into long-term repopulating HSCs, mesenchymal stem cells (MSCs), lung epithelial cells, cardiomyocytes and gametes. In this review, we discuss the most recent data from our laboratory and other groups regarding the optimal isolation procedures and describe the updated molecular characteristics of VSELs.
Keywords: VSEL, Igf2-H19 locus, Oct4, Sall4
Introduction
The field of regenerative medicine is currently searching for a reliable source of pluripotent stem cells (PSCs) that could give rise to cells from all three germ layers.1 For almost 20 years, researchers have been attempting to harness embryonic stem cells (ESCs) that can be isolated from the embryos generated by in vitro fertilization2, 3 or therapeutic cloning.4 However, this strategy is burdened by ethical considerations. A promising source of PSCs can be generated by the genetic modification of adult tissues—induced PSCs5, 6—but this strategy is still under development and risks the formation of teratomas in the injected cells, in addition to rejection by the host immune system.7
Various potential types of adult stem and progenitor cells can now be isolated from bone marrow (BM), mobilized peripheral blood and umbilical cord blood (UCB) or derived from expanded in vitro cultures of adherent cells (such as mesenchymal stem cells (MSCs) and multipotent adult progenitor cells (MAPCs)) and are being investigated in clinical trials to determine their ability to regenerate damaged organs (for example, heart, kidney and neural tissues).8 Rare cases of chimerism after the infusion of unmanipulated donor BM, UCB or mobilized peripheral blood cells have been reported by some investigators; however, these results can be explained by cell fusion9, 10 or presence of rare populations of stem cells that are endowed with multi-tissue differentiation abilities.8
Thus, two of the most intriguing questions in stem cell biology are (1) if adult tissues contain PSCs or multipotent stem cells and (2) if these cells can differentiate into cells from more than one germ layer. Several groups of investigators have employed various isolation protocols, surface marker detection systems and experimental in vitro and in vivo models and have reported the presence of cells that possess pluripotent/multipotent characteristics in various adult organs. Such cells have been assigned various operational abbreviations and names in the literature, such as MAPCs,11 multipotent adult stem cells (MASCs),12, 13 unrestricted somatic stem cells,14 marrow-isolated adult multilineage-inducible cells15 and multilineage-differentiating stress-enduring stem (Muse) cells.16 However, this raises the basic question: are these truly distinct cells or instead just overlapping populations of the same primitive stem cell? In fact, taking into consideration the common features described in the literature, it is very likely that various investigators have described overlapping populations of developmentally early stem cells that are closely related. Unfortunately, these cells were never characterized side-by-side in order to address this important issue. Moreover, the rare and quiescent population of so-called very small embryonic-like stem cells (VSELs), which was initially isolated from murine tissues and human UCB by our group17, 18 (and subsequently confirmed by other laboratories19, 20, 21, 22, 23), expresses several PSC markers and, in addition, shares some characteristics with the abovementioned cell populations.
VSELs circulate in PB under steady-state conditions; however, the number of cells is very low. In our recent study, we provide evidence that VSELs can mobilize into PB in mice and adult patients who have been injected with granulocyte colony-stimulating factor.24 This observation laid the foundation for the concept that granulocyte colony-stimulating factor mobilization can be employed to harvest VSELs from patients for therapeutic purposes. Furthermore, our studies on VSEL mobilization into PB reveal that VSELs are mobilized not only in patients suffering from myocardial infarct25 and stroke26 but also in patients suffering from skin burns,27 active inflammatory bowel disease28 and cancer.29 In a recently published paper, Taichman et al.19 demonstrated that VSELs circulating in PB in tumor-bearing mice may give rise to tumor-associated fibroblasts involved in tumor expansion.29 More importantly, based on our preliminary data, we also believe that the number of VSELs circulating in PB could be prognostic of myocardial infarct or stroke.25, 26 This notion, however, requires further study and long-term clinical confirmation.
In this article, we discuss the most recent data from our laboratory and others on the optimization of VSEL isolation from adult tissues, provide an update on the molecular characteristics of VSELs, and describe the potential roles of these rare cells in regenerative medicine.
The presence of potential pluripotent stem cells in adult tissues
For many years, it has been accepted that adult tissues contain only tissue-committed stem cells (TCSCs), such as epidermal, hematopoietic or skeletal muscle stem cells, which demonstrate a limited, single, lineage-restricted potential for differentiation. However, recent evidence demonstrates that BM-isolated cells may possess broader differentiation potentials than the hematopoietic stem cells (HSCs) or MSCs present in this tissue.8 If we exclude the possibility of stem cell plasticity and HSC transdifferentiation, one has to consider the possibility that BM contains other, more primitive populations of PSCs and multipotent stem cells that are deposited in BM during embryogenesis.
Accordingly, we consider two scenarios that could occur during early embryogenesis and the development of lineage-restricted TCSCs.1 In the first scenario, PSCs develop in the inner cell mass of the blastocyst, or later on in the epiblast, and after giving rise to more differentiated lineage-restricted TCSCs gradually disappear in the developing tissues. In contrast, in the second scenario—which we believe is more likely—some PSCs giving rise to TCSCs survive in developing adult tissues as a backup population of PSCs that renews the pool of TCSCs over time. In this scenario, PSCs are not only precursors of TCSCs during organ/tissue rejuvenation but also serve as a source of PSCs during emergency situations when these organs are damaged (for example, myocardial infarction and stroke). This scenario, however, requires the PSC population in adult tissues to be kept under control and in a quiescent state essential for preventing uncontrolled proliferation that could lead to the formation of teratomas. We believe that VSELs are a population of such cells, and the mechanism that keeps them quiescent in adult tissues is similar to the one described in migrating primordial germ cells (PGCs). This mechanism requires the epigenetic modification of the regulatory regions of some developmentally crucial genes that are, themselves, regulated by parental imprinting.30
Our recent research revealed that murine VSELs modulate the expression of parentally imprinted genes (for example, Igf2-H19, RasGRF1 and Igf2R (insulin-like growth factor receptor 2)) via epigenetic changes, which may have an important role in insulin/insulin-like growth factor signaling (IIS).31 It is well known that imprinted genes have a crucial role in embryogenesis, fetal growth, the totipotential state of the zygote, and the pluripotency of developmentally early stem cells.32 Thus, modification of imprinting within the regulatory regions (that is, differentially methylated regions; DMRs) of these genes, which occurs in VSELs, is crucial for maintaining quiescence in the pools of these cells residing in adult tissues.31, 33 Accordingly, we observed that murine BM-sorted VSELs erase paternally methylated imprints within the DMRs of Igf2-H19 and RasGrf1, whereas they hypermethylate the maternally methylated DMRs of Igf2R and KCNQ1-p57KIP2. As paternally expressed imprinted genes (Igf2 and RasGrf1) enhance embryonic growth and maternally expressed genes (H19, Igf2R and p57KIP2) inhibit cell proliferation,34 the unique genomic imprinting pattern observed in VSELs represses the growth of these cells.31
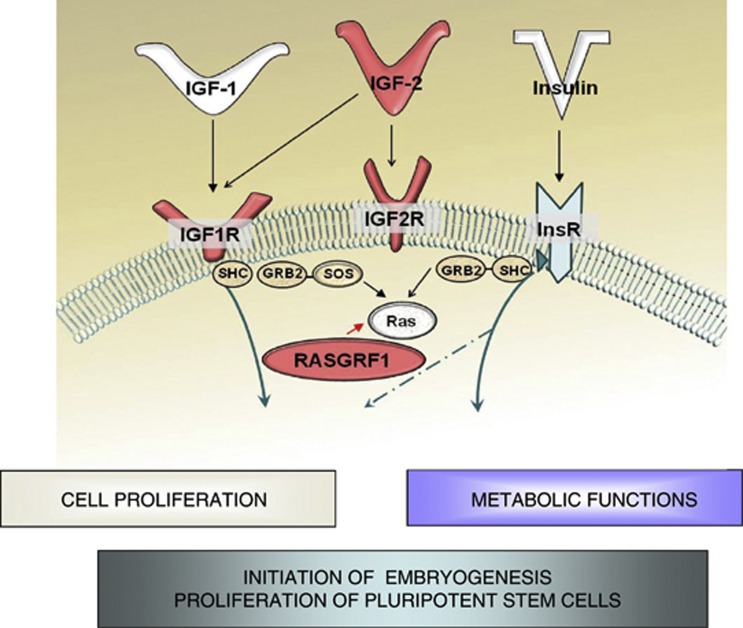
How do epigenetic changes in gene expression translate into changes in the state of VSELs? In murine VSELs (Figure 1), the abovementioned epigenetic changes lead to perturbation of IIS by downregulating (1) insulin-like growth factor 2 (Igf2), which is an autocrine factor involved in the proliferation of VSELs, and (2) RasGrf1, which is a GTP-exchange factor (GEF) crucial for signaling from activated insulin-like growth factor 1 receptor (IgfIR) and insulin receptor.35 In addition, hypermethylation of the DMRs on the maternal chromosome that encode Igf2R results in an additional negative effect on IIS in VSELs. As previously described, Igf2R serves as a decoy receptor that prevents Igf2 from binding to IgfIR.36 This epigenetic reprogramming of genomic imprinting negatively affects IIS signaling, maintains the quiescent state of murine VSELs (Figure 1), and, thus, protects VSELs from premature depletion from the tissues and prevents their involvement in tumor formation. Our most recent unpublished data on the methylation status of DMRs at the Igf2-H19 locus in human VSELs suggest that a similar mechanism may also operate in human VSELs.
Figure 1.
IIS signaling and imprinted genes. In mammals, there are three insulin factors (insulin, Igf1 and Igf2) that bind to two tyrosine kinase receptors (insulin receptor (InsR) and Igf1 receptor (Igf1R)). Igf2R is a non-signaling mannose-type sink receptor for Igf2. Activation of InsR and Igf1R leads to metabolic or proliferative responses depending on the cell type. RasGrf1 is a small GEF that is involved in signaling from InsR and Igf1R. VSELs demonstrate a decrease in Igf2 and RasGrf1 expression and the overexpression of Igf2R (shown in red) due to changes in the epigenetic state of the imprinted genes. These epigenetic changes in genes regulate IIS and maintain quiescence in VSELs in adult tissues that are somehow resistant to in vitro expansion. We hypothesize that chronic exposure to IIS accelerates the premature depletion of VSELs from adult tissues.
Next, based on the published studies reporting that IIS signaling negatively affects the lifespan of experimental animals,37, 38 we proposed a hypothesis that relates aging, longevity and IIS to the abundance and function of pluripotent VSELs that are present in adult murine tissues.39 Accordingly, we postulated that a prolonged IIS may negatively affect the pool of VSELs and, subsequently, TCSCs in various organs, thereby having an impact on tissue rejuvenation and lifespan.33 In support of this notion, we reported a significantly higher number of VSELs in BM of long-living murine strains of mice (for example, Laron and Ames dwarfs), whose longevity is explained by low levels of circulating IGF1 and, thus, a decrease in IIS.40 In contrast, the number of VSELs is reduced in mice with high levels of circulating Igf1 and enhanced IIS (for example, growth hormone-overexpressing transgenic mice) in comparison with normally aging littermates.41
We are currently investigating the influence of calorie restriction and the effects of the prolonged administration of drugs that modulate IIS and extend lifespan, such as metformin and rapamycin, on the pool of VSELs residing in BM and other tissues.
Optimization of isolation strategies
VSELs are very rare (∼1 per 105 monocular BM cells), and careful follow-up isolation protocols are required to purify these cells. Moreover, more specific markers are needed to isolate these cells. Nevertheless, at this point, the best positive surface markers that allow purification by multisorting analysis are the Sca1 antigen for murine VSELs and the CD133 antigen for human counterparts. However, these markers are also expressed by several other types of stem cells.
Current strategies for isolating murine VSELs
The current strategy for purifying murine VSELs from BM, as well as other adult tissues, is based on sorting any nucleated cells that are slightly smaller than erythrocytes (4–5 μm) and express the Sca1+Lin−CD45− phenotype. These cells also express SSEA1 antigen on their surface, and some VSELs also express receptors for FGF2 (FGF-2R), PDGF (PDGF-R), SDF1 (CXCR4) and KL (c-kit). We are aware that the lack of specific positive markers for VSELs leads to isolation by cell sorter of rare events that are enriched for VSELs but also contain some cell debris and even bare nuclei. Moreover, when isolating VSELs from murine BM, the most important factor is proper maintenance that consists of a cocktail of lineage antibodies and a sufficient titer against erythroid cells (Ter119) in order to avoid contamination of the sorted VSELs with Lin−CD45− erythroblasts that may acquire low expression of Sca1 antigen by microvesicles shed from Sca1+ cells in BM. Thus, to avoid enrichment of the erythroblasts, it is also important to set up a proper gate that excludes small Sca1dimLin−CD45− cells, which could become contaminated by erythroblasts.
As mentioned above, some murine VSELs express FGF-2R, PDGF-R, CXCR4 and c-kit on their surface. We still do not know whether this expression of various receptors reflects the commitments of some VSELs to different lineages. In support of this possibility, we recently isolated three different types of mRNA from singly purified VSELs that differ in terms of the expression of pluripotency, germline-specific and various other types of genes.42
As murine BM is a relatively easy tissue for Fluorescence-activated cell sorting (FACS) analysis and cell sorting, most molecular data are obtained using BM-derived VSELs. However, as there are corresponding populations of cells in other murine solid organs,43 further work is needed to compare the molecular signatures of solid organ-purified and BM-isolated VSELs. Similar studies also have to be performed on human VSELs. Nevertheless, in a recent report, an independent group of investigators performed mRNA gene array analysis on human ovarian surface epithelium-derived VSELs and observed similar patterns of expression in genes regulating stem cell pluripotency and germ line specification,44 as we described in murine BM-isolated VSELs.17 Interestingly, these small human VSELs were efficiently sorted using FACS by employing antibodies against human SSEA4 antigen.
Current strategies for isolating human VSELs
As described, VSELs from human UCB and mobilized peripheral blood were initially purified as small CD133+ CD45− Lin− cells by employing multiparameter sorting from an erythrocyte depleted by hypotonic lysis population of nucleated cells.18 Unfortunately, this isolation procedure is time consuming, and the sorting time required to process an entire unit of cord blood (∼50–100 ml), which is needed to isolate rare VSELs from mononucleated UCB cells, would be 3–4 days. Therefore, it has become clear that a faster and less expensive method for isolating these cells must be established. To develop a more efficient method for purifying VSELs from UCB, we proposed a three-step isolation strategy based on (1) removing erythrocytes by hypotonic lysis, (2) immunomagnetic separation of CD133+ cells, and (3) FACS-based isolation of CD133+Lin−CD45− cells. Processing 100-ml UCB requires only 2–4 h using this procedure.45, 46 As with murine VSELs, we do not have a specific marker for VSELs, and our sorted cells may also contain other CD133+ cells. Furthermore, as with murine VSELs, it is very important to employ antihuman erythroid lineage antibodies (CD235a) to avoid contamination with small Lin−CD45− erythroblasts.
As an alternative strategy, we also attempted exposing erythrocyte-depleted, immunomagnetic bead-selected CD133+ cells to Aldefluor, followed by staining with anti-CD133 antibodies conjugated with fluorochromes and two lineage-specific antibodies—one against pan-hematopoietic antigen CD45 (anti-CD45 MoAbs) and second one against erythroid marker glycophorin-A (anti-GlyA MoAbs).47 Aldefluor employed in this alternative sorting strategy is a substrate for aldehyde dehydrogenase (ALDH), a cytosolic enzyme that is highly expressed in less-differentiated hematopoietic cells. In the presence of ALDH, Aldefluor is modified to a fluorescent molecule that maybe used to mark ALDH-expressing cells. Using this novel strategy, we sort CD133+ cells enriched for VSELs and obtain from 100 ml of UCB, on average, ∼103 CD133+CD45−GlyA−ALDHlow and 4 × 103 CD133+CD45−GlyA−ALDHhigh VSELs from 100 ml of UCB.47 These numbers demonstrate how rare these cells are in UCB. Moreover, when we compared both fractions of VSELs using RT-PCR, we found that CD133+CD45−GlyA−ALDHlow VSELs demonstrated a higher expression of the pluripotency marker Oct4 than the CD133+CD45−GlyA−ALDHhigh fraction.47
However, we are aware that there is still room for improvement when it comes to sorting by employing, for example, metabolic fluorochromes to see whether VSELs are enriched among Rh123dull, Pyronin Ylow and Hoechst 33342low cells. Moreover, we expect our proteomic data analysis of UCB-derived VSELs, which is in progress, will reveal the presence of new positive markers that could be employed for sorting these cells. However, as mentioned above, some groups have successfully employed antibodies against SSEA antigens to purify human22 and rat VSELs.48
Nevertheless, in order to further enrich human VSELs, we recently employed a novel strategy in which we deplete the lineage to thereby enrich CD133+ UCB-derived mononuclear cells, which are subsequently permeabilized and stained with anti-Oct4 antibodies. Using this strategy, we isolated a highly purified population of VSELs that was suitable for molecular studies at the DNA (imprinting analysis) and mRNA (gene expression analysis) levels (manuscript in preparation).
The molecular signature of VSELs—the key to understand quiescence and pluripotency
As mentioned above, we performed most of our molecular studies on murine BM-purified VSELs at the DNA, mRNA and, to some extent, the protein levels.31, 42, 49 These DNA studies revealed that promoters for pluripotency markers, such as Oct4 and Nanog, have an open chromatin structure and are associated with transcription-promoting histones.31, 49 This finding strongly supports the authenticity of the expression levels of these pluripotency-regulating transcription factor genes in murine VSELs. Furthermore, as expected from the definition of PSC, we reported that VSELs possess so-called bivalent domains in developmentally crucial homeobox genes.42 As mentioned above, murine VSELs also possess a unique methylation pattern at the DMRs of some paternally imprinted genes (for example, erasure of imprinting at the Igf2-H19 and RasGrf1 loci and hypermethylation at the Igf2R and KCNQ1 loci) that is responsible for the quiescent state of these cells (Figure 1) and the decrease in IIS. These changes in the expression levels of paternally imprinted genes were subsequently confirmed using mRNA analysis.31
Furthermore, our genome-wide analysis of murine BM-derived VSELs revealed that the attenuation of mitogenic growth factor signaling pathways also has a crucial role in quiescence and ageing.50 Specifically, VSELs downregulate the genes involved in the responses to UV radiation, mRNA processing and mitogenic growth factor signaling (for example, from Igf1 and TRKA receptors involved in the ERK and PI3K pathways). Using leading-edge subset analysis and real-time quantitative PCR assays, we observed that several genes, such as Grb2, Sos1, Shc1, Map2k1, Akt3, Elk1, Rps6ka3, Gsk3β and Csnk2a1, which are involved in mitogenic growth factor signaling pathways, are commonly downregulated in VSELs. In contrast, we observed that Oct4+ VSELs upregulate tissue-specific gene sets and a gene set that encodes the complement-coagulation cascade.
These results suggest that the epigenetic reprogramming of genomic imprinting maintains the quiescence of the Oct4+ epiblast/germ line-derived VSELs that are deposited in the adult body and protects them from premature ageing and uncontrolled proliferation.51 On the other hand, reversal of this mechanism will be crucial for employing VSELs as a population of PSCs for use in regenerative medicine. Currently, we are investigating how the downregulation of the expression of H19 enhances VSEL expansion, as recently demonstrated in parthenogenesis-derived PSCs.52
As briefly mentioned above, our mRNA expression studies revealed that VSELs express several epiblast and germline markers, which is the basis for the hypothesis that VSELs originate from early epiblast-derived migrating PGC-like cells. We envision that VSELs are deposited in adult tissues during development as a source of TCSCs and have a role in organ rejuvenation. In support of this notion, molecular analysis of murine BM-derived VSELs revealed that these cells express several genes that are characteristic of epiblast stem cells such as Gbx2, Fgf5 and Nodal and germline specification of stem cells including Blimp1, Prdm14, Fragilis, Stella, Nanos3 and Dnd1.49 Recently, we also found that both murine and human VSELs highly express Sall4, a marker of germline cells. We also demonstrated that murine and human VSELs (1) are diploid, (2) are viable, as shown by their ability to exclude viability dye (7-aminoactinomycin D), and (3) highly express telomerase.17
We are currently assessing the expression of various miRNA species in VSELs, as well as how to perform proteomic analysis on these rare cells. We hope the latter approach will allow us to identify new and unique markers for these cells that will allow optimal and rapid purification.
Data supporting the presence of very small stem cells in adult murine and human tissues
As mentioned above, several primitive cells with the characteristics for pluripotency or multipotency were isolated from adult murine and human BM, UCB and adult solid organs, enzymatically processed using proteolytic enzymes, and expanded and cultured in vitro.14, 53, 54, 55, 56 From these cultures, in which the cells were allowed to grow and adhere to plastic or fibronectin, several populations of primitive cells were isolated, expanded and assigned different operational names such as MAPCs, MASCs, marrow-isolated adult multilineage-inducible cells, Muse cells or unrestricted somatic stem cells.11, 12, 13, 14, 15, 16, 57 Unfortunately, the phenotype of the stem cell that initiated these cultures has never been clearly described. Furthermore, very small cells, which are most likely human VSELs, can also be directly purified from neonatal UCB.22, 58 In addition to multiparameter sorting, other isolation strategies have been employed, and, for example, an interesting population of small cells that is able to differentiate into epithelial and HSCs was isolated from murine BM by elutriation, lineage depletion and the ability to home (H) to BM, has been described as ELH stem cells.59, 60, 61 Another group reported presence in adult mammalian tissues of small cells, which are able to differentiate into cells from all germ layers and have been isolated from adult mammalian tissues (known as ‘spore-like stem cells'). However, the purification strategy and expressed surface markers were not described in the original report.62 Nevertheless, the presence of ELH stem cells and ‘spore-like stem cells' residing in adult BM has somehow challenged the concept of HSC plasticity.
The most common features of VSELs are their very primitive morphology and relatively small size.17, 18 Recently, several reports were published that support the existence of small, primitive VSELs and VSEL-like cells in adult tissues (the most important are listed in Table 1). In addition to ELH cells and the spore-like stem cells mentioned above, VSELs or VSEL-like stem cells have been isolated by other independent groups. For example, murine BM-derived VSELs have been shown to give rise to type 2 pneumocytes, which produce lung surfactant protein after being transplanted into surfactant-deficient mice.21 Furthermore, small Oct4+SSEA1+Lin−CD45− cells isolated from rat BM gave rise in vivo to cardiomyocytes and endothelial cells in an experimental model of rodent acute myocardial infarction.48 Moreover, cells from human-mobilized PB expressing the SSEA4+CD133+CXCR4+Lin− and CD45− phenotypes that were isolated using FACS formed human bones when embedded in gelatin sponges and implanted into immunodeficient mice; this bone-forming activity exceeded the activity of the other populations of BM-purified cells that were evaluated using the same assay. Based on this finding, it has been proposed that these PB-purified VSELs are at the top of the hierarchy for the mesenchymal and endothelial lineages.23 Cells similar to BM-derived VSELs have been reported to reside in the ovarian surface epithelium in postmenopausal ovaries63 and normal testes64 and differentiate into gametes.44 Furthermore, human PB-VSELs have been successfully purified by other researchers,23, 65 and very small Oct4+Sox2+ cells corresponding to UCB-derived VSELs described by us were purified from UCB by other investigators, who described them as a population of UCB-derived embryonic-like stem cells.58 Interestingly, these cells were able to differentiate in vitro into neural progenitor cells. Finally, a corresponding population of primitive Oct4+ stem cells that resembles VSELs—named omnicytes—was also described to be circulating in UCB and was capable of migrating into the maternal circulatory system.66
Table 1. Summary of the findings from a selection of reports on stem cells that are attributable to VSELs or closely related cells.
| Cells originally named in the paper | Isolation procedure and properties described in the original paper | Reference |
|---|---|---|
| ELH cells | Isolated by elutriation, lineage depletion and recovery after homing (H) to the BM. Differentiate to epithelial cells and hematopoietic cells. | 59, 60, 61 |
| Spore-like stem cells | Small, ∼5 μm in diameter, isolated from various murine tissues, resistant to freeze/thawing, express Oct4 and demonstrating broad differentiation. Isolation procedures not indicated. | 62 |
| Small nonhematopoietic Sca1+ Lin− CD45− cells | Isolated using FACS from murine BM, differentiate into type II pneumocytes, produce surfactant in the lung alveolar epithelium. Recently these cells were confirmed as VSELs. | 21 |
| Rat VSELs | Isolated using FACS from rat bone marrow as SSEA+ Lin− CD45− cells and endowed with cardiomyogenic and endothelial potential. | 48 |
| Human PB-derived VSELs | Isolated using FACS as SSEA4+ CD133+ CXCR4+ Lin− and CD45− cells, described as being at the top of the mesenchymal lineage hierarchy. Develop into human bones in immunodeficient mice. | 23, 65 |
| Ovarian VSELs | Small Oct4+ SSEA+ cells were isolated using FACS from ovarian surface epithelium (OSE) obtained from mice and humans (precursors of female gametes). Human OSE-derived VSELs were extensively characterized using mRNA expression array analysis. | 44, 63 |
| Testicular VSELs | Small Oct4+ SSEA+ cells identified in murine and human testes (precursors of male gametes). | 64 |
| Embryonic-like stem cells from UCB | Small CD45−/CD33−/CD7−/CD235a− pluripotent stem cells (2–3 μm in diameter) coexpressing embryonic stem cell markers, including Oct4 and Sox2, able to differentiate into neuronal cells. | 58 |
| Omnicytes | Small Oct4+ stem cells identified in UCB, able to establish fetal–maternal chimerism. | 66 |
Abbreviations: BM, bone marrow; FACS, 5 Fluorescence-activated cell sorting; UCB, umbilical cord blood; VSEL, very small embryonic-like stem cell.
In addition to the cells listed in Table 1, in one of recent reports small cells with some of VSEL markers have also been identified in murine neonatal retina.67 In another study, small nonhematopoietic lineage-negative (Lin−) cells that have been isolated from adult BM by elutriation (Fraction 25 or Fr25) were involved in retinal regeneration following the induction of anterior ischemic optic neuropathy and optic nerve crush injury in a rodent model.68 Moreover, a similar population of small nonhematopoietic CD45− stem cells harvested from BM via elutriation was recently shown to differentiate in vivo into functional insulin-producing cells in chemically induced diabetic mice.69 Finally, several features of VSELs are present in Muse cells that were recently isolated from murine and human BM populations.16, 57 As postulated by the authors, Muse cells may have a major role in populations of cells that preferentially become transformed and give rise to immortal induced PSCs when BM-derived stromal cells are induced to pluripotency by genetic manipulation. Thus, VSELs and VSEL-resembling cells are currently purified in several laboratories worldwide, and the coming years will bring more information regarding their biology and in vitro and in vivo differentiation potential.
In vitro and in vivo criteria for stem cell pluripotentiality—comparison with VSELs
Several criteria have been proposed for classifying stem cell as PSCs (Table 2). These criteria were established by embryologists working on ESCs and induced PSCs. However, some of these stringent criteria of pluripotency listed in (Table 2), such as complementing blastocyst development and teratoma formation, are not always applicable for pluripotent epiblast stem cells (EpiSCs) as well as PGCs.70, 71
Table 2. In vitro and in vivo criteria for pluripotent stem cells.
| VSELs | |
|---|---|
| In vitro criteria for PSCs | |
| Undifferentiated morphology, euchromatin and high nucleus/cytoplasm ratio | Yes |
| PSC markers (for example, Oct4, Nanog, SSEA), open chromatin at the Oct4 promoter, bivalent domains and reactivation of the X chromosome in female PSCs | Yes |
| Broad multilineage differentiation into cells from all three germ layers (meso-, ecto- and endoderm) | Yes |
| In vivo criteria for PSCs | |
| Complement blastocyst development | No |
| Teratoma formation after inoculation into immunodeficient mice | No |
Abbreviations: PSC, pluripotent stem cell; VSEL, very small embryonic-like stem cell.
Our experimental data support that murine VSELs fulfill all the in vitro criteria listed in Table 2. In particular, as discussed in this review, murine VSELs not only possess the primitive morphology of early developmental cells (for example, high nuclear/cytoplasmic ratio, presence of euchromatin in the nucleus) but also express the marker characteristics for PSCs (for example, Oct4, Nanog, Rex1). Significantly, we recently confirmed the authentic expression of Oct4 in murine VSELs by demonstrating that the Oct4 promoter have an open chromatin by direct analysis of methylation state (hypomethylation in DNA) and its high association with transcription-favorable histone modifications (for example, the acetylation of histone H3 and trimethylation of lysine 4 on histone 3) and relatively low association with the transcription-restricting ones (for example, dimethylation of lysine 9 on histone 3).31 We also studied the epigenetic state of another pluripotency-related transcription factor, Nanog. In comparison to Oct4, the promoter of Nanog has a higher level of methylation in VSELs (∼50%), and the quantitative chromatin immunoprecipitation experiments performed in parallel revealed that the H3Ac/H3K9me2 ratio favors transcription and maintains its active state.72, 73, 74 Based on these results, we conclude that murine VSELs truly express both Oct4 and Nanog.
In addition, murine VSELs fulfill also other in vitro criteria of pluripotency (Table 2) such as presence of bivalent domains in promoters that encode developmentally important homeobox-containing transcription factors (Sox21, Nkx2.2, Dlx1, Lbx1h, Hlxb9, Pax5 and HoxA3)42 and VSELs derived from female mice reactivate the X chromosome. Finally, we and other groups have successfully differentiated VSELs in vitro into cells from all three germ layers. Accordingly, recently published studies confirm that murine BM-derived VSELs can differentiate in vivo into HSCs,47 MSCs,19 endothelial cells,48 epithelial cells of the lung,21 oocytes,44 tumor stroma cells29 and cardiomyocytes.20
However, murine VSELs do not complete blastocyst development and do not develop teratomas, which are included in the in vivo criteria of PSCs (Table 2). This obvious discrepancy between the in vitro and in vivo PSC criteria for VSELs was recently explained by the effects of epigenetic changes in the expression of some paternally imprinted genes,31 as described above. Moreover, one has to take into consideration that, although all these in vivo criteria apply very well for ESCs and iPSc, they are not always applicable as mentioned above for EpiSCs and PGCs.
Conclusions
New data from our and other independent laboratories have provided more convincing evidence for the existence and biological role of VSELs in murine adult tissues and their potential roles in (1) tissue organ rejuvenation, (2) regulation of lifespan, and (3) the regeneration/repair of damaged organs. We have to emphasize that, although the molecular nature of murine BM-derived VSELs has been extensively characterized, more research is needed to better characterize these small cells in humans. If we can confirm that a similar mechanism based on the epigenetic changes in somatic-imprinted genes operates in human VSELs, perhaps the controlled modulation of this imprinting state to produce proper de novo methylation of the regulatory regions in these genes on the maternal and paternal chromosomes could increase the regenerative power of these cells. This would allow for application of VSELs in clinical medicine. Finally, in order to avoid mistakes in isolation of VSELs we recommend for further reading our most recent paper75 in which we address in more details potential pitfalls in FACS sorting strategy. We also recommend an excellent recent review on VSELs published by Dr Krause group.76
Acknowledgments
This work was supported by a grant from the National Institutes of Health (2R01 DK074720), the European Union's Innovative Economy Operational Programme (POIG.01.01.02-00-109/09-01), and an endowment from Stella and Henry Hoenig that were awarded to MZR, and by a grant from the Asian Institute for Life Sciences, Seoul, Korea (2013-528) and the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A120301) that were awarded to DMS.
References
- Ratajczak MZ, Liu R, Ratajczak J, Kucia M, Shin D-M. The role of pluripotent embryonic-like stem cells residing in adult tissues in regeneration and longevity. Differentiation. 2011;81:153–161. doi: 10.1016/j.diff.2011.01.006. [DOI] [PubMed] [Google Scholar]
- Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292:154–156. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- Tachibana M, Amato P, Sparman M, Gutierrez Nuria M, Tippner-Hedges R, Ma H, et al. Human embryonic stem cells derived by somatic cell nuclear transfer. Cell. 2013;153:1228–1238. doi: 10.1016/j.cell.2013.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, Hochedlinger K, et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature. 2007;448:318–324. doi: 10.1038/nature05944. [DOI] [PubMed] [Google Scholar]
- Zhao T, Zhang Z-N, Rong Z, Xu Y. Immunogenicity of induced pluripotent stem cells. Nature. 2011;474:212–215. doi: 10.1038/nature10135. [DOI] [PubMed] [Google Scholar]
- Ratajczak MZ, Machalinski B, Wojakowski W, Ratajczak J, Kucia M. A hypothesis for an embryonic origin of pluripotent Oct-4+ stem cells in adult bone marrow and other tissues. Leukemia. 2007;21:860–867. doi: 10.1038/sj.leu.2404630. [DOI] [PubMed] [Google Scholar]
- Terada N, Hamazaki T, Oka M, Hoki M, Mastalerz DM, Nakano Y, et al. Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature. 2002;416:542–545. doi: 10.1038/nature730. [DOI] [PubMed] [Google Scholar]
- Ying Q-L, Nichols J, Evans EP, Smith AG. Changing potency by spontaneous fusion. Nature. 2002;416:545–548. doi: 10.1038/nature729. [DOI] [PubMed] [Google Scholar]
- Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418:41–49. doi: 10.1038/nature00870. [DOI] [PubMed] [Google Scholar]
- Pochampally RR, Smith JR, Ylostalo J, Prockop DJ. Serum deprivation of human marrow stromal cells (hMSCs) selects for a subpopulation of early progenitor cells with enhanced expression of OCT-4 and other embryonic genes. Blood. 2004;103:1647–1652. doi: 10.1182/blood-2003-06-1967. [DOI] [PubMed] [Google Scholar]
- Beltrami AP, Cesselli D, Bergamin N, Marcon P, Rigo S, Puppato E, et al. Multipotent cells can be generated in vitro from several adult human organs (heart, liver, and bone marrow) Blood. 2007;110:3438–3446. doi: 10.1182/blood-2006-11-055566. [DOI] [PubMed] [Google Scholar]
- Kögler G, Sensken S, Airey JA, Trapp T, Müschen M, Feldhahn N, et al. A new human somatic stem cell from placental cord blood with intrinsic pluripotent differentiation potential. J Exp Med. 2004;200:123–135. doi: 10.1084/jem.20040440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Ippolito G, Diabira S, Howard GA, Menei P, Roos BA, Schiller PC. Marrow-isolated adult multilineage inducible (MIAMI) cells, a unique population of postnatal young and old human cells with extensive expansion and differentiation potential. J Cell Sci. 2004;117:2971–2981. doi: 10.1242/jcs.01103. [DOI] [PubMed] [Google Scholar]
- Kuroda Y, Kitada M, Wakao S, Nishikawa K, Tanimura Y, Makinoshima H, et al. Unique multipotent cells in adult human mesenchymal cell populations. Proc Natl Acad Sci USA. 2010;107:8639–8643. doi: 10.1073/pnas.0911647107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kucia M, Reca R, Campbell FR, Zuba-Surma E, Majka M, Ratajczak J, et al. A population of very small embryonic-like (VSEL) CXCR4+SSEA-1+Oct-4+ stem cells identified in adult bone marrow. Leukemia. 2006;20:857–869. doi: 10.1038/sj.leu.2404171. [DOI] [PubMed] [Google Scholar]
- Kucia M, Halasa M, Wysoczynski M, Baskiewicz-Masiuk M, Moldenhawer S, Zuba-Surma E, et al. Morphological and molecular characterization of novel population of CXCR4+ SSEA-4+ Oct-4+ very small embryonic-like cells purified from human cord blood - preliminary report. Leukemia. 2006;21:297–303. doi: 10.1038/sj.leu.2404470. [DOI] [PubMed] [Google Scholar]
- Taichman RS, Wang Z, Shiozawa Y, Jung Y, Song J, Balduino A, et al. Prospective identification and skeletal localization of cells capable of multilineage differentiation in vivo. Stem Cells Dev. 2010;19:1557–1570. doi: 10.1089/scd.2009.0445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawn B, Tiwari S, Kucia MJ, Zuba-Surma EK, Guo Y, SanganalMath SK, et al. Transplantation of bone marrow-derived very small embryonic-like stem cells attenuates left ventricular dysfunction and remodeling after myocardial infarction. Stem Cells. 2008;26:1646–1655. doi: 10.1634/stemcells.2007-0715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kassmer SH, Jin H, Zhang P-X, Bruscia EM, Heydari K, Lee J-H, et al. Very small embryonic-like stem cells from the murine bone marrow differentiate into epithelial cells of the lung STEM CELLS 2013. doi: 10.1002/stem.1413 [DOI] [PMC free article] [PubMed]
- Bhartiya D, Shaikh A, Nagvenkar P, Kasiviswanathan S, Pethe P, Pawani H, et al. Very small embryonic-like stem cells with maximum regenerative potential get discarded during cord blood banking and bone marrow processing for autologous stem cell therapy. Stem Cells Dev. 2012;21:1–6. doi: 10.1089/scd.2011.0311. [DOI] [PubMed] [Google Scholar]
- Havens AM, Shiozawa Y, Jung Y, Sun H, Wang J, McGee S, et al. Human very small embryonic-like cells generate skeletal structures, in vivo. Stem Cells Dev. 2013;22:622–630. doi: 10.1089/scd.2012.0327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kucia MJ, Wysoczynski M, Wu W, Zuba-Surma EK, Ratajczak J, Ratajczak MZ. Evidence that very small embryonic-like stem cells are mobilized into peripheral blood. STEM CELLS. 2008;26:2083–2092. doi: 10.1634/stemcells.2007-0922. [DOI] [PubMed] [Google Scholar]
- Wojakowski W, Tendera M, Kucia M, Zuba-Surma E, Paczkowska E, Ciosek J, et al. Mobilization of bone marrow-derived oct-4+ ssea-4+ very small embryonic-like stem cells in patients with acute myocardial infarction. J Am Coll Cardiol. 2009;53:1–9. doi: 10.1016/j.jacc.2008.09.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paczkowska E, Kucia M, Koziarska D, Halasa M, Safranow K, Masiuk M, et al. clinical evidence that very small embryonic-like stem cells are mobilized into peripheral blood in patients after stroke. Stroke. 2009;40:1237–1244. doi: 10.1161/STROKEAHA.108.535062. [DOI] [PubMed] [Google Scholar]
- Drukała J, Paczkowska E, Kucia M, Młyńska E, Krajewski A, Machaliński B, et al. stem cells, including a population of very small embryonic-like stem cells, are mobilized into peripheral blood in patients after skin burn Injury. Stem Cell Rev Rep. 2012;8:184–194. doi: 10.1007/s12015-011-9272-4. [DOI] [PubMed] [Google Scholar]
- Marlicz W, Zuba-Surma E, Kucia M, Blogowski W, Starzynska T, Ratajczak MZ. Various types of stem cells, including a population of very small embryonic-like stem cells, are mobilized into peripheral blood in patients with Crohn's disease. Inflamm Bowel Dis. 2012;18:1711–1722. doi: 10.1002/ibd.22875. [DOI] [PubMed] [Google Scholar]
- Jung Y, Kim JK, Shiozawa Y, Wang J, Mishra A, Joseph J, et al. Recruitment of mesenchymal stem cells into prostate tumours promotes metastasis. Nat Commun. 2013;4:1795. doi: 10.1038/ncomms2766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratajczak M, Shin D-M, Liu R, Marlicz W, Tarnowski M, Ratajczak J, et al. Epiblast/germ line hypothesis of cancer development revisited: lesson from the presence of oct-4+ cells in adult tissues. Stem Cell Rev Rep. 2010;6:307–316. doi: 10.1007/s12015-010-9143-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin DM, Zuba-Surma EK, Wu W, Ratajczak J, Wysoczynski M, Ratajczak MZ, et al. Novel epigenetic mechanisms that control pluripotency and quiescence of adult bone marrow-derived Oct4+ very small embryonic-like stem cells. Leukemia. 2009;23:2042–2051. doi: 10.1038/leu.2009.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reik W, Walter J. Genomic imprinting: parental influence on the genome. Nat Rev Genet. 2001;2:21–32. doi: 10.1038/35047554. [DOI] [PubMed] [Google Scholar]
- Ratajczak MZ, Shin DM, Schneider G, Ratajczak J, Kucia M. Parental imprinting regulates insulin-like growth factor signaling: a Rosetta Stone for understanding the biology of pluripotent stem cells, aging and cancerogenesis. Leukemia. 2013;27:773–779. doi: 10.1038/leu.2012.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratajczak MZ, Kucia M, Liu R, Shin DM, Bryndza E, Masternak MM, et al. RasGrf1: genomic imprinting, VSELs, and aging. Aging (Albany NY) 2011;3:692–697. doi: 10.18632/aging.100354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Font de Mora J, Esteban LM, Burks DJ, Nunez A, Garces C, Garcia-Barrado MJ, et al. Ras-GRF1 signaling is required for normal beta-cell development and glucose homeostasis. EMBO J. 2003;22:3039–3049. doi: 10.1093/emboj/cdg280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludwig T, Eggenschwiler J, Fisher P, D'Ercole AJ, Davenport ML, Efstratiadis A. Mouse mutants lacking the type 2 IGF receptor (IGF2R) are rescued from perinatal lethality inIgf2andIgf1rNull backgrounds. Dev Biol. 1996;177:517–535. doi: 10.1006/dbio.1996.0182. [DOI] [PubMed] [Google Scholar]
- Bartke A. Insulin and aging. Cell Cycle. 2008;7:3338–3343. doi: 10.4161/cc.7.21.7012. [DOI] [PubMed] [Google Scholar]
- Piper MDW, Bartke A. Diet and aging. Cell Metabolism. 2008;8:99–104. doi: 10.1016/j.cmet.2008.06.012. [DOI] [PubMed] [Google Scholar]
- Ratajczak MZ, Shin DM, Ratajczak J, Kucia M, Bartke A. A novel insight into aging: are there pluripotent very small embryonic-like stem cells (VSELs) in adult tissues overtime depleted in an Igf-1-dependent manner. Aging (Albany NY) 2010;2:875–883. doi: 10.18632/aging.100231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratajczak J, Shin DM, Wan W, Liu R, Masternak MM, Piotrowska K, et al. Higher number of stem cells in the bone marrow of circulating low Igf-1 level Laron dwarf mice[mdash]novel view on Igf-1, stem cells and aging. Leukemia. 2011;25:729–733. doi: 10.1038/leu.2010.314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kucia M, Shin DM, Liu R, Ratajczak J, Bryndza E, Masternak MM, et al. Reduced number of VSELs in the bone marrow of growth hormone transgenic mice indicates that chronically elevated Igf1 level accelerates age-dependent exhaustion of pluripotent stem cell pool: a novel view on aging. Leukemia. 2011;25:1370–1374. doi: 10.1038/leu.2011.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin DM, Liu R, Wu W, Waigel SJ, Zacharias W, Ratajczak MZ, et al. Global gene expression analysis of very small embryonic-like stem cells reveals that the Ezh2-dependent bivalent domain mechanism contributes to their pluripotent state. Stem Cells Dev. 2012;21:1639–1652. doi: 10.1089/scd.2011.0389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuba-Surma EK, Kucia M, Wu W, Klich I, Lillard JW, Jr, Ratajczak J, et al. Very small embryonic-like stem cells are present in adult murine organs: ImageStream-based morphological analysis and distribution studies. Cytometry Part A. 2008;73A:1116–1127. doi: 10.1002/cyto.a.20667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parte S, Bhartiya D, Telang J, Daithankar V, Salvi V, Zaveri K, et al. Detection, characterization, and spontaneous differentiation in vitro of very small embryonic-like putative stem cells in adult mammalian ovary. Stem Cells Dev. 2011;20:1451–1464. doi: 10.1089/scd.2010.0461. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Zuba-Surma EK, Ratajczak MZ. Overview of Very Small Embryonic-Like Stem Cells (VSELs) and Methodology of Their Identification and Isolation by Flow Cytometric Methods. John Wiley & Sons, Inc.; 2010. [DOI] [PubMed] [Google Scholar]
- Ratajczak MZ, Liu R, Marlicz W, Blogowski W, Starzynska T, Wojakowski W, et al. Chapter 3-Identification of Very Small Embryonic/Epiblast-Like Stem Cells (VSELs) Circulating in Peripheral Blood During Organ/Tissue InjuriesIn: EHAOWT Zbigniew Darzynkiewicz, Donald W (eds). Methods in Cell Biology, vol Volume 103Academic Press; 201131–54. [DOI] [PubMed] [Google Scholar]
- Ratajczak J, Zuba-Surma E, Klich I, Liu R, Wysoczynski M, Greco N, et al. Hematopoietic differentiation of umbilical cord blood-derived very small embryonic/epiblast-like stem cells. Leukemia. 2011;25:1278–1285. doi: 10.1038/leu.2011.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu JH, Wang HJ, Tan YZ, Li ZH. Characterization of rat very small embryonic-like stem cells and cardiac repair after cell transplantation for myocardial infarction. Stem Cells Dev. 2012;21:1367–1379. doi: 10.1089/scd.2011.0280. [DOI] [PubMed] [Google Scholar]
- Shin DM, Liu R, Klich I, Wu W, Ratajczak J, Kucia M, et al. Molecular signature of adult bone marrow-purified very small embryonic-like stem cells supports their developmental epiblast/germ line origin. Leukemia. 2010;24:1450–1461. doi: 10.1038/leu.2010.121. [DOI] [PubMed] [Google Scholar]
- Mierzejewska K, Heo J, Kang JW, Kang H, Ratajczak J, Ratajczak MZ, et al. Genome-wide analysis of murine bone marrowderived very small embryonic-like stem cells reveals that mitogenic growth factor signaling pathways play a crucial role in the quiescence and ageing of these cells. Int J Mol Med. 2013;32:281–290. doi: 10.3892/ijmm.2013.1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin DM, Kucia M, Ratajczak MZ. Nuclear and chromatin reorganization during cell senescence and aging - a mini-review. Gerontology. 2011;57:76–84. doi: 10.1159/000281882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ragina NP, Schlosser K, Knott JG, Senagore PK, Swiatek PJ, Chang EA, et al. Downregulation of H19 improves the differentiation potential of mouse parthenogenetic embryonic stem cells. Stem Cells Dev. 2012;21:1134–1144. doi: 10.1089/scd.2011.0152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T, et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275:964–966. doi: 10.1126/science.275.5302.964. [DOI] [PubMed] [Google Scholar]
- Prockop DJ. Marrow Stromal Cells as Stem Cells for Nonhematopoietic Tissues. Science. 1997;276:71–74. doi: 10.1126/science.276.5309.71. [DOI] [PubMed] [Google Scholar]
- Zuba-Surma EK, Kucia M, Ratajczak J, Ratajczak MZ. ‘Small stem cells' in adult tissues: very small embryonic-like stem cells stand up! Cytometry Part A. 2009;75A:4–13. doi: 10.1002/cyto.a.20665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGuckin CP, Forraz N, Baradez MO, Navran S, Zhao J, Urban R, et al. Production of stem cells with embryonic characteristics from human umbilical cord blood. Cell Prolif. 2005;38:245–255. doi: 10.1111/j.1365-2184.2005.00346.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wakao S, Kitada M, Kuroda Y, Shigemoto T, Matsuse D, Akashi H, et al. Multilineage-differentiating stress-enduring (Muse) cells are a primary source of induced pluripotent stem cells in human fibroblasts. Proc Natl Acad Sci USA. 2011;108:9875–9880. doi: 10.1073/pnas.1100816108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGuckin C, Jurga M, Ali H, Strbad M, Forraz N. Culture of embryonic-like stem cells from human umbilical cord blood and onward differentiation to neural cells in vitro. Nat Protoc. 2008;3:1046–1055. doi: 10.1038/nprot.2008.69. [DOI] [PubMed] [Google Scholar]
- Jones R, Collector M, Barber J, Vala M, Fackler M, May W, et al. Characterization of mouse lymphohematopoietic stem cells lacking spleen colony-forming activity. Blood. 1996;88:487–491. [PubMed] [Google Scholar]
- Jones RJ, Wagner JE, Celano P, Zicha MS, Sharkis SJ. Separation of pluripotent haematopoietic stem cells from spleen colony-forming cells. Nature. 1990;347:188–189. doi: 10.1038/347188a0. [DOI] [PubMed] [Google Scholar]
- Krause DS. Bone marrow-derived cells and stem cells in lung repair. Proc Am Thorac Soc. 2008;5:323–327. doi: 10.1513/pats.200712-169DR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vacanti MP, Roy A, Cortiella J, Bonassar L, Vacanti CA. Identification and initial characterization of spore-like cells in adult mammals. J Cell Biochem. 2001;80:455–460. [PubMed] [Google Scholar]
- Virant-Klun I, Zech N, Rožman P, Vogler A, Cvjetičanin B, Klemenc P, et al. Putative stem cells with an embryonic character isolated from the ovarian surface epithelium of women with no naturally present follicles and oocytes. Differentiation. 2008;76:843–856. doi: 10.1111/j.1432-0436.2008.00268.x. [DOI] [PubMed] [Google Scholar]
- Bhartiya D, Kasiviswanathan S, Unni SK, Pethe P, Dhabalia JV, Patwardhan S, et al. Newer insights into premeiotic development of germ cells in adult human testis using Oct-4 as a stem cell marker. J Histochem Cytochem. 2010;58:1093–1106. doi: 10.1369/jhc.2010.956870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sovalat H, Scrofani M, Eidenschenk A, Pasquet S, Rimelen V, Hénon P. Identification and isolation from either adult human bone marrow or G-CSF−mobilized peripheral blood of CD34+/CD133+/CXCR4+/ Lin−CD45− cells, featuring morphological, molecular, and phenotypic characteristics of very small embryonic-like (VSEL) stem cells. Exp Hematol. 2011;39:495–505. doi: 10.1016/j.exphem.2011.01.003. [DOI] [PubMed] [Google Scholar]
- Mikhail MA, M'Hamdi H, Welsh J, Levičar N, Marley SB, Nicholls JP, et al. High frequency of fetal cells within a primitive stem cell population in maternal blood. Hum Reprod. 2008;23:928–933. doi: 10.1093/humrep/dem417. [DOI] [PubMed] [Google Scholar]
- Liu Y, Gao L, Zuba-Surma EK, Peng X, Kucia M, Ratajczak MZ, et al. Identification of small Sca-1+, Lin−, CD45− multipotential cells in the neonatal murine retina. Exp Hematol. 2009;37:1096–1107.e1. doi: 10.1016/j.exphem.2009.05.014. [DOI] [PubMed] [Google Scholar]
- Goldenberg-Cohen N, Avraham-Lubin BC, Sadikov T, Goldstein RS, Askenasy N. Primitive stem cells derived from bone marrow express glial and neuronal markers and support revascularization in injured retina exposed to ischemic and mechanical damage. Stem Cells Dev. 2012;21:1488–1500. doi: 10.1089/scd.2011.0366. [DOI] [PubMed] [Google Scholar]
- Iskovich S, Goldenberg-Cohen N, Stein J, Yaniv I, Fabian I, Askenasy N. Elutriated stem cells derived from the adult bone marrow differentiate into insulin-producing cells in vivo and reverse chemical diabetes. Stem Cells Dev. 2012;21:86–96. doi: 10.1089/scd.2011.0057. [DOI] [PubMed] [Google Scholar]
- Brons IGM, Smithers LE, Trotter MWB, Rugg-Gunn P, Sun B, Chuva de Sousa Lopes SM, et al. Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature. 2007;448:191–195. doi: 10.1038/nature05950. [DOI] [PubMed] [Google Scholar]
- Tesar PJ, Chenoweth JG, Brook FA, Davies TJ, Evans EP, Mack DL, et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature. 2007;448:196–199. doi: 10.1038/nature05972. [DOI] [PubMed] [Google Scholar]
- Shin DM, Liu R, Klich I, Ratajczak J, Kucia M, Ratajczak MZ. Molecular characterization of isolated from murine adult tissues very small embryonic/epiblast like stem cells (VSELs) Mol Cell. 2010;29:533–538. doi: 10.1007/s10059-010-0081-4. [DOI] [PubMed] [Google Scholar]
- Shin DM, Liu R, Klich I, Wu W, Ratajczak J, Kucia M, et al. Molecular signature of adult bone marrow-purified very small embryonic-like stem cells supports their developmental epiblast/germ line origin. Leukemia. 2010;24:1450–1461. doi: 10.1038/leu.2010.121. [DOI] [PubMed] [Google Scholar]
- Shin DM, Zuba-Surma EK, Wu W, Ratajczak J, Wysoczynski M, Ratajczak MZ, et al. Novel epigenetic mechanisms that control pluripotency and quiescence of adult bone marrow-derived Oct4(+) very small embryonic-like stem cells. Leukemia. 2009;23:2042–2051. doi: 10.1038/leu.2009.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratajczak MZ, Zuba-Surma E, Wojakowski W, Suszynska M, Mierzejewska K, Liu R, et al. Very Small Embryonic Like Stem Cells (VSELs) represent a real challenge in stem cell biology. Recent pros and cons in the midst of a lively debate Leukemia 2013. doi: 10.1038/leu.2013.255 [DOI] [PMC free article] [PubMed]
- Kassmer SH, Krause DS. Very small embryonic like stem cells: Biology and function of these potential endogenous pluripotent stem cells in adult tissues. Mol Reprod Dev. 2013;80:677–690. doi: 10.1002/mrd.22168. [DOI] [PMC free article] [PubMed] [Google Scholar]