Summary
The release of membrane vesicles from the surface of cells into their surrounding environment is now recognized as an important pathway for the delivery of proteins to extracellular sites of biological function. Membrane vesicles of this kind, termed exosomes and ectosomes, are the result of active processes and have been shown to carry a wide array of biological effector molecules that can play roles in cell-to-cell communication and remodeling of the extracellular space [1–7]. Degradation of the extracellular matrix (ECM) through the regulated release of proteolytic enzymes is a key process for development, morphogenesis and cell migration in animal and plant cells. Here we show that the unicellular alga, Chlamydomonas, achieves the timely degradation of its mother cell wall, a type of ECM, through the budding of ectosomes directly from the membranes of their flagella. Using a combination of immunoelectron microscopy, immunofluorescence microscopy, and functional analysis, we demonstrate that these vesicles, which we term ciliary ectosomes, act as carriers of the proteolytic enzyme necessary for the liberation of daughter cells following mitosis [8, 9]. Chlamydomonas has proven to be the key unicellular model for the highly conserved mechanisms of mammalian cilia, and our results suggest that cilia may be an under-appreciated source of bioactive, extracellular membrane vesicles.
Results
Ciliary ecotosomes observed by TEM
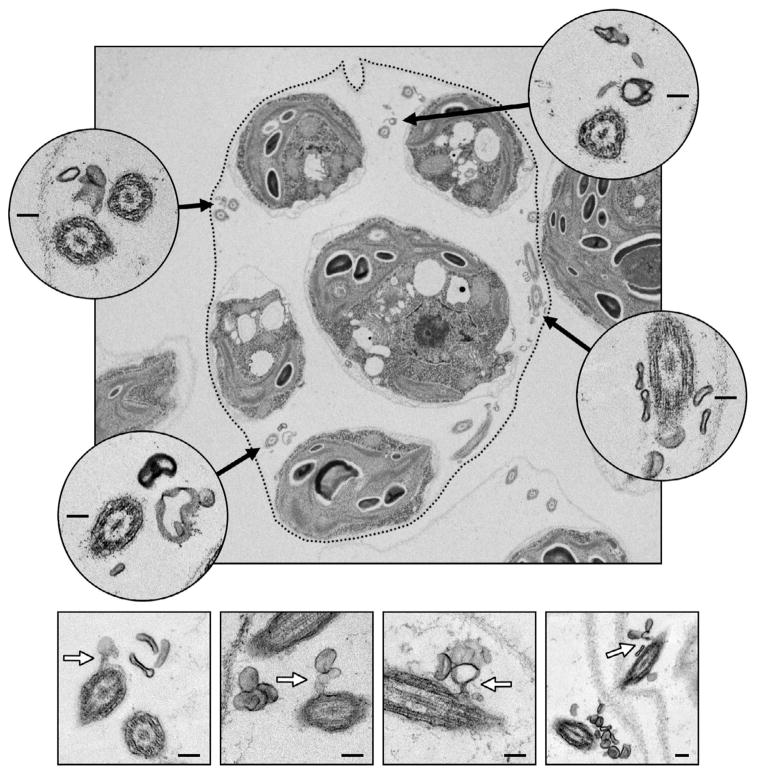
The fine structure of mature Chlamydomonas sporangia (see Figure 1 for an illustration of sporangium formation) was examined by transmission electron microscopy (TEM). The top panel in Figure 2 displays a TEM micrograph of an ultrathin section through a typical sporangium harvested at a stage just prior to hatching (stage V as illustrated in Figure 1). The daughter cells of the sporangium are flagellated by this time, and cross sections through several flagella are apparent. Most striking is the observation that the flagella are surrounded by numerous membrane vesicles ranging in diameter from approximately 50 nm – 200 nm (Figure 2, insets). In Figure 2’s four lower panels, examples are shown of vesicles caught in the process of budding directly from the membranes of daughter cell flagella. Given that daughter cells are encased in their own individual cell walls at this stage, totally restricting access of their plasma membranes to the surrounding medium, the membrane vesicles observed are evidently of flagellar origin. A vesicle released from the membrane of a cilium or flagellum is termed here a ciliary ectosome.
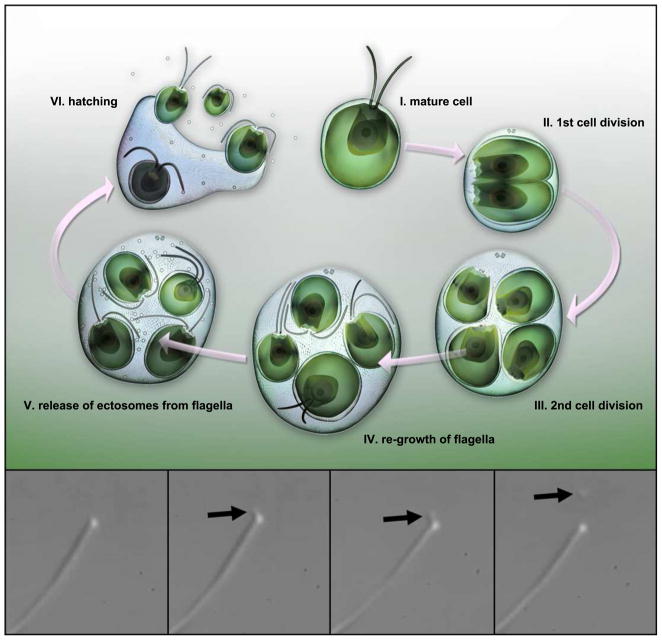
Figure 1. Illustration of the Chlamydomonas life-cycle.
A cell grows to a threshold size (I), resorbs its flagella, then typically undergoes multiple rounds of cell division (II – III) within the original, mother cell wall. The resulting ball of cells is termed a sporangium. Daughter cells re-grow flagella while they are still inside of the sporangium (IV). Ectosomes carrying protease are released from the flagella of daughter cells (V). Hatching occurs when the ectosome-associated protease digests the mother cell wall liberating daughter cells from the sporangium (VI). The four lower panels display sequential frames taken from a DIC video micrograph of a flagellum of a vegetative Chlamydomonas cell. Black arrows indicate a ciliary ectosome budding from the tip of the flagellum. The source video, Movie S1, can be found in the supplemental data.
Figure 2. Ciliary ectosomes observed in the mature sporangium by electron microscopy.
In the upper panel, an ultrathin section through a mature sporangium reveals five of the daughter cells within the mother cell wall prior to hatching. The location of the mother cell wall is emphasized by a dotted line. Four insets show higher magnification views of cross sections through flagella (black arrows indicate their position inside the sporangium). Numerous ectosomes are observed clustering around the flagella. The four lower panels display examples of ectosomes caught in the process of budding directly from the membranes of sporangial daughter cell flagella (white arrows). Inset scale bars indicate 100 nm.
A previous study reported that vegetative lytic enzyme (VLE), the enzyme responsible for breakdown of the mother cell wall, is found in the flagella of daughter cells prior to hatching [8]. Left unknown, however, was the means by which VLE is delivered from flagella to its eventual site of action on the inner surface of the mother cell wall. The release of ectosomes from the flagella could potentially mediate such a process. Using the same antibody preparation, specific for the catalytic region of VLE protease, that was employed by Kubo et al., immunogold labeling was performed in situ with mature sporangia (Figure 3). Ciliary ectosomes were readily labeled with VLE-specific gold particles indicating that the VLE protease is released from the flagella with ectosomes capable of carrying the enzyme to its site of function.
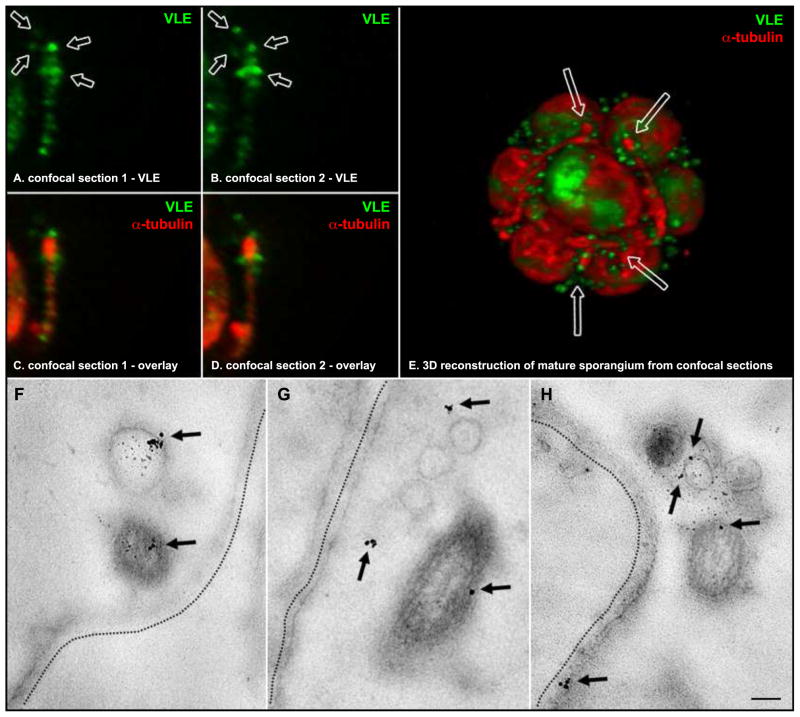
Figure 3. Localization of VLE protease on ciliary ectosomes in the mature sporangium.
A and B are sequential, 250 nm, confocal sections through a flagellum of a daughter cell within a mature sporangium. VLE-specific fluorescence appears in green, and arrows point to discreet puncta clustered around the distal end of the flagellum. C and D display the same confocal sections as A and B, respectively, overlaying an α-tubulin-specific fluorescence channel in red (the flagellar axoneme). E shows a view from a 3D reconstruction of a mature sporangium consisting of 8 daughter cells preparing to hatch from their mother cell wall. VLE-specific fluorescence appears as green discreet puncta populating the interior space of the sporangium, between the daughter cells and the mother cell wall. Arrows point to the distal regions of four flagella surrounded by clusters of VLE puncta. Representative images from in situ immunogold labeling of mature sporangia with VLE antibodies are shown in the bottom panels. F – H are electron micrographs of ultrathin sections through three different sporangia at a stage just prior to hatching. Flagella (F) emanating from daughter cells within the mother cell wall (emphasized by dotted lines) have released ectosomes with VLE protease on their surface. Arrows point to the location of VLE-specific gold particles. A scale bar in panel H indicates 100 nm.
Immunofluorescence localization of VLE
Laser scanning confocal fluorescence microscopy was used to reconstruct the pattern of VLE localization in three dimensions. At the time of hatching, VLE accumulates in the sporangial flagella and appears as a series of discreet puncta along their length (Figure 3). A clustering of VLE puncta was often observed toward the distal end of flagella (Figure 3, indicated by arrows in single confocal sections A, B; and in 3D reconstruction E). The interior space between daughter cells and the mother cell walls of mature sporangia is populated by numerous discreet VLE puncta (Figure 3, E). This pattern of fluorescence is consistent with the ectosomal localization observed by in situ gold labeling (Figure 3, lower panels). VLE does not appear in the flagella of cells that are liberated from the sporangium (data not shown). This is in agreement with a previously published expression profile indicating that VLE protease is only expressed during the cell division and hatching phase [8].
Isolated ciliary ectosomes are enzymatically active
Ciliary ectosomes were isolated by differential centrifugation of media from synchronously hatching sporangia. Intact, isolated ciliary ectosomes were examined by immunogold labeling using antibodies specific for VLE (Figure 4). Negative staining of the preparation revealed an array of membrane vesicles ranging in diameter from approximately 50 nm to 200 nm, consistent with the dimensions and appearance of ciliary ectosomes in situ (Figures 2 and 3). Isolated ciliary ectosomes were highly amenable to immunogold labeling with VLE-specific antibodies. Prominent decoration of the outside surfaces of intact ciliary ectosomes with gold particles in this experiment indicates that the active region of VLE protease resides on the outside of the ectosome membranes, and by extension the outside of the flagellar membranes from which they derive.
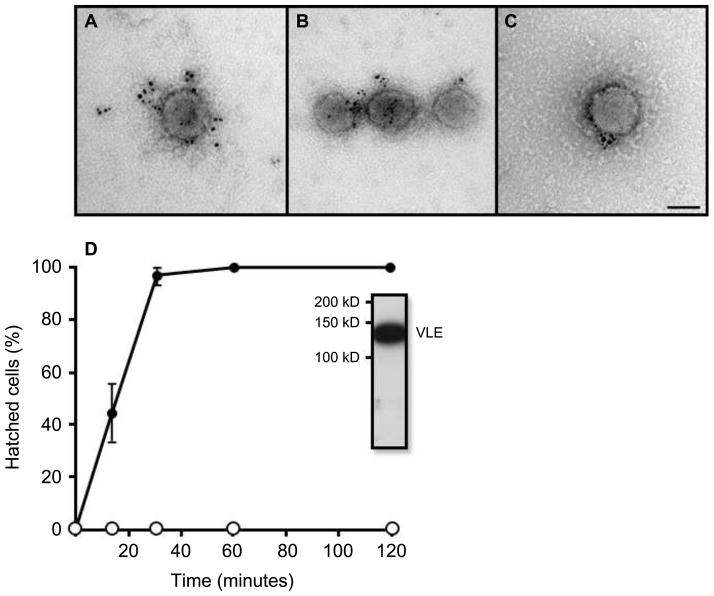
Figure 4. Isolated ciliary ectosomes carry VLE protease and induce hatching when added to hatching-defective mutants.
A – C show numerous VLE-specific gold particles associated with the outer surfaces of isolated, intact ciliary ectosomes. The scale bar in C indicates 100 nm. D is a graph showing that ciliary ectosomes isolated from the medium of hatched pf1 cells induce complete hatching of ift88 mutants over a time course of ~30 minutes. Black circles indicate the data from ciliary ectosome addition. Open circles indicate control data from addition of the final supernatant from the ciliary ectosome preparation. Immunoblot analysis of isolated ectosomes with VLE antibody shows a band at ~125 kD.
To examine the activity of the ectosomal VLE protease, isolated ciliary ectosomes were added to flagella-less, hatching-defective, IFT88 null mutant cells. An incubation time course of approximately 30 minutes was sufficient to induce hatching of daughter cells from 100% of IFT88 sporangia verifying that the VLE protease carried by ciliary ectosomes is active (Figure 4). A control experiment performed in parallel using an equal protein amount of the supernatant that remained after pelleting of ciliary ectosomes was unable to induce any hatching of IFT88 cells over a time course of 2 hours. Immunoblot analysis of the isolated ciliary ectosomes with VLE antibody shows a strong signal at the expected molecular weight of ~125 kD (Figure 4, inset).
Discussion
Documented here is the ability of the cilium to function as a secretory organelle via the release of ciliary ectosomes. The Chlamydomonas model system offers a combination of advantages uniquely suited for the study of this phenomenon. The cell is fully encased within a cell wall with the exception of two anteriorly placed channels through which the flagella extend. As a consequence, the only membrane surface exposed to the surrounding milieu is that of the flagella. This effectively narrows down the source of extracellular vesicles to the flagellar membrane and alleviates what would be a non-trivial problem of experimentally differentiating between plasma membrane-derived ectosomes and those of ciliary origin. Adding to this advantage in the case of this study is the nature of sporangium formation during Chlamydomonas cell division. As illustrated in Figure 1, at the time of cell division multiple fissions take place within the original cell wall. The result is a cluster of flagellated daughter cells encapsulated in an expanded mother cell wall. Thus, each mature sporangium is a set of cells with the capacity to release functional ectosomes into an enclosed microenvironment amenable to study. Previous work revealed that in order for daughter cells to be liberated from the sporangium, they must release a protease, VLE, from their flagella into the interior space between the cells and the mother cell wall [8, 10]. Here, we show that the cells accomplish this by packaging the VLE protease with its active site on the outer surface of ectosomes that bud directly from the flagellar membrane. These ectosomes then diffuse through the interior space of the sporangium where they can transport the protease from the cell to the mother cell wall where it carries out its degradative function. Using a polyclonal antibody raised to the catalytic region of VLE protease [8] with in situ immunogold labeling and confocal fluorescence microscopy procedures (Figure 3), we have made a definitive determination of the enzyme’s localization to ciliary ectosomes at the time of hatching. Ciliary ectosomes were then isolated from the medium of synchronously hatching sporangia by differential centrifugation. Immunogold labeling of intact, whole-mounted ciliary ectosomes demonstrates that the catalytic region of VLE protease is present on their outer membrane surface (Figure 4). Western blot analysis of isolated ciliary ectosome protein content verifies the presence of VLE (Figure 4), and other known flagellar membrane proteins such as the flagella membrane glycoprotein (FMG-1) and polycystic kidney disease 2 protein (PKD2) (Figure S1).
The failure or inefficiency of sporangial hatching is a common phenotype expressed by the many flagellar assembly mutants of Chlamydomonas [11, 12]. To test the activity of the ectosome-associated VLE protease, we exposed sporangia of IFT88 null mutants, which lack flagella and cannot hatch, to isolated ciliary ectosomes. The ciliary ectosomes were capable of inducing hatching with high efficiency, verifying that the ectosomes carry the VLE protease activity (Figure 4). This result suggests that the sporangial hatching difficulties typically observed with daughter cells of flagella-defective mutants may be due to the necessity of the flagellum as a secretory organelle. By this logic, daughter cells with absent or misassembled flagella would be defective in the release of ectosomes that mediate the delivery of the VLE protease to the mother cell wall. An interesting aspect of this finding is the fact that the isolated ciliary ectosomes induced hatching of the IFT88 null sporangia from the outside of their mother cell walls in our experiments. This means that the VLE protease can degrade a mother cell wall from either side, yet does not seem to degrade individual daughter cell walls, whose outer surface would be exposed to VLE within the mature sporangia. Perhaps an initially resistant vegetative cell wall undergoes some change in constitution during the process of becoming a mother cell wall that makes it competent for degradation by VLE protease. Previous studies report that a soluble form of active VLE protease, not associated with membrane, can also be isolated from the medium of hatched sporangia [8, 10]. This suggests that the ectosome-associated VLE may be cleaved at a site downstream of its transmembrane domain freeing it from the ectosome at some stage following its release from the flagella. The hatching enzyme, VheA, of the related alga Volvox cateri [13], as well as the gamete lytic enzyme (GLE) of Chlamydomonas [14], are expressed initially as proenzymes which require a proteolytic processing event in order to take on their final active form. One possibility is that the VLE protease is cleaved by a second, activating protease that awaits the ectosome at its destination on the surface of the mother cell wall.
The expression of VLE protease is tightly regulated, with transcription and subsequent accumulation of the protein occurring precisely at the time of hatching [8]. Such regulation at the level of protein expression allows for at least two general models by which secretion of a specific protein via ciliary ectosomes might be temporally regulated. In one model, the cell would localize the protein to cilia and control its secretion by temporally regulating the mechanism of ciliary ectosome formation. In another model, ectosome release from cilia would take place constitutively and the timed secretion of a specific protein would be controlled at the level of its synthesis and delivery to the ciliary compartment. The release of ectosomes is thought to be a constitutive, on-going process for a number of different cell types including osteoblasts [15], endothelial cells [16] and glomerular epithelial cells [17, 18]. A variety of different cancerous cells have also been shown to release ectosomes in an apparently constitutive manner in the absence of any stimulus [4, 19–21]. The release of ectosomes from the flagella of Chlamydomonas appears to be an on-going process even in vegetative cells (Figure 1 and supplemental video).
Having been regarded classically as an organelle of primarily motile function, the cilium has undergone a renaissance of appreciation in the past decade touched off by research in Chlamydomonas that revealed its role as a ubiquitous sensory antenna [22, 23]. The ensuing growth of a vibrant research community around the topic has given rise to what is now an extensive literature on the role of cilia in sensing and transducing signals from the extracellular space. The importance of cilia in this context is underscored most potently by the expanding list of ciliopathies - a classification of diseases and syndromes associated with abnormal formation or function of cilia [24, 25]. Despite its relatively small surface area compared with that of the cells from which it protrudes, the cilium acts as the obligate site of action for a variety of membrane receptors and signal transduction modules critical to basic cellular processes regulating growth, development and homeostasis [26, 27]. This capacity of the cilium to act as a specialized region of the cell where specific proteins can be readily concentrated for sensory function, also makes it an ideal organelle to employ in the sending of signals out from the cell. That is, the same mechanisms that give rise to the selective delivery and retention of sensory proteins at the ciliary compartment could conceivably generate an equally efficient and regulable signal release platform. Given the evident phenomenon of a ciliary ectosome mechanism reported here, we hypothesize that this is in fact the case and that cilia should be scrutinized not only as antennae capable of receiving signals from the environment, but also as an instrument of signal release.
Experimental Procedures
Cell culturing and synchronous hatching conditions
Chlamydomonas reinhardtii wild type strain cc1009, pf1 mutant strain (Chlamydomonas Culture Center), ift88 mutant strain (from Witman, UMass Med School), and Chlamydomonas moewusii strain cc957 (Chlamydomonas Culture Center) were used for experiments. For generation of synchronously hatching sporangia, Chlamydomonas reinhardtii were grown phototrophically in M1 medium [11] and synchronized hatching was induced as follows: 400 mL cultures or 8 L cultures were grown at 24 C and bubbled continuously with 0.5% CO2 in air under a 12 hr/12 hr light/dark regime with white light illumination provided by fluorescent bulbs. Culture density was kept between 105 and 106 cells ml−1. Under these conditions cc1009 cells begin to divide before the lights go off, but do not hatch, forming sporangia generally containing 8 cells. After several hours in the dark, the cells are almost all in such sporangia and can be induced to hatch by exposing the cells to light.
DIC video microscopy
Chlamydomonas moewusii strain cc957 was used for DIC microscopy of ectosome release because their flagella are paralyzed and approximately 50% longer than those of Chlamydomonas reinhardtii. Cells were prepared and visualized according to methods detailed previously. [28, 29]
Electron Microscopy
To look for the release of vesicles from flagella during hatching, cc1009 cells were grown synchronously as described above. Three hours after the lights went off most of the cells had divided but were still encased in their mother cell wall. Cells were centrifuged at 500 g in the dark, brought up in fresh medium and were put under a light source to stimulate hatching. Cells were fixed at 2 min intervals for TEM and light microscopy. Hatching was obvious after 8 min and complete by 20 min. For TEM, 0.5 ml of 5% glutaraldehyde was added to 0.5 ml of cells and fixed for 1 – 2 h at room temperature. Tannic acid was then added to 0.1% and fixation continued for 30 min. Cell pellets were washed in 50 mM Hepes pH 7.4, fixed for 40 min in 1% osmium tetroxide in 50 mM Hepes, washed, stained in 2% uranyl acetate for 1 hr, washed, dehydrated in ethanol and embedded in Embed 812 (Electron Microscopy Sciences). Sections were viewed with a JEOL 1230 microscope and digital images were captured with a Hamamatsu Orca HR digital camera. For detailed immunogold labeling methods please see supplemental experimental procedures.
Immunofluorescence microscopy
Sporangia of Chlamydomonas strain cc1009 were harvested at the time of hatching as above and fixed for 30 min at room temperature in MI growth medium containing 1% paraformaldehyde. The cells were then washed with phosphate buffered saline (PBS) and pipetted onto 0.1% polyethyleneimine-coated cover slips where they sank to the surface and attached over the course of 20 minutes at room temperature. To permeabilize and fix additionally, cover slips with attached cells were submerged in methanol at −20 C for 5 minutes, then transferred to fresh methanol for an additional 5 minutes at −20 C. Cells were rehydrated for 2 minutes in PBS then incubated at room temperature for 1 hour in blocking buffer (5% BSA, 1% cold water fish gelatin, and 10% goat serum in PBS). Cells were overlayed with a mixture of primary antibodies in blocking buffer and incubated in a moist chamber at 4 C overnight. After washing away primary antibodies by repeatedly dipping the coverslips in PBS, cells were overlayed with a mixture of fluorophore-conjugated secondary antibodies (Alexa Fluor 488 goat anti-rabbit IgG and Alexa Fluor 594 goat anti-mouse IgG, Invitrogen Molecular Probes) in blocking buffer and incubated at room temperature for 1 hour. Coverslips were washed by dipping in PBS as before and mounted to slides with SlowFade Antifade reagent (Invitrogen Molecular Probes). Laser scanning confocal microscopy was performed using a Zeiss LSM-510 system with an Axio Observer inverted microscope. 3D reconstructions were performed using Zeiss Zen software and individual images were adjusted using Adobe Photoshop.
Isolation of ciliary ectosomes and activity assay
The radial spoke head mutant strain, pf1, was ultimately chosen for ectosome isolation because it tends to yield higher densities of sporangia in large culture volumes. Cultures of 16 L were grown to a density of 5×106 cells/ml in tris-acetate phosphate (TAP) media (Harris, 1989). To initiate hatching, sporangia were harvested by centrifugation at low speed and placed to 10 mM Hepes buffer (pH 7.2) at a density of ~4×107 cells/ml with aeration under light. Sporangia were observed to hatch over a time course of ~20 minutes under these conditions. Differential centrifugation of the hatched cell preparation began with 3000 g for 5 min at RT to pellet the daughter cells. The resulting supernatant was subjected to an additional clarification by centrifugation at 16,000 g for 20 minutes. The ciliary ectosomes and some remaining cell wall material were pelleted from the clarified supernatant by centrifugation at 200,000 g for 45 minutes at 4 C. The resulting pellet had an upper, transparent layer composed of leftover cell wall material and a lower white layer composed of ciliary ectosomes. The transparent cell wall-containing layer was manually removed using a pipette and the remaining ciliary ectosome-containing layer was resuspended in 2 ml of 10 mM HEPES buffer. To perform a final density gradient step, the 2 ml ciliary ectosome specimen was mixed with a preparation of 2.1 g of cesium chloride dissolved in 2 ml of 10 mM Hepes buffer and raised by addition of Hepes buffer to a final volume of 4.5 ml. The final concentration of cesium chloride was 2.8 M. The mixture was subjected to centrifugation at 250,000 g for 14 hours. A major white band, enriched for ciliary ectosomes, and two minor bands containing cell wall material and other debris were the result. The ciliary ectosome band was isolated using a pipette and was analyzed by negative staining and immunogold labeling.
For use in functional analysis, the above ciliary ectosome isolation procedure was simplified to avoid exposure of the enzyme to cesium. The supernatant following centrifugation at 16,000 g was subjected to centrifugation at 46,000 g for 30 minutes. The resulting supernatant was collected and centrifuged again at 200,000 g for 45 minutes. The white ciliary ectosome pellet was harvested at this point and resuspended in 0.2 ml of 10 mM Hepes buffer. Protein concentrations of the ciliary ectosome specimen and the final supernatant were determined by using amido black (Popov et al., 1975). Equal protein amounts of ciliary ectosome specimen and supernatant were adjusted to the same volume of 100 μl. The ift88 mutant strain was cultured in TAP medium and all of the cells accumulated as sporangia that could not hatch due to the absence of flagella (Pazour et al., 2000). The ift88 sporangia were harvested when they reached a density of 5×106/ml. To perform the hatching activity assay, 100 μl of ift88 sporangia was mixed with 100 μl of ciliary ectosome specimen or supernatant. Aliquots of 25μl were removed at time intervals of 15 min, 30 min, 60 min and 120 min. Counts of liberated daughter cells and unhatched sporangia were made using a hemacytometer.
Supplementary Material
Ciliary ectosomes isolated by differential centrifugation were compared to isolated flagellar membranes by equal protein loads to SDSPAGE and western blotting with antibodies specific for the flagellar membrane glycoprotein (FMG-1) and polycystic kidney disease 2 protein (PKD2). Both of these known proteins of the flagellar membrane are found prominently in ciliary ectosomes.
A DIC video micrograph depicts the release of ciliary ectosomes from a Chlamydomonas flagellum. The still frames shown in Figure 1 derive from this video.
Highlights.
Ectosomes can bud directly from the membranes of cilia
Ciliary ectosomes carry the protease necessary for hatching in Chlamydomonas
Cilia may function to release signals in addition to their role as sensory antennae
Acknowledgments
We thank Keith Kozminski for the use of unpublished results (PhD thesis) on vesicle release in real time from the tips of Chlamydomonas flagella.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Mathivanan S, Ji H, Simpson RJ. Exosomes: extracellular organelles important in intercellular communication. Journal of proteomics. 2010;73:1907–1920. doi: 10.1016/j.jprot.2010.06.006. [DOI] [PubMed] [Google Scholar]
- 2.Taraboletti G, D’Ascenzo S, Borsotti P, Giavazzi R, Pavan A, Dolo V. Shedding of the matrix metalloproteinases MMP-2, MMP-9, and MT1-MMP as membrane vesicle-associated components by endothelial cells. The American journal of pathology. 2002;160:673–680. doi: 10.1016/S0002-9440(10)64887-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ginestra A, Monea S, Seghezzi G, Dolo V, Nagase H, Mignatti P, Vittorelli ML. Urokinase plasminogen activator and gelatinases are associated with membrane vesicles shed by human HT1080 fibrosarcoma cells. J Biol Chem. 1997;272:17216–17222. doi: 10.1074/jbc.272.27.17216. [DOI] [PubMed] [Google Scholar]
- 4.Dolo V, Ginestra A, Cassara D, Violini S, Lucania G, Torrisi MR, Nagase H, Canevari S, Pavan A, Vittorelli ML. Selective localization of matrix metalloproteinase 9, beta1 integrins, and human lymphocyte antigen class I molecules on membrane vesicles shed by 8701-BC breast carcinoma cells. Cancer Res. 1998;58:4468–4474. [PubMed] [Google Scholar]
- 5.Giusti I, D’Ascenzo S, Millimaggi D, Taraboletti G, Carta G, Franceschini N, Pavan A, Dolo V. Cathepsin B mediates the pH-dependent proinvasive activity of tumor-shed microvesicles. Neoplasia. 2008;10:481–488. doi: 10.1593/neo.08178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gutwein P, Stoeck A, Riedle S, Gast D, Runz S, Condon TP, Marme A, Phong MC, Linderkamp O, Skorokhod A, et al. Cleavage of L1 in exosomes and apoptotic membrane vesicles released from ovarian carcinoma cells. Clin Cancer Res. 2005;11:2492–2501. doi: 10.1158/1078-0432.CCR-04-1688. [DOI] [PubMed] [Google Scholar]
- 7.Stoeck A, Keller S, Riedle S, Sanderson MP, Runz S, Le Naour F, Gutwein P, Ludwig A, Rubinstein E, Altevogt P. A role for exosomes in the constitutive and stimulus-induced ectodomain cleavage of L1 and CD44. Biochem J. 2006;393:609–618. doi: 10.1042/BJ20051013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kubo T, Kaida S, Abe J, Saito T, Fukuzawa H, Matsuda Y. The Chlamydomonas hatching enzyme, sporangin, is expressed in specific phases of the cell cycle and is localized to the flagella of daughter cells within the sporangial cell wall. Plant Cell Physiol. 2009;50:572–583. doi: 10.1093/pcp/pcp016. [DOI] [PubMed] [Google Scholar]
- 9.Schlösser UG. Release of reproduction cells by action of cell wall autolytic factors in Chlamydomonas and Germinella. Ber Deutsch Bot Ges. 1981;94:373–374. [Google Scholar]
- 10.Matsuda Y, Koseki M, Shimada T, Saito T. Purification and characterization of a vegetative lytic enzyme responsible for liberation of daughter cells during the proliferation of Chlamydomonas reinhardtii. Plant Cell Physiol. 1995;36:681–689. [PubMed] [Google Scholar]
- 11.Harris EH. A Comprehensive Guide to Biology and Laboratory Use. San Diego, CA: Academic Press; 1989. The Chlamydomonas Sourcebook. [DOI] [PubMed] [Google Scholar]
- 12.Goodenough UW, St Clair HS. BALD-2: a mutation affecting the formation of doublet and triplet sets of microtubules in Chlamydomonas reinhardtii. J Cell Biol. 1975;66:480–491. doi: 10.1083/jcb.66.3.480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kirk DL, Harper JF. Genetic, biochemical, and molecular approaches to Volvox development and evolution. Int Rev Cytol. 1986;99:217–293. doi: 10.1016/s0074-7696(08)61428-x. [DOI] [PubMed] [Google Scholar]
- 14.Buchanan MJ, Imam SH, Eskue WA, Snell WJ. Activation of the cell wall degrading protease, lysin, during sexual signalling in Chlamydomonas: the enzyme is stored as an inactive, higher relative molecular mass precursor in the periplasm. J Cell Biol. 1989;108:199–207. doi: 10.1083/jcb.108.1.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Anderson HC. Matrix vesicles and calcification. Current rheumatology reports. 2003;5:222–226. doi: 10.1007/s11926-003-0071-z. [DOI] [PubMed] [Google Scholar]
- 16.Freyssinet JM. Cellular microparticles: what are they bad or good for? Journal of thrombosis and haemostasis : JTH. 2003;1:1655–1662. doi: 10.1046/j.1538-7836.2003.00309.x. [DOI] [PubMed] [Google Scholar]
- 17.Pascual M, Steiger G, Sadallah S, Paccaud JP, Carpentier JL, James R, Schifferli JA. Identification of membrane-bound CR1 (CD35) in human urine: evidence for its release by glomerular podocytes. J Exp Med. 1994;179:889–899. doi: 10.1084/jem.179.3.889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lescuyer P, Pernin A, Hainard A, Bigeire C, Burgess JA, Zimmermann-Ivol C, Sanchez JC, Schifferli JA, Hochstrasser DF, Moll S. Proteomic analysis of a podocyte vesicle-enriched fraction from human normal and pathological urine samples. Proteomics Clinical applications. 2008;2:1008–1018. doi: 10.1002/prca.200800033. [DOI] [PubMed] [Google Scholar]
- 19.Sadallah S, Eken C, Schifferli JA. Ectosomes as modulators of inflammation and immunity. Clin Exp Immunol. 2011;163:26–32. doi: 10.1111/j.1365-2249.2010.04271.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Al-Nedawi K, Meehan B, Micallef J, Lhotak V, May L, Guha A, Rak J. Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells. Nat Cell Biol. 2008;10:619–624. doi: 10.1038/ncb1725. [DOI] [PubMed] [Google Scholar]
- 21.Johnstone RM. Exosomes biological significance: A concise review. Blood cells, molecules & diseases. 2006;36:315–321. doi: 10.1016/j.bcmd.2005.12.001. [DOI] [PubMed] [Google Scholar]
- 22.Pazour GJ, Witman GB. The vertebrate primary cilium is a sensory organelle. Curr Opin Cell Biol. 2003;15:105–110. doi: 10.1016/s0955-0674(02)00012-1. [DOI] [PubMed] [Google Scholar]
- 23.Pazour GJ, Dickert BL, Vucica Y, Seeley ES, Rosenbaum JL, Witman GB, Cole DG. Chlamydomonas IFT88 and its mouse homologue, polycystic kidney disease gene tg737, are required for assembly of cilia and flagella. J Cell Biol. 2000;151:709–718. doi: 10.1083/jcb.151.3.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Waters AM, Beales PL. Ciliopathies: an expanding disease spectrum. Pediatr Nephrol. 2011;26:1039–1056. doi: 10.1007/s00467-010-1731-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sharma N, Berbari NF, Yoder BK. Ciliary dysfunction in developmental abnormalities and diseases. Curr Top Dev Biol. 2008;85:371–427. doi: 10.1016/S0070-2153(08)00813-2. [DOI] [PubMed] [Google Scholar]
- 26.Seeger-Nukpezah T, Golemis EA. The extracellular matrix and ciliary signaling. Curr Opin Cell Biol. 2012;24:652–661. doi: 10.1016/j.ceb.2012.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Christensen ST, Pedersen LB, Schneider L, Satir P. Sensory cilia and integration of signal transduction in human health and disease. Traffic. 2007;8:97–109. doi: 10.1111/j.1600-0854.2006.00516.x. [DOI] [PubMed] [Google Scholar]
- 28.Kozminski KG. High-resolution imaging of flagella. Methods Cell Biol. 1995;47:263–271. doi: 10.1016/s0091-679x(08)60819-5. [DOI] [PubMed] [Google Scholar]
- 29.Kozminski KG, Johnson KA, Forscher P, Rosenbaum JL. A motility in the eukaryotic flagellum unrelated to flagellar beating. Proc Natl Acad Sci USA. 1993;90:5519–5523. doi: 10.1073/pnas.90.12.5519. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Ciliary ectosomes isolated by differential centrifugation were compared to isolated flagellar membranes by equal protein loads to SDSPAGE and western blotting with antibodies specific for the flagellar membrane glycoprotein (FMG-1) and polycystic kidney disease 2 protein (PKD2). Both of these known proteins of the flagellar membrane are found prominently in ciliary ectosomes.
A DIC video micrograph depicts the release of ciliary ectosomes from a Chlamydomonas flagellum. The still frames shown in Figure 1 derive from this video.