Abstract
Introduction
Placental glutamine synthesis has been demonstrated in animals and is thought to increase the availability of this metabolically important amino acid to the fetus. Glutamine is of fundamental importance for cellular replication, cellular function and inter-organ nitrogen transfer. The objective of this study was to investigate the role of glutamate/glutamine metabolism by the isolated perfused human placenta in the provision of glutamine to the fetus.
Methods
Glutamate metabolism was investigated in the isolated dually perfused human placental cotyledon. U–13C-glutamate was used to investigate the movement of carbon and 15N-leucine to study movement of amino-nitrogen. Labelled amino acids were perfused via maternal or fetal arteries at defined flow rates. The enrichment and concentration of amino acids in the maternal and fetal veins were measured following 5 h of perfusion.
Results
Glutamate taken up from the maternal and fetal circulations was primarily converted into glutamine the majority of which was released into the maternal circulation. The glutamine transporter SNAT5 was localised to the maternal-facing membrane of the syncytiotrophoblast. Enrichment of 13C or 15N glutamine in placental tissue was lower than in either the maternal or fetal circulation, suggesting metabolic compartmentalisation within the syncytiotrophoblast.
Discussion
Placental glutamine synthesis may help ensure the placenta's ability to supply this amino acid to the fetus does not become limiting to fetal growth. Glutamine synthesis may also influence placental transport of other amino acids, metabolism, nitrogen flux and cellular regulation.
Conclusions
Placental glutamine synthesis may therefore be a central mechanism in ensuring that the human fetus receives adequate nutrition and is able to maintain growth.
Keywords: Amino acid, Metabolism, Glutamate, Glutamine, System N
1. Introduction
Glutamine and glutamate fulfil important metabolic functions underpinning cellular growth [1,2]. During gestation the growing fetus will require substantial amounts of these amino acids and evidence from animals suggests that placental synthesis of glutamine helps meet fetal demand. However the extent to which placental glutamate and glutamine metabolism occur in human placenta and its importance to fetal nutrition is not well understood.
Studies in the isolated perfused human placenta suggest that glutamate is converted to another amino acid, probably glutamine, as it crosses the placenta [3]. This is in keeping with studies in sheep and other species which show that the placenta synthesises glutamine from glutamate [4–7]. In sheep, production of glutamine has been shown to incorporate nitrogen from branched chain amino acids [8]. Furthermore, in sheep placental–fetal glutamine–glutamate cycling has been demonstrated suggesting that metabolic interaction between the placenta and other fetal organs is important in providing the fetus with the nutrients it requires [4]. However, despite their importance for provision of amino acids to the fetus these processes are not well characterised in humans and as we have demonstrated previously it cannot be assumed that metabolic processes occurring in other species also occur in humans [9].
Fetal nutrition is not simply about the amount of nutrients reaching the fetus, the balance of nutrients is also important. The relative availability of specific amino acids may determine the pace and the nature of fetal growth and development. Thus the relative composition of amino acids transferred to the fetus may be as important as their quantity. For instance, high rates of cellular replication in the rapidly growing fetus create an enhanced demand for nucleotide synthesis and hence the preferential utilisation of glutamine, an absolute requirement for actively dividing cells [10]. Glutamine is now regarded as a conditionally essential amino acid and fetal development may be one of those times when its provision is essential [11].
Placental synthesis may provide an important source of the conditionally essential amino acid glutamine to the growing fetus. The aim of this study is therefore to investigate the role of placental glutamate–glutamine metabolism in the provision of glutamine to the human fetus.
2. Methods
Human placentas were collected from daytime normal term deliveries from uncomplicated pregnancies at the Princess Anne Hospital in Southampton. Ethical approval from the Southampton and Southwest Hampshire Regional Ethics Committee (REC approval number 308/03/w).
2.1. Perfusions
Placentas were perfused using the methodology of Schneider et al. [12], as adapted in our laboratory [13,14]. Placentas were collected within 30 min of delivery and placed on ice for transport to the laboratory where fetal side perfusion was established within approximately 30 min of collection. The fetal and maternal circulations were perfused with Earle's bicarbonate buffer ((EBB) (mm): 1.8 CaCl2, 0.4 MgSO4, 116.4 NaCl, 5.4 KCl, 26.2 NaHCO3, 0.9 NaH2PO4, 5.5 glucose, containing 0.1% bovine serum albumin, and 5000 IU L−1 heparin, equilibrated with 95% O2 – 5% CO2) at 6 and 14 ml/min, respectively. Perfusion of the fetal circulation was established first, and, if fetal venous outflow was ≥95% of fetal arterial inflow, the maternal arterial perfusion was established 15 min later. Perfusion was performed for 5 h and samples were collected from the maternal and fetal veins every hour. At the end of this perfusion period the maternal and fetal circulations were washed out with EBB for 15 min, the cotyledon trimmed of non-perfused areas (perfused areas become white) and the cotyledon frozen for analysis of intracellular amino acids. In 6 placentas where fetal side recovery did not reach 95% maternal and fetal perfusion was performed for 15–20 min to wash out blood and the tissue was trimmed of non-perfused areas and frozen for analysis of baseline intracellular amino acid levels.
Four experimental protocols for placental perfusion with stable isotope labelled amino acids were performed as described in Table 1. 15N-leucine was added to the maternal circulation as the placenta in vivo takes up leucine from the maternal circulation and releases leucine into the fetal circulation. 15N-glutamate was added to both maternal and fetal circulations as the placenta takes up glutamate from both the maternal and fetal circulations.
Table 1.
Experimental parameters and uptake of labelled amino acids.
| Protocol | Maternal artery flow 14 ml/min | Fetal artery flow 6 ml/min | Perfused tissue weight (g) | Labelled amino acid uptake at 5 h (nmol/min/g) | n |
|---|---|---|---|---|---|
| 1. Fetal 13C-glutamate | Buffer only | U–13C-glutamate 100 μmol/L (≈physiological) | 27.5 (6) | 13.3 (2.07) | 5 |
| 2. Maternal 15N-leucine |
15N-leucine 146 μmol/L (≈2 × physiological) |
Buffer only | 39.3 (8.3) | 16.7 (2.1) | 5 |
| 3. Fetal 15N-glutamate | Buffer only | 15N-glutamate 200 μmol/L (≈2 × physiological) | 38.8 (10.1) | 14.8 (4.5) | 4 |
| 4. Maternal 15N-glutamate |
15N-glutamate 200 μmol/L (≈2 × physiological) |
Buffer only | 34.0 (8.3) | 17.5 (3.5) | 4 |
Data are mean (SEM).
The U–13C-glutamate (97–99 atoms percent excess), 15N-leucine (98 atoms percent excess) and 15N-glutamate (98 atoms percent excess) were all obtained from Cambridge Isotope Laboratories, MA, USA. Perfusion of 13C-glutamate was performed at near physiological concentrations while the 15N amino acid perfusions were at approximately double physiological concentrations. Perfusion with stable isotope labelled glutamate or leucine was performed for 5 h and samples were collected from the maternal and fetal veins every hour. At the end of this perfusion period the maternal and fetal circulations were washed out with EBB for 15 min and the cotyledon collected for analysis of intracellular amino acids.
2.2. Intracellular amino acids
Frozen cotyledons were pulverised and a sample homogenised in 3 volumes of ice cold EBB. The homogenate was centrifuged and the supernatant stored at −80 °C until analysed by High Pressure Liquid Chromatography (HPLC) and Gas Chromatography Mass Spectrometry (GCMS) as described below.
2.3. Analysis of amino acids by gas chromatography mass spectrometry
Samples were mixed with an equal volume of 1 mol/L acetic acid, applied to a column containing Dowex 50X8 (H+) cation exchanger (Sigma Aldrich Chemical Co., Gillingham, UK) and washed with 10 volumes of distilled water. The amino acids were eluted in 3 mol/L ammonium hydroxide, dried and derivatised at 80 °C for 1 h with a mixture of equal volumes of N-methyl-N-tert-butyldimethylsilyltrifluoroacetamide and acetonitrile. GCMS measurements were carried out with an Agilent 6890/5973 system (Agilent Technologies, Wokingham, UK) on a BPX5 30m × 250 μm column in electron impact mode. Isotopomer abundances were calculated based on the principles described by Wolfe [15].
2.4. Analysis of amino acids by HPLC
Amino acid concentrations were measured by HPLC with fluorescence detection using nor-valine as an internal standard [16]. Perfusate protein was removed by centrifugation following addition of an equal volume of 6% sulphosalicylic acid containing 100 μmol/L nor-valine (Sigma, UK). Immediately following automated pre-column derivatization of the supernatant with o-phthaldialdehyde/3-mercaptopropionic acid at pH 9.2 for 100 s at room temperature a 20 μL sample was injected into the HPLC system (Gilson HPLC, Anachem, Luton, UK). A binary solvent system was used to separate the amino acid derivatives on a Supelcosil C18 (25 × 0.46 cm, 5.0 μm) column (Sigma Aldrich, Gillingham, UK). Solvent A consisted of 100 mmol/L di-sodium hydrogen phosphate adjusted to pH 6.2 with propionic acid, methanol and tetrahydrofuran in ratio 460:40:5; Solvent B consisted of water, methanol, acetonitrile (Fisher Scientific, Loughborough, UK) in ratio 4:3:3. The eluted components were measured by fluorescence (λex = 335 nm, λem = 455 nm). The amino acid:internal standard (nor-valine) peak area ratio was calculated and samples quantified by comparison to the area ratios of known amino standards. The coefficient of variation of the amino acid analysis was 2–5%.
2.5. Analysis of keto acids by HPLC
Concentrations of the keto acids in the maternal and fetal venous outflow and placental tissue homogenates were measured by HPLC using a modified method of Pailla [17]. Briefly, 0.5 ml samples and standards were deproteinated by mixing with an equal volume of 6% w/v sulphosalicylic acid containing 15 μmol/L ketovaleric acid. An aliquot of the supernatant was mixed with o-phenylenediamine in hydrochloric acid, heated at 80 °C, cooled, extracted into ethyl acetate and reduced to dryness under nitrogen. The residue was dissolved in methanol and a 50 μL sample was analysed by HPLC (Beckman System Gold, Beckman–Coulter Ltd., High Wycombe, UK) using a Supelcosil C18 (25 × 0.46 cm, 5.0 μm) column.
The eluted components were measured by fluorescence (λex = 350 nm, λem = 410 nm). The response ratio of each keto acid peak was calculated as the area ratio to the internal standard peak. Concentrations of the individual keto acids were calculated by comparison to the area ratios of known keto acid standards. The limit of detection for the keto acids was 0.08 μmol/L for pyruvate, 0.60 μmol/L for α-ketoisocaproate, 0.03 for α-ketoisovalerate and 0.04 for α-keto-β-methylvalerate.
2.6. Reverse transcriptase PCR
Total RNA was extracted from placental tissue and reverse transcribed into cDNA as described previously [14]. Intron spanning primers to human SLC38A3 and SLC38A5 designed using Primer Express (Applied Bio-systems, California, USA). SNAT3 (SLC38A3; NM_006841) forward 5′-TGCAGACAGAGATGGTGGAG-3′, reverse 5′-GTAGCCCAGCTGCTCATAGG-3′; SNAT5 (SLC38A5; NM_033518.2) forward 5′-CTGAGCTCCCCCTGGTTATC-3′, reverse 5′-CAAAAGCCATAATGGGCACT-3′. PCR was performed under the following conditions: 94 °C for 3 min; 40 cycles at 94 °C for 30 s, 60 °C for 30 s and 72 °C for 30 s; and then 72 °C for 7 min (Mastercycler, Eppendorf AG, Hamburg). PCR products were isolated and sequenced to confirm their identity (GATC Biotech, Konstanz, Germany).
2.7. Western blotting
Microvillous (MVM) and basal membrane (BM) preparations of placental syncytiotrophoblast were isolated as described previously [14,18]. MVM and BM protein samples (45 μg membrane protein, n = 4) were mixed with loading buffer (RunBlue LDS Sample Buffer with dithiothreitol at 10 mg/ml; Expedeon Ltd., Harston, Cambridgeshire, UK) and reduced at 70 °C for 10 min. Samples were separated by electrophoresis in 4–12% SDS-PAGE gels and blotted on PVDF-membranes (Expedeon). The membranes were blocked overnight at 4 °C with 5% Advanced Blocking Reagent (GE Healthcare, Little Chalfont, UK) in phosphate-buffered saline–0.1% Tween-20 (PBS-T, pH 7.4). Blots were incubated with primary antibody against SNAT5 (rabbit, anti-human, ab72717, AbCAM, Cambridge, UK) at a 1:1000 dilution overnight at 4 °C. After washing, blots were incubated for 2 h at room temperature with 1:100,000 goat HRP-anti-rabbit secondary antibody. Immunoreactive signals were visualised using enhanced chemiluminescence (SuperSignal West Femto, Thermo Scientific, UK). A negative control membrane probed only with secondary antibody was also used in all cases. Membranes were stripped and incubated with monoclonal anti-β-actin peroxidise (A3854, Sigma Aldrich, UK) to assess protein loading.
2.8. Glutamate disposal rate calculations
To derive a value for the rate of glutamate disposal the M+5 isotopomer was used for glutamate and glutamine and the M+4 isotopomer was used for aspartate. The uptake of 13C-glutamate by the placenta was estimated from the fetal venous–arterial difference. The transfer of 13C-glutamate from the placenta to the maternal circulation was calculated as the maternal venous–arterial concentration difference multiplied by the rate of perfusion to derive uptake in nmol/min. The rate of accumulation of free M+5 13C-glutamate or M+5 13C-glutamine within the placenta was estimated as the final pool size of the M+5 isotopomer for the relevant 13C-amino acid (nmol/cotyledon) divided by the duration of the experiment in minutes to give accumulation rate of the M+5 isotopomer in nmol/min. Other isotopomers were not included in these calculations. The placental conversion of glutamate to glutamine was considered to be at least equal to, or greater, than the measured release of glutamine into the maternal and fetal circulations plus the accumulation per minute within tissue. Disposal of 13C-glutamate into the Krebs cycle was considered to be at least equal to, or greater, than the measured release and accumulation of M+4 13C-aspartate: although likely to be an underestimate as we did not measure other metabolites or CO2. Disposal into the protein and proline pools was taken to be the balance of glutamate not accounted for by these other routes.
2.9. Analysis of 13C incorporation into lipid pools by LCMS
Samples of placental tissue (100–150 mg) were taken before and after perfusion (n = 4 each) for lipidomic analysis. Briefly, tissue was homogenised in 1.5 ml of 0.9% saline on ice and total lipids extracted using dichloromethane and methanol [19]. After drying under nitrogen gas, the lipid residue was dissolved in 1 ml dichloromethane:methanol:water:concentrated ammonia (30:66:3:1 v:v) and introduced into a Waters Xevo TQS mass spectrometer by direct infusion from a Harvard syringe pump at a flow rate of 8 μl/min. Positive and negative ionisation mass spectra were collected, followed by diagnostic scans for phosphatidylcholine (PC), phosphatidylethanolamine (PE), phosphatidylserine (PS), phosphatidylinositol (PI), cholesterol esters (CE) and diacylglycerol (DAG) (1). Peak intensities determined in MassLynx software were converted to Excel files and the ratio of M0:M+1 peak intensities for selected species calculated.
2.10. Statistics
Enrichment above baseline was determined using a one sample t-test. Specific comparisons between placental tissue, maternal vein and fetal vein were performed by a paired t test. Analysis of amino acid concentrations in tissue under different conditions was determined by 2 way ANOVA. Where differences were indicated by two way ANOVA individual amino acids were analysed by one way ANOVA with Bonferroni correction. Significance was assumed at the 0.05 level. Data are presented as mean and SEM.
3. Results
The weight of the perfused cotyledons in each experimental group is reported in Table 1. The fetal venous recovery rate in placentas used for the studies was 5.9 ± 0.1 ml/min (98 ± 2% of fetal inflow). Labelled amino acid uptake from the donor circulation after 5 h of perfusion in the 4 experimental protocols is reported in Table 1.
3.1. Tissue amino acid concentrations
Concentrations of amino acids were determined in placental tissue perfused for only 15 min to remove maternal and fetal blood (baseline tissue concentration) and in the cotyledons following the 5 h perfusions in the U–13C-glutamate or 15N-leucine experimental groups (Table 2). Analysis by 2 way ANOVA (group vs amino acid) indicated that there were significant differences in the concentrations of individual amino acids between the experimental groups (P < 0.001). When individual amino acids levels in the three groups were compared this indicated that levels of many amino acids in the 15N-leuicne perfused cotyledons were significantly lower than in baseline or 13C-glutamate perfused cotyledons (Table 2).
Table 2.
Tissue amino acids in perfused and non perfused placentas.
| Amino acids | Baseline after 15–20 min perfusion (μmol/kg tissue) n = 6 | 13C-glu fetal after 5 h perfusion (μmol/kg tissue) n = 5 | 15N-leu maternal after 5 h perfusion (μmol/kg tissue) n = 5 |
|---|---|---|---|
| Glutamate | 1025 (976) | 1293 (81)a | 731 (36)a,b |
| Taurine | 719 (69) | 788 (51) | 368 (64)b |
| Glycine | 449 (40) | 366 (15) | 245 (29)a |
| Aspartate | 348 (38) | 496 (97) | 230 (21)b |
| Alanine | 296 (42) | 200 (11) | 134 (8)b |
| Serine | 246 (29) | 232 (14) | 139 (16)b |
| Leucine | 120 (12) | 87 (10) | 80 (9) |
| Lysine | 168 (33) | 52 (27)a | 78 (4) |
| Threonine | 155 (15) | 115 (4) | 64 (5)a,b |
| Arginine | 104 (11) | 108 (7) | 59 (8)a,b |
| Valine | 107 (13) | 73 (3) | 53 (4)a |
| Glutamine | 94 (21) | 140 (14) | 42 (8)b |
| Tyrosine | 41 (4) | 38 (2) | 26 (3) |
| Phenylalanine | 51 (5) | 45 (5) | 24 (2)a,b |
| Isoleucine | 49 (6) | 41 (5) | 19 (2)a |
| Asparagine | 39 (4) | 53 (4) | 21 (2)a,b |
| Methionine | 22 (3) | 19 (2) | 15 (2) |
| Histidine | 37 (7) | 36 (2) | 17 (1)b |
Data is mean (SEM). Overall analysis by 2 way ANOVA found significant effects of experiment (P < 0.001), amino acid (P < 0.001) and interactions between these (P < 0.001). Amino acids were compared individually by one way ANOVA followed by Bonferroni posthoc test.
Indicates a significant difference from initial.
Indicates that the 15N group is significantly different from the 13C group.
3.2. 13C-glutamate experiments
Following infusion of U–13C-glutamate into the fetal circulation of the perfused placenta the recovery of the 13C label (other than as M+5 13C-glutamate) as the M+5 isotopomer was mainly as 13C-glutamine which was preferentially released into the maternal circulation (Fig. 1a). As outlined below recovery of 13C label was also observed in isotopomers of glutamate, aspartate, glutamine and proline (Fig. 2).
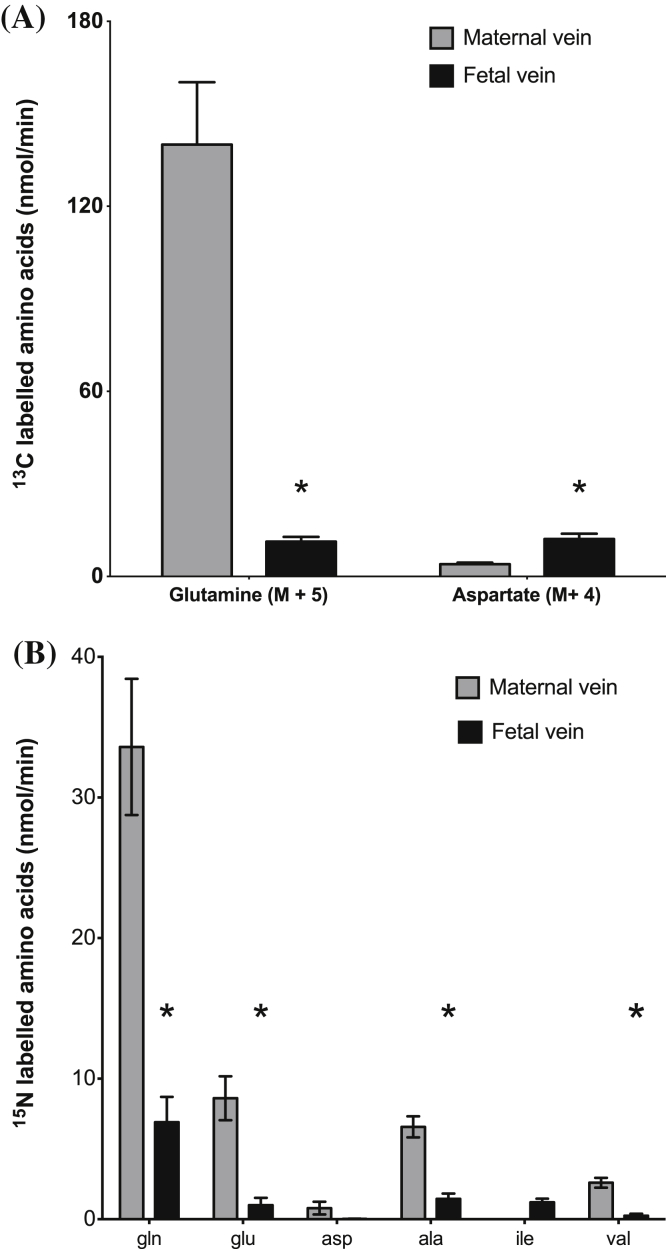
Fig. 1.
Glutamine synthesised within the placenta is primarily released into the maternal circulation. (A) Following perfusion of the fetal circulation with 13C-glutamate, 13C labelled glutamine, aspartate, and proline (not shown) were released from placental tissue into fetal vein and maternal vein. (B) Following perfusion of the maternal circulation with 15N-leucine 15N-labelled glutamine, glutamate, aspartate, alanine, isoleucine, valine and proline (not shown) were released from placental tissue into the fetal vein and maternal vein. Data is mean and SEM. N = 5, *P < 0.05 vs maternal vein.
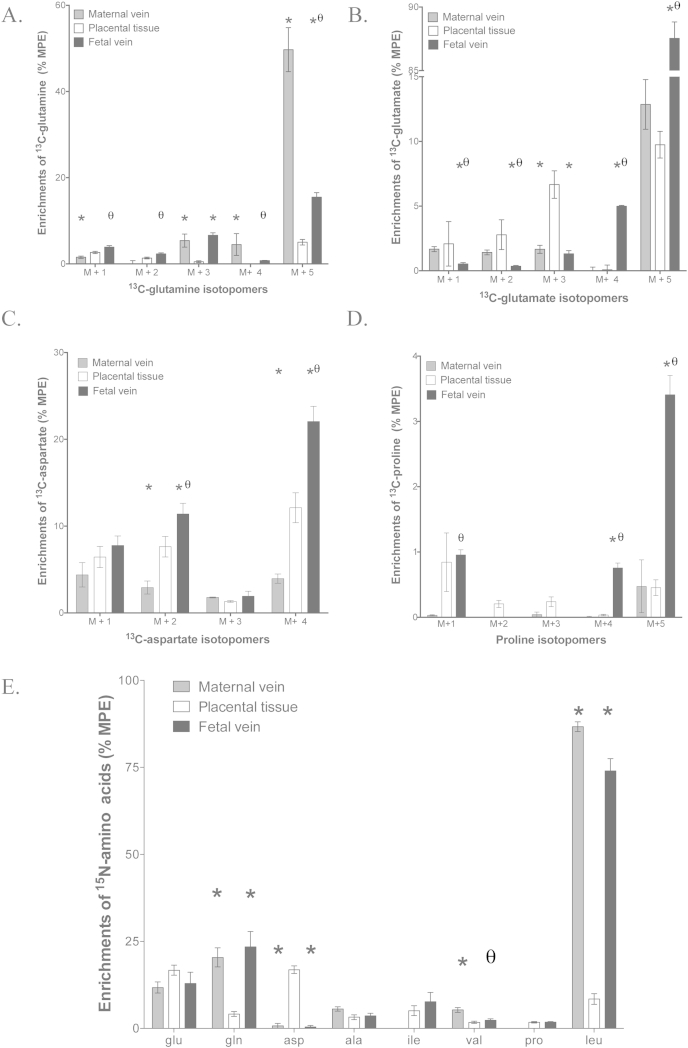
Fig. 2.
Amino acid enrichment and concentrations in the fetal vein and placental tissue following 5 h perfusion of 100 μmol/L 13C-glutamate into the fetal vein of the isolated perfused placenta (A–D) or 15N-leucine into the maternal arterial circulation (E). (A) Isotopomers of 13C-glutamate appearing in placental tissue, fetal vein and maternal vein samples. (B) Isotopomers of 13C-glutamine appearing in placental tissue, fetal vein and maternal vein samples. (C) Isotopomers of 13C-aspartate appearing in placental tissue, fetal vein and maternal vein samples. (D) Isotopomers of 13C-proline appearing in placental tissue, fetal vein and maternal vein samples. (E) Distribution of 15N label following maternal arterial perfusion of 15N-leucine (N = 5). Data is mean and SEM. N = 5, *P < 0.05 vs placental tissue, θP < 0.05 vs maternal vein.
Following infusion of U–13C-glutamate enrichment of the 13C-glutamine M+5, M+4, M+3, M+2 and M+1 isotopomers was observed in placental tissue, maternal vein and fetal vein (Fig. 2a). Enrichment of 13C-glutamine in the maternal vein was significantly greater than in the fetal vein for M+5 and M+4 isotopomers, not different from the fetal vein for M+3 isotopomer and lower than in the fetal vein for M+2 and M+1 isotopomers (Fig. 2a). For the M+5, M+4 and M+3 isotopomers of 13C-glutamine enrichment in the maternal and fetal circulations was higher than in placental tissue, but this was not the case for M+2 and M+1 isotopomers (Fig. 2a).
Following infusion of U–13C-glutamate in addition to the recovery in the placenta of the M+5 glutamate perfused into the placenta, the M+4, M+3, M+2, and M+1 isotopomers of 13C-glutamate were detected in placental tissue, the fetal vein and the maternal vein (Fig. 2b). The release of M+4 13C-glutamate from the placenta into the fetal circulation was significantly greater than into the maternal circulation (P < 0.05, Fig. 1a).
Following infusion of U–13C-glutamate enrichment of the 13C-aspartate isotopomers M+4, M+3, M+2 and M+1 was observed in placental tissue, maternal vein and fetal vein (Fig. 2c). Enrichment of the aspartate isotopomers M+4 and M+2 was significantly greater in the fetal vein compared with placental tissue and significantly greater in placental tissue compared with the maternal vein (P < 0.05, Fig. 2c). There were no significant differences in the enrichment of M+3 and M+1 13C-aspartate amongst the three pools (Fig. 2c). There was significantly more M+4 13C-aspartate released into the fetal vein than the maternal vein (P < 0.05, Fig. 1a).
Following infusion of U–13C-glutamate enrichment of the 13C-proline isotopomers M+5, M+4 was observed in placental tissue, maternal vein and fetal vein (Fig. 2d). Enrichment of M+5 and M+4 proline was significantly greater in the fetal vein than in the placenta or maternal vein (P < 0.05, Fig. 2d). Enrichment of M+3 and M+2 13C-proline was only observed in the placenta. Enrichment of M+1 13C-proline in fetal vein and in placenta was significantly greater than in the maternal vein where there was no detectable enrichment (Fig. 2d). Proline was not detected by HPLC and hence the release of 13C-proline into the maternal or fetal circulations was not determined.
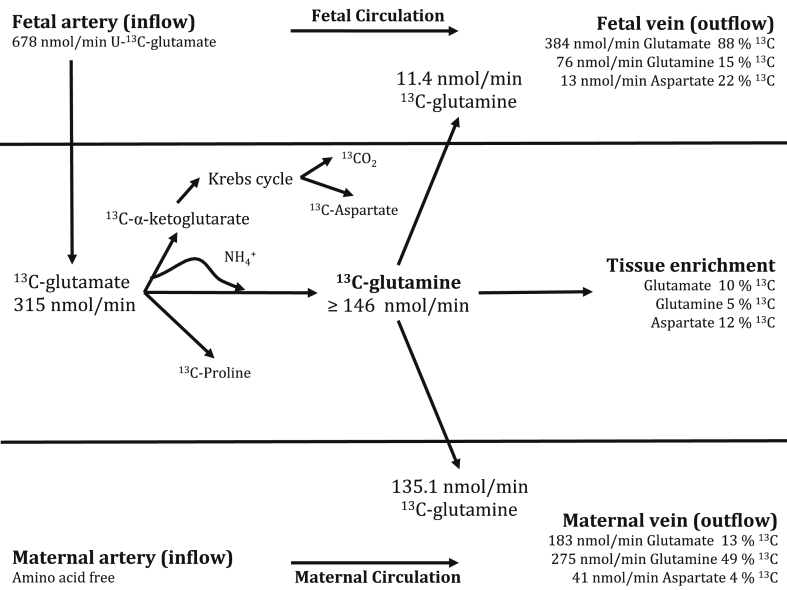
Estimates for disposal of 13C-glutamate into different amino acid pools after 5 h perfusion in a quasi isotopic and metabolic steady state are presented in Fig. 3.
Fig. 3.
13C-Glutamate disposal in the isolated perfused placenta perfused with 678 nmol/min (100 μmol/L) 98% 13C–U-glutamate for 300 min. 13C-Glutamine production was the major route of glutamate disposal. For simplicity these estimates are based on M+5 glutamate and glutamine rather than including all isotopomers.
Following infusion with 13C-glutamate there was no detectable enrichment in alanine, glycine, valine, leucine, isoleucine, asparagine, serine, threonine, lysine, phenylalanine and tyrosine. The method did not allow measurement of enrichment in arginine or cysteine.
3.3. 15N-leucine experiments
Following perfusion of 15N-leucine into the maternal artery, 15N-labelled amino acids were released into fetal vein as glutamate, glutamine, aspartate, alanine, leucine and valine (Fig. 1b). Following perfusion of 15N-leucine into the maternal artery labelled amino acids were recovered from placental tissue and one or both of the placental circulations: glutamate, glutamine, aspartate, alanine, isoleucine, valine and proline. There were significant differences in the levels of enrichment of specific amino acids recovered from the placenta compared to the maternal and fetal veins (Fig. 2e). Following perfusion of 15N-leucine into the maternal artery the greatest recovery of 15N label (other than as 15N-leucine) was as 15N-glutamine in the maternal vein, similar to the results following infusion with 13C-glutamate into the fetal artery (Fig. 1).
3.4. Branched chain keto-acid and pyruvate concentrations in the 15N-leucine experiments
Following perfusion of 15N-leucine into the maternal artery, the concentrations of the keto analogues of the branched chain amino acids as α-keto-isocaproic acid, α-keto-isovaleric acid and α-keto-β-methylvaleric acid were measured in placental tissue and maternal vein, however the concentrations in the fetal vein were near the limit of detection. In placental tissue the concentrations (n = 5, mean (SEM)) were: α-ketoisovalerate, 790 nmol/L (237); α-ketoisocaproate, 149 nmol/L (78) and α-keto-β-methylvalerate, 465 nmol/L (154). From the maternal vein the appearance of branched chain keto acids was: α-ketoisovalerate 49 nmol/min (4), α-ketoisocaproate, 234 nmol/min (36) and α-keto-β-methylvalerate, 34 nmol/min (10). From the fetal vein (n = 5) the values were near the limit of detection: α-ketoisovalerate, 1 nmol/min (1); α-ketoisocaproate, 4 nmol/min (4) and α-keto-β-methylvalerate, 1 nmol/min (1). Release of pyruvate was significantly greater into the maternal vein (2.7 (0.43) μmol/min) than the fetal vein (0.12 (0.12) μmol/min) (P < 0.001).
3.5. 15N-glutamate experiments
Following perfusion of 15N-glutamate into either the fetal artery (protocol 3) or the maternal artery (protocol 4), 15N label was recovered in the fetal vein, maternal vein and placental tissue as 15N-glutamine, 15N-aspartate, 15N-alanine, 15N-proline, 15N-leucine, 15N-valine and 15N-isoleucine (Supplemental data Figs. S1 and S2). Production of 15N amino acids from maternal or fetal 15N-glutamate reached steady state at 3 h and remained essentially constant to 5 h (Supplemental data Fig. S3). This data is not available for the 13C-glutamate experiment.
3.6. Immuno-localisation of SNAT5 in placental syncytiotrophoblast plasma membranes
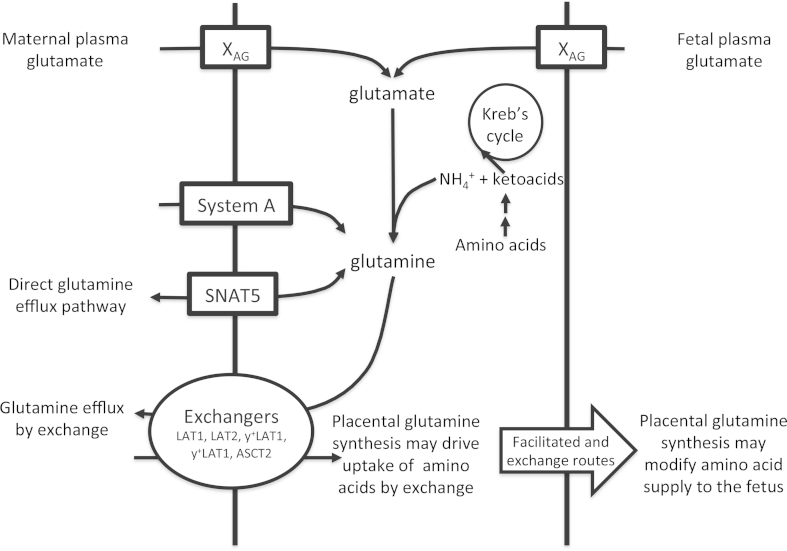
rtPCR analysis demonstrated placental expression of SNAT3 (SLC38A3) and SNAT5 (SLC38A5) mRNA which was confirmed by sequencing of the PCR product (data not shown). SNAT5 was immuno-localised to the maternal facing microvillous membrane but not fetal facing basal membrane of the placental syncytiotrophoblast (Fig. 4).
Fig. 4.
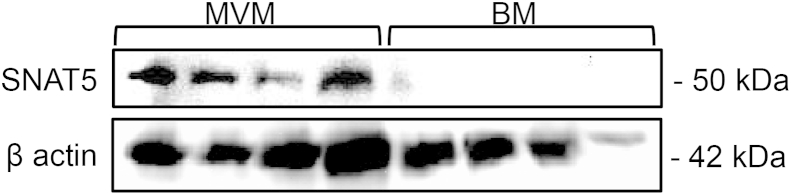
Western blot demonstrating localisation of the SNAT5 (SLC38A5), which can mediate glutamine efflux, to the maternal facing microvillous membrane of placental syncytiotrophoblast. There was no apparent SNAT5 localisation in the fetal facing basal membrane.
3.7. 13C from glutamate was not incorporated into the lipid pool
Lipid incorporation of 13C-acetyl-CoA derived from 13C-glutamate would be expected to generate mass isotopomer distributions with increased relative intensities of M+1 and M+2 ion peaks. Supplementary Fig. 3 shows that for selected lipid species exclusively containing palmitate, the end product of fatty acid synthesis, mass isotopomer distributions were identical before and after perfusion (Fig. 5). More detailed calculation of M0:M+1 peak intensities for all detected species of PC, PE, PS, PI, CE and DAG similarly found no increased relative M+1 ion intensities after 13C-glutamate for any lipid species. These results suggest that under these experimental conditions there was negligible conversion of 13C-glutamate to any of these lipids.
Fig. 5.
Placental glutamine synthesis may drive uptake of amino acids for transport to the fetus. Glutamine is a substrate of 5 exchangers whose activity have been clearly demonstrated in the placenta and is the only substrate transported by all these amino acid exchangers. System N (SNAT5) can mediate both uptake and efflux of glutamine depending on the glutamine and H+ ion gradients. For simplicity only selected transporters are shown on the MVM and BM.
3.8. Estimated whole placental glutamine release into the maternal circulation
After 5 h perfusion in a quasi-isotopic and metabolic steady state, glutamine was released into the maternal circulation at a rate of 140 nmol/min of 49% enriched glutamine from an average 27.5 g cotyledon. Assuming an average placental weight of 470 g, and that in vivo glutamine release occurs at a similar rate as the perfused cotyledon then placental glutamine release into the maternal circulation would equate to 7.0 mmol/day.
4. Discussion
This study demonstrates that the isolated perfused human placenta can synthesise glutamine and that it can do so in biologically relevant amounts. An unexpected finding was that most of this glutamine is transferred to the maternal rather than the fetal circulation. We suggest that high rates of placental glutamine production ensure that there is always sufficient glutamine to meet fetal requirements for growth and metabolism. In addition, placentally derived glutamine may play metabolic and regulatory roles within the placenta.
In the isolated perfused placenta net uptake of glutamate was observed from both the maternal and fetal circulations indicating a large placental metabolic demand. These observations are consistent with the umbilical V–A difference for glutamate [20]. While this glutamate was disposed of into a number of different pools, the major product of 13C-glutamate was 13C-glutamine. Assuming that the ammonia required for glutamine production was derived from the same metabolic pool, then an equivalent amount of 13C-glutamate is likely to have been converted to α-ketoglutarate and ammonia; in which case the glutamate used for the carbon backbone and for the provision of nitrogen for glutamine production would account for around 90% of glutamate uptake by the isolated perfused placental cotyledon. The appearance of 13C-aspartate as well as M+1–4 isotopomers of glutamate and glutamine demonstrates that this α-ketoglutarate is entering the Kreb's cycle where it will produce intermediary metabolites and energy. Incorporation of 13C from glutamate was also observed in the proline pool and may also be entering the placental protein pool, and together these pathways may account for around 10% of glutamate disposal. However, as the largest proportion of the 13C-glutamate is going to glutamine and to α-ketoglutarate the underlying driver for placental uptake and metabolism of glutamate is likely to be to produce one or both of these compounds.
It should be noted that these experiments were undertaken in the absence of physiological plasma concentrations of amino acids. As substrate concentrations may directly and indirectly affect the activity of metabolic pathways we cannot assume that our results will be quantitatively similar to those obtained in vivo.
13C-Glutamine release from the perfused placenta was 7 mmol/day which we estimate to be around 2% of maternal glutamine flux [21] but around 80% of placental delivery of glutamine to the fetus [22,23]. Even if glutamine synthesis in vivo was higher it is not likely that this would impact greatly on maternal metabolism however it is clearly significant for the fetus. If so, the question arises of why this placental to maternal flux occurs? We suggest that placental glutamine production may serve to ensure that there is always glutamine available for transfer to the fetus or that glutamine synthesis serves a different metabolic requirement.
Data from sheep suggest that the fetus derives a significant proportion of its energy from amino acid catabolism [24]. This fetal amino acid consumption may in part reflect the metabolic requirement of rapidly growing cells for glutamine [1,2]. It is possible that a greater proportion of placentally derived glutamine is destined for the fetus in vivo but that the placenta requires a fetal signal to initiate this transport which is missing in the perfused placenta. Whether SNAT5 mediates glutamine uptake or efflux depends on hydrogen ion gradients and in vivo this may change depending on metabolic status [25].
Consumption of amino acids by the placenta and the fetus will produce amino nitrogen, which must be dealt with to prevent the accumulation of ammonia and glutamine transfer may be trafficking excess nitrogen to the mother. While the fetus can synthesise urea, the placenta cannot, and production of glutamine may traffic excess placental nitrogen back to the mother [24]. As much of the glutamate converted to glutamine will come from the fetus it is possible that, in addition to fetal urea production, placental glutamate uptake may remove excess fetal nitrogen.
Production of the intermediary metabolite α-ketoglutarate could provide an energy substrate for the placenta or may meet a specific metabolic requirement. It may be that glutamine synthesis occurs as a result of α-ketoglutarate production in order to transfer the ammonia to the mother. In rapidly growing cells α-ketoglutarate from glutamine is incorporated into lipid [2]. However, no evidence for incorporation of 13C into the lipid pool was observed here and we have no evidence that this is a major pathway in the placenta. While this particular pathway was not apparent in the placenta α-ketoglutarate may meet some other metabolic requirement.
Another possibility is that glutamine produced within the placenta may facilitate uptake of other amino acids by exchange. Glutamine is a substrate for 5 key amino acid exchangers expressed in the human placenta: LAT1, LAT2, y+LAT1, y+LAT2 and ASCT2 [14,26] (Fig. 4). This is particularly interesting as glutamine efflux by exchange has been shown to facilitate uptake of substrates which activate the mTOR pathway [27]. Glutamate levels in maternal and fetal plasma are indicative of metabolic processes and glutamate uptake may provide a link between maternal and fetal metabolic status and placental mTOR activation.
4.1. Tissue amino acids levels
The tissue amino acid levels in the 13C-glutamate group were maintained compared to baseline however in the 15N-leucine experiment amino acid levels were reduced. One explanation for the difference between the 13C-glutamate and 15N-leuicine experiments is that plasma leucine, but not glutamate, stimulates efflux of intracellular amino acids from the syncytiotrophoblast by amino acid exchange and that the leucine taken up by exchange does not accumulate as it diffuses out of the placenta via the basal membrane facilitated transporters [14].
4.2. Metabolic and transporter compartmentalisation within the syncytiotrophoblast
The data indicate that there is compartmentalisation of both transport and metabolism within the placenta and more specifically in placental syncytiotrophoblast. 13C-Glutamine enrichment within the placenta was 5% and if this was the same in all placental cell types then the enrichment of 13C-glutamine released into the maternal and fetal veins could not exceed 5%. As release of glutamine in to the maternal circulation had a 13C enrichment of 49% this indicates that at least one cellular or subcellular compartment within the placenta has a 13C-glutamine enrichment ≥49%. Other cells or compartments must have a 13C-enrichment <5% such that the average of the ≥49% and <5% enriched compartments is 5% 13C-glutamine. Similar evidence of metabolic compartmentalisation within the placenta can be observed in the 15N-leucine and 15N-glutamate experiments where tissue enrichment of specific amino acids is lower than that released into fetal plasma.
The only placental cell type in direct contact with the maternal circulation is the syncytiotrophoblast. For this reason the observation that the enrichment of glutamine secreted into the maternal circulation was significantly higher than into the fetal circulation suggests that it was derived from the syncytiotrophoblast. If this 13C-glutamine were not made within the syncytiotrophoblast then it would need to be transported through the syncytiotrophoblast to reach the maternal circulation without becoming diluted by endogenous 12C-glutamine within the syncytiotrophoblast. It is not clear how enriched 13C-glutmaine from another cell type could pass through the syncytiotrophoblast to reach the maternal circulation while still having a significantly greater enrichment than that reaching the fetal circulation. For this reason we argue that the synthesis of glutamine from glutamate within the placenta is primarily occurring in the syncytiotrophoblast.
Our observation that SNAT5, a transporter capable of mediating glutamine efflux, is localised to the maternal facing MVM is consistent with the suggestion that the syncytiotrophoblast is the most highly enriched compartment within the placenta.
To explain our observations the low enrichment compartments would need to contain 4–9 fold as much amino acid as the highly enriched compartment in order to dilute the 50% enriched 13C-glutamine within the highly enriched compartment to an overall tissue 5% enrichment. Given the relatively small volume of the other placental cellular compartments (primarily endothelial cells and cytotrophoblast) we suggest the syncytiotrophoblast itself must be functionally compartmentalised [28,29]. Accumulation of glycogen in first trimester villi closest to the openings of uterine glands provides an example of compartmentalisation within the villi [30].
Further evidence of compartmentalisation may come from the observation that 13C from glutamate did not appear in the alanine pool. This was despite the fact that the appearance of M+3 aspartate suggests that 13C-pyruvate is being produced and recycled into the Kreb's cycle and that 15N-alanine was produced by transamination of pyruvate in the 15N experiments [31].
Exchange of 15N by aminotransferases observed within the amino acid pool was limited to aspartate, alanine and the branched chain amino acids. Serine and glycine can both be formed by transamination via the alanine-glyoxylate (serine-pyruvate) aminotransferase but consistent with our previous findings there was no evidence for the transfer of 15N to these amino acids [32,33].
The relatively low tissue amino acid enrichments observed at the end of the glutamate and leucine experiments suggests that the free amino acid pool is being diluted by unlabelled amino acids. This may reflect turnover of the protein pool, with protein breakdown releasing unlabelled amino acids into the free amino acid pool and protein synthesis removing labelled and unlabelled amino acids from this pool. The effect of protein turnover on the placental amino acid pool deserves more attention, in particular because it may have the capacity to buffer fetal amino acid supply in the event of reduced maternal delivery.
4.3. Conclusion
The isolated perfused human placenta has a significant capacity for glutamine synthesis and the substantive transfer of glutamine to the mother suggests that placental synthesis may routinely exceed fetal requirements. This may ensure that there is always sufficient glutamine to meet fetal requirements for growth. In addition, placental synthesis may provide glutamine for other roles including mediating the transfer of other amino acids by exchange, regulation of placental function via mTOR and trafficking of nitrogen to the mother. Placental glutamine metabolism provides further evidence that the placenta is not a passive conduit for maternal nutrients and future research needs to address whether altered placental amino acid metabolism is a cause of fetal growth restriction.
Acknowledgements
This work was funded by a grant from the Gerald Kerkut Charitable Trust. MAH is supported by the British Heart Foundation.
Footnotes
This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial-No Derivative Works License, which permits non-commercial use, distribution, and reproduction in any medium, provided the original author and source are credited.
Supplementary data related to this article can be found at http://dx.doi.org/10.1016/j.placenta.2013.10.003.
Appendix A. Supplementary data
The following is the supplementary data related to this article:
References
- 1.Newsholme P., Lima M.M., Procopio J., Pithon-Curi T.C., Doi S.Q., Bazotte R.B. Glutamine and glutamate as vital metabolites. Braz J Med Biol Res. 2003;36(2):153–163. doi: 10.1590/s0100-879x2003000200002. [DOI] [PubMed] [Google Scholar]
- 2.DeBerardinis R.J., Lum J.J., Hatzivassiliou G., Thompson C.B. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 2008;7(1):11–20. doi: 10.1016/j.cmet.2007.10.002. [DOI] [PubMed] [Google Scholar]
- 3.Schneider H., Mohlen K.H., Challier J.C., Dancis J. Transfer of glutamic acid across the human placenta perfused in vitro. Br J Obstet Gynaecol. 1979;86(4):299–306. doi: 10.1111/j.1471-0528.1979.tb11260.x. [DOI] [PubMed] [Google Scholar]
- 4.Geddie G., Moores R., Meschia G., Fennessey P., Wilkening R., Battaglia F.C. Comparison of leucine, serine and glycine transport across the ovine placenta. Placenta. 1996;17(8):619–627. doi: 10.1016/s0143-4004(96)80080-4. [DOI] [PubMed] [Google Scholar]
- 5.Chung M., Teng C., Timmerman M., Meschia G., Battaglia F.C. Production and utilization of amino acids by ovine placenta in vivo. Am J Physiol. 1998;274(1 Pt 1):E13–E22. doi: 10.1152/ajpendo.1998.274.1.E13. [DOI] [PubMed] [Google Scholar]
- 6.Bloxam D.L., Tyler C.F., Young M. Foetal glutamate as a possible precursor of placental glutamine in the guinea pig. Biochem J. 1981;198(2):397–401. doi: 10.1042/bj1980397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Self J.T., Spencer T.E., Johnson G.A., Hu J., Bazer F.W., Wu G. Glutamine synthesis in the developing porcine placenta. Biol Reprod. 2004;70(5):1444–1451. doi: 10.1095/biolreprod.103.025486. [DOI] [PubMed] [Google Scholar]
- 8.Jozwik M., Teng C., Meschia G., Battaglia F.C. Contribution of branched-chain amino acids to uteroplacental ammonia production in sheep. Biol Reprod. 1999;61(3):792–796. doi: 10.1095/biolreprod61.3.792. [DOI] [PubMed] [Google Scholar]
- 9.Lewis R., Godfrey K., Jackson A., Cameron I., Hanson M. Low serine hydroxymethyltransferase activity in the human placenta has important implications for fetal glycine supply. J Clin Endocrinol Metab. 2005;90(3):1594–1598. doi: 10.1210/jc.2004-0317. [DOI] [PubMed] [Google Scholar]
- 10.Eagle H. Nutrition needs of mammalian cells in tissue culture. Science. 1955;122(3168):501–514. doi: 10.1126/science.122.3168.501. [DOI] [PubMed] [Google Scholar]
- 11.Parimi P.S., Kalhan S.C. Glutamine supplementation in the newborn infant. Semin Fetal Neonatal Med. 2007;12(1):19–25. doi: 10.1016/j.siny.2006.10.003. [DOI] [PubMed] [Google Scholar]
- 12.Schneider H., Panigel M., Dancis J. Transfer across the perfused human placenta of antipyrine, sodium and leucine. Am J Obstet Gynecol. 1972;114(6):822–828. doi: 10.1016/0002-9378(72)90909-x. [DOI] [PubMed] [Google Scholar]
- 13.Cleal J.K., Brownbill P., Godfrey K.M., Jackson J.M., Jackson A.A., Sibley C.P. Modification of fetal plasma amino acid composition by placental amino acid exchangers in vitro. J Physiol. 2007;582(2):871–882. doi: 10.1113/jphysiol.2007.130690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cleal J.K., Glazier J.D., Ntani G., Crozier S.R., Day P.E., Harvey N.C. Facilitated transporters mediate net efflux of amino acids to the fetus across the basal membrane of the placental syncytiotrophoblast. J Physiol. 2011;589(4):987–997. doi: 10.1113/jphysiol.2010.198549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wolfe R.R. Wiley-Liss; New York: 1992. Radioactive and stable isotope tracers in biomedicine: principles and practice of kinetic analysis. [Google Scholar]
- 16.Godel H., Graser T., Foldi P., Pfaender P., Furst P. Measurement of free amino acids in human biological fluids by high-performance liquid chromatography. J Chromatogr. 1984;297:49–61. doi: 10.1016/s0021-9673(01)89028-2. [DOI] [PubMed] [Google Scholar]
- 17.Pailla K., Blonde-Cynober F., Aussel C., De Bandt J.P., Cynober L. Branched-chain keto-acids and pyruvate in blood: measurement by HPLC with fluorimetric detection and changes in older subjects. Clin Chem. 2000;46(6 Pt 1):848–853. [PubMed] [Google Scholar]
- 18.Lewis R.M., Glazier J., Greenwood S.L., Bennett E.J., Godfrey K.M., Jackson A.A. L-serine uptake by human placental microvillous membrane vesicles. Placenta. 2007;28(5–6):445–452. doi: 10.1016/j.placenta.2006.06.014. [DOI] [PubMed] [Google Scholar]
- 19.Pulfer M., Murphy R.C. Electrospray mass spectrometry of phospholipids. Mass Spectrom Rev. 2003;22(5):332–364. doi: 10.1002/mas.10061. [DOI] [PubMed] [Google Scholar]
- 20.Cetin I., de Santis M.S., Taricco E., Radaelli T., Teng C., Ronzoni S. Maternal and fetal amino acid concentrations in normal pregnancies and in pregnancies with gestational diabetes mellitus. Am J Obstet Gynecol. 2005;192(2):610–617. doi: 10.1016/j.ajog.2004.08.011. [DOI] [PubMed] [Google Scholar]
- 21.Kurpad A.V., Dwarkanath P., Thomas T., Mhaskar A., Thomas A., Mhaskar R. Comparison of leucine and dispensable amino acid kinetics between Indian women with low or normal body mass indexes during pregnancy. Am J Clin Nutr. 2010;92(2):320–329. doi: 10.3945/ajcn.2010.29205. [DOI] [PubMed] [Google Scholar]
- 22.Cetin I., Marconi A.M., Bozzetti P., Sereni L.P., Corbetta C., Pardi G. Umbilical amino acid concentrations in appropriate and small for gestational age infants: a biochemical difference present in utero. Am J Obstet Gynecol. 1988;158(1):120–126. doi: 10.1016/0002-9378(88)90792-2. [DOI] [PubMed] [Google Scholar]
- 23.Gerson A.G., Wallace D.M., Stiller R.J., Paul D., Weiner S., Bolognese R.J. Doppler evaluation of umbilical venous and arterial blood flow in the second and third trimesters of normal pregnancy. Obstet Gynecol. 1987;70(4):622–626. [PubMed] [Google Scholar]
- 24.Gresham E.L., James E.J., Raye J.R., Battaglia F.C., Makowski E.L., Meschia G. Production and excretion of urea by the fetal lamb. Pediatrics. 1972;50(3):372–379. [PubMed] [Google Scholar]
- 25.Mackenzie B., Erickson J.D. Sodium-coupled neutral amino acid (system N/A) transporters of the SLC38 gene family. Pflugers Arch. 2004;447(5):784–795. doi: 10.1007/s00424-003-1117-9. [DOI] [PubMed] [Google Scholar]
- 26.Cleal J.K., Lewis R.M. The mechanisms and regulation of placental amino acid transport to the human foetus. J Neuroendocrinol. 2008;20(4):419–426. doi: 10.1111/j.1365-2826.2008.01662.x. [DOI] [PubMed] [Google Scholar]
- 27.Nicklin P., Bergman P., Zhang B., Triantafellow E., Wang H., Nyfeler B. Bidirectional transport of amino acids regulates mTOR and autophagy. Cell. 2009;136(3):521–534. doi: 10.1016/j.cell.2008.11.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Teasdale F. Histomorphometry of the human placenta in pre-eclampsia associated with severe intrauterine growth retardation. Placenta. 1987;8(2):119–128. doi: 10.1016/0143-4004(87)90015-4. [DOI] [PubMed] [Google Scholar]
- 29.Mayhew T.M., Wadrop E., Simpson R.A. Proliferative versus hypertrophic growth in tissue subcompartments of human placental villi during gestation. J Anat. 1994;184(Pt 3):535–543. [PMC free article] [PubMed] [Google Scholar]
- 30.Burton G.J., Scioscia M., Rademacher T.W. Endometrial secretions: creating a stimulatory microenvironment within the human early placenta and implications for the aetiopathogenesis of preeclampsia. J Reprod Immunol. 2011;89(2):118–125. doi: 10.1016/j.jri.2011.02.005. [DOI] [PubMed] [Google Scholar]
- 31.Olstad E., Olsen G.M., Qu H., Sonnewald U. Pyruvate recycling in cultured neurons from cerebellum. J Neurosci Res. 2007;85(15):3318–3325. doi: 10.1002/jnr.21208. [DOI] [PubMed] [Google Scholar]
- 32.Bennett F.I., Jackson A.A. Glycine is not formed through the amino transferase reaction in human or rat placenta. Placenta. 1998;19(4):329–331. doi: 10.1016/s0143-4004(98)90066-2. [DOI] [PubMed] [Google Scholar]
- 33.Hayasaka K., Tada K., Fueki N., Takahashi I., Igarashi A., Takabayashi T. Feasibility of prenatal diagnosis of nonketotic hyperglycinemia: existence of the glycine cleavage system in placenta. J Pediatr. 1987;110(1):124–126. doi: 10.1016/s0022-3476(87)80305-0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.