Abstract
Bacterial pericarditis is a rare disease in the era of antibiotics. Purulent pericarditis is most often caused by Staphylococcus aureus, Streptococcus pneumoniae, or Haemophilus influenzae. The number of H. parainfluenzae infections has been increasing; in rare cases, it has caused endocarditis. We report a case of purulent pericarditis caused by H. parainfluenzae in a 62-year-old woman who reported a recent upper respiratory tract infection. The patient presented with signs and symptoms of pericardial tamponade. Urgent pericardiocentesis restored her hemodynamic stability. However, within 24 hours, fluid reaccumulation led to recurrent pericardial tamponade and necessitated the creation of a pericardial window. Cultures of the first pericardial fluid grew H. parainfluenzae. Levofloxacin therapy was started, and the patient recovered.
Haemophilus parainfluenzae should be considered in a patient who has signs and symptoms of purulent pericarditis. Prompt diagnosis, treatment, and antibiotic therapy are necessary for the patient's survival. To our knowledge, this is the first report of purulent pericarditis caused by H. parainfluenzae.
Key words: Endocarditis, bacterial/diagnosis/microbiology/pathology; haemophilus/isolation & purification; haemophilus infections/diagnosis/drug therapy; haemophilus parainfluenzae; pericarditis/complications/diagnosis/etiology/microbiology/therapy; suppuration/diagnosis; treatment outcome
Purulent pericarditis is a disease process that is usually described as a secondary infection from a primary site in the respiratory tract. The condition has been associated with respiratory disease processes such as pneumonia or empyema, but it can be a sequela of endocarditis, chest trauma, chest surgery, or the hematogenous spread of infection from elsewhere in the body.1 Haemophilus influenzae has been suspected as a cause of purulent pericarditis; however, H. parainfluenzae has not previously been reported as a cause. Haemophilus parainfluenzae organisms are considered to be normal respiratory flora with low pathogenicity. However, H. parainfluenzae is being more frequently implicated in a variety of infections.2,3 We present what we think is the first report of purulent pericarditis caused by H. parainfluenzae.
Case Report
In December 2011, a 62-year-old woman was transferred from another hospital to our institution with pleuritic chest pain, worsening dyspnea, and a pericardial effusion. Her medical history was notable for hypertension, hyperlipidemia, hypothyroidism, and gastroesophageal reflux disease. She had no history of alcohol abuse or tobacco use. She had been in apparently good health until approximately 2 weeks before admission, when she developed a sore throat, headaches, and bilateral ear pain. She had been prescribed azithromycin by her primary care physician, and her symptoms had improved after a few days. Two days before admission to the other hospital, she had begun experiencing pleuritic chest pain that radiated to both shoulders, along with dyspnea on exertion; however, she reported no fever, sore throat, or cough at the time of admission.
Evaluation at the Referring Hospital
Upon presentation at the referring hospital, the patient was in mild distress. Her vital signs were as follows: blood pressure, 136/74 mmHg; respiratory rate, 20 breaths/min; heart rate, 102 beats/min; pulse oximetry, 87% on room air; and temperature, 98.6 °F (37 °C). Physical examination revealed lungs clear to auscultation, sinus tachycardia, no jugular venous distention, and no murmurs or rubs on cardiac auscultation.
The patient's peripheral white blood cell count was 14.3 k/μL with 85.5% polymorphonuclear neutrophils, no bands, 4% lymphocytes, 10% monocytes, and 0.1% eosinophils. Chest radiographs showed a moderate-to-large left pleural effusion with cardiomegaly but without consolidations (Fig. 1). Chest computed tomograms with intravenous contrast enhancement revealed a moderate-to-large pericardial effusion (Fig. 2) and atelectatic changes at the left lung base. No pulmonary embolism was seen. Deep vein thrombosis was not clinically evident. An electrocardiogram showed T-wave inversions in leads I and aVR with no evidence of electrical alternans (Fig. 3). The patient was transferred to our hospital.
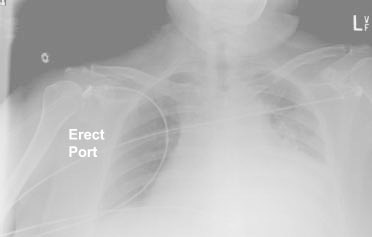
Fig. 1 Chest radiograph shows a moderate-to-large left pleural effusion with cardiomegaly, without consolidations.
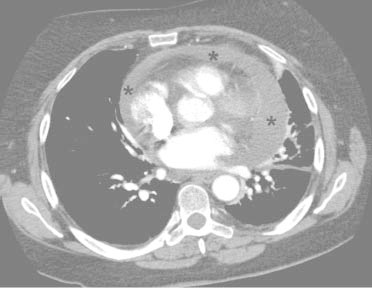
Fig. 2 Contrast-enhanced computed tomogram reveals a moderate-to-large pericardial effusion (asterisks).
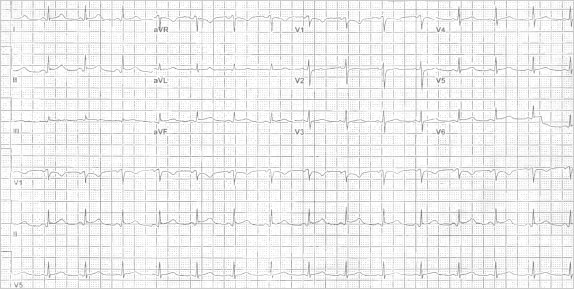
Fig. 3 Electrocardiogram shows sinus tachycardia at 102 beats/min, normal axis, and T-wave inversions in leads I and aVR with no evident electrical alternans.
Evaluation and Treatment after Referral
When the patient arrived at our hospital, an urgent echocardiogram revealed a large, circumferential pericardial effusion with marked respiratory variation of mitral and tricuspid inflow velocities, plethora of the inferior vena cava, and diastolic compression of the right ventricle, consistent with cardiac tamponade (Fig. 4). Hemodynamic evaluation yielded an initial pulsus paradoxus of approximately 50 mmHg on invasive aortic pressure monitoring, a mean right atrial pressure of 18 mmHg, and a mean pericardial pressure of 18 mmHg. Urgent pericardiocentesis yielded 650 mL of serosanguineous fluid, which was taken to our laboratory in blood-culture bottles. The patient's symptoms and hemodynamic status improved markedly after pericardiocentesis. However, 24 hours after admission, she reported recurrent dyspnea, and echocardiograms showed the reaccumulation of a large pericardial effusion with signs of tamponade. The patient underwent the surgical creation of a pericardial window, from which 500 mL of purulent bloody fluid was removed. Numerous loculations were noted.
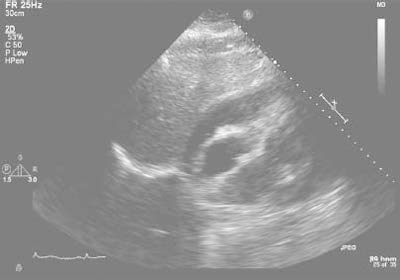
Fig. 4 Echocardiogram reveals a large circumferential pericardial effusion with marked respiratory variation of mitral and tricuspid inflow velocities, plethora of the inferior vena cava, and diastolic compression of the right ventricle, consistent with cardiac tamponade.
A blood culture taken upon admission revealed no organisms. No pathogen grew in the fluid from the pericardial window. However, gram-staining of the initial pericardiocentesis fluid revealed gram-negative coccobacilli, and a culture of that fluid yielded β-lactamase–negative H. parainfluenzae after 6 days. Pathologic analysis of the patient's pericardial tissue yielded granulation tissue and no malignancy. On hospital day 3, the patient was started on intravenous vancomycin and levofloxacin, but levofloxacin alone was continued on hospital day 6 when the culture results became known. Her respiratory status improved on hospital day 3, and serial echocardiograms on days 3 and 6 showed no reaccumulation of pericardial fluid.
The patient was discharged from the hospital on day 8 with instructions to complete a 28-day course of oral levofloxacin therapy. Upon outpatient examination 30 days after discharge, she was doing well clinically. An echocardiogram 60 days after discharge showed a normal left ventricular ejection fraction, no constrictive pericarditis, and no reaccumulation of pericardial effusion.
Discussion
We searched for reported cases of H. parainfluenzae pericarditis and reviewed the MEDLINE database records from 1946 through 2012. The key words used in the search were pericarditis, purulent pericarditis, cardiac tamponade, and H. parainfluenzae. We found no other cases of purulent pericarditis caused by H. parainfluenzae.
Haemophilus parainfluenzae is being identified frequently in a range of infections.4 A member of the Pasteurellaceae family, H. parainfluenzae is a gram-negative coccobacillus that is frequently found as normal flora in the oropharynx of human hosts. Its growth requirement of exogenous nicotinamide adenine dinucleotide (NAD) enables its distinction from H. influenzae, which requires hemin (factor X) in addition to NAD. Either organism is difficult to culture in the laboratory, and identification at the species level can take a week or longer.5 Haemophilus parainfluenzae is more readily cultured when aspirated fluid is transported in blood-culture bottles.2 When a culture is negative, broad-range polymerase chain reaction on tissue specimens can be used to identify the organism within hours.6,7 Haemophilus parainfluenzae is very unlikely to be a contaminant, a conclusion reinforced by a lack of growth of other organisms.8
Purulent pericarditis, first described by Galen almost 2,000 years ago,9 has been reported less often since antibiotics came into widespread use; however, it is still an important sequela of infectious diseases.10 Early diagnosis is essential for complete recovery, and delayed diagnosis carries a high mortality rate.11–13 Because the presenting symptoms are often insidious, diagnosis can be difficult, and a high degree of clinical suspicion is necessary.11,14
Purulent pericarditis is associated with many infectious organisms and portals of entry. The usual pathogens are Staphylococcus aureus (in 36% of cases reported during a 60-year period), Streptococcus pneumoniae (21%), and H. influenzae (12%).14 Before the antibiotic era, most patients with purulent pericarditis reportedly had an underlying infectious disease; currently, most typically present with noninfectious predisposing conditions such as immunosuppression, alcohol abuse, rheumatoid arthritis, and malignancy.11,14–16
Tuberculous pericarditis is the most frequent cause of pericarditis in Africa and in countries where tuberculosis is a major public-health problem, and its prevalence there is increasing.17 In contrast, tuberculous pericarditis constitutes only 4% of pericarditis cases in developed countries. Pericardial involvement usually develops by way of retrograde lymphatic spread or hematogenous spread from primary tuberculous infection. The clinical presentation of tuberculous pericarditis varies, and the symptoms are usually nonspecific. A major complication is effusive constrictive pericarditis. The definitive diagnosis of tuberculous pericarditis is centered around the presence of tubercle bacilli in pericardial fluid.17,18 Our patient was not immunocompromised, and we had a low clinical suspicion of tuberculosis. The pericardial fluid acid-fast smear was negative, and acid-fast bacilli were not isolated in the pericardial fluid after 6 weeks of growth.
The portal of entry for our patient's H. parainfluenzae is not definitively known. The chief portal for infection into the pericardial space is thought to be direct extension from a primary lung source. Other modes of infection include local extension from the myocardium itself, perforating injury to the chest wall, and hematogenous spread.15 In a review of H. parainfluenzae endocarditis cases in France,16 recent dental work was thought to be the primary portal of entry in 17.5% of previously reported cases (7 of 40 patients), and upper respiratory tract infection in 2.5% (1/40). Our patient had not recently undergone a dental procedure, and she displayed no clinical indication of endocarditis, meningitis, genitourinary tract infection, or biliary tract infection.19 However, she had reported a recent upper respiratory infection, and initial chest radiographs revealed a left pleural effusion without associated consolidation. These findings in her case perhaps indicate a pulmonary process with direct extension into the pericardial space.
With or without purulent pericardial effusion, the clinical presentation of purulent pericarditis varies. In a review of 68 cases,1 the most sensitive clinical marker was fever, in 88% of the patients. Dyspnea was present in 61% of the patients, and 57% reported pleuritic or nonpleuritic chest pain. Late complications of purulent pericarditis include constrictive pericardial disease from thickening and scarring of the pericardium or pericardial empyema.1
Patients with purulent pericarditis have poor clinical outcomes: despite treatment, up to 40% of patients die of tamponade, constriction, or toxicity.20 Our patient presented with subjective dyspnea and pleuritic chest pain with sinus tachycardia; however, she was notably afebrile. Despite the purulent pericardial effusion with associated pericardial tamponade, prompt evaluation and percutaneous drainage resulted in her rapid return to normal hemodynamic status. Clinical evaluation in the coronary care unit enabled us to identify the rapid reaccumulation of the effusion and recurrent cardiac tamponade. Definitive surgical drainage and prompt antimicrobial therapy helped to prevent further recurrence. The fluid that was drained from the pericardial window grew no organism for 2 possible reasons: the fastidious nature of H. parainfluenzae, and because the fluid samples were not transported to the microbiology laboratory in blood-culture bottles.
Transthoracic echocardiography facilitates the detection and quantification of pericardial fluid, and it enables a visual evaluation of chamber collapse and the appropriateness of percutaneous versus surgical pericardial drainage.
Because of its fastidious nature, H. parainfluenzae can cause purulent pericarditis and yet be difficult to isolate in culture.8,21 Haemophilus parainfluenzae should be considered in a patient who has signs and symptoms of purulent pericarditis, with or without associated pericardial tamponade. Tamponade necessitates emergent treatment. Definitive diagnosis depends upon a high degree of suspicion and careful microbiologic analysis, and, in certain cases, confirmation by means of polymerase chain reaction. Targeted antibiotic therapy can be curative. In our patient, the prompt diagnosis and identification of this novel infectious cause of purulent pericarditis, as well as the associated pericardial tamponade, resulted in a favorable outcome.
Footnotes
Address for reprints: Eron Sturm, MD, MS 108, Division of Cardiovascular Diseases, Department of Internal Medicine, Drexel University College of Medicine, 230 N. Broad St., 7th fl., Philadelphia, PA 19102.
E-mail: eron.sturm@gmail.com
References
- 1.Hall IP. Purulent pericarditis. Postgrad Med J 1989;65(765):444–8. [DOI] [PMC free article] [PubMed]
- 2.Mora A, Marimon I, Mesquida J, Perez A. Haemophilus parainfluenzae septic arthritis: report of a case and review of the literature. Enferm Infecc Microbiol Clin 2011;29(6):472–3. [DOI] [PubMed]
- 3.Pillai A, Mitchell JL, Hill SL, Stockley RA. A case of Haemophilus parainfluenzae pneumonia. Thorax 2000;55 (7):623–4. [DOI] [PMC free article] [PubMed]
- 4.Fink DL, St Geme JW 3rd. The genus Haemophilus. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E, editors. The prokaryotes: a handbook on the biology of bacteria. 3rd ed. New York: Springer; 2006: p. 1034–61.
- 5.Hamed KA, Dormitzer PR, Su CK, Relman DA. Haemophilus parainfluenzae endocarditis: application of a molecular approach for identification of pathogenic bacterial species. Clin Infect Dis 1994;19(4):677–83. [DOI] [PubMed]
- 6.Kelesidis T, Kelesidis I, Lewinski MA, Humphries R. Establishing diagnosis of Haemophilus parainfluenzae as etiology of culture-negative endocarditis using DNA sequence analysis on tissue specimen. Am J Med 2011;124(7):e9–e10. [DOI] [PMC free article] [PubMed]
- 7.Marin M, Munoz P, Sanchez M, del Rosal M, Alcala L, Rodriguez-Creixems M, et al. Molecular diagnosis of infective endocarditis by real-time broad-range polymerase chain reaction (PCR) and sequencing directly from heart valve tissue. Medicine (Baltimore) 2007;86(4):195–202. [DOI] [PubMed]
- 8.Auten GM, Levy CS, Smith MA. Haemophilus parainfluenzae as a rare cause of epidural abscess: case report and review. Rev Infect Dis 1991;13(4):609–12. [DOI] [PubMed]
- 9.Galen C. De anatomicus administrationibus (Singer C, transl). New York: Oxford University Press; 1956. p. 192–3.
- 10.Rubin RH, Moellering RC Jr. Clinical, microbiologic and therapeutic aspects of purulent pericarditis. Am J Med 1975; 59(1):68–78. [DOI] [PubMed]
- 11.Sagrista-Sauleda J, Barrabes JA, Permanyer-Miralda G, Soler-Soler J. Purulent pericarditis: review of a 20-year experience in a general hospital. J Am Coll Cardiol 1993;22(6):1661–5. [DOI] [PubMed]
- 12.Boyle JD, Pearce ML, Guze LB. Purulent pericarditis: review of literature and report of eleven cases. Medicine (Baltimore) 1961;40(2):119–44.
- 13.Gould K, Barnett JA, Sanford JP. Purulent pericarditis in the antibiotic era. Arch Intern Med 1974;134(5):923–7. [PubMed]
- 14.Parikh SV, Memon N, Echols M, Shah J, McGuire DK, Keeley EC. Purulent pericarditis: report of 2 cases and review of the literature. Medicine (Baltimore) 2009;88(1):52–65. [DOI] [PubMed]
- 15.Klacsmann PG, Bulkley BH, Hutchins GM. The changed spectrum of purulent pericarditis: an 86 year autopsy experience in 200 patients. Am J Med 1977;63(5):666–73. [DOI] [PubMed]
- 16.Darras-Joly C, Lortholary O, Mainardi JL, Etienne J, Guillevin L, Acar J. Haemophilus endocarditis: report of 42 cases in adults and review. Haemophilus Endocarditis Study Group. Clin Infect Dis 1997;24(6):1087–94. [DOI] [PubMed]
- 17.Mayosi BM, Burgess LJ, Doubell AF. Tuberculous pericarditis. Circulation 2005;112(23):3608–16. [DOI] [PubMed]
- 18.Pankuweit S, Ristic AD, Seferovic PM, Maisch B. Bacterial pericarditis: diagnosis and management. Am J Cardiovasc Drugs 2005;5(2):103–12. [DOI] [PubMed]
- 19.Cardines R, Giufre M, Ciofi degli Atti ML, Accogli M, Mastrantonio P, Cerquetti M. Haemophilus parainfluenzae meningitis in an adult associated with acute otitis media. New Microbiol 2009;32(2):213–5. [PubMed]
- 20.Maisch B, Ristic AD. Practical aspects of the management of pericardial disease. Heart 2003;89(9):1096–103. [DOI] [PMC free article] [PubMed]
- 21.Jemsek JG, Greenberg SB, Gentry LO, Welton DE, Mattox KL. Haemophilus parainfluenzae endocarditis. Two cases and review of the literature in the past decade. Am J Med 1979;66(1):51–7. [DOI] [PubMed]


