Abstract
To explore the effects of adipose tissue-derived stem cells (ADSCs) on the proliferation and invasion of pancreatic cancer cells in vitro and the possible mechanism involved, ADSCs were cocultured with pancreatic cancer cells, and a cell counting kit (CCK-8) was used to detect the proliferation of pancreatic cancer cells. ELISA was used to determine the concentration of stromal cell-derived factor-1 (SDF-1) in the supernatants. RT-PCR was performed to detect the expression of the chemokine receptor CXCR4 in pancreatic cancer cells and ADSCs. An in vitro invasion assay was used to measure invasion of pancreatic cancer cells. SDF-1 was detected in the supernatants of ADSCs, but not in pancreatic cancer cells. Higher CXCR4 mRNA levels were detected in the pancreatic cancer cell lines compared with ADSCs (109.3±10.7 and 97.6±7.6 vs 18.3±1.7, respectively; P<0.01). In addition, conditioned medium from ADSCs promoted the proliferation and invasion of pancreatic cancer cells, and AMD3100, a CXCR4 antagonist, significantly downregulated these growth-promoting effects. We conclude that ADSCs can promote the proliferation and invasion of pancreatic cancer cells, which may involve the SDF-1/CXCR4 axis.
Keywords: Adipose tissue-derived stem cells, Pancreatic cancer, Proliferation, Invasion, SDF-1
Introduction
It has been increasingly recognized that cancer cells actively recruit stromal cells into tumors and that this recruitment is essential for the generation of a microenvironment that promotes tumor growth (1). In recent years, it has been suggested that stromal cells are not only associated with pancreatic cancer, but also play an active role in cancer progression. Pancreatic stroma cells can stimulate tumor growth and invasiveness (2,3), induce chemotherapy resistance, and inhibit apoptosis of cancer cells (4,5). The pancreas has abundant stroma surrounding it, and tumor-stroma interactions play a critical role in tumor progression (6). Although the exact mechanisms of tumor-stroma interactions are unclear, some chemokines, including hepatocyte growth factor, epidermal growth factor, vascular endothelial growth factor, and stromal cell-derived factor-1 (SDF-1), have been shown to be involved in the interaction (7,8).
SDF-1 belongs to the CXC chemokine family and is a ligand for CXCR4. SDF-1 is expressed in stromal cells (9), and CXCR4 expression has been found in many cancer types, including breast, ovary, kidney, prostate, and stomach cancer tissue as well as cell lines (10-12). In addition, it has been demonstrated that the SDF-1/CXCR4 receptor-ligand system plays an important role in pancreatic cancer progression by promoting tumor cell migration and angiogenesis (13,14).
The interactions between mesenchymal stem cells from bone marrow and malignant tumors from breast, prostate, ovaries, and stomach have been described (10,15). Compared to bone marrow-derived stem cells, adipose tissue-derived stem cells (ADSCs) are locally adjacent to the tumor tissues and may interact with tumor cells directly. However, the ability of ADSCs to promote tumor cell proliferation remains controversial. It has been reported that ADSCs can promote breast, prostate, non-small lung, and glioblastoma cancer cell growth in vivo (16-18). In contrast, several reports have shown an anti-tumor effect of mesenchymal stem cells. Khakoo et al. (19) used systemic injection of mesenchymal stem cells to inhibit the growth of a subcutaneous Kaposi sarcoma xenotransplant. In addition, the coimplantation of breast cancer cells with mesenchymal stem cells results in tumor growth inhibition and a reduction of metastasis in vivo (20). However, the impact of ADSCs, which are a type of local mesenchymal stem cell, on proliferation and invasion of pancreatic cancer has not been reported to date. Therefore, in this study, we determined whether ADSCs can promote the growth and metastasis of pancreatic cancer cells and sought to determine the mechanism responsible for these observed effects.
Material and Methods
Cell lines and reagents
Human pancreatic cancer (PaCa) cell lines SW1990 and PANC-1 were purchased from the Chinese Academy of Sciences (Shanghai, China). Dulbecco's modified Eagle's medium (DMEM) and fetal bovine serum (FBS) were provided by Gibco (USA), recombinant human CXCL12 (SDF-1) was purchased from R&D Systems (USA), and AMD3100 and Matrigel were obtained from Sigma-Aldrich (USA).
Isolation of human ADSCs
Adipose tissue was obtained from healthy people undergoing bariatric surgery who had provided informed written consent. The protocol of all procedures was approved by the institutional review board of the First Affiliated Hospital of Wenzhou Medical College. Fresh visceral fat tissue was washed with phosphate-buffered saline (PBS) and immediately processed. The tissue was minced into pieces less than 2 mm in diameter and then digested with collagenase type IV at 37°C for 1 h. The digested tissue was then centrifuged at 400 g for 10 min. The supernatant containing adipocytes and debris was discarded, and the pelleted cells were washed with Hanks balanced salt solution. Finally, cells were resuspended and grown in 1000 mg/L low-glucose DMEM (L-DMEM) containing 20% FBS, 100 µg/L penicillin, and 100 µg/L streptomycin in a humidified environment with 5% CO2 at 37°C. After 24 h, red blood cells and unattached cells were washed and removed. The cells were then grown in a humidified incubator at 37°C. After the cells were cultured for three to five passages, cell surface markers, such as CD45, CD73, and CD90, were analyzed for characterization of the ADSCs.
Differentiation of human ADSCs into adipocytes
ADSCs were cultured to confluence on 35-mm dishes containing DMEM. The medium was then removed and fresh DMEM was added containing 0.5 mM IBMX, 1.0 µM dexamethasone, and 300 nM insulin. The cells were cultured in this differentiation medium for 2 days, and then the medium was changed every 2 days with DMEM containing only 300 nM insulin for a total of two times. After this step, the cells were incubated in DMEM without any additives, with a change of medium every 10 days. Fully differentiated adipocytes were observed by light microscopy based on morphology. Oil red O staining was used to detect fat droplets for the various treatments as described earlier. Using parallel time and conditions, ADSCs were incubated in DMEM without any additives as a negative control.
Transwell coculture of ADSCs and PaCa cells
ADSCs were cultured in the apical compartments of Transwells (Transwell insert 0.4 µM; Millipore, USA) with PaCa cells grown in the basal compartment of a 6-well plate (Millipore). ADSCs were seeded onto the upper layer of Transwells at a density of 2×105 cells/well and were not in direct contact with PaCa cells. PaCa cells were seeded onto the lower layer of Transwells at a density of 2×104 cells/well. All cells were cultured in L-DMEM supplemented with 10% FBS at 37°C in 5% CO2. As a control, the same PaCa cells were seeded instead of ADSCs onto the upper layer. After the cells were cultured for 72 h, cell proliferation was analyzed using a cell counting kit (CCK-8, Dojindo, Japan) following the manufacturer's instructions. Absorption (A) was measured at 450 nm using a spectrophotometer. The proliferation rate (%) of PaCa cells was calculated using the following equation: 1−(A of control−A of ADSCs)/(A of control−A of PaCa)×100%. The experiments were performed in triplicate and repeated twice.
Invasion assay
An in vitro invasion assay was performed using Boyden chambers with inserts (pore size 8 µm) coated with Matrigel on 24-well plates. Briefly, PaCa cells (4×105 cells/mL) were suspended in 250 µL medium containing 2% FBS in the upper chamber, which was precoated with 15 µL Matrigel (0.5 µg/mL). ADSCs were seeded onto the lower layer of Transwells at a density of 2×105 cells/well. The coculture system was incubated at room temperature for 15 min. The control group consisted of plates with medium containing 2% FBS and PaCa cells in the upper chamber only. The plates were incubated for 24 h at 37°C in 5% CO2. Cells on the upper side of the filters were removed with cotton-tipped swabs, and invaded cells were fixed with 4% paraformaldehyde, washed with PBS, air dried, and stained with crystal violet for 30 min. They were then rinsed several times with distilled water. The number of invading cells was counted in five random microscopic fields. Each experiment was repeated three times.
SDF-1 ELISA
After the cells were cultured for 72 h at 37°C with 5% CO2 in a humidified incubator, the culture medium was collected separately and centrifuged at 800 g for 5 min to remove debris. The supernatants were then frozen at −80°C for further assessment by ELISA. ELISA was performed using a human CXCL12/SDF-1 kit (R&D Systems) according to the manufacturer's instructions.
SDF-1 and CXCR4 qRT-PCR
Total RNA was extracted from cultured cells using Trizol (Invitrogen, USA). The cDNA was synthesized using a random primer from 1 µg total RNA with the RevertAid First Strand cDNA Synthesis Kit according to the manufacturer's instructions (Fermentas, USA). The following primers were used for qRT-PCR: human SDF-1 (sense: 5′-gctttgagtgactgggtt-3′, antisense: 5′-gtggcaagatgatggttt-3′), PCR product size 124 bp; human CXCR4 (sense: 5′-gaagctgttggctgaaaagg-3′, antisense: 5′-gagtcgatgctgatcccaat-3′), PCR product size 345 bp; human β-actin (sense: 5′-actcttccagccttccttc-3′, antisense: 5′-tgtcaccttcaccgttcc-3′), PCR product size 516 bp. PCR was performed following the manufacturer's instructions. The cycling conditions were 3 min at 94°C, followed by 30 cycles of denaturation at 94°C for 30 s, annealing at 54°C for 30 s, and extension at 72°C for 60 s. Amplified DNA fragments were resolved by electrophoresis on 1% agarose gels containing ethidium bromide.
Proliferation assay
PaCa cells were seeded into culture medium containing 10% FBS on a 96-well plate at a cell density of 5000 cells/well. Cells were incubated with the indicated concentration of SDF-1 for 72 h and then counted using a CCK-8 kit. Absorbance was measured with a microplate reader at a wavelength of 490 nm. In another set of experiments, cells were first treated with the indicated concentration of AMD3100 (100 ng/mL) for 6 h and then refed with medium containing SDF-1 (1 ng/mL). After 72 h, the number of cells was determined using the CCK-8 kit and absorbance was measured with a microplate reader at a wavelength of 490 nm. Each condition was assessed with five independent samples, and the experiments were repeated three times.
Transwell coculture of ADSCs and PaCa cells with AMD3100
PaCa cells were seeded in culture medium containing 10% FBS on a 96-well plate at a density of 5000 cells/well. AMD3100 (100 ng/mL) was added to the wells and the same Transwell cocultured system was set up as described above. Cells were cultured for 72 h and then the number of PaCa cells was counted using the CCK-8 kit. Absorbance was measured with a microplate reader at 490 nm. Each condition was assessed with five independent samples, and the experiments were repeated three times.
Statistical analysis
Data are reported as means±SD and analyzed using analysis of variance. A value of P<0.05 was considered to be statistically significant.
Results
Identification of ADSCs
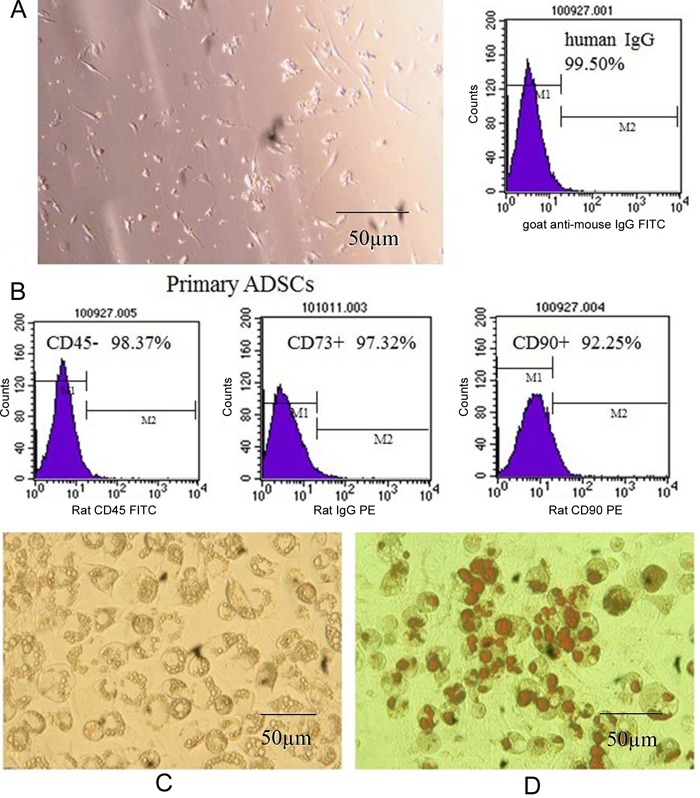
Morphologically, primary human ADSCs were spindle shaped and adherent to the dish. Several ADSCs markers were confirmed by flow cytometric analysis, including CD73, CD90, and CD45 (97.32%, 92.25%, and 98.37% positivity, respectively; Figure 1). The CD marker analysis of ADSCs was consistent with typical mesenchymal stem cells. After differentiation, the accumulated adipocytes in ADSCs were detected by oil red O staining for the various treatments. Most of the ADSCs differentiated into adipocytes successfully, and oil red O staining detected numerous fat droplets in the cytoplasm of adipocytes. However, in the negative control, no adipocytes were observed in ADSCs (Figure 1).
Figure 1. A, Phenotype of adipose tissue-derived stem cells (ADSCs) isolated from human adipose tissue (on the 1st day). B, Expression of CD45, CD73, and CD90 in isolated ADSCs. C, Fat droplets were observed in the cytoplasm of adipocytes at 100X magnification. D, Fat droplets were stained with oil red O.
Effect of ADSCs on the proliferation and invasion of PaCa cells
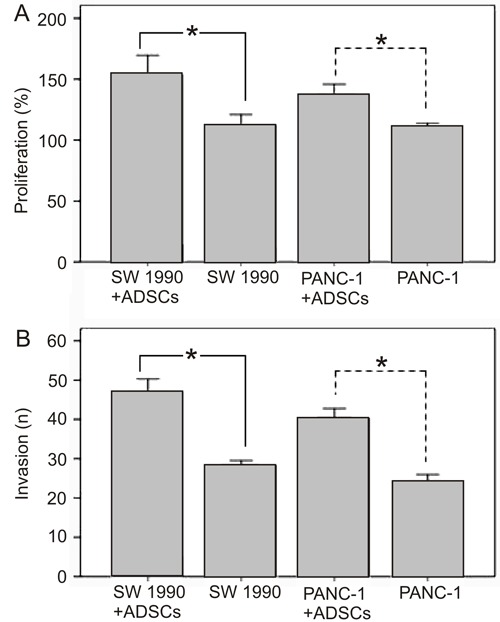
Using a cell coculture system, we next found that ADSCs promote the proliferation of PaCa cells (Figure 2A). The number of pancreatic SW1990 and PANC-1 cells cultured with ADSCs in the system was significantly higher than the cells grown in the absence of ADSCs (P<0.05). Moreover, the invasive behavior of PaCa cells incubated with ADSC supernatant was enhanced (P<0.05; Figure 2B).
Figure 2. Effect of adipose tissue-derived stem cells (ADSCs) on the proliferation and invasion of human pancreatic cancer (PaCa) cell lines (SW1990 and PANC-1). A, After coculturing ADSCs with PaCa cells for 72 h, the proliferation of PaCa cells was examined using the CCK-8 assay. The proliferation of SW1990 and PANC-1 cells cocultured with ADSCs was significantly increased compared to the respective cells cultured alone (*P<0.05, t-test). B, ADSCs were cocultured with PaCa cells for 72 h and the percentage of PaCa cell invasion was examined under a microscope. The invasion of SW1990 and PANC-1 cells was significantly greater in the presence of ADSCs compared to control cells (*P<0.05, t-test).
Secretion of SDF-1 by ADSCs
The amount of SDF-1 secreted by ADSCs and PaCa cells was next determined by ELISA. The concentration of SDF-1 in the ADSC supernatant was 1110.8±91.5 pg/mL (range: 1007.3-1201.2 pg/mL). Importantly, a very low concentration of SDF-1 was detected in the supernatant of PaCa cells. These data confirmed that ADSCs, but not PaCa cell lines, secrete SDF-1.
Expression of SDF-1 and CXCR4 in cultured PaCa cell lines and ADSCs
We evaluated the expression of SDF-1 and CXCR4 by qRT-PCR in cultured cells. ADSCs exhibited a higher expression of SDF-1 compared to SW1990 and PANC-1 cells (69±5 vs 0 and 0, respectively; P<0.01). However, these two PaCa cell lines showed a higher expression of CXCR4 compared to ADSCs (109.3±10.7 and 97.6±7.6 vs 18.3±1.7, respectively; P<0.01).
Effects of SDF-1 on the proliferation of PaCa cell lines
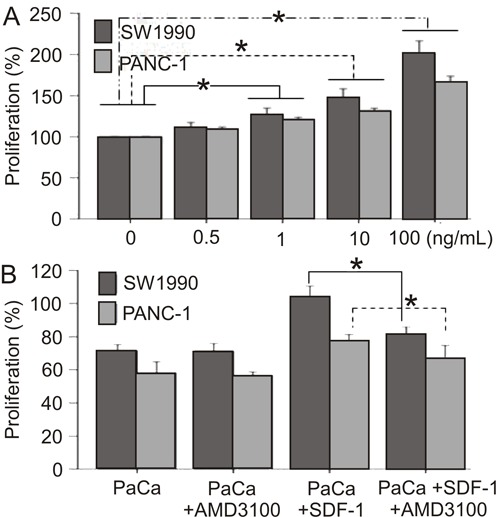
The effect of SDF-1 on SW1990 and PANC-1 cell growth was determined using the CCK-8 assay. We found that SDF-1 enhanced the proliferation of PaCa cells in a dose-dependent manner 1 (0, 0.5, 1, 10, 100 ng/mL) compared to controls (P<0.05; Figure 3A). Importantly, the CXCR4 antagonist AMD3100 significantly blocked SDF-1-induced proliferation (P<0.05) but did not inhibit SW1990 and PANC-1 basal cell growth in the absence of SDF-1 (Figure 3B).
Figure 3. Effect of SDF-1 on the proliferation of human pancreatic cancer (PaCa) cell lines (SW1990 and PANC-1) using the CCK-8 assay. A, SDF-1 enhanced the proliferation of PaCa cells in a dose-dependent manner (*P<0.05, t-test). B, AMD3100 blocked SDF-1-mediated proliferation (*P<0.05, t-test).
Effects of ADSCs on the proliferation of PaCa cells in the presence of AMD3100
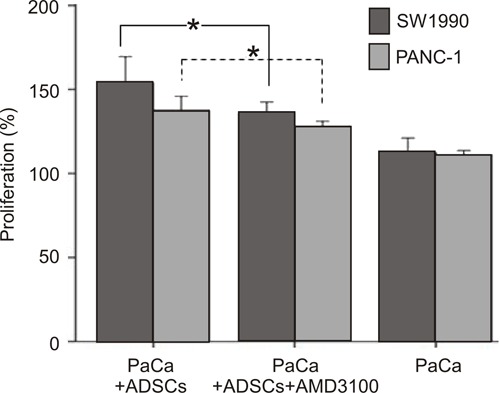
The effect of ADSCs on SW1990 and PANC-1 cell growth in the presence of AMD3100 was also determined using the CCK-8 assay. We found that ADSCs enhanced the proliferation of both PaCa cell lines, which was significantly reduced by the addition of 100 ng/mL AMD3100 (P<0.05; Figure 4).
Figure 4. Effect of adipose tissue-derived stem cells (ADSCs) on the proliferation of human pancreatic cancer (PaCa) cell lines (SW1990 and PANC-1) in the presence of AMD3100. AMD3100 downregulated the effects of ADSCs on PaCa cell line proliferation (*P<0.05, t-test).
Discussion
The interaction between a tumor and tumor-associated stroma has been reported in many studies, and the importance of the local tumor microenvironment for tumor progression has been recognized for many years (21,22). In many solid tumors, the stroma is critically important for promoting tumor growth. Our study demonstrated that ADSCs promote pancreatic cancer cell proliferation and invasion in vitro. Furthermore, we demonstrated that the SDF-1/CXCR4 axis plays an important role in mediating the tumor-promoting effect.
Our findings that ADSCs promote the growth of PaCa cells are of particular importance, because ADSCs are derived from adipose tissue that is abundant in the tissues adjacent to the pancreas. ADSCs are ubiquitously distributed in all tissues and may interact with tumors directly (10). We first found that ADSCs, but not PaCa cells, secrete SDF-1. SDF-1 is a secreted chemokine that is released into the interstitial space, where it acts on cells in the local microenvironment in a paracrine fashion to stimulate directional migration of hematopoietic and nonhematopoietic normal and malignant cells (23). SDF-1 exerts a variety of biological functions, including the regulation of angiogenesis and inhibition of apoptosis, as well as tumor growth, migration, and invasion-promoting effects (24-26) through the SDF-1/CXCR4 receptor ligand axis. Furthermore, Brand et al. (27) demonstrated that SDF-1 stimulation induces a significant increase in vascular endothelial growth factor protein levels in the colorectal cancer line HT-29. SDF-1 has also been shown to play a role in tumors from breast, melanoma, ovaries, stomach, and other carcinomas (12).
We also assessed the expression of CXCR4 in the two PaCa cell lines using qRT-PCR and found that the mRNA levels were elevated compared to controls. In contrast, CXCR4 mRNA levels were very low in ADSCs. Importantly, these results were in agreement with previous studies (13,28). We also found that the SDF-1/CXCR4 axis plays an important role in the proliferation of PaCa cells. In this study, we demonstrated that recombinant SDF-1 could significantly promote the proliferation of PaCa cells. Blocking the receptor with AMD3100 was sufficient for preventing SDF-1-mediated proliferation in PANC-1 and SW1990 cells. It has also been reported that CXCR4 signaling in pancreatic cancer cell lines can promote PaCa proliferation (29). Therefore, taken together, these observations indicate that CXCR4 is a potential therapeutic target in pancreatic cancer.
In this study, we also found that ADSCs stimulate the migratory and invasive behavior of PaCa cells. In a Transwell chamber migration assay, the presence of ADSCs significantly increased migration of PaCa cells. An invasion assay using Matigel-coated invasion chambers also showed that ADSCs stimulated invasion of PaCa cells and that these effects were inhibited by the addition of the CXCR4 inhibitor AMD3100. Therefore, SDF-1 plays a key role in promoting the migration and invasion of PaCa cells. However, although the ADSC-induced proliferation was inhibited by AMD3100, proliferation was still higher than the control. Therefore, other factors, such as matrix metalloproteinase (MMP)-9, MMP-2, and vascular endothelial growth factor (10,30), may also be involved in promoting the proliferation, migration, and invasion of PaCa cells. It has been shown that myofibroblast-derived SDF-1 recruits endothelial progenitor cells to sites of carcinomas and thereby enhances angiogenesis and tumor growth (31). Therefore, ADSC-derived SDF-1 may be partially responsible for the proliferation and invasion of PaCa cells.
The aim of this study was to gain a better understanding of the mechanisms related to pancreatic cancer growth and invasion. Despite advances in surgical and medical therapies, pancreatic cancer continues to have a very poor prognosis. The poor prognosis of pancreatic cancer is attributable to late presentation, aggressive local invasion, early metastases, and poor response to chemotherapy (32,33). Therefore, a better understanding of the fundamental nature of this cancer is needed to improve clinical outcomes. Our finding that local stem cells adjacent to pancreatic cancer cells are involved in tumor growth and metastasis may be useful for pancreatic tumor resection, because these results suggest that the adipose tissue surrounding pancreatic tissue should be resected completely. However, future studies are needed to confirm these findings.
Acknowledgments
Research supported in part by a grant from Zhejiang Province Key Surgery projects (Zhejiang High-Tech #2008-255), China.
Footnotes
First published online September 23, 2013.
References
- 1.Bhowmick NA, Neilson EG, Moses HL. Stromal fibroblasts in cancer initiation and progression. Nature. 2004;432:332–337. doi: 10.1038/nature03096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Armstrong T, Packham G, Murphy LB, Bateman AC, Conti JA, Fine DR, et al. Type I collagen promotes the malignant phenotype of pancreatic ductal adenocarcinoma. Clin Cancer Res. 2004;10:7427–7437. doi: 10.1158/1078-0432.CCR-03-0825. [DOI] [PubMed] [Google Scholar]
- 3.Bachem MG, Schunemann M, Ramadani M, Siech M, Beger H, Buck A, et al. Pancreatic carcinoma cells induce fibrosis by stimulating proliferation and matrix synthesis of stellate cells. Gastroenterology. 2005;128:907–921. doi: 10.1053/j.gastro.2004.12.036. [DOI] [PubMed] [Google Scholar]
- 4.Miyamoto H, Murakami T, Tsuchida K, Sugino H, Miyake H, Tashiro S. Tumor-stroma interaction of human pancreatic cancer: acquired resistance to anticancer drugs and proliferation regulation is dependent on extracellular matrix proteins. Pancreas. 2004;28:38–44. doi: 10.1097/00006676-200401000-00006. [DOI] [PubMed] [Google Scholar]
- 5.Muerkoster S, Wegehenkel K, Arlt A, Witt M, Sipos B, Kruse ML, et al. Tumor stroma interactions induce chemoresistance in pancreatic ductal carcinoma cells involving increased secretion and paracrine effects of nitric oxide and interleukin-1beta. Cancer Res. 2004;64:1331–1337. doi: 10.1158/0008-5472.CAN-03-1860. [DOI] [PubMed] [Google Scholar]
- 6.Ohuchida K, Mizumoto K, Murakami M, Qian LW, Sato N, Nagai E, et al. Radiation to stromal fibroblasts increases invasiveness of pancreatic cancer cells through tumor-stromal interactions. Cancer Res. 2004;64:3215–3222. doi: 10.1158/0008-5472.CAN-03-2464. [DOI] [PubMed] [Google Scholar]
- 7.Daly AJ, McIlreavey L, Irwin CR. Regulation of HGF and SDF-1 expression by oral fibroblasts - implications for invasion of oral cancer. Oral Oncol. 2008;44:646–651. doi: 10.1016/j.oraloncology.2007.08.012. [DOI] [PubMed] [Google Scholar]
- 8.Kajita T, Ohta Y, Kimura K, Tamura M, Tanaka Y, Tsunezuka Y, et al. The expression of vascular endothelial growth factor C and its receptors in non-small cell lung cancer. Br J Cancer. 2001;85:255–260. doi: 10.1054/bjoc.2001.1882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pablos JL, Amara A, Bouloc A, Santiago B, Caruz A, Galindo M, et al. Stromal-cell derived factor is expressed by dendritic cells and endothelium in human skin. Am J Pathol. 1999;155:1577–1586. doi: 10.1016/S0002-9440(10)65474-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Muehlberg FL, Song YH, Krohn A, Pinilla SP, Droll LH, Leng X, et al. Tissue-resident stem cells promote breast cancer growth and metastasis. Carcinogenesis. 2009;30:589–597. doi: 10.1093/carcin/bgp036. [DOI] [PubMed] [Google Scholar]
- 11.Taichman RS, Cooper C, Keller ET, Pienta KJ, Taichman NS, McCauley LK. Use of the stromal cell-derived factor-1/CXCR4 pathway in prostate cancer metastasis to bone. Cancer Res. 2002;62:1832–1837. [PubMed] [Google Scholar]
- 12.Schrader AJ, Lechner O, Templin M, Dittmar KE, Machtens S, Mengel M, et al. CXCR4/CXCL12 expression and signalling in kidney cancer. Br J Cancer. 2002;86:1250–1256. doi: 10.1038/sj.bjc.6600221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Marchesi F, Monti P, Leone BE, Zerbi A, Vecchi A, Piemonti L, et al. Increased survival, proliferation, and migration in metastatic human pancreatic tumor cells expressing functional CXCR4. Cancer Res. 2004;64:8420–8427. doi: 10.1158/0008-5472.CAN-04-1343. [DOI] [PubMed] [Google Scholar]
- 14.Mori T, Doi R, Koizumi M, Toyoda E, Ito D, Kami K, et al. CXCR4 antagonist inhibits stromal cell-derived factor 1-induced migration and invasion of human pancreatic cancer. Mol Cancer Ther. 2004;3:29–37. [PubMed] [Google Scholar]
- 15.Wang J, Wang J, Sun Y, Song W, Nor JE, Wang CY, et al. Diverse signaling pathways through the SDF-1/CXCR4 chemokine axis in prostate cancer cell lines leads to altered patterns of cytokine secretion and angiogenesis. Cell Signal. 2005;17:1578–1592. doi: 10.1016/j.cellsig.2005.03.022. [DOI] [PubMed] [Google Scholar]
- 16.Kucerova L, Altanerova V, Matuskova M, Tyciakova S, Altaner C. Adipose tissue-derived human mesenchymal stem cells mediated prodrug cancer gene therapy. Cancer Res. 2007;67:6304–6313. doi: 10.1158/0008-5472.CAN-06-4024. [DOI] [PubMed] [Google Scholar]
- 17.Prantl L, Muehlberg F, Navone NM, Song YH, Vykoukal J, Logothetis CJ, et al. Adipose tissue-derived stem cells promote prostate tumor growth. Prostate. 2010;70:1709–1715. doi: 10.1002/pros.21206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yu JM, Jun ES, Bae YC, Jung JS. Mesenchymal stem cells derived from human adipose tissues favor tumor cell growth in vivo . Stem Cells Dev. 2008;17:463–473. doi: 10.1089/scd.2007.0181. [DOI] [PubMed] [Google Scholar]
- 19.Khakoo AY, Pati S, Anderson SA, Reid W, Elshal MF, Rovira II, et al. Human mesenchymal stem cells exert potent antitumorigenic effects in a model of Kaposi's sarcoma. J Exp Med. 2006;203:1235–1247. doi: 10.1084/jem.20051921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Qiao L, Xu ZL, Zhao TJ, Ye LH, Zhang XD. Dkk-1 secreted by mesenchymal stem cells inhibits growth of breast cancer cells via depression of Wnt signalling. Cancer Lett. 2008;269:67–77. doi: 10.1016/j.canlet.2008.04.032. [DOI] [PubMed] [Google Scholar]
- 21.de Wever O, Mareel M. Role of tissue stroma in cancer cell invasion. J Pathol. 2003;200:429–447. doi: 10.1002/path.1398. [DOI] [PubMed] [Google Scholar]
- 22.Liotta LA, Kohn EC. The microenvironment of the tumour-host interface. Nature. 2001;411:375–379. doi: 10.1038/35077241. [DOI] [PubMed] [Google Scholar]
- 23.Burger JA, Kipps TJ. CXCR4: a key receptor in the crosstalk between tumor cells and their microenvironment. Blood. 2006;107:1761–1767. doi: 10.1182/blood-2005-08-3182. [DOI] [PubMed] [Google Scholar]
- 24.Balkwill F. Cancer and the chemokine network. Nat Rev Cancer. 2004;4:540–550. doi: 10.1038/nrc1388. [DOI] [PubMed] [Google Scholar]
- 25.Homey B, Muller A, Zlotnik A. Chemokines: agents for the immunotherapy of cancer? Nat Rev Immunol. 2002;2:175–184. doi: 10.1038/nri748. [DOI] [PubMed] [Google Scholar]
- 26.Murphy PM. Chemokines and the molecular basis of cancer metastasis. N Engl J Med. 2001;345:833–835. doi: 10.1056/NEJM200109133451113. [DOI] [PubMed] [Google Scholar]
- 27.Brand S, Dambacher J, Beigel F, Olszak T, Diebold J, Otte JM, et al. CXCR4 and CXCL12 are inversely expressed in colorectal cancer cells and modulate cancer cell migration, invasion and MMP-9 activation. Exp Cell Res. 2005;310:117–130. doi: 10.1016/j.yexcr.2005.07.006. [DOI] [PubMed] [Google Scholar]
- 28.Koshiba T, Hosotani R, Miyamoto Y, Ida J, Tsuji S, Nakajima S, et al. Expression of stromal cell-derived factor 1 and CXCR4 ligand receptor system in pancreatic cancer: a possible role for tumor progression. Clin Cancer Res. 2000;6:3530–3535. [PubMed] [Google Scholar]
- 29.Billadeau DD, Chatterjee S, Bramati P, Sreekumar R, Shah V, Hedin K, et al. Characterization of the CXCR4 signaling in pancreatic cancer cells. Int J Gastrointest Cancer. 2006;37:110–119. doi: 10.1007/s12029-007-0011-7. [DOI] [PubMed] [Google Scholar]
- 30.Nagakawa Y, Aoki T, Kasuya K, Tsuchida A, Koyanagi Y. Histologic features of venous invasion, expression of vascular endothelial growth factor and matrix metalloproteinase-2 and matrix metalloproteinase-9, and the relation with liver metastasis in pancreatic cancer. Pancreas. 2002;24:169–178. doi: 10.1097/00006676-200203000-00008. [DOI] [PubMed] [Google Scholar]
- 31.Iyengar P, Combs TP, Shah SJ, Gouon-Evans V, Pollard JW, Albanese C, et al. Adipocyte-secreted factors synergistically promote mammary tumorigenesis through induction of anti-apoptotic transcriptional programs and proto-oncogene stabilization. Oncogene. 2003;22:6408–6423. doi: 10.1038/sj.onc.1206737. [DOI] [PubMed] [Google Scholar]
- 32.Matsuo Y, Ochi N, Sawai H, Yasuda A, Takahashi H, Funahashi H, et al. CXCL8/IL-8 and CXCL12/SDF-1alpha co-operatively promote invasiveness and angiogenesis in pancreatic cancer. Int J Cancer. 2009;124:853–861. doi: 10.1002/ijc.24040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Niedergethmann M, Alves F, Neff JK, Heidrich B, Aramin N, Li L, et al. Gene expression profiling of liver metastases and tumour invasion in pancreatic cancer using an orthotopic SCID mouse model. Br J Cancer. 2007;97:1432–1440. doi: 10.1038/sj.bjc.6604031. [DOI] [PMC free article] [PubMed] [Google Scholar]


