Abstract
Cerebellar and frontal cortical pathologies have been commonly reported in schizophrenia, autism, and other developmental disorders. Whether there is a relationship between prefrontal and cerebellar pathologies is unknown. Using fixed potential amperometry, dopamine (DA) efflux evoked by cerebellar or, dentate nucleus electrical stimulation (50 Hz, 200 μA) was recorded in prefrontal cortex of urethane anesthetized lurcher (Lc/+) mice with 100% loss of cerebellar Purkinje cells and wildtype (+/+) control mice. Cerebellar stimulation with 25 and 100 pulses evoked prefrontal cortex DA efflux in +/+ mice that persisted for 12 and 25 s poststimulation, respectively. In contrast, 25 pulse cerebellar stimulation failed to evoke prefrontal cortex DA efflux in Lc/+ mice indicating a dependency on cerebellar Purkinje cell outputs. Dentate nucleus stimulation (25 pulses) evoked a comparable but briefer (baseline recovery within 7 s) increase in prefrontal cortex DA efflux compared to similar cerebellar stimulation in +/+ mice. However, in Lc/+ mice 25 pulse dentate nucleus evoked prefrontal cortex DA efflux was attenuated by 60% with baseline recovery within 4 s suggesting that dentate nucleus outputs to prefrontal cortex remain partially functional. DA reuptake blockade enhanced 100 pulse stimulation evoked pre-frontal cortex responses, while serotonin or norepinephrine reuptake blockade were without effect indicating the specificity of the amperometric recordings to DA. Results provide neurochemical evidence that the cerebellum can modulate DA efflux in the prefrontal cortex. Together, these findings may explain why cerebellar and frontal cortical pathologies co-occur, and may provide a mechanism that accounts for the diversity of symptoms common to multiple developmental disorders.
Keywords: cerebellum, dentate nucleus, prefrontal cortex, amperometry, lurcher mice
INTRODUCTION
Although structural or functional abnormalities of the cerebellum are known to occur in a variety of disorders including schizophrenia, depression, mania, and autism, traditionally little attention has been paid to whether they could play a major role in the pathogenesis of these disorders (Konarski et al., 2005; Phillips et al., 2003). This attitude may be changing, in that neurocognitive theories have emphasized that disruptions in neural networks and distributed parallel processing are likely the basis of many brain diseases (e.g., Goldman-Rakic, 1990; Mesulam, 1990). Left cerebellar peduncle white matter comprising the fibers connecting the cerebellum with the cerebral cortex is reduced in early onset schizophrenia (Kyriakopoulos et al., 2007). Thus, it has been proposed that the diverse symptoms of schizophrenia may result from developmental abnormalities in prefrontal cortico-cerebellar-thalamo-prefrontal cortical circuits (Andreasen et al., 1998).
It is possible that the diverse symptoms of autism are related to similar circuitry. Autism spectrum disorders are one of the most common of the developmental disabilities (Kuehn, 2007). Neuropsychological testing has revealed that impairments in attention, memory, and a marked tendency to perseverate in the face of changing contingencies (executive function) underlie many of the symptoms of autism (Boucher and Lewis, 1989; Courchesne et al., 1994; Dawson and Lewey, 1989; Happe and Frith, 1996).
The multitude of cognitive deficits common to autism has been associated with neural pathology in several brain regions including cerebral and cerebellar cortices. The most reproducible pathology has been found in the cerebellum, and includes cerebellar hypoplasia and reduced neuron numbers (Bauman, 1991). In MRI analyses, lobules of the cerebellum were found to be hypoplastic in high functioning autistic patients (Courchesne et al., 1988), along with substantial reductions in cerebellar Purkinje cell number (Carper and Courchesne, 2000; Ritvo et al., 1986). Frontal lobe structural abnormalities have also been reported (see for review, Courchesne et al., 2005). Thus, fMRI has shown that the degree of frontal lobe abnormality correlated with the degree of cerebellar abnormality in autistic children (Carper and Courchesne, 2000). The structural abnormalities in the frontal cortex may have functional significance. As measured by positron emission tomography (PET), autistic children exhibit low medial prefrontal cortex (PFC) dopaminergic activity compared to age and gender matched controls (Ernst et al., 1997).
Relationships between cerebellar pathology and PFC function are unknown. That they are closely related is consistent with the hypothesis that autism is a disconnection syndrome (Geshwind and Levitt, 2007). As such, we explored a functional relationship between these structures using lurcher and wildtype mice. Lurcher mice were selected because they lose all cerebellar Purkinje cells between the 2nd and 4th weeks of life (Wetts and Herrup, 1982); a time that is analogous to the loss of cells in autistic individuals (Pickett and London, 2005). The lurcher gene codes for the δ2-glutamateδ2 glutamate receptor that is expressed in cerebellar Purkinje cells (Wetts and Herrup, 1982; Zuo et al., 1997). To determine if loss of cerebellar Purkinje cells affected functioning in the PFC we used in vivo fixed potential amperometry to monitor evoked PFC dopamine (DA) efflux following electrical stimulation of the cerebellar Purkinje cell layer and dentate nucleus.
MATERIALS AND METHODS
Subjects
Wildtype and lurcher mice (gene symbol: +/+ and Lc/+, respectively) were derived from mating pairs (B6CBACaAw-J/A-Grid2Lc, Jackson Laboratory, ME) and maintained at the University of Tennessee and the University of Memphis Animal Care Facility. Lc/+ (n = 10) and +/+ (n = 10) mice were used for cerebellar Purkinje cell layer (PCL) and dentate nucleus (DN) stimulation studies and 15 +/+ mice for presynaptic transporter inhibition studies involving PCL stimulation (see below). Experiments were approved by a local Institutional Animal Care and Use Committee and conducted in compliance with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals.
Surgery
Mice were anesthetized with urethane (1.5 g/kg, i.p.) and placed in a stereotaxic (David Kopf, CA) with a mouse head-holder (Stoelting, IL) within a Faraday cage. Body temperature was maintained at 37 ± 0.5°C with a temperature-regulated heating pad (TC-1000, CWE, NY). Three holes were drilled through the animal’s skull for an Ag/AgCl reference/auxiliary electrode (positioned at the surface of the cortex), a concentric bipolar stimulating electrode (CBARD75, 125 μm outer and 25 μm inner pole diam., FHC, ME), and a DA recording electrode (carbon fiber 10 μm o.d., 250 μm length, Thornel Type P, Union Carbide, PA) (Forster and Blaha, 2003; Lee et al., 2006). The stimulating electrode tip was positioned in mm with respect to dura and bregma in the PCL (+/+ mice: AP −6.25, ML −0.75, DV −0.2 to −0.6; Lc/+ mice: AP −5.85, ML −0.35, DV −0.1 to −0.3) and another in the DN (+/+ mice: AP −6.25, ML −2.1, DV −2.1; Lc/+ mice: AP −5.85, ML −1.0, DV −1.0), both contralateral to the DA recording electrode positioned in mm with respect to dura and bregma in the PFC (+/+ and Lc/+ mice: AP +2.35, ML +0.25, DV −1.0) (Franklin and Paxinos, 1997). For reuptake inhibition studies, only one stimulating electrode was implanted into the PCL of +/+ mice using the coordinates above.
Fixed potential amperometry and electrical stimulation
Following implantation of all electrodes, a fixed positive potential (+0.8 V) was applied to the recording electrode and oxidation current monitored continuously (10K samples/s) with an electrometer (e-corder/picostat system, eDAQ, CO), filtered at 10 Hz low pass (Forster and Blaha, 2003). Electrical stimulation of the PCL or DN (counterbalanced within individual +/+ and Lc/+ mice) consisted of 50 Hz trains of 25 cathodic monophasic pulses (200 μA intensity, 0.5 ms pulse duration) applied every 30 s over a 6-min period via an optical stimulus isolator and programmable pulse generator (Iso-Flex/Master-8; AMPI, Jerusalem, Israel). Similar stimulation of the PCL with 100 pulses were used in +/+ mice to determine the selectivity of the amperometric recordings to DA efflux in the PFC (see below).
Selectivity of amperometric recordings to DA efflux
Amperometric recordings in combination with bare carbon fiber electrodes exhibit poor chemical resolution, but can be used successfully to monitor DA efflux evoked by brief electrical stimulations of less than 4–5 s (Dugast et al., 1994). The selectivity of amperometric recordings to DA efflux in the PFC evoked by electrical stimulation of the PCL (50 Hz train of 100 cathodic monophasic pulses, 200 μA intensity, 0.5-ms pulse duration, applied every 60 s over a 60-min period) was confirmed in separate studies with selective reuptake inhibitors of the presynaptic transporters for DA (nomifensine alone, 20 mg/kg, n = 6 +/+ mice), serotonin, and norepinephrine (fluoxetine + desipramine combination, 20 mg/kg each, n = 9 +/+ mice; Sigma-Aldrich, MO), all dissolved in saline and injected systemically (i.p.) 10 min following initiation of the stimulation protocol described above (Lee et al., 2006). The maximal effects of these drugs on PCL stimulation evoked oxidation current in the PFC of +/+ mice were assessed 30-min postinjection.
Data analysis
Amperometric currents (0.25 s before each of the trains of 25 or 100 pulses of stimulation applied to the PCL or DN of individual mice) were normalized to zero current values. These responses were then averaged first within and then between each strain of mice (+/+ and Lc/+) with respect to the brain region stimulated (PCL and DN) and drug treatment. The mean peak oxidation current values poststimulation were then used to statistically compare differences between mouse strains, stimulated brain regions, and drug treatments using repeated measures analysis of variance (RANOVA). In these analyses, the between subjects factor was either mouse Strain (+/+ and Lc/+), stimulation Site (Purkinje cell layer and Dentate) or Drug treatment (baseline or nomifensine, and fluoxetine + desipramine combination), while the within subjects factor was always the oxidation current values at various time points poststimulation. Depending on the RANOVA, oxidation current at either 0.5 or 1.0 s intervals was used. If the RANOVA indicated a significant interaction with time, Dunnett’s t-tests were used to further identify significant differences.
Histology
After recordings, a direct current (100 μA for 10 s; +5 V for 5 s) was passed through the stimulating electrode in the PCL and DN to leave iron deposits and through the recording electrode in the striatum to lesion tissue, respectively. Mice were then euthanized via an intracardial injection of urethane, the brains removed, and placed overnight in 0.1% potassium ferricyanide/10% formalin to mark the stimulation site with a Prussian blue spot (resulting from redox reaction of ferricyanide). Coronal sections were cut (40 μm) on a cryostat and placements of PCL, DN, and PFC electrodes determined by light microscopy and recorded on representative coronal diagrams (Franklin and Paxinos, 1997).
RESULTS
Stereotaxic placements of electrodes
The stimulating electrode tips were confined within the PCL of the lateral portion of the 4th and 5th cerebellar lobules (+/+ mice, n = 21 electrodes, range −5.8 to −6.3, 0.6–1.1, and 0.25–0.8; Lc/+ mice, n = 10 electrodes, range −5.75 to −5.90, 0.2–0.5, and 0.1–0.4) and within the boundaries of the DN (+/+ mice, n = 10 electrodes, −5.8 to −6.3, 2.0–2.3, and 2.0–2.6; Lc/+ mice, n = 10 electrodes, range −5.75 to −5.90, 0.9–1.25, and 0.75–1.25) (Figs. 1A and 1B). All of these coordinates in mm were posterior to bregma, lateral to midline, and ventral from dura, respectively. Electrochemical recording electrode surfaces were confined within the PFC (prelimbic region) (+/+ mice, n = 25 electrodes, range +2.1 to +2.4, 0.15–0.5 and 0.75–1.5; Lc/+ mice, n = 10 electrodes, range +2.1 to +2.4, 0.15–0.4, and 0.65–1.4) (Figs. 1C and 1D). All of these coordinates in mm were anterior to bregma, lateral to midline, and ventral from dura, respectively.
Fig. 1.

Representative coronal sections of the wildtype (+/+) and lurcher (Lc/+) mouse cerebellum (A,B) and forebrain (C,D) (adapted from the mouse atlas of Franklin and Paxinos, 1997), with gray shaded areas illustrating placements of stimulating electrodes in the Purkinje cell layer (PCL) and dentate nucleus (DN) and electrochemical recording electrodes in the prefrontal cortex (PFC) (numbers correspond to mm from bregma). Note that the cerebellum of Lc/+ mice is approximately 50% the size of +/+ mice due to the 100% loss of Purkinje cells (Martin et al., 2003, 2006). Thus, based on preliminary histological analysis the stereotaxic coordinates for the PCL and DN were adjusted accordingly for these mice.
Effects of PCL stimulation in +/+ and Lc/+ mice
Electrical stimulation (25 pulses, 50 Hz) of the PCL in +/+ mice evoked a significant increase in PFC oxidation current (+34.2 ± 1.36 pA; Fig. 2A) above prestimulation baseline levels (+1.5 ± 1.2 pA) compared to that observed in Lc/+ animals (Strain X Time, F = 5.82, df = 31,434, P < 0.001). In +/+ mice, current values peaked within 0.35–0.4 s of stimulation and slowly declined thereafter to prestimulation levels within 11–12 s. Dunnett’s t-tests conducted on current values sampled every 0.5 s indicated that the oxidation current in +/+ mice was significantly elevated from 0.5 to 12 s following stimulation, compared to that observed in Lc/+ mice. Similar stimulation of the cerebellar cortex in Lc/+ mice failed to evoke an increase in PFC oxidation current (+1.6 ± 1.5 pA at 0.35 s poststimulation; Fig. 2A) above prestimulation baseline levels (+1.2 ± 1.3 pA).
Fig. 2.
Electrical stimulation (black bar, 25 pulses at 50 Hz, 200 μA) of the (A) Purkinje cell layer (PCL) or (B) dentate nucleus (DN) evokes a long lasting increase in prefrontal cortex DA efflux of wildtype (+/+) mice; an effect that is absent and markedly attenuated, respectively, in PCL and DN stimulated lurcher (Lc/+) mice. In (A and B) the thick black lines and outer thin black lines are the mean ± SEM, respectively (n = 6 and 10 for PCL and DN stimulation in +/+ mice, respectively, n = 10 and 10 for Lc/+ mice, respectively). * indicates significant difference from 0.5 to 12 s between +/+ and Lc/+ mice following PCL stimulation and significant difference from 0.5 to 5 s between +/+ and Lc/+ mice following DN stimulation.
Effects of DN stimulation in +/+ and Lc/+ mice
Compared to PCL stimulation (25 pulses, 50 Hz) in +/+ mice, similar DN stimulation in these mice evoked a comparable mean peak increase in PFC oxidation current (+34.1 ± 1.9 pA; Fig. 2B) above prestimulation baseline levels (−1.1 ± 1.9 pA) within 0.35–0.4 s of stimulation, but this response recovered to prestimulation levels more rapidly (within 7–8 s). RANOVA confirmed that the time course of oxidation current was significantly different in +/+ mice following PCL or DN stimulation (Site X Time, F = 2.11, df = 31,43, P < 0.001). Dunnett’s t-tests further indicated that, although the peak responses were non-significantly different, PCL stimulation evoked a more prolonged increase in oxidation current than that observed following DN stimulation which lasted from 2.5 to 12 s poststimulation.
In contrast, oxidation current evoked by PCL or DN stimulation (25 pulses, 50 Hz) in Lc/+ mice showed an opposite pattern of change (Site X Time, F = 2.61, df = 30,540, P < 0.001). Thus, in comparison to stimulation of the PCL in Lc/+ mice, similar DN stimulation showed a larger increase in oxidation current between 0.5- and 2.5-s poststimulation (Dunnett’s t-tests).
Control (+/+) and Lc/+ mice also differed significantly in response to this DN stimulation (Strain X Time, F = 5.82 df = 31,434, P < 0.001). Dunnett’s t-tests showed that oxidation current was significantly larger in +/+ mice between 0.5 and 5.0 s after stimulation of the DN, compared to that observed in Lc/+ animals.
Effects of selective reuptake inhibition on PCL stimulation in +/+ mice
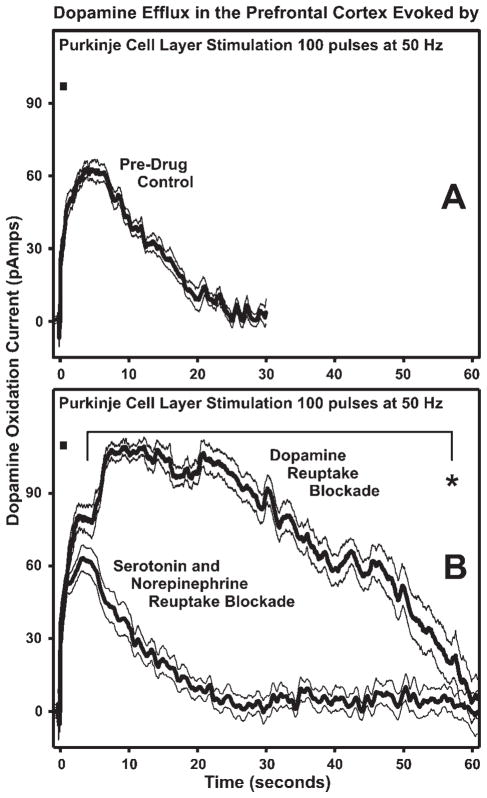
Figure 3A shows the time course of evoked changes in oxidation current in undrugged +/+ mice during 30 s following PCL stimulation (100 pulses, 50 Hz). The response to this stimulation of the PCL was then compared to those of +/+ mice following either DA reuptake blockade (nomifensine 20 mg/kg) or combined blockade of serotonin and norepinephrine (fluoxetine + desipramine combination, 20 mg/kg each). As indicated in Figure 3B, blockade of DA reuptake massively prolonged the stimulation-evoked increase in oxidation current compared to that observed in undrugged mice (Drug X Time F = 12.40, df = 30,300, P < 0.001). Dunnett’s t-tests indicated that throughout the 30 s comparison interval oxidation current following DA reuptake blockade was significantly increased over baseline from 3 to 30 s following stimulation.
Fig. 3.
Electrical stimulation (black bar, 100 pulses at 50 Hz, 200 μA) of the Purkinje cell layer evokes a long lasting increase in prefrontal cortex DA efflux of wildtype (+/+) mice (A); an effect that is significantly enhanced by selective blockade of DA reuptake with systemic administration of nomifensine, but unaffected by selective blockade of serotonin and norepinephrine reuptake with systemic administration of fluoxetine and desipramine, respectively (B). In (A and B) the thick black lines and outer thin black lines are the mean ± SEM, respectively (n = 6 +/+ mice for Pre-Drug Control and DA Reuptake Blockade groups and n = 9 +/+ mice for Serotonin and Norepinephrine Reuptake Blockade group). * indicates significant difference from 4 to 57 s between DA Reuptake Blockade group and Pre-Drug Control and Serotonin and Norepinephrine Reuptake Blockade groups.
Combined blockade of serotonin and norepinephrine reuptake did not produce similar results. RANOVA comparing baseline against the combined blockade indicated that the time course of stimulation-evoked oxidation current did not change significantly across these two conditions (Drug X Time, F = 1.27, df = 30,390, P = ns). As might be expected, the time course of stimulation-evoked oxidation current following DA reuptake blockade differed significantly from that observed following combined blockade of serotonin and norepinephrine (Drug X Time, F = 16.65, df = 60,780, P < 0.001). In this case oxidation current was significantly larger following DA reuptake blockade from 4 to 57 s poststimulation (Dunnett’s t-tests).
DISCUSSION
Electrical stimulation of the PCL in the cerebellar cortex of +/+ mice with 25 and 100 pulses at 50 Hz evoked significant stimulus train-dependent increases in PFC DA efflux, effects that persisted after the stimulations had ended. Similar stimulation of the deep cerebellar DN in these mice also evoked PFC DA efflux of a comparable magnitude, but briefer duration. In contrast, stimulation of the cerebellar cortex in Lc/+ mice with a complete loss of Purkinje cells failed to evoke a significant increase in PFC DA efflux; however, DN stimulation in these mice evoked a significant increase in PFC DA efflux. Compared to +/+ mice, this response was significantly attenuated. Selective detection of PFC DA efflux was confirmed by showing that blockade of norepinephrine and serotonin reuptake failed to alter PCL evoked PFC DA efflux, while inhibition of DA reuptake selectively enhanced the magnitude and recovery of this response in the PFC. Altogether, these results are the first to indicate the dependency of PFC DA release on functional cerebellar Purkinje cell outputs and further indicate that dentate output to the PFC remains partially functional in Lc/+ mice. These results are consistent with recent immunohistochemical and electrophysiological studies showing partial compensatory restoration in DN function as a consequence of primary Purkinje cell loss (Baurle et al., 1997; Sultan et al., 2002).
Similarities between the cognitive deficits seen with cerebellar or PFC lesions in humans suggest that damage to the DN or PFC yield similar deficits because they both interrupt cerebellar to PFC inputs (Middleton and Strick, 2001). Similar to those observed in autism, symptoms of frontal cortical damage include deficits in movement programming, losses in divergent thinking, behavioral spontaneity and response inhibition, poor temporal memory as well as impairments in social behavior (Kolb and Whishaw, 1990). Thus, it is possible that a disconnection between cerebellar Purkinje cell output to the PFC could result in aberrant DA signaling in the PFC which, in turn, would lead to abnormal behavior. In this regard it is noteworthy that we have previously observed cognitive deficits in mice that were correlated with the degree of developmental loss of cerebellar Purkinje cells (Martin et al., 2003, 2004).
Functional significance
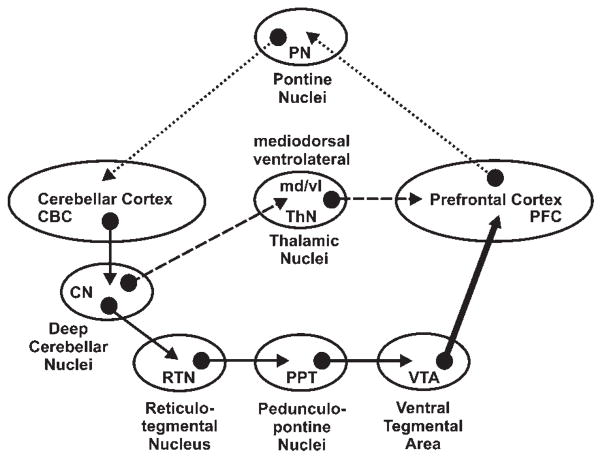
Disruption in the circuitry underlying cerebellar modulation of PFC DA release could represent a main mechanism of cerebellar involvement in various cognitive dysfunctions including autism and schizophrenia. DA neurons in the ventral tegmental area (VTA) comprise the mesocortical dopaminergic pathway. A possible circuit for cerebellar modulation of this pathway may involve cerebellar projections to reticulotegmental nuclei in the pons (RTN) which receives glutamatergic input from the DN and interposed deep cerebellar nuclei (CN) (Schwarz and Schmitz, 1997) and pedunculopontine tegmental nuclei (PPT), which receive input from and project back to the RTN (Garcia-Rill et al., 2001) and also project to VTA dopaminergic neurons (Forster and Blaha, 2003) (Fig. 4). A second pathway may involve CN inputs to the PFC via mediodorsal (md) and ventrolateral (vl) thalamic nuclei (ThN). In turn, the PFC projects back to the cerebellum via pontine nuclei (PN) (Middleton and Strick, 2001).
Fig. 4.
Proposed neuronal circuitry underlying cerebellar modulation of prefrontal cortex (PFC) DA neurotransmission. Cerebellar modulation of DA efflux in the PFC may occur via polysynaptic inputs from cerebellar nuclei to DA-containing cells in the ventral tegmental area (solid arrows) or via a monosynaptic input to thalamic projections making presynaptic contact with DA terminals in the PFC (dashed arrows). Glutamatergic pathways are represented as thin solid and dashed arrows and the dopaminergic pathway as a thick solid arrow. Dotted arrows represent PFC feedback to cerebellum via pontine nuclei. Nuclei abbreviations are shown in the ovals.
Neurons in the RTN respond with prolonged spiking activity (up to 12 s) to PPT stimulation (Garcia-Rill et al., 2001). Considering this, the observed prolonged DA efflux that outlasted cerebellar stimulation for several seconds could be due to sustained activity of RTN neurons. Sustained RTN activity could drive PPT neurons which in turn would directly excite midbrain DA neurons. This notion is consistent with our recent findings that either chemical or electrical stimulation of mesopontine nuclei in rats and mice activates midbrain DA neurons that, in turn, lead to an enhancement in DA efflux in forebrain target structures (Blaha et al., 1996; Forster and Blaha, 2000, 2003; Forster et al., 2001). Alternatively, cerebellar modulation of PFC DA release could involve thalamic glutamatergic projections forming presynaptic inputs to DA varicosities in the PFC (Fig. 4). Morphological studies have shown that glutamate-containing terminals are in close apposition to DA-containing terminals in the PFC (Del Arco and Mora, 2005), suggesting that glutamate exerts a local modulation of PFC DA release. Indeed, increases in mesocortical dopaminergic transmission by activation of ionotropic glutamatergic receptors in the PFC have been reported (Feenstra et al., 1995; Jedema and Moghaddam, 1996; Jones et al., 1993), as well as following electrical or chemical stimulation of excitatory thalamic afferents to the PFC (Feenstra et al., 1993; Jones et al., 1987). An additional mechanism may involve thalamic activation of descending PFC glutamatergic projections to the PPT and/or VTA that, in turn, stimulate mesocortical dopaminergic neurons to release DA in the PFC.
CONCLUSION
Cerebellar and frontal cortical pathologies have been commonly reported in schizophrenia, autism, and other developmental disorders. The relationship between neuropathologies of the PFC and the cerebellum is unknown. The present study provides a unique, new framework for understanding how these seemingly disparate pathologies may be related, by showing modulation of PFC DA efflux that is functionally dependent on an intact cerebellar cortex. Future studies will be required to elucidate the neuronal pathways whereby the cerebellum modulates PFC DA, as well as how this modulation is affected by varying numbers of Purkinje cells.
Acknowledgments
Contract grant sponsors: Cure Autism Now, Autism Speaks.
References
- Andreasen NC, Paradiso S, O’Leary DS. “Cognitive dysmetria” as an integrative theory of schizophrenia: A dysfunction in cortical-subcortical-cerebellar circuitry. Schizophr Bull. 1998;24:203–218. doi: 10.1093/oxfordjournals.schbul.a033321. [DOI] [PubMed] [Google Scholar]
- Bauman ML. Microscopic neuroanatomic abnormalities in autism. Pediatrics. 1991;87:791–796. [PubMed] [Google Scholar]
- Baurle J, Helmchen C, Grusser-Cornehls U. Diverse effects of Purkinje cell loss on deep cerebellar and vestibular nuclei neurons in Purkinje cell degeneration mutant mice: A possible compensatory mechanism. J Comp Neurol. 1997;384:580–596. [PubMed] [Google Scholar]
- Blaha CD, Allen LF, Das S, Inglis WL, Latimer MP, Vincent SR, Winn P. Modulation of dopamine efflux in the nucleus accumbens after cholinergic stimulation of the ventral tegmental area in intact, pedunculopontine tegmental nucleus-lesioned, and laterodorsal teg-mental nucleus-lesioned rats. J Neurosci. 1996;16:714–722. doi: 10.1523/JNEUROSCI.16-02-00714.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boucher J, Lewis V. Memory impairments and communication in relatively able autistic children. J Child Psychol Psychiatry. 1989;30:99–122. doi: 10.1111/j.1469-7610.1989.tb00771.x. [DOI] [PubMed] [Google Scholar]
- Carper RA, Courchesne E. Inverse correlation between frontal lobe and cerebellum sizes in children with autism. Brain. 2000;123:836–844. doi: 10.1093/brain/123.4.836. [DOI] [PubMed] [Google Scholar]
- Courchesne E, Yeung-Courchesne R, Press GA, Hesselink JR, Jernigan TL. Hypoplasia of cerebellar vermal lobules VI and VII in autism. N Engl J Med. 1988;318:1349–1354. doi: 10.1056/NEJM198805263182102. [DOI] [PubMed] [Google Scholar]
- Courchesne E, Townsend J, Akshoomoff NA, Saitoh O, Yeung-Courchesne R, Lincoln AJ, James HE, Haas RH, Schreibman L, Lau L. Impairment in shifting attention in autistic and cerebellar patients. Behav Neurosci. 1994;108:848–865. doi: 10.1037//0735-7044.108.5.848. [DOI] [PubMed] [Google Scholar]
- Courchesne E, Redcay E, Morgan JT, Kennedy DP. Autism at the beginning: Microstructural and growth abnormalities underlying the cognitive and behavioral phenotype of autism. Dev Psychopathol. 2005;17:577–597. doi: 10.1017/S0954579405050285. [DOI] [PubMed] [Google Scholar]
- Dawson G, Lewey A. Arousal, attention, and the socioemotional impairments of individuals with autism. In: Dawson G, editor. Autism: Nature, diagnosis and treatment. New York: Guilford Press; 1989. pp. 49–74. [Google Scholar]
- Del Arco A, Mora F. Glutamate-dopamine in vivo interaction in the prefrontal cortex modulates the release of dopamine and acetylcholine in the nucleus accumbens of the awake rat. J Neural Transm. 2005;112:97–109. doi: 10.1007/s00702-004-0172-5. [DOI] [PubMed] [Google Scholar]
- Dugast C, Suaud-Chagny MF, Gonon F. Continuous in vivo monitoring of evoked dopamine release in the rat nucleus accumbens by amperometry. Neuroscience. 1994;62:647–654. doi: 10.1016/0306-4522(94)90466-9. [DOI] [PubMed] [Google Scholar]
- Ernst M, Zametkin AJ, Matochik JA, Pascualvaca D, Cohen RM. Low medial pre-frontal dopaminergic activity in autistic children. Lancet. 1997;350:638–639. doi: 10.1016/s0140-6736(05)63326-0. [DOI] [PubMed] [Google Scholar]
- Feenstra MGP, van der Weij W, Hamstra JJ, Botterblom MHA, Buijs RM. In vivo dopamine release in the rat prefrontal cortex is increased by stimulation of mediodorsal thalamic nucleus. Soc Neurosci Abstr. 1993;19:1382. [Google Scholar]
- Feenstra MGP, van der Weij W, Botterblom MHA. Concentration-dependent dual action of locally applied N-methyl-D-aspartate on extracellular dopamine in the rat prefrontal cortex in vivo. Neurosci Lett. 1995;201:175–178. doi: 10.1016/0304-3940(95)12164-1. [DOI] [PubMed] [Google Scholar]
- Forster G, Blaha CD. Laterodorsal tegmental stimulation elicits dopamine efflux in the rat nucleus accumbens by activation of acetylcholine and glutamate receptors in the ventral tegmental area. Eur J Neurosci. 2000;12:3596–3604. doi: 10.1046/j.1460-9568.2000.00250.x. [DOI] [PubMed] [Google Scholar]
- Forster GL, Blaha CD. Pedunculopontine tegmental stimulation evoked striatal dopamine efflux by activation of acetylcholine and glutamate receptors in the midbrain and pons of the rat. Eur J Neurosci. 2003;17:751–762. doi: 10.1046/j.1460-9568.2003.02511.x. [DOI] [PubMed] [Google Scholar]
- Forster GL, Yeomans JS, Takeuchi J, Blaha CD. M5 muscarinic receptors are required for prolonged accumbal dopamine release after electrical stimulation of the pons in mice. J Neurosci. 2001;21:RC190. doi: 10.1523/JNEUROSCI.22-01-j0001.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franklin KB, Paxinos G. The mouse brain in stereotaxic coordinates. San Diego: Academic Press; 1997. [Google Scholar]
- Garcia-Rill E, Skinner RD, Miyazato H, Homma Y. Pedunculopontine stimulation induces prolonged activation of pontine reticular neurons. Neuroscience. 2001;104:455–465. doi: 10.1016/s0306-4522(01)00094-x. [DOI] [PubMed] [Google Scholar]
- Geschwind DH, Levitt P. Autism spectrum disorders: Developmental disconnection syndromes. Curr Opin Neurobiol. 2007;17:103–111. doi: 10.1016/j.conb.2007.01.009. [DOI] [PubMed] [Google Scholar]
- Goldman-Rakic PS. Cellular and circuit basis of working memory in prefrontal cortex of nonhuman primates. In: Uylings HBM, Van Eden CG, De Bruin JPC, Corner MA, Feenstra MGP, editors. Progress in brain research: The prefrontal cortex - its structure, function, and pathology. New York, NY: Elsevier Science Publishers; 1990. pp. 325–335. [DOI] [PubMed] [Google Scholar]
- Happe F, Frith U. The neuropsychology of autism. Brain. 1996;119:1377–1400. doi: 10.1093/brain/119.4.1377. [DOI] [PubMed] [Google Scholar]
- Jedema HP, Moghaddam B. Characterization of excitatory amino acid modulation of dopamine release in the prefrontal cortex of conscious rats. J Neurochem. 1996;66:1448–1453. doi: 10.1046/j.1471-4159.1996.66041448.x. [DOI] [PubMed] [Google Scholar]
- Jones MW, Kilpatrick IC, Phillipson OT. Regulation of dopamine function in the prefrontal cortex of the rat by the thalamic mediodorsal nucleus. Brain Res Bull. 1987;19:9–17. doi: 10.1016/0361-9230(87)90159-6. [DOI] [PubMed] [Google Scholar]
- Jones CA, Zempleni E, Davis B, Reynolds GP. Glutamate stimulates dopamine release from cortical and limbic rat brain in vitro. Eur J Pharmacol. 1993;242:183–187. doi: 10.1016/0014-2999(93)90078-v. [DOI] [PubMed] [Google Scholar]
- Kolb B, Whishaw IQ. Fundamentals of human neuropsychology. New York: WH Freeman and Company; 1990. [Google Scholar]
- Konarski JZ, McIntyre RS, Grupp LA, Kennedy SH. Is the cerebellum relevant in the circuitry of neuropsychiatric disorders? J Psychiatry Neurosci. 2005;30:178–186. [PMC free article] [PubMed] [Google Scholar]
- Kuehn BM. CDC: Autism spectrum disorders common. JAMA. 2007;297:940. doi: 10.1001/jama.297.9.940. [DOI] [PubMed] [Google Scholar]
- Kyriakopoulos M, Vyas NS, Barker GJ, Chitnis XA, Frangou S. A diffusion tensor imaging study of white matter in early-onset schizophrenia. Biol Psychiatry. 2008;63:519–523. doi: 10.1016/j.biopsych.2007.05.021. [DOI] [PubMed] [Google Scholar]
- Lee KH, Blaha CD, Cooper S, Hitti FL, Leiter JC, Roberts DW, Kim U. Dopamine efflux in the rat striatum evoked by electrical stimulation of the subthalamic nucleus: Potential mechanism of action in Parkinson’s disease. Eur J Neurosci. 2006;23:1005–1014. doi: 10.1111/j.1460-9568.2006.04638.x. [DOI] [PubMed] [Google Scholar]
- Martin LA, Goldowitz D, Mittleman G. The cerebellum and spatial ability: Dissection of motor and cognitive components with a mouse model system. Eur J Neurosci. 2003;18:2002–2010. doi: 10.1046/j.1460-9568.2003.02921.x. [DOI] [PubMed] [Google Scholar]
- Martin LA, Escher T, Goldowitz D, Mittleman G. A relationship between cerebellar Purkinje cells and spatial working memory demonstrated in a lurcher/chimera mouse model system. Genes Brain Behav. 2004;3:158–166. doi: 10.1111/j.1601-183x.2004.00067.x. [DOI] [PubMed] [Google Scholar]
- Martin LA, Goldowitz D, Mittleman G. Sustained attention in the mouse: A study on the relationship with the cerebellum. Behav Neurosci. 2006;120:477–481. doi: 10.1037/0735-7044.120.2.477. [DOI] [PubMed] [Google Scholar]
- Mesulam MM. Large-scale neurocognitive networks and distributed processing for attention, language, and memory. Ann Neurol. 1990;28:597–613. doi: 10.1002/ana.410280502. [DOI] [PubMed] [Google Scholar]
- Middleton FA, Strick PL. Cerebellar projections to the pre-frontal cortex of the primate. J Neurosci. 2001;21:700–712. doi: 10.1523/JNEUROSCI.21-02-00700.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phillips ML, Drevets WC, Rauch SL, Lane R. Neurobiology of emotion perception II: Implications for major psychiatric disorders. Biol Psychiatry. 2003;54:515–528. doi: 10.1016/s0006-3223(03)00171-9. [DOI] [PubMed] [Google Scholar]
- Pickett J, London E. The neuropathology of autism: A review. J Neuropathol Exp Neurol. 2005;64:925–935. doi: 10.1097/01.jnen.0000186921.42592.6c. [DOI] [PubMed] [Google Scholar]
- Ritvo ER, Freeman BJ, Scheibel AB. Lower Purkinje cell counts in the cerebella of four autistic subjects: Initial findings of the UCLA-NSAC autopsy research report. Am J Psychiatry. 1986;143:862–866. doi: 10.1176/ajp.143.7.862. [DOI] [PubMed] [Google Scholar]
- Schwarz C, Schmitz Y. Projection from the cerebellar lateral nucleus to precerebellar nuclei in the mossy fiber pathway is glutamatergic: A study combining anterograde tracing with immunogold labeling in the rat. J Comp Neurol. 1997;381:320–334. doi: 10.1002/(sici)1096-9861(19970512)381:3<320::aid-cne5>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- Sultan F, Konig T, Mock M, Thier P. Quantitative organization of neurotransmitters in the deep cerebellar nuclei of the Lurcher mutant. J Comp Neurol. 2002;452:311–323. doi: 10.1002/cne.10365. [DOI] [PubMed] [Google Scholar]
- Wetts R, Herrup K. Interaction of granule, Purkinje, and inferior olivary neurons in lurcher chimeric mice. I. Qualitative studies. J Embryo Exp Morphol. 1982;68:87–98. [PubMed] [Google Scholar]
- Zuo J, De Jager PL, Takahashi KA, Jiang W, Linden DJ, Heintz N. Neurodegeneration in Lurcher mice caused by mutation in δ2 glutamate receptor gene. Nature. 1997;388:769–773. doi: 10.1038/42009. [DOI] [PubMed] [Google Scholar]