Abstract
Prenatal stress might increase cardiometabolic disease risk. We measured prenatal stress due to an ice storm in 1998, and measured glucose tolerance among a subsample of 32 exposed adolescents in 2011. Severity of stress was positively associated with insulin secretion, suggesting that prenatal stress independently predicts metabolic outcomes in adolescence.
Keywords: Developmental origins of health and disease, metabolism, pregnancy
Introduction
Prevalence of childhood metabolic disorders has recently escalated dramatically (1). Research in the Developmental Origins of Health and Disease suggests that features of the prenatal environment, such as poor nutrition, might “program” key aspects of growth or metabolism and thereby predispose offspring to adverse cardiometabolic outcomes (2). High levels of prenatal maternal stress (PNMS) can also have long-term programming effects that might contribute to childhood metabolic disorders. PNMS exposure negatively impacts fetal growth, which increases risk for later cardiometabolic diseases, and can also disrupt the fetal hypothalamic pituitary adrenal axis, which is involved in metabolic pathways. Animal studies suggest that prenatal stress or glucocorticoid exposure is associated with alterations in glucose-insulin metabolism such as insulin resistance, hyperglycemia, and hyperinsulinaemia (3, 4). Unfortunately, evidence from humans is limited (5, 6). Retrospective case-control studies indicate increased risk of insulin resistance among adults whose mothers experienced stressors during pregnancy (5). Furthermore, risk of Type 2 Diabetes Mellitus has been shown to be elevated among children and young adults whose mothers experienced bereavement during pregnancy (7). Further human studies are needed to determine what aspect of PNMS, the objective exposure or the subjective distress, is the stronger predictor of metabolic outcomes.
Since 1998, we have studied effects of two components of PNMS (i.e., objective hardship and subjective distress) among children of women who were pregnant during a severe ice storm. The storm affected women randomly regarding socioeconomic status and physical and mental health. Analyses from Project Ice Storm indicate that PNMS due to the storm negatively impacted birth outcomes (8), and objective maternal hardship increased risk for obesity at age 5½ (9). Based on these patterns, we expected effects of PNMS on glucose-insulin metabolism.
Methods
This study was approved by the Research Ethics Board of the Douglas Hospital Research Center. We obtained written informed consent from parents and written informed assent from adolescents.
Project Ice Storm (8–10) includes 176 women who were pregnant during the 1998 Quebec (Canada) ice storm, and their children. In 1998, we assessed PNMS due to the storm using an objective hardship questionnaire, which addressed loss (e.g. damage to residence), scope (e.g. days without electricity), and change (e.g. time in a shelter); and a validated French version of the Impact of Events Scale–Revised (11), which addressed subjective distress due to the storm. We collected demographic and health data for the women, including household socioeconomic status (Hollingshead social position criteria) (12) at recruitment, and maternal anxiety (General Health Questionnaire) (13) and exposure to stressful life events (Life Experiences Survey) (14) at both recruitment and when the children were 13½ years old.
In 2011, we invited families to participate in a study of glucose-insulin metabolism. A subset of 18 boys and 14 girls (mean age 13.4 years) completed the assessment. Their mothers were in their 3rd (n=8), 2nd (n=9), or 1st (n=10) trimester of pregnancy during the storm, or conceived within one month of the storm (n=5) when stress hormones could still be elevated. Participating families did not differ from the rest of the families on any key maternal or child characteristics such as socioeconomic status, levels of objective hardship or subjective distress, or birth weight.
We measured height, weight, and percent body fat (%BF) through air displacement plethysmography; %BF was missing for 4 participants. We collected venous blood samples after an overnight fast, followed by collection 30 minutes after an oral glucose challenge (1.75g/kg, maximum 75g). Adolescents completed the Puberty Development Scale (15), and parents completed a survey regarding family history of diabetes. During a separate assessment period at age 13½, adolescents completed the Life Experiences Survey (14), as well as the Perceived Stress Scale (16), Mental Health Continuum (17), and Eating Attitudes Test (EAT-26) (18).
Assays for fasting and stimulated glucose (G0, G30; mmol/L) and insulin (I0, I30; mU/L) were conducted at St. Mary’s Hospital, Montreal. Insulin secretion was estimated using the insulinogenic index [(I30−I0)/(G30−G0); mU/mmol], one of the best indices for first-phase insulin secretion in youth (19).
Hierarchical linear regression was used to test associations among predictor variables and insulin secretion. We tested models including PNMS (objective or subjective) and key control variables (child’s sex, birth weight, body mass index (BMI, kg/m2), pubertal stage, and number of family members with diabetes) individually, as well as a full model including all key predictor variables. We tested potential sex effects using an interaction term (PNMS×Sex), and tested variations of the model including %BF and maternal gestational diabetes. Finally, we conducted additional analyses to test effects of other prenatal and postnatal characteristics such as life events and perceived stress. Data were analyzed using SPSS 20.0.
Results
No adolescents had diagnosed diabetes or G0≥7.0 (the cutoff for diagnosis). Three outliers for insulin secretion were detected and Winsorized. There were no sex differences in mean objective hardship scores (boys: mean 9.8, SD 3.8; girls: mean 9.1, SD 4.1; p=0.66), subjective distress scores (10.9, 10.5; 9.3, 8.4; p=0.64), birth weight (g) (3289, 657; 3466, 564; p=0.43), pubertal development indices (2.6, 0.4; 2.6, 0.3; p=0.96), BMI (23.2, 6.5; 20.6, 4.1; p=0.21), %BF (23.7, 11.5; 24.1, 5.5; p=0.92), number of relatives with diabetes (1.0, 1.0; 0.6, 0.9; p=0.32), G30 (7.4, 1.7; 7.2, 1.7; p=0.72), I0 (111.8, 139.3; 51.0, 23.8; p=0.12), I30 (595.2, 483.9; 547.6, 349.8; p=0.76), insulin secretion (31.6, 20.9; 25.6, 23.7; p=0.45), or percentage of adolescents exposed to gestational diabetes (11.1; 14.3, Chi-square p=1.00). The only variable exhibiting sex differences was G0, which was higher among boys (5.3, 0.4) than girls (4.8, 0.4) (p<0.01). No variables differed by trimester of exposure.
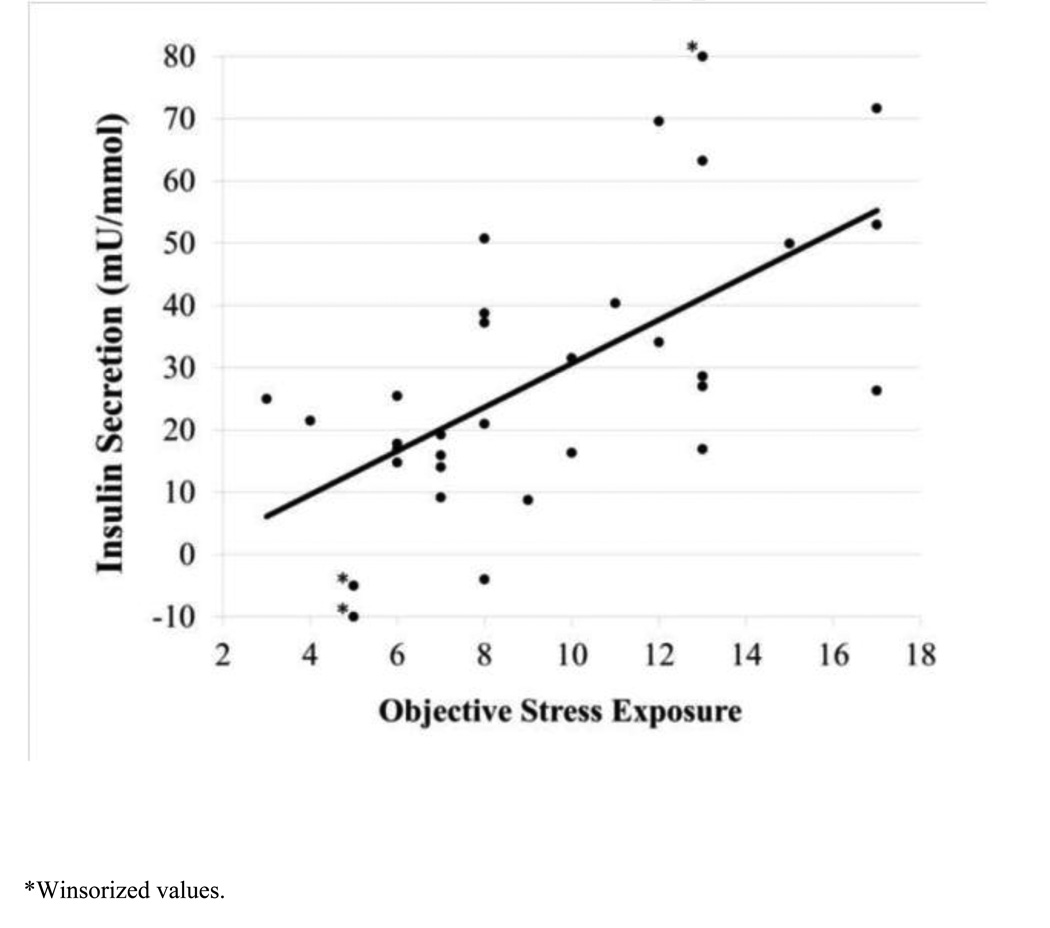
Objective hardship was significantly positively correlated with insulin secretion (r=0.62, p<0.01) (Figure 1), as well as with BMI (r=0.39, p=0.03) and BMI Z-score (r=0.40, p=0.02; based on World Health Organization growth references, (20)), and showed a trend with %BF (r=0.33, p=0.09). In contrast, subjective distress was not significantly correlated with insulin secretion (r=0.15, p=0.42), BMI (r=0.12, p=0.51), BMI Z-score (r=0.13, p=0.47), or %BF (r=− 0.30, p=0.90). Analyses were thus focused on objective hardship. Correlation coefficients (r) for insulin secretion and key independent variables are shown in Table 1.
Figure 1.
Relationship between prenatal maternal stress exposure (objective hardship due to the storm) and insulin secretion (r=0.62, p<0.01)
*Winsorized values.
Table 1.
Relationships among insulin secretiona and key independent variables: Zero-order correlations (r)
| Insulin secretion |
Objective hardship |
BMI | # Fam. w/ Diab. |
Birth weight |
Sex | Pubertal Index |
|
|---|---|---|---|---|---|---|---|
| Insulin secretion | 1 | ||||||
| Objective hardship | 0.62** | 1 | |||||
| BMI | 0.41* | 0.39* | 1 | ||||
| # Fam. w/Diab. | 0.35† | 0.05 | 0.27 | 1 | |||
| Birth weight | −0.26 | −0.05 | 0.21 | 0.07 | 1 | ||
| Sex a | −0.14 | −0.08 | −0.23 | −0.18 | 0.15 | 1 | |
| Pubertal Index | 0.06 | 0.07 | 0.12 | −0.10 | 0.16 | −0.01 | 1 |
Insulinogenic index [(I30−I0)/(G30−G0); mU/mmol]
Boys=1, Girls=2
p<0.01
p<0.05
p<0.1
In regression analyses (Table 2), higher insulin secretion was associated with greater objective hardship (p<0.01) irrespective of which control variables were included in the model. A greater number of family members with diabetes, and lower birth weight, both predicted higher insulin secretion independently of objective hardship. While objective stress alone explained 38.8% of the variance in insulin secretion, the addition of other predictors individually explained up to 10.0% additional variance, with 58.2% explained by the full model.
Table 2.
Relationships among insulin secretiona and key independent variables: Results of linear regression models
| Standardized coefficients | ||||||
|---|---|---|---|---|---|---|
| Independent variable |
Model 1 | Model 2 | Model 3 | Model 4 | Model 5 | Model 6 |
| Objective hardship | 0.55** | 0.61** | 0.61** | 0.62** | 0.62** | 0.52** |
| BMI | 0.19 | 0.18 | ||||
| # Fam. w/Diab. | 0.32* | 0.31* | ||||
| Birth weight | −0.23 | −0.31* | ||||
| Sex b | −0.09 | 0.05 | ||||
| Pubertal Index | 0.02 | 0.08 | ||||
| Model R2 | 0.42 | 0.49 | 0.40 | 0.35 | 0.35 | 0.58 |
Insulinogenic index [(I30−I0)/(G30−G0); mU/mmol]
Boys=1, Girls=2
p<0.01
p<0.05
The addition of the interaction term Objective hardship×Sex indicated no evidence for sex differences in the association between objective hardship and insulin secretion (data not shown). Results were unchanged when controlling for gestational diabetes, or when replacing BMI with %BF (not shown).
We assessed relationships among insulin secretion and a number of additional postnatal household, maternal, and child characteristics. Insulin secretion was unrelated to household socioeconomic status (r=0.26, p=0.16), maternal life events at recruitment(r=−0.01, p=0. 94), and maternal anxiety at recruitment (r=0.11, p=0.56) or at 13½ years (r=−0.14, p=0.47). Furthermore, there were no significant correlations between insulin secretion and adolescents’ scores on the Perceived Stress Scale (r=0.12, p=0.52), Mental Health Continuum (r=−0.09, p=0.64), or life events (r=−0.04, p=0.84). We observed positive correlations between insulin secretion and maternal life events at the 13½-year assessment (r=0.36, p=0.05), as well as adolescents’ EAT-26 scores (r=0.35, p=0.05). Maternal life events did not retain significance in regression models including the key covariates (BMI, number of family members with diabetes, birth weight, sex, pubertal index scores), and results for objective hardship were unchanged (p<0.01). However, EAT-26 scores remained significant in the final regression model (p=0.01) including BMI (p=0.40), number of family members with diabetes (p=0.03), birth weight (p=0.09), sex (p=0.45), pubertal index scores (p=0.87), and objective hardship (p<0.01). Trimming this model of non-significant variables, 34.8% of variance in insulin secretion was explained by covariates (number of family members with diabetes, B=0.33, p<0.01; birth weight, B=−0.30, p=0.02; and EAT-26 scores, B=0.29, p=0.02), and objective hardship accounted for a further 28.4% of variance (B=0.54, p<0.01).
Discussion
Increased insulin secretion is an early feature of insulin resistance (21). The relationship between PNMS due to the ice storm and increased insulin secretion supports recent studies suggesting that PNMS negatively affects metabolic health, and highlights that these effects can be manifest in adolescence. Furthermore, effects were independent of other maternal and child characteristics that might be expected to correlate with insulin secretion. The relationships between insulin secretion and adolescents’ number of family members with diabetes and birth weight follow expected patterns. Furthermore, other researchers have observed positive relationships between EAT-26 scores and obesity among adolescents (22) and young adults (23), consistent with the positive relationship between EAT-26 scores and insulin secretion in the current sample. That the effects of objective hardship remained significant despite small sample sizes, and taking into account these important covariates, lends support to our conclusions.
PNMS might have direct effects on metabolic pathways, as well as indirect effects through early growth patterns or adiposity (5, 6). Exposure to the ice storm was associated with shorter length at birth (8) and with childhood obesity (9), as well as with BMI in the current sample. However, effects on insulin secretion persisted even when controlling for these growth patterns, suggesting potential effects on central mediators of metabolism. Unfortunately, our sample is not large enough to test mediating pathways, and our findings must be replicated in larger samples. Studies of PNMS and cardiometabolic health planned by the Amsterdam Born Children and their Development study (24) could refine our knowledge of underlying mechanisms.
Our results further suggest that it is the woman’s exposure to hardship, rather than her distress, that predicts glucose-insulin metabolism. Other studies indicate differing effects of objective and subjective PNMS depending on the outcome assessed. For example, in Project Ice Storm, we have observed associations between objective hardship, but not subjective distress, and cognitive and linguistic functioning at ages 2 (10) and 5½ (25), and with childhood BMI and obesity (9). In contrast, effects of PNMS on dermatoglyphic asymmetry (26) and head circumference at birth (8) appear to be more strongly related to subjective than objective PNMS. The effects of PNMS likely reflect a number of interacting mechanistic pathways, including hormonal cascades (27), physiological responses such as maternal heart rate change (28), and epigenetic changes (29). Objective hardship and subjective distress might act through different pathways, which could account for the differing effects seen based on type of PNMS. Our results highlight the need for more research in this area.
Considering the lifelong consequences of childhood metabolic disorders (1), studies identifying preventable or treatable risk factors are increasingly necessary. The growing body of evidence suggests that any assistance we can provide pregnant women to reduce stress is important not only for their own health, but also for the long-term metabolic health of their children.
Acknowledgements
This research was supported by a grant from the Canadian Institute of Health Research (CIHR) to Suzanne King and David Laplante. Kelsey Dancause was supported by fellowships from CIHR and the National Institutes of Health (NIH). We are grateful to families for their continued participation in Project Ice Storm, and to Chunbo Yu and Aihua Liu for advice on statistical methods.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
This study was approved by the Research Ethics Board of the Douglas Hospital Research Center.
Conflict of interest statement
The authors declare no conflicts of interest
All authors participated in the design, execution, and analysis of the manuscript, and have seen and approved the final version. The manuscript has neither been published nor submitted elsewhere.
References
- 1.Lee JM, Okumura MJ, Davis MM, Herman WH, Gurney JG. Prevalence and determinants of insulin resistance among U.S. adolescents: a population-based study. Diabetes Care. 2006 Nov;29(11):2427–2432. doi: 10.2337/dc06-0709. Epub 2006/10/27. eng. [DOI] [PubMed] [Google Scholar]
- 2.Entringer S, Buss C, Wadhwa PD. Prenatal stress and developmental programming of human health and disease risk: concepts and integration of empirical findings. Curr Opin Endocrinol Diabetes Obes. 2010 Dec;17(6):507–516. doi: 10.1097/MED.0b013e3283405921. Epub 2010/10/22. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brunton PJ, Sullivan KM, Kerrigan D, Russell JA, Seckl JR, Drake AJ. Sex-specific effects of prenatal stress on glucose homoeostasis and peripheral metabolism in rats. J Endocrinol. 2013;217(2):161–173. doi: 10.1530/JOE-12-0540. Epub 2013/02/23. eng. [DOI] [PubMed] [Google Scholar]
- 4.Lesage J, Del-Favero F, Leonhardt M, Louvart H, Maccari S, Vieau D, et al. Prenatal stress induces intrauterine growth restriction and programmes glucose intolerance and feeding behaviour disturbances in the aged rat. The Journal of endocrinology. 2004 May;181(2):291–296. doi: 10.1677/joe.0.1810291. Epub 2004/05/07. eng. [DOI] [PubMed] [Google Scholar]
- 5.Entringer S, Buss C, Swanson JM, Cooper DM, Wing DA, Waffarn F, et al. Fetal programming of body composition, obesity, and metabolic function: the role of intrauterine stress and stress biology. J Nutr Metab. 2012;2012:632548. doi: 10.1155/2012/632548. Epub 2012/06/02. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rinaudo P, Wang E. Fetal programming and metabolic syndrome. Annu Rev Physiol. 2012;74:107–130. doi: 10.1146/annurev-physiol-020911-153245. Epub 2011/09/14. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li J, Olsen J, Vestergaard M, Obel C, Kristensen JK, Virk J. Prenatal exposure to bereavement and type-2 diabetes: a Danish longitudinal population based study. PLoS ONE. 2012;7(8):e43508. doi: 10.1371/journal.pone.0043508. Epub 2012/09/07. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dancause KN, Laplante D, Oremus C, Fraser S, Brunet A, King S. Disaster-related prenatal maternal stress influences birth outcomes: Project Ice Storm Early Human Development. 2011;87:813–820. doi: 10.1016/j.earlhumdev.2011.06.007. [DOI] [PubMed] [Google Scholar]
- 9.Dancause KN, Laplante DP, Fraser S, Brunet A, Ciampi A, Schmitz N, et al. Prenatal exposure to a natural disaster increases risk for obesity in 5 ½ year old children. Pediatr Res. 2012;71:126–131. doi: 10.1038/pr.2011.18. [DOI] [PubMed] [Google Scholar]
- 10.Laplante DP, Barr RG, Brunet A, Galbaud du Fort G, Meaney ML, Saucier JF, et al. Stress during pregnancy affects general intellectual and language functioning in human toddlers. Pediatric Research. 2004 Sep;56(3):400–410. doi: 10.1203/01.PDR.0000136281.34035.44. Epub 2004/07/09. eng. [DOI] [PubMed] [Google Scholar]
- 11.Brunet A, St-Hilaire A, Jehel L, King S. Validation of a French version of the Impact of Event Scale - Revised. Canadian Journal of Psychiatry. 2003;48:55–60. doi: 10.1177/070674370304800111. [DOI] [PubMed] [Google Scholar]
- 12.Hollingshead AB. Four-factor index of social status. New Haven: Yale University Press; 1973. [Google Scholar]
- 13.Goldberg DP. The Detection of Psychiatric Illness by Questionnaire: A Technique for the Identification and Assessment of Non-Psychiatric Illness. London: Oxford University Press; 1972. [Google Scholar]
- 14.Sarason IG, Johnson JH, Siegel JM. Assessing the impact of life changes: Development of the Life Experience Survey. Journal of Consulting & Clinical Psychology. 1978;46(5):932–946. doi: 10.1037//0022-006x.46.5.932. [DOI] [PubMed] [Google Scholar]
- 15.Petersen AC, Crockett L, Richards M, Boxer A. A self-report measure of pubertal status: Reliability, validity, and initial norms. Journal of Youth and Adolescence. 1988;17(2):117–133. doi: 10.1007/BF01537962. [DOI] [PubMed] [Google Scholar]
- 16.Cohen S, Kamarck T, Mermelstein R. A global measure of perceived stress. Journal of Health & Social Behavior. 1983 Dec;24(4):385–396. 1983. [PubMed] [Google Scholar]
- 17.Keyes CL. The mental health continuum: from languishing to flourishing in life. Journal of health and social behavior. 2002 Jun;43(2):207–222. [PubMed] [Google Scholar]
- 18.Garner DM, Olmsted MP, Bohr Y, Garfinkel PE. The eating attitudes test: psychometric features and clinical correlates. Psychol Med. 1982 Nov;12(4):871–878. doi: 10.1017/s0033291700049163. Epub 1982/11/01. eng. [DOI] [PubMed] [Google Scholar]
- 19.Henderson M, Baillargeon JP, Rabasa-Lhoret R, Chiasson JL, Hanley J, Lambert M. Estimating insulin secretion in youth using simple indices derived from the oral glucose tolerance test. Diabetes Metab. 2012 Mar 23; doi: 10.1016/j.diabet.2012.02.002. Epub 2012/03/27. Eng. [DOI] [PubMed] [Google Scholar]
- 20.World Health Organization. Growth reference data for 5–19 years. Geneva: World Health Organization; 2010. [Google Scholar]
- 21.Weiss R. Impaired glucose tolerance and risk factors for progression to type 2 diabetes in youth. Pediatr Diabetes. 2007 Dec;8(Suppl 9):70–75. doi: 10.1111/j.1399-5448.2007.00336.x. Epub 2007/12/06. eng. [DOI] [PubMed] [Google Scholar]
- 22.Pastore DR, Fisher M, Friedman SB. Abnormalities in weight status, eating attitudes, and eating behaviors among urban high school students: correlations with self-esteem and anxiety. J Adolesc Health. 1996 May;18(5):312–319. doi: 10.1016/1054-139X(95)00321-I. Epub 1996/05/01. eng. [DOI] [PubMed] [Google Scholar]
- 23.Desai MN, Miller WC, Staples B, Bravender T. Risk factors associated with overweight and obesity in college students. J Am Coll Health. 2008 Jul-Aug;57(1):109–114. doi: 10.3200/JACH.57.1.109-114. Epub 2008/08/07. eng. [DOI] [PubMed] [Google Scholar]
- 24.van Dijk AE, van Eijsden M, Stronks K, Gemke RJ, Vrijkotte TG. Cardio-metabolic risk in 5-year-old children prenatally exposed to maternal psychosocial stress: the ABCD study. BMC Public Health. 2010 May 14;10(1):251. doi: 10.1186/1471-2458-10-251. Epub 2010/05/18. Eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Laplante DP, Brunet A, Schmitz N, Ciampi A, King S. Project Ice Storm: Prenatal maternal stress affects cognitive and linguistic functioning in 5½-year-old children. Journal of the American Academy of Child and Adolescent Psychiatry. 2008 Sep;47(9):1063–1072. doi: 10.1097/CHI.0b013e31817eec80. Epub 2008/07/31. eng. [DOI] [PubMed] [Google Scholar]
- 26.King S, Mancini-Marie A, Brunet A, Walker E, Meaney MJ, Laplante DP. Prenatal maternal stress from a natural disaster predicts dermatoglyphic asymmetry in humans. Dev Psychopathol. 2009 Spring;21(2):343–353. doi: 10.1017/S0954579409000364. Epub 2009/04/03. eng. [DOI] [PubMed] [Google Scholar]
- 27.Lazinski MJ, Shea AK, Steiner M. Effects of maternal prenatal stress on offspring development: a commentary Archives of Women's Mental Health. 2008;11:363–375. doi: 10.1007/s00737-008-0035-4. [DOI] [PubMed] [Google Scholar]
- 28.Dipietro JA. Maternal stress in pregnancy: considerations for fetal development. J Adolesc Health. 2012 Aug;51(2 Suppl):S3–S8. doi: 10.1016/j.jadohealth.2012.04.008. Epub 2012/07/20. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Monk C, Spicer J, Champagne FA. Linking prenatal maternal adversity to developmental outcomes in infants: the role of epigenetic pathways. Dev Psychopathol. 2012 Nov;24(4):1361–1376. doi: 10.1017/S0954579412000764. Epub 2012/10/16. eng. [DOI] [PMC free article] [PubMed] [Google Scholar]