Abstract
A critical shortage of donor organs for treating end-stage organ failure highlights the urgent need for generating organs from human induced pluripotent stem cells (iPSCs). Despite many reports describing functional cell differentiation, no studies have succeeded in generating a three-dimensional vascularized organ such as liver. Here we show the generation of vascularized and functional human liver from human iPSCs by transplantation of liver buds created in vitro (iPSC-LBs). Specified hepatic cells (immature endodermal cells destined to track the hepatic cell fate) self-organized into three-dimensional iPSC-LBs by recapitulating organogenetic interactions between endothelial and mesenchymal cells. Immunostaining and gene-expression analyses revealed a resemblance between in vitro grown iPSC-LBs and in vivo liver buds. Human vasculatures in iPSC-LB transplants became functional by connecting to the host vessels within 48 hours. The formation of functional vasculatures stimulated the maturation of iPSC-LBs into tissue resembling the adult liver. Highly metabolic iPSC-derived tissue performed liver-specific functions such as protein production and human-specific drug metabolism without recipient liver replacement. Furthermore, mesenteric transplantation of iPSC-LBs rescued the drug-induced lethal liver failure model. To our knowledge, this is the first report demonstrating the generation of a functional human organ from pluripotent stem cells. Although efforts must ensue to translate these techniques to treatments for patients, this proof-of concept demonstration of organ-bud transplantation provides a promising new approach to study regenerative medicine.
Successful isolation of human embryonic stem cells and, more recently, development of induced pluripotent stem cells (iPSC) has created the ability to generate cells representing almost any lineage with the hope of modeling diseases in vitro, and developing new therapies. This potential has been validated through generation of PSC-derived cells with characteristics of cardiomyocytes, pancreatic beta cells, blood vessels, hematopoietic cells, neurons, and hepatocytes, to name just a few. It is now possible to envisage a time when cells could be generated for transplantation to correct genetic abnormalities or replace damaged parenchymal cells.
Despite significant progress over the last decade in deriving hepatocytes from PSCs, differentiation to a fully mature phenotype has remained elusive. While human iPSC-derived hepatocytes recapitulate many characteristics of adult hepatocytes, some critical ones, such as mature inducible CYP450 metabolizing capacity (e.g. CYP3A4), appropriate responsiveness to hepatic proliferation signals in immune-deficient mouse models, and the ability to correct liver disease have not been demonstrated. Furthermore, most forms of cell therapy, other than hematopoietic stem cell transplantation, have not yet proven to be effective in the clinic, and whether hepatocyte transplantation could treat degenerative liver disease remains questionable. As a result, a major aspiration for PSCs has been generation of donor organs, where limited availability has been a major barrier to transplantation. Towards this end, Takebe et al, in a recent paper in Nature (1), attempted to create an iPSC-derived organ by generating an “embryonic liver bud” in vitro from PSCs. Following transplantation in immune deficient mice, the liver bud-like structure became quickly vascularized and exhibited many human hepatocyte functions for a period of weeks.
Takebe et al generated hepatocyte-specific definitive endoderm, expressing the liver-enriched transcription factor HNF4α, from human iPSC using previously published protocols (2). The resulting cells were then cultured with human umbilical vein endothelial cells (HUVECs) and mesenchymal stem cells (MSCs). Such cells have previously been shown to be important for organogenesis (3, 4), and aggregates formed in culture containing these cells have been shown to improve the survival and physiological function of iPSC–derived cardiomyocytes and pancreatic cells (5, 6). The mixture of cells formed into three-dimensional clusters in vitro, where the iPSC-derived cells stained for AFP and albumin, and expressed many liver-specific genes by qPCR, indicating that cluster formation supported maturation toward a hepatocyte phenotype. The clusters were then implanted into a cranial window, the small bowel mesentery, or under the kidney capsule of immune deficient mice, where they became vascularized within 48 hours (Figure 1). As reported previously following transplantation of embryonic (ED28) porcine liver fragments (7), the engrafted cell clusters formed chimeric vascular connections and exhibited marked proliferation for 2 months in a setting where host liver cells were not induced to divide. FACs analysis revealed that about 4% of the cells stained for both AFP and albumin, while approximately 33% developed a more mature phenotype, staining for albumin only. AFP and albumin was undetectable in 65% of the cells, but whether these cells represented iPSCs that failed to differentiate or were HUVECs or stromal cells was not defined. The engrafted cell clusters secreted human albumin and alpha-1-antitrypsin in the peripheral blood at levels of 1–2 microgram per ml, exhibited human cytochrome P450 activity, and improved the survival of mice in a toxic hepatic injury model. The level of human albumin in the blood of transplanted animals was consistent and 5–10 fold greater than that described in all but one previously published study (8). As dissociation of single hepatocytes from the extracellular matrix can lead to loss of function and reduced survival, the authors, by generating cell clusters incorporating endothelial and mesenchymal cells, induced the iPS-derived cells to mature toward a hepatocyte phenotype, and to engraft, expand, and function in vivo following transplantation at extrahepatic sites.
Figure 1.
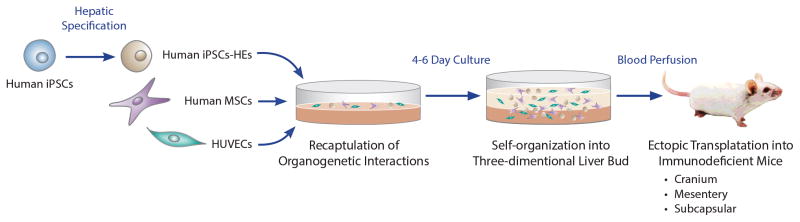
Generation of structures resembling liver buds from human iPSCs. Schematic diagram outlining the strategy used by Takebe et al to generate and implant clusters of endothelial cells, mesenchymal cells, and hepatic progenitors that were derived from human iPSCs (induced pluripotent stem cells). HE, hepatic endoderm; HUVECs, human umbilical vein endothelial cells; MSCs, human mesenchymal stem cells.
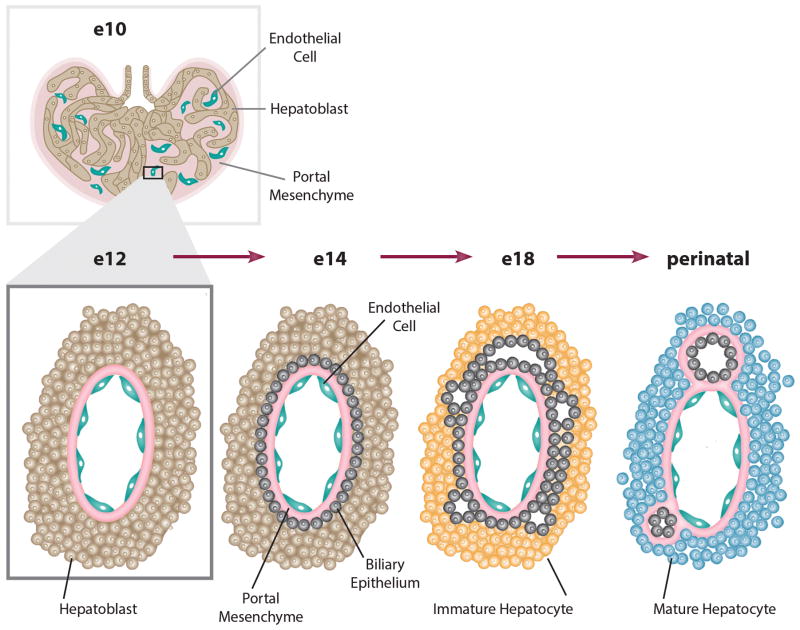
While the findings are encouraging, it is perhaps premature to characterize the engrafted clusters as liver organoids. First, the studies of gene expression and hepatic function, although extensive, did not unequivocally demonstrate that the human iPSC-derived hepatocytes were differentiated any further toward mature hepatocytes than what has been previously published. Second, because the engrafted cell clusters did not develop cholangiocytes or biliary structures (Figure 2), they did not truly generate authentic liver tissue, as do embryonic porcine implants, which initially contain no biliary structures but develop mature biliary cells after transplantation in immune deficient mice (9). Since embryonic porcine liver organogenesis is critically dependent on gestational age at the time of transplantation in immune deficient mice, it is possible that the iPSC-derived hepatic endoderm or the supporting endothelial and mesenchymal cells were insufficiently capable of providing the signals necessary for complete liver development. It is known that extensive development of embryonic tissue is possible following transplantation in some circumstances since peritoneal implantation of embryonic kidney tissue results in the formation of functioning nephrons, and a collecting system that can prolong the survival of anephric rats (10). Finally, although the human iPS-derived clusters improved the survival of mice with severe toxic injury of the liver, it is difficult to extrapolate this result to the clinic. The cause of death from the toxic liver injury was not characterized, correction of any specific liver function by the engrafted cells was not demonstrated, and whether the iPSC-derived hepatocytes responded appropriately to proliferation signals following loss of hepatocyte mass was not tested.
Figure 2.
Embryonic development of the liver. The “liver bud”, as referenced in the paper, resembles the structure of the E10 (mouse) fetal liver. At this developmental stage hepatic progenitor cells migrate into the septum transversum mesenchyme and rudimentary vascular structures begin to develop. The majority of cells within the fetal liver are hematopoietic cells, which are not shown. As illustrated in the figure, if the cells follow a normal developmental process, one would expect the generation of a biliary system as the hepatic progenitor cells differentiate to form both hepatocytes and cholangiocytes.
In summary, the recent findings by Takebe et al offer encouragement for the use of PSC-derived hepatocytes for tissue engineering. Nevertheless is important to recognize that a combination of vasculature, stromal cells, and hepatocytes does not an organ make. Liver architecture, including biliary structures, is critical for the liver’s exocrine function. Liver-like tissues have been generated from primary rodent and human hepatocytes (11, 12), and primary hepatocytes that have colonized lymph nodes can produce enough ectopic liver mass to rescue a mouse from a lethal metabolic disease, indicating that the lymph node contains enough structure for primary cells to support liver tissue and life-sustaining organ function. It may also be important to consider that non-parenchymal cells could have organ–specific characteristics that potentially have unique roles in controlling organ development and function (14). Transplant of iPSC–derived liver buds into the native hepatic environment may therefore prove to be efficacious and could potentially promote cholangiocyte differentiation. The potential to generate organs from iPSCs is exciting and could have substantial ramification for the treatment of liver disease; however, more complete differentiation of iPSCs to a mature hepatic phenotype with the capacity to expand as robustly as primary human hepatocytes will be required. Long-term engraftment in host animals has long been thought to be the most likely way to produce near-complete maturation of iPSC-derived hepatocytes. A significant finding from this study is that, unlike iPSC-derived pancreatic beta cells, the iPSC–derived hepatocytes in the cell clusters failed to completely differentiate following transplantation in immune deficient adult hosts (13). Thus, many important positive and negative lessons can be taken from this work, and, with them, many more questions will need answers.
Acknowledgments
We thank Dr. Jayanta Roy-Chowdhury for careful review of the manuscript and helpful suggests. IJF is supported by RO1 DK48794, R01 DK099320, and PO1 DK096990 from the NIH, and W81XWH-09-1-0658 and W81XWH-11-1-0803 from the DOD and SAD is supported by RO1 DK55743, PO1 HL094857, and UO1 HG006398.
References
- 1.Takebe T, Sekine K, Enomura M, Koike H, Kimura M, Ogaeri T, Zhang RR, et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature. 2013;499:481–484. doi: 10.1038/nature12271. 410.1038/nature12271. Epub 12013 Jul 12273. [DOI] [PubMed] [Google Scholar]
- 2.Si-Tayeb K, Noto FK, Nagaoka M, Li J, Battle MA, Duris C, North PE, et al. Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology. 2010;51:297–305. doi: 10.1002/hep.23354. 210.1002/hep.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Si-Tayeb K, Lemaigre FP, Duncan SA. Organogenesis and development of the liver. Dev Cell. 2010;18:175–189. doi: 10.1016/j.devcel.2010.01.011. 110.1016/j.devcel.2010.1001.1011. [DOI] [PubMed] [Google Scholar]
- 4.Zaret KS, Grompe M. Generation and regeneration of cells of the liver and pancreas. Science. 2008;322:1490–1494. doi: 10.1126/science.1161431. 1410.1126/science.1161431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Banerjee I, Sharma N, Yarmush M. Impact of co-culture on pancreatic differentiation of embryonic stem cells. J Tissue Eng Regen Med. 2011;5:313–323. doi: 10.1002/term.317. 310.1002/term.1317. Epub 2010 Aug 1017. [DOI] [PubMed] [Google Scholar]
- 6.Stevens KR, Kreutziger KL, Dupras SK, Korte FS, Regnier M, Muskheli V, Nourse MB, et al. Physiological function and transplantation of scaffold-free and vascularized human cardiac muscle tissue. Proc Natl Acad Sci U S A. 2009;106:16568–16573. doi: 10.1073/pnas.0908381106. 16510.11073/pnas.0908381106. Epub 0908382009 Sep 0908381117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eventov-Friedman S, Katchman H, Shezen E, Aronovich A, Tchorsh D, Dekel B, Freud E, et al. Embryonic pig liver, pancreas, and lung as a source for transplantation: optimal organogenesis without teratoma depends on distinct time windows. Proc Natl Acad Sci U S A. 2005;102:2928–2933. doi: 10.1073/pnas.0500177102. Epub 2005 Feb 2914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Basma H, Soto-Gutierrez A, Yannam GR, Liu L, Ito R, Yamamoto T, Ellis E, et al. Differentiation and transplantation of human embryonic stem cell-derived hepatocytes. Gastroenterology. 2009;136:990–999. doi: 10.1053/j.gastro.2008.10.047. 910.1053/j.gastro.2008.1010.1047. Epub 2008 Oct 1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Katchman H, Tal O, Eventov-Friedman S, Shezen E, Aronovich A, Tchorsh D, Cohen S, et al. Embryonic porcine liver as a source for transplantation: advantage of intact liver implants over isolated hepatoblasts in overcoming homeostatic inhibition by the quiescent host liver. Stem Cells. 2008;26:1347–1355. doi: 10.1634/stemcells.2007-0631. 1310.1634/stemcells.2007-0631. Epub 2008 Mar 1313. [DOI] [PubMed] [Google Scholar]
- 10.Rogers SA, Hammerman MR. Prolongation of life in anephric rats following de novo renal organogenesis. Organogenesis. 2004;1:22–25. doi: 10.4161/org.1.1.1009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen AA, Thomas DK, Ong LL, Schwartz RE, Golub TR, Bhatia SN. Humanized mice with ectopic artificial liver tissues. Proc Natl Acad Sci U S A. 2011;108:11842–11847. doi: 10.1073/pnas.1101791108. 11810.11073/pnas.1101791108. Epub 1101792011 Jul 1101791111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Komori J, Boone L, DeWard A, Hoppo T, Lagasse E. The mouse lymph node as an ectopic transplantation site for multiple tissues. Nat Biotechnol. 2012;30:976–983. doi: 10.1038/nbt.2379. 910.1038/nbt.2379. Epub 2012 Sep 1023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kroon E, Martinson LA, Kadoya K, Bang AG, Kelly OG, Eliazer S, Young H, et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat Biotechnol. 2008;26:443–452. doi: 10.1038/nbt1393. 410.1038/nbt1393. Epub 2008 Feb 1020. [DOI] [PubMed] [Google Scholar]
- 14.Nolan DJ, Ginsberg M, Israely E, et al. Molecular signatures of tissue-specific microvascular endothelial cell heterogeneity in organ maintenance and regeneration. Dev Cell. 2013;26:204–219. doi: 10.1016/j.devcel.2013.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]