The prevalence of neuropathy in patients with immunoglobulin M (IgM) monoclonal gammopathies ranges from 5% to 31%.1 The most frequent neuropathy is associated with monoclonal IgM reacting with myelin-associated glycoprotein (MAG) and is usually a chronic demyelinating disorder that typically presents with progressive ataxia and painful paresthesias.2 The clinical features of neuropathies associated with Waldenstrom’s macroglobulinemia (WM) and IgM-monoclonal gammopathy of unknown significance are similar.3 Treatment is only warranted in case of significant disability and should not be based on the IgM level or bone marrow infiltration.4 Intravenous immunoglobulin (IVIg), interferon alpha, plasma exchange and immunosuppressive therapy have all been used, but there is no consensus treatment.5 More effective therapy is therefore needed as 30–40% of patients are disabled by severe, progressive neuropathies that undermine their quality of life.6 Several open-label trials of rituximab, a chimeric mouse-human monoclonal antibody directed against the B-cell surface protein CD20, have given encouraging results.7,8 Moreover, even though no randomized controlled trials have provided evidence of improvement in primary outcome measures, several secondary outcomes were improved.9,10 In WM and other indolent B-cell lymphomas, rituximab combined with nucleoside analogs or with nucleoside analogs plus alkylating agents, yields better responses than rituximab monotherapy.4 Based on these results, we wondered whether patients with anti-MAG neuropathy might benefit more from rituximab plus chemotherapy (immunochemotherapy) than from rituximab alone (immunotherapy).
In this retrospective study, we therefore compared our experience with immunochemotherapy and rituximab monotherapy in 45 patients treated for anti-MAG neuropathy at Salpêtrière Hospital, Paris, France, from 1996 to 2011. Apart from symptomatic neuropathy, none of these patients met the criteria for treatment initiation defined by the second WM international workshop. The treatment choice was based on the aggressiveness of the neuropathy and its rate of progression. To evaluate the treatment response we used the Rankin Score (RS) that measures the degree of disability or dependence for daily activities. The scale ranges from 0 to 5, as described in Table 1. Improvement was defined as a 1 point or more decrease in the RS score, stabilization as an unchanged RS, and progression as a 1 point or more increase.
Table 1.
Rankin score

Table 2 shows patients’ characteristics. Nineteen patients received immunochemotherapy. Rituximab was given intravenously at a dose of 375 mg/m2 once every three weeks for 6 cycles. In addition, 8 of these 19 patients received oral cyclophosphamide 300 mg/m2 from Day 1 to 5 plus dexamethasone 20 mg on Day 1 (RDC regimen), 7 patients received oral fludarabine 40 mg/m2 from Day 1 to 5 (RF regimen), and 4 patients received oral fludarabine 40 mg/m2 from Day 1 to 5 plus cyclophosphamide 250 mg/m2 from Day 1 to 3 (RFC regimen). Median follow up was 30 months (range 8–45). Sixteen patients (84%) improved, 2 patients (10%) stabilized and one patient (5%) deteriorated. The median time to response was five months (range 2–20). There was no difference in the median time to response between patients receiving first-line treatment (6 months, range 2–17) and previously treated patients (6.5 months, range 3–20). The base-line Rankin score was similar in the 3 groups. Because of the small size of the groups, however, it was difficult to assess the benefit of each combination individually. One patient relapsed after a median of 24 months. Three patients developed cytopenia requiring blood and platelet transfusion and one patient had repeated infections due to severe hypogammaglobulinemia requiring gammaglobulin replacement therapy.
Table 2.
Patients’ characteristics.
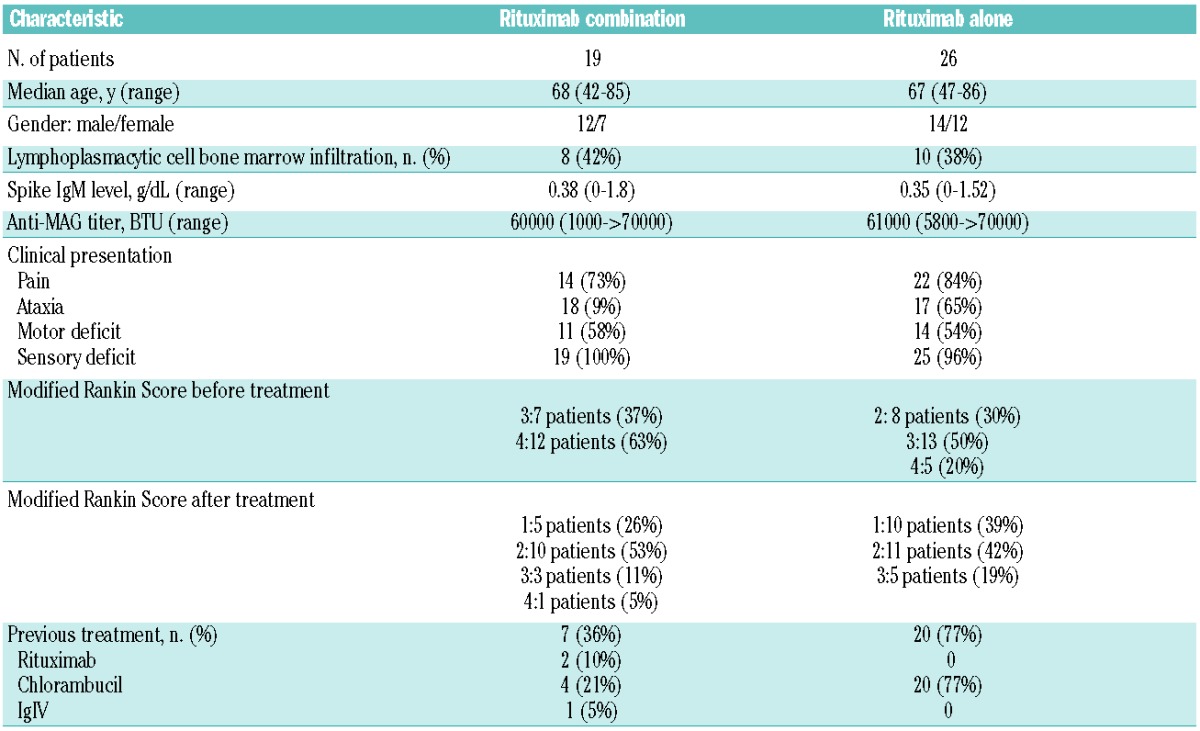
On the other hand, 26 were treated with rituximab alone at a dose of 375 mg/m2, once a week for four weeks. Clinical status was less severe than in the immunochemotherapy group (Table 2). The median follow up of patients treated with rituximab alone was 30 months (range 4–76). Twenty-one patients (80%) improved, 4 (15%) stabilized and one (4%) deteriorated. The median time to response was 9.5 months (range 3–33) overall, 11.9 months (range 6.7–22.1) among patients receiving first-line treatment and 9.7 months in patients previously treated with chlorambucil. One patient relapsed, after a median time of 32 months. No adverse effects were reported.
Interestingly, the median time to response was significantly shorter in the combination therapy group compared to the immunotherapy group, 5 months and 9.5 months, P=0.03. Moreover, anti-MAG titers and IgM level at diagnosis were not associated with disease severity. We observed no significant difference between pre- and post-treatment anti-MAG titers in patients who responded clinically (P=0.64), possibly because we could not measure serum antibody activity at levels above 70,000 BTU. On the contrary, the IgM level fell in patients with a clinical response (P<0.029) and might thus be used as a surrogate marker of treatment efficacy. The electrophysiological evaluations in 23 responder patients confirmed the clinical improvement observed, significant improvements were observed in the mean median nerve distal latencies and in the mean compound muscle amplitude potential of peroneal nerve on tibialis anterior (7.73 ms vs. 7.14 ms, P=0.04 and 1.44 mV vs. 2.37 mV, P=0.02, respectively).
In our study, we found that over 80% of patients responded to both immunotherapy and immunochemotherapy. However, clinical responses were significantly more rapid (median 5 months) with combination therapy than with immunotherapy alone (median 9 months; P=0.03). Patients who did not respond or who relapsed after rituximab alone responded to rituximab plus chemotherapy, confirming the ability of combination therapy to reduce clonal B-cell expansion in anti-MAG neuropathies, as in indolent B-cell lymphoma. However, combination treatments are more myelotoxic, especially in elderly patients with comorbidities, and late adverse effects of chemotherapy, including myelodysplasia/acute myelogenous leukemia, must be taken into account before starting treatment.11,12 These treatments, therefore, must only be used in fit patients with severe, progressive neuropathy.
In conclusion, this retrospective study suggests that rituximab, combined with a purine analog or cyclophosphamide, is effective in patients with severe anti-MAG neuropathy and a Rankin score of 3 or over, and that responses are obtained more rapidly than with rituximab monotherapy. Rituximab combination therapy was well tolerated and may, therefore, be suitable for fit elderly patients requiring rapid disease control.
Footnotes
Information on authorship, contributions, and financial & other disclosures was provided by the authors and is available with the online version of this article at www.haematologica.org.
References
- 1.Steck AJ, Stalder AK, Renaud S. Anti-myelin-associated glycoprotein neuropathy. Curr Opin Neurol. 2006;19(5): 458–63 [DOI] [PubMed] [Google Scholar]
- 2.Chassande B, Leger JM, Younes-Chennoufi AB, Bengoufa D, Maisonobe T, Bouche P, et al. Peripheral neuropathy associated with IgM monoclonal gammopathy: correlations between M-protein antibody activity and clinical/electrophysiological features in 40 cases. Muscle Nerve. 1998;21(1): 55–62 [DOI] [PubMed] [Google Scholar]
- 3.Klein CJ, Moon JS, Mauermann ML, Zeldenrust SR, Wu Y, Dispenzieri A, et al. The neuropathies of Waldenstrom’s macroglobulinemia (WM) and IgM-MGUS. Can J Neurol Sci. 2011;38(2): 289–95 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dimopoulos MA, Gertz MA, Kastritis E, Garcia-Sanz R, Kimby EK, Leblond V, et al. Update on treatment recommendations from the Fourth International Workshop on Waldenstrom’s Macroglobulinemia. J Clin Oncol. 2009;27(1): 120–6 [DOI] [PubMed] [Google Scholar]
- 5.Lunn MP, Nobile-Orazio E. Immunotherapy for IgM anti-myelin-associated glycoprotein paraprotein-associated peripheral neuropathies. Cochrane Database Syst Rev. 2006(2):CD002827. [DOI] [PubMed] [Google Scholar]
- 6.Joint Task Force of the EFNS and the PNS European Federation of Neurological Societies/Peripheral Nerve Society Guideline* on management of paraproteinemic demyelinating neuropathies. Report of a Joint Task Force of the European Federation of Neurological Societies and the Peripheral Nerve Society – first revision. Journal of the Peripheral Nervous System. 2010;15(3): 185–95 [DOI] [PubMed] [Google Scholar]
- 7.Niermeijer JM, Eurelings M, Lokhorst HL, van der Pol WL, Franssen H, Wokke JH, et al. Rituximab for polyneuropathy with IgM monoclonal gammopathy. J Neurol Neurosurg Psychiatry. 2009;80(9): 1036–9 [DOI] [PubMed] [Google Scholar]
- 8.Gruson B, Ghomari K, Beaumont M, Garidi R, Just A, Merle P, et al. Long-term response to rituximab and fludarabine combination in IgM anti-myelin-associated glycoprotein neuropathy. J Peripher Nerv Syst. 2011;16(3): 180–5 [DOI] [PubMed] [Google Scholar]
- 9.Dalakas MC, Rakocevic G, Salajegheh M, Dambrosia JM, Hahn AF, Raju R, et al. Placebo-controlled trial of rituximab in IgM anti-myelin-associated glycoprotein antibody demyelinating neuropathy. Ann Neurol. 2009;65(3): 286–93 [DOI] [PubMed] [Google Scholar]
- 10.Leger JM, Viala K, Nicolas G, Creange A, Vallat JM, Pouget J, et al. Placebo-controlled trial of rituximab in IgM anti-myelin-associated glycoprotein neuropathy. Neurology. 2013;80(24): 2217–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Leleu X, Tamburini J, Roccaro A, Morel P, Soumerai J, Levy V, et al. Balancing risk versus benefit in the treatment of Waldenstrom’s Macroglobulinemia patients with nucleoside analogue-based therapy. Clin Lymphoma Myeloma. 2009;9(1): 71–3 [DOI] [PubMed] [Google Scholar]
- 12.Tam CS, Seymour JF, Prince HM, Kenealy M, Wolf M, Januszewicz EH, et al. Treatment-related myelodysplasia following fludarabine combination chemotherapy. Haematologica. 2006;91(11): 1546–50 [PubMed] [Google Scholar]


