Abstract
Purpose
Autosomal recessive retinitis pigmentosa (arRP) is a genetically heterogeneous disease resulting in progressive loss of photoreceptors that leads to blindness. To date, 36 genes are known to cause arRP, rendering the molecular diagnosis a challenge. The aim of this study was to use homozygosity mapping to identify the causative mutation in a series of inbred families with arRP.
Methods
arRP patients underwent standard ophthalmic examination, Goldman perimetry, fundus examination, retinal OCT, autofluorescence measurement, and full-field electroretinogram. Fifteen consanguineous families with arRP excluded for USH2A and EYS were genotyped on 250 K SNP arrays. Homozygous regions were listed, and known genes within these regions were PCR sequenced. Familial segregation and mutation analyzes were performed.
Results
We found ten mutations, seven of which were novel mutations in eight known genes, including RP1, IMPG2, NR2E3, PDE6A, PDE6B, RLBP1, CNGB1, and C2ORF71, in ten out of 15 families. The patients carrying RP1, C2ORF71, and IMPG2 mutations presented with severe RP, while those with PDE6A, PDE6B, and CNGB1 mutations were less severely affected. The five families without mutations in known genes could be a source of identification of novel genes.
Conclusions
Homozygosity mapping combined with systematic screening of known genes results in a positive molecular diagnosis in 66.7% of families.
Introduction
Photoreceptor degeneration is the leading cause of inherited blindness [1]. This is partly explained by the extreme genetic heterogeneity of these conditions as more than 200 genes are currently registered in the RetNet database, reflecting the vast repertoire of genes necessary for photoreceptor or other retinal cell function. The most frequent clinical entity, nonsyndromic retinitis pigmentosa (RP; OMIM 268000), is also the most genetically heterogeneous with more than 50 disease-causing genes currently associated with this condition. Among these, 36 are known to be responsible for autosomal recessive (ar) inheritance, accounting for 50–60% of all arRP cases [2]. Two major genes are responsible for ar inheritance, USH2A [3,4] and EYS [5-8].
Homozygosity mapping in inbred multiplex families or isolated cases with presumed ar inheritance has proven successful for finding novel genes [9-19] and identifying mutations in previously described genes [20-35]. Homozygosity mapping saves time as it readily highlights regions containing already known disease-causing genes or new genes/loci. This strategy has also been successful in a variable proportion of cases from outbred families who carry a homozygous mutation due to a high level of inbreeding encountered in some populations [21,22,36]. In this study, we applied this strategy to a series of 15 families with consanguineous parents and found that two-thirds of the families carried a mutation in a known arRP gene.
Methods
Patients and clinical investigations
Consanguineous arRP families were selected from 423 families with arRP. Informed written consent and peripheral blood samples were obtained for genetic analysis from all family members according to approved protocols of the Montpellier University Hospital, in agreement with the Declaration of Helsinki.
Patients underwent standard ophthalmologic examination (refractometry, visual acuity, slit-lamp examination, applanation tonometry, funduscopy). Kinetic visual fields were determined with a Goldman perimeter with targets V4e, III4e, and I4e. OCT measurement of the macula was performed using an OCT-3 system (Stratus model 3000; Carl Zeiss Meditec, Dublin, CA) with software version 3.0. Autofluorescence measurements were obtained with the HRA2 Heidelberg retinal confocal angiograph (Heidelberg Engineering, Dossenheim, Germany), and fundus pictures were taken. Full-field ERG was recorded using a Ganzfeld apparatus (Metrovision, Pérenchies, France) with a bipolar contact lens electrode on maximally dilated pupils according to the ISCEV protocol [37].
Single nucleotide polymorphism genotyping and Sanger sequencing of candidate genes
Genomic DNA was isolated from leucocytes using a proteinase K digestion, followed by an ethanol precipitation [38]. DNA samples were quantified by a spectrophotometer, aliquoted and stored at +4 °C and -20 °C until use. From 31 consanguineous families (26 multiplex, five sporadic), 15 families that were not homozygous for EYS and USH2A microsatellite markers were selected and genotyped with 262,270 single nucleotide polymorphisms (SNPs; GeneChip Mapping 250 K Nsp Array; Affymetrix, Santa Clara, CA) at DNAVision, Charleroi, Belgium. Results were analyzed using the common homozygosity regions test of the transmitted allele search engine (TASE) [39]. TASE was designed to screen for common homozygous genotypes in all affected individuals that are heterozygous or wild type in unaffected individuals. Candidate chromosomal regions of homozygosity larger than 2 Mb were compared to the position of known genes and loci for retinal inherited diseases according to the RetNet database. All exons and exon–intron boundaries of the candidate genes were then sequenced. Each PCR was performed in a 25-µl reaction mix containing 50 ng of genomic DNA, 2 mM MgCl2, 200 µM deoxyribo nucleotide triphosphate (dNTPs), 0.2 µM of each primer (designed with Primer 3 software), and 1 U of AmpliTaq Gold DNA Polymerase (Applied Biosystems, Foster City, CA) in a buffer composed of 100 mM Tris-HCl, pH 8.3 and 500 mM KCl. Following the first denaturation at 95 °C for 9 min, amplification was carried out for 35 cycles at 95 °C for 30 s, at the melting temperature (Tm) of the primers (56 °C-60 °C) for 30 sec and at 72 °C for 1 min, ending with a final extension step at 72 °C for 10 min. PCR products were purified with ExoStar 1-step clean up (GE Healthcare, Little Chalfont, UK) and sequenced using the BigDye Terminator cycle sequencing ready reaction kit V3.1 on an Applied Biosystems 3130xL genetic analyzer, following the manufacturer’s instructions. Sequencing results were analyzed with Sequencing Analysis v5.2 software (Applied Biosystems).
Mutation validation
Mutations identified were validated by reading the existing literature, performing familial segregation whenever possible, consulting the Human Gene Mutation Database (HGMD), and interrogating databases with mutation frequencies (1000 genomes, Exome Variant Server [EVS], dbSNP). Missense variations were systematically analyzed using Polyphen-2 and SIFT softwares.
Results
Genotyping and mutation detection
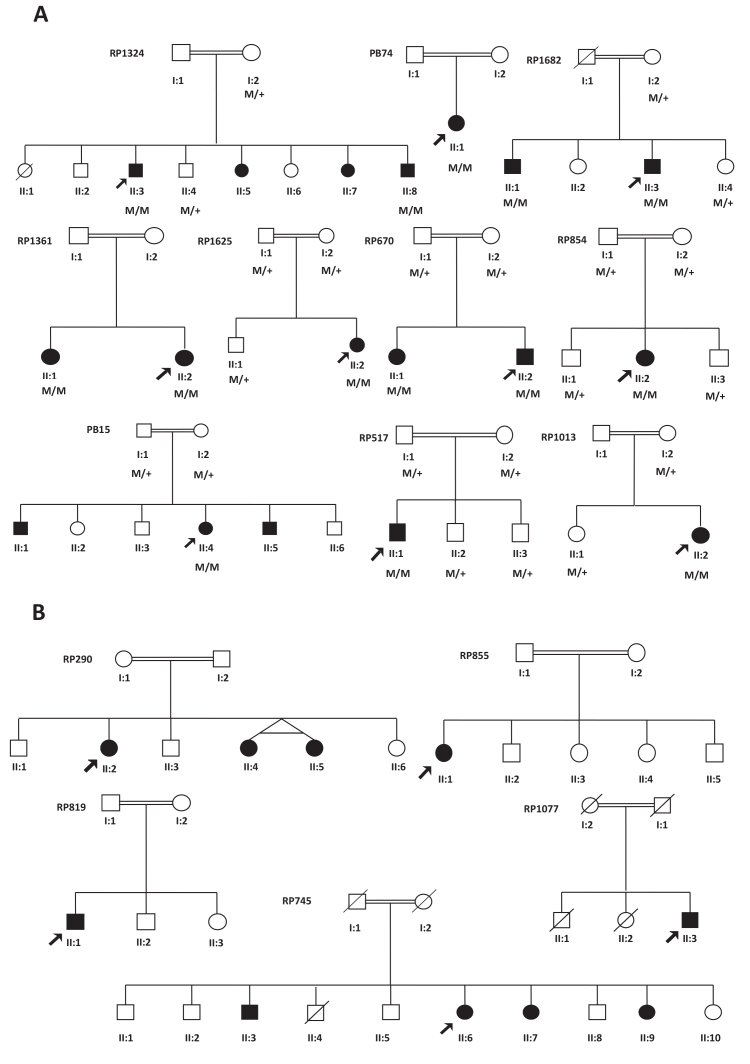
From the 15 consanguineous families, eight families were simplex and seven were multiplex (Figure 1). An average of two SNP arrays per family was performed (range one to three arrays). The quality control (QC) call rate for all samples was always above 90%.
Figure 1.
Pedigrees of 15 consanguineous families with autosomal recessive retinitis pigmentosa. A: Pedigrees of families with mutations in a known RP gene, and co-segregation in available family members. Filled symbols, affected; unfilled, unaffected persons. Squares, males; circles, females; arrows, index patients; M, mutated allele; +, wild-type allele. B: Pedigrees of unsolved families.
For each family, homozygous regions were classified as a function of SNP coverage (number of SNPs/Mb; Table 1). There were on average 7.7 homozygous regions (range 1–18 regions) with a mean size of 15.3 Mb (range 2.2–53 Mb) per family. The homozygous regions were compared with the position of the genes listed in the RetNet database, and the corresponding genes were systematically sequenced, with priority given to known nonsyndromic arRP genes. A total of 32 genes were sequenced for the 15 families. For eight of these families, only one gene was sequenced and this revealed a causative mutation.
Table 1. Homozygous regions and candidate genes found in the studied families.
| Family # | Chromosome | Length* | SNP/Mb | Start | Stop | RANK | Genes |
|---|---|---|---|---|---|---|---|
| PB15 |
15 |
8 |
97 |
94,755,230 |
103,000,000 |
1 |
|
| 3 |
20 |
69 |
81,385,867 |
101,821,068 |
2 |
IMPG2, BBS3 |
|
| 11 |
8 |
48 |
48,019,397 |
56,670,113 |
3 |
||
| 8 |
6 |
18 |
42,604,791 |
49,328,524 |
4 |
||
| PB74 |
7 |
2.8 |
192 |
14,305,994 |
17,109,926 |
1 |
|
| 9 |
2.6 |
147 |
23,912,009 |
26,502,533 |
2 |
||
| 11 |
25.2 |
110 |
8,268,949 |
33,487,251 |
3 |
TEAD1, USH1C |
|
| 6 |
2.4 |
104 |
75,641,951 |
78,011,282 |
4 |
||
| 14 |
24.4 |
99 |
33,161,305 |
57,535,645 |
5 |
OTX2 |
|
| 13 |
20 |
92 |
41,472,025 |
61,458,472 |
6 |
RB1 |
|
| 2 |
11.3 |
77 |
229,188,012 |
240,448,167 |
7 |
SAG, KCNJ13 |
|
| 3 |
30.2 |
73 |
86,088,197 |
116,313,537 |
8 |
IMPG2, BBS3 |
|
| 1 |
2.9 |
47 |
49,428,303 |
52,348,388 |
9 |
||
| 4 |
3.6 |
45 |
0 |
3,586,240 |
10 |
PDE6B |
|
| 11 |
5.6 |
42 |
51,538,030 |
57,168,706 |
11 |
||
| 19 |
2.2 |
25 |
17,232,499 |
19,446,936 |
12 |
||
| 10 |
5.6 |
24 |
37,264,807 |
42,897,610 |
13 |
||
| RP290 |
4 |
22 |
111 |
164,572,108 |
1,886,612,779 |
1 |
|
| 13 |
24 |
110 |
19,611,203 |
43,979,500 |
2 |
||
| 4 |
17 |
109 |
14,126,510 |
31,208,706 |
3 |
PROM1 |
|
| 14 |
18 |
95 |
72,775,668 |
90,693,572 |
4 |
BBS8 |
|
| 11 |
37 |
78 |
27,547,379 |
64,200,379 |
5 |
BEST1, ROM1 |
|
| 9 |
22 |
71 |
115,228,455 |
137,266,704 |
6 |
||
| 11 |
16 |
51 |
64,202,427 |
80,653,055 |
7 |
BBS1, CABP4, EVR4, MYO7A |
|
| RP517 |
18 |
10 |
147 |
64,702,698 |
75,485,034 |
1 |
|
| 10 |
15 |
104 |
102,878,609 |
124,720,093 |
2 |
||
| 15 |
6 |
99 |
88,605,547 |
94,989,511 |
3 |
RLBP1 |
|
| 4 |
27 |
93 |
142,603,130 |
170,091,790 |
4 |
LRAT |
|
| 6 |
5 |
93 |
134,536,698 |
139,731,589 |
5 |
AHI1, PEX7 |
|
| 10 |
42 |
92 |
13,549,280 |
55,658,110 |
6 |
RBP3, PCDH15 |
|
| 10 |
16 |
90 |
73,315,427 |
90,014,680 |
7 |
PCDH21, RGR |
|
| 2 |
19 |
88 |
215,924,486 |
243,717,669 |
8 |
SAG, KCNJ13 |
|
| 2 |
7 |
85 |
161,288,182 |
168,767,322 |
9 |
||
| 7 |
10 |
77 |
139,122,579 |
149,147,283 |
10 |
||
| 16 |
22 |
68 |
54,368,954 |
76,163,081 |
11 |
CNGB1, CDH3 |
|
| RP670 |
15 |
20 |
67 |
70,670,532 |
91,626,291 |
1 |
RLBP1, NR2E3 |
| 12 |
5 |
35 |
33,095,301 |
38,783,937 |
2 |
||
| 11 |
7 |
28 |
47,884,847 |
55,182,977 |
3 |
||
| RP745 |
18 |
15 |
131 |
59,022,364 |
74,333,187 |
1 |
|
| 7 |
13 |
96 |
36,733,691 |
49,999,517 |
2 |
||
| 5 |
27 |
81 |
38,657,850 |
65,932,343 |
3 |
||
| 7 |
32 |
74 |
70,138,345 |
102,504,683 |
4 |
PEX1 |
|
| 7 |
20 |
61 |
50,006,472 |
70,113,409 |
5 |
||
| 11 |
8 |
34 |
47,873,883 |
55,620,859 |
6 |
||
| 17 |
17 |
33 |
14,152,804 |
30,823,007 |
7 |
HRG4/UNC119 |
|
| RP819 |
16 |
7 |
145 |
6,137,184 |
12,877,511 |
1 |
|
| 11 |
9 |
106 |
76,325,821 |
85,436,868 |
2 |
MYO7A |
|
| 3 |
17 |
93 |
100,042,849 |
116,695,553 |
3 |
IMPG2 |
|
| 10 |
11 |
89 |
99,038,933 |
110,389,800 |
4 |
ARL3 |
|
| 5 |
20 |
82 |
72,943,817 |
92,857,172 |
5 |
||
| RP854 |
8 |
7 |
125 |
16,489,491 |
23,287,191 |
1 |
|
| 12 |
6 |
113 |
102,972,070 |
108,720,576 |
2 |
||
| 1 |
9 |
97 |
167,407,517 |
176,453,828 |
3 |
||
| 8 |
24 |
98 |
102,458,515 |
126,789,312 |
4 |
||
| 8 |
13 |
96 |
53,880,054 |
67,044,043 |
5 |
RP1, TTPA |
|
| 2 |
7 |
81 |
183,870,146 |
191,325,255 |
6 |
||
| 2 |
8 |
74 |
26,097,607 |
34,429,224 |
7 |
ZNF513, C2ORF71 |
|
| 5 |
16 |
67 |
38,414,330 |
54,391,555 |
|||
| RP855 |
11 |
12 |
119 |
85,869,911 |
97,383,737 |
1 |
|
| 15 |
5 |
117 |
33,415,288 |
38,491,401 |
2 |
||
| 10 |
19 |
114 |
105,391,725 |
123,911,345 |
3 |
||
| 13 |
6 |
106 |
84,581,120 |
90,263,015 |
4 |
||
| 6 |
8 |
104 |
117,641,016 |
125,607,559 |
5 |
||
| 5 |
23 |
103 |
101,320,305 |
123,846,658 |
6 |
||
| 7 |
15 |
103 |
155,645,303 |
170,683,241 |
7 |
||
| 18 |
9 |
101 |
38,011,865 |
47,209,971 |
8 |
||
| 5 |
5 |
101 |
26,045,009 |
31,423,339 |
9 |
||
| 14 |
14 |
96 |
34,194,336 |
48,552,897 |
10 |
||
| 4 |
25 |
93 |
111,252,455 |
136,022,295 |
11 |
BBS12 |
|
| 11 |
10 |
89 |
110,685,226 |
120,548,318 |
12 |
MFRP |
|
| 2 |
16 |
88 |
148,875,566 |
164,388,639 |
13 |
||
| 10 |
29 |
87 |
34,317,035 |
63,401,422 |
14 |
RBP3, PCDH15 |
|
| 20 |
9 |
87 |
53,622,497 |
63,000,000 |
15 |
PRPF6 |
|
| 6 |
20 |
84 |
23,895,019 |
44,154,325 |
16 |
TULP1, PRPH2 |
|
| 21 |
5 |
73 |
42,506,107 |
48,000,000 |
17 |
||
| 11 |
48 |
72 |
35,991,050 |
83,847,610 |
18 |
BEST1, ROM1, BBS1, CABP4, MYO7A |
|
| RP1013 |
7 |
15 |
112 |
19,386,915 |
34,648,783 |
1 |
KLHL7,PAP1 |
| 5 |
15 |
110 |
106,422,649 |
121,734,393 |
2 |
||
| 16 |
15 |
104 |
1,889,821 |
16,730,604 |
3 |
ABCC6 |
|
| 8 |
7 |
103 |
25,445,326 |
32,079,101 |
4 |
||
| 7 |
9 |
92 |
120,144,923 |
128,748,534 |
5 |
TSPAN12, IMPDH1, OPN1SW |
|
| 2 |
36 |
89 |
188,652,307 |
224,429,597 |
6 |
||
| 8 |
11 |
86 |
58,758,850 |
69,756,364 |
7 |
TTPA |
|
| 3 |
53 |
85 |
68,243,465 |
121,132,268 |
8 |
BBS3, IMPG2 |
|
| 8 |
30 |
85 |
73,152,837 |
102,974,357 |
9 |
PEX2, CNGB3, C8ORF37 |
|
| 5 |
48 |
84 |
26,788,362 |
74,365,757 |
10 |
||
| 7 |
10 |
81 |
137,476,571 |
147,969,602 |
11 |
||
| 11 |
48 |
70 |
32,076,832 |
80,155,054 |
12 |
BEST1, ROM1, BBS1, LRP5 |
|
| 2 |
11 |
68 |
23,712,008 |
34,302,743 |
13 |
ZNF513, C2ORF71 |
|
| RP1077 |
1 |
25 |
106 |
217,257,837 |
241,982,430 |
1 |
|
| 21 |
21 |
98 |
9,764,385 |
31,185,292 |
2 |
||
| 4 |
5 |
20 |
48,094,534 |
53,233,482 |
3 |
||
| 5 |
6 |
16 |
44,334,983 |
50,066,049 |
4 |
||
| 20 |
5 |
10 |
25,506,582 |
30,680,225 |
5 |
||
| 16 |
15 |
4 |
31,905,355 |
46,831,180 |
6 |
||
| 2 |
7 |
3 |
88,965,501 |
96,239,773 |
7 |
||
| 9 |
33 |
2 |
38,703,364 |
71,244,025 |
8 |
||
| RP1324 |
16 |
7 |
100 |
54,776,161 |
62,457,005 |
1 |
CNGB1, BBS2 |
| RP1361 |
5 |
16 |
112 |
145,313,228 |
162,831,477 |
1 |
PDE6A |
| 20 |
11 |
112 |
49,172,235 |
60,352,153 |
2 |
||
| 16 |
16 |
107 |
65,223,172 |
81,678,049 |
3 |
OPA8, CDH3 |
|
| 8 |
25 |
77 |
37,328,843 |
63,110,766 |
4 |
RP1, ADAM9 |
|
| 20 |
22 |
66 |
22,782,904 |
45,359,408 |
5 |
||
| RP1625 |
8 |
18 |
142 |
5,057,818 |
23,559,224 |
1 |
RP1L1 |
| 1 |
13 |
120 |
58,394,231 |
72,327,802 |
2 |
RPE65 |
|
| 4 |
5 |
98 |
31,636,008 |
36,732,290 |
3 |
||
| 7 |
7 |
91 |
123,580,016 |
131,407,605 |
4 |
OPN1SW, IMPDH1 |
|
| 12 |
13 |
90 |
117,118,028 |
130,497,472 |
5 |
||
| 2 |
7 |
79 |
233,929,283 |
241,117,231 |
6 |
SAG |
|
| 8 |
32 |
79 |
40,071,825 |
72,824,945 |
7 |
RP1, TTPA |
|
| 12 |
5 |
30 |
34,142,799 |
39,744,369 |
8 |
||
| 3 |
5 |
27 |
88,365,050 |
93,558,926 |
9 |
||
| 7 |
7 |
17 |
56,665,370 |
63,796,171 |
10 |
||
| RP1682 |
15 |
13 |
89 |
81,632,077 |
95,169,873 |
1 |
RLBP1 |
| 5 | 5 | 74 | 172,682,382 | 177,906,494 | 2 |
For each family, homozygous regions were classified according to the size of the region in Mb, and the coverage (number of SNP per Mb) to give a rank for prioritizing the molecular screening. Each region is then defined by its position on the corresponding chromosome (Start and Stop sections). The causative gene was bolded for families with positive molecular diagnosis. *: In Megabase pair.
We found that ten out of 15 families had a causative homozygous mutation in one of the genes screened (Table 2). In eight of the ten families, segregation analysis could be performed and the results obtained were in accordance with the autosomal recessive inheritance of the disease (Figure 1). Among the ten mutations, three were previously described (one mutation was only described in the dbSNP database). Seven mutations were either nonsense, frameshift, or large deletions and were presumed to lead to loss of protein function.
Table 2. Mutations found in this study.
| Family # | Gene | cDNA change | Protein change | Prediction | Allele frequency | Previously described |
|---|---|---|---|---|---|---|
| PB15 |
IMPG2 |
c.636delA |
p.Glu213ArgfsX17 |
deletion of the last 1012 AA |
0 |
novel |
| PB74 |
PDE6B |
c.1568T>G |
p.Met523Arg |
possibly damaging |
0 |
novel |
| RP517 |
RLBP1 |
DelExons7–9 |
p.Ile176_Phe317del |
deletion of the last 142 AA |
nd* |
[62] |
| RP670 |
NR2E3 |
c.364C>T |
p.Arg122Cys |
probably damaging |
0 |
novel |
| RP854 |
RP1 |
c.3418delGG |
p.Gly1140LysfsX4 |
deletion of the last 1013 AA |
0 |
novel |
| RP1013 |
C2ORF71 |
c.403G>T |
p.E135X |
deletion of the last 1154 AA |
0 |
novel |
| RP1324 |
CNGB1 |
c.2284C>T |
p.Arg762Cys |
probably damaging |
0 |
novel |
| RP1361 |
PDE6A |
c.769C>T |
p.Arg257X |
deletion of the last 603 AA |
0.000093** |
[40] |
| RP1625 |
RP1 |
c.1186C>T |
p.Arg396X |
deletion of the last 1760 AA |
0.000076** |
rs201493928 |
| RP1682 | RLBP1 | c.488insA | p.Ile163AsnfsX1 | deletion of the last 154 AA | 0 | novel |
For each family with a positive molecular result, we indicate the name of the causative gene, the cDNA change, the protein change and its prediction, the allele frequency of the mutation, and when possible the reference of the mutation found. *nd: not determined ** based on Exome Variant Server database
Three mutations were amino acid changes. The c.364C>T (p.Arg122Cys) in NR2E3 is not found in the HGMD database nor in the EVS. It is located two amino acids downstream of the DNA-binding domain of the protein and is considered to be probably damaging with a score of 1.000 by Polyphen-2. The c.2284C>T (p.Arg762Cys) in exon 23 of CNGB1 affects arginine 762 located in the extracellular domain, which is found in two isoforms of the protein but is missing in the third isoform (named GARP2, for glutamic acid-rich protein-2). This amino acid is conserved in all species in the Polyphen-2 software. It is not described in the EVS, 1000 genomes, and HGMD databases. It is predicted to be probably damaging by Polyphen-2 and damaging by SIFT. The c.1568T>G (p.Met523Arg) in exon 12 of PDE6B is located between the cGMP binding domain and the catalytic domain where many mutations known to cause RP are found. This mutation is not described in the EVS, 1000 genomes, and HGMD databases and is predicted to be possibly damaging by Polyphen-2 and damaging by SIFT.
In total, we found that ten out of 15 families, i.e., 66.7% of the sequenced families, had a mutation in a known gene. For the remaining five families (33.3%), all RP genes in homozygous regions were sequenced but no mutation was detected, suggesting that their proband could carry a mutation in a novel gene. We cannot, however, exclude that mutations in noncoding genomic sequences have been missed. These negative families will ultimately undergo exome sequencing.
Clinical findings in families with identified mutations
Clinical description was available for 12 patients from the ten families in which a mutation was found (Appendix 1). The clinical features were variable depending on the gene involved, although all patients had night blindness (not known for II:4 from PB15), retinal vessel attenuation and retinal atrophy in fundus, and strongly decreased ERG responses.
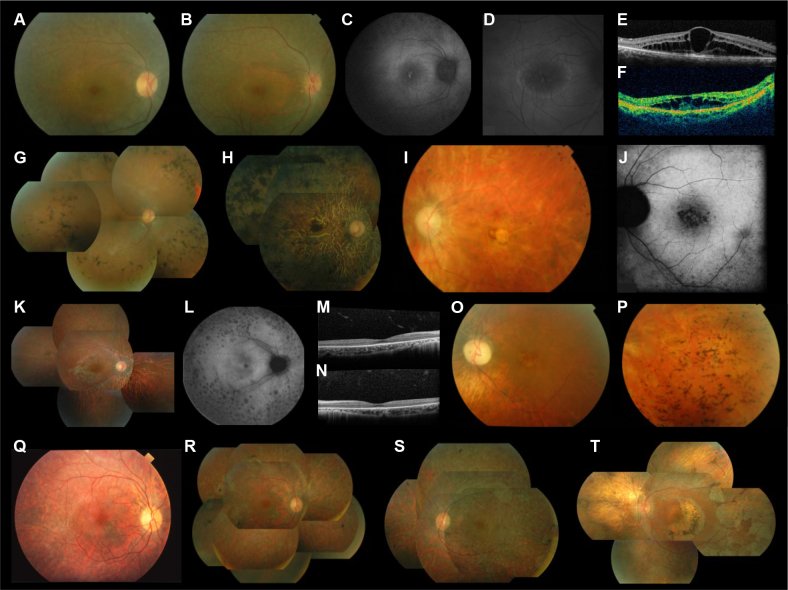
The 19- and 17-year-old II:1 and II:2 sisters from RP1361 had the same clinical presentation, revealing severe RP, which was consistent with the homozygous PDE6A null mutation that they carried as the produced protein is expected to be unstable and degraded by nonsense-mediated decay [40]. They showed few pigment deposits in the fundus (Figure 2A, B) and few atrophic spots in peripheral retina (Figure 2C). The retinal arterioles, however, were already narrow and the macular area showed a typical ring of autofluorescence best seen in patient II:2 (Figure 2D). The sisters also had an important bilateral macular edema (Figure 2E, F) with decreased visual acuity between 0.5 and 0.7. They had no scotopic ERG responses but both still had minimal photopic responses. In comparison, 46-year-old patient II:1 from PB74, who had a missense mutation in PDE6B, retained relatively good visual acuity (0.8 in both eyes). The 44- and 57-year-old II:8 and II:3 brothers from RP1324 also had severe RP due to a missense homozygous mutation in CNGB1. The younger brother had ocular trauma on the left eye in infancy with no light perception, while the contralateral eye had decreased visual acuity at 0.3 (in part due to a cataract as shown by the blurred fundus image [Figure 2G]), bone spicule pigment deposits in the retinal periphery, and narrowing of retinal vessels without atrophy of the optic disc. The IS/OS line was still present in the foveal area. He had tunnel vision at 20–30°, and ERG responses were absent. The elder brother had advanced RP with bare light perception in both eyes, atrophy of both peripheral retina and macula, and large pigment deposits distributed throughout the retina (Figure 2H).
Figure 2.
Clinical features of patients with mutations in autosomal recessive retinitis pigmentosa. A-F: Family RP1361 with PDE6A mutation; fundus photographs of right eyes of subjects II:1 (A) and II:2 (B), retinal autofluorescence in right eyes of subjects II:1 (C) and II:2 (D), OCT scan of the macula of the right eye of subjects II:1 (E) and II:2 (F). G-H: Family RP1324 with CNGB1 mutation; fundus photographs of right eyes of subjects II:8 (G) and II:3 (H). I-J: Family RP1013 with C2ORF71 mutation; fundus photograph (I) and retinal autofluorescence (J) of the left eye of subject II:2. K-P: Families RP1625 (K-N) and RP854 (O, P) with RP1 mutations; fundus photograph (K) and retinal autofluorescence (L) of the right eye of subject II:2 of family RP1625, OCT scan in the macula of the right (M) and left (N) eyes of subject II:2 of family RP1625, fundus photographs of the left eye of subject II:2 of family RP854 showing the macula (O) and the temporal periphery (P). Q-T: Families RP517 (Q-S) and RP1682 (T) with RLBP1 mutations; fundus photographs of the right eye at 32 years (Q) and 40 years (R) and of the left eye at 40 years (S) of subject II:1 of family RP517, and of the left eye (T) of subject II:3 of family RP1682.
The 29-year-old III:2 patient from RP1013 carried a null mutation in C2ORF71, was myopic, and had severe RP; visual acuity was decreased at 0.3 OD and 0.4 OS. Fundus examination showed bilateral, round, foveal atrophy with narrowed retinal vessels and atrophic optic discs (Figure 2I). Retinal autofluorescence testing revealed small atrophic spots grouped in the foveal area (Figure 2J). The visual field was tubular at 30°, and ERG photopic responses were still recordable although very low.
Both patients with a homozygous RP1 mutation also had severe RP, were myopic, and showed bilateral macular involvement. The younger 10-year-old patient II:2 from RP1625 had decreased visual acuity at 0.6 on both eyes. The fundus showed an abnormal foveal reflex, a dark perifoveal area, narrowed retinal arterioles, atrophy of the peripheral retina, and a few small pigment deposits (Figure 2K). Retinal autofluorescence testing revealed many atrophic spots in the peripheral retina and a slightly increased autofluorescence around the fovea (Figure 2L). The outer nuclear layer and the IS/OS were absent except in the foveola where they remained only partly preserved (Figure 2M, N). The patient had tunnel visual field (10–20°), and ERG responses were absent. The older 37-year-old patient III:2 from RP854 had hand motion in both eyes. Fundus examination showed a bilateral round atrophy of the macula, narrowed retinal vessels, atrophic optic discs, and many bone spicule pigment deposits in the retinal periphery (Figure 2O, P). The visual field was undetectable, and ERG responses were absent. The 13-year-old patient from PB15 with a null mutation in IMPG2 also had severe RP with myopia, macular involvement, decreased visual acuity at 0.2 in both eyes, and no ERG response.
Both patients with homozygous RLBP1 mutations had less severe RP than the ten other patients and both had early onset night blindness. The 32-year-old patient from RP517 family had decreased visual acuity in accordance with foveal thinning, but the retinal vessels were moderately narrowed and the optic discs were not atrophic (Figure 2Q). The fundus had a whitish aspect and the retinal periphery showed rare clumps of pigment deposits. When this patient was examined at the age of 40, larger atrophic spots were visible (Figure 2R, S). The 58-year-old II:3 from RP1682 had a similar presentation although with more advanced disease. Visual acuity was still at 0.4 OD and 0.3 OS. The fundus showed large scallop-shaped spots of atrophy covering the mid-periphery of the retina (Figure 2T). There were semicircular atrophic spots around the fovea in both eyes that were secondary to previous laser treatment of macular edema.
Discussion
With the advent of clinical trials for inherited retinal dystrophies, the causative gene needs to be identified. Molecular identification permits the diagnosis of the RP subtype, improved patient follow up, and prediction of disease course. Gene identification is also necessary for gene therapy and to organize patient series for clinical trials. However, molecular diagnosis in arRP, the most genetically heterogeneous form of inherited retinal disease, currently requires screening 36 genes, a process which has never been completed by any research group by Sanger sequencing because it is time and money consuming.
As a preliminary approach to exome sequencing, we used SNP genotyping for homozygosity mapping of consanguineous families and found that two-thirds of the 15 families carried a homozygous causative mutation in a known gene. At the time of our study, it was more economical to perform homozygosity mapping with SNP 250 K arrays to select for families negative for known genes. Presently, the cost of targeted sequencing of RP genes or exome sequencing using next generation sequencing (NGS) is equivalent to that of mapping with SNP arrays, therefore making this approach more affordable.
Given that an average of 13% of arRP cases (range 5% to 18%) may have a mutation in EYS [5-8] and that 16% (range 12% to 20%) may have a mutation in USH2A [3,4], then 76% of patients with arRP are estimated to have a mutation in a known gene, meaning that about a quarter of the arRP patients would have mutations in yet undiscovered genes. In this series, we did not find any homozygous or heterozygous mutations in candidate genes within homozygous regions for the five negative families. These regions represent good candidates to find novel genes by whole exome sequencing. Future analyzes will then be shortened by directly exploring the homozygous regions. Our results showed a high percentage (70%) of novel mutations, indicating that there is considerable allelic heterogeneity in arRP. Similar results were found in a recent study in which 63% of novel mutations were found by targeted sequencing using NGS in a Chinese patient cohort with arRP [41].
The analysis of patient phenotypes showed some variations in disease severity. We found that the three patients with mutations in a connecting cilium gene, i.e., RP1 and C2ORF71, had severe RP with early macular degeneration, while the patients with mutations in a phototransduction gene, i.e., PDE6A, PD6B, and CNGB1, had less severe RP [40,42-45]. Indeed, patients with arRP due to RP1 mutations were frequently reported with legal blindness by their twenties and thirties. Previous case reports described onset in childhood, flat ERG by 18 years, macular involvement before 20 years, or even total blindness before 20 [46-53]. Macular involvement is found earlier in RP due to cilia-associated genes, such as Bardet–Biedl syndrome genes and the recently described ARL2BP gene [54]. Conversely, PDE6A and PDE6B phenotypes show great variation in the severity of disease and frequent macular edema [55]. Yet, it remains difficult to preselect genes for screening based only on macular involvement and severity of the disease.
It is of note that the two patients with a mutation in RLBP1 were not diagnosed as having retinitis punctata albescens (RPA). Apart from typical RPA, RLBP1 mutations have been reported in two subclinical forms of RPA, Bothnia retinal dystrophy [56] and Newfoundland rod–cone dystrophy [57], as well as in rare cases of arRP [58]. In our patients, the specific, small, white, dot-like deposits usually observed on the fundus were not present [59]. It is possible that the dots were present at early stages and had progressively vanished in the course of the disease, thus preventing the correct diagnosis, as previously reported [60]. It is also important to mention that the two patients with this RPA form of arRP had the least severe phenotype among the 12 patients examined. It is known that signs of retinal degeneration (retinal vessel attenuation, optic disc pallor) progress more slowly in RPA than in typical arRP [61].
Acknowledgments
We thank the patients and their families and Vasiliki Kalatzis for critical review of the manuscript. Funding: This work was supported by private foundations (Fondation des Aveugles et Handicapés Visuels de France, Formicoeur, Information Recherche sur la Rétinite Pigmentaire, Retina France, SOS Rétinite, and UNADEV).
Appendix 1. Summary of clinical features.
To access the data, click or select the words “Appendix 1.” *: apparent age at onset; Ch: childhood; fl: flickers; NB: night blindness; nd: not determined; ND: not done; nl: normal; PP: photophobia; PV: peripheral vision impairment; OD: ocular dextra; OS: ocular sinistra; HM: hand motion; LP: light perception.
References
- 1.Hartong DT, Berson EL, Dryja TP. Retinitis pigmentosa. Lancet. 2006;368:1795–809. doi: 10.1016/S0140-6736(06)69740-7. [DOI] [PubMed] [Google Scholar]
- 2.Bocquet B, Lacroux A, Surget MO, Baudoin C, Marquette V, Manes G, Hebrard M, Sénéchal A, Delettre C, Roux AF, Claustres M, Dhaenens CM, Rozet JM, Perrault I, Bonnefont JP, Kaplan J, Dollfus H, Amati-Bonneau P, Bonneau D, Reynier P, Audo I, Zeitz C, Sahel JA, Paquis-Flucklinger V, Calvas P, Arveiler B, Kohl S, Wissinger B, Blanchet C, Meunier I, Hamel CP. Relative Frequencies of Inherited Retinal Dystrophies and Optic Neuropathies in Southern France: Assessment of 21-year Data Management. Ophthalmic Epidemiol. 2013;20:13–25. doi: 10.3109/09286586.2012.737890. [DOI] [PubMed] [Google Scholar]
- 3.McGee TL, Seyedahmadi BJ, Sweeney MO, Dryja TP, Berson EL. Novel mutations in the long isoform of the USH2A gene in patients with Usher syndrome type II or non-syndromic retinitis pigmentosa. J Med Genet. 2010;47:499–506. doi: 10.1136/jmg.2009.075143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lenassi E, Saihan Z, Li Z, Hawlina M, Moore A, Luxon L, Steel K, Bitner-Glindzicz M, Webster A. A comprehensive screen of the USH2A gene in 185 patients with autosomal recessive retinal disease. ARVO 2013. [Google Scholar]
- 5.Hosono K, Ishigami C, Takahashi M, Park DH, Hirami Y, Nakanishi H, Ueno S, Yokoi T, Hikoya A, Fujita T, Zhao Y, Nishina S, Shin JP, Kim IT, Yamamoto S, Azuma N, Terasaki H, Sato M, Kondo M, Minoshima S, Hotta Y. Two novel mutations in the EYS gene are possible major causes of autosomal recessive retinitis pigmentosa in the Japanese population. PLoS ONE. 2012;7:e31036. doi: 10.1371/journal.pone.0031036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barragán I, Borrego S, Pieras JI, González-del Pozo M, Santoyo J, Ayuso C, Baiget M, Millan JM, Mena M, Abd El-Aziz MM, Audo I, Zeitz C, Littink KW, Dopazo J, Bhattacharya SS, Antiñolo G. Mutation spectrum of EYS in Spanish patients with autosomal recessive retinitis pigmentosa. Hum Mutat. 2010;31:E1772–800. doi: 10.1002/humu.21334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Littink KW, van den Born LI, Koenekoop RK, Collin RW, Zonneveld MN, Blokland EA, Khan H, Theelen T, Hoyng CB, Cremers FP, den Hollander AI, Klevering BJ. Mutations in the EYS gene account for approximately 5% of autosomal recessive retinitis pigmentosa and cause a fairly homogeneous phenotype. Ophthalmology. 2010;117:2026–33. doi: 10.1016/j.ophtha.2010.01.040. [DOI] [PubMed] [Google Scholar]
- 8.Audo I, Sahel JA, Mohand-Saïd S, Lancelot ME, Antonio A, Moskova-Doumanova V, Nandrot EF, Doumanov J, Barragan I, Antinolo G, Bhattacharya SS, Zeitz C. EYS is a major gene for rod-cone dystrophies in France. Hum Mutat. 2010;31:E1406–35. doi: 10.1002/humu.21249. [DOI] [PubMed] [Google Scholar]
- 9.Abd El-Aziz MM, Barragan I, O'Driscoll CA, Goodstadt L, Prigmore E, Borrego S, Mena M, Pieras JI, El-Ashry MF, Safieh LA, Shah A, Cheetham ME, Carter NP, Chakarova C, Ponting CP, Bhattacharya SS, Antinolo G. EYS, encoding an ortholog of Drosophila spacemaker, is mutated in autosomal recessive retinitis pigmentosa. Nat Genet. 2008;40:1285–7. doi: 10.1038/ng.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Collin RW, Littink KW, Klevering BJ, van den Born LI, Koenekoop RK, Zonneveld MN, Blokland EA, Strom TM, Hoyng CB, den Hollander AI, Cremers FP. Identification of a 2 Mb human ortholog of Drosophila eyes shut/spacemaker that is mutated in patients with retinitis pigmentosa. Am J Hum Genet. 2008;83:594–603. doi: 10.1016/j.ajhg.2008.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.den Hollander AI, McGee TL, Ziviello C, Banfi S, Dryja TP, Gonzalez-Fernandez F, Ghosh D, Berson EL. A homozygous missense mutation in the IRBP gene (RBP3) associated with autosomal recessive retinitis pigmentosa. Invest Ophthalmol Vis Sci. 2009;50:1864–72. doi: 10.1167/iovs.08-2497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Collin RW, Safieh C, Littink KW, Shalev SA, Garzozi HJ, Rizel L, Abbasi AH, Cremers FP, den Hollander AI, Klevering BJ, Ben-Yosef T. Mutations in C2ORF71 cause autosomal-recessive retinitis pigmentosa. Am J Hum Genet. 2010;86:783–8. doi: 10.1016/j.ajhg.2010.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dvir L, Srour G, Abu-Ras R, Miller B, Shalev SA, Ben-Yosef T. Autosomal-recessive early-onset retinitis pigmentosa caused by a mutation in PDE6G, the gene encoding the gamma subunit of rod cGMP phosphodiesterase. Am J Hum Genet. 2010;87:258–64. doi: 10.1016/j.ajhg.2010.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bandah-Rozenfeld D, Collin RW, Banin E, van den Born LI, Coene KL, Siemiatkowska AM, Zelinger L, Khan MI, Lefeber DJ, Erdinest I, Testa F, Simonelli F, Voesenek K, Blokland EA, Strom TM, Klaver CC, Qamar R, Banfi S, Cremers FP, Sharon D, den Hollander AI. Mutations in IMPG2, encoding interphotoreceptor matrix proteoglycan 2, cause autosomal-recessive retinitis pigmentosa. Am J Hum Genet. 2010;87:199–208. doi: 10.1016/j.ajhg.2010.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Langmann T, Di Gioia SA, Rau I, Stöhr H, Maksimovic NS, Corbo JC, Renner AB, Zrenner E, Kumaramanickavel G, Karlstetter M, Arsenijevic Y, Weber BH, Gal A, Rivolta C. Nonsense mutations in FAM161A cause RP28-associated recessive retinitis pigmentosa. Am J Hum Genet. 2010;87:376–81. doi: 10.1016/j.ajhg.2010.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bandah-Rozenfeld D, Mizrahi-Meissonnier L, Farhy C, Obolensky A, Chowers I, Pe'er J, Merin S, Ben-Yosef T, Ashery-Padan R, Banin E, Sharon D. Homozygosity mapping reveals null mutations in FAM161A as a cause of autosomal-recessive retinitis pigmentosa. Am J Hum Genet. 2010;87:382–91. doi: 10.1016/j.ajhg.2010.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ostergaard E, Batbayli M, Duno M, Vilhelmsen K, Rosenberg T. Mutations in PCDH21 cause autosomal recessive cone-rod dystrophy. J Med Genet. 2010;47:665–9. doi: 10.1136/jmg.2009.069120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zelinger L, Banin E, Obolensky A, Mizrahi-Meissonnier L, Beryozkin A, Bandah-Rozenfeld D, Frenkel S, Ben-Yosef T, Merin S, Schwartz SB, Cideciyan AV, Jacobson SG, Sharon D. A missense mutation in DHDDS, encoding dehydrodolichyl diphosphate synthase, is associated with autosomal-recessive retinitis pigmentosa in Ashkenazi Jews. Am J Hum Genet. 2011;88:207–15. doi: 10.1016/j.ajhg.2011.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Estrada-Cuzcano A, Neveling K, Kohl S, Banin E, Rotenstreich Y, Sharon D, Falik-Zaccai TC, Hipp S, Roepman R, Wissinger B, Letteboer SJ, Mans DA, Blokland EA, Kwint MP, Gijsen SJ, van Huet RA, Collin RW, Scheffer H, Veltman JA, Zrenner E, European Retinal Disease Consortium den Hollander AI, Klevering BJ, Cremers FP. Mutations in C8orf37, encoding a ciliary protein, are associated with autosomal-recessive retinal dystrophies with early macular involvement. Am J Hum Genet. 2012;90:102–9. doi: 10.1016/j.ajhg.2011.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kondo H, Qin M, Mizota A, Kondo M, Hayashi H, Hayashi K, Oshima K, Tahira T, Hayashi K. A homozygosity-based search for mutations in patients with autosomal recessive retinitis pigmentosa, using microsatellite markers. Invest Ophthalmol Vis Sci. 2004;45:4433–9. doi: 10.1167/iovs.04-0544. [DOI] [PubMed] [Google Scholar]
- 21.den Hollander AI, Lopez I, Yzer S, Zonneveld MN, Janssen IM, Strom TM, Hehir-Kwa JY, Veltman JA, Arends ML, Meitinger T, Musarella MA, van den Born LI, Fishman GA, Maumenee IH, Rohrschneider K, Cremers FP, Koenekoop RK. Identification of novel mutations in patients with Leber congenital amaurosis and juvenile RP by genome-wide homozygosity mapping with SNP microarrays. Invest Ophthalmol Vis Sci. 2007;48:5690–8. doi: 10.1167/iovs.07-0610. [DOI] [PubMed] [Google Scholar]
- 22.Harville HM, Held S, Diaz-Font A, Davis EE, Diplas BH, Lewis RA, Borochowitz ZU, Zhou W, Chaki M, MacDonald J, Kayserili H, Beales PL, Katsanis N, Otto E, Hildebrandt F. Identification of 11 novel mutations in eight BBS genes by high-resolution homozygosity mapping. J Med Genet. 2010;47:262–7. doi: 10.1136/jmg.2009.071365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Abu Safieh L, Aldahmesh MA, Shamseldin H, Hashem M, Shaheen R, Alkuraya H, Al Hazzaa SA, Al-Rajhi A, Alkuraya FS. Clinical and molecular characterisation of Bardet-Biedl syndrome in consanguineous populations: the power of homozygosity mapping. J Med Genet. 2010;47:236–41. doi: 10.1136/jmg.2009.070755. [DOI] [PubMed] [Google Scholar]
- 24.Littink KW, Koenekoop RK, van den Born LI, Collin RW, Moruz L, Veltman JA, Roosing S, Zonneveld MN, Omar A, Darvish M, Lopez I, Kroes HY, van Genderen MM, Hoyng CB, Rohrschneider K, van Schooneveld MJ, Cremers FP, den Hollander AI. Homozygosity mapping in patients with cone-rod dystrophy: novel mutations and clinical characterizations. Invest Ophthalmol Vis Sci. 2010;51:5943–51. doi: 10.1167/iovs.10-5797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Collin RW, van den Born LI, Klevering BJ, de Castro-Miró M, Littink KW, Arimadyo K, Azam M, Yazar V, Zonneveld MN, Paun CC, Siemiatkowska AM, Strom TM, Hehir-Kwa JY, Kroes HY, de Faber JT, van Schooneveld MJ, Heckenlively JR, Hoyng CB, den Hollander AI, Cremers FP. High-resolution homozygosity mapping is a powerful tool to detect novel mutations causative of autosomal recessive RP in the Dutch population. Invest Ophthalmol Vis Sci. 2011;52:2227–39. doi: 10.1167/iovs.10-6185. [DOI] [PubMed] [Google Scholar]
- 26.Siemiatkowska AM, Arimadyo K, Moruz LM, Astuti GD, de Castro-Miro M, Zonneveld MN, Strom TM, de Wijs IJ, Hoefsloot LH, Faradz SM, Cremers FP, denHollander AI, Collin RW. Molecular genetic analysis of retinitis pigmentosa in Indonesia using genome-wide homozygosity mapping. Mol Vis. 2011;17:3013–24. [PMC free article] [PubMed] [Google Scholar]
- 27.Ksantini M, Lafont E, Bocquet B, Meunier I, Hamel CP. Homozygous mutation in MERTK causes severe autosomal recessive retinitis pigmentosa. Eur J Ophthalmol. 2012;22:647–53. doi: 10.5301/ejo.5000096. [DOI] [PubMed] [Google Scholar]
- 28.Rizel L, Safieh C, Shalev SA, Mezer E, Jabaly-Habib H, Ben-Neriah Z, Chervinsky E, Briscoe D, Ben-Yosef T. Novel mutations of MYO7A and USH1G in Israeli Arab families with Usher syndrome type 1. Mol Vis. 2011;17:3548–55. [PMC free article] [PubMed] [Google Scholar]
- 29.Kannabiran C, Singh H, Sahini N, Jalali S, Mohan G. Mutations in TULP1, NR2E3, and MFRP genes in Indian families with autosomal recessive retinitis pigmentosa. Mol Vis. 2012;18:1165–74. [PMC free article] [PubMed] [Google Scholar]
- 30.Ajmal M, Khan MI, Micheal S, Ahmed W, Shah A, Venselaar H, Bokhari H, Azam A, Waheed NK, Collin RW, den Hollander AI, Qamar R, Cremers FP. Identification of recurrent and novel mutations in TULP1 in Pakistani families with early-onset retinitis pigmentosa. Mol Vis. 2012;18:1226–37. [PMC free article] [PubMed] [Google Scholar]
- 31.Avila-Fernandez A, Corton M, Nishiguchi KM, Muñoz-Sanz N, Benavides-Mori B, Blanco-Kelly F, Riveiro-Alvarez R, Garcia-Sandoval B, Rivolta C, Ayuso C. Identification of an RP1 prevalent founder mutation and related phenotype in Spanish patients with early-onset autosomal recessive retinitis. Ophthalmology. 2012;119:2616–21. doi: 10.1016/j.ophtha.2012.06.033. [DOI] [PubMed] [Google Scholar]
- 32.Cohen B, Chervinsky E, Jabaly-Habib H, Shalev SA, Briscoe D, Ben-Yosef T. A novel splice site mutation of CDHR1 in a consanguineous Israeli Christian Arab family segregating autosomal recessive cone-rod dystrophy. Mol Vis. 2012;18:2915–21. [PMC free article] [PubMed] [Google Scholar]
- 33.Davidson AE, Sergouniotis PI, Mackay DS, Wright GA, Waseem NH, Michaelides M, Holder GE, Robson AG, Moore AT, Plagnol V, Webster AR. RP1L1 variants are associated with a spectrum of inherited retinal diseases including retinitis pigmentosa and occult macular dystrophy. Hum Mutat. 2013;34:506–14. doi: 10.1002/humu.22264. [DOI] [PubMed] [Google Scholar]
- 34.Beryozkin A, Zelinger L, Bandah-Rozenfeld D, Harel A, Strom TA, Merin S, Chowers I, Banin E, Sharon D. Mutations in CRB1 are a relatively common cause of autosomal recessive early-onset retinal degeneration in the Israeli and Palestinian populations. Invest Ophthalmol Vis Sci. 2013;54:2068–75. doi: 10.1167/iovs.12-11419. [DOI] [PubMed] [Google Scholar]
- 35.Roosing S, van den Born LI, Hoyng CB, Thiadens AA, de Baere E, Collin RW, Koenekoop RK, Leroy BP, van Moll-Ramirez N, Venselaar H, Riemslag FC, Cremers FP, Klaver CC, den Hollander AI. Maternal Uniparental Isodisomy of Chromosome 6 Reveals a TULP1 Mutation as a Novel Cause of Cone Dysfunction. Ophthalmology. 2013;120:1239–46. doi: 10.1016/j.ophtha.2012.12.005. [DOI] [PubMed] [Google Scholar]
- 36.Hildebrandt F, Heeringa SF, Rüschendorf F, Attanasio M, Nürnberg G, Becker C, Seelow D, Huebner N, Chernin G, Vlangos CN, Zhou W, O'Toole JF, Hoskins BE, Wolf MT, Hinkes BG, Chaib H, Ashraf S, Schoeb DS, Ovunc B, Allen SJ, Vega-Warner V, Wise E, Harville HM, Lyons RH, Washburn J, Macdonald J, Nürnberg P, Otto EA.A systematic approach to mapping recessive disease genes in individuals from outbred populations. PLoS Genet 20095e1000353Epub 2009 Jan 23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Marmor MF, Holder GE, Seeliger MW, Yamamoto S. Standard for clinical electroretinography (2004 update). Doc Ophthalmol. 2004;108:107–14. doi: 10.1023/b:doop.0000036793.44912.45. [DOI] [PubMed] [Google Scholar]
- 38.Miller SA, Dykes DD, Polesky HF. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988;16:1215. doi: 10.1093/nar/16.3.1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hebrard M, Manes G, Bocquet B, Meunier I, Coustes-Chazalette D, Hérald E, Sénéchal A, Bolland-Augé A, Zelenika D, Hamel CP. Combining gene mapping and phenotype assessment for fast mutation finding in non-consanguineous autosomal recessive retinitis pigmentosa families. Eur J Hum Genet. 2011;19:1256–63. doi: 10.1038/ejhg.2011.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Riazuddin SA, Zulfiqar F, Zhang Q, Yao W, Li S, Jiao X, Shahzadi A, Amer M, Iqbal M, Hussnain T, Sieving PA, Riazuddin S, Hejtmancik JF. Mutations in the gene encoding the alpha-subunit of rod phosphodiesterase in consanguineous Pakistani families. Mol Vis. 2006;12:1283–91. [PubMed] [Google Scholar]
- 41.Fu Q, Wang F, Wang H, Xu F, Zaneveld JE, Ren H, Keser V, Lopez I, Tuan HF, Salvo JS, Wang X, Zhao L, Wang K, Li Y, Koenekoop RK, Chen R, Sui R. Next-generation sequencing-based molecular diagnosis of a Chinese patient cohort with autosomal recessive retinitis pigmentosa. Invest Ophthalmol Vis Sci. 2013;54:4158–66. doi: 10.1167/iovs.13-11672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dryja TP, Rucinski DE, Chen SH, Berson EL. Frequency of mutations in the gene encoding the alpha subunit of rod cGMP-phosphodiesterase in autosomal recessive retinitis pigmentosa. Invest Ophthalmol Vis Sci. 1999;40:1859–65. [PubMed] [Google Scholar]
- 43.Tsang SH, Tsui I, Chou CL, Zernant J, Haamer E, Iranmanesh R, Tosi J, Allikmets R. A novel mutation and phenotypes in phosphodiesterase 6 deficiency. Am J Ophthalmol. 2008;146:780–8. doi: 10.1016/j.ajo.2008.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jacobson SG, Sumaroka A, Aleman TS, Cideciyan AV, Danciger M, Farber DB. Evidence for retinal remodelling in retinitis pigmentosa caused by PDE6B mutation. Br J Ophthalmol. 2007;91:699–701. doi: 10.1136/bjo.2006.104463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ali S, Riazuddin SA, Shahzadi A, Nasir IA, Khan SN, Husnain T, Akram J, Sieving PA, Hejtmancik JF, Riazuddin S. Mutations in the β-subunit of rod phosphodiesterase identified in consanguineous Pakistani families with autosomal recessive retinitis pigmentosa. Mol Vis. 2011;17:1373–80. [PMC free article] [PubMed] [Google Scholar]
- 46.Riazuddin SA, Zulfiqar F, Zhang Q, Sergeev YV, Qazi ZA, Husnain T, Caruso R, Riazuddin S, Sieving PA, Hejtmancik JF. Autosomal recessive retinitis pigmentosa is associated with mutations in RP1 in three consanguineous Pakistani families. Invest Ophthalmol Vis Sci. 2005;46:2264–70. doi: 10.1167/iovs.04-1280. [DOI] [PubMed] [Google Scholar]
- 47.Khaliq S, Abid A, Ismail M, Hameed A, Mohyuddin A, Lall P, Aziz A, Anwar K, Mehdi SQ. Novel association of RP1 gene mutations with autosomal recessive retinitis pigmentosa. J Med Genet. 2005;42:436–8. doi: 10.1136/jmg.2004.024281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Singh HP, Jalali S, Narayanan R, Kannabiran C. Genetic analysis of Indian families with autosomal recessive retinitis pigmentosa by homozygosity screening. Invest Ophthalmol Vis Sci. 2009;50:4065–71. doi: 10.1167/iovs.09-3479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chen LJ, Lai TY, Tam PO, Chiang SW, Zhang X, Lam S, Lai RY, Lam DS, Pang CP. Compound heterozygosity of two novel truncation mutations in RP1 causing autosomal recessive retinitis pigmentosa. Invest Ophthalmol Vis Sci. 2010;51:2236–42. doi: 10.1167/iovs.09-4437. [DOI] [PubMed] [Google Scholar]
- 50.Zhang X, Chen LJ, Law JP, Lai TY, Chiang SW, Tam PO, Chu KY, Wang N, Zhang M, Pang CP. Differential pattern of RP1 mutations in retinitis pigmentosa. Mol Vis. 2010;16:1353–60. [PMC free article] [PubMed] [Google Scholar]
- 51.Al-Rashed M, Abu Safieh L, Alkuraya H, Aldahmesh MA, Alzahrani J, Diya M, Hashem M, Hardcastle AJ, Al-Hazzaa SA, Alkuraya FS. RP1 and retinitis pigmentosa: report of novel mutations and insight into mutational mechanism. Br J Ophthalmol. 2012;96:1018–22. doi: 10.1136/bjophthalmol-2011-301134. [DOI] [PubMed] [Google Scholar]
- 52.Avila-Fernandez A, Corton M, Nishiguchi KM, Muñoz-Sanz N, Benavides-Mori B, Blanco-Kelly F, Riveiro-Alvarez R, Garcia-Sandoval B, Rivolta C, Ayuso C. Identification of an RP1 prevalent founder mutation and related phenotype in Spanish patients with early-onset autosomal recessive retinitis. Ophthalmology. 2012;119:2616–21. doi: 10.1016/j.ophtha.2012.06.033. [DOI] [PubMed] [Google Scholar]
- 53.Siemiatkowska AM, Astuti GD, Arimadyo K, den Hollander AI, Faradz SM, Cremers FP, Collin RW. Identification of a novel nonsense mutation in RP1 that causes autosomal recessive retinitis pigmentosa in an Indonesian family. Mol Vis. 2012;18:2411–9. [PMC free article] [PubMed] [Google Scholar]
- 54.Davidson AE, Schwarz N, Zelinger L, Stern-Schneider G, Shoemark A, Spitzbarth B, Gross M, Laxer U, Sosna J, Sergouniotis PI, Waseem NH, Wilson R, Kahn RA, Plagnol V, Wolfrum U, Banin E, Hardcastle AJ, Cheetham ME, Sharon D, Webster AR. Mutations in ARL2BP, Encoding ADP-Ribosylation-Factor-Like 2 Binding Protein, Cause Autosomal-Recessive Retinitis Pigmentosa. Am J Hum Genet. 2013;93:321–9. doi: 10.1016/j.ajhg.2013.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tsang SH, Tsui I, Chou CL, Zernant J, Haamer E, Iranmanesh R, Tosi J, Allikmets R. A novel mutation and phenotypes in phosphodiesterase 6 deficiency. Am J Ophthalmol. 2008;146:780–8. doi: 10.1016/j.ajo.2008.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Burstedt M, Jonsson F, Köhn L, Burstedt M, Kivitalo M, Golovleva I. Genotype-phenotype correlations in Bothnia dystrophy caused by RLBP1 gene sequence variations. Acta Ophthalmol (Copenh) 2013;91:437–44. doi: 10.1111/j.1755-3768.2012.02431.x. [DOI] [PubMed] [Google Scholar]
- 57.Eichers ER, Green JS, Stockton DW, Jackman CS, Whelan J, McNamara JA, Johnson GJ, Lupski JR, Katsanis N. Newfoundland rod-cone dystrophy, an early-onset retinal dystrophy, is caused by splice-junction mutations in RLBP1. Am J Hum Genet. 2002;70:955–64. doi: 10.1086/339688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Singh HP, Jalali S, Narayanan R, Kannabiran C. Genetic analysis of Indian families with autosomal recessive retinitis pigmentosa by homozygosity screening. Invest Ophthalmol Vis Sci. 2009;50:4065–71. doi: 10.1167/iovs.09-3479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Burstedt MS, Sandgren O, Golovleva I, Wachtmeister L. Effects of prolonged dark adaptation in patients with retinitis pigmentosa of Bothnia type: an electrophysiological study. Doc Ophthalmol. 2008;116:193–205. doi: 10.1007/s10633-007-9082-9. [DOI] [PubMed] [Google Scholar]
- 60.Burstedt MS, Forsman-Semb K, Golovleva I, Janunger T, Wachtmeister L, Sandgren O. Ocular phenotype of bothnia dystrophy, an autosomal recessive retinitis pigmentosa associated with an R234W mutation in the RLBP1 gene. Arch Ophthalmol. 2001;119:260–7. [PubMed] [Google Scholar]
- 61.Dessalces E. Béatrice Bocquet, Jérôme Bourien, Xavier Zanlonghi, Robert Verdet, Isabelle Meunier, and Christian P. Hamel. Early onset foveal involvement in retinitis punctata albescens with mutations in RLBP1. JAMA Ophthalmol. 2013;131:1314. doi: 10.1001/jamaophthalmol.2013.4476. : - [DOI] [PubMed] [Google Scholar]
- 62.Humbert G, Delettre C, Sénéchal A, Bazalgette C, Barakat A, Bazalgette C, Arnaud B, Lenaers G, Hamel CP. Homozygous deletion related to Alu repeats in RLBP1 causes retinitis punctata albescens. IOVS. 2006;47:4719–24. doi: 10.1167/iovs.05-1488. [DOI] [PubMed] [Google Scholar]