Abstract
Optogenetics is a research field that uses gene therapy to deliver a gene encoding a light-activated protein to cells providing light-regulated control of targeted cell pathways. The technology is a popular tool in many fields of neuroscience, used to transiently switch cells on and off, for example, to map neural circuits. In inherited retinal degenerative diseases, where loss of vision results from the loss of photoreceptors, optogenetics can be applied to either augment the function of surviving photoreceptors or confer light sensitivity to naturally nonlight sensitive retinal cells, such as a bipolar cells. This can be achieved either by the light sensitive protein integrating with native internal signaling pathways, or by using a dual function membrane protein that integrates light signaling with an ion channel or pump activity. Exposing treated cells to light of the correct wavelength activates the protein, resulting in cellular depolarization or hyperpolarization that triggers neurological signaling to the visual cortex.
While there is a lot of interest in optogenetics as a pan-disease clinical treatment for end-stage application in the inherited degenerative diseases of the retina, research to date has been limited to nonhuman clinical studies. To address the clinical translational needs of this technology, the Foundation Fighting Blindness and Massachusetts Eye and Ear Infirmary cohosted an International Optogenetic Therapies for Vision Workshop, which was held at Massachusetts Eye and Ear Infirmary, Boston, Massachusetts on June 1, 2012.
Keywords: optogenetics, vision, retina, gene therapy
Introduction
The first International Optogenetic Therapies for Vision Workshop was held at Massachusetts Eye and Ear Infirmary, Boston, Massachusetts on June 1, 2012 and streamed live over the internet to registered participants for the widest audience. The meeting was triggered, at least in part, by two companies, RetroSense Therapeutics LLC, Ann Arbor, MI and Eos Neurosciences, Los Angeles, CA, that had approached the Foundation Fighting Blindness for funding support to conduct investigative new drug (IND)-enabling studies leading to phase I human clinical trials using optogenetic therapy. Following partial support from the Foundation, RetroSense Therapeutics LLC had recently held a pre-IND meeting with the Food and Drug Administration (FDA) to understand the regulatory path forward, and the Foundation sought to share and discuss pertinent information arising from the meeting with the scientific community. The meeting was cohosted by the Foundation Fighting Blindness and Massachusetts Eye and Ear Infirmary and chaired by Stephen Rose PhD, Chief Research Officer of the Foundation Fighting Blindness. The format was a series of sessions addressing specific aspects of optogenetics, with each session consisting of invited presentations. Audience participation and questions were addressed at the end of each talk, with a wider discussion following each session. All meeting attendees, including those participating by webinar, were able to ask questions. The audience included members of the public, scientists, physicians, venture capitalists, philanthropists, and members of the United States (US) FDA. A full listing of the conference presenters is provided in the Appendix. This article provides a synopsis of the conference proceedings and should not be considered a review article of the field.1–9
The purpose of the meeting was to review the current state-of-the-art science in optogenetics, and make recommendations to the vision research community regarding those steps necessary to move vision restoration by optogenetic therapy into the clinic. The symposium addressed the following key areas: (1) What is the current thinking and planning for clinical trials? (2) Which patients are the best candidates for the treatment? (3) Which are the best optogenetic sensors to use? (4) What are the best cellular targets? In addition, the meeting addressed the level of vision that might be anticipated in treated humans, and whether additional assistive devices might be needed to provide a useful visual restoration when using optogenetic therapy.
Overview
The vision science community is pursuing five broad approaches to vision restoration for retinal degenerative disease: (1) gene therapy to correct a known, causative gene mutation,10 (2) optogenetics to restore function using optogenetic gene therapy,1 (3) stem cell therapy to repair or replace specific subsets of retinal cells,11 (4) visual prosthetic devices,12 and (5) chemical reanimation of the retina.13
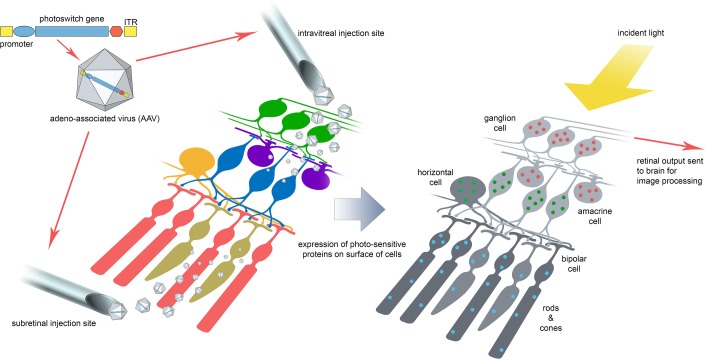
The basic concept of the optogenetic strategy in the retinal degenerative diseases (Fig. 1) is to either enhance the survival and supplement the signaling ability of residual light-sensitive photoreceptors, or to impart a light sensitive property to nonlight sensitive retinal neurons as a means of restoring signaling to the visual cortex. The clinical translation of optogenetic therapy faces challenges. Several challenges are shared with ocular gene therapy as a whole, such as optimizing retinal delivery approaches, targeting the desired cells, attaining sufficient transduction efficiency, increasing the extent of the retina transduced, controlling and sustaining gene expression, controlling tissue specificity, and, in the instances where expression cannot be sustained, being able to provide repeat and fellow eye treatment.
Figure 1.
Optogenetics for vision restoration. The current concept is to use a serotype or mutated version of AAV to deliver to the retina a gene encoding a photoswitch protein, for example channelrhodopsin-2 or halorhodopsin. The intravitreal approach is preferred where cells in the inner retinal layers are to be targeted and the subretinal route for the outer retina. Cell-specific promoters are also being explored to further refine the expression of photoswitch proteins to selected cell types (highlighted by the different colored dots in the cells). For the most part, photoswitch proteins are expressed on the cell membrane where light incident upon the retina opens a channel in the protein allowing ion flux across the cell membrane with resultant depolarization or hyperpolarization of the retinal cell and transmission of the visual impulse to the occipital cortex via the optic nerve.
Other challenges are unique to molecular photoswitches. For example, the switches must be activated at light intensities relevant to lifestyle and be responsive to wavelengths in the human visible range. Targeting different sets of retinal cells may provide different levels of neural processing, such as motion detection, edge effects, and contrast sensitivity that influence the information content of the resulting signaling. Similarly, photoswitch channel response kinetics need to be favorable toward physiologic neuronal firing because this might dictate the quality of vision achievable. While some aspects of these challenges may be augmented by vision processing goggles that project processed images onto the retina, much optimization of these components remains to be understood. It is also important to note that with the exception of melanopsin, the photoswitches are either nonhuman proteins, like the algal/bacterial channelrhodopsins and halorhodopsins, or completely artificial, such as the ionotropic glutamate receptor (LiGluR), with the inherent uncertainty of whether they might present an increased immunogenicity, especially in the context of a degenerating retina, which may have less structural integrity than the immune-privileged healthy retina. A recent study14 has attempted to address this issue in regard to the channelrhodopsin-2 gene (which originates from the algae, Chlamydomonas reinhardtii) delivered to the rodent retina using the adeno-associated virus (AAV)-2 vector. The authors concluded that although antibodies to rAAV and channelrhodopsin were detectable, their levels were too low for rejection. In addition, cellular immune responses were transient and limited. Recently, it has been suggested that the mere intraocular injection of a photoswitch, active on K+ channels, is sufficient to restore visual functions in blind mice. In this case, this therapeutic treatment did not require any cell transfection using viral vectors, but instead only the intraocular injection of the chemical photoswitch AAQ.13
While it is clear from published data that optogenetic therapy can result in visual behavioral responses in small animal models, it is difficult to judge the quality of vision, such as visual fields and acuity, so it is currently unclear how predictive these responses might be of human efficacy. The lack of nonhuman primates with retinitis pigmentosa (RP) prevents evaluation in these higher species. Thus, designing better visual endpoint measures in mammals may be a challenge that the optogenetic field has to meet in the near future or perhaps concede that human testing may be the only way to evaluate efficacy.
There are strong parallels between the questions currently being raised about the challenges facing optogenetics today and those that faced retinal prosthesis development several years ago.15 Encouragingly, these have been clearly answered by human studies16–19 that have shown significant adaptability of the human visual system to process a wide variety of inputs from the retina into functional, meaningful, and useful vision.
The following sections discuss the presentations given at the symposium. Each presenter is listed by their presentation and their full affiliation is provided in the Appendix. Readers interested in more specific details of the field are referred to the published reviews on the subject of optogenetics and visual restoration.1–9 An issue of Nature in 2010 focused on optogenetics as “Method of the Year” also contains an excellent explanatory video of the technique in the following link: http://www.youtube.com/watch?v=I64X7vHSHOE.
Proof of Concept and State-of-the-Art in Vision Restoration Using OptogeneticTools (Richard Masland)
Richard Masland discussed the seminal publications in the field that have lead researchers to conclude that optogenetics has an exciting translational potential for vision restoration. He expressed a personal view that “one of these (optogenetic) molecules is likely to work in restoring vision.” Additionally, evidence supports the observation that the primate/human brain retains significant plasticity and adaptability in processing and perception of visual stimuli, even as an adult.
Richard Masland indicated that there is now unequivocal evidence that optogenetic therapy can restore vision in animal models, as follows:
-
1.
Firstly, the initial proof-of-principle paper from Pan et al.,20 which showed that if you transduce the ganglion cells in rd1 mice with channelrhodopsin-2 the animals become electrophysiologically responsive to light. The rd1 model is one in which photoreceptor degeneration progresses rapidly and at an early age. In a later presentation, Tamai21 corroborated this work providing a detailed discussion on similar behavioral findings in mice and rats.
-
2.
The next paper was from Roska's group who used a specific promoter to transduce a subset of bipolar cells, only the ON bipolar cells. Expressing channelrhodopsin2 selectively in ON bipolar cells restored the light-induced depolarization of these bipolar cells without affecting OFF bipolar cells, which are normally hyperpolarized by light. They showed, for the first time, restoration of visuobehavioral responses by optokinetic testing. Masland commented that the spatial tuning was good and the result outstanding for the field.22
-
3.
The next key experiment was done by Richard Masland's group. A small population of normal retinal ganglion cells (RGCs) expresses melanopsin making them intrinsically photosensitive. These RGCs project their axons to brain centers involved in nonvisual tasks such as the circadian rhythm. In the Masland experiment, mice with advanced retinal degeneration were treated with an AAV vector that over-expressed melanopsin, targeted at the surviving RGCs,23 including those projecting to the visual cortex. The result was a restoration of both electrophysiological and behavioral responses (as demonstrated in the modified Morris water maze test) about 1-week post treatment. Richard Masland did not examine optokinetic responses because the slower response kinetics of melanopsin relative to channelrhodopsin-2 is such that mice would be unlikely to be able to detect motion. Indeed, he highlighted the contrasts in the photoswitches being evaluated: melanopsin is a native protein and can be activated at lower light levels, but has disadvantageous response kinetics in contrast to, for example, channelrhodopsins, which are nonmammalian proteins that require more light for activation but have better responsiveness;
-
4.
Roska's group explored whether transducing surviving cones with hyperpolarizing halorhodopsin might restore vision.24 Cone photoreceptors naturally hyperpolarize to light and cones do persist to a degree in rod photoreceptor degenerations. Their data obtained in two mouse models that differed in the rate of progression of retinal degeneration, rd1 (fast) and rho-/-cng3-/- (slow), show that this is indeed the case, from both electrophysiological and behavioral perspectives;
-
5.
Synthetic photoswitches are also being evaluated. LiGluR25 combines a naturally-occurring glutamate receptor with a photoactive switching mechanism (azobenzene). When this switch changes conformation in the presence of light, the glutamate receptor is opened producing cell depolarization. When RGCs are transduced, and express LiGluR, blind mice with retinal degeneration become visually aware, as shown behaviorally in the 5-arm swimming maze test; and
-
6.
The final experiment from Hauswirth's group, although not an optogenetic approach, addressed whether the adult mammalian brain has the ability to accept and process novel visual information. It focused on whether a dichromatic retina, such as in the monkey, can be made trichromatic. Retinas were transduced with AAV vectors containing the human red opsin gene under the control of the human red/green opsin promoter. In transduced cones, depending on the level of vector expressed red opsin relative to endogenous monkey green opsin in individual cones, novel red responses were measurable in treated monkeys, thus, red sensitivity was added to the animal's color response spectrum. The intriguing finding is that the brain can process new visual information it receives from the retina that now contains a novel subset of red sensitive cones such that these animals can now perform trichromatic tests (modified Cambridge Color Vision test).26
Photoswitches
Sensitivity of Currently Developed Photoswitches (Michael Tri H. Do)
Michael Do presented a synopsis of the comparative sensitivity of published optical switches. This is a major consideration for translation of optogenetics to treat vision loss. If the light intensity required to activate the receptor is high, this would reduce efficacy of the therapy and introduces the potential for light toxicity. Michael Do noted that a significant limitation in current optogenetic technology is the lack of ability to adapt to a wide dynamic range. He also highlighted inconsistency in the way light intensity is reported in the literature, which makes it difficult to interpret, or indeed reproduce experiments, using different equipment. Articles frequently use photometric units that are psychophysical measures of spectral sensitivity (i.e., relying on the scotopic or photopic luminosity function) and as a result photometric units (e.g., lux) are relevant to normal human visual perception but not to optogenetic tools. It would be preferable instead to use radiometric measures in which physical units (e.g., photons cm−2 sec−1 nm−1) are reported precisely.27 In principle, radiometric units allow one to predict the level of activation of any optogenetic tool in any lighting condition.
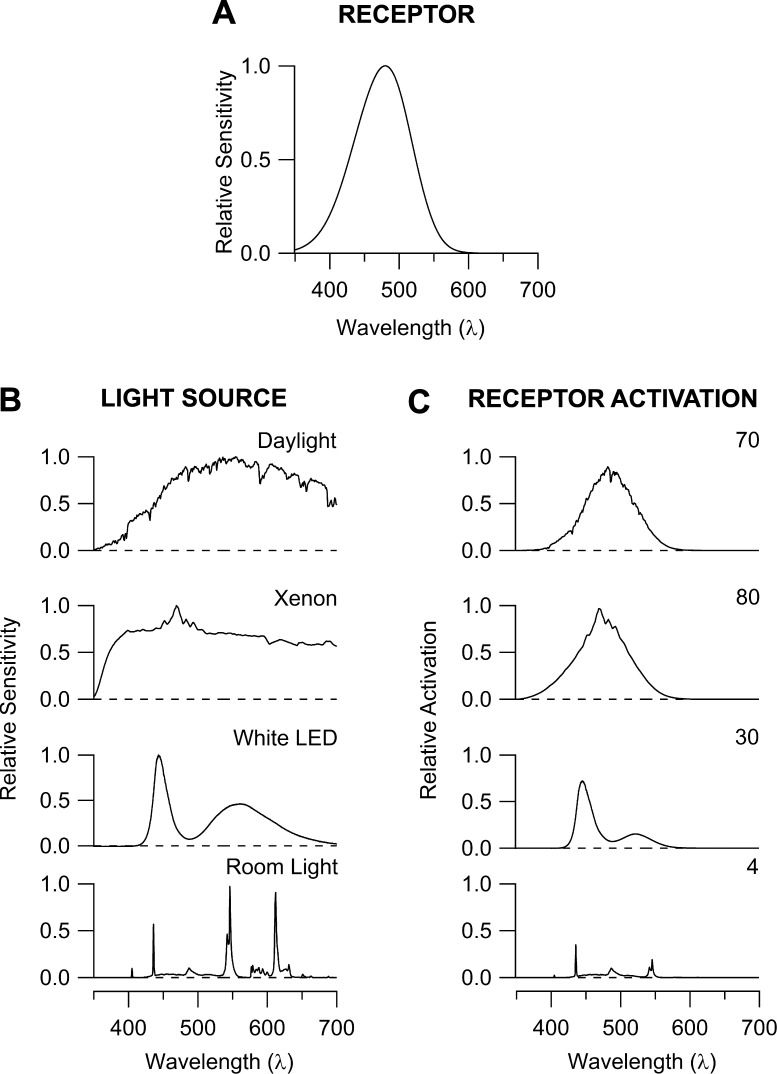
In making comparisons between different optogenetic tools, there are a large number parameters that need to be considered in terms of the stimulus (spectrum, intensity, geometry), stimulus capture (e.g., attenuation by the optical media, receptor density, receptor properties, such as extinction coefficient, path length), response of the receptor (unitary response size, time course, adaptation), and measurement of the response (e.g., at molecular, cellular, or behavioral levels).27 Figure 2 illustrates an aspect of this complexity by showing how one receptor is differentially activated by light sources that appear similarly white to human observers. By measuring and reporting the relevant parameters, investigators can accelerate the development of optogenetic tools and permit careful selection of the tool that is most suitable for a given application.
Figure 2.
Differential activation of an opsin molecule by lighting that appears psychophysically similar to human observers. (A) The spectral sensitivity of melanopsin, a visual pigment in the human retina that has been used as an optogenetic tool.64,65 Shown is the principal, alpha band.66 (B) Spectra of common, white light sources normalized to their peaks. Daylight is measured through a window on a clear day in Boston, xenon is the output of an arc lamp projected through a microscope, room light is fluorescent laboratory light, and white, light emitting diode (LED) is the beam of a flashlight. (C) Product of the spectrum of each light source with that of melanopsin. The level of melanopsin activation is obtained by integrating under the curve, and is given on the right.
Channelrhodopsins (Brian Chow)
The microbially-derived rhodopsins differ from human rhodopsins in that they are not G-protein coupled receptors and lack signal amplification, having evolved their response kinetics for creating electrochemical gradient for adenosine triphosphate (ATP) synthesis and motility. A major purpose of ongoing research for vision restoration is to re-engineer these proteins for greater sensitivity and/or signal amplification. Improving sensitivity is not about improving quantum efficiency since microbial rhodopsins are not photochemically inefficient per se, but instead requires a deeper understanding of how many photons are required to depolarize a neuron and obtain reliable spike responses. This physiological spiking response (versus purely photochemical response of photon absorption) depends on a variety of factors such as light propagation through tissue, expression levels and trafficking, off-kinetics, and conductance per photocycle, among others. Accordingly, additional re-engineering goals will be to produce proteins that have a longer wavelength (red-shifted) spectral sensitivity, larger conductances, and understand and meet the optimal off-kinetics for smooth visual perception.28–30 Other issues to overcome include potential complexities of the heterologous gene expression, such as inaccurate protein folding, cellular trafficking, and an immune response; and the impact of high levels of channel-rhodopsin on heterologous cell membrane stability and function.
Several approaches are being employed to engineer new depolarizing and hyperpolarizing ion channels, including continued evaluations of phylogenetic diversity and intelligent design through site directed mutagenesis and chimeric protein development. For each engineered improvement in function, there are, however, trade-offs. For example, slowing down the channel kinetics, especially increasing its open time, can increase the light sensitivity of channelrhodopsin, but the associated drawback is to slow down the light response kinetics of photosensitized cells. Similarly, increasing the calcium conductance of the channel to improve light sensitivity also increases intracellular calcium levels that may prove cytotoxic.
Melanopsin (Satchidananda Panda)
Satchidananda Panda discussed current approaches to using melanopsin as an optical switch for optogenetic therapy. Melanopsin is an endogenous mammalian retinal protein, first identified in intrinsically-sensitive RGCs.31 Melanopsin is involved in relaying signals for pupillary responsiveness as well as entrainment of circadian rhythms.32,33 As a proof of concept, Panda and colleagues23 used AAV2-mediated melanopsin expression to transduce RGCs in mice and were able to observe both single ganglion cell electrophysiological responses and visual behavioral responses.
While melanopsin is sensitive to relatively low light levels, it produces sustained depolarization with off-rate kinetics that is too slow for functional vision restoration. C-terminal truncations can improve this kinetics, probably by modulating the large number of phosphorylation sites on the melanopsin protein. The systematic evaluation of these potential phosphorylation sites by a variety of in vitro expression studies and calcium release assays, along with in vivo measurements, such as restoration of pupillary responses and circadian entrainment in rd1 melanopsin double-knockout mice, has revealed several candidates that have the potential for clinical translation, though more work is needed. Additional studies have examined whether co-expression with arrestin-1 and/or -2 (a signal transduction regulator that assists in switching off the melanopsin signal) will improve melanopsin performance as an optogenetic tool.
Photoreceptive Ionotropic Glutamate Receptors (John Flannery)
John Flannery presented research being undertaken by the National Institutes of Health (NIH) Nanomedicine Development Center for Optical Control of Biological Function, a group of eight laboratories in which endogenous mammalian receptors are being re-engineered to add intrinsic light sensitivity. One project is the LiGluR,34 which has similar kinetics to channelrhodopsins in terms of speed and conductance. LiGluR is a two-component system comprising a naturally occurring glutamate receptor covalently attached to an azobenzene arm that is covalently attached to a glutamate molecule. Exposure to 380 nm light results in a conformational change in the azobenzene arm that brings the glutamate molecule to bind with the receptor, forming an ion channel pore, resulting in ion (Na+, K+, and Ca2+) flow and depolarization of the cell. The azobenzene arm will thermally relax back to the inactive trans conformation or can be rapidly isomerized to the inactive conformation by 500 nm light. This optical switch is fast enough to generate reproducible 50 Hz spike trains. Functional studies using multielectrode arrays (MEA) of LiGluR have shown in vivo that multiple RGCs can be rendered photosensitive, such that these cells are capable of delivering a signal to the visual cortex, as confirmed in vivo by measuring a visual evoked potential, pupillary light responses, and a functional, behavioral response (Morris water maze) in rodent models of retinal degenerative disease.25 John Flannery indicated further studies are underway to evaluate the visual performance and behavior of LiGluR expressed in dog and primates and to develop red-shifted photoswitches to function in the visible light range. He also speculated that the field should consider the development of more complex molecular systems to emulate the physiological phototransduction cascade. In this way, the light sensor could be separated from the actuator with resultant improvements in sensitivity, adaptation, and kinetics.
Synthetic Photoswitches (Russell Van Gelder)
Russell Van Gelder spoke about synthetic small molecule photoswitches. While not genetic, and therefore not strictly optogenetic tools, the chemicals function in an analogous manner to the optogenetic tools, by conferring photosensitivity on RGCs. The first compound developed is named AAQ13 and comprises three moieties: quaternary ammonium, which blocks voltage-gated potassium channels; azobenzene, the photo-responsive switch; and acrylamide, to bind the ion channel. In similar fashion to LiGluR, the activity of AAQ is driven by light-induced conformational changes in azobenzene. As a proof of concept, the properties of AAQ were investigated in retina explants from melanopsin knock-out mice, at age P8, using a multielectrode array. With direct exposure to AAQ in solution, RGCs could be depolarized in response to the correct wavelength of light (380 nm, 1015 photons mm−2 sec−1). In vivo, AAQ was able to restore both the pupillary response and a positive behavioral response in the light aversion test to rd/rd;opn4-/- mice. A limitation of AAQ is its short half-life in vivo, which means any visual benefit is transient. The next generation of AAQ-derivatives is already now in testing and these may have greater clinical translational applicability. An example is PHeNAQ, which can produce photosensitization for approximately 8 hours in response to a white light stimulus, has much better response kinetics, inactivates spontaneously in the dark, and is activated at lower light levels. Preliminary evidence also suggests PHeNAQ can produce behavioral responses in treated mice (Nemargut JP III, et al. IOVS 2012;358:ARVO E-Abstract 3639). Further work is necessary to improve formulation, assess toxicity, formulate for sustained release, and better vitreous penetration. Russell Van Gelder suggested that one potential early application of this technology might be to harness the transient effect to evaluate whether conventional gene-based optogenetic therapy might be of benefit to a candidate patient.
Clinical Translational Challenges and Opportunities
Understanding Morphological Changes in Retinal Degenerations: Remodeling and Its Consequences
Enrica Strettoi, Robert Marc, and Christine Curcio detailed the histo- and molecular pathologic changes that accompany photoreceptor loss in conditions such as RP and age-related macular degeneration (AMD). While most of what is known comes from animal models, the findings in mice, rats, and rabbits closely mirror the limited data available in humans.
In the broadest terms, three stages can be recognized in the degenerative process: photoreceptor stress, photoreceptor loss, and then tissue remodeling.35,36 Remodeling describes the gradual and morphological, functional, and cellular reprogramming37 events that affect all retinal components, such as neurons, glia, and blood vessels. The process partially reflects the consequence of deafferentation of remaining retinal cells as photoreceptors die38 and appears stereotyped to all forms of RP though the kinetics vary likely dependent upon genotype. The end stage of RP is characterized by cell death and gliosis, obliteration of the subretinal space, intraretinal migration of the retinal pigment epithelium (RPE), and blood vessel attenuation. There is no evidence that ischemic cytotoxicity also occurs in RP.
Photoreceptors and the Retinal Pigment Epithelium
In RP, rod photoreceptor death typically precedes cone photoreceptor loss. In some forms of retinal degeneration, cone photoreceptors are decimated early in the disease process, while in others cones can survive late into the disease process. Inner retinal remodeling appears to accompany and follow both the death of the rods and cones. Migration and loss of surviving RPE cells is also observed congruent with the rod and cone loss.
The disappearance of both photoreceptors and RPE and the formation of a gliotic scar obliterates the subretinal space present in a nondiseased retina, and this poses potential challenges for obtaining surgical access to this area for subretinal delivery of gene therapeutic agents.39
Remodeling and the Bipolar Cells
Some of the earliest signs of remodeling appear in rod bipolar cells, which show dendrite reduction, retraction, and mislocation combined with displacement of their cell bodies and more overt migration.38 The process is reminiscent of transynaptic atrophy. Neuritogenesis and axonogenesis of bipolar cells are observed with the establishment of de novo connections and signaling networks. Surface contacts are formed, but do not comprise the ribbon synapses characteristic of a nondiseased retina. At the molecular level, during retinal degeneration there is loss of mGluR6 expression, activation of the retinoic acid pathways,40 and a change in polarity with the conversion of ON-bipolar cells to OFF-bipolar cells. This is significant for any gene therapy that might target the ON-bipolar cells. For these reasons, targeting the bipolar cell selectively with optogenetic therapy may not optimal. Cell-specific promoters are available for ON cells,41 but not yet characterized for OFF cells.
Remodeling of the RGCs and Amacrine Cells
The RGCs are the innermost layer of cells in the retina and extend their processes from the retina back through the optic nerve to the brain, being the ultimate connection between the light signaling processed by the retinal layers and the brain. The retinal ganglion and amacrine cells appear the most resistant to degenerative changes.42 Indeed, survival appears fairly complete even late in the disease. Additionally, RGCs maintain their inner laminal position unchanged and functionally maintain anterograde transport and depolarization characteristics.43 In Enrica Strettoi's opinion, “the most stable retinal cell during retinal degeneration is the ganglion cell”. Current evidence suggests this might be the most universal target for optogenetic therapy, but signaling from these layers would lack the additional interpretation provided by signal processing in the outer layers of the retina.
Approaches to Evaluate Candidate Patients for Optogenetic Treatments (Artur V. Cideciyan)
Artur Cideciyan's presentation examined different noninvasive methods that are currently available and can be used to perform quantitative measurements to evaluate candidacy of patients with retinal degenerations for potential optogenetic treatments. Initially the capabilities of optical coherence tomography (OCT) were reviewed. OCT is a noninvasive, noncontact method that produces cross-sectional images of the retinal architecture. In healthy eyes, OCT provides micron-scale resolution for 10 or more boundaries formed by scattering changes and retinal layers distinguishable between these boundaries. Unbiased comparisons between histology and OCT in animal models have defined the identity of some of the layers.44 In terms of inherited retinal degenerations, primary consequences of disease are found in the outer retina, which includes the outer nuclear layer (ONL) where the nuclei of rod and cone photoreceptors reside; predegenerative photoreceptor stress results in abnormal thickening of the ONL, whereas loss of photoreceptors leading to degeneration results in abnormal thinning.45 Quantitative aspects of ONL thickness abnormalities were presented from patients with RP due to RHO, MAK, or RPGR mutations,46–49 those with macular degenerations and cone–rod dystrophies caused by ABCA4 and CERKL mutations,49,50 Usher syndrome patients with USH3A or USH2A mutations,51 and those with Leber congenital amaurosis due to RPE65, CRB1, CEP290, IQCB1, and AIPL1 mutations.52–55 More recently, it has been possible to distinguish between rod outer segments (ROS) and cone outer segments (COS), and several examples of retinal degenerations illustrating abnormalities of the ROS and COS lengths were presented.47,52,56 Artur Cideciyan suggested that primary candidates for optogenetics therapies would be expected to lack detectable ONL, ROS, and COS.
OCT also affords the opportunity to evaluate structural abnormalities at the outer plexiform layer (OPL) where photoreceptors synapse to bipolar and horizontal cells, and at the inner plexiform layer (IPL) where bipolar cells synapse to amacrine and ganglion cells. In addition, inner nuclear layer (INL) and ganglion cell layer (GCL) are distinguishable with their hyper-scattering bands and represent cell nuclei. A characteristic finding in retinal degenerative conditions is thickening of the inner retinal layers overlying areas of photoreceptor loss.46,51,55 The thickening of inner retinal layers is thought to represent a form of reactive retinal remodeling in response to photoreceptor degeneration. An important contributor to inner retinal thickening is thickening of the INL, which may result from a combination of Müller glial activation and major synaptic abnormalities at the level of the bipolar cells. In Artur Cideciyan's opinion, such changes detectable noninvasively would not bode well for optogenetic approaches targeting the bipolar cells. GCL and retinal nerve fiber layer (RNFL) is also measurable on OCT even in advanced stages of retinal degeneration.50 Cideciyan suggested that different molecular forms of retinal degenerations be examined quantitatively for inner retinal structural abnormalities in order to detect the stages of disease that show remnant GCL population that could be targeted with optogenetic approaches.
Importantly, Artur Cideciyan pointed out that it is relatively easy to image and evaluate the central retina, but evaluation of the more peripheral retinal regions is constrained by ocular optics. For this purpose, visual function tests with a large dynamic range, such as dark-adapted perimetry, can be used as surrogates of peripheral retinal structure. Additionally, it will be important to determine the structural and functional evaluation of extra-retinal visual pathways. For this purpose, structural magnetic resonance imaging (MRI) is very useful in assessing the optic nerve, lateral geniculate nucleus, and occipital cortex.53,57,58 Functional MRI offers the opportunity to study cortical responses to visual stimuli and has been adapted to allow evaluation of patients with low vision.57 Further studies are necessary to determine test–retest variability of these measures in specific patient populations en route to their use in a potential clinical trial.
Delivery of Optogenetic Agents to the Retina (William Hauswirth)
William Hauswirth presented an overview of AAV-based technology, retinal cell targeting, and ongoing efforts to identify and engineer retinal cell-specific expression. There are two conventional surgical routes of vector administration: subretinal injection, that preferentially targets the outer retinal cells and intravitreal injection for targeting inner retinal cells. A key consideration for intravitreal delivery is that ‘the mouse is not a primate' and transduction efficiency seen in rodents does not necessarily translate to the experience in primates, including man. This appears largely as a result in differences in the internal limiting membrane (ILM), which forms the interface between the vitreous body and the RNFL. In rodents, the ILM appears permeable to AAV serotype 2 (AAV2) but in primates, retinal penetration is reduced probably due to both the increased number of heparin sulphate binding sites in the ILM that bind virus and its increased thickness relative to the mouse. Specific regions of the primate ILM are however permeable to AAV2, most notably within the fovea of the central macula and around retinal arterioles.
Rod and Cone Photoreceptors
A number of AAV serotypes transduce photoreceptor cells efficiently when delivered subretinally, for example AAV2, AAV5, and AAV9.59 AAV2 vectors are currently being employed in human clinical trials. In addition, cell specific promoters are also available (rhodopsin kinase for rods, cone arrestin, and cone opsin for cones).
Bipolar Cells
Targeting these cells is still in development, but AAV serotypes delivered subretinally carrying the bipolar cell-specific GRM6 promoter afford an opportunity to selectively express genes in the ON-cell subtype, though OFF-bipolar cell selectivity is not yet achievable.
Retinal Ganglion Cells
Approximately 50% of these cells can be transduced in rodents from a single intravitreal injection. The challenge in the primate is, however, to overcome the barrier to AAV diffusion presented by the ILM. Critically, efficient foveal and parafoveal transduction is seen following intravitreal delivery of conventional AAV serotypes; this has translational applicability to optogenetics since the macula is so important for high-resolution vision. In addition, multifocal perivascular transduction is also observed in the macula and retinal midperiphery, presumably providing access through the ILM at points where the vasculature also crosses. William Hauswirth highlighted ongoing research efforts to selectively mutate AAV to gain better retinal penetration and transduction efficiency by altering viral surface tyrosine moieties.60 With this work currently making good progress, he identified that the current major hindrance to retinal cell targeting as the lack of new retina-specific promoters. In this regard, he discussed the Pleiades Promoter Project (http://www.pleiades.org/) that was attempting to identify all central nervous system–related promoters. Mining the current dataset may have identified 17 mini-promoters relevant to the retina that are compatible with AAV size constraints. It must be hoped that the combination of improved AAV tropism and cell-specific promoters will alleviate (or at least substantially reduce) nontarget cell expression of the gene.
Optogenetic Human Clinical Trials: Best Candidates and Appropriate Efficacy Endpoints (Eric Pierce)
Eric Pierce noted that optogenetic therapy affords the opportunity to treat all forms of outer retinal degenerative disease independent of genotype, however, the best candidates for initial clinical evaluation were likely to be those patients with advanced RP given their phenotypic homogeneity in the late stages of disease.
Eric Pierce acknowledged that identifying the best efficacy endpoints for human optogenetic trials is challenging. The currently FDA-approved endpoints of visual acuity, visual field, and retinal lesion size have limited applicability to early phase optogenetic studies since those with advanced or end-stage retinal disease have very low vision. There is also a need to perform tests at suitable lighting levels. Eric Pierce reviewed the symptomatology observed in these conditions and then critically evaluated the utility of currently available tests. He drew from clinical experiences gained from ongoing Leber's congenital amaurosis RPE65 gene therapy clinical trials to observe:
-
1.
The electroretinogram (ERG) measures mostly photoreceptor responses and some inner retinal responses. Therefore, current ERG protocols would not be useful in the absence of photoreceptors;
-
2.
Retinal imaging, for example OCT, may be useful in identifying those individuals with outer retinal pathology or persisting RGCs but would be not useful as an outcome measure for functional vision in optogenetic therapy;
-
3.
Measurement of full field threshold sensitivity has been used to show improved cone function in the RPE65 gene therapy trials and would definitely have potential as a quantitative evaluation of visual restoration and visual function in optogenetic trials;
-
4.
Threshold visual fields have been reported to provide a noticeable increase in visual field sensitivity in some patients treated with RPE65 gene replacement, but in Eric Pierce's experience this is neither uniform nor reliable and difficult to apply to profoundly visually-impaired individuals;
-
5.
Visual acuity might be regarded as the gold standard and although some RPE65 patients do get sustained improvements in acuity, this is not a uniform observation and does not necessarily correlate with restored visual function in certain patients. As a result, it may not be suitable as a primary endpoint in optogenetic trials;
-
6.
Microperimetry may have some utility, however, it would be challenging to perform in profoundly visually-impaired individuals due to fixation instability;
-
7.
Pupillary light reflex is a good integrator of the light response and has the advantage of being objective; and
-
8.
Quantitative mobility testing might have the greatest utility since it integrates all aspects of visual function (visual acuity, visual field, and light sensitivity) and relates to quality of life. However, this test is not yet reliably quantifiable or validated. As part of the LCA2, RPE65 gene therapy clinical trial, there are studies striving to validate these and have them accepted by the FDA as a primary endpoint.
Discussion from the attendees focused on whether other electrophysiological methods might have use such as the visual evoked potential (VEP). It was suggested that in the untreated individual the VEP would be unrecordable, although possibly combined with the electrically-evoked response (EER) these measures might have utility in identifying potential participants. If therapy restored this response, both in latency and amplitude of the waveform generated, it would be a sign of efficacy although it cannot tell whether the patient is actually seeing (i.e., had functional vision). However, it was also acknowledged that the visual evoked potential is generated by a mass response from all retinal regions so the amplitude is dependent on variables, such as electrode replacement and so may lack either the sensitivity or specificity to be of great value. Additional evaluation using a pattern VEP was mentioned as one way of providing some measure of the level of visual acuity.
Challenges and Opportunities for High-Resolution Vision Following Optogenetic Therapy (Frank Werblin)
Frank Werblin provided insight into the processing of visual information by the retina and what type of image preprocessing might be needed to achieve optimal vision following optogenetic therapy.61–63 At the very least, luminance gain control will be required in any system because none of the artificial approaches employs a photoreceptor-like gain control or adaptation. He acknowledged that while there is strong logic to suggest that preprocessing technology, for example a pair of specially-designed goggles or spectacles might optimize both gain and visual resolution, experience with cochlear and, to a degree, retinal implants have demonstrated the remarkable capacity and adaptability of human sensory systems and cortical processing to receive and interpret unnatural information after adaptation has occurred. Conceptually, if preprocessing is needed, it will be needed at all levels of transduction, from the photoreceptor to the ganglion cells. Preprocessing of the image would require manipulation of a number features of the signal including brightness, contrast, saturation, and adaptation (http://mcb.berkeley.edu/labs/werblin/). A key component of such a system would be the bio-inspired mechanism for each of these as the image and environment changed. Frank Werblin noted that existing camera technology was already sufficiently advanced to perform preprocessing automatically or to allow the user to adjust the system manually to optimize the visual experience. He also noted that it is not yet known if other functions of the retina, including contrast gain control, fixational stability, and saccadic suppression, will need to be addressed at a preprocessing level or whether the remaining visual system can itself compensate and accommodate these functions following optogenetic therapy.
The audience raised questions on the difficulty to code visual information when stimulating RGCs because there is no retinotopy in the foveal area at this level. Frank Werblin answered that the visual cortex should solve this difficulty.
A Proposed Human Clinical Trial
Thomas Rea and Peter Francis of RetroSense Therapeutics LLC discussed the company's drug development plans for their first proposed human clinical optogenetic application for vision restoration. RetroSense proposes using an AAV2-based vector to deliver a cDNA encoding a truncated version of channelrhodopsin-2 (which retains its functionality as a light sensor) to RGCs via intravitreal injection. Both the vector, including the promoter and polyadenylation signal, and the serotype, have been used safely in a number of human clinical trials, including retinal gene replacement trials for Leber's congenital amaurosis. While animal models have proven useful in exploring the visual restorative promise of optogenetic therapy, the field lacks a single animal model that embodies all the necessary biological features with which to best predict the complex phenotypic outcome in humans (e.g., human responses to therapy). To date, in the literature, all efficacy and toxicology evaluations14 have been performed with the truncated channelrhodopsin-2 fused with a reporter such as green flourescent protein or Venus.
RetroSense is proposing that their first human study will be an open label, single eye phase Ib/IIa dose escalation study of intravitreally administered agent to individuals with advanced RP. One week before the symposium, the company held a formal type B (preIND) meeting with the US FDA in which a preclinical path to the human clinical trial was identified. The FDA was very receptive to the concept of optogenetic therapy, though it is clear that the regulatory approval environment is still evolving. The potential regulatory agency expectations in the preclinical setting, for groups considering clinical translation of optogenetics may include:
-
1.
Demonstration of efficacy in multiple animal models with careful documentation of the light levels needed for channel activation;
-
2.
The need for IND-enabling combined/hybrid pharmacology/toxicology studies, which allow the assessment of toxicity in the setting of the disease process in an environment where the agent is at a dose(s) where its efficacious effect is exerted;
-
3.
Long-term human safety monitoring;
-
4.
The development of a potency assay for later phase clinical development;
-
5.
A clinical development plan that considers repeat and/or fellow eye injection; and
-
6.
Light-activation requirements specific to their optogenetic therapy.
Conclusions
The symposium provided an excellent forum for thought-leaders in the field to share their ideas on the best path forward for optogenetics in vision restoration. While there was a general consensus that the field has matured to a point that clinical translation is a realistic proposition, the discussion also led to the identification of key areas requiring more research, development, and funding. Specifically, the attendees concluded that visual outcomes will be optimized by improved photoswitch sensitivity and channel kinetics, enhanced gene delivery technology, and the development of image-enhancing visual aids. Furthermore, the assessment and prediction of visual efficacy will be best achieved by the generation of new animal models, both small and large eye, better animal visuobehavioral testing, and validated clinical endpoints designed to optimally assess real-world visual function. Ambitious though this is, accomplishing such an agenda will ensure a promising future for this technology and the realistic prospect of restoring meaningful vision to those affected by inherited retinal degenerative disease.
Acknowledgments
The symposium thanks the Foundation Fighting Blindness Clinical Research Institute for financial support, the Massachusetts Eye and Ear facilities, to all speakers (listed in Appendix), and attendees of the symposium. In particular, thanks are extended to the organizers (Richard Masland, PhD, Massachusetts Eye and Ear Infirmary and John Flannery, PhD, University of California, Berkeley), and the session moderators.
Disclosure: P.J. Francis, RetroSense Therapeutics (E); B. Mansfield, Foundation Fighting Blindness (E); S. Rose, Foundation Fighting Blindness (E)
Appendix
Presenters
Brian Chow, Department of Bioengineering, University of Pennsylvania, Philadelphia, PA
Artur V. Cideciyan, Scheie Eye Institute, Department of Ophthalmology, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA
Christine Curcio, Department of Ophthalmology, University of Alabama School of Medicine, EyeSight Foundation of Alabama Vision Research Labs, Birmingham, AL
Michael Tri H. Do, F.M. Kirby, Neurobiology Center and Department of Neurology of Boston Children's Hospital and Harvard Medical School, Boston, MA
William W. Hauswirth, Department of Ophthalmology, University of Florida, Gainesville, FL
Robert E. Marc, Moran Eye Center, Salt Lake City, UT
Satchidananda Panda, Salk Institute for Biological Studies, La Jolla, CA
Eric Pierce, Massachusetts Eye and Ear Infirmary, Boston, MA
Thomas Rea, RetroSense Therapeutics LLC, Ann Arbor, MI
Enrica Strettoi, Istituto di Neuroscienze del CNR, Pisa, Italy
Hiroshi Tomita, Department of Chemistry and Bioengineering, Graduate School of Engineering, Iwate University Japan
Russell N. Van Gelder, Department of Ophthalmology, University of Washington, Seattle, WA
Frank Werblin, Graduate School Division of Neurobiology, University of California, Berkeley, CA
John G. Flannery, Helen Wills Neuroscience Institute, University of California, Berkeley, CA
Richard Masland, Massachusetts Eye and Ear Infirmary, Boston, MA
Grant Funding and Conflicts of Interest
AVC: National Eye Institute grant EY 013203. AVC is an RPB Senior Scientific Scholar.
JGF: Foundation Fighting Blindness, National Institutes of Health 5PN2EY018241-08 Nanomedicine Development Center for the Optical Control of Biological Function
SP: National Institutes of Health grant EY016807
MTD: Research grant (Whitehall Foundation), Basil O'Connor Starter Scholar Research Award (March of Dimes). Spectra of light sources in Figure 2 were obtained by Alan Emanuel (Program in Neuroscience, Harvard Medical School).
WWH and the University of Florida have a financial interest in the use of AAV therapies, and own equity in a company (AGTC Inc.) that might, in the future, commercialize some aspects of this work. WWH also owns equity in BionicSight.
References
- 1.Busskamp V, Picaud S, Sahel JA, Roska B. Optogenetic therapy for retinitis pigmentosa. Gene Ther. 2012;19:169–175. doi: 10.1038/gt.2011.155. [DOI] [PubMed] [Google Scholar]
- 2.Busskamp V, Roska B. Optogenetic approaches to restoring visual function in retinitis pigmentosa. Curr Opin Neurobiol. 2011;21:942–946. doi: 10.1016/j.conb.2011.06.001. [DOI] [PubMed] [Google Scholar]
- 3.den Hollander AI, Black A, Bennett J, Cremers FP. Lighting a candle in the dark: advances in genetics and gene therapy of recessive retinal dystrophies. J Clin Invest. 2010;120:3042–3053. doi: 10.1172/JCI42258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Do MT, Yau KW. Intrinsically photosensitive retinal ganglion cells. Physiol Rev. 2010;90:1547–1581. doi: 10.1152/physrev.00013.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dowling J. Artificial human vision. Expert Rev Med Devices. 2005;2:73–85. doi: 10.1586/17434440.2.1.73. [DOI] [PubMed] [Google Scholar]
- 6.Fehrentz T, Schonberger M, Trauner D. Optochemical genetics. Angew Chem Int Ed Engl. 2011;50:12156–12182. doi: 10.1002/anie.201103236. [DOI] [PubMed] [Google Scholar]
- 7.Gross M. Shining new light on the brain. Curr Biol. 2011;21:R831–R833. doi: 10.1016/j.cub.2011.10.007. [DOI] [PubMed] [Google Scholar]
- 8.Hellstrom M, Harvey AR. Retinal ganglion cell gene therapy and visual system repair. Curr Gene Ther. 2011;11:116–131. doi: 10.2174/156652311794940746. [DOI] [PubMed] [Google Scholar]
- 9.Tomita H, Sugano E, Isago H, Tamai M. Channelrhodopsins provide a breakthrough insight into strategies for curing blindness. J Genet. 2009;88:409–415. doi: 10.1007/s12041-009-0062-6. [DOI] [PubMed] [Google Scholar]
- 10.Lipinski DM, Thake M, Maclaren RE. Clinical applications of retinal gene therapy. Prog Retin Eye Res. 2012;32:22–47. doi: 10.1016/j.preteyeres.2012.09.001. [DOI] [PubMed] [Google Scholar]
- 11.Ong JM, da Cruz L. A review and update on the current status of stem cell therapy and the retina. Br Med Bull. 2012;102:133–146. doi: 10.1093/bmb/lds013. [DOI] [PubMed] [Google Scholar]
- 12.Dagnelie G. Retinal implants: emergence of a multidisciplinary field. Curr Opin Neurol. 2012;25:67–75. doi: 10.1097/WCO.0b013e32834f02c3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Polosukhina A, Litt J, Tochitsky I, et al. Photochemical restoration of visual responses in blind mice. Neuron. 2012;75:271–282. doi: 10.1016/j.neuron.2012.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sugano E, Isago H, Wang Z, Murayama N, Tamai M, Tomita H. Immune responses to adeno-associated virus type 2 encoding channelrhodopsin-2 in a genetically blind rat model for gene therapy. Gene Ther. 2011;18:266–274. doi: 10.1038/gt.2010.140. [DOI] [PubMed] [Google Scholar]
- 15.Zrenner E. Will retinal implants restore vision? Science. 2002;295:1022–1025. doi: 10.1126/science.1067996. [DOI] [PubMed] [Google Scholar]
- 16.Wilke R, Gabel VP, Sachs H, et al. Spatial resolution and perception of patterns mediated by a subretinal 16-electrode array in patients blinded by hereditary retinal dystrophies. Invest Ophthalmol Vis Sci. 2011;52:5995–6003. doi: 10.1167/iovs.10-6946. [DOI] [PubMed] [Google Scholar]
- 17.Zrenner E, Bartz-Schmidt KU, Benav H, et al. Subretinal electronic chips allow blind patients to read letters and combine them to words. Proc Biol Sci. 2011;278:1489–1497. doi: 10.1098/rspb.2010.1747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Humayun MS, Dorn JD, da Cruz L, et al. Interim results from the international trial of Second Sight's visual prosthesis. Ophthalmology. 2012;119:779–788. doi: 10.1016/j.ophtha.2011.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.da Cruz L, Coley BF, Dorn J, et al. The Argus II epiretinal prosthesis system allows letter and word reading and long-term function in patients with profound vision loss. Br J Ophthalmol. 2013;97:632–636. doi: 10.1136/bjophthalmol-2012-301525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bi A, Cui J, Ma YP, et al. Ectopic expression of a microbial-type rhodopsin restores visual responses in mice with photoreceptor degeneration. Neuron. 2006;50:23–33. doi: 10.1016/j.neuron.2006.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tomita H, Sugano E, Fukazawa Y, et al. Visual properties of transgenic rats harboring the channelrhodopsin-2 gene regulated by the thy-1.2 promoter. PLoS One. 2009;4:e7679. doi: 10.1371/journal.pone.0007679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lagali PS, Balya D, Awatramani GB, et al. Light-activated channels targeted to ON bipolar cells restore visual function in retinal degeneration. Nat Neurosci. 2008;11:667–675. doi: 10.1038/nn.2117. [DOI] [PubMed] [Google Scholar]
- 23.Lin B, Koizumi A, Tanaka N, Panda S, Masland RH. Restoration of visual function in retinal degeneration mice by ectopic expression of melanopsin. Proc Natl Acad Sci U S A. 2008;105:16009–16014. doi: 10.1073/pnas.0806114105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Busskamp V, Duebel J, Balya D, et al. Genetic reactivation of cone photoreceptors restores visual responses in retinitis pigmentosa. Science. 2010;329:413–417. doi: 10.1126/science.1190897. [DOI] [PubMed] [Google Scholar]
- 25.Caporale N, Kolstad KD, Lee T, et al. LiGluR restores visual responses in rodent models of inherited blindness. Mol Ther. 2011;19:1212–1219. doi: 10.1038/mt.2011.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mancuso K, Hauswirth WW, Li Q, et al. Gene therapy for red-green colour blindness in adult primates. Nature. 2009;461:784–787. doi: 10.1038/nature08401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Johnsen S. The Optics of Life: A Biologist's Guide to Light in Nature. Princeton: Princeton University Press;; 2012. [Google Scholar]
- 28.Chow BY, Han X, Boyden ES. Genetically encoded molecular tools for light-driven silencing of targeted neurons. Prog Brain Res. 2012;196:49–61. doi: 10.1016/B978-0-444-59426-6.00003-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Han X, Chow BY, Zhou H, et al. A high-light sensitivity optical neural silencer: development and application to optogenetic control of non-human primate cortex. Front Syst Neurosci. 2011;5:18. doi: 10.3389/fnsys.2011.00018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chow BY, Chuong AS, Klapoetke NC, Boyden ES. Synthetic physiology strategies for adapting tools from nature for genetically targeted control of fast biological processes. Methods Enzymol. 2011;497:425–443. doi: 10.1016/B978-0-12-385075-1.00018-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hattar S, Liao HW, Takao M, Berson DM, Yau KW. Melanopsin-containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science. 2002;295:1065–1070. doi: 10.1126/science.1069609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Panda S, Sato TK, Castrucci AM, et al. Melanopsin (Opn4) requirement for normal light-induced circadian phase shifting. Science. 2002;298:2213–2216. doi: 10.1126/science.1076848. [DOI] [PubMed] [Google Scholar]
- 33.Panda S. Multiple photopigments entrain the Mammalian circadian oscillator. Neuron. 2007;53:619–621. doi: 10.1016/j.neuron.2007.02.017. [DOI] [PubMed] [Google Scholar]
- 34.Szobota S, Gorostiza P, Del Bene F, et al. Remote control of neuronal activity with a light-gated glutamate receptor. Neuron. 2007;54:535–545. doi: 10.1016/j.neuron.2007.05.010. [DOI] [PubMed] [Google Scholar]
- 35.Jones BW, Kondo M, Terasaki H, Lin Y, McCall M, Marc RE. Retinal remodeling. Jpn J Ophthalmol. 2012;56:289–306. doi: 10.1007/s10384-012-0147-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Curcio CA, Medeiros NE, Millican CL. Photoreceptor loss in age-related macular degeneration. Invest Ophthalmol Vis Sci. 1996;37:1236–1249. [PubMed] [Google Scholar]
- 37.Marc RE, Jones BW, Anderson JR, et al. Neural reprogramming in retinal degeneration. Invest Ophthalmol Vis Sci. 2007;48:3364–3371. doi: 10.1167/iovs.07-0032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Strettoi E, Pignatelli V. Modifications of retinal neurons in a mouse model of retinitis pigmentosa. Proc Natl Acad Sci U S A. 2000;97:11020–11025. doi: 10.1073/pnas.190291097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Adler R, Curcio C, Hicks D, Price D, Wong F. Cell death in age-related macular degeneration. Mol Vis. 1999;5:31. [PubMed] [Google Scholar]
- 40.Lin Y, Jones BW, Liu A, et al. Retinoid receptors trigger neuritogenesis in retinal degenerations. FASEB J. 2012;26:81–92. doi: 10.1096/fj.11-192914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nakajima Y, Moriyama M, Hattori M, Minato N, Nakanishi S. Isolation of ON bipolar cell genes via hrGFP-coupled cell enrichment using the mGluR6 promoter. J Biochem. 2009;145:811–818. doi: 10.1093/jb/mvp038. [DOI] [PubMed] [Google Scholar]
- 42.Medeiros NE, Curcio CA. Preservation of ganglion cell layer neurons in age-related macular degeneration. Invest Ophthalmol Vis Sci. 2001;42:795–803. [PubMed] [Google Scholar]
- 43.Mazzoni F, Novelli E, Strettoi E. Retinal ganglion cells survive and maintain normal dendritic morphology in a mouse model of inherited photoreceptor degeneration. J Neurosci. 2008;28:14282–14292. doi: 10.1523/JNEUROSCI.4968-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Huang Y, Cideciyan AV, Papastergiou GI, et al. Relation of optical coherence tomography to microanatomy in normal and rd chickens. Invest Ophthalmol Vis Sci. 1998;39:2405–2416. [PubMed] [Google Scholar]
- 45.Jacobson SG, Cideciyan AV. Treatment possibilities for retinitis pigmentosa. N Engl J Med. 2010;363:1669–1671. doi: 10.1056/NEJMcibr1007685. [DOI] [PubMed] [Google Scholar]
- 46.Aleman TS, Cideciyan AV, Sumaroka A, et al. Retinal laminar architecture in human retinitis pigmentosa caused by Rhodopsin gene mutations. Invest Ophthalmol Vis Sci. 2008;49:1580–1590. doi: 10.1167/iovs.07-1110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sakami S, Maeda T, Bereta G, et al. Probing mechanisms of photoreceptor degeneration in a new mouse model of the common form of autosomal dominant retinitis pigmentosa due to P23H opsin mutations. J Biol Chem. 2011;286:10551–10567. doi: 10.1074/jbc.M110.209759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Stone EM, Luo X, Heon E, et al. Autosomal recessive retinitis pigmentosa caused by mutations in the MAK gene. Invest Ophthalmol Vis Sci. 2011;52:9665–9673. doi: 10.1167/iovs.11-8527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cideciyan AV, Swider M, Aleman TS, et al. Reduced-illuminance autofluorescence imaging in ABCA4-associated retinal degenerations. J Opt Soc Am A Opt Image Sci Vis. 2007;24:1457–1467. doi: 10.1364/josaa.24.001457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Aleman TS, Soumittra N, Cideciyan AV, et al. CERKL mutations cause an autosomal recessive cone-rod dystrophy with inner retinopathy. Invest Ophthalmol Vis Sci. 2009;50:5944–5954. doi: 10.1167/iovs.09-3982. [DOI] [PubMed] [Google Scholar]
- 51.Herrera W, Aleman TS, Cideciyan AV, et al. Retinal disease in Usher syndrome III caused by mutations in the clarin-1 gene. Invest Ophthalmol Vis Sci. 2008;49:2651–2660. doi: 10.1167/iovs.07-1505. [DOI] [PubMed] [Google Scholar]
- 52.Maeda T, Cideciyan AV, Maeda A, et al. Loss of cone photoreceptors caused by chromophore depletion is partially prevented by the artificial chromophore pro-drug, 9-cis-retinyl acetate. Hum Mol Genet. 2009;18:2277–2287. doi: 10.1093/hmg/ddp163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Aleman TS, Cideciyan AV, Aguirre GK, et al. Human CRB1-associated retinal degeneration: comparison with the rd8 Crb1-mutant mouse model. Invest Ophthalmol Vis Sci. 2011;52:6898–6910. doi: 10.1167/iovs.11-7701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Cideciyan AV, Rachel RA, Aleman TS, et al. Cone photoreceptors are the main targets for gene therapy of NPHP5 (IQCB1) or NPHP6 (CEP290) blindness: generation of an all-cone Nphp6 hypomorph mouse that mimics the human retinal ciliopathy. Hum Mol Genet. 2011;20:1411–1423. doi: 10.1093/hmg/ddr022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jacobson SG, Cideciyan AV, Aleman TS, et al. Human retinal disease from AIPL1 gene mutations: foveal cone loss with minimal macular photoreceptors and rod function remaining. Invest Ophthalmol Vis Sci. 2011;52:70–79. doi: 10.1167/iovs.10-6127. [DOI] [PubMed] [Google Scholar]
- 56.Mustafi D, Kevany BM, Genoud C, et al. Defective photoreceptor phagocytosis in a mouse model of enhanced S-cone syndrome causes progressive retinal degeneration. FASEB J. 2011;25:3157–3176. doi: 10.1096/fj.11-186767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Aguirre GK, Komaromy AM, Cideciyan AV, et al. Canine and human visual cortex intact and responsive despite early retinal blindness from RPE65 mutation. PLoS Med. 2007;4:e230. doi: 10.1371/journal.pmed.0040230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cideciyan AV, Aleman TS, Jacobson SG, et al. Centrosomal-ciliary gene CEP290/NPHP6 mutations result in blindness with unexpected sparing of photoreceptors and visual brain: implications for therapy of Leber congenital amaurosis. Hum Mutat. 2007;28:1074–1083. doi: 10.1002/humu.20565. [DOI] [PubMed] [Google Scholar]
- 59.Vandenberghe LH, Bell P, Maguire AM, et al. AAV9 targets cone photoreceptors in the nonhuman primate retina. PLoS One. 2013;8:e53463. doi: 10.1371/journal.pone.0053463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kay CN, Ryals RC, Aslanidi GV, et al. Targeting photoreceptors via intravitreal delivery using novel, capsid-mutated AAV vectors. PLoS One. 2013;8:e62097. doi: 10.1371/journal.pone.0062097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chen X, Hsueh HA, Werblin FS. Amacrine-to-amacrine cell inhibition: Spatiotemporal properties of GABA and glycine pathways. Vis Neurosci. 2011;28:193–204. doi: 10.1017/S0952523811000137. [DOI] [PubMed] [Google Scholar]
- 62.Werblin FS. Six different roles for crossover inhibition in the retina: correcting the nonlinearities of synaptic transmission. Vis Neurosci. 2010;27:1–8. doi: 10.1017/S0952523810000076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Werblin F, Roska B, Balya D. Parallel processing in the mammalian retina: lateral and vertical interactions across stacked representations. Prog Brain Res. 2001;131:229–238. doi: 10.1016/s0079-6123(01)31019-1. [DOI] [PubMed] [Google Scholar]
- 64.Lin B, Koizumi A, Tanaka N, Panda S, Masland RH. Restoration of visual function in retinal degeneration mice by ectopic expression of melanopsin. Proc Natl Acad Sci. 2008;105:16009–16014. doi: 10.1073/pnas.0806114105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Koizumi A, Tanaka KF, Yamanaka A. The manipulation of neural and cellular activities by ectopic expression of melanopsin. Neurosci Res. 2013;75:3–5. doi: 10.1016/j.neures.2012.07.010. [DOI] [PubMed] [Google Scholar]
- 66.Govardovskii VI, Fyhrquist N, Reuter T, Kuzmin DG, Donner K. In search of the visual pigment template. Vis Neurosci. 2000;17:509–528. doi: 10.1017/s0952523800174036. [DOI] [PubMed] [Google Scholar]