Abstract
OBJECTIVE
: The I/D polymorphism of angiotensin-converting enzyme (ACE) and R577X of the α-actinin-3 (ACTN3) is related to changes in skeletal muscle function. The aim of this study was to evaluate the distribution of these polymorphisms in a family with multiple members with adolescent idiopathic scoliosis (AIS).
METHODS
: Evaluated 25 subjects from a family with multiple members with AIS, by collecting 10mL of blood for DNA isolation. The genotyping of the I/D polymorphism of the ACE gene and the R577X of the ACTN3 gene was performed using two specific primers to classify individuals as homozygous or heterozygous.
RESULTS
: Regarding the ACE polymorphism it was found that 19 (76%) subjects were DD and 6 (24%) ID. The prevalence of the D allele was 88% and the I allele was 12%. Regarding the ACTN3 polymorphism there were 6 subjects RR (24%), 11 RX (44%) and 8 XX (32%). The prevalence of the R allele was 23 (46%) and the X allele was 27 (54%).
CONCLUSION
: There was a difference between the distribution of the polymorphism of ACE and ACTN3 in the family studied. When assessing the ACE polymorphism a higher prevalence of the D allele was observed as compared with the I allele. Level of Evidence III, Cross-sectional, Clinical Trial.
Keywords: Scoliosis, Polymorphism, genetic, Spine
INTRODUCTION
Idiopathic scoliosis is one of the most frequent diseases of the spinal column, with reports of its incidence in 0.5% to 10% of the world population. 1 It isdefined as lateral spinal column deviation associated with rotation, with no known cause, in individuals who do not present neurological/muscle disorders or other diseases. 2 The radiographic study does not demonstrate vertebral alterations and presents curves of more than 10 degrees, measured using the Cobb method. 3 The deformity progresses during the growth phase, and can be classified in three categories, according to the age at which the deformity is noticed: infantile under the age of three, juvenile between three and ten years of age (or beginning of puberty) and adolescent when it appears after age ten or after puberty. 4 , 5
The prevalence of idiopathic scoliosis in radiographic studies with school populations ranged between 0.3% and 15.3%, yet when only curves above ten degrees were considered, the rates dropped to values between 1.5% and 3%. In curves above 20 degrees Cobb, the prevalence is between 0.3% and 0.5% and in curves above 30 degrees the rate is from 0.2% to 0.3%. 5
The etiology of idiopathic scoliosis is still unknown. Nowadays several factors are suggested as causal, including deviation of the growth pattern, neuromuscular or connective tissue alterations, asymmetric growth of the limbs and trunk, alterations of the sagittal configuration of the spine and factors linked to the environment. 5 - 7
Hereditary factors also are determinants in the etiology of this disease, but the inheritance pattern is not yet known. Multifactorial inheritances linked to the X chromosome and autosomal dominant inheritances were also suggested. 2 , 5 , 6 , 8 , 9 Various authors, by means of family studies, have suggested possible chromosomal regions related to the etiology of idiopathic scoliosis. 2 , 6 , 8 - 11 In 2010 Wajchenberg et al. 12 failed to find, in a genetic linkage study of a family with multiple members affected, a particular chromosomal region related to AIS.
Nowadays studies of genetic polymorphisms are carried out on patients with AIS in an attempt to correlate alterations in certain proteins that might be related to the disease. The polymorphisms reported were related to the genes MATN1 in region 1p35 and CHD7 in region 8q12.1 and IL-6. 13 , 14
It is known that the human genetic map contains at least 170 variant sequences of genes and of genetic markers that are related to the phenotypes of physical performance and of fitness related to health. These genes include the angiotensin-converting enzyme (ACE) gene, which is located on chromosome 17q23, composed of 26 exons and 25 introns. A common genetic variant in the ACE gene was described as absence or deletion (D allele) and presence or insertion (I allele) of 287 base pairs in intron 16. 15 Genetic association studies have associated this polymorphism with health, sports and also with the genesis and maintenance of various diseases. Recent studies show that the I allele is more frequent in endurance athletes, while the D allele appears more often in strength and muscular explosion athletes. Studies of our group have been showing that genotype DD presents greater activity of plasma ACE when compared with genotypes II and ID. 16
Another polymorphism related to physical performance, by means of the basic composition of the muscle fibers, is related to the α-actinin protein. It is a component of the sarcomeric Z-line, belonging to the family of the actin-bonding proteins, important in the anchoring of actin myofilaments and maintenance of the myofibrillar arrangement.Four genes for α-actinin were described in humans (ACTN1, 2, 3 and 4), while isoforms 2 and 3 are constituents of the muscular cytoskeleton. 17
It is known that the ACTN3 isoform is specific to the fast twitch fibers (type II) responsible for the generation of contractile force at high speed. It was identified in the ACTN3 gene that there is a nucleotide C → T transition in position 1,747 in exon 16, i.e., a mutation resulting in the conversion of the arginine amino acid (R) into a premature stop codon (X) at residue 577 (R577X).The lack of the protein in XX individuals interestingly fails to present a pathological phenotype such as muscular dystrophy or myopathies. However, a reduction was observed in all the muscles that normally express ACTN3 in knockout animals for the ACTN3 gene, and also in humans. 17
In this study, we examine the possible association between polymorphisms of the ACE gene and of ACTN 3 in a family of individuals containing multiple members with AIS.
METHODS
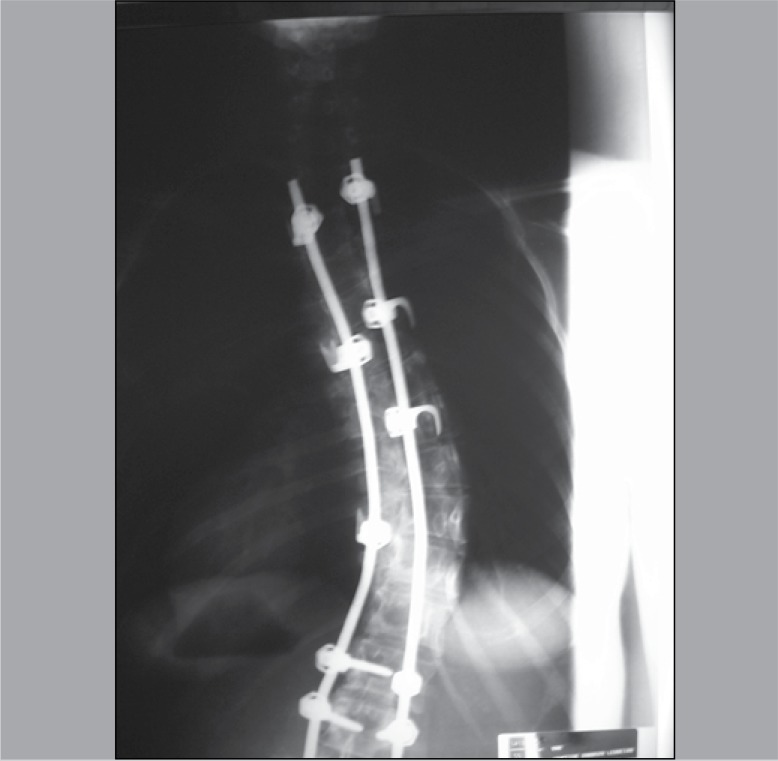
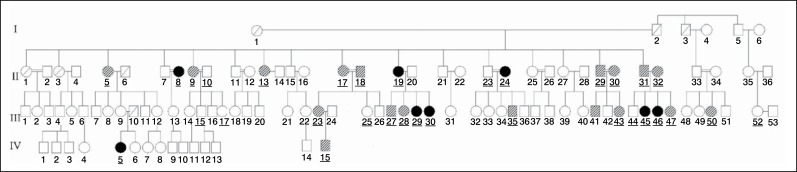
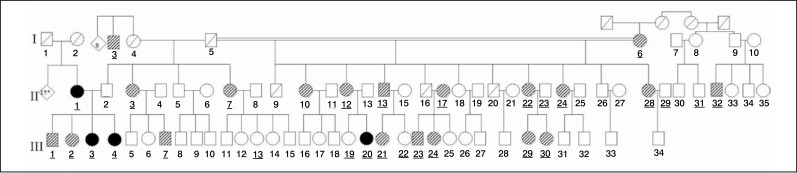
A cross-sectional study of the analysis of polymorphism of ACE and of ACTN 3 in individuals from a family with multiple members affected by idiopathic scoliosis related to a patient (proband) operated by the Spinal Column Group of the Department of Orthopedics and Traumatology of Universidade Federal de São Paulo. (Figure 1) Such patient has adolescent idiopathic scoliosis with right thoracic curve of 75 degrees, measured by the Cobb method, before surgery. Due to the large number of members, the family was divided into maternal (Figure 2) and paternal. (Figure 3)
Figure 1. PA radiography of postoperative spinal column of patient IBF 1.
Figure 2. Genogram of the maternal family of patient IBF1.
Figure 3. Genogram of the paternal family of patient IBF1.
The sample included 25 individuals, 16 of whom were female and 9 male. This family was evaluated clinically and by radiography with frontal view of the spinal column. This was followed by the drawing of 10 ml of blood for DNA extraction, after authorization by means of the Informed Consent Form (ICF). 16 The total DNA of the leukocytes was extracted using ChargeSwitch(r) gDNABlood Kits (Invitrogen(tm)) employed for purification of genomic DNA from small volumes of human blood, following the steps recommended by the manufacturer.
The genotyping of the I/D polymorphism of the ACE gene was conducted using a system of 2 specific primers (Primer ACES 5' -CTGGAGACCACTCCCATCCTTTCT-3' ), (Primer ACEAS 5' -GATGTGGCCATCACATTCGTCAGAT- 3' ) that open the sequence where polymorphism occurs in the gene, with the purpose of amplifying them and classifying the individuals as II or DD homozygous or as ID heterozygous.
The reagents that were used in the first PCR for identification of the genotypes were: 10µL of the extracted DNA, 5µL of PCR buffer (10x), 2.5µLof MgCl2 (50 mM), 1.0µL of dNTPs (10 mM), 0.5µL of ACE primer, 0.5µL of ACEAS primer, 0.5 of Taq DNA polymerase (5 U/µL), 5µL of autoclaved milli-Q H2O, for a final 25µL.
The reagents that were used in the second PCR for confirmation of the genotype of the DD volunteers, with the use of the internal primer (Internal primer ACE 5' -GTCTCGATCTCCTGACCTCGTG-3' ), (Primer ACEAS 5' -GATGTGGCCATCACATTCGTCAGAT- 3' ): 10µL of the extracted DNA, 5µL of PCR buffer (10x), 2.5µL of MgCl2 (50 mM), 1.0µL of dNTPs (10 mM), 0.5µL of ACESint primer,
0.5µL of ACEAS primer, 0.5µL of Taq DNA polymerase (5 U/µL), 5µL of autoclaved milliQ H2O, for a final 25µL.
The following condition was used for the PCR: Start at 95°C for 5 minutes, followed by 95°C for 45 seconds, 60°C for 45 seconds, 72°C for 45 seconds, returning to 95°C for 45 seconds 35 times, 72°C for 7 minutes and finishing with 4°C for an indefinite time.
The genotyping of the alleles of the ACTN3R577X polymorphism was performed with a system of two specific primers (Primer Actn3f - 5' -CAG CTGGAGGATGGCCTGG- 3' ), (Primer Actn3r - 5' -GTC CAG GTATTT CTC TGCCAC C- 3' ) that opened the sequence where polymorphism occurs in the gene, with the purpose of amplifying them and classifying the individuals as RR, RX and XX.
The reagents that were used in the PCR for genotype identification after the extraction of 10µL of the DNA were: 2.5µL of PCR buffer (10x), 1.25µL of MgCl2 (50 mM), 1.0µL of dNTPs (10 mM), 0.5µL of Actn3f primer, 0.5µL of Actn3r primer, 0.5µL of Taq DNA polymerase (5 U/µL), 5µL of autoclaved milliQ H2O, 25µL total.
The following condition was used for the PCR: Start at 95°C for 7 minutes, followed by 95°C for 45 seconds, 60°C for 45 seconds, 72°C for 45 seconds, returning to 95°C for 45 seconds 35 times, 72°C for 10 minutes and finishing with 4°C for an indefinite time. The fragments amplified in the reactions were analyzed by electrophoresis in 4% agarose gel containing SYBR(r) Safe DNA gel stain (Invitrogen) (0.2µg/mL). After a check of the DNA integrity and of the gene amplification in the PCR, digestion was promoted by the DdeI restriction enzyme, and it was incubated at 37°C for 2 hours, using 8µL of DNA (≈6µg) of the sample, 1µL of REACT 3 buffer (10X), 1µL of DdeI (10 U/µL) for a final volume of 10µL. Once again, the result was analyzed by electrophoresis, this time visualized in 3% agarose gel containing SYBR(r) Safe DNA gel stain (Invitrogen) (0.2µg/mL).
RESULTS
Among the 25 family members, 22 had some degree of scoliosis and nine had curves with 15 or more degrees of deformity. Individuals with curves above 15 degrees, measured by the Cobb method, were considered affected (A) by the disease. The members with a curve between 5 and 14 degrees were considered suspect (S) and normal (N) when they had a curve of up to 4 degrees, or absence of rotation.
As regards ACE polymorphism, it was found that 19 individuals were DD (76%) and 6 ID (24%). The prevalence of the D allele was 88% while that of the Iallele was 12%. As regards ACTN3 polymorphism, it was found that 6 individuals were RR (24%), 11 RX (44%) and 8 XX (32%). The prevalence of the R allele was 23 (46%) and that of the X allele was 27 (54%).
The characteristics of each family member and their location inside the family are described in Table 1.
Table 1. Characteristics of the members of the family studied.
| Patient | Age | Sex | Disease | Curve | Angle | Genogram | ACE | ACTN 3 |
|---|---|---|---|---|---|---|---|---|
| 1 | 50 | F | A | R Thoracic | 10 | II-12 maternal | DD | RX |
| 2 | 13 | F | A | R Thoracic | 22 | III-20 paternal | DD | RR |
| 3 | 39 | F | S | R Thoracolumbar | 5 | II-12 paternal | ID | RR |
| 4 | 18 | F | A | R Thoracic and L Lumbar | 12/12 | III-23 maternal | DD | RX |
| 5 | 23 | F | A | R Thoracolumbar | 75 | III-3 paternal and III- 29 maternal | DD | XX |
| 6 | 27 | F | S | R Thoracic and L Lumbar | 10/14 | III-21 maternal | DD | RR |
| 7 | 26 | F | S | R Thoracolumbar | 5 | III-22 maternal | DD | XX |
| 8 | 20 | F | A | L Thoracolumbar | 30 | III-4 paternal and II-33 maternal | DD | XX |
| 9 | 52 | F | A | R Thoracic | 20 | II-paternal and II-19 maternal | DD | RX |
| 10 | 22 | F | A | R Thoracic and L Lumbar | 16/10 | III-35 maternal | DD | RX |
| 11 | 28 | M | S | R Thoracic | 13 | III-20 maternal | DD | XX |
| 12 | 30 | M | N | Absent | 0 | III 19-maternal | DD | XX |
| 13 | 56 | M | S | R Thoracic | 10 | II-2 paternal and II-20 maternal | ID | RX |
| 14 | 33 | M | S | L Thoracolumbar | 10 | II-32 paternal | II-32 paternal | RR |
| 15 | 52 | F | A | L Lumbar and R Thoracic | 25 and 20 | II-8 maternal | DD | XX |
| 16 | 40 | M | S | R Thoracic | 10 | II-31 maternal | DD | RX |
| 17 | 14 | F | A | R Thoracic and L Lumbar | 17 and 10 | III-49 maternal | ID | RX |
| 18 | 17 | F | A | L Lumbar and R Thoracic | 22 and 15 | III-48 maternal | ID | RX |
| 19 | 11 | F | S | R Thoracolumbar | 10 | III-50 maternal | ID | XX |
| 20 | 16 | M | S | L Lumbar and R Thoracic | 6 and 4 | III-38 maternal | DD | RX |
| 21 | 42 | F | A | L Thoracic and R Lumbar | 20 and 12 | II-24 maternal | DD | RX |
| 22 | 29 | F | S | L Thoracolumbar | 8 | III-2 paternal and III-31 maternal | DD | RR |
| 23 | 30 | M | S | L Thoracolumbar | 5 | III-1 paternal and III-30 maternal | DD | RX |
| 24 | 45 | M | N | absent | 0 | II-23 maternal | DD | XX |
| 25 | 19 | M | N | absent | 0 | III-34 maternal | ID | RR |
DISCUSSION
The etiology of adolescent idiopathic scoliosis remains unknown. Today different factors have been suggested, such as deviation of the growth pattern, neuromuscular or connective tissue alterations, asymmetric growth of limbs and trunk, alterations of the sagittal configuration of the spine and factors related to the environment. 4 , 6 , 7
In 1997 Meier et al. 18 analyzed muscular reaction in patients using a jacked for the treatment of adolescent idiopathic scoliosis, by means of muscle biopsies. These authors cited the cause of spinal distortion as paraspinal muscle imbalance and reported that in the curve convexity in the dorsal region, the distribution of muscular fibers follows the normal pattern, with type I fibers predominating. A decrease of type I fibers was noted on concavity, with substitution by type IIC fibers. It was also reported that the patients with more accentuated curves had fewer type I muscle fibers, both on the convexity and on the concavity in comparison to the patients with less severe curves. It was concluded in that study that the use of the jacket made the muscle alteration noted at the apex of deformity, distributed through the scoliosis region, suggesting that the muscle alterations found in scoliosis are in fact secondary.
The inheritance pattern for the transmission of adolescent idiopathic scoliosis is not yet fully known. Multifactorial inheritances linked to the X chromosome and autosomal dominant inheritances have already been suggested. 1 , 2 , 4 , 7 , 9 As of 2000, genetic linkage studies involving families with multiple members affected, suggested possible chromosomal regions related to the etiology of adolescent idiopathic scoliosis. Using this technique, Wise et al., 8 in 2000, described the first regions related to adolescent idiopathic scoliosis, in the chromosomes 6p,10q and 18q, when studying a large family with seven members affected by this disease.
Some authors found regions linking adolescent idiopathic scoliosis to chromosome 17. 2 , 9 , 11 Salehi et al. 9 investigated three generations of a family of Italian origin with 11 members affected, who presented curves between 10 and 20 degrees and autosomal dominant inheritance pattern with complete penetrance. This study mapped a region of approximately 20 cm on chromosome 17p11 linked to idiopathic scoliosis. Another study conducted by Clough et al., 11 in 2010, confirmed, by means of the linkage study, the relationship between the region described by Salehi et al. 9 and familial idiopathic scoliosis, when studying 17 families with one member (proband) affected, of the male sex, who required surgical treatment to correct the deformity. Ocaka et al. found chromosomal regions in chromosomes 9q34 and 17q25. 2 Wajchenberget al. 12 did not find any connection when studying a family from upstate Paraíba, with nine members affected.
The greatest uncertainty when conducting genetic linkage studies, for adolescent idiopathic scoliosis, occurs when indicating which patients are affected, since the measurement of the curve by the Cobb method, described in 1948, is dependent on the examiner and minor variations in the measurement can interfere in the individual' s classification as affected or normal. Thus individuals with a difference of only two degrees (one with nine and another with 11 degrees) are classified differently. 3
Another alternative for genetic studies is the use of molecular markers, chosen previously and with known positioning in the human genome, such as the SNPs. Authors such as Chen et al. 13 and Aulisa et al. 14 conducted genetic polymorphism studies in an attempt to relate adolescent idiopathic scoliosis with alterations in particular genes.
This study was aimed at relating ACE and ACTN3 polymorphisms to AIS. The ACE gene is located on chromosome 17q23, close to region 17q25, described by Ocaka et al., 2 in 2008 and the I/D polymorphism of the ACE gene is still one of the most widely studied genetic markers. Recent studies show that the I allele is more frequent in endurance athletes, while the D allele appears more often in strength and muscular explosion athletes. Accordingly, the variability of this polymorphism can affect the performance of certain muscle groups that act as a means of support for the spinal column. 16
The paraspinal musculature plays a trunk support and movement role, undergoing suffering significant changes in patients with AIS, who have trunk deformity in the three planes and significant rotation. The multifidus muscles are rotators of the spinal column and just like the longissimus has as a histochemical pattern, in normal individuals (20 to 30 years of age), predominant type I fibers (57-62%).Type II-A fibers appeared from 20 to 22% and those of type IIB are present in the proportion of 18 to 22%. 19 These muscles, as well as the soleus, tibialis anterior, adductor pollicis, diaphragm and extraocular muscles, have a higher energy demand than the others, with a fundamentally aerobic metabolism, related to type I fibers.
The reduction of ACE activity, associated with the I allele, has been reported with better muscular efficiency and consequently greater muscle resistance capacity in healthy individuals. In 2003, Zhang et al. 20 associated muscular efficiency with the superior presence of type I fiber, influenced by the I allele, while the DD genotype was allegedly related to the presence of type II fibers, proving maximum strength in the muscles where this type of fiber predominates, such as the quadriceps. Moreover, ACE is responsible for the genesis of the vasoconstrictor peptide, angiotensin II, and for the degradation of bradykinin.Thus high concentrations of ACE (related to the "D" allele) are apparently linked to reduced levels of bradykinin, capturing less glucose and producing a decrease in the muscle blood flow, consequently impairng muscle groups where type I muscle fibers predominate, such as the multifidus and erector spinae muscles.
Predominance of the D allele was noted in the family studied (88%). Such a fact can have a negative effect on the paraspinal musculature, mainly the multifidus muscles, which, as reported previously, have a larger quantity of type I muscle fibers, with aerobic characteristics, and anti-gravitational resistance function.
The distribution of these variations in the population is 25% II, 50% ID and 25% DD. None of the members of the aforesaid family were found to be genotype II, and homozygotes predominated for the D allele (76%). This result could be explained by the fact that we studied only one family, where there is less genetic variability. Hence we decided to study the polymorphism of another protein to evaluate its distribution in this family. Moreover, we could also observe the prevalence of another polymorphism that could influence muscle performance opposite the paraspinal musculature, i.e., with a predominance of type II fibers (anaerobic metabolism).
Knowing of its influence on type II muscle fibers, the ACTN3 polymorphism was studied next. The individuals homozygous for the 577X allele (XX) do not express α-actinin-3, yet the deficiency of this proteindoes not result in a pathological phenotype such as muscular dystrophy or myopathies, suggesting that the ACTN2 isoform (81% ofhomology in the amino acid sequence) could compensate the absence of the α-actinin-3. 17 It is known, however, that individuals who express the ACTN3 gene (genotypes RR or RX) can present an advantage in activities that require explosion and muscle strength when compared with individuals with genotype XX.
In the abovementioned family, unlike the polymorphism of ACE, greater balance was found in the distribution between RR (24%) and XX (32%) homozygotes and RX heterozygotes (44%), as well as the R (46%) and X (54%) alleles, with distribution similar to the control population.
These observations may be an indication that there is a relationship between ACE polymorphism and AIS, due to the prevalence of the DD genotype and of the D allele, according to the data found.
CONCLUSION
In this study a difference was observed between the distribution of ACE polymorphism and ACTN3 in a family with multiple members diagnosed with adolescent idiopathic scoliosis. When evaluating the polymorphism of ACE we did not find any individuals homozygous for genotype II among the members, which led to a greater prevalence of the D allele in comparison to the I allele.
Footnotes
Study conducted in the Spinal Column Group of the Department of Orthopedics and Traumatology of Universidade Federal de São Paulo, São Paulo, SP, Brazil.
Citation: Wajchenberg M, Luciano RP, Araújo RC, Martins DE, Puertas EB, Almeida SS. Polymorphism of the ace gene and the α-actinin-3 gene in adolescent idiopathic scoliosis. Acta Ortop Bras. [online]. 2013;21(3):170-4. Available from URL: http://www.scielo.br/aob.
REFERENCES
- 1.Lonstein JE. Idiopathicscoliosis. In: Lonstein JE, Bradfud DS, Winter RB, Ogilive JW, editors. Moe's textbook of scoliosis and other spinal deformities. 3rd ed. Philadelphia: Saunders; 1995. pp. 219–256. [Google Scholar]
- 2. Ocaka L, Zhao C, Reed JA, Ebenezer ND, Brice G, Morley T, et al. Assignment of two loci for autosomal dominant adolescent idiopathic scoliosis to chromosomes 9q31.2-q34.2 and 17q25.3-qtel. J Med Genet. 2008;45(2):87–92. doi: 10.1136/jmg.2007.051896. [DOI] [PubMed] [Google Scholar]
- 3. Cobb JR. Outline for the study of scoliosis. Am AcadOrthopSurgInstr Course Lect. 1948;5:261–265. [Google Scholar]
- 4. Wynne-Davies R. Familial (idiopathic) scoliosis. A family survey. J Bone Joint Surg Br. 1968;50(1):24–30. [PubMed] [Google Scholar]
- 5. Lonstein JE. Adolescent idiopathic scoliosis. Lancet. 1994;344(8934):1407–1412. doi: 10.1016/s0140-6736(94)90572-x. [DOI] [PubMed] [Google Scholar]
- 6. Alden KJ, Marosy B, Nzegwu N, Justice CM, Wilson AF, Miller NH. Idiopathic scoliosis: identification of candidate regions on chromosome 19p13. Spine (Phila Pa 1976) 2006;31(16):1815–1819. doi: 10.1097/01.brs.0000227264.23603.dc. [DOI] [PubMed] [Google Scholar]
- 7. Wajchenberg M, Puertas EB, Zatz M. Estudo da prevalência da escoliose idiopática do adolescente em pacientes brasileiros. Coluna. 2005;4(3):127–131. [Google Scholar]
- 8. Wise CA, Barnes R, Gillum J, Herring JA, Bowcock AM, Lovett M. Localization of susceptibility to familial idiopathic scoliosis. Spine (Phila Pa 1976) 2000;25(18):2372–2380. doi: 10.1097/00007632-200009150-00017. [DOI] [PubMed] [Google Scholar]
- 9. Salehi LB, Mangino M, De Serio S, De Cicco D, Capon F, Semprini S, et al. Assignment of a locus for autosomal dominant idiopathic scoliosis (IS) to human chromosome 17p11. Hum Genet. 2002;111(4-5):401–404. doi: 10.1007/s00439-002-0785-4. [DOI] [PubMed] [Google Scholar]
- 10. Justice CM, Miller NH, Marosy B, Zhang J, Wilson AF. Familial idiopathic scoliosis: evidence of an X-linked susceptibility locus. Spine (Phila Pa 1976) 2003;28(6):589–594. doi: 10.1097/01.BRS.0000049940.39801.E6. [DOI] [PubMed] [Google Scholar]
- 11. Clough M, Justice CM, Marosy B, Miller NH. Males with familial idiopathic scoliosis: a distinct phenotypic subgroup. Spine (Phila Pa 1976) 2010; 35(2):162–168. doi: 10.1097/BRS.0b013e3181b7f1a7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Wajchenberg M, Lazar M, Cavaçana N, Martins DE, Licinio L, Puertas EB, et al. Genetic aspects of adolescent idiopathic scoliosis in a family with multiple affected members: a research article. Scoliosis. 2010;7;5:7–7. doi: 10.1186/1748-7161-5-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Chen Z, Tang NL, Cao X, Qiao D, Yi L, Cheng JC, et al. Promoter polymorphism of matrilin-1 gene predisposes to adolescent idiopathic scoliosis in a Chinese population. Eur J Hum Genet. 2009;17(4):525–532. doi: 10.1038/ejhg.2008.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Aulisa L, Papaleo P, Pola E, Angelini F, Aulisa AG, Tamburrelli FC, et al. Association between IL-6 and MMP-3 gene polymorphisms and adolescent idiopathic scoliosis: a case-control study. Spine (Phila Pa 1976) 2007;32(24):2700–2702. doi: 10.1097/BRS.0b013e31815a5943. [DOI] [PubMed] [Google Scholar]
- 15. Wolfarth B, Bray MS, Hagberg JM, Pérusse L, Rauramaa R, Rivera MA, et al. The human gene map for performance and health-related fitness phenotypes: the 2004 update. Med Sci Sports Exerc. 2005;37(6):881–903. [PubMed] [Google Scholar]
- 16. Almeida SS, Barros CC, Moraes MR, Russo FJ, Haro AS, Rosa TS, et al. Plasma Kallikrein and Angiotensin I-converting enzyme N- and C-terminal domain activities are modulated by the insertion/deletion polymorphism. Neuropeptides. 2010;44(2):139–143. doi: 10.1016/j.npep.2009.12.003. [DOI] [PubMed] [Google Scholar]
- 17. North KN, Yang N, Wattanasirichaigoon D, Mills M, Easteal S, Beggs AH. A common nonsense mutation results in alpha-actinin-3 deficiency in the general population. Nat Genet. 1999;21(4):353–354. doi: 10.1038/7675. [DOI] [PubMed] [Google Scholar]
- 18. Meier MP, Klein MP, Krebs D, Grob D, Müntener M. Fiber transformations in multifidus muscle of young patients with idiopathic scoliosis. Spine (Phila Pa 1976) 1997;22(20):2357–2364. doi: 10.1097/00007632-199710150-00008. [DOI] [PubMed] [Google Scholar]
- 19. Thorstensson A, Carlson H. Fibre types in human lumbar back muscles. Acta Physiol Scand. 1987;131(2):195–202. doi: 10.1111/j.1748-1716.1987.tb08226.x. [DOI] [PubMed] [Google Scholar]
- 20. Zhang B, Tanaka H, Shono N, Miura S, Kiyonaga A, Shindo M, et al. The I allele of the angiotensin-converting enzyme gene is associated with an increased percentage of slow-twitch type I fibers in human skeletal muscle. Clin Genet. 2003;63(2):139–144. doi: 10.1034/j.1399-0004.2003.00029.x. [DOI] [PubMed] [Google Scholar]