Abstract
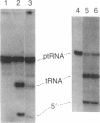
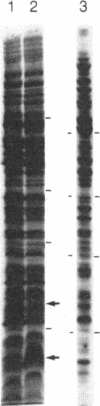
M1 RNA, the catalytic subunit of Escherichia coli RNase P, has been covalently linked at its 3' terminus to agarose beads. Unlike M1 RNA, which is active in solution in the absence of the protein component (C5) of RNase P, the RNA linked to the beads is active only in the presence of C5 protein. Affinity chromatography of crude extracts of E. coli on a column prepared from the beads to which the RNA has been crosslinked results in the purification of C5 protein in a single step. The protein has been purified in this manner from cells that contain a plasmid, pINIIIR20, which includes the gene that codes for C5 protein. A 6-fold amplification of the expression of C5 protein is found in these cells after induction as compared to cells that do not harbor the plasmid.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burrell H. R., Horowitz J. Binding of ribosomal proteins to RNA covalently coupled to agarose. Eur J Biochem. 1977 May 16;75(2):533–544. doi: 10.1111/j.1432-1033.1977.tb11554.x. [DOI] [PubMed] [Google Scholar]
- Cech T. R., Bass B. L. Biological catalysis by RNA. Annu Rev Biochem. 1986;55:599–629. doi: 10.1146/annurev.bi.55.070186.003123. [DOI] [PubMed] [Google Scholar]
- Davydov R. M., Vesterman B. G., Genkin M. V., Bliumenfel'd L. A., Krylov O. V. Vliianie immobilizatsii na konformatsionnuiu dinamiku i reaktsionnuiu sposobnost' tsitokhroma C pri okislitel'no-vosstanovitel'nykh prevrashcheniiakh. Dokl Akad Nauk SSSR. 1981;256(2):491–494. [PubMed] [Google Scholar]
- Dijk J., Littlechild J. Purification of ribosomal proteins from Escherichia coli under nondenaturing conditions. Methods Enzymol. 1979;59:481–502. doi: 10.1016/0076-6879(79)59109-5. [DOI] [PubMed] [Google Scholar]
- Fahnestock S. R., Nomura M. Activity of ribosomes containing 5S RNA with a chemically modified 3'-terminus. Proc Natl Acad Sci U S A. 1972 Feb;69(2):363–365. doi: 10.1073/pnas.69.2.363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardiner K. J., Marsh T. L., Pace N. R. Ion dependence of the Bacillus subtilis RNase P reaction. J Biol Chem. 1985 May 10;260(9):5415–5419. [PubMed] [Google Scholar]
- Ghrayeb J., Kimura H., Takahara M., Hsiung H., Masui Y., Inouye M. Secretion cloning vectors in Escherichia coli. EMBO J. 1984 Oct;3(10):2437–2442. doi: 10.1002/j.1460-2075.1984.tb02151.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gold H. A., Altman S. Reconstitution of RNAase P activity using inactive subunits from E. coli and HeLa cells. Cell. 1986 Jan 31;44(2):243–249. doi: 10.1016/0092-8674(86)90758-0. [DOI] [PubMed] [Google Scholar]
- Guerrier-Takada C., Altman S. Catalytic activity of an RNA molecule prepared by transcription in vitro. Science. 1984 Jan 20;223(4633):285–286. doi: 10.1126/science.6199841. [DOI] [PubMed] [Google Scholar]
- Guerrier-Takada C., Altman S. M1 RNA with large terminal deletions retains its catalytic activity. Cell. 1986 Apr 25;45(2):177–183. doi: 10.1016/0092-8674(86)90381-8. [DOI] [PubMed] [Google Scholar]
- Guerrier-Takada C., Altman S. Structure in solution of M1 RNA, the catalytic subunit of ribonuclease P from Escherichia coli. Biochemistry. 1984 Dec 18;23(26):6327–6334. doi: 10.1021/bi00321a006. [DOI] [PubMed] [Google Scholar]
- Guerrier-Takada C., Gardiner K., Marsh T., Pace N., Altman S. The RNA moiety of ribonuclease P is the catalytic subunit of the enzyme. Cell. 1983 Dec;35(3 Pt 2):849–857. doi: 10.1016/0092-8674(83)90117-4. [DOI] [PubMed] [Google Scholar]
- Guerrier-Takada C., Haydock K., Allen L., Altman S. Metal ion requirements and other aspects of the reaction catalyzed by M1 RNA, the RNA subunit of ribonuclease P from Escherichia coli. Biochemistry. 1986 Apr 8;25(7):1509–1515. doi: 10.1021/bi00355a006. [DOI] [PubMed] [Google Scholar]
- Hansen F. G., Hansen E. B., Atlung T. Physical mapping and nucleotide sequence of the rnpA gene that encodes the protein component of ribonuclease P in Escherichia coli. Gene. 1985;38(1-3):85–93. doi: 10.1016/0378-1119(85)90206-9. [DOI] [PubMed] [Google Scholar]
- Kole R., Baer M. F., Stark B. C., Altman S. E. coli RNAase P has a required RNA component. Cell. 1980 Apr;19(4):881–887. doi: 10.1016/0092-8674(80)90079-3. [DOI] [PubMed] [Google Scholar]
- Kruger K., Grabowski P. J., Zaug A. J., Sands J., Gottschling D. E., Cech T. R. Self-splicing RNA: autoexcision and autocyclization of the ribosomal RNA intervening sequence of Tetrahymena. Cell. 1982 Nov;31(1):147–157. doi: 10.1016/0092-8674(82)90414-7. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lerman L. S. A Biochemically Specific Method for Enzyme Isolation. Proc Natl Acad Sci U S A. 1953 Apr;39(4):232–236. doi: 10.1073/pnas.39.4.232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marsh T. L., Pace N. R. Ribonuclease P catalysis differs from ribosomal RNA self-splicing. Science. 1985 Jul 5;229(4708):79–81. doi: 10.1126/science.4012313. [DOI] [PubMed] [Google Scholar]
- Reed R. E., Baer M. F., Guerrier-Takada C., Donis-Keller H., Altman S. Nucleotide sequence of the gene encoding the RNA subunit (M1 RNA) of ribonuclease P from Escherichia coli. Cell. 1982 Sep;30(2):627–636. doi: 10.1016/0092-8674(82)90259-8. [DOI] [PubMed] [Google Scholar]
- Robertson H. D., Altman S., Smith J. D. Purification and properties of a specific Escherichia coli ribonuclease which cleaves a tyrosine transfer ribonucleic acid presursor. J Biol Chem. 1972 Aug 25;247(16):5243–5251. [PubMed] [Google Scholar]
- Stark B. C., Kole R., Bowman E. J., Altman S. Ribonuclease P: an enzyme with an essential RNA component. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3717–3721. doi: 10.1073/pnas.75.8.3717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahara M., Hibler D. W., Barr P. J., Gerlt J. A., Inouye M. The ompA signal peptide directed secretion of Staphylococcal nuclease A by Escherichia coli. J Biol Chem. 1985 Mar 10;260(5):2670–2674. [PubMed] [Google Scholar]
- Ulbrich N., Lin A., Wool I. G. Identification by affinity chromatography of the eukaryotic ribosomal proteins that bind to 5.8 S ribosomal ribonucleic acid. J Biol Chem. 1979 Sep 10;254(17):8641–8645. [PubMed] [Google Scholar]
- Ulbrich N., Wool I. G. Identification by affinity chromatography of the eukaryotic ribosomal proteins that bind to 5 S ribosomal ribonucleic acid. J Biol Chem. 1978 Dec 25;253(24):9049–9052. [PubMed] [Google Scholar]
- Vioque A., Palacián E. Partial reconstitution of 60S ribosomal subunits from yeast. Mol Cell Biochem. 1985 Feb;66(1):55–60. doi: 10.1007/BF00231823. [DOI] [PubMed] [Google Scholar]
- Wilchek M., Miron T., Kohn J. Affinity chromatography. Methods Enzymol. 1984;104:3–55. doi: 10.1016/s0076-6879(84)04082-9. [DOI] [PubMed] [Google Scholar]