Abstract
In this study, we designed a method to manufacture elevated fermented green tea by using Monascus pilosus, which is known as a functional microbe, and observe its antioxidant abilities and quality characteristics. The water-soluble substance (WSS) content of the fermented tea by M. pilosus (FTM) was lower than that of the non-fermented tea (NFT), although the alcohol-insoluble substance (AIS) content of the FTM was higher than that of NTM. On the other hand, the fractionated distilled water-soluble polysaccharide (DWSP), CDTA-soluble polysaccharides (CDSP), sodium carbonate-soluble polysaccharide (SCSP) and KOH soluble hemicellulose (HC) obtained from the AIS of the FTM was markedly higher than that of NFT. In the antioxidant parameters, the electron donating ability of all fractions, except HC, extracted from FTM was higher than that of NFT, and iron chelating ability of all fractions, except CDSP, extracted from FTM was higher than that of NFT. Whereas the DWSP and SCSP obtained from the FTM were higher than that of NFT, the activity of the HC fraction from both NFT and the FTM could not be detected. In addition, the xanthin oxidase (XO) inhibitory activities of the DWSP, CDSP and the SCSP obtained from the NFT were significantly higher than that of FTM, the aldehyde oxidase (AO) inhibitory activities of the DWSP and SCSP extracted from the FTM were markedly higher than that of the NFT. Meanwhile, the acceptance of NFT and FTM had no significant difference, while the quality of aroma, taste and mouthfeel of the FTM was higher than that of NFT. These results suggest that the post-fermented tea by Monascus microorgan-isms may be responsible for functional components as well as contribute to the improvement of the tea quality.
Keywords: post-fermented tea, Monascus pilosus, tea polysaccharides, sensory evaluation, color
INTRODUCTION
Tea is the most widely consumed beverage in the world since tea was planted during the ancient three Kingdom period in Korea, its also been used during palace feasts and Buddhist events (1). Currently, 80% of the cultivated tea in the world is made primarily into fermented tea products (2). Depending on production methods, tea products are generally divided into non-fermented green tea, semi-fermented tea (oolong tea), fermented tea (black tea) and post-fermented tea (Pu-erh tea), and depending on the color, white tea (degree of fermentation 10~20%), yellow tea (lightly fermented), green tea (non-fermented), blue tea (semi-fermented, degree of fermentation 30~60%), red tea (fully fermented tea) and black tea (post-fermented) (3). In addition, the polyphenols in the post-fermented tea, which is not oxidized by tea phytoenzymes, is fermented by micro-organisms and oxidized polyphenols, including new products such as theaflavins and thearubigins, as well as microbial ingredients such as statins (4,5).
Chinese Pu-erh tea, which is a famous post-fermented tea, is divided into dry-stored and wet-stored tea depending on the production method. Dry-stored tea is produced by long-term fermentation under low humidity, and wet- stored tea is relatively quickly fermented under moderate temperatures and high humidity. Microorganisms such as Aspergillus niger, Aspergillus gloucus, Penicillium, Rhizopus, Saccharomyces and Bacterium grow during fermentation (6). On the other hand, microorganisms such as Monascus produce statin-related components including monacolin K, which is an inhibitor of cholesterol biosynthesis in the body (7), as well as producing color ingredients such as rubropuntain and monascin, which exhibit anti-cancer activitites (8). As previously reported, Monascus extracts have an anti-obesity effect (9) as well as a hepatoprotective effect on obese rats induced with a high-fat diet (10). Functional studies and experimentation of tea focuses mainly on polyphenol antioxidant activity. However, a lack of functional studies on tea polysaccharides exists (11).
In this study, we produced high quality fermented tea using Monascus pilosus, and determined the antioxidant quality on the tea extracts of hot water or ethanol from different fermentation periods (12). Also, with each fraction of tea polysaccharides derived from various fermented teas, we determined the antioxidant activity and compared the activity of each fraction.
MATERIALS AND METHODS
Microorganism, medium and tea fermentation
Green tea (Camellia sinensis) leaves from Hadong-gun in Gyeongsangnam-do, Korea were harvested and sun dried in July, 2011. Monascus pilosus IFO4480 were obtained from the Korean Culture Center of Microorganisms (Seoul, Korea) for fermentation. M. pilosus seed culture was prepared, as described previously by Lee et al. (9), and used as a starter. The fermented tea by Monascus (FTM) and the non-fermented tea (NFT) were prepared according to Fig. 1.
Fig. 1.
Preparation procedure of non-fermented tea (NFT) and fermented tea by M. pilosus (FTM).
Color evaluation of tea infusion
Two grams of dried NFT and FTM samples were infused with 200 mL of boiled distilled water for 3 min using a Tea Extractor (Damian Tea Co., Gyeonggi, Korea). The color of the tea infusion was measured for L* (lightness), a* (redness), b* (yellowness) and H∘ (hue angle) using a Chromameter (CR-200, Minolta, Tokyo, Japan).
Content of WSS, AIS, and measurement of brix degree
The content of the water soluble substance (WSS) in tea infusions was measured by weight of the freeze dried tea infusion. Dried tea powder (10 g) was added to 200 mL of 80% ethanol solution and boiled to prepare the alcohol insoluble substance (AIS). The extracted solution and precipitate were filtered and washed three times using a Whatman No 1 filter paper (Whatman International Ltd., Maidstone, England) and the residual AIS was freeze-dried and weighed according to Yamaki et al. (13). The brix degree of the tea infusion samples was measured using a Hand Refractometer (Atago, Tokyo, Japan).
Fractionation of tea polysaccharides and sample preparation
Tea polysaccharides (TPs) were fractionated using distilled water (distilled water soluble polysaccharide; DWSP), 0.05 M 1,2-diaminocyclohexane tetra acetate (CDTA) soluble polysaccharide (CDSP), 0.05 M Na2CO3 (sodium carbonate soluble polysaccharide; SCSP), or 8 M KOH soluble hemicellulose (HC) by the method of Redgwell et al. (14).
Measurements of antioxidant activities
The electron donating ability was measured as described by Blois (15). Iron chelating and superoxide dismutase-like (SOD) activities were measured using methods by Dinis et al. (16) and Martin et al. (17), respectively. The xanthine oxidase (XO) inhibitory activity was measured by the use of partially purified milk containing XO (18,19), whereas the aldehyde oxidase (AO) inhibitory activity was evaluated using partially purified rabbit hepatic enzyme (20).
Sensory evaluation
The brewed tea (1%) was prepared by boiling NFT and FTM in water for 3 minutes. Sensory evaluation of tea infusion was evaluated by the well-trained 25 members of the Woori-tea Culture Association (Daegu, Korea) for aroma, taste, color, brightness and mouth feel by a 5 point method as follows: very poor=1, poor=2, moderate=3, good=4 and very good=5.
Statistical analysis
Values for analysis were represented as mean±standard deviations of triplicate determinations. Sensory values were represented as mean±standard deviation of 25 panels. Significance tests were carried out by performing a two-way analysis of variance (ANOVA) test followed by Duncan’s multiple range test and t-test using SPSS statistical software (version 12.0, SPSS Inc., Chicago, IL, USA). A p value <0.05 denotes statistically significant differences.
RESULTS AND DISCUSSION
Color of tea infusion
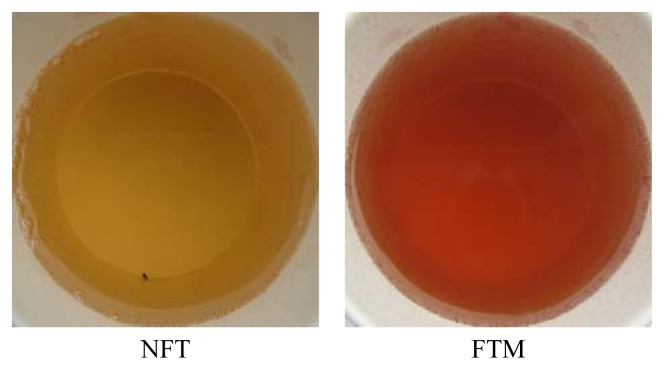
Hunter color values and photographs of 1% tea (NFT and FTM) infusions are showed in Table 1 and Fig. 2. Whereas the L* (lightness) value of the NFT infusion (59.03) was higher than that of FTM (40.25), the a* (redness) value of the FTM infusion (26.17) was markedly higher than that of NFT (8.42). On the other hand, the b* (yellowness) value of FTM infusion (15.17) was lower than that of NFT (25.71). In addition, while the hue angle in FTM infusion was 39.23 (reddish orange), the NFT infusion value was 68.92 (yellowish red). These results indicated that color of FTM infusion may be due to theaflavins and thearubigins derived from polyphenols, including catechins, via oxidation and polymerization, which also produce pigments such as rubropuntain and monascin during fermentation by M. pilosus(21). The color of the NFT infusion may be due to the production of relatively lower amounts of theaflavins and thearubigins. Therefore, the above results suggest that a considerable part of polyphenols could convert to flavonoids as thearubigins during fermentation by M. pilosus.
Table 1.
Color of tea infusions with and without fermentation by M. pilosus
| Plots | Hunter values and hue angle | |||
|---|---|---|---|---|
|
| ||||
| L* | a* | b* | H∘ | |
| NFT1) | 59.03±2.34a3) | 8.42±1.30b | 25.71±1.41a | 68.93±2.45a |
| FTM2) | 40.25±2.21b | 26.17±2.28a | 15.17±1.22b | 39.23±2.32b |
See Fig. 1.
Values are mean±SD of triplicate determinations, and different superscripts within a column (a,b) indicate significantly different at p<0.05.
Fig. 2.
Photographs of 1% tea fusion. Abbreviations: See Fig. 1.
Content of WSS and brix degree of tea infusion
The WSS content and the brix degree of 1% tea infusions are shown in Fig. 3. WSS content of FTM (299.30 mg/g, dry basis) was 17.60% lower than that of NFT (363.20 mg/g, dry basis). Furthermore, brix degree of the FTM infusion (0.84) was also lower than that of the NFT (0.98). This result may be caused by a high reduction of water-soluble components, such as carbohydrates, despite newly formed water-soluble color components during tea fermentation (Fig. 2). Zuo et al. (22) reported that catechins make up 30~42% of tea leaves; furthermore, 25% of catechins are oxidized and 6~14% are converted to water-soluble flavonoids in the post-fermented tea manufactured by microorganisms. Also, carbohydrates within raw tea leaves are being used as microorganism nutrients. Therefore, these results suggest that water-soluble components are consumed by the growth of microorganisms during fermentation.
Fig. 3.
Content of water soluble substance (WSS) (A) and brix degree of tea infusion (B). Abbreviations: See Fig. 1. Values are mean±SD of triplicate determinations, and different superscripts on the bars (a,b) indicate significant differences at p<0.05.
Content of tea polysaccharides
Tea polysaccharides (TPs) content fermented by M. pilosus are shown in Fig. 4. AIS content in FTM (910.11 mg/g, dry basis) is significantly higher than that of NFT (830.03 mg/g). Also, fractionated DWSP, CDSP, SCSP and HC from AIS in FTM is 2.15, 2.40, 2.68 and 1.57 folds higher than that of NFT, respectively. AIS in plants are well-known as cell wall constituents (23) and consist of polysaccharides such as cellulose, hemicellulose and pectin (24). Furthermore, these polysaccharides exist as binding forms with color components and polyphenolic antioxidant materials in plants (25); therefore, according to well documented research, these TPs have various functions such as antioxidant, antidiabetic, hypolipidemic, immunomodulatory and anticancer activities (26–28). Generally, water-insoluble components in the cell wall are greater than water-soluble components; however, the function of water-insoluble TPs in related studies have not been established. On the other hand, water- soluble or CDTA-soluble TPs are mainly components of middle lamellae, and sodium carbonate-soluble fractions are of primary cell walls (29). In addition, most components of AIS are also cell wall materials (23); distilled water or CDTA-soluble polysaccharides are components of middle lamellae, and sodium carbonate-soluble fractions are of primary cell walls (29). Potassium hydroxide-soluble fractions, known as hemicellulose, are bridged between the middle and primary cell walls (30). Wang et al. (28) reported that the contents of water-soluble polysaccharides in fermented tea are increased according to the degree of fermentation. Therefore, the above results indicate that increased extractable polysaccharides in FTM may be due to changes of cell wall structure by the fermentation in this study.
Fig. 4.
Content of alcohol insoluble solids (AIS) and tea polysaccharides in NFT and FTM. NFT and FTM: See Fig. 1. DWSP: water soluble polysaccharide, CDSP: 0.05 M CDTA-soluble polysaccharides, SCSP: 0.05 M sodium carbonate soluble poly-saccharide, HC: 8 M KOH soluble hemicellulose. Values are mean±SD of triplicate determinations, and different superscripts on the same bars (a-d) and different bars (A,B) indicates significant differences at p<0.05.
Antioxidant activities of tea polysaccharides
Antioxidant activities of fractions of TPs separated by various solvents are shown in Table 2. Whereas, the electron donating ability of DWSP, CDSP and SCSP extracted from FTM fermented by M. pilosus was 15~ 58% higher than that of NFT, the HC fraction obtained from NFT was higher (43.89%) than that of FTM (9.94%). Although, iron-chelating ability of the CDSP extracted from NFT was higher (86.80%) than that of FTM (28.90%), DWSP and HC from FTM was significantly higher than that of NFT. On the other hand, SOD-like activity of the CDSP extracted from both NFT and FTM was similar, although DWSP and SCSP obtained from FTM was 27~82% higher than that of NFT. The activity of HC fraction from both NFT and FTM could not be detected. Compared to the XO inhibitory activities of DWSP, CDSP and SCSP obtained from NFT (2.46~49.18%), the inhibitory activity of DWSP, CDSP and SCSP from FTM could not be detected. On the other hand, the inhibitory activity of HC fraction from NFT was undetectable, while that of FTM was 19.06%. The AO inhibitory activity of the DWSP and SCSP extracted from FTM was 3.22~3.85 fold strikingly higher than that of NFT; however, the inhibitory activity of CDSP from NFT did not appear, while that of FTM was 19.65%. Furthermore, the inhibitory activity of HC from NFT was 99.42%, although that of FTM could not be detected. Interestingly, we observed the XO inhibitory activities of all polysaccharide fractions, except HC, were decreased, whereas the AO inhibitory activities were increased due to the fermentation by M. pilosus in this study. These results indicated that newly formed components from fermentation may be responsible for this phenomenon, but further studies are needed.
Table 2.
Antioxidant related activities of tea polysaccharides
| Teas1) | DWSP2) | CDSP3) | SCSP4) | HC5) | |
|---|---|---|---|---|---|
| Electron | NFT | 43.47±1.65aB6) | 36.51±2.14bB | 30.82±2.25cB | 43.89±2.87aA |
| donating ability (%) | FTM | 49.86±2.08bA | 57.53±3.00aA | 48.58±3.01bA | 9.94±2.40cB |
|
| |||||
| Iron chelating | NFT | 12.36±2.66bB | 86.80±4.71aA | 9.11±2.85NS | 3.44±1.04cB |
| activity (%) | FTM | 21.10±1.95bA | 28.90±2.45aB | 9.85±1.74 | 24.72±2.38aA |
|
| |||||
| SOD-like activity | NFT | 0.69±0.03aB | 0.22±0.02NS7) | 0.39±0.03bB | 0.00±0.00NS |
| (U) | FTM | 0.88±0.07aA | 0.23±0.02 | 0.71±0.04bA | 0.00±0.00 |
|
| |||||
| XO inhibitory | NFT | 12.09±2.97bA | 2.46±1.88cA | 49.18±2.32aA | 0.00±0.00dB |
| activity (%) | FTM | 0.00±0.00bB | 0.00±0.00bB | 0.00±0.00bB | 19.06±3.12aA |
|
| |||||
| AO inhibitory | NFT | 10.41±1.98bB | 0.00±0.00cB | 7.51±2.58bB | 99.42±4.09aA |
| activity (%) | FTM | 33.53±2.23aA | 19.65±1.45cA | 28.90±1.82bA | 0.00±0.00dB |
Wang et al. (28) reported that oolong tea also has a higher content of DWSP, depending on the degree of fermentation, as well as higher antioxidant activities such as electron donating ability, ABTS radical scavenging and ferric iron reducing power, and higher α-glucosidase inhibitory activity. The above results suggest that markedly increased content of polysaccharides in FTM compared to NFT may be responsible for an overall increase in antioxidant activities.
Sensory evaluation
Evaluating sensory quality of infused tea by extraction from tea powder (1 g) with hot water (100 mL) for 3 min is shown in Table 3. Appearance for the acceptability of the NFT and FTM are 2.63 and 2.55 points, respectively, but not significantly different. Whereas, aroma and taste of NFT are 3.15 and 2.84 points, respectively, those of FTM are 4.11 and 3.95 points, respectively; the value of FTM is significantly higher than that of NFT. On the other hand, in the color of brewed tea, NFT is a high yellow color, while FTM as a post-fermented tea is red-brown due to theaflavins and thearubigins as maroon and pink color ingredients produced by Monascus microorganisms (Fig. 2). Preference for the brightness and mouth feel of the NFT are 2.85 and 2.88 points, respectively, although those of FTM are 4.26 and 4.25 points, respectively; the value of FTM is significantly higher than that of NFT.
Table 3.
Sensory evaluation of tea infusions with and without fermentation by M. pilosus
| Attributes1) | NFT2) | FTM3) |
|---|---|---|
| Appearance | 2.63±0.31NS4) | 2.55±0.37 |
| Aroma | 3.15±0.28b5) | 4.11±0.34a |
| Taste | 2.84±0.37b | 3.95±0.36a |
| Color | 2.78±0.32b | 4.15±0.37a |
| Brightness | 2.85±0.35b | 4.26±0.41a |
| Mouth feel | 2.88±0.45b | 4.25±0.44a |
All attributes were evaluated from very poor (1 point) to very good (5 points).
See Fig. 1.
Not significant.
Values are mean±SD of 25 panels, and different superscripts within a row (a,b) indicate significantly different at p<0.05.
The widely-accepted important attributes as quality determinant factors of post-fermented tea are appearance, aroma and taste as well as color, brightness and mouth feel in tea infusion (31,32). In general, low-quality tea is almost a bad color or appearance due to fermentation technology. In other words, the appearance of optimized fermented tea is greenish black due to the increased contents of theaflavin (golden yellow) thearubigins (orange-brown), undegradated chlorophyll, and pheophytin and pheophorbide derived from chlorophyll. As mentioned above, it is widely accepted that various important attributes of tea also affect flavor, taste and mouth feel. Therefore, these results indicate that the sensory evaluation of tea could be improved with fermentation by M. pilosus.
In summary, the present study provides evidence that post-fermented tea by Monascus microorgansims may be responsible for functional components as well as contributing to improvement of the tea quality, specifically the tea-manufacturing period, which could be shortened.
ACKNOWLEDGMENTS
This research was supported by Industrialization Support Program for Bio-technology of Agriculture and Forestry (810007-03-1-SU000), Ministry for Food, Agriculture, Forestry and Fisheries, Republic of Korea.
REFERENCES
- 1.Matsumoto S, Kiriiwa Y, Yamaguchi S. The Korean tea plant (Camellia sinensis): RFLP analysis of genetic diversity and relationship to Japanese tea. Breeding Sci. 2004;54:231–237. [Google Scholar]
- 2.Halder B, Pramanick S, Mukhopadhyay S, Giri AK. Anticlastogenic effects of black tea polyphenols theaflavins and thearubigins in human lymphocytes in vitro. Toxicol In Vitro. 2006;20:608–613. doi: 10.1016/j.tiv.2005.10.010. [DOI] [PubMed] [Google Scholar]
- 3.Muramatsu K, Oguni I, Isemura M, Sugiyama K, Yamamoto M. Health Science of Tea–New possibility for physiological function. Gakkai Shuppan Center; Tokyo, Japan: 2002. pp. 17–23. [Google Scholar]
- 4.Yang DJ, Hwang LS. Study on the conversion of three natural statins from lactone forms to their corresponding hydroxy acid forms and their determination in Pu-erh tea. J Chromatogr A. 2006;1119:277–284. doi: 10.1016/j.chroma.2005.12.031. [DOI] [PubMed] [Google Scholar]
- 5.Jeng KC, Chen CS, Fang YP, Hou RC, Chen YS. Effect of microbial fermentation on content of statin, GABA, and polyphenols in Pu-erh tea. J Agric Food Chem. 2007;55:8787–8792. doi: 10.1021/jf071629p. [DOI] [PubMed] [Google Scholar]
- 6.Chen YS, Liu BL, Chang YN. Bioactivities and sensory evaluation of Pu-erh teas made from three tea leaves in an improved pile fermentation procss. J Biosci Bioeng. 2010;109:557–563. doi: 10.1016/j.jbiosc.2009.11.004. [DOI] [PubMed] [Google Scholar]
- 7.Endo A. Monacolin K, a new hypocholesterolemic agent that specifically inhibits 3-hydroxy-3-methylglutaryl coenzyme A reductase. J Antibiot (Tokyo) 1980;33:334–336. doi: 10.7164/antibiotics.33.334. [DOI] [PubMed] [Google Scholar]
- 8.Pyo YH, Lee TC. The potential antioxidant capacity and angiotensin I-converting enzyme inhibitory activity of Monascus-fermented soybean extracts: Evaluation of Monascus-fermented soybean extracts as multifunctional food additives. J Food Sci. 2007;72:S218–S223. doi: 10.1111/j.1750-3841.2007.00312.x. [DOI] [PubMed] [Google Scholar]
- 9.Lee SI, Kim JW, Lee YK, Yang SH, Lee IA, Suh JW, Kim SD. Anti-obesity effect of Monascus pilosus mycelial extract in high fat diet induced-obese rats. J Appl Biol Chem. 2011;54:197–205. [Google Scholar]
- 10.Lee SI, Kim JW, Lee YK, Yang SH, Lee IA, Suh JW, Kim SD. Protective effect of Monascus pilosus mycelial extract on hepatic damage in high-fat diet induced- obese rats. J Appl Biol Chem. 2011;54:206–213. [Google Scholar]
- 11.Guoa L, Dua X, Lanb J, Lianga Q. Study on molecular structural characteristics of tea polysaccharide. Int J Biol Macromol. 2010;47:244–249. doi: 10.1016/j.ijbiomac.2010.03.026. [DOI] [PubMed] [Google Scholar]
- 12.Lee YK, Lee SI, Kim JS, Yang SW, Lee IA, Kim SD, Suh JW. Antioxidant activity of green tea fermented with Monascus pilosus. J Appl Biol Chem. 2012;55:19–25. [Google Scholar]
- 13.Yamaki S, Machida Y, Kakiuchi N. Changes in cell wall polysaccharides and monosaccharides during development and ripening of Japanese pear fruit. Plant Cell Physiol. 1979;20:311–321. [Google Scholar]
- 14.Redgwell RJ, Melton LD, Brasch DJ. Cell wall changes in kiwifruit following postharvest ethylene treatment. Phytochem. 1990;29:399–407. [Google Scholar]
- 15.Blois MS. Antioxidant determination by the use of a stable free radical. Nature. 1958;181:1199–1200. [Google Scholar]
- 16.Dinis TCP, Madeira VMC, Almeida LM. Action of phenolic derivatives (acetaminophen, salicylate, and 5-amino salicylate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch Biochem Biophy. 1994;315:161–169. doi: 10.1006/abbi.1994.1485. [DOI] [PubMed] [Google Scholar]
- 17.Martin JP, Dailey M, Sugarman E. Negative and positive assays of superoxide dismutase based on hema-toxylin autoxidation. Arch Biochem Biophys. 1987;255:329–336. doi: 10.1016/0003-9861(87)90400-0. [DOI] [PubMed] [Google Scholar]
- 18.Stirpe F, Della Corte E. The regulation of rat liver xanthine oxidase: Conversion in vitro of the enzyme activity from dehydrogenase (type D) to oxidase (type O) J Biol Chem. 1969;244:3855–3863. [PubMed] [Google Scholar]
- 19.Özer N, Müftüoglu M, Ataman D, Ercan A, Ögüs IH. Simple, high-yield purification of xanthine oxidase from bovine milk. J Biochem Biophys Methods. 1999;39:153–159. doi: 10.1016/s0165-022x(99)00012-3. [DOI] [PubMed] [Google Scholar]
- 20.Rajagopalan KV, Fridovich I, Handler P. Hepatic aldehyde oxidase. I: Purification and properties. J Biol Chem. 1962;237:922–928. [PubMed] [Google Scholar]
- 21.Ngure MF, Wanyokob JK, Mahungua SM, Shitandia AA. Catechins depletion patterns in relation to theaflavin and thearubigins formation. Food Chem. 2009;115:8–14. [Google Scholar]
- 22.Zuo YG, Chen H, Deng YW. Simultaneous determination of catechins, caffeine and gallic acids in green, oolong, black and Pu-erh teas using HPLC with a photodiode array detector. Talanta. 2002;57:307–313. doi: 10.1016/s0039-9140(02)00030-9. [DOI] [PubMed] [Google Scholar]
- 23.Shewfelt AL, Payter VA, Jen JJ. Textural changes and molecular characteristics of pectin constituent in ripening peaches. J Food Sci. 1971;36:573–575. [Google Scholar]
- 24.Grassin C, Herweijer M. Enzymes in fruit juice production and fruit processing. In: Aehle W, editor. Enzymes in Industry. WILEY-VCH; Weinheim, Germany: 2004. pp. 113–125. [Google Scholar]
- 25.Kang YH, Parker CC, Smith AC, Waldron KW. Characterization and distribution of phenolics in carrot cell walls. J Agric Food Chem. 2008;56:8558–8564. doi: 10.1021/jf801540k. [DOI] [PubMed] [Google Scholar]
- 26.Chen HX, Zhang M, Qu ZS, Xie BJ. Antioxidant activities of different fractions of polysaccharides conjugates from green tea (Camellia sinensis) Food Chem. 2008;106:559–563. [Google Scholar]
- 27.Nie SP, Xie MY. A review on the isolation and structure of tea polysaccharides and their bioactivities. Food Hydrocolloids. 2011;25:144–149. [Google Scholar]
- 28.Wang Y, Shao S, Xu P, Chen H, Lin-Shiau SY, Deng YT, Lin JK. Fermentation process enhanced production and bioactivities of oolong tea polysaccharides. Food Res Int. 2012;46:158–166. [Google Scholar]
- 29.Cutillas-Iturralde A, Zarra I, Lorences EP. Metabolism of cell wall polysaccharides from persimmon fruit: Pectin solubilization during fruit ripening occur in apparent absence of polygalacturonase activity. Physiol Plant. 1993;89:369–375. [Google Scholar]
- 30.Gross KC. Fractionation and partial characterization of cell walls from normal and non-ripening mutant tomato fruit. Physiol Plant. 1984;62:25–32. [Google Scholar]
- 31.Owuor PO, Obanda M, Nyirenda HE, Mphangwe NIK, Wright LP, Apostolides Z. The relationship between some chemical parameters and sensory evaluations for plain black tea (Camellia sinensis) produced in Kenya and comparison with similar teas from Malawi and South Africa. Food Chem. 2006;97:644–653. [Google Scholar]
- 32.Chen CH, Chan HC, Chang YN, Liu BL, Chen YS. Effects of bacterial strains on sensory quality of Pu-erh tea in an improved pile-fermentation process. J Sens Stud. 2000;24:534–553. [Google Scholar]