Why has an entire BBA issue been devoted to triglyceride? During the past several years the BBA has devoted special issues to lipid droplets, lipotoxicity, lipids and Alzheimer’s disease, and lipid pathways and human disease. Several of these state of the art reviews have touched on the central molecule of this current issue; triglyceride. The centrality of triglyceride to cellular biology, organ function, and several human diseases is a fast moving scientific and clinical research field and the objective of this issue is to present a birth to death biography of this molecule (Figure 1). In so doing, I tried to recruit authors who could review issues in triglyceride production, storage, and catabolism, triglyceride and organ dysfunction in animal models, and the correlation of plasma and tissue triglyceride levels with human diseases. This issue is divided into the sections that follow.
Figure 1. Triglyceride transport in the blood.
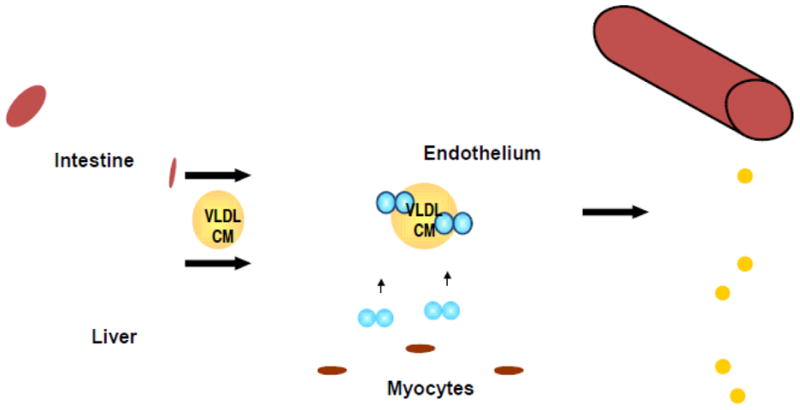
Triglycerides are synthesized in either the intestine or liver. Chylomicrons are secreted into the lymph and then enter the circulation via the superior vena cava. VLDL are directly secreted into the blood. Both particles are metabolized via their interaction with lipoprotein lipase on the luminal surface; a reaction that coverts the triglyceride into free fatty acids that are taken-up by peripheral tissues. Oversupply leads to triglyceride accumulation in heart, skeletal muscle, and liver. In addition, triglyceride-rich lipoproteins can be either a source of atherosclerotic lipids or triglyceride lipolysis might lead to production of lipids that are toxic to the vessels.
The first reviews in this issue begin with triglyceride production. Dietary triglyceride is almost 100% absorbed unless there is a defect in pancreatic enzyme production or in enterocyes. Chylomicron triglyceride does not, however, directly enter the bloodstream; rather it is routed via the lymphatics and enters the bloodstream when the thoracic duct empties into the superior vena cava. The reason for this rather circuitous route is uncertain but could be a means to protect the liver from a large lipid load or to allow greater delivery of fatty acids to the heart, lungs and peripheral tissues. Although the process of triglyceride absorption was known to take several hours, recent kinetic analyses reviewed by Drs. Lambert and Parks suggest that enterocyte storage of triglyceride occurs and that the sequence of meals, not just an individual meal, can affect the appearance of dietary chylomicron triglycerides. Major advances in understanding chylomicron assembly have also come from studies of microsomal triglyceride transfer protein (MTTP). Drs. Pan and Hussain discuss what we have learned about the basic pathways of MTTP regulation and how they, in turn, affect gut lipid secretion. Genetic defects in MTTP are the cause of abetalipoproteinemia in humans. However, other factors can lead to a more subtle regulation of triglyceride absorption. Most importantly, Drs. Xiao and Lewis note how insulin can affect chylomicron production, as well as clearance, in humans.
The ancients invented the term melancholia, black bile, which occurs with biliary obstruction. Although we longer view the liver as the seat of human passion or emotion, it is clearly the central organ for control of lipid homeostasis. What are the steps regulating the production and secretion of triglyceride from the liver? The primary liver enzyme responsible for de novo hepatic synthesis of triglyceride from glucose is fatty acid synthetase. The biology of this enzyme, much of it learned from genetically modified mice, is reviewed by Drs. Jensen-Urstad and Semenkovich. The liver must allocate its newly formed triglyceride and triglyceride that arrives via lipoprotein and free fatty acid uptake to oxidation, storage or secretion. Phospholipid production, as reviewed by Cole and the Vances, is required both for lipoprotein and lipid droplet production. Drs. Quiroga and Lehner discuss the several enzymes capable of liberating stored triglyceride and Dr. Liu and colleagues describe the surprising observation that liver triglyceride secretion is also modulated by the ABC-A1 cholesterol transporter. Finally in this section, Dr. Fisher reviews the factors affecting the production of apoB, particularly those that cause its pre-secretory degradation. ApoB has a complex intracellular life that is shortened by both an excess or deficiency of certain dietary fatty acids, reactive oxygen species, ER-stress, and structural mutations.
Almost 70 years ago, Han first learned that intravenous heparin led to release of a “clearing factor” that eliminated chylomicrons from the circulation of dogs 1. Havel and Gordon linked lipoprotein lipase (LpL) deficiency with human fasting chylomicronemia in 1960 2. Ten years ago it was generally felt that regulation of lipolysis and LpL has been “worked out”. We were wrong! A number of new molecules in the lipolysis reaction were initially discovered in genetically modified mice and shown to regulate circulating triglyceride levels (Figure 2). LpL activity in adipose increases with feeding and decreases with fasting and this regulation is on the posttranslational level. Drs. Mattijssen and Kersten describe how angiopoietin like proteins 3 and 4 are regulated and, in turn, inhibit LpL and appear to affect LpL actions even in the lymph. Another unexplained lipolysis defect was that found more than 3 decades ago when severe hypertriglyceridemia and neonatal death was observed in the combined lipase deficiency mouse3. A multi-decade study of these mice culminated with the identification of lipase maturation factor 1; this saga is reviewed by Dr. Peterfy. A third protein, apoA5 was initially shown in the mouse to reduce plasma triglyceride and its absence to increase triglyceride. The biochemistry of apoA5 and how it might regulate triglyceride metabolism is discussed by Drs. Sharma, Ryan and Forte.
Figure 2. The lipolysis reaction.
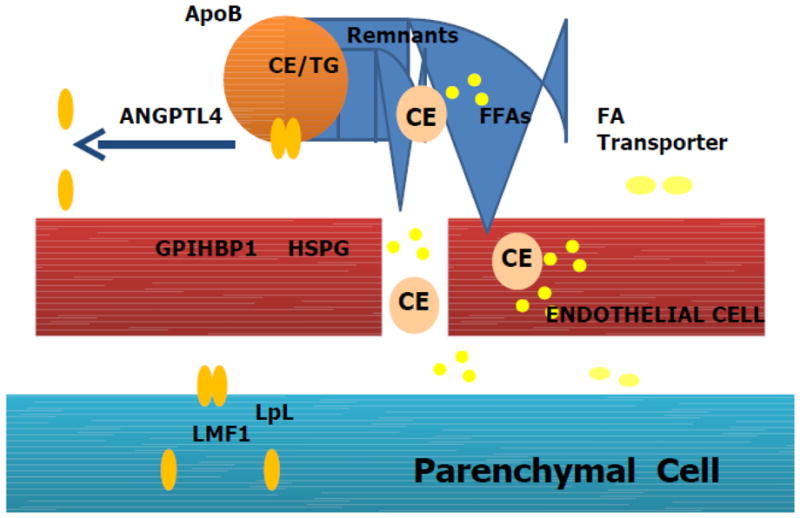
Triglyceride-rich lipoproteins contain apoproteins including apoCII, apoCIII and perhaps apoA5 that regulate lipolysis. The lipoproteins must contact the vessel wall to allow the reaction to occur. Lipoprotein lipase (LpL) probably associated in part with heparan sulfate proteoglycans (HSPG) has its actions modulated by a number of factors including lipase maturation factor (LMF)1 that is needed to synthesize active enzyme, glycosylphosphatidylinositol-anchored high density lipoprotein-binding protein (GPIHBP) 1 that is necessary for securing active enzyme on the endothelial surface, and angioipoietin like protein 4 (ANGPTL4) that inhibits activity probably by dissociating the LpL dimer. Released fatty acids (FAs) enter cells either by non-receptor mediated pathways or via protein transporters such as FATPs, FABPpm, or CD36.
Two additional papers in this section focus on extracellular lipolysis and lipid uptake by specific tissues. Drs. Kim, Wang and Rodrigues focus on LpL regulation in the heart. And Drs. Kazantzis and Stahl discuss the FATP family of proteins, in which tissues they are expressed, and how their deletion alters cellular lipid uptake.
Humans develop hypertriglyceridemia for a variety of reasons. Severe hypertriglyceridemia causes pancreatitis, which then triggers treatment of triglycerides. Excess caloric intake leading to obesity and type 2 diabetes is the most common cause of hypertriglyceridemia. This association and the reasons for it are discussed by Drs. Subramanian and Chait. Subsequent articles by Dr. Hassing and colleagues and Johanssen and Hegele discuss the pathology and genetics of several genetic human hypertriglyceridemias.
Fibric acids and omega 3 fatty acids are currently the first choice treatments for severe hypertriglyceridemia. The former is discussed in the next section by Dr. Boullart et al. and the latter by Drs. Shearer, Savinova and Harris. There is no doubt that omega 3 fatty acids are an option for treatment of human hypertriglyceridemia. Whether the cellular and animals studies that have argued for use of these drugs as anti-inflammatory treatment in a variety of human conditions will prove correct is not known.
Complications of obesity that were described in unusual patients are rapidly becoming major public health issues. For that reason our final subsection focuses on tissue triglycerides and human disease. Non-alcoholic fatty liver disease (NAFLD) is rapidly becoming the leading cause of liver dysfunction in overweight societies. How excess liver fat affects liver metabolism and gene regulation is discussed by Drs. Tailleux, Wouters, and Stahl.
In another BBA special issue the causes of lipotoxicity were addressed. For that reason, the remaining articles in this section focus on the putative role of circulating triglycerides as a risk factor for cardiovascular disease. This relationship has been known for decades 4. As triglyceride-rich particles are lipolysed in the circulation especially along arterial surfaces, they produce potentially toxic products that, as reviewed by Drs. Schwartz and Reaven, could alter the biology of the artery. But, are triglycerides an independent risk for cardiovascular disease and does triglyceride reduction reduce cardiovascular events? This has been the focus of several recent position papers in the US 5 and Europe 6. Drs. Boullart, de Graaf, and Stalenhoef review the clinical trial data and its interpretation that have led to much uncertainty in the approach to moderate hypertriglyceridemia and its treatment.
As a final note, I am grateful to these authors who took time from their busy schedules to contribute to this issue. Also I wish to thank the unmentioned reviewers of these articles who, I think the authors will agree, have improved each paper. I hope that the readers enjoy my efforts and those of the BBA staff to splash a number of topics on the same canvas as a means to create a landscape, much of it in yellow, of the creation and life of triglycerides in mammals. Finally, as with any issue of this type, there are many topics that were not discussed and that can be the focus of future issues of BBA-Lipids.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Hahn PF. Abolishment of alimentary lipemia following injection of heparin. Science. 1943;98:19–20. doi: 10.1126/science.98.2531.19. [DOI] [PubMed] [Google Scholar]
- 2.Havel RJ, Gordon RS., Jr Idiopathic hyperlipemia: Metabolic studies in an affected family. J Clin Invest. 1960;39:1777–1790. doi: 10.1172/JCI104202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Paterniti JR, Jr, Brown WV, Ginsberg HN, Artzt K. Combined lipase deficiency (cld): A lethal mutation on chromosome 17 of the mouse. Science. 1983;221:167–169. doi: 10.1126/science.6857276. [DOI] [PubMed] [Google Scholar]
- 4.Goldstein JL, Hazzard WR, Schrott HG, Bierman EL, Motulsky AG. Hyperlipidemia in coronary heart disease. I. Lipid levels in 500 survivors of myocardial infarction. J Clin Invest. 1973;52:1533–1543. doi: 10.1172/JCI107331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Miller M, Stone NJ, Ballantyne C, Bittner V, Criqui MH, Ginsberg HN, Goldberg AC, Howard WJ, Jacobson MS, Kris-Etherton PM, Lennie TA, Levi M, Mazzone T, Pennathur S. Triglycerides and cardiovascular disease: A scientific statement from the american heart association. Circulation. 2011;123:2292–2333. doi: 10.1161/CIR.0b013e3182160726. [DOI] [PubMed] [Google Scholar]
- 6.Chapman MJ, Ginsberg HN, Amarenco P, Andreotti F, Boren J, Catapano AL, Descamps OS, Fisher E, Kovanen PT, Kuivenhoven JA, Lesnik P, Masana L, Nordestgaard BG, Ray KK, Reiner Z, Taskinen MR, Tokgozoglu L, Tybjaerg-Hansen A, Watts GF. Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: Evidence and guidance for management. Eur Heart J. 2011;32:1345–1361. doi: 10.1093/eurheartj/ehr112. [DOI] [PMC free article] [PubMed] [Google Scholar]


