Clinical Points
▪ Understanding how structural and functional neuroimaging techniques work, and what their risks are, can help the clinician order these tests in a rational and cost-effective manner.
▪ For patients presenting with cognitive, behavioral, or emotional symptoms, the use of neuroimaging can be of major relevance when certain conditions apply.
Have you ever wondered which neuroimaging techniques can facilitate making a neuropsychiatric diagnosis? Have you been uncertain about the indications for and the risks of various neuroimaging modalities? Have you been perplexed by which test to order first? If you have, then the following case presentation and discussion should prove useful.
CASE VIGNETTE
Mr A, a 35-year-old man with an unremarkable medical and psychiatric history, presented to the emergency department complaining of 1 month of worsening intrusive thoughts, insomnia, and mood symptoms (involving both the depressive and manic spectrum).
Mr A described himself as “very low-key” and said he rarely became anxious and never had problems sleeping. Four weeks before his arrival at the emergency department, he developed insomnia (primarily a difficulty with the initiation of sleep but also with its maintenance) that became problematic over a few days. Then, Mr A developed vivid, intrusive, and hostile thoughts (that were insulting toward his loved ones and to himself); these thoughts frightened him. There was no readily apparent trigger, and nothing seemed to make the thoughts better or worse. Although these thoughts were distressing, Mr A did not develop compulsions or rituals, and he did not develop panic attacks or phobias. No hallucinations, delusions, or ideas of reference were reported. However, when Mr A closed his eyes, he saw a knife rapidly chopping pizza or stabbing a teddy bear. These images were never noted when his eyes were open; he knew that these images were not real.
Mr A’s wife described him as “abulic” and recounted a number of situations when his expected affect and reactivity were missing. Despite these observations, Mr A endorsed a fearful and dysphoric mood, with an extreme sense of internal restlessness, despite no observable motor component. He described racing thoughts, disorganization, and impaired concentration but denied irritable or elated mood, impulsivity, grandiosity, increased goal-oriented activity, or reckless behavior. Mr A denied neurovegetative symptoms of depression (other than prominent insomnia) or concerns about safety.
Mr A went to see a psychiatrist for the first time 2 weeks before coming to the emergency department. He was diagnosed as having a thought disorder and was started on low-dose risperidone (1 mg/d). Prior to that visit, his primary care physician prescribed alprazolam (for anxiety) and zolpidem (for sleep). None of these treatments were effective, and his symptoms continued to worsen.
The emergency department evaluation led to a psychiatric admission. On the inpatient unit, Mr A was withdrawn (he stayed in his room and avoided contact with other patients and staff), fearing that he would act on or verbalize his thoughts and either injure or insult someone. Although distressed, he was always appropriate, cordial, and cooperative. His speech was robotic at times, but it was neither rapid nor pressured. He was detail oriented and, at times, tangential. His physical examination (including a comprehensive neurologic examination) was essentially normal, as were his basic laboratory results.
Shortly after his arrival to the inpatient unit, Mr A’s preadmission medications (alprazolam, zolpidem, risperidone) were stopped due to lack of efficacy and a sense that they were making his intrusive thoughts worse. He was started on clonazepam (1 mg 3 times daily); this medication reduced his intrusive thoughts and anxiety. Citalopram was also started (and quickly titrated to 60 mg/d), as was quetiapine for insomnia and mood stabilization (titrated to 200 mg/d). These changes coincided with a gradual improvement. Psychological testing was obtained given the acute yet atypical nature of his symptoms and an uncertain diagnosis, with intrusive thoughts (without compulsions), severe insomnia, and mood symptoms (on both the depressed and manic spectrum that did not qualify for a mixed episode). Testing showed signs of mild cognitive impairment and an atypical Personality Assessment Inventory profile, which suggested the possibility of an “organic” etiology. An electroencephalogram (EEG) failed to reveal abnormalities (including epileptiform activity). A head computerized tomography (CT) scan (without contrast) was within normal limits. A subsequent magnetic resonance imaging (MRI) scan of the head (without contrast) showed an ill-defined T2 hyperintense cortical and subcortical lesion in the right precentral gyrus with local mass effect suggestive of an infiltrative astrocytoma. An MRI with gadolinium was also obtained; significant enhancement of the lesion was shown.
HOW ARE NEUROIMAGING TESTS CONDUCTED AND WHAT CAN THEY TELL US?
Plain films of the skull follow the same principle as standard x-rays of other organ systems (a single source of radiation and a single sensor or film), but they have limited utility. CT images (Figure 1) are created by serially acquiring x-rays in a rotating axial plane. As with x-rays, different body tissues have specific attenuation properties with CT images; this makes water, fat, bone, and other tissue types appear differently on the film or digital sensor. The higher the attenuation, the lighter the material will appear on the CT scan. Because CT scanners measure serial x-rays in an axial plane, CT images are typically presented in axial slices (with a typical in-plane resolution of less than 1 mm). Although computational reconstruction algorithms can present CT images in a sagittal or coronal plane, such data manipulation loses spatial resolution and detail.1
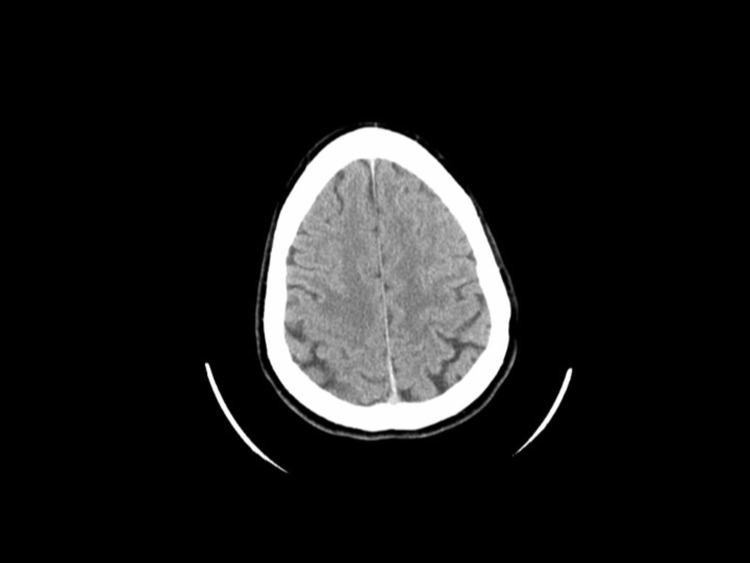
Figure 1.
Head Computerized Tomography (CT) Without Contrasta
aHead CT obtained in the course of the emergency room evaluation. This brain scan was read to be within normal limits. In retrospect, one can use the T2-weighted magnetic resonance imaging scan to guide the search and identify an area of very mild hypoattenuation in the right frontal lobe corresponding to the tumor.
The CT technology can be enhanced by the use of a contrast material. Contrast agents for CT are radiopaque and have a high x-ray attenuation; they appear white on CT images. Contrast is usually injected intravenously to enable imaging of vascular structures (eg, CT angiography) or lesions that disrupt the blood-brain barrier (eg, secondary to inflammation, bleeds, certain tumors). CT contrast agents can be ionic or nonionic, although current neuroimaging applications rely almost exclusively on use of nonionic contrasts because they have a better safety profile.2
The MRI techniques do not use radiation; instead, the magnetic properties of hydrogen ions in the body are used. A patient inside the MRI scanner is under the influence of a strong magnet in standard clinical applications, usually 1.5 or 3 Tesla. This magnetic force aligns a significant proportion of the body’s hydrogen atoms in the direction of the magnetic field. A brief radiofrequency pulse is then applied to shift and misalign the vectors of the hydrogen atoms. However, after the pulse ends, hydrogen atoms return to their original aligned position by a process called proton relaxation that releases energy. During the course of a scan, multiple radiofrequency pulses are applied (pulse sequence), and electromagnetic receiver coils measure the energy emitted by the protons.3 Different variables determine the proton relaxation process, which is responsible for the type of signal that each hydrogen atom emits. Two main factors are relevant to the clinician: the milieu where the hydrogen atom is found and the pulse sequence applied.
During the process of realignment (proton relaxation), the atoms emit energy, but this varies as a function of the physical and chemical environment of the atom. Therefore, protons will send different signals depending on the tissue type in which they are found (eg, bone, gray matter, white matter, cerebrospinal fluid [CSF]). In addition to the tissue type, the parameters in the MRI pulse sequence will also determine the properties of the images seen. The mechanism of proton relaxation has 2 time constants: T1 and T2. These constants reflect related but independent physical mechanisms of the proton relaxation process: T1 explains the relaxation toward the original plane aligned with the magnetic field, and T2 explains the relaxation away from the plane forced by the radiofrequency pulse. T1 and T2 relaxation components can be manipulated to force protons to realign in ways that maximize T1 or T2 times.4 These different MRI protocols change the proton relaxation process and the signal that is emitted; therefore, different types of images are formed that highlight specific features of tissue structure. T1-weighted images (Figure 2A) present with gray matter being darker than white matter (reflecting tissue appearance) and with dark-appearing CSF. These images are ideal to visualize the normal structure of the brain, as well as pathological atrophy, cortical dysplasias, and sclerosis. T2 images (Figure 3A) show a pattern opposite to T1-weighted scans, with gray matter being lighter than white matter (opposite to the tissue) and with bright CSF. These images are more sensitive to pathological processes such as vascular changes (including chronic microvascular insults), demyelination, general inflammation, and edema.1
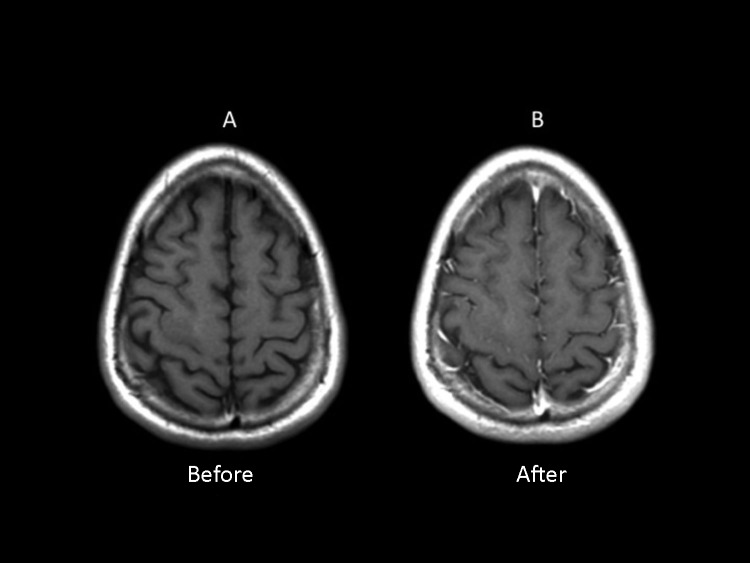
Figure 2.
T1 Magnetic Resonance Image (MRI) Before (A) and After (B) Gadolinium Contrast Injectiona
aThis T1-weighted MRI image (A) shows the anatomy in great detail, and the tumor as a heterogeneous and mildly hypointense lesion. Note that, compared to the T2 sequences, T1 offers lower detection sensitivity for this lesion. After gadolinium contrast injection, the postcontrast image (B) shows no gadolinium enhancement, suggesting intact blood-brain barrier at this point.
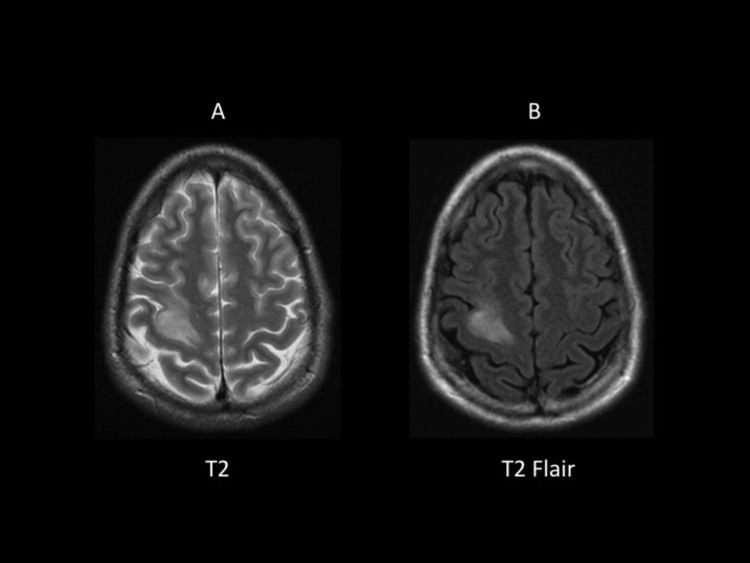
Figure 3.
T2 (A) and T2-FLAIR (B) Magnetic Resonance Image Sequencesa
aThis pulse sequence clearly identifies the right frontal tumor as a hyperintense lesion in both the T2 (A) and T2-FLAIR (B). Notice the contrast difference between the 2 images: once the fluid hyperintense signal is suppressed with FLAIR, the lesion increases its contrast.
Abbreviation: FLAIR = fluid attenuated inverted recovery.
T1- and T2-weighted images can be altered to increase their diagnostic resolution with protocols that suppress specific sources of signal. For example, one can suppress the hyperintense signal of CSF in T2-weighted images with the fluid attenuated inverted recovery or FLAIR sequence (Figure 3B). These T2 images therefore present with light-appearing gray matter, and with darker white matter, and they offer high diagnostic value for pathological process (such as inflammation, edema, or ischemia). Nevertheless, CSF appears black because its usually hyperintense signal is suppressed, and this greatly increases the contrast and visibility of pathological lesions, particularly, but not exclusively, in territories in contact with CSF (eg, cortical rim or periventricular regions). With a similar strategy, different fat-suppressing MRI approaches exist and are used when structures containing fat are obscuring the visualization of a potential lesion (eg, perivascular fat around a dissected vessel or thrombus). These approaches can be used with both T1- and T2-weighted images.3 Table 1 provides a summary of the visual presentation of the different MRI sequences.
Table 1.
Visual Appearance of Magnetic Resonance Image Sequences (T1, T2, T2-FLAIR)
| Sequence | Gray Matter | White Matter | Cerebrospinal Fluid |
| T1 | Dark gray (hypointense) | Light gray (hyperintense) | Black |
| T2 | Light gray (hyperintense) | Dark gray (hypointense) | White |
| T2-FLAIR | Light gray (hyperintense) | Dark gray (hypointense) | Black |
Abbreviation: FLAIR = fluid attenuated inverted recovery.
Diffusion-weighted imaging (DWI) is a distinct type of MRI acquisition method that measures the movements of water molecules in the brain. A water molecule in a glass of water has isotropic kinetics, that is, it diffuses freely in all possible directions. Water molecules in the brain do not diffuse randomly; their motion is limited by the constraints of the cerebral anatomy and the histologic structure. Diffusion-weighted imaging is able to measure water diffusivity in each defined voxel (or volume element) in the brain, and the more anisotropy (ie, limited diffusivity), the more hyperintense the voxel appears. This approach has been used to map the structural anatomy of white matter tracts with diffuse tensor imaging. This noninvasive tool is of great significance for the scientific study of human anatomy in vivo, but diffuse tensor imaging also has growing clinical applications, particularly in neurosurgical planning.
More commonly, DWI is used to diagnose a number of pathological conditions in which water molecules present with decreased diffusivity. The most common and clinically relevant application is that of diagnosing acute ischemic strokes. The lack of oxygen causes cytotoxic injury to cells in the affected region, inducing swelling and edema. Under these conditions, anisotropy increases due to the increased density of molecules (causing voxels to appear brighter in DWI within the first hour after an acute ischemic stroke). Other pathological processes (such as abscesses, hypercellular tumors [eg, lymphomas, high-grade gliomas], or excitotoxicity) also present limitations to water diffusivity due to the increased density of the tissue structure and can be identified as hyperintense lesions in DWI scans.1,3
The DWI scans are sensitive not only to changes in anisotropy, but also to T1 and T2 relaxation mechanisms. Therefore, T1 and T2 signal changes may also be seen in DWI maps and could be misidentified as changes in anisotropy. This phenomenon is of great relevance when differentiating acute and chronic strokes. In order to avoid misidentification, DWI images are always compared with quantitative images of the diffusion coefficient, known as apparent diffusion coefficient maps. That is to say that DWI scans have limited diagnostic value if analyzed independently of apparent diffusion coefficient images, and one must always compare the 2 scans. True reduction in diffusivity, stemming from any pathophysiologic process, will always present as hyperintense voxels in DWI and hypointense voxels in apparent diffusion coefficient scans. If this pattern is not observed, the mechanism driving the effect is likely to be different from increased anisotropy. A well-known example, T2-shine through, occurs in chronic ischemic strokes. These lesions present with T2 hyperintensity and may also appear as hyperintensities in DWI but with normal or more commonly hyperintense signals in apparent diffusion coefficient maps. If one were to look at the DWI image in isolation, the lesion could be confused with an acute stroke, and the patient might be given thrombolytic therapy. But, if one looks at all images together, it could be concluded that the changes in T2 signal from the old stroke “shine through” the DWI scan and can be observed as hyperintense lesions in both the DWI and apparent diffusion coefficient maps, which cannot reflect increased anisotropy.4
Gadolinium is the most commonly used MRI contrast material due to its paramagnetic properties. Like contrast agents for CT imaging, gadolinium is injected intravenously and used to detect or rule out lesions that break the blood-brain barrier. MR angiography of the head does not use contrast agents as are used in CT angiography, but instead, specific MR pulse sequences allow for the noninvasive visualization of the vasculature.1 Neck MR angiography may use either gadolinium or the same pulse sequence used for head MR angiography.1
Functional MRI (fMRI) is an imaging modality that, until recently, had been used exclusively as a research tool; however, it has now been developed for certain limited, but growing, clinical applications5 (Figure 4). In contrast to the above-mentioned MRI modalities, fMRI is optimized to measure the function (not the structure) of brain areas and circuits. Its MRI pulse sequences are designed to detect the ratio between oxyhemoglobin and deoxyhemoglobin. When a brain area increases its activity, for example, in the context of a certain task, it also increases its metabolic and oxygenation needs. In this context, 2 phenomena happen in parallel. First, because more oxygen is used, more oxyhemoglobin is turned into deoxyhemoglobin, and the absolute quantity of deoxyhemoglobin increases. Second, because more oxygen is needed, a coupled neurovascular mechanism is activated that induces activity-dependent local vasodilation that increases the regional flow of blood with oxyhemoglobin. The summation of the 2 processes induces an absolute and relative increase of oxyhemoglobin that correlates with the increase in brain activity. Therefore, fMRI can detect changes in regional blood flow and oxyhemoglobin concentration dynamically, and through these measures, it reflects changes in brain activity with good spatial resolution.6
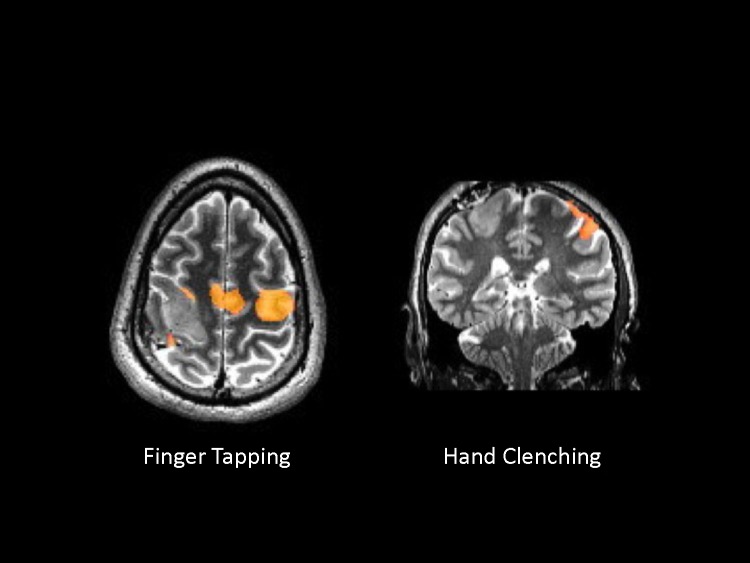
Figure 4.
Functional Magnetic Resonance Image Showing Finger-Tapping (A) and Hand-Clenching (B) Tasksa
aTwo images in different planes were selected to illustrate the activation of the finger-tapping (axial slice) and hand-clenching tasks (coronal slice). Note the tumor on the right precentral gyrus. The activation for this left motor task is atypical: primarily in the left precentral gyrus (ipsilateral to the movement and contralesional). Activation in the supplementary motor area (medial frontal) is also noted.
Magnetic resonance spectroscopy is an MRI-based application used to measure the relaxation properties of specific chemical bonds beyond hydrogen atoms. Unlike the previous methods, it does not measure the whole brain but selects a predefined region and measures the relative concentrations of certain chemical elements or molecules. Magnetic resonance spectroscopy is therefore not used to measure the structure or function of the brain, but its chemical composition. The method is widely used in research but is slowly finding its place in the clinical setting for the detection of tumors, epileptic foci, vascular lesions, or areas of demyelination.7
Positron emission tomography (PET) is a nuclear medicine diagnostic technique used to obtain functional brain scans, similar to fMRI and different from standard CT and MRI scans that provide structural information. The PET technique can be used to measure 3 primary variables: regional blood flow, metabolic changes, and neurotransmitter dynamics. New experimental approaches are in development to identify more sophisticated biological mechanisms, such as protein synthesis, second messenger systems, and gene expression.8 Unlike fMRI, which is also a functional neuroimaging modality, PET requires the injection of a radioactive substance or radiopharmaceutical that will be selectively distributed in the brain (and all other organs), while emitting energy in the form of γ photons. The regional uptake, distribution, and washout of these photons can be quantified using the special receptor coils present in the scanner, and the information computed can be used to obtain tomographic images of the brain that identify the neurobiological variable of interest (eg, blood flow, glucose absorption, dopamine receptor density).
Positron emission tomography requires positron-emitting isotopes of chemical elements called radioactive nuclides. Nuclides are created in a cyclotron by adding positive charges to the nucleus of chemical elements commonly found in organic molecules, such as 11-carbon (11C), 15-oxygen (15O), 18-fluorine (18F), and 13-nitrogen (13N). With these nuclides, one can then create radiopharmaceuticals, which are molecules of biological significance that carry 1 of these radioactive elements and therefore emit radioactive energy (γ photons). Since the nuclide has an excess of protons, it releases a positively charged particle (a positron) in order to return to a more stable state. This positron collides with the negatively charged electrons that surround the nucleus, and, as a consequence of this collision (an annihilation event), 2 γ photons are created. These 2 γ photons are propelled in opposite directions (180°) from each other after the collision until they arrive at the detectors in the PET camera. Detectors in the PET camera that are located opposite from one another are connected and synchronized in a coincidence circuit, so that when they both receive a γ photon within a given time window, it can be identified as the result of an annihilation event that occurred at a specific point within the vector that connects the 2 detectors. Image-reconstruction algorithms can identify the exact position where the collision occurred and illustrate it in the tomographic brain image.
The nature and chemical design of the radiopharmaceutical are what determine the biological function that can be measured. In order to measure blood flow, one can choose 15O that has a short half-life (approximately 2 minutes) and can be used to create radioactive water molecules (H215O) that are injected intravenously. One can also use 15O to create radioactive carbon dioxide (C15O2), which can be inhaled. To measure metabolic activity, one can create and radioactively label a compound that cells will confuse with glucose (18F-fluorodeoxyglucose or 18F-FDG). The FDG will be absorbed and phosphorylated in cells just like glucose but will not be processed further in metabolic pathways and therefore remains trapped in the cell. Importantly, FDG will be absorbed proportionally to the metabolic needs of cells, just like glucose. As a result, metabolically hyperactive neurons (like those of an ictal focus) will trap more radioactive compound, and hypoactive neurons (like those in areas of neurodegeneration) will emit a lower proportion of γ photons. These changes will be reflected in the brain maps.
Changes in regional blood flow or metabolism can be used as indirect measures of brain activity, which can also be measured with alternative modalities (such as fMRI). Nevertheless, the application that is unique to nuclear medicine techniques is the assessment of neurotransmitter dynamics. A radioligand is a specific type of radiopharmaceutical designed to have great affinity for a target of interest and much lower affinity for all other targets, so that it will be cleared from the bloodstream and other structures rapidly but will remain attached (and detectable) to the target, usually a neurotransmitter receptor. The radioligand must also be able to cross the blood-brain barrier and be biologically inactive.8
Single photon emission computed tomography (SPECT) is also a nuclear medicine modality, but it differs from PET in the physical reactions and the particles that are emitted. The SPECT nuclides themselves (as opposed to emitted positrons) will collide with local electrodes to become more stable, and that reaction will emit a single photon (not 2 photons as in PET). The SPECT technique has worse spatial resolution and sensitivity than PET, and this is most evident in deep structures in which PET is superior. Also, SPECT is less versatile, as it cannot use the rich variety of nuclides that allow PET the measurement of a wide array of biological processes. Nevertheless, SPECT is much cheaper and generally more commonly available. The higher costs of PET are driven by different variables but most significantly by the need to have a cyclotron and radiopharmaceutical synthesis capabilities onsite (given the short half-life of its products). In contrast, SPECT compounds can be synthesized offsite. Commonly used elements in SPECT are technetium (99 mTc), iodine (123I), or xenon (133Xe). These nuclides can be attached to biological molecules to create SPECT radiopharmaceuticals, but the fit is more difficult, as technetium, iodine, or xenon are not naturally present in common biochemicals.8
HOW MUCH DO NEUROIMAGING TESTS COST?
The costs of neuroimaging tests vary across countries, states, cities, and hospitals. Still, some relative differences remain constant and illustrate price differences. Table 2 presents the Medicare reimbursement rates for these tests in Massachusetts.
Table 2.
Medicare Reimbursement Rates of Neuroimaging Tests in Massachusetts
| Examination | Current Procedural Terminology Code | Price, US$ |
| X-ray examination of skull, less than 4 views | 70250 | 42.06 |
| X-ray examination of skull, minimum of 4 views | 70260 | 53.73 |
| CT head/brain without contrast | 70450 | 207.46 |
| CT head/brain with contrast | 70460 | 270.97 |
| CT head/brain without and with contrast | 70470 | 289.24 |
| CT angiography head | 70496 | 477.92 |
| CT angiography neck | 70498 | 477.92 |
| MRI brain without contrast | 70551 | 463.37 |
| MRI brain with contrast | 70552 | 591.46 |
| MRI brain without and with contrast | 70553 | 733.14 |
| MR angiography head without contrast | 70544 | 449.52 |
| MR angiography head with contrast | 70545 | 560.98 |
| MR angiography head without and with contrast | 70546 | 704.79 |
| MR angiography neck without contrast | 70547 | 449.52 |
| MR angiography neck with contrast | 70548 | 561.77 |
| MR angiography neck without and with contrast | 70549 | 704.52 |
| fMRI brain by technician | 70554 | 610.94 |
| Brain imaging (PET), metabolic evaluation | 78608 | 1,265.43 |
Abbreviations: CT = computed tomography, fMRI = functional magnetic resonance image, MR = magnetic resonance, MRI = magnetic resonance image, PET = positron emission tomography.
WHAT ARE THE RISKS ASSOCIATED WITH NEUROIMAGING TESTS?
Although neuroimaging tests have a very good safety record, a number of complications should be considered by clinicians who order these tests. The complications (including the risk of radiation and contrast agents, the effects of magnetic fields on metallic implants, and the impact of claustrophobia) are specific to the imaging modalities.
Radiation
Computerized tomography scans have become a usual tool in multiple clinical specialties; it is estimated that 62 million scans are performed in the United States each year, including 4 million in children.9 The risks of radiation are dose dependent and have the potential to cause genetic mutations, including heritable changes and cancer. A standard CT scan has an effective dose of 10 mSv, which is thought to be associated with an increase in the risk of fatal cancer of 1 in 2,000. Since the natural incidence of fatal cancer in the United States is 1 in 5, the risk of radiation-induced cancer is much smaller than the natural risk for the disease.9 While these risks are small at the individual level, the high (and increasing) number of CT scans performed (including screening exams in healthy subjects) may have a true measurable impact at the population level.9
No significant risks have been identified for brain PET studies, particularly FDG-PET for the workup of neurodegenerative disorders,10 with an average dose of radiation of 8 mSv. Other diagnostic applications of PET, particularly whole-body PET-CT for the workup of abdominal or thoracic malignancies, present with higher rates of radiation (23–25 mSv) and risk for malignancies.11
Contrast
The most commonly used contrast agent for MRI imaging is gadolinium; it is generally well tolerated, with infrequent and mild complications. The focus of contrast-induced complications is on CT agents. The majority of these complications are mild and resolve completely with simple interventions. Nevertheless, severe and life-threatening reactions do exist and tend to manifest within 20 minutes of the injection.
The 2 types of CT imaging contrast materials are ionic (high-osmolar, most iodine-based) and nonionic (low-osmolar, eg, Iohexol).12 Nonionic contrast agents are the most widely used for neuroimaging applications (despite their higher cost) due to their better safety profile. Since the introduction of low-osmolar agents and the increased vigilance and sophistication of safety guidelines, the incidence and severity of contrast reactions have been dramatically reduced. However, the occurrence of life-threatening events has not changed; their occurrence is often unpredictable.12,13
The mechanism by which contrast agents cause complications is called “anaphylactoid,” because it is similar to the anaphylactic reaction caused by drugs or allergens. Idiosyncratic reactions (including nausea, flushing, hypotension, urticaria, and sometimes frank anaphylaxis) occur in approximately 5% of cases in which ionic agents are used14; being younger (< 1 year of age) and older (> 60 years of age) convey an increased risk, as does having a history of a contrast reaction, cerebrovascular disease, asthma, or allergies.13 Ionic agents are also linked to an increased risk for chemotoxic reactions, which occur primarily in the kidney and the brain. Renal chemotoxicity is manifest by reduced renal function or by renal failure, and the primary risk factors for it are renal insufficiency and dehydration.15 Ionic agents convey a higher risk of inducing cerebral vasospasm and seizures. Seizures occur in 1 in 10,000 cases, but their incidence can be as high as 1%–5% in those with impairment of the blood-brain barrier.16 The longer an ionic agent is in contact with the blood vessels, the higher the risk for vasospasm or seizure. Therefore, the rate and duration of the infusion should be kept to the shortest time necessary.
The potential of nonionic agents to cause coagulation and clotting is under review. Ionic agents have an anticoagulant effect that nonionic agents do not have. Therefore, when using nonionic agents, it is important to flush the syringes used with heparinized saline and to be mindful of the potential for clot formation. Clotting tends to be peripheral around the site of the injection.
The risk of death from use of ionic agents is 0.9 for every 100,000 administrations, and the risk for nonfatal complications is 157 for 100,000 procedures.1,17 For nonionic agents, the risk of death is similar but that of nonfatal complications is lower (126 per 100,000 doses).1,17 The risk of death from use of gadolinium is 1 in 5 million doses.18 Gadolinium has recently been associated with renal fibrosis, which is usually delayed by a few days to 2 months and has an unspecified but rare frequency. Published guidelines for the prevention and treatment of contrast-induced complications have been prepared by The American College of Radiology.12
Magnetic Fields
While MRI modalities typically do not convey a risk due to radiation or contrast reactions, other factors need to be taken into consideration. The main dangers for the patient undergoing an MRI are magnetic forces (torque, dislodgement) and heating of metallic foreign bodies or devices. Safety precautions in the MRI environment are a fundamental aspect of its use, and strict procedures and checklists have been established and are enforced by MRI technicians.14 While the detailed knowledge of these procedures is less relevant for clinicians, several key aspects should be considered when ordering a test. Patients with paramagnetic metallic implants (such as aneurism clips, brain stimulators [deep brain stimulator or vagus nerve stimulator], or pacemakers) are ineligible for MRI. These metallic foreign bodies could move or heat up, resulting in injuries to surrounding tissues. Certain tattoos (including those made with permanent eyeliner) use paramagnetic metallic components (suspended subcutaneously) as part of the injected ink and have the potential to absorb heat and burn the patient while in the scanner. In addition, one has to consider that the MRI magnet creates a very tight space, and patients with claustrophobia may be unable to remain still in the scanner for the duration of the examination because of intolerable anxiety. Similarly, children or patients with cognitive or behavioral impairments may not be able to undergo the test. This obstacle can sometimes be overcome with the use of sedation or with use of newer open MRIs. Open MRIs are also useful for patients who are too large or too heavy for standard scanners.1
HOW SHOULD NEUROIMAGING TESTS BE SELECTED?
A number of neuroimaging tests are available to the clinician, but often the question is which one to select to obtain the greatest diagnostic value with the best safety and lowest cost. Both technical and practical variables need to be considered.
Plain films of the skull are rarely used given their relatively low yield, although they can show fractures of the skull, erosions/hyperostosis, changes in the basal foramina, and inflammation of the sinuses and mastoids. Head CT has almost entirely replaced the use of plain films; it is as good, if not much better, for these indications, has an equally low risk of radiation, is widely available, and is relatively cheap.
Head CTs allow very good visualization of the skull and bone processes (including fractures), of calcium deposits (in the parenchyma or vessels), and of fresh blood (intraparenchymal, as in hemorrhagic strokes, or perimeningeal bleeds, as in epidural, subdural, or subarachnoid bleeds). Modern CT devices also allow appropriate visualization of midline structures and the ventricular system, providing sufficient diagnostic yield for cases of herniation and hydrocephalus. Edema, some tumors, and some abscesses can be visualized with CT, although MRI is clearly superior for these indications. CT is generally suboptimal for imaging of the posterior fossa and brain stem. For all other indications, MRI is superior.
MRI is the most sensitive imaging modality to examine the structure of brain parenchyma and white matter tracts, the gray-white matter boundary, the posterior fossa, and the brain stem. It is therefore the neuroimaging study of choice to visualize most lesions in patients. But some practical variables make CT better suited in certain cases.
The greatest practical advantage of CT, particularly over MRI, is its greater availability, much faster imaging time, lower cost, and good safety profile (particularly in the context of known or suspected metallic foreign bodies, when MRI is contraindicated). It is therefore the test of choice in emergency settings and in clinical environments in which access to an MRI scanner is limited.
Clinically, one should think of ordering a CT scan when a fracture or emergent mass effect is suspected, and CT continues to be the diagnostic modality of choice for the diagnosis of acute bleeds. Nevertheless, modern DWI sequences provide comparable sensitivity and specificity, although they require longer imaging times and are more costly. Disorders due to calcium deposits can also be visualized with CT better than with MRI, since calcium has weak paramagnetic properties. Scanning with CT will therefore provide important diagnostic information in calcifying tumors (eg, craniopharyngioma, oligodendroglioma, ependymomas, meningiomas, some metastasis), metabolic alterations (eg, parathyroidism), congenital disorders (eg, tuberous sclerosis, TORCH infections [toxoplasmosis, other infections, such as coxsackievirus, syphilis, varicella-zoster virus, HIV, parvovirus B19, rubella, cytomegalovirus, herpes simplex virus-2]), and idiopathic processes (such as Fahr’s disease). For all other indications, MRI is the modality of choice (Table 3).
Table 3.
Comparison of Advantages and Disadvantages of CT and MRI
| Variable | Head CT | MRI |
| Technical | Head trauma/skull fracture | Better for most examinations |
| Acute bleed | Parenchyma and | |
| Calcifications | white matter lesions | |
| Emergent mass effect/herniation process | Posterior fossa and brain stem | |
| Emergent hydrocephalus MRI contraindications (metallic foreign bodies, claustrophobia, size/weight) | CT contraindications (generally related to radiation risks: pregnancy, children < 3 y) | |
| Practical | Quick imaging time (emergencies) | Longer imaging time |
| Broad availability (emergencies) | Higher cost (although becoming less costly) | |
| Low cost | No radiation, safer for repeated studies |
Abbreviations: CT = computed tomography, MRI = magnetic resonance image.
PET and SPECT are slowly finding their space in the diagnostic workup of neuropsychiatric disorders. PET imaging is always preferable, and the only reasons why one would choose SPECT over PET is lack of access to a PET facility or because of SPECT’s lower cost.19 The primary application of these nuclear medicine techniques is in the workup of neurodegenerative dementias and seizures.
The definitive diagnosis of Alzheimer’s disease requires pathological analysis of brain tissue, typically done postmortem. The clinical diagnosis is based on history, physical examination, and neuropsychological testing. Nevertheless, diagnostic certainty can be difficult in many cases, particularly in the early phases of diagnosis, in atypical presentations, in younger patients, in patients with a high cognitive baseline, and in patients with comorbidities (such as mood and anxiety disorders or traumatic brain injury). In these cases, PET can be of great help in clarifying the diagnosis. Once a dementia has been diagnosed clinically, PET can also be of help in differentiating among the various subtypes such as Alzheimer’s disease, the different frontotemporal dementia variants, vascular dementia, and Creutzfeld-Jacob disease. This differentiation is of importance for prognosis and family planning, and as new treatments emerge (particularly disease-modifying drugs), it will also have therapeutic relevance, as it can influence the choice of medications.20
The most common PET modality used for the workup of dementias is FDG-PET, which measures neuronal glucose metabolism as a marker of synaptic function and density. The typical Alzheimer’s dementia pattern is hypometabolism of parietal and temporal regions (usually bilaterally), including the precuneus and posterior cingulate, with sparing of the primary somatosensory and visual cortices, the basal ganglia, and the cerebellum. As the disease progresses, frontal regions can also be affected. These changes can be observed in FDG-PET before any structural changes (eg, atrophy) can be detected with MRI and even in the early stages of mild cognitive impairment.10 The detailed description of the patterns of FDG-PET hypometabolism in the different dementias is beyond the scope of this review. A newer PET modality of increasing utility in the diagnosis of neurodegenerative processes uses a radioligand named Pittsburgh compound B (PiB). The PiB radioligand has great affinity for amyloid, and after crossing the blood-brain barrier, attaches to amyloid plaques in the brain. Therefore, PiB-PET identifies a different pathophysiologic mechanism, more directly linked to the disease process, by mapping the density of amyloid plaques in the brain. The diagnostic yield of PiB-PET and FDG-PET is similar,21 but the cost of PiB-PET is still superior.
The diagnosis of seizures is fundamentally based on the history, examination, and EEG. But in many instances, the diagnosis remains unclear. The EEG may be normal in the interictal or postictal phase and when the focus is deep. In such cases, PET or SPECT can be extremely helpful in identifying a seizure focus. FDG-PET is usually used and will typically show glucose hypermetabolism during the ictal phase and hypometabolism in the postictal phase. PET has an important role in the localization of the focus for cases in which surgical treatment is considered.22,23 Other less common applications of nuclear medicine techniques in neuropsychiatry are the evaluation of brain tumors (which tend to be hypermetabolic) and head trauma (particularly in the chronic phase if affective, behavioral, and cognitive symptoms are present and structural imaging is unrevealing).24,25
The emerging role of fMRI and diffuse tensor imaging as clinical modalities also needs to be considered. While these techniques remain primarily neuroscience research tools, their use in neurosurgical planning is expanding.7 Functional MRI allows for the mapping of functional areas (such as motor, language, and vision). This is of great relevance in cases in which the functional and structural anatomy do not overlay in the usual pattern due to congenital developmental factors or plastic restructuring in the context of a lesion. Diffuse tensor imaging maps the structural anatomy of white matter pathways in the brain, which need to be preserved to avoid disconnection syndromes.
WHEN SHOULD CLINICIANS CONSIDER ORDERING A NEUROIMAGING TEST?
A number of studies have looked at the use of CT to detect abnormalities in psychiatric patients. Across all studies, 12% of patients presented with abnormalities, although these tended to be nonspecific.26 The risk of presenting with an anomaly increases with age, with an abnormal neurologic examination, with acute change in mental status, with a history of brain trauma, or a history of alcohol abuse.26 Weinberger26 proposed 6 criteria for the use of brain imaging: confusion and/or dementia of unknown cause, first episode of a psychosis of unknown etiology, a movement disorder of an unknown etiology, anorexia nervosa, prolonged catatonia, and the first episode of major affective disorder or a personality change after the age of 50 years.
With the advent and popularization of MRIs, the focus shifted from CT to this more descriptive imaging modality. Many studies reported white matter changes, but as the field of MRI advanced, these changes were also described with comparable prevalence in asymptomatic subjects.14 Rauch and Renshaw27 reported the findings of a study that analyzed 6,200 patients hospitalized in a psychiatric institution over the course of 5 years and described that 1.6% had an unexpected and treatable finding. They described a high prevalence of white matter lesions, but these were present in 30% of the normal population.27 A smaller study by Moles and colleagues28 looked at psychiatrically hospitalized patients who received a CT scan in their institution and described that 53% had abnormalities, 11% had findings that influenced patient care, and 2% had reversible lesions.
Given the wide array of neuroimaging tools available to the clinician, the question becomes knowing when it is appropriate to order a test and what benefit one can expect. Unfortunately, there is an overwhelming lack of evidence to help answer this question and to guide the clinician evaluation of psychiatric patients. The decision remains a matter of clinical judgment, but a few principles can be useful. Decisions about ordering neuroimaging are based on the findings of a thorough history and examination, including good cognitive and neurologic examinations, which are critical in the decision process.
Clinicians should order neuroimaging when they suspect that an intracranial process causing a structural (or functional) observable change is responsible for the presenting signs and symptoms. Some would argue that neuroimaging should only be ordered if the results have the potential to change the treatment plan. Nevertheless, we believe that confirming or ruling out significant or life-threatening etiologies, avoiding further testing, and providing prognostic information is of sufficient clinical value to justify the test (particularly if the associated risks are low).
One should consider neuroimaging for a patient with an abrupt (within minutes) onset of symptoms, which is likely to be caused by vascular causes, migraines, or seizures (with the exception of idiopathic panic attacks). A patient presenting with a change in the level of consciousness (which could in fact be an emergency) will also be a candidate for neuroimaging. If the neurologic or cognitive examinations show deficits, the anatomic correlations of these findings to the primary psychiatric symptoms should be considered, and neuroimaging should be ordered. Also, patients with a history of head trauma (particularly with loss of consciousness), neurologic comorbidities, or whole-brain radiation are more likely to present with anatomic abnormalities related to the chief complaint. Testing should also be considered for patients with a history of cancer. Atypical presentations may reflect a structural cause of psychiatric symptoms, but a thorough differential should be considered. In atypically old age of onset, even with a standard clinical presentation, a structural process (eg, vascular, neurodegenerative, paraneoplastic, inflammatory) should be considered and ruled out with a thorough examination and neuroimaging. We lack sufficient evidence to suggest what age should be the threshold, but prior reports have suggested 50 years of age14; this recommendation remains reasonable.
The use of neuroimaging in the workup of first-break psychosis and prior to administration of electroconvulsive therapy (ECT) remains a topic under debate. While structural neuroimaging (CT or MRI) is common in multiple clinics and emergency departments to evaluate a first psychotic break, and it has been proposed in many guidelines, the evidence to support this imaging is mixed. The goal of imaging is to identify intracranial abnormalities causing the first psychotic symptoms that would require a treatment different than the standard of care. The percentage of cases that present with abnormal neuroimaging findings is low, and it is even lower for those abnormalities that would require an intervention. Nevertheless, given the very good safety profile of these tests, it is often argued that it is worth scanning to identify those few cases and to confirm the lack of brain structural abnormalities in all other patients. The debate is often centered around the cost-effectiveness of routine scanning, which is affected by multiple variables.29 The question of whether to scan or not is still unresolved, but clinicians should identify (according to the above-mentioned criteria) those cases in which neuroimaging may have a higher yield.
Patients requiring ECT are usually affected by severe and treatment-resistant symptoms (eg, depression, mania, psychosis, catatonia). These refractory cases have a higher likelihood of being caused by a disease mechanism that changes the structure of the brain (and can be identified with imaging); however, most are not. Another potential reason to use neuroimaging in the evaluation of patients undergoing ECT is to rule out intracranial pathology (eg, masses, aneurysms, arteriovenous malformations), which can lead to complications. The final decision to use neuroimaging will usually be made on an individual basis.30 TABLE 4 summarizes the proposed criteria for neuroimaging.
Table 4.
Guidelines for the Use of Neuroimaging in Psychiatric Patients
| Abrupt (minutes) onset of symptoms |
| Change in level of consciousness or arousal |
| Deficits in the neurologic examination (with anatomic relation to the chief complaint) |
| Deficits in the cognitive examination (with anatomic relation to the chief complaint) |
| History of: |
| Head trauma (with loss of consciousness) |
| Whole-brain radiation |
| Neurologic comorbidities |
| Cancer |
| Late onset of symptoms (> 50 y) |
| Atypical presentation (to be determined on an individual basis) |
| First-break psychosis (to be determined on an individual basis) |
| Electroconvulsive therapy candidates (to be determined on an individual basis) |
CASE VIGNETTE CONTINUED
Given the atypical nature of Mr A’s symptoms, their unusually quick onset, the description of cognitive deficits in the neuropsychological evaluation, and the corroboration by results of the Personality Assessment Inventory of an atypical profile suggestive of a nonidiopathic etiology, a more extensive workup was started (including ordering of an MRI), resulting in the identification of a nonenhancing T2 hyperintense mass in the right precentral gyrus (Figure 2 and 3). Following that test result, the neuro-oncology and neurosurgery departments were consulted. Thoracic, abdominal, and pelvic CT scans were obtained, and a lumbar puncture was performed; all were normal. Mr A’s symptoms progressively improved with the medications prescribed in the inpatient unit, and he became asymptomatic. Although he was concerned about the diagnosis of a brain tumor and the uncertainty of his prognosis, he felt relieved because he had a tangible explanation for his psychiatric symptoms. He was discharged, and a multidisciplinary outpatient clinic follow-up was planned. To confirm the diagnosis, a frameless biopsy was obtained that showed normal tissue; this was followed by a more precise biopsy with a stereotactic frame. The pathology report noted a grade II/III astrocytoma. Given the location of the mass over the right precentral gyrus (where the motor strip is usually located), the neurosurgery team decided that it was nonresectable due to the high risk of iatrogenic motor deficits. A second opinion was obtained, and a fMRI was done to map the location of the right motor cortex. To do this, fMRI scans were obtained while Mr A performed 4 separate tasks: lip pursing, finger tapping, hand clenching, and toe wiggle (Figure 4). To the team’s surprise, the functional anatomy of the primary motor cortex did not correspond to the expected structural anatomy (ie, the motor control of the left hemibody was not found in the right precentral gyrus where the tumor was found). Instead, activation was observed primarily in the ipsilateral left precentral gyrus, and to a lesser extent in the anterior margin of the tumor, with minimal (10%–15%) overlap. This remapping phenomenon could be explained by neural plasticity induced by a slow-growing tumor (similar to what has been described after successful recovery from stroke), and it justified the lack of motor symptoms despite having a tumor over the precentral gyrus. With this functional map in hand, the second neurosurgical team decided that the tumor was operable, and they proceeded. The surgery was successful and Mr A developed only minimal coordination and strength difficulties immediately postoperatively (these recovered within a few months). Mr A continued to receive outpatient neuropsychiatric follow-up, and his psychotropics were slowly tapered off and discontinued over the next 18 months (without a relapse of symptoms). He returned to work 2 weeks after surgery and has remained fully functional at a professional and personal level.
Understanding the nature of neuroimaging tests can guide their selection. This understanding leads to differential treatments and more targeted interventions for patients.
Footnotes
LESSONS LEARNED AT THE INTERFACE OF MEDICINE AND PSYCHIATRY
The Psychiatric Consultation Service at Massachusetts General Hospital sees medical and surgical inpatients with comorbid psychiatric symptoms and conditions. Such consultations require the integration of medical and psychiatric knowledge. During their twice-weekly rounds, Dr Stern and other members of the Consultation Service discuss the diagnosis and management of conditions confronted. These discussions have given rise to rounds reports that will prove useful for clinicians practicing at the interface of medicine and psychiatry.
Dr Camprodon is director of the Laboratory for Neuropsychiatry and Neuromodulation and of Translational Research, Division of Neurotherapeutics at Massachusetts General Hospital, Boston. Dr Stern is chief of the Psychiatric Consultation Service at Massachusetts General Hospital, Boston, and a professor of psychiatry at Harvard Medical School, Boston, Massachusetts.
Dr Stern is an employee of the Academy of Psychosomatic Medicine, has served on the speaker’s board of Reed Elsevier, is a stock shareholder in WiFiMD (Tablet PC), and has received royalties from Mosby/Elsevier and McGraw Hill. Dr Camprodon reports no conflicts of interest related to the subject of this article.
References
- 1.Yousem DM, Grossman RI. Techniques in neuroimaging. In: Yousem DM, Grossman RI, editors. Neuroradiology: The Requisites. 3rd ed. Philadelphia, PA: Mosby; 2010. [Google Scholar]
- 2.de Lucas EM, Sánchez E, Gutiérrez A, et al. CT protocol for acute stroke: tips and tricks for general radiologists. Radiographics. 2008;28(6):1673–1687. doi: 10.1148/rg.286085502. [DOI] [PubMed] [Google Scholar]
- 3.Goldstein MA, Price BH. Magnetic resonance imaging. In: Darin D, Dougherty DD, Rauch SL, et al., editors. Essentials of Neuroimaging for Clinical Practice. American Psychiatric Publishing, Inc; Washington, DC: pp. 21–74. [Google Scholar]
- 4.Bitar R, Leung G, Perng R, et al. MR pulse sequences: what every radiologist wants to know but is afraid to ask. Radiographics. 2006;26(2):513–537. doi: 10.1148/rg.262055063. [DOI] [PubMed] [Google Scholar]
- 5.Orringer DA, Vago DR, Golby AJ. Clinical applications and future directions of functional MRI. Semin Neurol. 2012;32(4):466–475. doi: 10.1055/s-0032-1331816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Logothetis NK. What we can do and what we cannot do with fMRI. Nature. 2008;453(7197):869–878. doi: 10.1038/nature06976. [DOI] [PubMed] [Google Scholar]
- 7.Kuhnt D, Bauer MH, Ganslandt O, et al. Functional imaging: where do we go from here? J Neurosurg Sci. 2013 Mar;57(1):1–11. [PubMed] [Google Scholar]
- 8.Dougherty DD, Rauch SL, Fischman AJ. Positron emission tomography and single photon emission computed tomography. In: Dougherty DD, Rauch SL, Rosenbaum JF, editors. Essentials of Neuroimaging for Clinical Practice. Washington, DC: American Psychiatric Publishing; 2004. pp. 75–92. [Google Scholar]
- 9.Brenner DJ, Hall EJ. Computed tomography: an increasing source of radiation exposure. N Engl J Med. 2007;357(22):2277–2284. doi: 10.1056/NEJMra072149. [DOI] [PubMed] [Google Scholar]
- 10.Bohnen NI, Djang DS, Herholz K, et al. Effectiveness and safety of 18F-FDG PET in the evaluation of dementia: a review of the recent literature. J Nucl Med. 2012;53(1):59–71. doi: 10.2967/jnumed.111.096578. [DOI] [PubMed] [Google Scholar]
- 11.Brix G, Lechel U, Glatting G, et al. Radiation exposure of patients undergoing whole-body dual-modality 18F-FDG PET/CT examinations. J Nucl Med. 2005;46(4):608–613. [PubMed] [Google Scholar]
- 12.American College of Radiology. ACR practice guidelines for the use of intravascular contrast media. http://www.acr.org/∼/media/ACR/Documents/PGTS/guidelines/IV_Contrast.pdf . Updated 2007. Accessed February 11, 2013.
- 13.Shehadi WH. Contrast media adverse reactions: occurrence, recurrence, and distribution patterns. Radiology. 1982;143(1):11–17. doi: 10.1148/radiology.143.1.7063711. [DOI] [PubMed] [Google Scholar]
- 14.Dougherty DD, Rauch SL. Neuroimaging in psychiatry. In: Stern TA, Rosenbaum JF, Fava M, et al., editors. Massachusetts General Hospital Comprehensive Clinical Psychiatry. 1st ed. Philadelphia, PA: Mosby; 2008. [Google Scholar]
- 15.Hayman LA, Evans RA, Fahr LM, et al. Renal consequences of rapid high dose contrast CT. AJR Am -J Roentgenol. 1980;134(3):553–555. doi: 10.2214/ajr.134.3.553. [DOI] [PubMed] [Google Scholar]
- 16.Witten DM, Hirsch FD, Hartman GW. Acute reactions to urographic contrast medium: incidence, clinical characteristics and relationship to history of hypersensitivity states. Am J Roentgenol Radium Ther Nucl Med. 1973;119(4):832–840. doi: 10.2214/ajr.119.4.832. [DOI] [PubMed] [Google Scholar]
- 17.Caro JJ, Trindade E, McGregor M. The risks of death and of severe nonfatal reactions with high- vs low-osmolality contrast media: a meta-analysis. AJR Am J Roentgenol. 1991;156(4):825–832. doi: 10.2214/ajr.156.4.1825900. [DOI] [PubMed] [Google Scholar]
- 18.Dougherty DD, Rauch SL, Luther K. Use of neuroimaging techniques. In: Stern TA, Herman JB, Slavin PL, editors. Massachusetts General Hospital Guide to Primary Care Psychiatry. 2nd ed. New York, NY: McGraw Hill; 2004. [Google Scholar]
- 19.Herholz K, Schopphoff H, Schmidt M, et al. Direct comparison of spatially normalized PET and SPECT scans in Alzheimer’s disease. J Nucl Med. 2002;43(1):21–26. [PubMed] [Google Scholar]
- 20.Dickerson BC. Diagnostic tests for Alzheimer disease: judicious use can be helpful in clinical practice. Neurol Clin Pract. 2012;2(2):154–157. doi: 10.1212/CPJ.0b013e31825a77ef. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lowe VJ, Kemp BJ, Jack CR, Jr, et al. Comparison of 18F-FDG and PiB PET in cognitive impairment. J Nucl Med. 2009;50(6):878–886. doi: 10.2967/jnumed.108.058529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Alavi A, Yakir S, Newberg AB. Positron emission tomography in seizure disorders. Ann N Y Acad Sci. 2011;1228:E1–E12. doi: 10.1111/j.1749-6632.2011.06161.x. [DOI] [PubMed] [Google Scholar]
- 23.Juhász C. The impact of positron emission tomography imaging on the clinical management of patients with epilepsy. Expert Rev Neurother. 2012;12(6):719–732. doi: 10.1586/ern.12.48. [DOI] [PubMed] [Google Scholar]
- 24.Hunter JV, Wilde EA, Tong KA, et al. Emerging imaging tools for use with traumatic brain injury research. J Neurotrauma. 2012;29(4):654–671. doi: 10.1089/neu.2011.1906. 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Edlow BL, Wu O. Advanced neuroimaging in traumatic brain injury. Semin Neurol. 2012;32(4):374–400. doi: 10.1055/s-0032-1331810. 10.1055/s-0032-1331810 Advanced neuroimaging in traumatic brain injury. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Weinberger DR. Brain disease and psychiatric illness: when should a psychiatrist order a CAT scan? Am J Psychiatry. 1984;141(12):1521–1527. doi: 10.1176/ajp.141.12.1521. [DOI] [PubMed] [Google Scholar]
- 27.Rauch SL, Renshaw PF. Clinical neuroimaging in psychiatry. Harv Rev Psychiatry. 1995;2(6):297–312. doi: 10.3109/10673229509017151. [DOI] [PubMed] [Google Scholar]
- 28.Moles JK, Franchina JJ, Sforza PP. Increasing the clinical yield of computerized tomography for psychiatric patients. Gen Hosp Psychiatry. 1998;20(5):282–291. doi: 10.1016/s0163-8343(98)00040-1. [DOI] [PubMed] [Google Scholar]
- 29.Albon E, Tsourapas A, Frew E, et al. Structural neuroimaging in psychosis: a systematic review and economic evaluation. Health Technol Assess. 2008;12(18):iii–iv. doi: 10.3310/hta12180. ix-163. [DOI] [PubMed] [Google Scholar]
- 30.Lisanby SH. Electroconvulsive therapy for depression. N Engl J Med. 2007;357(19):1939–1945. doi: 10.1056/NEJMct075234. 8. [DOI] [PubMed] [Google Scholar]