Abstract
In lek mating systems, females choose mates through indicators of quality, which males may exhibit by their performance of courtship displays. In temperate regions, displaying seasons are brief (one to two months), whereas in the tropics courtship seasons may be prolonged. Moreover, in temperate-breeding animals lekking behaviour can be energetically demanding, but little is known about the energy costs of lekking in tropical animals. Daily, over the course of a nearly seven-month-long breeding season, male golden-collared manakins (Manacus vitellinus) of Panamanian rainforests perform acrobatic courtship displays that markedly elevate heart rates, suggesting that they require high energy investment. Typically, animals of tropical lowland forests (such as manakins) exhibit a ‘slow pace of life’ metabolic strategy. We investigated whether male manakin courtship is indeed metabolically costly or whether the birds retain a low daily energy expenditure (DEE), as seen in other tropical species. To assess these questions, we calibrated manakin heart rate against metabolic rate, examined daily lek activity and, using telemetry, obtained heart rates of individual wild, lekking male manakins. Although metabolic rates peak during courtship displays, we found that males actually invest minimal time (only approx. 5 min d−1) performing displays. As a consequence, the DEE of approximately 39 kJ d−1 for male manakins is comparable to other lowland tropical species. The short, intense bursts of courtship by these birds make up only approximately 1.2% of their total DEE. Presumably, this cost is negligible, enabling them to perform daily at their arenas for months on end.
Keywords: energetics, heart rate telemetry, courtship, tropics, manakins
1. Introduction
Males of many species provide no direct benefits to their mates, nor help with parental care, but aggregate in leks to court females for purposes of copulating [1]. Under these conditions, females choose a partner among many males, presumably assessing indicators of quality, such as male ornaments, behaviour or position in the lek [1,2]. Such intensive male–male competition and/or selective female choice can lead to the evolution of exaggerated secondary sexual phenotypic and/or behavioural traits [3,4]. In temperate regions, lekking species may display across a one- to two-month-long season when the males are thought to expend considerable energy in courtship, here defined as time on the lek when many individual ‘courtship displays’ are performed. In birds, for example, male great snipe (Gallinago media) [5] and sage grouse (Centrocercus urophasianus) [6] spend many hours per day at their leks. Their courtship activity increases their metabolic rates, causing significant weight loss in some individuals (usually the less successful males) [6]. The large investment in courtship, however, can be rewarded by high reproductive success, as a few males monopolize most matings [1,7].
Many species of lekking birds live in the tropics [1], but studies of their energy investment in courtship are scarce. Compared with birds breeding at high latitudes, lowland tropical birds tend to develop slowly, invest less in reproduction each year, and display slow senescence and extended longevity, as well as a low metabolic rate; a series of life-history traits that collectively reflect a tropical ‘slow pace of life’ [8–17]. By tropical, we refer to the wet, lowland forest environment, as different metabolic life-history strategies appear to apply to tropical animals living at high elevations or in deserts [18].
Golden-collared (GC) manakins (18.1 g; Manacus vitellinus) are tropical passerine birds that are known for their elaborate courtship displays. GC manakins live in lowland forests of Panama. During a lengthy breeding season (up to seven months in duration), adult males spend much of the day at their leks engaged in male–male and male–female interactions that include performance of courtship displays. Courtship is dominated by the ‘jump–snap’ display, which involves vigorous and rapid jumping between young saplings interspersed with loud snaps [19–21]. Individual ‘wingsnaps’ as well as high frequency ‘rollsnaps’ are produced as the wings strike one another after being lifted rapidly over the birds’ back [22,23]. When displaying, heart rates are profoundly elevated in males, even briefly exceeding 1300 beats min−1 [24], one of the highest heart rates recorded for any avian/mammalian species [25,26]. This elevated heart rate suggests that male manakins expend considerable energy in the performance of courtship activities. Outside of courtship, however, GC manakins exhibit life-history traits typical of the ‘slow pace’ of tropical life, producing small clutches (one to two eggs [10,27]) and living as long as 14 years [9,28,29]. Thus, it is unclear whether, during the breeding season, male GC manakins are able to sustain a tropical slow pace of life in the face of the putative high energy demands of courtship or whether GC manakins are an exception to the general rule that tropical birds have a lower energy expenditure than temperate birds.
Efforts to assess organismal energy metabolism have used various measures that describe overlapping and distinct states of metabolism, including basal (BMR) and resting metabolic rate (RMR), as well as peak metabolic rate (PMR), which are determined under controlled conditions in the laboratory [11,30,31]. Field metabolic rate (FMR), or daily energy expenditure (DEE) is used to assess the average metabolic rate of an animal under natural conditions. Whereas the tropical ‘slow pace of life’ is associated with a relatively low BMR and PMR [11–17], there are insufficient data to assign a definitive FMR measure for lowland tropical birds, though a trend towards decreased FMR has been described [18].
Although these measures describe average energy expenditure, they are less useful for assessing energy demands of discrete behaviours. One can determine the absolute amount of energy required for a given behaviour by accounting for the duration of the behaviour (‘joule cost’) or as the increase in energy expenditure per unit time with an emphasis on the peak demand required (joules per sec; ‘power’ [32]). The latter measure is usually applied to behaviours involving rapid bursts of high energy. During a chase, for example, cheetahs are thought to display a brief 50-fold increase in their RMR [33]. This behaviour requires high power but low joule cost. By contrast, while running, a marathoner displays a minimal increase in metabolic rate, but for an extended time period. This involves a high joule cost but less power [34,35]; ‘joule cost’ is not to be confused with the evolutionary ‘cost of a trait’ [3,4,36]. Both types of behaviour might require the animal to develop specialized physiological and morphological adaptations, one for bouts of PMR and one for extended metabolic demands (reviewed in [37,38]). Such distinctions are especially valuable for understanding energy expenditure of discrete behaviours such as courtship.
Gathering accurate data on energy utilization of natural animal behaviours can be difficult, so captive animals are often studied, as was the case for cheetahs described above (by measuring O2 consumption on captive cats breathing into ventilated masks while running on treadmills [33]). Using telemetry and miniature transmitters, heart rate measures can be obtained from wild animals. They provide excellent temporal resolution, such that estimates of metabolic rate can be obtained in the order of seconds to minutes. This contrasts with more traditional techniques (e.g. the doubly labelled water method) that provide estimates across days [39–42]. Heart rate stands as a proxy for energy expenditure [42], so the application of heart rate telemetry can be a valuable technique to identify moment-to-moment energy expenditure [3,4,36].
Previous studies of GC manakins obtained estimates of BMR that suggested that they maintain metabolic rates comparable to other tropical species that lack vigorous courtship displays [12,43,44]. As the relationship between BMR and DEE is not clearly defined [45,46], we sought to obtain diurnal and nocturnal measures of energy expenditure of wild lekking males during their peak months of courtship. Heart rate telemetry, in combination with respirometry to estimate energy expenditure, enabled us for the first time to obtain estimates of energy expenditure during various hours of the day, as well as during specific courtship behaviours in free-living birds, something not previously possible (cf. [47,48]). Energy expenditure was estimated in relation to measures of daily lek activity as well as focal observations of individual males (this study and [24]). In addition, we compared these estimates to comparable data obtained previously from a similar, though non-lekking, tropical bird, the spotted antbird (Hylophylax naevioides) [49]. Our work shows that male manakins exhibit a tropical slow pace of life despite the performance of energetically costly displays. These data offer new insight into the energetics of tropical courtship.
2. Material and methods
Studies were conducted during the dry season (February–March) in forests around Gamboa, Panama (9°7′ N, 79°42′ W) and in facilities maintained by the Smithsonian Tropical Research Institute (STRI).
(a). Heart rate telemetry
Heart rates were recorded via telemetry using miniature transmitters as described previously for small birds [47,49–51], including GC manakins [24]. Using mistnets, eight adult male GC manakins were captured from five leks. Birds were given coloured leg-bands for individual identification. Out of eight birds, four were transferred to a laboratory facility within 10 km of their capture (and release) site, where transmitters were attached and respirometry calibration was performed. On the remaining four birds, transmitters were attached in the field and birds were released within approximately 10 min at the site of capture. Each transmitter (approx. 1 g; Sparrow Systems, Fisher, USA) has a 9 cm flexible wire antenna and two wire electrodes, and was glued to trimmed feathers on the centre of the bird's back. The two electrodes were inserted beneath the skin (for details, see [49,50]). The heart rate transmitters emitted a continuous, amplitude-modulated carrier signal that was frequency-modulated by heart muscle potentials. Transmitters attached in this way fall off after approximately 14 days, eliminating the need for recapture of the birds [52]. Every 10 min for 30 s, using a Yagi antenna connected to an AR8000 receiver (AOR Ltd., Japan), heart rates were recorded as sound files (mp3) on a digital recorder connected to the receiver (mp3 recorder, Edirol R09, Roland Inc., Japan). In this way, heart rate measures were obtained randomly with respect to behaviour, providing an accurate reflection of the bird's daily commitment to different energetic demands. Sound files were analysed using CoolEdit v. 2000 software (Syntrillium Software, USA). Bandpass filters (Fast Fourier transform filters) were applied to increase the signal-to-noise ratio between heart rate frequency and carrier frequency. Heart rate was measured by identifying 10 consecutive amplitude spikes, and then averaging the time intervals between each spike pair [51]; skeletal muscle potentials were irregular and easily distinguished from iterative potentials emanating from the heart.
(b). Calibration of heart rate versus O2 consumption
Energy metabolism is assumed to be equivalent to O2 consumption, as aerobic respiration restocks depleted ATP. Fick's equation relates heart rate to O2 consumption using stroke volume and the arterio-venous difference in O2 content, the product of which is defined as O2 pulse (OP) [53]. If OP is constant or varies in a predictable manner, a calibration between O2 consumption and heart rate can define this relationship [42,54]. If CO2 production is known, the respiratory exchange ratio (RER) of O2 consumption and CO2 production can be calculated in order to estimate metabolic rate [55]. Heart rate is not a measure of absolute energy expenditure as a potential error is associated with the metabolic rate estimates based on the RER obtained in the calibration equation [42,54]. Heart rate was calibrated against O2 consumption in four male GC manakins. Once fitted with a transmitter, the male was placed in an open flow respirometer consisting of an airtight plastic 2 l volume chamber that enabled simultaneous measurement of O2 inflow and CO2 outflow. Flow rate was adjusted to 1400 ml min−1, allowing for 95% equilibrium within 1.26 min or 99% equilibrium within 3.3 min, and ambient temperatures were 28±1°C. Heart rate was averaged across the 5 s preceding the respirometry measurement, after ensuring that heart rate was stable in the 60 s preceding the measurements. Birds could freely move around in the chamber, but when placed in a dark location, they were generally quiescent in the chambers. If individuals did not move vigorously to achieve high heart rates, we approached the chambers and birds were enticed to move. For each bird, RER was determined at seven to eight different heart rates. Instantaneous O2 consumption was calculated using the equation of Bartholomew et al. [56], after empirically determining its denominator. Following Withers [57], we obtained the rate of O2 consumption (for detailed methodological description, see [47,57]). These measurements were then used to obtain an equation relating heart rate to metabolic rate as described below.
(c). Comparative analyses of manakin energetics
Heart rate measures and estimates of DEE were compared with those obtained using a similar method in another lowland tropical bird of similar size, the spotted antbird (17.6 g) [49]. From six adult male antbirds, heart rates were recorded continuously and measurements were obtained every 5 min for 24 h (for additional details, see [49]). Additionally, we compared GC manakin DEE with estimates derived from several other models [18,58,59]. GC manakin minimal MR, the best estimate of BMR or RMR in wild birds in which environmental conditions and feeding status cannot be controlled, was compared with RMR estimates obtained from the equation of Aschoff & Pohl [60].
(d). Courtship activity
In a previous study, we found that courtship displays (in particular the production of mechanical wing sounds) are associated with extremely high heart rates, hence they are the most energy-demanding behaviours performed on the lek [24]. Pointing to their importance, wingsnap and rollsnap frequencies are also significant predictors of mating success [24]. We calculated average on-lek activity across the day using audio recordings (06.30 to 17.30). During our study period, sunrise occurred between 06.03 and 06.34, with sunset occurring between 18.10 and 18.35. A microphone attached to a digital recorder with more than 12 h of battery life was placed in a central position within five different leks (with 4±1 adult courting males) from which we obtained three 1-day audio recordings (see electronic supplementary material, table S1). To estimate daily social and courtship activity, audio files were analysed using CoolEdit v. 2000 software. Dividing the file into 1 h intervals, starting at 06.30, we counted the number of wingsnaps and rollsnaps between 06.30 and 07.30, which represented activity at 07.00, and so on. To estimate activity of individual males on a lek, we divided the hourly sums of mechanical sounds produced by the number of males occupying that lek. Breeding male GC manakins have very high lek attendance and on the day of recording, all males were known to be present on the lek. In addition, we used data from previously obtained video recordings (n = 17) and focal observations (n = 31) of individual males, to estimate mating success in relation to the time spent by males performing courtship displays [24]. High-speed videos (n = 87) of 17 courting males were used to measure the average duration of a courtship display. With focal observations, we measured the number of displays and rollsnaps performed by males as well as their mating success.
(e). Statistical analyses
SPSS v. 19 (IBM Inc., USA) was used for statistical analyses. Calibration relationships between heart rate and O2 consumption of birds in the respirometer were determined by least-square regressions. A generalized linear model (GLM) with metabolic rate as the dependent variable, individual as a random factor and heart rate as a covariate was used to determine the overall relationship between heart rate and O2 consumption based on the data collected for the four birds during calibration. The standard error of the estimate (SEE) and the coefficient of variation (COV = 100 × SEE/estimate) were calculated after Green [54]. To investigate the pattern of courtship activity, we used a GLM with hour of the day as the repeated measure (daytime) and leks as the between-subject factor. The pattern of heart rate across the day was examined using a GLM with hour of day as the repeated measure (daytime). Heart rates were log-transformed to obtain normality. Post hoc tests with Bonferroni corrections were used to identify differences of activity or heart rate between different time points. Differences between antbirds and GC manakins were investigated using t tests. If not indicated differently, values in the text are given as mean±s.e.m.
3. Results
(a). Patterns of daily courtship activity
Activity levels varied significantly across the day and also between leks (daytime: F3.5,34.6 = 9.159, p < 0.001; lek: F4,10 = 5.643, p = 0.012; interaction: F13.8,34.6 = 1.743, p = 0.266; figure 1). Thus, despite differences between leks in the magnitude of activity, leks were similar with respect to their patterns of daily courtship. Activity was generally elevated in the morning, declining to a nadir at 12.00, after which activity levels increased significantly to a peak at 14.00, and then declined significantly by 17.00 (figure 2).
Figure 1.
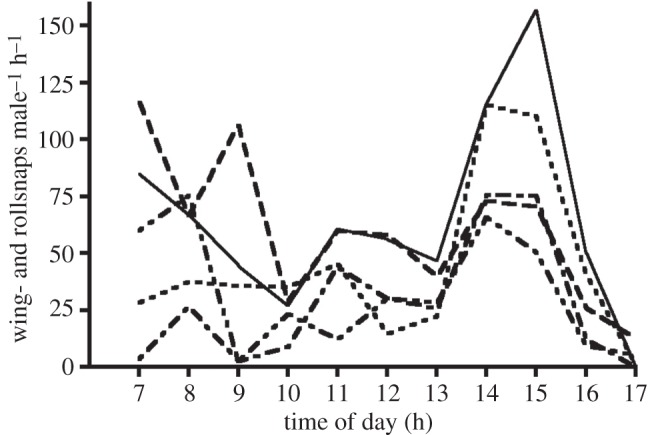
Diurnal pattern of courtship activity of male GC manakins. Mechanically produced sounds (wingsnaps and rollsnaps), measures of manakin social and courtship behaviour at the lek, were recorded for five different leks in three 1-day recordings. Hourly means were calculated including 30 min before and after the time point. Values were then divided by the number of males (4±1) on the corresponding lek, which was determined with direct behavioural observations. Mean values of sounds produced male−1 h−1 on each of the five leks are shown.
Figure 2.
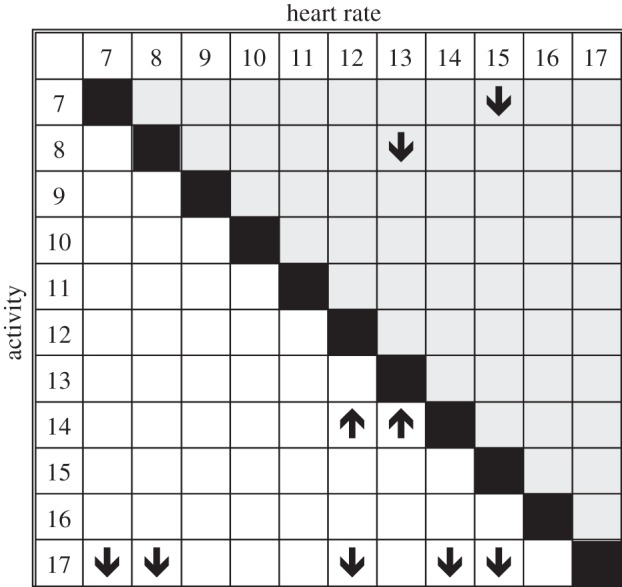
Hourly differences in activity and heart rate levels. Results of Bonferroni-corrected post hoc tests comparing activity and heart rate levels between different hours of the day (07.00 to 17.00). Arrows indicate significant differences. The shaded area shows values for heart rates and arrows refer to the hours on the top. For example, heart rate is lower at 13.00 compared with 08.00. The non-shaded area of the table refers to activity and arrows refer to hours on the left. An example is the significantly lower activity at 17.00 compared with 07.00 and 08.00.
(b). Patterns of daily heart rate
Telemetry transmitters added approximately 5.5% of a bird's body mass (18.1 g) and appeared to have minimal impact on the birds: males with transmitters were regularly observed courting and sometimes even copulating with females (see the electronic supplementary material, video S1, of [24]). Nevertheless, we cannot rule out the possibility that they have some impact on the birds, including increasing energy expenditure, leading to an overestimation of natural energy expenditure (reviewed in [61]).
For six birds, we measured heart rates across a complete 24 h span; heart rates during daylight hours were measured on two additional birds (61±12 diurnal recordings versus 17±4 nocturnal recordings; electronic supplementary material, table S2). When comparing hourly individual averages between 07.00 and 17.00, we found a significant effect of daytime on heart rate (F10,40 = 6.180, p < 0.001; figure 3). Heart rates rose steeply after 05.00, reaching a significant peak of 596±37 beats min−1 at 07.00, where they remained for another hour (figure 2). Following this early morning peak, heart rates significantly decreased to 394±14 beats min−1 at 13.00. For the remainder of the afternoon, heart rates were slightly elevated, reaching a peak at 18.00 (500±16 beats min−1), a time that corresponded with decreased activity on the lek (figure 2).
Figure 3.
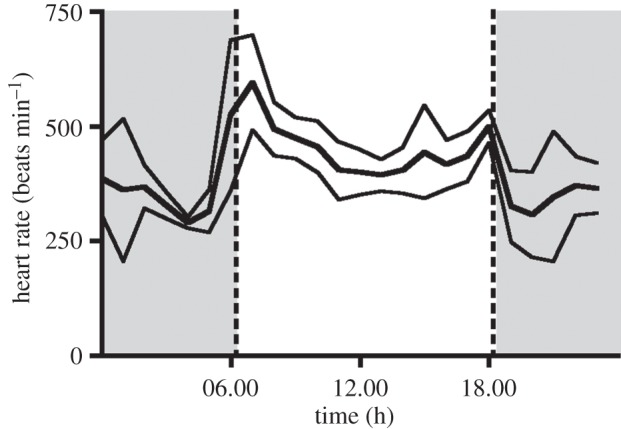
Daily heart rate pattern. The thick line represents the hourly mean heart rates of eight male GC manakins. Hourly averages were calculated for the 30 min before and after the time point. During the night, we obtained heart rate measurements of only six manakins. No measure was taken at 3.00 in the morning, thus we averaged values of 2.00 and 4.00 to complete the graph. Thin lines represent the 95% CIs (1 s.d.). Shaded areas indicate night time (18.12 until 06.15).
At sunset, heart rates markedly declined, with nocturnal rates generally low, decreasing to a min of 290±8 beats min−1 at 04.00. This result corresponds with previous findings of low nocturnal metabolic activity in GC manakins such that the birds appear to exhibit heterothermia, achieving a minimum nocturnal body temperature of 26.8°C [44]. Nevertheless, GC manakins retain the capacity to be responsive at night [44] as we observed discrete surges in nocturnal heart rate (approx. 700 beats min−1) in one bird, likely to indicate reactions to disturbance.
Taken together, these data allow us to calculate mean diurnal (active) heart rate of 450±15 beats min−1, calculated from 06.00 to 18.30, and a mean nocturnal heart rate of 332±23 beats min−1. Overall, the mean daily heart rate (24 h period) was 398±13 beats min−1.
(c). Calibration of heart rate versus O2 consumption
Heart rate calibration measurements spanned 382–943 beats min−1 (see electronic supplementary material, table S3), coinciding roughly with the range of heart rates recorded in the field (195–979 beats min−1). During courtship displays, average heart rates exceeded these ranges by 38 beats min−1 [24], so there may be some error in estimating metabolic rate at those elevated heart rates, as calibration of such high energy levels cannot be obtained in the laboratory. The RER was on average 0.88±0.01, which lies within the predicted range of 0.7–1 [30].
A linear relationship between O2 consumption and heart rate of captive birds is required to estimate energy expenditure from heart rates recorded in the wild. We found such a relationship in GC manakins (figure 4 and table 1), confirming what had been observed previously in other small birds [47,49,51]. When comparing the regressions obtained from the four birds, we found that intercepts were significantly different between individual regressions (F4,28 = 11.77, p < 0.001), but the slopes were the same (table 1). Following Green et al. [41], we obtained a final regression equation using the slope from the pooled data with an average of the individual intercepts to predict energy expenditure in the wild: MR = (ƒH × 0.148) − 19.796, where MR is the metabolic rate in kJ day−1 and ƒH is the heart rate in beats min−1. Eqn (11) from [41] was used to calculate the standard error of the estimated MR. The error associated with the variation between GC manakins (d2) was 48.5 (kJ2)(beats min−1)−2 and the error associated with the scatter around the regression lines (e2) was 8.1 × 10−5 (beats min−1)2.
Figure 4.
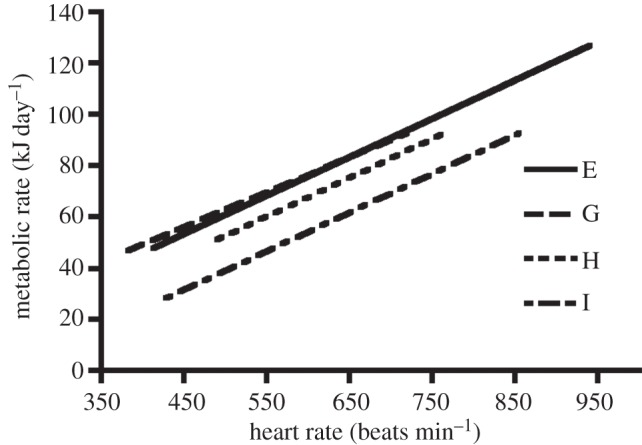
Relationship between heart rates and metabolic rates. Least-square regressions of heart rates against metabolic rates for four birds determined by calibration measurements: bird E (p < 0.001), bird G (p = 0.013), bird H (p < 0.001) and bird I (p < 0.001). See table 1 for statistical details.
Table 1.
Parameters of least-square regressions for heart rate against metabolic rate of four male golden-collared manakins. Birds equipped with a heart rate transmitter were placed in a respirometer, and heart rate was measured simultaneously with O2 consumption and CO2 production. The RER is used to estimate metabolic rate, following Walsberg & Hoffman [55]. bird, birds used for calibrations; n, number of measurements that have been taken at different heart rates; a, intercept; b, slope.
| bird | n | a | b | r2 | p |
|---|---|---|---|---|---|
| E | 7 | −13.8 | 0.15 | 0.97 | < 0.001 |
| G | 7 | −4.76 | 0.14 | 0.71 | 0.017 |
| H | 7 | −22.55 | 0.15 | 0.93 | <0.001 |
| I | 8 | −35.18 | 0.15 | 0.94 | <0.001 |
(d). Daily energy metabolism and comparative analyses
From the average daily heart rate, we estimated DEE to be 39.1±2.5 kJ day−1 (table 2). We are confident of these estimates given the relatively low 4–6% COV we obtained for each estimate. The lowest recorded nocturnal heart rate was used to estimate minimal MR, calculated to be 14.1±4.7 kJ day−1.
Table 2.
Heart rate measures and estimates of DEE of two tropical birds. DEE obtained with heart rate telemetry was compared between the golden-collared manakin (18.1 g) and the spotted antbird (17.6 g). Columns 2 and 3 are mean values for manakins and antbirds for the following variables: average diurnal heart rate (day), average minimal heart rate (night), average daily heart rate (24 h) and average DEE. Measures are reported as mean±s.e. (sample size). Columns 4 and 5 show the results of t-tests comparing the different variables between the two species. Manakins have lower heart rates for two of the three measures, but no difference is found in DEE. Also when comparing mass corrected DEE, we did not find a significant difference. Data on antbirds are taken from [49].
| variable | manakin | antbird | t (d.f.) | p |
|---|---|---|---|---|
| day (b min−1) | 450±15 (8) | 601±24 (6) | −5.534(12) | 0.000 |
| night (b min−1) | 332±23 (6) | 384±15 (6) | −1.896(10) | 0.087 |
| 24 h (b min−1) | 397±12 (6) | 492±17 (6) | −4.525(10) | 0.001 |
| DEE (kJ day−1) | 39.1±2.5 (6) | 37.4±2.4 (6) | 0.609(10) | 0.556 |
Manakins had significantly lower day (t12 = −5.534, p < 0.001) and 24 h (t10 = −4.525, p = 0.001) heart rates than those of spotted antbirds but no difference was found in DEE between the two species (t10 = 0.609, p = 0.556; table 2). Similar oxygen consumption combined with a lower heart rate suggests that manakins have an increased cardiac output, that is they eject a greater volume of blood with each heartbeat. This conclusion is supported by the limited available data on heart weight in these species. A higher cardiac output is typically associated with a larger heart mass; when heart weight to body weight ratios are calculated for these two species (dry heart weights over wet body weights from [62]), that of manakins (0.046/15 g = 3.1 × 10−3) exceeds that of antbirds (0.044/16.1 g = 2.7 × 10−3).
Our estimates of DEE in manakins obtained from heart rate measures compare favourably with those based on a general model for tropical birds [18], whereas estimates obtained from models for passeriformes in general [59] or songbirds from arid and mesic climates [58] were almost twice that of the GC manakins (table 3). Moreover, applying the equation of Aschoff & Pohl [60] that estimates BMR of temperate zone birds, a manakin-sized bird is predicted to have a BMR of 26 kJ day−1, a value twice the minimal MR of 14 kJ day−1 that we estimated in this study.
Table 3.
Estimates of DEE based on the heart rate method from this study and three different models based on birds living in the tropics, passeriformes, and passerines from arid and mesic climates. All model estimates are based on the body weight of manakins (18.1 g).
(e). Energy requirements for the courtship display
Male GC manakin heart rates rose during the performance of courtship displays and rollsnaps to 1017±28 and 1062±102 beats min−1, respectively [24]. For courtship displays, this rate corresponded to 131 kJ day−1 (5.5 kJ h−1) with a COV of 3% (corrected from [24]), which is equivalent to the PMR of manakins induced by exercise measured by Wiersma et al. [11]. Although overall lek activity lasted for many hours per day, males varied greatly in the total number of displays they performed, ranging from 1.6 to 140 displays day−1, producing an average of 32±5 displays day−1 (n = 31). Importantly, each display lasts on average 9.5±0.5 s (n = 17), thus males engage in courtship displays for only 5±1 min day−1 (0.25–22.2 min day−1; data from [24]). Consequently, they spent on average 0.46 kJ in courtship displays, which corresponds to 1.2% of their DEE. On average, males also produce 10.8±1.5 rollsnaps h−1, expending an additional 0.04 kJ day−1 on rollsnaps. Thus, the performance of the two most demanding courtship behaviours requires on average 0.5 kJ day−1. If we assume that the power required for a courtship display is similar across males, then the least active males would spend approximately 0.1% of their DEE in courtship displays compared with approximately 5.2% of the most active ones (see electronic supplementary material, figure S1). Although we have not measured energy investment in courtship in relation to mating success in GC manakins, it is likely that from least active to most active and successful males, GC manakins expend a minimal amount of energy in their courtship displays including the production of mechanical sounds.
4. Discussion
Despite their high-power-demanding courtship displays, heart rate measures of free-living birds indicate that GC manakins maintain a low DEE typical for tropical birds. Similar results are obtained by several independent analytic approaches. First, using data collected via similar techniques, we found no difference between DEE of GC manakins and the spotted antbird, a non-lekking sympatric bird with a low DEE [49]. Second, when we apply several models designed to provide general estimates of avian energy expenditure, manakins' energy expenditure is approximately 50% of what is expected [58,59]. Only the model by Anderson & Jetz [18] that is designed for tropical birds provides an estimate that is similar to what we obtained (table 3). In addition, compared with temperate-breeding birds, BMR is lower in tropical birds, including the GC manakin [12,43,44]. Our data confirm the measurements of BMR obtained by Vleck & Vleck [43] (21 kJ day−1) and Bartholomew et al. [44] (20 kJ day−1), which are both lower than what would be estimated by the equation of Aschoff & Pohl [60] for birds of the temperate zone (26 kJ day−1). Thus, our estimates of DEE as well as minimal MR together with previous estimates of BMR strongly suggest that GC manakins with their high-power-demanding courtship displays fall in the life history strategy classification of ‘slow pace of life’, typical for tropical birds [10,12,17,49].
We were surprised to discover that overall daily courtship activity in GC manakins lacks high joule costs. Although individual courtship displays per se have high power demands, reaching PMRs ninefold higher than minimal MR and threefold higher than DEE [11,24], males actually spend minimal time per day in courtship performance, using on average only 1.2% of their DEE on courtship dances. Short, highly intensive exercise bouts are thought to rely on anaerobic energy production, though there is good evidence that anaerobic metabolism plays a limited role in fuelling manakin courtship. First, heart rates decrease the moment the courtship display ends [24], evidence that the birds have not accumulated an O2 deficit. Second, one can estimate aerobic scope, the maximum O2 consumption of an animal during exercise over the animal's BMR, which for birds of 18.1 g such as manakins is estimated to be 36 [63]. We find that manakins reach only about nine times BMR during courtship, which is well within the animal's aerobic scope, hence courtship is likely to be sustained by aerobic metabolism. Thus, we are confident that our measures are valid estimates of energy expenditure during GC manakin courtship.
Collectively, males on a lek produce considerable noise in order to attract females and engage in courtship bouts in hopes of mating. Yet each individual invests minimal energy in the lekking enterprise. Even the most active male, performing as many as 140 courtship displays per day, invests only 5% of his DEE, because each display only lasts approximately 9.5 s. This strategy of low joule costs could explain why manakins are able to display for up to seven months of the year and retain a lengthy life span.
In contrast to GC manakins, other lekking species engage in courtship involving high joule costs [5,62,64,65]. For example, sage grouse males perform on average 1680 displays during one or two bouts per day [63,66]. Thus, each bout of displaying (totalling about 1.4 h day−1) probably functions as an endurance exercise [6]. These energy demands may impose limits on the duration of the season of courtship, which lasts only one to two months.
Courtship enables females to assess male quality for purposes of mating [67]. Presumably, males that engage in courtship that exacts high joule cost do so, at least partly, to demonstrate their capacity to withstand the behaviour-added energy demands [2–4]. The females might then choose males based on their behavioural and/or physiological adaptations to accumulate and efficiently metabolize the energy reserves required for courtship activity. Rather than being limited by total energy, male GC manakins appear to be performance-limited; that is, the courtship display may be designed to demonstrate physiological and morphological qualities associated with their rapid, acrobatic performances. For example, male manakins have hypertrophied skeletal muscles and fibres modified for increased muscular contraction [68,69]. It is conceivable that the high heart rates achieved during courtship performance reflect optimal physiological function that females indirectly assess via these courtship displays.
Although there are many advantages to the use of heart rate telemetry for estimating metabolic rates of wild animals [35,39–42,49,51,62], there are also limitations. The signal detection range in dense forest placed limits on our ability to simultaneously observe some behaviours. For example, these studies would have benefitted from heart rate measures when focal males were interacting momentarily with other males or females. To overcome this difficulty, we combined heart rate data from one group of birds with estimates of activity from other birds. In addition, we were unable to stimulate peak heart rates during the calibration in captivity. Thus, when predicting energy expenditure during courtship, we use values that are outside the calibration curve. Bishop & Spivey [70] derived a model that estimated metabolic costs from heart-rate data that can be applied to our dataset: body weight 18.1 g, heart weight 0.273 g, heart rate during display 1017 beats min−1 and during 24 h 450 beats min−1. Plugging our data into the model, we find that GC manakins require 2.592 l of O2 per day and 49 ml during the 5 min of courtship display, which corresponds to 1.9% of the daily budget, which is similar to the 1.2% estimated in our study [70]. Despite experimental limitations, we are confident that this work applying heart rate telemetry to study the energetics of courtship behaviour provides valid estimates of the energetics of a tropical bird during its peak courtship period.
In conclusion, our data indicate that this tropical bird species with a lek mating system does not experience periods of courtship demanding high joule costs, as has been described for some temperate-breeding birds. Despite being present on the lek for much of the day, GC manakins show off their morphological and physiological proficiency in short display bouts across the day while maintaining a tropical ‘slow pace of life’ metabolic strategy [32,67,71]. Many species of neotropical manakins engage in lekking behaviour, with a variety of patterns of timing and frequency of courtship [72–74]. Additional research is required to determine whether the low joule cost strategy of GC manakins is common to other tropical lekking birds.
Acknowledgements
We thank the Smithsonian Tropical Research Institute for support in Panama. The Princeton University 2008 tropical biology field course, G. Terranova and E. Choi contributed to data collection and processing. We thank J. Green for his advice on data analysis, and D. Altshuler and K. Nagy as well as anonymous referees for valuable comments.
All procedures for animal use were approved by UCLA Chancellor's Animal Research Committee, STRI and the Panamanian Autoridad Nacional del Ambiente.
Funding statement
This research was supported by NSF-IBN-021319 to B.A.S.
References
- 1.Höglund J, Alatalo RV. 1995. Leks. Princeton, NJ: Princeton University Press [Google Scholar]
- 2.Andersson M. 1994. Sexual selection. Princeton, NJ: Princeton University Press [Google Scholar]
- 3.Fisher RA. 1930. The genetical theory of natural selection. Oxford, UK: Oxford University Press [Google Scholar]
- 4.Zahavi A. 1975. Mate selection: a selection for a handicap. J. Theor. Biol. 53, 205–214 (doi:10.1016/0022-5193(75)90111-3) [DOI] [PubMed] [Google Scholar]
- 5.Höglund J, Kålås JA, Fiske P. 1992. The costs of secondary sexual characters in the lekking great snipe (Gallinago media). Behav. Ecol. 30, 309–315 [Google Scholar]
- 6.Vehrencamp SL, Bradbury JW, Gibson RM. 1989. The energetic cost of display in male sage grouse. Anim. Behav. 38, 885–896 (doi:10.1016/S0003-3472(89)80120-4) [Google Scholar]
- 7.Vitousek MN, Rubenstein DR, Nelson KN, Wikelski M. 2008. Are hotshots always hot? A longitudinal study of hormones, behavior, and reproductive success in male marine iguanas. Gen. Comp. Endocrinol. 157, 227–232 (doi:10.1016/j.ygcen.2008.04.015) [DOI] [PubMed] [Google Scholar]
- 8.Ricklefs RE. 1968. Patterns of growth in birds. Ibis 110, 419–451 (doi:10.1111/j.1474-919X.1968.tb00058.x) [Google Scholar]
- 9.Snow DW, Lill A. 1974. Longevity records for some neotropical land birds. Condor 76, 262–267 (doi:10.2307/1366339) [Google Scholar]
- 10.Ricklefs RE. 1976. Growth rates of birds in the humid New World tropics. Ibis 118, 179–207 (doi:10.1111/j.1474-919X.1976.tb03065.x) [Google Scholar]
- 11.Wiersma P, Chappell MA, Williams JB. 2007. Cold- and exercise-induced peak metabolic rates in tropical birds. Proc. Natl Acad. Sci. USA 104, 10 866–10 871 (doi:10.1073/pnas.0707683104) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wiersma P, Muñoz-Garcia A, Walker A, Williams JB. 2007. Tropical birds have a slow pace of life. Proc. Natl Acad. Sci. USA 104, 9340–9345 (doi:10.1073/pnas.0702212104) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Williams JB, Miller RA, Harper JM, Wiersma P. 2010. Functional linkages for the pace of life, life-history, and environment in birds. Integr. Comp. Biol. 50, 855–868 (doi:10.1093/icb/icq024) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Weathers WW. 1979. Climatic adaptation in avian standard metabolic rate. Oecologia 42, 81–89 [DOI] [PubMed] [Google Scholar]
- 15.Hails CJ. 1983. The metabolic rate of tropical birds. Condor 85, 61–65 (doi:10.2307/1367889) [Google Scholar]
- 16.Weathers WW. 1997. Energetics and thermoregulation by small passerines of the humid, lowland tropics. Auk 114, 341–353 (doi:10.2307/4089237) [Google Scholar]
- 17.Wikelski M, Spinney L, Schelsky W, Scheuerlein A, Gwinner E. 2003. Slow pace of life in tropical sedentary birds: a common-garden experiment on four stonechat populations from different latitudes. Proc. R. Soc. Lond. B 270, 2383–2388 (doi:10.1098/rspb.2003.2500) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Anderson KJ, Jetz W. 2005. The broad-scale ecology of energy expenditure of endotherms. Ecol. Lett. 8, 310–318 (doi:10.1111/j.1461-0248.2005.00723.x) [Google Scholar]
- 19.Chapman F. 1935. The courtship of Gould's manakin (Manacus vitellinus vitellinus) on Barro Colorado Island, Canal Zone. Bull. Am. Museum Nat. History 68, 471–525 [Google Scholar]
- 20.Schlinger BA, Day LB, Fusani L. 2008. Behavior, natural history and neuroendocrinology of a tropical bird. Gen. Comp. Endocrinol. 157, 254–258 (doi:10.1016/j.ygcen.2008.05.015) [DOI] [PubMed] [Google Scholar]
- 21.Fusani L, Schlinger B. 2012. Proximate and ultimate causes of male courtship behavior in golden-collared manakins. J. Ornithol. 153, 119–124 (doi:10.1007/s10336-011-0809-8) [Google Scholar]
- 22.Bostwick KS, Prum RO. 2003. High-speed video analysis of wing-snapping in two manakin clades (Pipridae: Aves). J. Exp. Biol. 206, 3693–3706 (doi:10.1242/jeb.00598) [DOI] [PubMed] [Google Scholar]
- 23.Fusani L, Giordano M, Day LB, Schlinger BA. 2007. High-speed video analysis reveals individual variability in the courtship displays of male golden-collared manakins. Ethology 113, 964–972 (doi:10.1111/j.1439-0310.2007.01395.x) [Google Scholar]
- 24.Barske J, Schlinger B, Wikelski M, Fusani L. 2011. Female choice for male motor skills. Proc. R. Soc. B 278, 3523–3528 (doi:10.1098/rspb.2011.0382) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lasiewski RC. 1964. Body temperatures, heart and breathing rate, and evaporative water loss in hummingbirds. Physiol. Zool. 37, 212–223 [Google Scholar]
- 26.Jürgens KD, Fons R, Peters T, Sender S. 1996. Heart and respiratory rates and their significance for convective oxygen transport rates in the smallest mammal, the Etruscan shrew Suncus etruscus. J. Exp. Biol. 199, 2579–2584 [DOI] [PubMed] [Google Scholar]
- 27.Skutch F. 1985. Clutch size, nesting success, and predation on nests of neotropical birds, reviewed. Ornithol. Monogr. 36, 575–594 (doi:10.2307/40168306) [Google Scholar]
- 28.Karr JR, Nichols JD, Klimkiewicz MK, Brawn JD. 1990. Survival rates of birds of tropical and temperate forests: will the dogma survive? Am. Nat. 136, 277–291 (doi:10.1086/285098) [Google Scholar]
- 29.Brawn JD, Karr JR, Nichols JD. 1995. Demography of birds in a neotropical forest Effects of allometry, taxonomy, and ecology. Ecology 76, 41–51 (doi:10.2307/1940630) [Google Scholar]
- 30.Kleiber M. 1961. The fire of life: an introduction to animal energetics. New York, NY: John Wiley [Google Scholar]
- 31.Brody S. 1945. Bioenergetics and growth. New York, NY: Hafner [Google Scholar]
- 32.Clark CJ. 2012. The role of power versus energy in courtship: what is the ‘energetic cost’ of a courtship display? Anim. Behav. 84, 269–277 (doi:10.1016/j.anbehav.2012.04.012) [Google Scholar]
- 33.Taylor CR. 1974. Running in cheetahs, gazelles, and goats: energy cost and limb configuration. Am. J. Physiol. 227, 848–850 [DOI] [PubMed] [Google Scholar]
- 34.Gill RE, et al. 2009. Extreme endurance flights by landbirds crossing the Pacific Ocean: ecological corridor rather than barrier? Proc. R. Soc. B 276, 447–457 (doi:10.1098/rspb.2008.1142) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hawkes LA, et al. 2011. The trans-Himalayan flights of Bar-headed Geese (Anser indicus). Proc. Natl Acad. Sci. USA 108, 9516–9519 (doi:10.1073/pnas.1017295108) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kotiaho JS. 2001. Costs of sexual traits: a mismatch between theoretical considerations and empirical evidence. Biol. Rev. Camb. Philos. Soc. 76, 365–376 (doi:10.1017/S1464793101005711) [DOI] [PubMed] [Google Scholar]
- 37.Speakman JR. 2000. The cost of living: field metabolic rates of small mammals. Adv. Ecol. Res. 30, 177–297 [Google Scholar]
- 38.Piersma T. 2011. Why marathon migrants get away with high metabolic ceilings: towards an ecology of physiological restraint. J. Exp. biol. 214, 295–302 (doi:10.1242/jeb.046748) [DOI] [PubMed] [Google Scholar]
- 39.Bevan RM, Woakes AJ, Butler PJ. 1994. The use of heart rate to estimate oxygen consumption of free-ranging balck-browed albatrosses Diomedea melanophrys. J. Exp. Biol. 193, 119–137 [DOI] [PubMed] [Google Scholar]
- 40.Bevan RM, Speakman JR, Butler PJ. 1995. Daily energy expenditure of tufted ducks: a comparison between indirect calorimetry, doubly labelled water and heart rate. Funct. Ecol. 9, 40–47 (doi:10.2307/2390088) [Google Scholar]
- 41.Green J, Butler PJ, Woakes AJ, Boyd IL, Holder R. 2001. Heart rate and rate of oxygen consumption of exercising macaroni pinguins. J. Exp. Biol. 204, 673–684 [DOI] [PubMed] [Google Scholar]
- 42.Butler PJ, Green JA, Boyd IL, Speakman JR. 2004. Measuring metabolic rate in the field: the pros and cons of the doubly labelled water and heart rate methods. Funct. Ecol. 18, 168–183 (doi:10.1111/j.0269-8463.2004.00821.x) [Google Scholar]
- 43.Vleck CM, Vleck D. 1979. Metabolic rate in five tropical bird species. Condor 81, 89–91 (doi:10.2307/1367864) [Google Scholar]
- 44.Bartholomew GA, Vleck CM, Bucher TL. 1983. Energy metabolism and nocturnal hypothermia in two tropical passerine frugivores, Manacus vitellinus and Pipra mentalis. Physiol. Zool. 56, 370–379 [Google Scholar]
- 45.Nagy KA. 2005. Field metabolic rate and body size. J. Exp. Biol. 208, 1621–1625 (doi:10.1242/jeb.01553) [DOI] [PubMed] [Google Scholar]
- 46.Ricklefs RE, Konarzewski M, Daan S. 1996. The relationship between basal metabolic rate and daily energy expenditure in birds and mammals. Am. J. Physiol. 147, 1047–1071 [Google Scholar]
- 47.Bisson I-A, Butler LK, Hayden TJ, Romero LM, Wikelski MC. 2009. No energetic cost of anthropogenic disturbance in a songbird. Proc. R. Soc. B 276, 961–969 (doi:10.1098/rspb.2008.1277) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bowlin MS, et al. 2010. Grand challenges in migration biology. Comp. Gen. Pharmacol. 50, 261–279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Steiger SS, Kelley JP, Cochran WW, Wikelski M. 2009. Low metabolism and inactive lifestyle of a tropical rain forest bird investigated via heart-rate telemetry. Physiol. Biochem. Zool. 82, 580–589 (doi:10.1086/605336) [DOI] [PubMed] [Google Scholar]
- 50.Cochran WW, Wikelski M. 2005. Individual migratory tactics of New World Catharus thrushes: current knowledge and future tracking options from space. In Birds of two worlds: the ecology and evolution of migration (eds Marra P, R. Greenberg), pp. 274–289 Baltimore, MD: Johns Hopkins University Press [Google Scholar]
- 51.Sapir N, Wikelski M, McCue MD, Pinshow B, Nathan R. 2010. Flight modes in migrating European Bee-eaters: heart rate may indicate low metabolic rate during soaring and gliding. PLoS ONE 5, e13956 (doi:10.1371/journal.pone.0013956) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Raim A. 1978. A radio transmitter attachment for small passerine birds. Bird Band. 49, 326–332 (doi:10.2307/4512391) [Google Scholar]
- 53.Fick A. 1870. Uber die Messung des Blutquantums in der Herzventrikeln. Sitz. Physik. Med. Ges. 2, 16 [Google Scholar]
- 54.Green JA. 2011. The heart rate method for estimating metabolic rate: review and recommendations. Comp. Biochem. Physiol. A 158, 287–304 (doi:10.1016/j.cbpa.2010.09.011) [DOI] [PubMed] [Google Scholar]
- 55.Walsberg GE, Hoffman TCM. 2005. Direct calorimetry reveals large errors in respirometric estimates of energy expenditure. J. Exp. Biol. 208, 1035–1043 (doi:10.1242/jeb.01477) [DOI] [PubMed] [Google Scholar]
- 56.Bartholomew GA, Vleck D, Vleck CM. 1981. Instantaneous measurements of oxygen consumption during pre-flight warm-up and post-flight cooling in sphingid and saturniid moths. J. Exp. Biol. 90, 17–32 [Google Scholar]
- 57.Withers PC. 1977. Measurement of VO2, VCO2, and evaopartive water loss with a flow-through mask. J. Appl. Physiol. 42, 120–123 [DOI] [PubMed] [Google Scholar]
- 58.Garamszegi LZ, Moreno J, Møller AP. 2006. Avian song complexity is associated with high field metabolic rate. Evol. Ecol. Res. 8, 75–90 [Google Scholar]
- 59.Nagy KA, Girard IA, Brown TK. 1999. Energetics of free-ranging mammals, reptiles and. Annu. Rev. Nutr. 19, 247–277 (doi:10.1146/annurev.nutr.19.1.247) [DOI] [PubMed] [Google Scholar]
- 60.Aschoff J, Pohl H. 1970. Der Ruheumsatz von Vögeln als Funktion der Tageszeit und der Körpergröße. J. Ornithol. 111, 38–47 (doi:10.1007/BF01668180) [Google Scholar]
- 61.Barron DG, Brawn JD, Weatherhead PJ. 2010. Meta-analysis of transmitter effects on avian behaviour and ecology. Methods Ecol. Evol. 1, 180–187 (doi:10.1111/j.2041-210X.2010.00013.x) [Google Scholar]
- 62.Kotiaho JS. 2000. Testing the assumptions of conditional handicap theory: costs and condition dependence of a sexually selected trait. Behav. Ecol. Sociobiol. 48, 188–194 (doi:10.1007/s002650000221) [Google Scholar]
- 63.Gibson RM, Bradbury JW. 1985. Sexual selection in lekking sage grouse: phenotypic correlates of male mating success. Behav. Ecol. Sociobiol. 18, 117–123 (doi:10.1007/BF00299040) [Google Scholar]
- 64.Kotiaho JS, Alatalo RV, Mappes J, Nielsen MG, Parri S, Rivero A. 1998. Energetic costs of size and sexual signalling in a wolf spider. Proc. R. Soc. Lond. B 265, 2203–2209 (doi:10.1098/rspb.1998.0560) [Google Scholar]
- 65.Thomas RJ. 2002. The costs of singing in nightingales. Anim. Behav. 63, 959–966 (doi:10.1006/anbe.2001.1969) [Google Scholar]
- 66.Wiley RH. 1973. The strut display of male sage grouse: a ‘fixed’ action pattern. Behaviour. 47, 129–152 (doi:10.1163/156853973X00319) [Google Scholar]
- 67.Byers J, Hebets E, Podos J. 2010. Female mate choice based upon male motor performance. Anim. Behav. 79, 771–778 (doi:10.1016/j.anbehav.2010.01.009) [Google Scholar]
- 68.Schultz JD, Hertel F, Bauch M, Schlinger BA. 2001. Adaptations for rapid and forceful contraction in wing muscles of the male golden-collared manakin: sex and species comparisons. J. Comp. Physiol. A 187, 677–684 (doi:10.1007/s00359-001-0239-9) [DOI] [PubMed] [Google Scholar]
- 69.Lowe RP. 1942. The anatomy of Gould's manakin (Manacus vitellinus) in relation to its display. Ibis 84, 50–83 (doi:10.1111/j.1474-919X.1942.tb03420.x) [Google Scholar]
- 70.Bishop CM, Spivey RJ. 2013. Integration of exercise response and allometric scaling in endotherms. J. Theor. Biol. 323, 11–19 (doi:10.1016/j.jtbi.2013.01.002) [DOI] [PubMed] [Google Scholar]
- 71.Irschick DJ, Herrel A, Vanhooydonck B, Van Damme R. 2007. A functional approach to sexual selection. Funct. Ecol. 21, 621–626 (doi:10.1111/j.1365-2435.2007.01281.x) [Google Scholar]
- 72.Bostwick KS. 2000. Display behaviors, mechanical sounds, and evolutionary relationships of the club-winged manakin (Machaeropterus deliciosus). Auk 117, 465–478 (doi:10.1642/0004-8038(2000)117[0465:DBMSAE]2.0.CO;2) [Google Scholar]
- 73.DuVal EH. 2007. Adaptive advantages of cooperative courtship for subordinate male lance-tailed manakins. Am. Nat. 169, 423–432 (doi:10.1086/512137) [DOI] [PubMed] [Google Scholar]
- 74.Shorey L. 2002. Mating success on white-bearded manakin (Manacus manacus) leks: male characteristics and relatedness. Behav. Ecol. Sociobiol. 52, 451–457 (doi:10.1007/s00265-002-0540-9) [Google Scholar]


