Abstract
The neuroendocrine state of new mothers may alter their neural processing of stressors in the environment through modulatory actions of oxytocin on the limbic system. We predicted that amygdala sensitivity to negatively arousing stimuli would be suppressed in postpartum compared to nulliparous women and that this suppression would be modulated by administration of oxytocin nasal spray. We measured brain activation (fMRI) and subjective arousal in response to negatively arousing pictures in 29 postpartum and 30 nulliparous women who received either oxytocin nasal spray or placebo before scanning. Pre- and post-exposure urinary cortisol levels were also measured. Postpartum women (placebo) demonstrated lower right amygdala activation in response to negative images, lower cortisol and lower negative photo arousal ratings to nulliparous women. Nulliparous women receiving oxytocin had lower right amygdala activation compared to placebo. Cortisol levels in the placebo group, and ratings of arousal across all women, were positively associated with right amygdala activation. Together, these findings demonstrate reductions in both amygdala activation and subjective negative arousal in untreated postpartum vs nulliparous women, supporting the hypothesis of an attenuated neural response to arousing stimuli in postpartum women. A causal role of oxytocin and the timing of potential effects require future investigation.
Keywords: postpartum, oxytocin, arousal, amygdale, cortisol
INTRODUCTION
Postpartum women undergo functional changes in the neuroendocrine circuitry that regulates emotional responding which may promote adaptive decreases in reactivity to non-infant-related stressors. The hormonal state of new mothers may alter their neural processing, and hence their perception of stressors in the environment through modulatory actions of oxytocin on the limbic system. The ability to distinguish effectively between threatening and harmless stimuli may help postpartum women balance the need to protect their infants from external threats against the negative consequences that enhanced vigilance in general might have on their own physiological stress response. Although the mechanisms underlying the postpartum changes in arousal response are not entirely clear, oxytocin is thought to play an important role in postpartum women’s heightened infant responsiveness and decreased stress response to non-infant stimuli (Carter et al., 2007; Mortimer, 2008; Strathearn et al., 2009). fMRI research in women (Strathearn et al., 2009) supports the central action of oxytocin in heightened maternal responsiveness to infant-related stimuli in postpartum women; however, it is not known whether there are oxytocin-related decreases in women’s neural responses to non-infant stressors.
During recent years, the neuropeptide oxytocin has earned considerable interest with regard to regulation of social interactions, attachment and stress in addition to its originally studied role in maternal physiology and behavior. Concurrent postpartum increases in oxytocin may serve as a protective factor for new mothers from the dramatic psychological and biological changes that accompany parturition (Carter et al., 2001). The neuropeptide may have a vital role in the regulation of postpartum women’s stress response via its actions on the hypothalamic–pituitary–adrenal (HPA) axis (Carter et al., 2001; Bartz and Hollander, 2006; Meyer-Lindenberg et al., 2011), which is itself an important regulating system of the stress response, mood and general arousal. Postpartum women demonstrate changes in HPA axis functioning, e.g. suppressed response to corticotrophin-releasing hormone (CRH), which may be related to breastfeeding (Altemus et al., 2001). Breastfeeding by itself has been shown to increase oxytocin (Drewett et al., 1982; Carter et al., 2007) and decrease stress (Mezzacappa and Katkin, 2002). Compared to bottle-feeding women, breastfeeding women with their possibly higher endogenous oxytocin levels (Drewett et al., 1982; Carter et al., 2007) show a blunted HPA response to physical and psychological stressors (Altemus, 1995; Altemus et al., 2001; Heinrichs et al., 2001; Groer et al., 2002; Tu et al., 2006). Animal models showed that oxytocin acts in the brain to decrease the stress response of new mothers by inhibiting CRH (Carter et al., 2001) and thereby suggest that oxytocin might mediate the stress-reducing effects of breastfeeding. Oxytocin administration is shown to lower cortisol in men and women (Ditzen et al., 2009), especially in conjunction with social support (Heinrichs et al., 2003); giving further evidence that oxytocin is complexly involved in postpartum women’s decreased stress responsivity.
The unique neuroendocrine state of new mothers may promote an altered perception and neural processing of the environment, possibly through inhibitory actions of oxytocin in the limbic system in response to stressors. Before the point of HPA axis activation, the brain must process the stimulus as a stressor (Tu et al., 2006), leaving open the possibility that oxytocin acts in the limbic system to modify the perception of stressors, in addition to possible direct oxytocin effects on the HPA axis. The reported decreases in HPA axis activity in new mothers could, therefore, reflect concurrent functional changes in the responsiveness of the prefrontal-limbic brain circuitry to environmental stressors. The amygdala is a likely site of this altered processing based on recent neuroimaging work demonstrating decreased right amygdala activation in response to emotional faces with oxytocin administration in men (Kirsch et al., 2005; Domes et al., 2007; Meyer-Lindenberg et al., 2011). However, a similar study in non-postpartum women found increased left amygdala (and to a lesser extent right amygdala) activation in response to fearful faces with nasal spray application (Domes et al., 2010). The authors argue that increased amygdala activation to oxytocin might facilitate child protection due to the sensitization toward possible threat. This interpretation is consistent with the hypotheses of the current study. We believe that infant-related threatening stimuli would produce a different response than the general non-infant-threatening negatively arousing stimuli used here. It is currently unknown whether the amygdala responds differently in postpartum vs nulliparous women in response to non-infant-related negatively arousing stimuli.
The current study aimed to characterize neural activation of postpartum women in response to negatively arousing non-infant-related stimuli and to ask whether oxytocin might modulate this activity. We hypothesized that oxytocin acts centrally in the amygdala to modulate neural processing of negatively arousing environmental stimuli, which may alter the subsequent behavioral and psychological responses of postpartum women to stressors. Using a between-subject double-blind placebo-controlled design in postpartum and nulliparous women, we measured brain activation (fMRI) to oxytocin/placebo in response to negatively arousing stimuli from the International Affective Picture System (IAPS). We predicted that postpartum women would show lower overall amygdala responses to negatively arousing non-infant-related stimuli since they were expected to be less responsive to the images given their generally attenuated stress response (Altemus et al., 2001). We also expected neural activity in the amygdala when viewing negative stimuli to be decreased in women receiving nasal oxytocin due to inhibitory influences of oxytocin on the HPA axis in response to negatively arousing images.
METHODS
Participants
Participants were 30 nulliparous and 29 postpartum women recruited through flyer, emails and local organizations. We included only heterosexual women currently in relationships, aged 20–40 years, who were not depressed, using antidepressants or other psychotropic medications, and not pregnant. Postpartum respondents were only eligible if they were primarily breastfeeding their infant (>75% breast vs bottle feeding) and had infants between 1 and 6 months old. Postpartum participants were phone-screened for postpartum depression using the Edinburgh Postnatal Depression Scale (EPDS; Cox et al., 1987). Only women scoring below 10 on the EPDS screening instrument were considered eligible. Due to the strong magnetic fields used for MR imagers, women who had magnetic life-support devices, metal prostheses or other metallic objects were excluded from this research.
Participants were assigned to either the placebo or oxytocin nasal spray group in a double-blind procedure. Specifically, at the time of scheduling and phone screening by the principal investigator (PI) and prior to mailing materials, participants were assigned to the next available subject ID number and group assignment (A or B) from a pre-generated list made by a co-investigator uninvolved with subject screening or testing. The PI and all experimenters and assistants who interacted with the participants and managed data did not know which spray was placebo or oxytocin (A and B). The code for spray group was broken only after all data had been collected and data analysis had begun.
Procedure
All procedures of the study were approved by the University’s (Indiana University) institutional review board committee and were in compliance with ethical treatment of human subjects guidelines. After the initial phone screening and scheduling, the women were mailed a participant packet for at-home completion including a questionnaire regarding demographics, health, menstrual cycle, motherhood and relationships with partners. Eligible nulliparous respondents were scheduled to come in for testing during the periovulatory phase of their menstrual cycle (Days 8–16) to control for general hormonal state. Participants were asked to abstain from alcohol, sexual activity and tobacco use the day of the test session. Postpartum women were requested to bring their infant with them to the test session and childcare was provided. At the test session, postpartum participants were asked to nurse their infant in a private adjacent room to enhance the comfort of the mothers and infants and to control for changes in oxytocin that occur with nursing (Altemus, 1995). Breastfeeding ended ∼1 h and 15 min before fMRI scanning. Following consent (and postpartum nursing), participants were administered a paper version of the 10-item EPDS. These scores as well as Likert scale ratings of self-reported feelings of anxiety (‘Do you currently feel anxious?’ Scale 0–7; 0 = not at all, 7 = very) were used as the mood variables in the current study. Following the questionnaire, participants provided a small (20 ml) urine sample for a baseline cortisol measurement. A second urine sample was collected following the scanning to monitor possible changes in cortisol across the session.
Next, participants were introduced to a laptop practice of the task that they would perform in the fMRI scanner. The task involved viewing negatively arousing pictures in addition to neutral control pictures. Pictures were taken from the IAPS (Lang et al., 2005), which includes photos with normative ratings of valence and arousal for each photo (1–9). Negative pictures used in the current study were low on valence (IAPS rating: mean ± s.d. = 1.78 ± 1.25) and high on arousal (IAPS rating: mean ± s.d. = 6.45 ± 2.20). Examples of negatively arousing pictures are a mutilated body, a gun pointed at the viewer, a growling dog or a car accident. Neutral pictures were mid-range on valance (IAPS rating: mean ± s.d. = 4.99 ± 1.15) and low on arousal (IAPS rating: mean ± s.d. = 2.76 ± 1.91). IAPS negative stimuli have been shown extensively to activate the amygdala (e.g. Garavan et al., 2001; Hariri et al., 2003; Britton et al., 2006) and can increase cortisol (Buchanan and Tranel, 2008). During photo presentation, participants performed a backward-matching task to ensure attention. After the task introduction, ∼30 min prior to the first run on fMRI scanning, participants received either nasal oxytocin or placebo nasal spray. Participants received an absolute dose of 24 IU, based on previous studies (Kosfeld et al., 2005; MacDonald et al., 2011). The oxytocin nasal spray (Syntocinon®, Novartis Pharma, Switzerland) contains a synthetic oxytocin nonapeptide (Kosfeld et al., 2005; MacDonald et al., 2011). The placebo spray contained only the inactive carrier found in the active oxytocin spray and is indistinguishable from the active spray (Meyer-Lindenberg et al., 2011). A recent study in primates demonstrated increased levels of oxytocin measured in CSF after oxytocin spray inhalation (Chang et al., 2012).
Imaging was carried out using a Siemens Magnetom Trio 3T whole-body MRI. The MRI session took ∼1 h, during which the following scans were acquired: (i) one 10-s three-plane scout/localizer used for choosing slice planes and volumes for the remaining scans, (ii) seven ∼5-min whole-brain functional blood-oxygenation-level-dependent (BOLD) scans and (iii) one 5-min high-resolution whole-brain anatomical scan. Each of the seven functional runs began with 12 s of rest to ensure a stable baseline signal. During each run, participants viewed 64 stimuli in a randomized order from each category of negative and neutral pictures. Each stimulus was presented for 2 s, followed by a variable inter-stimulus interval of 2–6 s. As described for the practice task, the participants performed a one-back matching task. Following the fMRI session, participants provided an additional urine sample. Participants then viewed the same stimuli as presented during the fMRI scanning on a laptop and rated them for how ‘aroused they make them feel’ (1–9).
Urine samples were frozen at −20°C until shipment for assay by the Assay Services laboratory at the University of Wisconsin National Primate Research Center. Cortisol levels were measured in 6 µl of urine by enzyme immunoassay (Ziegler et al., 1995). The assay/inter-assay coefficient of variation for a low pool was 2.5/3.8% and for a high pool was 4.1/4.7%.
Imaging parameters
Images were collected using a 32-channel phased-array head coil. The field of view was 220 × 220 mm, with an in-plane resolution of 128 × 128 pixels and 35 axial slices of 3.4 mm thickness per volume. These parameters produced voxels that were 1.7 × 1.7 × 3.4 mm. Functional images were collected using a gradient-echo BOLD echo-planar imaging sequence: TE = 24 ms, TR = 2000 ms, flip angle = 70°. Parallel imaging was used with a iPAT factor of 2. High-resolution T1-weighted anatomical volumes were acquired using a Turbo-flash 3D sequence: TI = 900 ms, TE = 2.67 ms, TR = 1800 ms, flip angle = 9°, with 192 sagittal slices of 1 mm thickness, a field of view of 224 × 256 mm and an isometric voxel size of 1 mm3.
Data analysis
Demographic information was compared using multivariate analysis of variance (ANOVA) with cohort and group and the between-subject factors. Cortisol data were analyzed using a repeated-measures ANOVA across pre- and post-time points with cohort and group as between-subject factors. We tested whether self-reported arousal ratings in response to images differed across cohort (nulliparous vs postpartum) or with nasal spray treatment group using a repeated-measures ANOVA across photo type with cohort and nasal spray as between-subject factors and age, percent body fat and EPDS score as covariates.
Imaging data were analyzed using BrainVoyager™ QX 2.2. Individual anatomical volumes were transformed into a common stereotactic space based on the reference of the Talairach atlas using an eight-parameter affine transformation. All functional volumes were re-aligned to a reference functional volume, which was the volume collected closest in time to the anatomical volume. Re-alignment was done using an intensity-based motion-correction algorithm. Functional volumes also underwent slice scan-time correction, 3D spatial Gaussian filtering (FWHM 6 mm) and linear trend removal. Functional volumes were co-registered to the anatomical volume using an intensity-based matching algorithm and normalized to the common stereotactic space using an eight-parameter affine transformation. During normalization, functional data were re-sampled to 3 mm3 isometric voxels. Whole-brain statistical parametric maps were calculated using a random-effect generalized linear model with predictors based on the timing protocol of the blocked stimulus presentation, convolved with a two-gamma hemodynamic response function. A whole-brain analysis was conducted using a three-factor 2 × 2 × 2 design with valence (negative and neutral), cohort and nasal spray as factors.
Since the amygdala is the most studied of these regions with respect to negative affect, we conducted a region of interest (ROI) analysis on this region. Performing analyses on a subset of the data can be more statistically sensitive than performing whole-brain analyses, because the correction for multiple tests is less harsh if there are fewer tests (Saxe et al., 2006; Poldrack, 2007). Thus, to further analyze the interaction between valence, cohort and nasal spray, we performed another ANOVA on the data from only the left and right amygdala clusters found with the main effect of the whole-brain analysis. Note that the main effect used to select the ROIs and the interaction effect tested in the ROI analysis are orthogonal contrasts, which protects the analysis from the problem of non-independent ROI analysis (Kriegeskorte et al., 2009). Beta weights were extracted from group ROIs using the analysis of covariance (ANCOVA) table tool in BrainVoyager’s volume of interest module. Statistical hypothesis testing was performed on the extracted beta weights using the repeated-measures ANOVA and multiple regression tools in SPSS. Specifically, data extracted from the right and left amygdala ROIs were examined using a 2 × 2 × 2 × 2 ANCOVA with valence (negative and neutral) and hemisphere as within-subject factors, cohort and nasal spray as between-subject factors and age, percent body fat and EPDS scores as covariates (due to group differences and effect of mood on amygdala activation). Follow-up post hoc analyses were conducted within cohorts and groups (multivariate ANOVAs with the same covariates used above).
In order to examine whether amygdala activation with negative images was significantly related to endogenous cortisol levels, we performed stepwise regression analyses on both activation in the right and left amygdala. Variables entered in as predictors were cortisol pre, cortisol post and cortisol change. Finally, we were interested in whether activation in the amygdala was related to women’s self-reports of arousal in response to the negative images. To examine this, we conducted one-tailed Spearman correlations between ratings of arousal for negative photos and average activation in the right and left amygdala. This test was used because we had a directional hypothesis and the subjective arousal ratings data were not normally distributed.
RESULTS
Sample
Participant characteristics by group are presented in Table 1. The majority of participants self-reported as White (n = 47, 80%; 22 nulliparous), six were Asian (four nulliparous), three were Black (all nulliparous), one was Hispanic/Latino (postpartum) and two participants self-described themselves as ‘Other’ (one nulliparous). The majority of women (n = 26, 44%) reported an education level at the Bachelor’s degree level, 17 reported completing up to high school (8 postpartum) and 16 had post-graduate degrees (11 postpartum). Average age of participants was 27 years old; however, nulliparous women were significantly younger than postpartum women (nulliparous: mean ± s.d. = 23.8 ± 3.74, postpartum: mean ± s.d. = 30.21 ± 4.44, P ≤ 0.001). Ten of the postpartum women reported currently using hormonal contraceptives, none of the nulliparous women did. Within postpartum women, eight women reported having resumed menstruation, 21 did not. There were no differences in questionnaire or neural responses based on hormonal contraceptive use and menstruation. All postpartum women were breastfeeding (average percent breast vs bottle feeding, mean ± s.d. = 87% ± 17.55). There was no difference in self-reported anxiety or depression between postpartum or nulliparous women, both reporting low levels (anxiety, Likert score 0–7, mean ± s.d. = 0.29 ± 0.49; EPDS score 1–9, mean ± s.d. = 4.78 ± 3.51). Women reported on average 7 h of sleep a night; there was not a significant difference between nulliparous (mean ± s.d. = 7.24 ± 1.46) and postpartum (mean ± s.d. = 7.45 ± 1.53) women. We did observe a significant difference in weight (P = 0.04) and percent body fat (P = 0.001) in which postpartum women were heavier (weight kgs, Mean SD nulliparous = 62.69 ± 12.27 kgs, Mean ± SD postpartum = 69.73 ± 11.81 kgs; for percent body fat: nulliparous: mean ± s.d. = 25.84 ± 7.76, postpartum: mean ± s.d. = 33.82 ± 8.89). None of the variables discussed above differed significantly by nasal spray assignment group.
Table 1.
Participant characteristics by group.
| Nulliparous | Postpartum | Total | |
|---|---|---|---|
| White (N) | 22 | 25 | 47 |
| Asian (N) | 4 | 2 | 6 |
| Black (N) | 3 | 0 | 3 |
| Hispanic/Latino (N) | 0 | 1 | 1 |
| Other Race (N) | 1 | 1 | 2 |
| High School (N) | 9 | 8 | 17 |
| Bachelors (N) | 16 | 10 | 26 |
| Post Graduate | 5 | 11 | 16 |
| Age (Mean Years ± SD)* | 23.8 ± 3.74 | 30.21 ± 4.44 | 26.95 ± 5.19 |
| Anxiety (Mean ± SD) | .32 ± .55 | .25 ± .44 | .29 ± .49 |
| Depression (Mean EPDS ± SD) | 5.42 ± 4.51 | 4.24 ± 2.36 | 4.78 ± 3.51 |
| Sleep (Mean hours ± SD) | 7.24 ± 1.46 | 7.45 ± 1.53 | 7.34 ± 1.48 |
| Weight (Mean kg ± SD)* | 62.69 ± 12.35 | 69.73 ± 11.81 | 66.15 ± 12.50 |
| % Body Fat (Mean ± SD)* | 25.84 ± 7.76 | 33.82 ± 8.89 | 29.76 ± 9.18 |
*Significant difference between groups (p < .05)
Cortisol
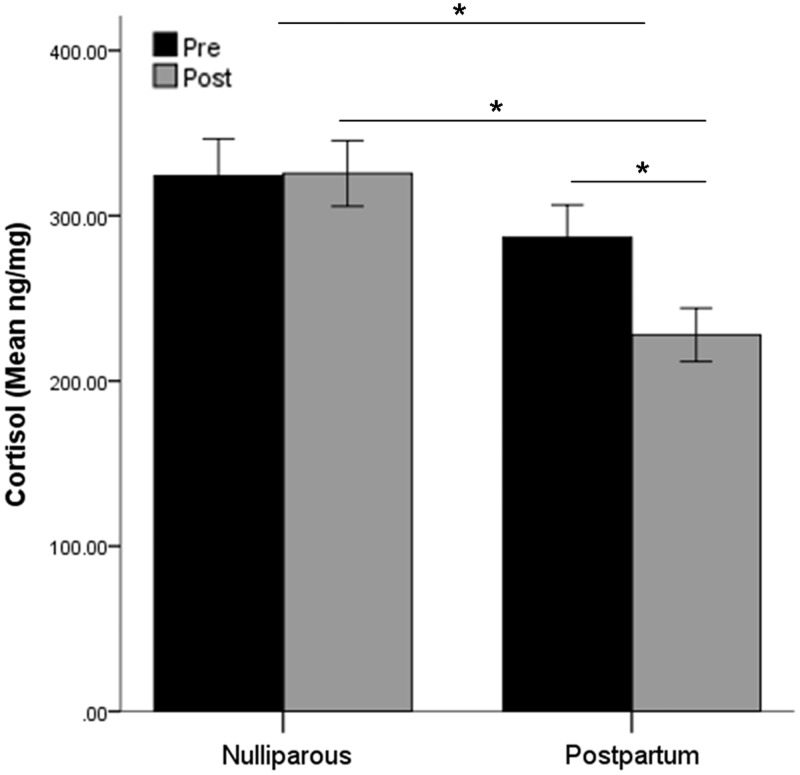
Data for two nulliparous women (pre and post for one participant and post for another) and one postpartum woman (pre) were excluded from analysis as outliers (>3 s.d. from the mean). Of the remaining participants, women’s average cortisol levels at baseline were 307.60 ng/mg (n = 57, s.d. = 112.26) and 278.12 ng/mg following fMRI scanning (n = 57, s.d. = 106.60). Nulliparous women had higher cortisol levels overall compared to postpartum women [repeated-measures ANOVA, F(1,54) = 7.02, P = 0.01]. However, there was an interaction of time and cohort [F(1,54) = 7.40, P = 0.009; Figure 1]. At baseline, nulliparous and postpartum women did not differ in cortisol levels [nulliparous: mean ± s.d. = 327.70 ± 117.84, postpartum: mean ± s.d. = 286.80 ± 104.19; F(1,56) = 1.92, P = 0.17]. There was a difference post-scan, however, in which nulliparous were significantly higher than postpartum [nulliparous: mean ± s.d. = 325.59 ± 115.95, postpartum: mean ± s.d. = 232.29 ± 87.09; F(1,56) = 13.31, P = 0.001]. For nulliparous women there was not a significant change in cortisol from before to after the MRI scan (P = 0.94, mean ± s.d. change = 1.40 ± 90.85 ng/mg), while for postpartum women cortisol decreased over time (P < 0.001, mean ± s.d. change = − 58.91 ± 74.26 ng/mg). There was no effect of nasal spray group on cortisol levels.
Fig. 1.
Average cortisol levels before and after MRI scanning in nulliparous and postpartum women. Nulliparous women had higher levels than postpartum women overall and post-scan. Only postpartum women demonstrated a decrease over time.
Photo ratings
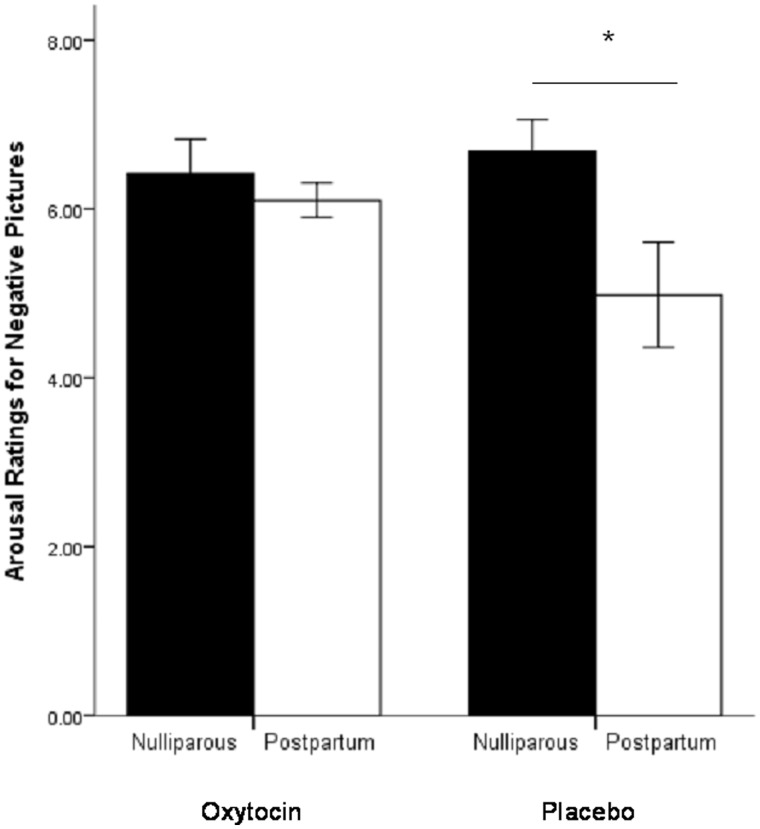
Three women were not included in the photo ratings analysis; two women (one in each cohort) rated all pictures as ‘1’ (which suggests that did not actually evaluate the stimuli) and one postpartum woman did not complete the ratings due to time constraints. There was a significant interaction of photo type, cohort and nasal spray [F(1,41) = 4.24, P = 0.05]. Post hoc analyses demonstrated that within the placebo group nulliparous women rated negative photos, but not neutral photos, to be more arousing than did postpartum women [nulliparous: mean ± s.d. = 6.68 ± 1.44, postpartum: mean ± s.d. = 4.98 ± 2.32, F(1,28) = 5.71, P = 0.02; Figure 2]. However, within the oxytocin group there was no difference between nulliparous and postpartum women in ratings of arousal for negative stimuli [nulliparous: mean ± s.d. = 6.42 ± 1.53, postpartum: mean ± s.d. = 6.11 ± .73, F(1,26) = 0.45, P = 0.51; Figure 2]. Further analyses within cohorts demonstrated no difference between oxytocin and placebo groups in either postpartum [placebo: mean ± s.d. = 4.98 ± 2.33, oxytocin: mean ± s.d. = 6.10 ± .73; F(1,26) = 2.77, P = 0.11] or nulliparous women [placebo: mean ± s.d. = 6.68 ± 1.44, oxytocin: mean ± s.d. = 6.42 ± 1.53; F(1,28) = 0.23, P = 0.63].
Fig. 2.
Average ratings of arousal for negative photos within nasal spray groups by cohort. Nulliparous women receiving the placebo spray rated the photos to be more arousing than postpartum women receiving the placebo.
Whole-brain contrasts
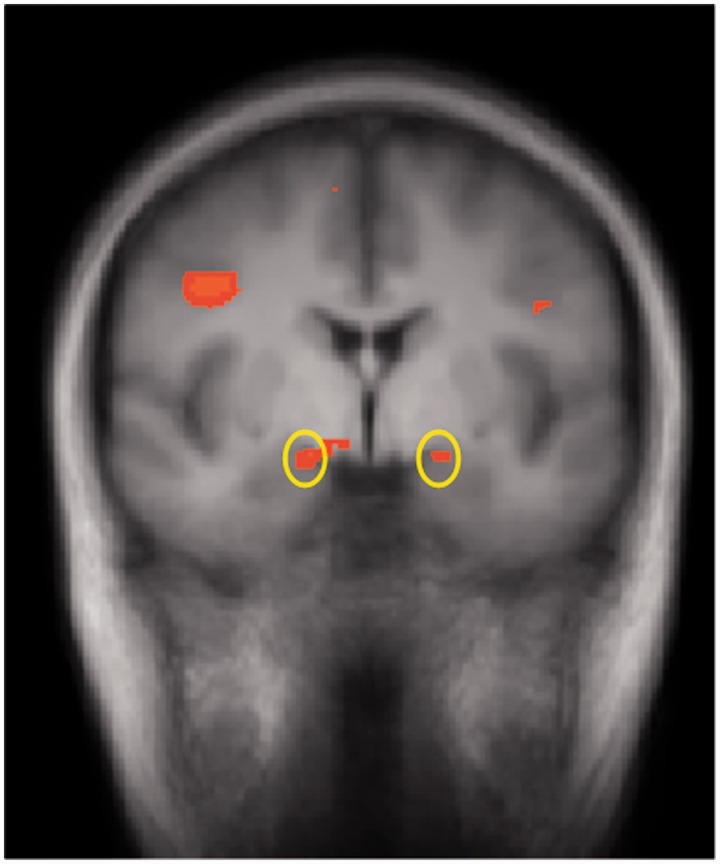
fMRI data from six of the women were not usable due to excessive motion artifacts or technical issues at the time of scanning, leaving a total sample of 53 (13 nulliparous placebo, 14 nulliparous oxytocin, 12 postpartum placebo and 14 postpartum oxytocin) for the brain imaging analysis. Contrary to our hypotheses, no clusters passed a false discovery rate corrected threshold (q = 0.05) for the main effect of cohort, the main effect of nasal spray or any of the interaction terms. Consistent with an extensive previous literature, there was a significant main effect of valence in the bilateral amygdala (Talairach coordinates: right, 13, −2, −9; left, −17, −3, −9; Figure 3), and also in four other brain regions, the right dlPFC (37, 4, 24), the midbrain (4, −26, −4), the thalamus (0, −13, 0) and the bilateral occipitotemporal cortex.
Fig. 3.
Coronal slice showing functionally defined amygdala ROIs (circled in yellow) determined from main effect of negative–neutral (15, −4, −11), i.e. collapsing across cohort and spray condition.
ROI: amygdala
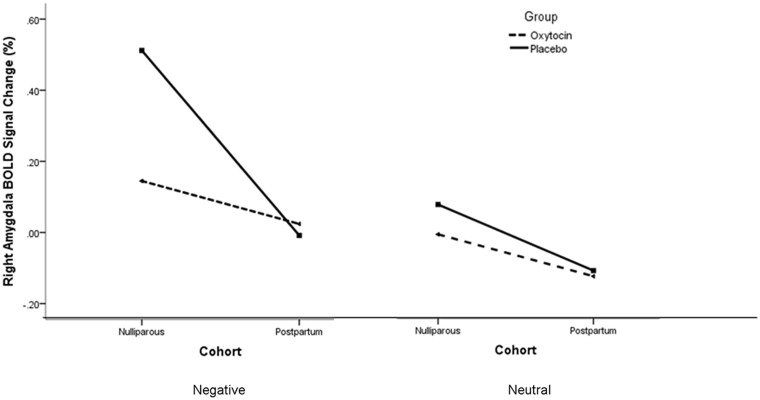
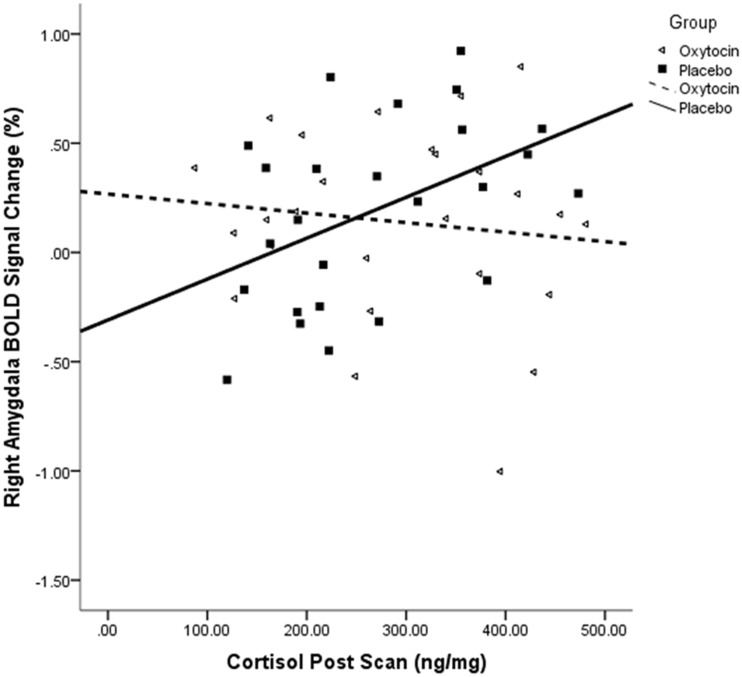
There was an interaction effect of hemisphere, valence and cohort [F(1,35) = 4.20, P = 0.05]. Follow-up post hoc analyses found a main effect of parturition within the placebo [F(1,21) = 5.64, P = 0.03], but not oxytocin spray, group [F(1,22) = 0.07, P = 0.79]. Within the placebo group, postpartum women had lower right amygdala activation than nulliparous women did in response to negative (Figure 4), but not neutral, images. The absence of group differences in response to negative images within the oxytocin group appears due to nulliparous women’s lower amygdala responses to negative stimuli with oxytocin vs placebo administration [F(1,19) = 5.83, P = 0.03], thus diminishing the difference between nulliparous and postpartum ratings that was observed in the placebo group (Figure 4). Regression analyses demonstrated a significant positive relationship between cortisol post with right amygdala activation in response to negative images within the placebo group (n = 25, R2 = 0.21, r = 0.45, P = 0.02; Figure 5). Correlation analysis also showed a positive association between right amygdala activation and women’s self-reports of arousal (n = 50, R = 0.24, P = 0.05).
Fig. 4.
BOLD signal change as a function of stimulus valence, cohort and nasal spray group. There was a significant effect of cohort in only the placebo group in which nulliparous women had a higher right amygdala response than postpartum did in response to negative stimuli. Nulliparous women in the oxytocin group had lower levels of activation to negative pictures than did nulliparous women in the placebo group. This pattern was not observed for neutral images.
Fig. 5.
BOLD signal change by nasal spray group in relation to cortisol levels taken following the fMRI scan. There was a significant positive relationship between cortisol (post) and right amygdala activation in response to negative images within the placebo, but not oxytocin, group.
DISCUSSION
The first aim of the study was to determine whether there were lower levels of arousal in postpartum women in response to negative images. This study did demonstrate differences in both subjective and amygdala responses to negatively arousing stimuli in postpartum vs nulliparous women. As predicted, postpartum women in the placebo group, when compared to nulliparous, demonstrated both decreased subjective ratings of arousal and decreased right amygdala activation in response to negatively arousing IAPS images. In conjunction with the findings demonstrating lower cortisol levels across the test session in placebo postpartum participants, the study supports the idea that there is a decreased perception of and reaction to environmental stressors in the postpartum period in healthy postpartum women (Altemus, 1995; Altemus et al., 2001; Heinrichs et al., 2001; Tu et al., 2006).
The second aim of the study was to examine whether observed differences in responses to arousing stimuli were related to oxytocin through the administration of a nasal oxytocin spray. We predicted that oxytocin would blunt responses to negatively arousing stimuli. This hypothesis was partially confirmed for both fMRI results and subjective assessments in that the parturitional differences reported above were only significant within the placebo, but not oxytocin spray groups. On their own, this pattern of findings suggests that differences between nulliparous and postpartum women are decreased with the administration and possible elevation of oxytocin. For the fMRI results, this interpretation is more strongly supported by findings showing lower right amygdala responses to negative stimuli in nulliparous women receiving the oxytocin spray vs placebo. This finding in nulliparous women of a decreased amygdala response with oxytocin administration is consistent with previous findings in which oxytocin was administered to men (Kirsch et al., 2005; Domes et al., 2007). However, this pattern is inconsistent with the Domes et al. (2010) which used fearful faces as stimuli, in contrast to the current study that used negative IAPS images. Additionally in contrast to the Domes et al. (2010) study, the nulliparous women in our study were all tested during the late follicular phase of their menstrual cycle, rather than the luteal phase, suggesting possibly that the existing hormonal state, specifically levels of progesterone in relation to estrogen, in which oxytocin is administered may mediate the observed effect in women. Similarly, postpartum women may not have responded to exogenous oxytocin spray in our study the same way as nulliparous women due to pre-existing postpartum changes in neuroendocrine function and their current hormonal state. Postpartum women may be ‘primed’ in a sense from breastfeeding and general maternal physiology to have a system that is either de-sensitized to the exogenous oxytocin, or possibly already saturated from endogenous elevations of this hormone.
Also of interest from this study were findings demonstrating direct relationships between amygdala activation to negative stimuli and cortisol. We observed a positive association between right amygdala activation in response to negative stimuli and endogenous cortisol levels (post-scan) in our placebo group, consistent with previous work (Buchanan and Tranel, 2008). The absence of this relationship in the oxytocin spray group further suggests a direct relationship between oxytocin and changes in HPA axis function. For the women in our study who received oxytocin, there was a disrupted relationship between amygdala responses to negative images and subsequent cortisol levels. We cannot be sure whether this is due to peripheral or central actions of the nasal oxytocin in the brain due to the limitations in our understanding and measurement of the relationship between central and peripheral oxytocin in humans. Wherever the target, however, it is interesting that increasing oxytocin had an immediate and detectable effect on the relationship between women’s amygdala responses and cortisol levels. These findings must be interpreted with caution; however, since time of day of the sessions and cortisol sampling was not controlled for and may be confounded by the diurnal rhythm of this hormone.
Additional limitations of our study warrant mention in order to inform future work and interpretation of the current results. Although the use of the oxytocin nasal spray methodology is a significant strength, its relative novelty allows for some outstanding questions. The dosage and timing used was based on previous studies in men and women (i.e. Kosfeld et al., 2005) rather than a dose or neuroendocrine response expected to be reflective of postpartum neuroendocrinology. It is not known whether lower or higher doses would produce different patterns of results, or for instance, whether there is some ‘threshold’ over which levels of oxytocin alter responsivity in women or reach a ceiling effect. We believe our timing of administration relative to fMRI scanning was delayed enough for action in the brain to be captured, but before it would have left the system. However, this can not be confirmed. Future work examining the dose response curve and timing of oxytocin action in humans or primates would be useful for future study designs using nasal oxytocin and for our understanding of oxytocin in the maternal brain. Additionally, all of our postpartum women were breastfeeding, which may have altered the sensitivity or responsiveness of these women to the nasal oxytocin. Future research should compare postpartum women who are not breastfeeding in order to separate out this possible effect. Finally, although our nulliparous and postpartum participants were very similar on many parameters, they did differ in age and use of hormonal contraception, which may have undetected impacts on women’s arousal response given associations of these variables with cortisol responsiveness (Carr et al., 1979; Kirschbaum et al., 1996). An ideal follow-up experiment would be a within-subjects design.
In sum, this study supports an emerging literature demonstrating functional changes in postpartum women’s neuroendocrine and behavioral response to stressors, and extends support for a causal role of oxytocin on this change. Nulliparous women in our study who received nasal oxytocin demonstrated decreased amygdala responsiveness to negative images and in that sense were pushed in the direction closer to the maternal brain. What is unknown is whether the observed induced changes in nulliparous women here can be thought of as representative of the ‘start’ of the maternal neuroendocrine change that accompanies parturition. We believe that the priority of future work should be to understand the changes in oxytocin responsiveness in postpartum women longitudinally to further understand the neuroendocrine timeline that initiates and subsequently maintains maternal behavior. Given our differing results in nulliparous women from Domes et al. (2010), other future work should also examine the possible mediation of other hormones, specifically progesterone. This would have implications not only for our understanding of the healthy maternal brain, but also for women susceptible to postpartum depression who may not demonstrate the observed changes in stress responsiveness with parturition.
Acknowledgments
We would like to thank Toni Zeigler and the Wisconsin National Primate Research Center for cortisol analysis and consultation. Diane Ebling at the Indiana University Health Center did safety monitoring, for which we are appreciative. We are grateful to Bloomington Area Birth Services for help with recruitment and guidance. The following are thanked for assistance with data collection: L. Reckley, F. Munoz, B. Keller, C. Anderson, C. White and Q. Class. Finally, we want to thank the MRI operators B. Ward and T. Atwood. This study was supported by grant #NIMH (R21MH082925 to H. Rupp and J. Heiman).
Conflict of Interest
None declared.
REFERENCES
- Altemus M. Neuropeptides in anxiety disorders—effects of lactation. Annals of the New York Academy of Sciences. 1995;771:697–707. doi: 10.1111/j.1749-6632.1995.tb44721.x. [DOI] [PubMed] [Google Scholar]
- Altemus M, Redwine LS, Leong Y, Frye CA, Porges SW, Carter SC. Responses to laboratory psychosocial stress in postpartum women. Psychosomatic Medicine. 2001;63:814–21. doi: 10.1097/00006842-200109000-00015. [DOI] [PubMed] [Google Scholar]
- Bartz JA, Hollander E. The neuroscience of affiliation: forging links between basic and clinical research on neuropeptides and social behavior. Hormones and Behavior. 2006;50:518–28. doi: 10.1016/j.yhbeh.2006.06.018. [DOI] [PubMed] [Google Scholar]
- Britton JC, Taylor SF, Sudheimer KD, Liberzon I. Facial expressions and complex IAPS pictures: common and differential networks. Neuroimage. 2006;31:906–19. doi: 10.1016/j.neuroimage.2005.12.050. [DOI] [PubMed] [Google Scholar]
- Buchanan TW, Tranel D. Stress and emotional memory retrieval: effects of sex and cortisol response. Neurobiology of Learning and Memory. 2008;89:134–41. doi: 10.1016/j.nlm.2007.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr BR, Parker CR, Madden JD, MacDonald PC, Porter JC. Plasma levels of adrenocorticotropin and cortisol in women receiving oral contraceptive steroid treatment. Journal of Clinical Endocrinology and Metabolism. 1979;49:346–9. doi: 10.1210/jcem-49-3-346. [DOI] [PubMed] [Google Scholar]
- Carter CS, Altemus M, Chrousos GP. Neuroendocrine and emotional changes in the post-partum period. Progress in Brain Research. 2001;133:241–9. doi: 10.1016/s0079-6123(01)33018-2. [DOI] [PubMed] [Google Scholar]
- Carter CS, Pournajafi-Nazarloo H, Kramer KM, et al. Oxytocin: behavioral associations and potential as a salivary biomarker. Annals of the New York Academy of Sciences. 2007;1098:312–22. doi: 10.1196/annals.1384.006. [DOI] [PubMed] [Google Scholar]
- Chang SW, Barter JW, Ebitz RB, Watson KK, Platt ML. Inhaled oxytocin amplifies both vicarious reinforcement and self reinforcement in rhesus macaques (Macacca mulatta) Proceedings of the National Academy of Sciences. 2012;109:959–64. doi: 10.1073/pnas.1114621109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox JL, Holden JM, Sagovsky R. Detection of postnatal depression: development of the 10-item Edinburgh Postnatal Depression Scale. British Journal of Psychiatry. 1987;150:782–6. doi: 10.1192/bjp.150.6.782. [DOI] [PubMed] [Google Scholar]
- Ditzen B, Schaer M, Gabriel B, Bodenmann G, Ehlert U, Heinrichs M. Intranasal oxytocin increases positive communication and reduces cortisol levels during couple conflict. Biological Psychiatry. 2009;65:728–31. doi: 10.1016/j.biopsych.2008.10.011. [DOI] [PubMed] [Google Scholar]
- Domes G, Heinrichs M, Glascher J, Buchel C, Braus DF, Herpertz SC. Oxytocin attenuates amygdala responses to emotional faces regardless of valence. Biological Psychiatry. 2007;62:1187–90. doi: 10.1016/j.biopsych.2007.03.025. [DOI] [PubMed] [Google Scholar]
- Domes G, Lischke A, Berger C, et al. Effects of intranasal oxytocin on emotional face processing in women. Psychoneuroendocrinology. 2010;35:83–93. doi: 10.1016/j.psyneuen.2009.06.016. [DOI] [PubMed] [Google Scholar]
- Drewett RF, Bowen-Jones A, Dogterom J. Oxytocin levels during breast-feeding in established lactation. Hormones and Behavior. 1982;16:245–8. doi: 10.1016/0018-506x(82)90023-x. [DOI] [PubMed] [Google Scholar]
- Garavan H, Pendergrass JC, Ross TJ, Stein EA, Risinger RC. Amygdala response to both positively and negatively valenced stimuli. Neuroreport. 2001;12:2779–83. doi: 10.1097/00001756-200108280-00036. [DOI] [PubMed] [Google Scholar]
- Groer MW, Davis MW, Hemphill J. Postpartum stress: current concepts and the possible protective role of breastfeeding. Journal of Obstetric, Gynecologic, and Neonatal Nursing. 2002;31:411–7. doi: 10.1111/j.1552-6909.2002.tb00063.x. [DOI] [PubMed] [Google Scholar]
- Hariri AR, Mattay VS, Tessitore A, Fera F, Weinberger DR. Neocortical modulation of the amygdala response to fearful stimuli. Biological Psychiatry. 2003;53:494–501. doi: 10.1016/s0006-3223(02)01786-9. [DOI] [PubMed] [Google Scholar]
- Heinrichs M, Baumgartner T, Kirschbaum C, Ehlert U. Social support and oxytocin interact to suppress cortisol and subjective responses to psychosocial stress. Biological Psychiatry. 2003;54:1389–98. doi: 10.1016/s0006-3223(03)00465-7. [DOI] [PubMed] [Google Scholar]
- Heinrichs M, Meinlschmidt G, Neumann I, et al. Effects of suckling on hypothalamic-pituitary-adrenal axis responses to psychosocial stress in postpartum lactating women. The Journal of Clinical Endocrinology and Metabolism. 2001;86:4798–804. doi: 10.1210/jcem.86.10.7919. [DOI] [PubMed] [Google Scholar]
- Kirsch P, Esslinger C, Chen Q, et al. Oxytocin modulates neural circuitry for social cognition and fear in humans. The Journal of Neuroscience. 2005;25:11489–93. doi: 10.1523/JNEUROSCI.3984-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirschbaum C, Platte P, Pirke K, Hellhammer D. Adrenocortical activation following stressful exercise: further evidence for attenuated free cortisol responses in women using oral contraceptives. Stress and Health. 1996;12:137–43. [Google Scholar]
- Kosfeld M, Heinrichs M, Zak PJ, Fischbacher U, Fehr E. Oxytocin increases trust in humans. Nature. 2005;435:673–76. doi: 10.1038/nature03701. [DOI] [PubMed] [Google Scholar]
- Kriegeskorte N, Simmons WK, Bellgowan PSF, Baker CI. Circular analysis in systems neuroscience: the dangers of double dipping. Nature Neuroscience. 2009;12:535–40. doi: 10.1038/nn.2303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lang PJ, Bradley MM, Cuthbert BN. Technical Report A-6. University of Florida: Gainesville; 2005. International Affective Picture System (IAPS): Affective Ratings of Pictures and Instruction Manual. [Google Scholar]
- MacDonald E, Dadds MR, Brennan JL, Williams K, Levy F, Cauchi AJ. A review of safety, side-effects and subjective reactions to intranasal oxytocin in human research. Psychoneuroendocrinology. 2011;36:1114–26. doi: 10.1016/j.psyneuen.2011.02.015. [DOI] [PubMed] [Google Scholar]
- Meyer-Lindenberg A, Domes G, Kirsch P, Heinrichs M. Oxytocin and vasopressin in the human brain: social neuropeptides for translational medicine. Nature. 2011;12:524–35. doi: 10.1038/nrn3044. [DOI] [PubMed] [Google Scholar]
- Mezzacappa ES, Katkin ES. Breastfeeding is associated with reduced perceived stress and negative mood in mothers. Health Psychology. 2002;21:187–93. [PubMed] [Google Scholar]
- Mortimer AM. The neuroscience of maternal behavior. Current Psychiatry Reviews. 2008;3:129–35. [Google Scholar]
- Poldrack RA. Region of interest analysis for fMRI. Social Cognitive and Affective Neuroscience. 2007;2:67–70. doi: 10.1093/scan/nsm006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saxe R, Brett M, Kanwisher N. Divide and conquer: a defense of functional localizers. Neuroimage. 2006;30:1088–96. doi: 10.1016/j.neuroimage.2005.12.062. [DOI] [PubMed] [Google Scholar]
- Strathearn L, Fonagy P, Amico J, Montague PR. Adult attachment predicts maternal brain and oxytocin response to infant cues. Neuropsychopharmacology. 2009;34:2655–66. doi: 10.1038/npp.2009.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tu MT, Lupien SJ, Walker CD. Multiparity reveals the blunting effect of breastfeeding on psychological reactivity to psychological stress. Journal of Neuroendocrinology. 2006;18:494–503. doi: 10.1111/j.1365-2826.2006.01441.x. [DOI] [PubMed] [Google Scholar]
- Ziegler TE, Scheffler G, Snowdon CT. The relationship of cortisol levels to social environment and reproductive functioning in female cotton-top tamarins, Saguinus oedipus. Hormones and Behavior. 1995;29:407–24. doi: 10.1006/hbeh.1995.1028. [DOI] [PubMed] [Google Scholar]