Abstract
OBJECTIVE:
The influence of dose of low power lasertherapy (AsGaAl, 830 nm) on the regeneration of the fibular nerve of rats after a crush injury was evaluated by means of the functional gait analysis and histomorphometric parameters.
METHODS:
Controlled crush injury of the right common fibular nerve, immediately followed by increasing doses (G1: no irradiation; G2: simulated; G3: 5 J/cm2; G4: 10 J/cm2; G5: 20 J/cm2) laser irradiation directly on the lesion site for 21 consecutive days. Functional gait analysis was carried out at weekly intervals by measuring the peroneal/fibular functional index (PFI). The animals were killed on the 21st postoperative day for removal of the fibular nerve, which was prepared for the histomorphometric analysis.
RESULTS:
The PFI progressively increased during the observation period in all groups, without significant differences between them (p>0.05). The transverse nerve area was significantly wider in group 2 than in groups 3 and 4, while fiber density was significantly greater in group 4 than in all remaining groups.
CONCLUSION:
The low power AsGaAl laser irradiation did not accelerate nerve recovery with any of the doses used. Level of Evidence I, Therapeutic Studies Investigating the Results of Treatment.
Keywords: Nerve regeneration, Crush syndrome, Peroneal nerve, Laser therapy
INTRODUCTION
Peripheral nerve injury recovery has been studied under experimental conditions, mainly through electrophysiological, histological, morphometric and functional gait assessment techniques. However, the correlation between morphometry, functional gait assessment and adequate laser dosage is not yet known.
On the other hand, it is widely known nowadays that physical agents, such as electricity, magnetic fields, therapeutic ultrasound and laser, can positively influence the result of the healing and regeneration process of different tissues, such as the skin, bone, muscles and tendons and the peripheral nerves. 1 - 3 Among the physical resources, laser is perhaps the least well known, as regards its effects on the biological tissues and on the usage parameters, such as dose, wavelength, continuous 2 - 4 or pulsed mode, 1 , 5 treatment duration 6 and application site.
The use of low-power 1 (wavelength of 904 nm, and dose of 4 J/cm2) pulsed laser irradiation (Gallium Arsenide, GaAs) on the sciatic nerve submitted to crush injury, in rats, led to the conclusion that low-power laser effectively accelerates the regeneration of the injured nerves, having the potential for clinical application in humans. Similar results were obtained 7 in investigations such as the use of irradiation with low-intensity GaAsAl laser (wavelength 830 nm, dose of 20 J/cm2, and continuous mode) on a model of injury to the right common fibular nerve of the rat, alternatively irradiating the injury site, the region of emergence of the nerve roots that form the sciatic trunk in the spinal column or both. They evaluated the results using functional gait analysis and demonstrated that laser irradiation, both in the region of the injury and in the spinal column, already accelerated regeneration on the 14th postoperative day, in comparison with non-treatment.
Conflicting results were found in relation to the use of therapeutic laser in a low dose and pulsed mode. 1 , 5 , 8 , 9
The dose of low-power laser irradiation to stimulate nerve regeneration is a highly variable parameter in the various investigations consulted. Hence dose ranges from 28 J/cm2, in longer daily applications (several minutes), up to 140 J/cm2, in shorter daily applications (a few seconds), and there is apparently no consensus regarding which is best. 4 , 5 , 7 , 9 , 10 Therefore, this parameter is still open, and needs to be clarified before we can use laser irradiation in clinical practice.
Functional gait analysis is currently used in several experiments to measure the rate of functional recovery and has presented satisfactory results with regard to its efficiency and reproducibility. 1 - 3 , 7 , 11
Functional assessment has become one of the methods for studying peripheral nerve regeneration in animals, since the description of the sciatic functional index 12 , 13 that was later modified. 14
This method has the advantage of not being invasive, besides showing close correlation with the degree of morphologic regeneration, measured by morphometry, and can be employed as a replacement for invasive methods. 15
The animals' rear footprints are obtained for its execution, with the measurement of some parameters that are introduced in a mathematical formula, whose result is the sciatic functional index (SFI), which is a negative indicator of nerve function, ranging from zero (0) to -100, with zero meaning absence of dysfunction and -100, complete dysfunction. The aim of this study was to find the most appropriate dose to obtain any beneficial effects of laser on the peripheral nerve tissue.
METHODS
This project was approved by the Committee of Ethics in Animal Experimentation (CETEA) of Faculdade de Medicina de Ribeirão Preto - USP, on February 11, 2008, under protocol no. 158/2007.
The study subjects were 50 Wistar (Rattus Norvegicus Albinus) rats, with body mass varying between 250 and 300g, provided by the Central Vivarium of the City Council of the Ribeirão Preto Campus, of Universidade de São Paulo. These animals were kept in groups of three in collective containment cages, at ambient temperature, in the Vivarium of the Bioengineering Laboratory of FMRP/USP, with unrestricted access to water and standard feed.
At the beginning of the experiment the animals were weighed, identified according to the type of preestablished experiment, and distributed in groups formed of 10 animals each, as follows: group 1: without treatment (n=10); group 2: sham, (n=10); group 3: crush injury, effective irradiation with intensity of 5 J/cm2 (N=10); group 4: crush injury, effective irradiation with intensity of 10 J/cm2; group 5: crush injury, effective irradiation with intensity of 20 J/cm2.
Surgery
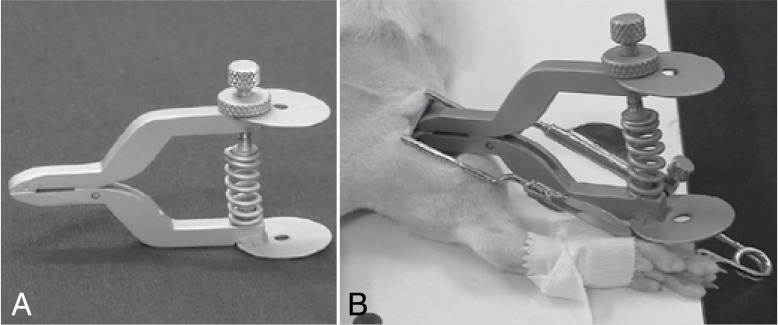
After weighing, the animals underwent general anesthesia, administered intraperitoneally, by means of a mixture of ketamine (1 ml/kg) and xylazine (1.5 ml/kg) followed by care procedures such as trichotomy and antisepsis at the surgical site of the right lower limb for us to approach the common fibular nerve and to perform the crushing technique. The animals were positioned in ventral decubitus, with their rear and fore paws fixed in abduction, and the incision was made in the lateral region of the thigh, from the greater trochanter up to the lateral condyle of the femur. The musculature was separated by divulsion until it was possible to visualize the sciatic nerve and its three branches: common fibular, tibial and sural. Fibular compression covered a length of 5 mm, with weight of 5Kgf and crushing time standardized at 10 minutes. After crushing the nerve was placed in its bed, and the surgeon did not suture the muscles, but only the skin, using 3-0 nylon thread and finalizing with hygiene and antisepsis care at the surgical site. The crushing of the fibular nerve was performing using locking forceps specially designed for this purpose. (Figures 1 A and B) These forceps produce a fixed static lesion of 5,000g, and were calibrated in advance in the Universal Testing Machine (brand EMIC®, model DL 10000).
Figure 1. (A) Crushing forceps; (B) Crushing of fibular nerve.
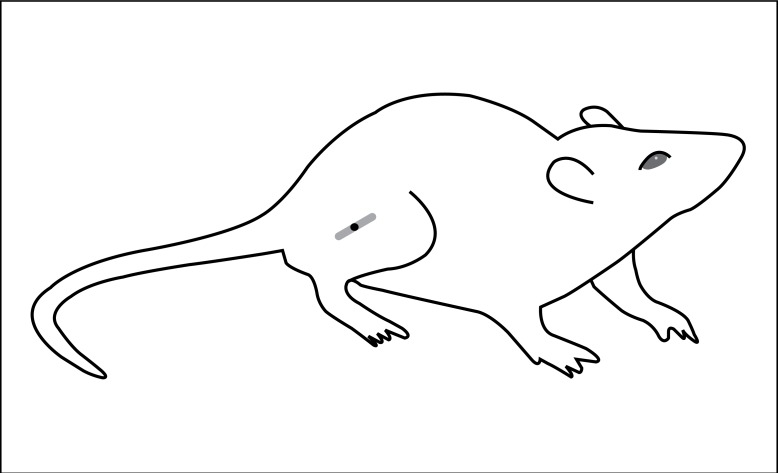
Laser irradiation was performed daily, directly on the fibular nerve lesion, (Figure 2) and the treatment protocol was started on the first postoperative day. The equipment employed was of current use in clinical practice, (Physiolux Dual, BIOSET®) with the following parameters: continuous power of 30 mW; beam area of 0.02 cm2; dosimetry of 5, 10 and 20 J/cm2 (according to the group of animals); and wavelength of 828.43 nm.
Figure 2. Diagram showing the effective and simulated irradiation site.
The laser was of low-intensity gallium arsenide and aluminum (GaAsAl), employing the direct punctual transcutaneous method, applied once a day, for 21 consecutive days.
Functional gait assessment
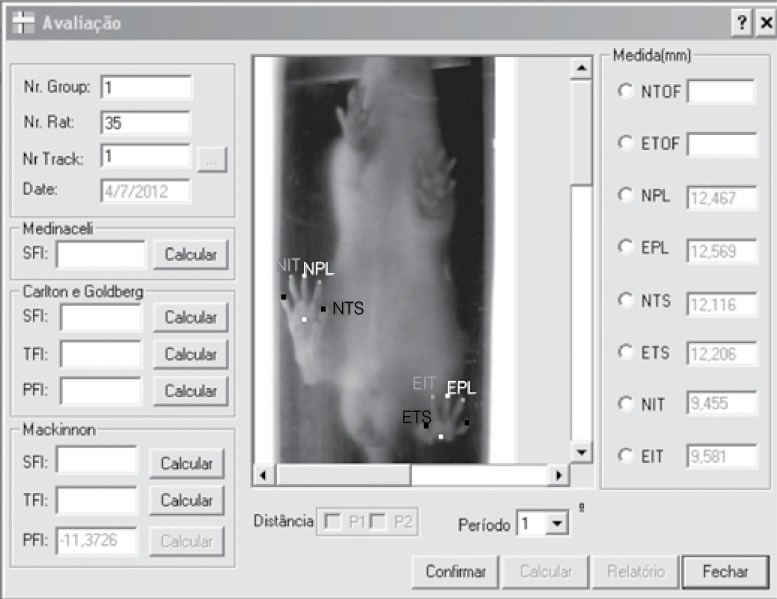
The footprints were analyzed for the functional gait assessment, 16 recorded by means of a webcam with capacity of 1.3 megapixels, fastened to an acrylic treadmill with controlled speed, made of transparent polycarbonate sheeting, with the following dimensions: corridor 850 mm in length, 1090 mm in height and 400 mm in width and a metal hutch at the end of the course (Insight®), coupled to a portable computer by means of a USB cable. The captured images were analyzed using the Functional Analysis of Peripheral Nerves - FAPN program (Figure 3), which calculated the predetermined parameters for the functional gait assessment.
Figure 3. AFNP (Functional Evaluation of Peripheral Nerves) program evaluation screen.
Before the experiment the animals were made to walk on the acrylic bridge up to the metal hutch, with speed standardized at 4 m/min, in order to adapt. The footprints were obtained in the preoperative period, on the 7th and 14th days after the initial injury. The following parameters were measured in the footprints: E: Experimental; N: Normal; PL: print length; TS: total toe spread; IT: intermediate toe spread. The same data were entered in the formula of Bain et al. 14 which provided the peroneal functional index (PFI):
 |
Morphometric Analysis
The morphometric analysis process, from preparation of the material to the analysis of results, was carried out at the Laboratory of Applied and Experimental Neurology of FMRP.
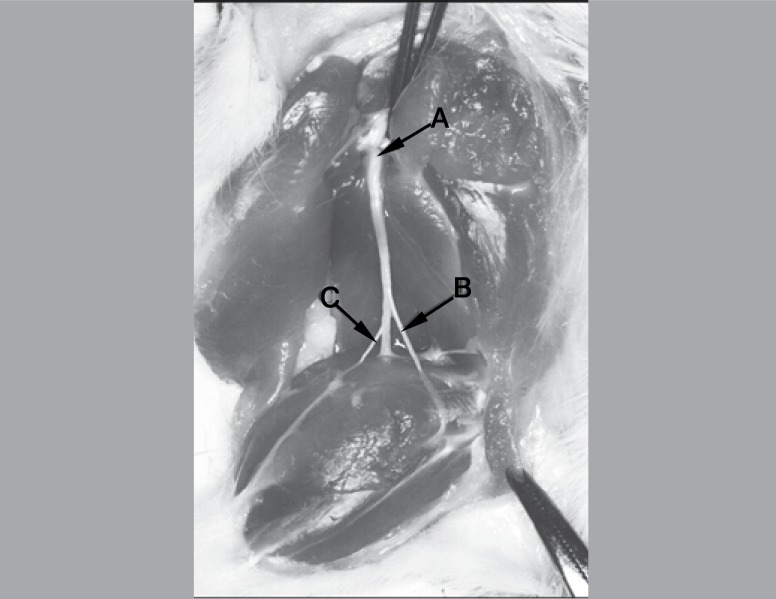
The animals were euthanized with a massive dose of pentobarbital (40 mg/kg) on the 21st postoperative day, according to the standards of AVMA (2007) for rats. The fibular nerve, smaller branch of the trunk and study subject, (Figure 4) was sectioned up to the most distal point possible, when it penetrates in the leg musculature, which consists of a segment of about 5 mm below the crush injury segment.
Figure 4. Trunk of the sciatic nerve and its 3 branches. (A) sciatic nerve; (b) fibular and (c) tibial.
The morphometric analysis was conducted using sections of the distal portion of the nerve in the animals from the experimental groups. These were analyzed under a light microscope (Zeiss Axiophoto II) equipped with a video camera (JVC-TK 1270) connected to a microcomputer using the KS 400 Measure Interactive program (v. 2.0).
After this the participants captured the images and measured the nerve area, also counting the total number of fibers to calculate the density of fibers (fibers/mm2). Other parameters, such as the density of blood vessels, maximum and minimum diameters of the fibers, myelin sheath area and g-ratio (quotient between the axonal diameter and the diameter of the nerve fiber) were calculated for each fiber diameter interval. After the data were obtained, these were analyzed by the SigmaPlot® application for graphic presentation. The morphometric analysis of the regenerated nerve fibers was carried out so that the analysis program randomly selected 30% of the images of each section. The mean of the values found in each group was used in the statistical analysis.
Statistical analysis
The data obtained from the gait analysis and from the morphometric parameters were submitted to the Shapiro-Wilk and Levene tests for verification, respectively, of the distribution of data and homogeneity of variances.
In the comparison between the different groups studied (independent samples), we conducted a variance analysis (ANOVA) followed by the Bonferroni test for parametric data or the Kruskal-Wallis test followed by the Dunn test for nonparametric data.
The comparison between evaluation periods (dependent samples) was carried out by means of the repeated measures analysis of variance (ANOVA).
Moreover, the two-way analysis of variance (Two-way ANOVA) was conducted to verify the evolution of the different groups throughout the evaluation period.
The data from the gait and morphometry analyses were also analyzed by means of the Pearson or Spearman correlation tests.
All the statistical tests were carried out with the help of Statistica 8.0 software (Statsoft, Tulsa, OK, 2008) and results that presented a significance level (p) below 0.05 were considered significant.
RESULTS
The study was conducted with a total of 150 footprint images in the preoperative period and on the 7th and 14th postoperative days. They were evaluated by the PFI formula 14 and the injury produced was crushing of the fibular nerve.
There was no significant difference (p>0.05) in the gait analysis values (PFI) between the groups without irradiation (G1), and the sham (G2), 5 J/cm2 (G3), 10 J/cm2 (G4) and 20 J/cm2 (G4) groups, in any of the periods evaluated. (Table 1)
Table 1. PFI comparisons in the preoperative period (pre-op), in week 1 (W1) and week 2 (W2).
| Group | Gait analysis | p-value (time) | Evolution | p-value (groups x time) | ||
|---|---|---|---|---|---|---|
| Pre-op | W1 | W2 | ||||
| No irradiation | -37.9 ± 14.0 | -39.2 ± 7.3 | -22.1 ± 26.8 | 0.091 | -10.8% ± 92.9% | 0.139 |
| Sham | -37.1 ± 17.3 | -32.5 ± 19.3 | -25.0 ± 13.6 | 0.238 | 9.2% ± 81.4% | |
| 5 J/cm2 | -36.2 ± 17.0 | -33.9 ± 31.0 | -37.1 ± 14.8 | 0.956 | -21.1% ± 60.7% | |
| 10 J/cm2 | -29.5 ± 11.0 | -40.8 ± 31.7 | -41.5 ± 21.2 | 0.381 | -68.7% ± 115.5% | |
| 20 J/cm2 | -22.0 ± 36.3 | -41.7 ± 13.6 | -39.6 ± 5.4 | 0.073 | -17.6% ± 52.7% | |
| p-value (groups) | 0.399 | 0.851 | 0.058 | 0.334 | ||
To compare the evolution of gait analysis over time between the different groups studied we calculated the variation in percentage between the end of the experiment (S2) and the evaluation carried out in the preoperative period.
Monitoring the absence of significant results between the groups as well as between the evaluation periods the comparison of the evolution of the gait analysis did not show significant variations between the groups either.
As regards the morphometric parameters evaluated (Table 2) the nerve area showed significant differences between the groups studied. The animals that received simulated irradiation (placebo) presented a larger area than those that received laser irradiation of 5J/cm2 and 10 J/cm2. Among the other groups evaluated there was no significant difference in relation to the fibular nerve area.
Table 2. Area, total density of fibers and vessel area.
| Group | Morphometric parameters | ||
|---|---|---|---|
| Area (µm2) | Total density | Vessel area | |
| No irradiation | 168174.1 ± 36896.6 ab | 15127.4 ± 2273.0 b | 1829.3 ± 1626.2 |
| sham | 288416.8 ± 228442.5 a | 11101.7 ± 3402.0 b | 4121.4 ± 4938.4 |
| 5 J/cm2 | 144951.0 ± 28170.1 b | 12678.6 ± 3015.1 b | 3535.5 ± 2368.5 |
| 10 J/cm2 | 131144.3 ± 21115.3 b | 26698.2 ± 15286.7 a | 3304.8 ± 1893.8 |
| 20 J/cm2 | 152786.8 ± 45383.2 ab | 12748.1 ± 4061.7 b | 2859.7 ± 1499.4 |
| p-value (groups)1 | 0.023 | < 0.001 | 0.468 |
As regards total density, the group that received 10J/cm2 of laser irradiation presented significantly higher values than all the other groups. No significant difference was observed in the vessel area.
The p-value (groups) represents the level of significance obtained after performance of the Kruskal-Wallis test or ANOVA in the comparison between the different treatments (Injury, Injury + placebo, Injury + 5 J/cm2, Injury + 10 J/cm2, Injury + 20 J/cm2) for each one of the morphometric parameters (Area, Total density and Vessel area). In the variables with p<0.05, equal letters indicate groups in which no significant differences were observed after the Dunn or Bonferroni multiple comparison test. 1
The correlation of the morphometric parameters and the gait analysis did not show any significant statistical difference. (Table 3)
Table 3. Correlation between morphometric parameters and PFI.
| Gait analysis | Morphometric parameters | ||
|---|---|---|---|
| Area (µm2) | Total density | Vessel area | |
| Pre-Op | -0.147 | 0.015 | -0.014 |
| p = 0.335 | p = 0.924 | p = 0.927 | |
| W1 | -0.002 | -0.102 | -0.023 |
| p = 0.992 | p = 0.505 | p = 0.880 | |
| W2 | 0.262 | -0.133 | -0.122 |
| p = 0.082 | p = 0.384 | p = 0.425 | |
N.B.: The values represent Pearson's or Spearman's correlation coefficient. Significant correlations (p<0.05) can be interpreted according to the correlation intensity by the value of the coefficient ®: |r|≤0.5 - weak correlation; 0.5<|r|≤0.8 - moderate correlation; r>0.8 - strong correlation.
DISCUSSION
Several resources have been used to accelerate or better obtain nerve regeneration. These can be used together with surgical repair techniques or separately, with special emphasis on therapeutic laser with diode diversification. The rat was chosen as the experimentation animal since the morphological structures of its peripheral nerves closely resemble those of the nerves of humans. Moreover, the natural nerve regeneration of these animals is very fast and can achieve normal levels. 17
The surgical procedure, to injure the fibular nerve using the forceps, is easy to learn without the need for microsurgical equipment, and lasted 20 minutes on average for each animal. The crushing injury was produced in all the animals by fixed load crush forceps (5 kgf) to make the lesions homogenous. 16 This method does not provoke nerve stretching, as the nerve does not need to be exposed, but is crushed in its own bed, thus diminishing another variable, that of nerve stretching.
Axon regeneration starts a few hours after the injury. Two to three sprouts project from each axon and will continue up to the target organ, mediated by trophic factors secreted by the Schwann cells, a phenomenon known in the literature. Accordingly, in this study, the irradiation was performed on the 1st PO day with the intention of assisting this mechanism, even though it is unknown. We used GaAsAl laser with a wavelength of 830 nm, continuous mode, with doses of 5 J/cm2, 10 J/cm2 and 20 J/cm2, as an isolated therapeutic resource, in order to promote the desirable regeneration.
There were no problems during laser application, since the rat was immobilized while the irradiation was executed according to the experimental group, and the maximum application time was 40 seconds. Low-intensity laser irradiation has been investigated by several authors who report its benefits in nerve tissue regeneration. Laser treatment promotes significant functional recovery, 10 increases the quantity of connective tissue (collagen), increases the number of fibroblasts at the site of the injury and that of myelinized axons, accelerating nerve conduction, 4 accelerates healing, induces faster recovery from muscular trophism 10 and acts both in regeneration and in the prevention of nerve fiber degeneration.
However, in other studies 5 , 8 no positive effects were observed after use of low-intensity laser on nerve injuries.
In this study, corroborating such findings, in relation to low-intensity laser irradiation, no statistical differences were observed between the groups irradiated with 5 J/cm2, 10 J/cm2 and 20 J/cm2, and there were no differences when these experimental groups were compared with the untreated injury and simulated (or placebo) irradiation injury groups.
Some factors such as the short laser application time, which was seven consecutive days and the pulsed emission of the low-intensity laser could have influenced the absence of any effect of the laser on nerve regeneration. 10
In our study we did not find any improvement in peripheral nerve regeneration through functional gait assessment and morphometric analysis in the groups treated with GaAsAl laser (830nm), perhaps due to the use of low doses, from 5 to 20 J/cm2.
The functional gait assessment is a method widely used in our field, in investigations of the sciatic and fibular nerve of rats, showing a clear correlation between this and the evaluation by morphometry of the nerve. 2 , 9 , 11 , 16 - 18
After the fibular nerve injury the gait patterns are changed so the toe extenders, the dorsoflexors and the paw eversors are denervated, causing the non-opposition of the toes and flexion of the paw that generates shortening of the print length. The distance between the intermediate toes is relatively unaffected due to the normal function of the intrinsic paw parts. Only a slight decrease of toe spread is noticed.
The functional gait assessment in the preoperative period did not reach the value zero, as was expected, or oscillation of around - 10 as found in other studies. 9 , 18
On the 7th and 14th days there was no significant difference between groups, although we have noted that groups G1 and G2, not submitted to laser therapy, presented lower mean PFI values than the other groups.
As regards the analysis of the morphometric parameters evaluated, the nerve area showed significant differences between the studied groups. Group G2, animals that received simulated irradiation (placebo), presented a larger area than those that received laser irradiation of 5 J/cm2 and 10 J/cm2. With regards to total density, the group that received 10 J/cm2 of laser irradiation presented significantly higher values when compared with all the other groups.
It can be inferred that although the use of low-intensity laser is widely used in clinical settings, its values and therapeutic efficacy are still controversial, since the lack of knowledge of the mechanisms increases this lack of standardization. Furthermore, most studies are not conclusive and indicate the need for further research in this area.
CONCLUSION
The use of low-intensity GaAs laser (830nm), in different doses, did not promote functional recovery in the fibular nerve submitted to crushing, while the results obtained by the functional gait analysis (PFI) and histomorphometric studies demonstrated that such a resource was not able to promote beneficial results in nerve regeneration.
Acknowledgments
ACKNOWLEDGMENTS
To the Laboratory of Applied and Experimental Neurology of Faculdade de Medicina de Ribeirão Preto (FMRP) of Universidade de São Paulo, for its availability in the performance of the morphometry of this study. To FAPESP for having provided the research grant.
Footnotes
Acta Ortop Bras. [online]. 2013;21(5): 266-70. Available from URL: http://www.scielo.br/aob.
Work performed at the Bioengineering Laboratory by the Graduate Program of the Department of Biomechanics, Medicine and Rehabilitation of the Musculoskeletal System of Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo, Ribeirão Preto, SP, Brazil.
REFERENCES
- 1.Endo C, Barbieri CH, Mazzer N, Fazan VS. A laserterapia de baixa intensidade acelera a regeneração de nervos periféricos. Acta Ortop Bras. 2008;16(5):305–310. [Google Scholar]
- 2.Marcolino AM, Barbosa RI, Neves LMS, Vinas TS, Duarte DTB, Mazzer N, et al. Laser de baixa intensidade (830 nm) na recuperação funcional do nervo isquiático de ratos. Acta Ortop Bras. 2010;18(4):207–211. [Google Scholar]
- 3.dos Reis FA, Belchior AC, de Carvalho Pde T, da Silva BA, Pereira DM, Silva IS, et al. Effect of laser therapy (660 nm) on recovery of the sciatic nerve in rats after injury through neurotmesis followed by epineural anastomosis. Lasers Med Sci. 2009;24(5):741–747. doi: 10.1007/s10103-008-0634-3. [DOI] [PubMed] [Google Scholar]
- 4.Rochkind S, Rousso M, Nissan M, Villarreal M, Barr-Nea L, Rees DG. Systemic effects of low-power laser irradiation on the peripheral and central nervous system, cutaneous wounds, and burns. Lasers Surg Med. 1989;9(2):174–182. doi: 10.1002/lsm.1900090214. [DOI] [PubMed] [Google Scholar]
- 5.Bagis S, Comelekoglu U, Coskun B, Milcan A, Buyukakilli B, Sahin G, et al. No effect of GA-AS (904 nm) laser irradiation on the intact skin of the injured rat sciatic nerve. Lasers Med Sci. 2003;18(2):83–88. doi: 10.1007/s10103-003-0258-6. [DOI] [PubMed] [Google Scholar]
- 6.Anders JJ, Geuna S, Rochkind S. Phototherapy promotes regeneration and functional recovery of injured peripheral nerve. Neurol Res. 2004; 26(2):233–239. doi: 10.1179/016164104225013914. [DOI] [PubMed] [Google Scholar]
- 7.Sousa FFA, Andraus RAC, Barbieri CH, Mazzer N. Influência da irradiação do laser na regeneração nervosa. Acta Ortop Bras. 2009; 17(6):331–335. [Google Scholar]
- 8.Wu WH, Ponnudurai R, Katz J, Pott CB, Chilcoat R, Uncini A, et al. Failure to confirm report of light-evoked response of peripheral nerve to low power helium-neon laser light stimulus. Brain Res. 1987;401(2):407–408. doi: 10.1016/0006-8993(87)91430-2. [DOI] [PubMed] [Google Scholar]
- 9.Cömelekoğlu U, Bagiş S, Büyükakilli B, Sahin G, Erdoğan C, Kanik A. Acute electrophysiological effect of pulsed gallium-arsenide low-energy laser irradiation on isolated frog sciatic nerve. Lasers Med Sci. 2002;17(1):62–67. doi: 10.1007/s10103-002-8268-3. [DOI] [PubMed] [Google Scholar]
- 10.Gigo-Benato D, Geuna S, de Castro Rodrigues A, Tos P, Fornaro M, Boux E, et al. Low-power laser biostimulation enhances nerve repair after end-to-side neurorrhaphy: a double-blind randomized study in the rat median nerve model. Lasers Med Sci. 2004;19(1):57–65. doi: 10.1007/s10103-004-0300-3. [DOI] [PubMed] [Google Scholar]
- 11.Monte-Raso VV, Barbieri CH, Mazzer N, Yamasita AC, Barbieri G. Is the Sciatic Functional Index always reliable and reproducible? J Neurosci Methods. 2008;170(2):255–261. doi: 10.1016/j.jneumeth.2008.01.022. [DOI] [PubMed] [Google Scholar]
- 12.de Medinaceli L, Freed WJ, Wyatt RJ. An index of the functional condition of rat sciatic nerve based on measurements made from walking tracks. Exp Neurol. 1982;77(3):634–643. doi: 10.1016/0014-4886(82)90234-5. [DOI] [PubMed] [Google Scholar]
- 13.de Medinaceli L, DeRenzo E, Wyatt RJ. Rat sciatic functional index data management system with digitized input. Comput Biomed Res. 1984;17(2):185–192. doi: 10.1016/0010-4809(84)90031-4. [DOI] [PubMed] [Google Scholar]
- 14.Bain JR, Mackinnon SE, Hunter DA. Functional evaluation of complete sciatic, peroneal, and posterior tibial nerve lesions in the rat. Plast Reconstr Surg. 1989;83(1):129–138. doi: 10.1097/00006534-198901000-00024. [DOI] [PubMed] [Google Scholar]
- 15.Oliveira EF, Mazzer N, Barbieri CH, Selli M. Correlation between functional index and morphometry to evaluate recovery of the rat sciatic nerve following crush injury: experimental study. J Reconstr Microsurg. 2001;17(1):69–75. doi: 10.1055/s-2001-12691. [DOI] [PubMed] [Google Scholar]
- 16.Monte-Raso VV, Barbieri G, Mazzer N, Fonseca Mde C, Barbieri CH. A new treadmill-type motorized walking belt machine for video recording of the rat's gait and sciatic functional index measurement. A comparative study with other methods. J Neurosci Methods. 2010;189(1):23–29. doi: 10.1016/j.jneumeth.2010.03.005. [DOI] [PubMed] [Google Scholar]
- 17.Mazzer PYCN, Barbieri CH, Mazzer N, Fazan VS. Avaliação qualitativa e quantitativa das lesões agudas por esmagamento do nervo isquático do rato. Acta Ortop Bras. 2006;14(4):220–225. [Google Scholar]
- 18.Mendonça AC, Barbieri CH, Mazzer N. Directly applied low intensity direct electric current enhances peripheral nerve regeneration in rats. J Neurosci Methods. 2003;129(2):183–190. doi: 10.1016/s0165-0270(03)00207-3. [DOI] [PubMed] [Google Scholar]