Abstract
The ability to achieve precisely tailored activation and inactivation of gene expression represents a critical utility for vertebrate model organisms. In this regard, Cre and other site-specific DNA recombinases have come to play a central role in achieving temporally regulated and cell type-specific genetic manipulation. In zebrafish, both Cre and Flp recombinases have been applied for inducible activation, inactivation and inversion of inserted genomic elements. Here we describe the addition of Dre, a heterospecific Cre-related site-specific recombinase, to the zebrafish genomic toolbox. Combining Dre-based recombination in zebrafish with established Cre/lox technology, we have established an effective strategy for transgene activation and inactivation using lox and rox (TAILOR). Using stable transgenic lines expressing tamoxifen-inducible CreERT2 and RU486-inducible DrePR fusions, we demonstrate that Cre and Dre retain non-overlapping specificities for their respective lox and rox target sites in larval zebrafish, and that their combinatorial and sequential activation can achieve precisely timed transgene activation and inactivation. In addition to TAILOR, the successful application of Dre/rox technology in zebrafish will facilitate a variety of additional downstream genetic applications, including sequential lineage labeling, complex genomic rearrangements and the precise temporal and spatial control of gene expression through the intersection of partially overlapping promoter activities.
Introduction
Site-specific DNA recombinases have become critical components of genome manipulation strategies in vertebrates. In zebrafish, both the Cre/lox and Flp/frt systems have been utilized for a variety of applications, including transgene activation, transgene excision, and transgene inversion [1]–[16]. Recently, an additional Cre-like site-specific recombinase known as Dre was identified by sequencing homologous genomic regions of D6, a transducing bacteriophage related to the P1 phage from which Cre was isolated [17]. Like Cre, Dre is a site-specific tyrosine recombinase capable of catalyzing both excision and integration events, with no requirement for other phage-encoded or bacterial proteins. While Cre displays specificity for lox target sites, Dre recognizes a different target sequence known as rox. While lox and rox sites are similar in size (34 bp vs. 32 bp), sequence (21 nucleotides in common) and structure (inverted repeats flanking an asymmetric spacer), Cre has no ability to recombine rox sites and Dre similarly has no activity at lox sites, either in bacteria or in mammalian cells [18], [19].
Based on the recent successful application of the Dre/rox system in mice [18], we sought to determine its activity in zebrafish embryos, with an emphasis on developing new techniques for sequential activation and inactivation of inserted transgenes.
Materials and Methods
Transgenesis
All experiments involving zebrafish were approved by the Johns Hopkins University Institutional Animal Care and Use Committee. Fish were raised and maintained under standard laboratory conditions. The following strains were established and/or utilized: Tg(ubb:Dre;cryaa:Venus) (herein ubb-Dre), Tg(ubb:Cre;cryaa:Venus) (herein ubb-Cre), Tg(ubb:lox-Nuc-mCherry-stop-lox-eGFP) (herein Lox-Nuc-mCherry-Lox-eGFP), Tg(ubb:rox-Nuc-mCherry-stop-rox-eGFP) (herein Rox-Nuc-mCherry-Rox-eGFP), Tg(ubb:DrePR;cryaa:eCFP) (herein ubb-DrePR), Tg(ubb:CreERT2;cmlc2:eGFP) (herein CreERT2) [20], Tg(ubb:lox-stop-lox-rox-nuc-mCherry-stop-rox-eGFP;cryaa:mCherry) (herein LSL-Rox-Nuc-mCherry-Rox-eGFP). Dre and DrePR cDNAs were kindly provided by Dr. Francis Stewart. All new transgenic lines were generated using the backbone of T2KXIGΔIN [21], as previously described [22]. Larvae were anaesthetized in 0.16% tricaine (3-aminobenzoic acid ethylester, A-5040, Sigma, pH 7.0). Adult zebrafish were euthanized by induction of tricaine anesthesia followed by placement in an ice bath, consistent with recommendations of the Panel on Euthanasia of the American Veterinary Association.
4-OHT Treatment for CreERT2 Induction and RU486 Treatment for DrePR Induction
4-Hydroxytamoxifen (4-OHT, H7904; Sigma, St Louis, MO, USA) and RU486 (H110-01; Invitrogen) were dissolved in ethanol at a final stock concentration of 10 mM and kept in single-use aliquots in the dark at –20°C. To induce Cre activity in CreERT2-expressing embryos, 25–30 stage-matched embryos were incubated with E3 medium freshly mixed with 5 µM 4-OHT. To induce Dre activity in DrePR-expressing embryos, 25–30 stage-matched embryos were incubated with E3 medium freshly mixed with 4 µM RU486. The treated embryos were immediately put into a closed and dark 28.5°C incubator for 24 hrs. The embryos were subsequently placed in a fresh E3 medium and grown as described previously.
Tissue Dissection, Confocal Microscopy, and Cell Counting
Tissue dissection and confocal microscopy were performed as described previously [22]. To quantify the efficiency of DrePR recombination in a RU486-dose dependent manner, 25–30 stage-matched embryos (n = 5) were treated with E3 medium freshly mixed with 0 µM, 1 µM, 2 µM, 4 µM, and 10 µM of RU486 between 24 and 48 hpf. Two days later, 4 dpf the larval zebrafish were fixed overnight in 4% PFA and the intestine was dissected and mounted in the mounting media (DAKO, S3023). To quantify the efficiency of sequential CreERT2- and DrePR-mediated recombination, larval zebrafish (n = 5) were treated sequentially with 4-OHT at 24 hpf and RU486 at 48 hpf, and fixed overnight in 4% PFA at 7 dpf. Following dissection, sections of intestine, liver, and pancreas were prepared. The total number of DAPI-labeled cells also labeled by either mCherry or eGFP was counted, with a minimum of 400 cells counted for each tissue.
Results
Dre Effectively Recombines Rox Sites but not Lox Sites in Larval Zebrafish
As an initial evaluation of Dre-based recombination in zebrafish, we utilized standard Tol2-mediated transgenesis to establish Dre-driver and a Dre-responder lines in which relevant elements are expressed under the control of zebrafish ubiquitin b (ubb) promoter (Fig. 1A). The ubb promoter drives transgene expression ubiquitously in the vast majority of cell types through all stages of zebrafish development, beginning at the mid-blastula transition [20]. The Dre driver transgene (ubb:Dre;cryaa:Venus, subsequently referred to as ubb-Dre) also incorporated a crystalline aa:Venus cassette [23], with resulting Venus fluorescence in the eye facilitating the identification of transgene-expressing embryos. To establish stable transgenic lines, we selected two independent male F1 founders whose F2 progeny displayed a ∼50% incidence of Venus eye fluorescence, suggesting a single ubb-Dre transgene integration. The Dre responder line contained a Nuc-mCherry-stop cassette flanked by 32 bp rox sites and followed by eGFP (ubb-rox-Nuc-mCherry-stop-rox-eGFP, subsequently referred to as Rox-Nuc-mCherry-Rox-eGFP). Stable Dre responder lines were selected based on ubiquitous expression of ubb promoter-driven nuclear mCherry, evident shortly following the mid-blastula transition.
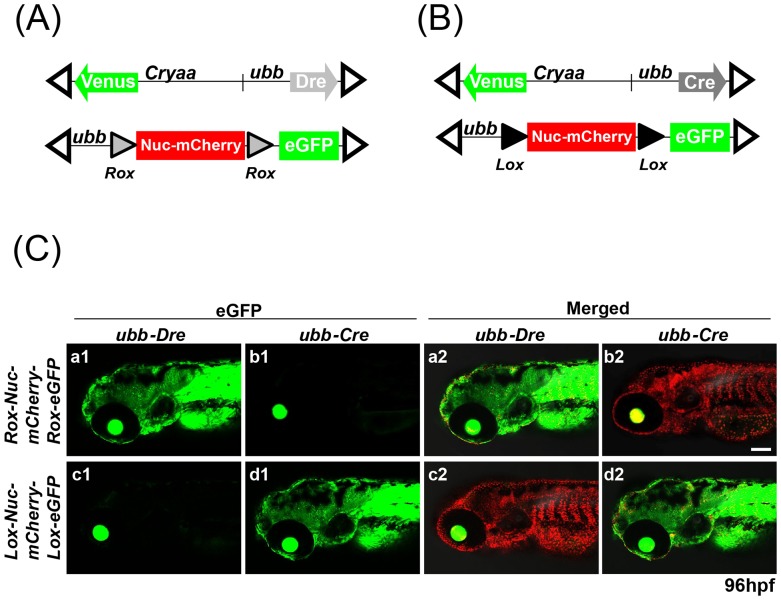
Figure 1. Heterospecific recombination of rox and lox sites by Dre and Cre in zebrafish embryos.
(A, B) Schematic of ubb-Dre (A) and ubb-Cre (B) driver lines and corresponding Rox-Nuc-mCherrys-Rox and Lox-Nuc-mCherry-Lox reporters. Additional cryaa:Venus cassette facilitates identification of transgene-expressing embryos. Open triangles indicate Tol2 arms. (C) Images from double transgenic embryos produced by indicated crosses of ubb-Dre and ubb-Cre driver lines with either Rox-Nuc-mCherry-Rox-eGFP or Lox-Nuc-mCherry-Lox-eGFP reporter lines. Activation of eGFP confirms Dre-specific recombination of Rox-Nuc-mCherry-Rox reporter and Cre-specific recombination of Lox-Nuc-mCherry-Lox reporter. Scale bar: 200 µm.
To confirm non-overlapping specificities for the Dre/rox and Cre/lox systems in larval zebrafish, we also established Cre-driver and Cre-responder lines under the control of the ubb promoter (Fig. 1B). As in the case of ubb-Dre, the ubb-Cre transgene also contained crystalline aa:Venus cassette. We established three independent male F1 founders carrying a single ubb-Cre transgene integration. A similar Cre responder line was also established, with a Nuc-mCherry-stop cassette flanked by lox sites and followed by eGFP (ubb-lox-Nuc-mCherry-stop-lox-eGFP, subsequently referred to as Lox-Nuc-mCherry-Lox-eGFP) (Fig. 1B).
To initially determine whether Dre was active in larval zebrafish, we crossed male ubb-Dre fish to female Rox-Nuc-mCherry-Rox-eGFP fish. These experiments confirmed efficient Dre-based deletion of the Rox-Nuc-mCherry-Rox cassette, as evidenced by activation of eGFP expression (Fig. 1C a1 and a2). To further confirm that Dre does not recognize related lox sites, we also crossed ubb-Dre males with Lox-Nuc-mCherry-Lox-eGFP females. In contrast to Rox-Nuc-mCherry-Rox-eGFP, no activation of eGFP expression was observed (Fig. 1C c1 and c2). Extending these studies to the evaluation of Cre activity, we similarly crossed ubb-Cre males to both Lox-Nuc-mCherry-Lox-eGFP and Rox-Nuc-mCherry-Rox-eGFP females. As expected, Cre effectively activated the conditional Lox-Nuc-mCherry-Lox-eGFP allele (Fig. 1C d1 and d2), but showed no ability to activate eGFP expression in the Rox-Nuc-mCherry-Rox-eGFP line (Fig. 1C b1 and b2). Thus Cre and Dre behave as heterospecific DNA recombinases when expressed as stable transgenes in larval zebrafish.
Induction of DrePR by RU486
In order to establish a temporally-inducible system for the regulation of Dre activity that would complement already established CreERT2 lines, we adapted an expression construct for DrePR [18], in which Dre is fused to a progesterone receptor (PR) ligand binding domain engineered to be selectively responsive to the synthetic ligand RU486. First, we established a DrePR-driver line under control of the ubb promoter. To facilitate the identification of DrePR-expressing embryos, this transgene (ubb:DrePR;cryaa:eCFP subsequently referred to as ubb-DrePR) also incorporated a crystalline aa:eCFP cassette [23] in the opposite direction of the ubb promoter (Fig. 2A). To establish stable transgenic lines, we selected three independent male F1 founders whose F2 progeny displayed a ∼50% incidence of eCFP eye fluorescence, suggesting a single ubb-DrePR transgene integration. To characterize ubb-driven DrePR function, we crossed a male ubb-DrePR line to a female Rox-Nuc-mCherry-Rox-eGFP line. To induce Dre activity in DrePR-expressing embryos, 25–30 stage-matched embryos were incubated in the absence and presence of 4 µM RU486 for 24 hrs. No eGFP was expressed in DrePR-expressing embryos incubated in the absence of RU486 (Fig. 2B a1 and a4), but strong eGFP activation was observed in RU486-treated embryos (Fig. 2B b1 and b4). To test for any unanticipated cross-reactivity between 4-OHT and RU486, we also treated DrePR-expressing embryos with 5 µM 4-OHT, and observed no induction of eGFP expression (Fig. 2B c1 and c4).
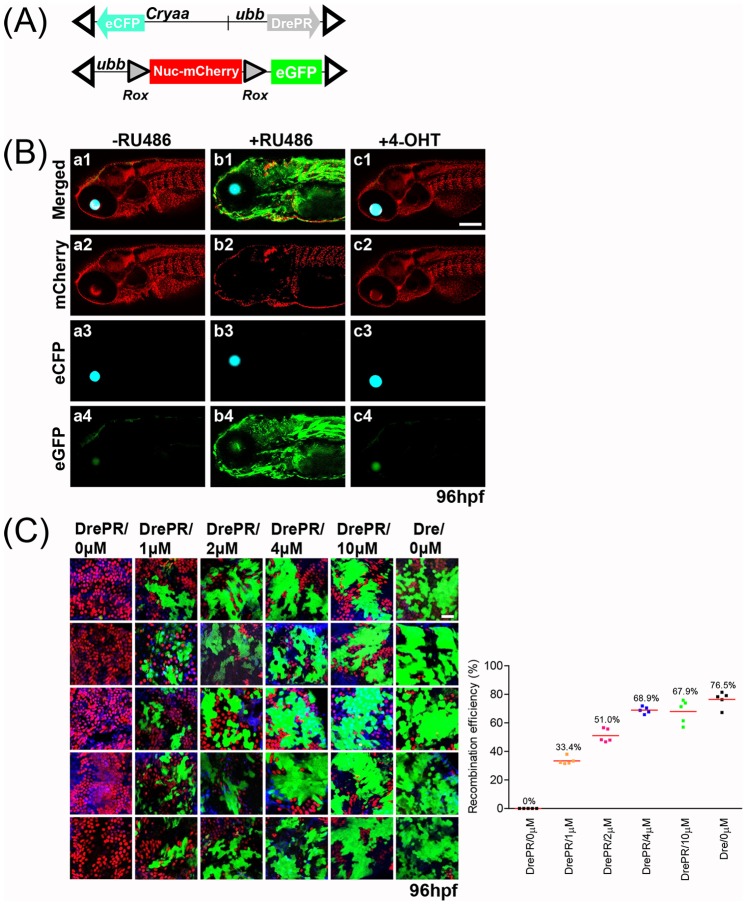
Figure 2. Induction of DrePR recombinase activity by RU486.
(A) Schematic of ubb-DrePR driver line and Rox-Nuc-mCherry-Rox reporter. Additional cryaa:eCFP cassette facilitates identification of transgene-expressing embryos. Open triangles indicate Tol2 arms. (B) Tight control of DrePR recombinase activity by RU486. ubb-DrePR; Rox-Nuc-mCherry-Rox-eGFP embryos were treated with and without 4 µM RU486 between 24 and 48 hpf, and imaged at 96 hpf. No expression of eGFP is observed in untreated (-RU486) or tamoxifen (4-OHT)-treated embryos, while treatment with RU486 results in potent induction of eGFP expression indicating successful recombination of Rox-Nuc-mCherry-Rox-eGFP allele. (C) To quantify the efficiency of Dre recombination in Dre-expressing embryos and DrePR recombination in DrePR-expressing embryos, the intestine of larval zebrafish (4 dpf) were dissected following treatment with RU486 at the indicated concentration between 24–48 hpf. DAPI-labeled cells also labeled by either nucleus mCherry or cytoplasmic eGFP were counted. Maximal recombination frequency is achieved an an RU486 concentration of 4 µM, at a level comparable with Dre lacking the PR fusion. Scale bar: 25 µm.
Prior studies have suggested that CrePR fusions display less effective recombinase activity compared to native Cre [24], [25]. To check whether the DrePR fusion might be associated with a similar reduction in Dre recombinase activity, we crossed both ubb-DrePR and ubb-Dre males to Rox-Nuc-mCherry-Rox-eGFP females. In the case of DrePR, 25–30 stage-matched embryos (n = 5) were treated with E3 medium freshly mixed with RU486-dose dependent manner, 0 µM, 1 µM, 2 µM, 4 µM, and 10 µM. To quantify the efficiency of Dre recombination in Dre-expressing embryos and DrePR recombination in DrePR-expressing embryos, the intestine of larval zebrafish (4 dpf) was dissected and DAPI-labeled cells also labeled by either nucleus mCherry or cytoplasmic eGFP was counted. A dose-response curve revealed that the efficiency of RU486-induced DrePR activation became saturated at between 2 and 4 µM of RU486. No cytoplasmic eGFP-labeled cells were observd in DrePR embryos in the absence of RU486 (Fig. 2C), further confirming the absence of leaky DrePR activity. Furthermore, the maximal recombination activity of DrePR (68.9% at 4 µM of RU486) was similar to that of non-inducible Dre lacking the PR fusion (76.5%), indicating that C-terminal fusion of Dre with the PR does not interfere greatly with recombinase functionality (Fig. 2C). These data confirm tight and efficient temporal control of DrePR activity by RU486.
Combinatorial Activation of DrePR and CreERT2
In order to further evaluate the combinatorial utilities of DrePR and CreERT2, we established an additional responder line incorporating fluorescent reporters of both Dre and Cre recombinase activity. To facilitate the identification of transgene expressing embryos, this dual reporter construct (ubb:lox-Stop-lox-rox-Nuc-mCherry-stop-rox-eGFP;cryaa:mCherry, subsequently referred to as LSL-Rox-Nuc-mCherry-Rox-eGFP) also incorporated a crystalline aa:mCherry cassette [23], placed in an opposite orientation relative to the ubb promoter. To generate a stable, temporally-regulated Cre driver line, we used a 4-OHT inducible CreERT2 element under the control of ubb promoter [20]. This line (ubb:CreERT2;cmlc2:eGFP, subsequently referred to as ubb-CreERT2) also incorporated cmlc2:eGFP cassette to facilitate the identification of transgene-expressing embryos based upon cardiac expression of eGFP. Following the creation of double-transgenic ubb-CreERT2; ubb-DrePR fish, additional crosses were completed to assemble these CreERT2 and DrePR driver alleles together with the LSL-Rox-Nuc-mCherry-Rox-eGFP reporter (Fig. 3A).
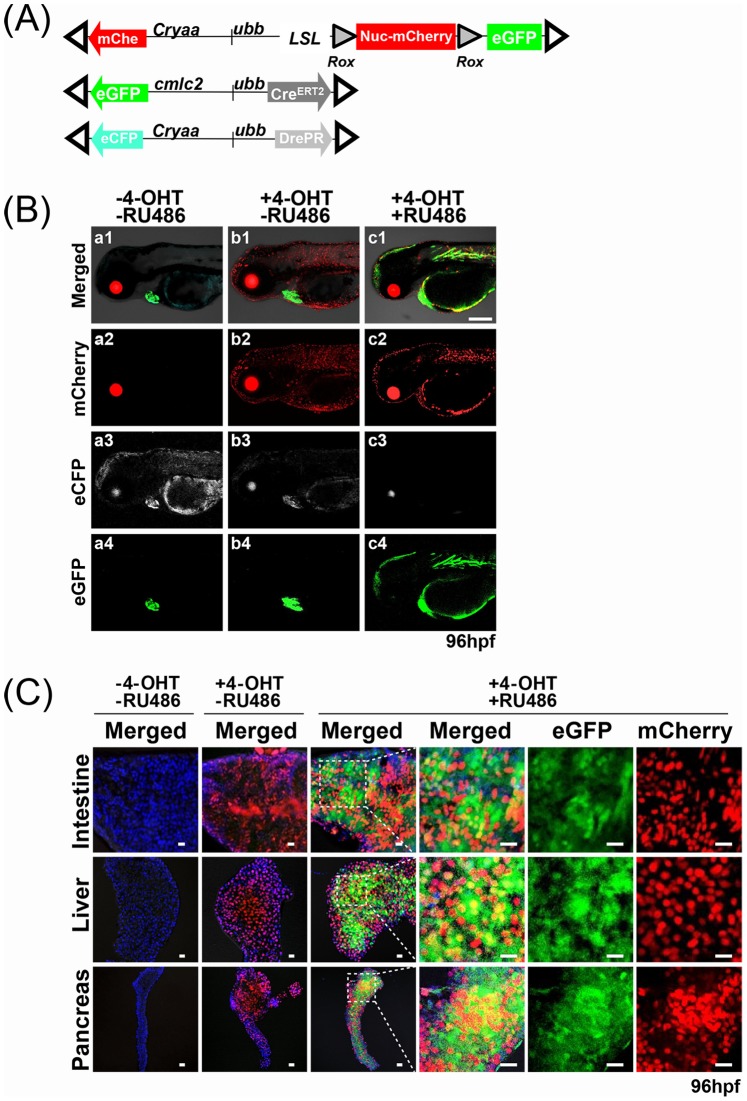
Figure 3. Combinatorial activation of DrePR and CreERT2.
(A) Schematic of ubb:lox-stop-lox-rox-Nuc-mCherry-stop-rox-eGFP dual reporter for assessment of both Cre- and Dre-mediated recombination, along with ubb-CreERT2 and ubb-DrePR driver lines. Ocular and cardiac fluorescence conveyed by additional cryaa:mCherry, cmlc2:eGFP and cryaa:eCFP cassettes facilitates identification of transgene-expressing embryos. Open triangles indicate Tol2 arms. (B) Triple transgenic fish were treated for 24 hrs with or without 4-OHT and RU486 as indicated, and imaged at 96 hpf. While untreated embryos showed no transgene-specific fluorescence besides that provided by the ocular mCherry, ocular eCFP and cardiac eGFP markers (Fig. 3B a1, a2, a3, and a4), embryos treated with only 4-OHT displayed widespread activation of nuc-mCherry, but no activation of eGFP (Fig. 3B b1, b2, b3, b4). In contrast, embryos simultaneously treated with both 4-OHT and RU486 displayed expression of both nuc-mCherry and eGFP (Fig. 3B c1, c2, c3, and c4). Scale bar: 200 µm. (C) Confocal imaging of dissected intestine, liver, and pancreas, confirming patterns of mCherry and eGFP expression observed in whole embryos. Following combined treatment with 4-OHT and RU486, a majority of cells in each tissue express either nuclear mCherry or cytoplasmic eGFP, with a smaller fraction of cells expressing both. Scale bar: 25 µm.
We then tested whether CreERT2 and DrePR could be combinatorially applied for the inducible activation of a tandem “LSL-Rox-Nuc-mCherry-Rox” allele, as would be required if transgene activation was desired only in a highly selective subset of cells expressing both Cre and Dre by virtue of the intersectional expression of two different promoters [26]–[28]. We treated 25–30 stage-matched LSL-Rox-Nuc-mCherry-Rox-eGFP; ubb-CreERT2; ubb-DrePR triple transgenic embryos with either 4-OHT alone or the combination of both 4-OHT and RU486, applied simultaneously. While untreated embryos showed no transgene-specific fluorescence besides that provided by the ocular mCherry, ocular eCFP and cardiac eGFP markers (Fig. 3B a1, a2, a3, and a4), embryos treated with only 4-OHT displayed widespread activation of nuc-mCherry, but no activation of eGFP (Fig. 3B b1, b2, b3, and b4). In contrast, embryos simultaneously treated with both 4-OHT and RU486 displayed expression of both nuc-mCherry and eGFP (Fig. 3B c1, c2, c3, and c4). To examine activation of nuc-mCherry and eGFP at the cellular level, we dissected internal organs (intestine, liver, and pancreas) for confocal imaging. These studies confirmed observations made in whole mount embryos, with a majority of cells in each tissue expressing either the nuclear mCherry or the cytoplasmic eGFP markers, and a smaller fraction of cells expressing both (Fig. 3C). Several mechanisms may underlie the mosaic expression patterns observed in embryos treated with both 4OHT and RU486. While cells expressing eGFP alone have obviously undergone recombination of both lox- and the rox-flanked elements, cells expressing only mCherry have presumably undergone effective 4-OHT induction of CreERT2 but failed to achieve RU486 induction of DrePR activity at threshold levels required for excision of the Rox-Nuc-mCherry-Rox cassette. Cells expressing both mCherry and eGFP may have undergone sequential CreERT2 and DrePR-mediated recombination, such that an interval of stable mCherry protein production precedes DrePR-mediated excision. Alternatively, expression of both mCherry and eGFP may reflect discordant recombination of more than one Tol2-based LSL-Rox-Nuc-mCherry-Rox-eGFP insert, in spite of our attempts to utilize only F1 founders passing a single LSL-Rox-Nuc-mCherry-Rox-eGFP allele.
Sequential Transgene Activation and Inactivation using Lox and Rox (TAILOR)
In addition to simultaneous recombination of lox and rox sites for selected activation of transgenes in cells expressing both CreERT2 and DrePR, many additional applications would be enabled by the staged induction of recombinase activity. Among these would be the sequential activation and inactivation of a specific transgene in a temporally-regulated, cell type-specific manner. We therefore conceived a strategy for transgene activation and inactivation using lox and rox , referred to as TAILOR. Using the same LSL-Rox-Nuc-mCherry-Rox-eGFP; ubb-CreERT2; ubb-DrePR triple transgenic embryos depicted in Fig. 3A, we sought to determine the feasibility and efficiency of tamoxifen-inducible LSL-Rox-Nuc-mCherry-Rox transgene activation, followed by RU486-inducible transgene deletion. For these studies, 24 hpf triple transgenic embryos were treated with 4-OHT for 24 hrs, after which 4-OHT was removed from the embryo medium and replaced with RU486. Individual embryos were serially imaged prior to (48 hpf) and after (96 hpf) treatment with RU486 (Fig. 4A).
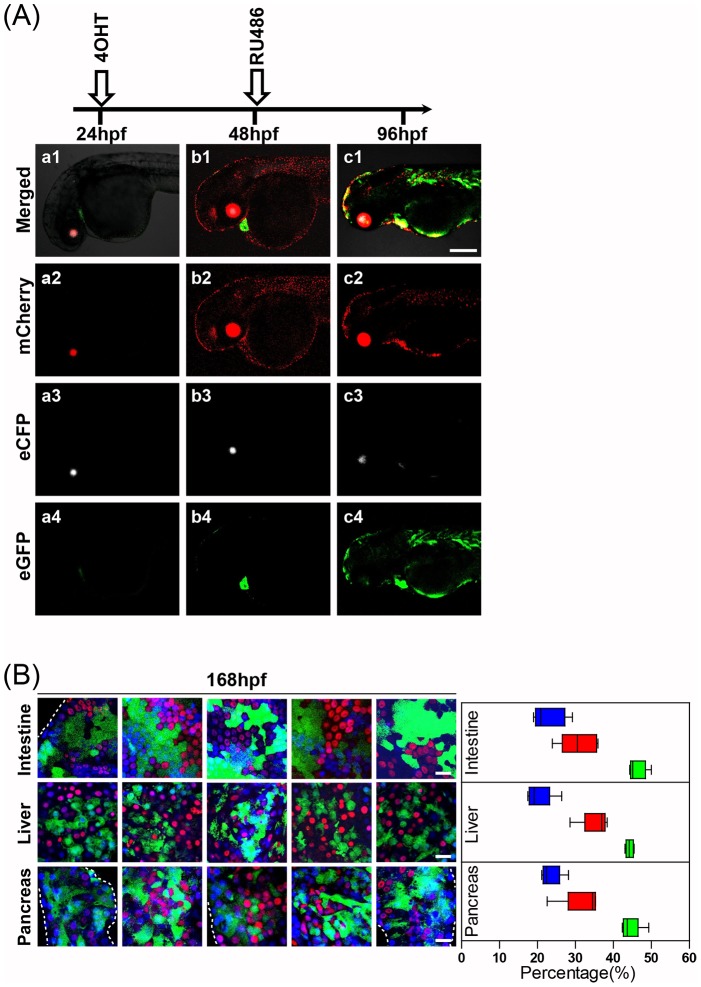
Figure 4. Sequential transgene activation and inactivation using lox and rox (TAILOR).
(A) ubb:lox-stop-lox-rox-Nuc-mCherry-stop-rox-eGFP; ubb-CreERT2; ubb-Dre triple transgenic fish were subjected to treatment with 4-OHT beginning at 24 hpf, followed by removal and replacement with RU486 at 48 hpf. The untreated triple transgenic embryos (24 hpf) showed no transgene-specific fluorescence besides that provided by the ocular mCherry, ocular eCFP and cardiac eGFP markers (Fig. 4A a1, a2, a3, and a4). The effective induction of nuclear mCherry expression was observed following 24 hrs of 4-OHT treatment (Fig. 4A b1 and b2). Following staged treatment with RU486 initiated at 48 hpf and left in place for 24 hrs, effective activation of eGFP expression was observed (Fig. 4A c1 and c4). Scale bar: 200 µm. (B) Quantification of relative numbers of intestinal, liver and pancreatic cells expressing mCherry (red), eGFP (green) or neither (blue) at 7 days following sequential 4OHT and RU486 exposure as above. Note high fraction of cells undergoing sequential CreERT2-mediated activation and DrePR-mediated inactivation of mCherry expression, as indicated by eGFP expression. Scale bar: 25 µm.
The untreated triple transgenic embryos (24 hpf) showed no transgene-specific fluorescence besides that provided by the ocular mCherry, ocular eCFP and cardiac eGFP markers (Fig. 4A a1, a2, a3, and a4). The effective induction of nuclear mCherry expression was observed following 24 hrs of 4-OHT treatment (Fig. 4A b1 and b2). Nuc-mCherry signal was detected by immunofluorescence within 6 hrs of 4-OHT treatment, and signal was widely expressed in all regions of embryo following 24 hrs of 4-OHT treatment. Following staged treatment with RU486 initiated at 48 hpf and left in place for 24 hrs, effective activation of eGFP expression was observed (Fig. 4A c1 and c4). The activation of eGFP fluorescence was evident within 6 hrs of RU486 application, indicating the rapid excision of rox-flanked mCherry in at least a subset of larval cells.
Not unexpectedly, embryos treated sequentially with 4-OHT and RU486 and harvested at 96 hpf continued to have a fraction of cells displaying ongoing red nuclear fluorescence (Fig 4A.c.2), potentially reflecting both perdurant mCherry protein and/or mosaic DrePR activity. In order to more formally document sequential transgene activation and inactivation, as well as to calculate the efficiency of sequential CreERT2- and DrePR-based recombination, we delayed harvest until 7 dpf and examined patterns of mCherry and eGFP fluorescence in intestine, liver and pancreas. This allowed us to precisely determine the number of cells undergoing either no recombination, single recombination mediated by CreERT2, or double recombination mediated by the sequential activity of CreERT2 and DrePR. Minimal variation in recombination frequencies was observed between these three tissues, with 20–25% of cells undergoing no recombination (blue DAPI fluorescence only), 30–40% of cells undergoing single recombination mediated by CreERT2 (red mCherry fluorescence only), and 40–50% of cells undergoing sequential transgene activation and inactivation mediated by both CreERT2 and DrePR (green eGFP fluorescence only).
Discussion
Together, these studies establish Dre/rox-based recombination as a new and effective method for temporally- and spatially-regulated genetic manipulation in zebrafish. In addition to this general utility, we present a new strategy for sequential activation and inactivation of inserted transgenes, referred to as TAILOR. Prior methods for temporally regulated transgene expression in zebrafish have included the use of thermally-inducible heat shock promoters [29], [30], the application of modified tetracycline-inducible systems [31], [32], and the development of hormone-regulated hybrid transcriptional activators [33]. More recently, DNA recombinases have been employed for temporal regulation of zebrafish transgene expression, and the power of combinatorial approaches utilizing multiple recombinases has become apparent [12], [13], [34].
The current addition of Dre/rox to the zebrafish genetic toolbox will enable a variety of experimental strategies requiring precise temporal and spatial control of transgene expression. New TAILOR transgenes employing additional “LSL-R-cDNA-R” cassettes will facilitate a wide variety of experiments requiring sequential and cell type-specific transgene activation and inactivation. As in the case of our ubb:lox-stop-lox-rox-mCherry-stop-rox-eGFP allele, recombinase-mediated activation and inactivation of any LSL-Rox-cDNA-Rox cassette is likely to be mosaic. However the incorporation of fluorescent proteins into LSL-Rox-cDNA-Rox alleles, either by in-frame fusion or by 2A peptide-based independent translation [35], will enable the identification of neighboring cells with and without residual transgene expression, allowing such mosaicism to be highly informative. Similarly, the generation of “LSL-RSR-cDNA” alleles, in which expression of a downstream transgene requires both Cre- and Dre-mediated recombination events, will allow ever more precise temporal and spatial regulation of transgene expression in highly selective cell types characterized by intersectional expression of both Cre and Dre. Dre/rox-based recombination will now also allow independent genetic manipulation of separate transgenes flanked by lox and rox sites, respectively. For example, Dre/rox-based recombination could be used to activate transgene expression in one cell population, with Cre/lox-mediated activation of a different transgene occurring in neighboring cells. As capabilities for gene targeting by endonuclease-facilitated homologous recombination [36], [37] become more widespread in zebrafish, the combinatorial application of Cre/lox and Dre/rox technology will also facilitate temporally regulated and cell type-specific manipulation of endogenous genetic loci.
Acknowledgments
The authors wish to thank Francis Stewart for providing Dre and DrePR cDNAs. The authors also wish to thank Marnie Halpern and Michael Parsons for many helpful discussions.
Funding Statement
This work was supported by NIH grants DK56211 and HD058530. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Mosimann C, Kaufman CK, Li P, Pugach EK, Tamplin OJ, et al. (2011) Ubiquitous transgene expression and Cre-based recombination driven by the ubiquitin promoter in zebrafish. Development 138: 169–177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Sinha DK, Neveu P, Gagey N, Aujard I, Le Saux T, et al. (2010) Photoactivation of the CreER T2 recombinase for conditional site-specific recombination with high spatiotemporal resolution. Zebrafish 7: 199–204. [DOI] [PubMed] [Google Scholar]
- 3. Collins RT, Linker C, Lewis J (2010) MAZe: a tool for mosaic analysis of gene function in zebrafish. Nat Methods 7: 219–223. [DOI] [PubMed] [Google Scholar]
- 4. Seok SH, Na YR, Han JH, Kim TH, Jung H, et al. (2010) Cre/loxP-regulated transgenic zebrafish model for neural progenitor-specific oncogenic Kras expression. Cancer Sci 101: 149–154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Speck NA, Iruela-Arispe ML (2009) Conditional Cre/LoxP strategies for the study of hematopoietic stem cell formation. Blood Cells Mol Dis 43: 6–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Le X, Langenau DM, Keefe MD, Kutok JL, Neuberg DS, et al. (2007) Heat shock-inducible Cre/Lox approaches to induce diverse types of tumors and hyperplasia in transgenic zebrafish. Proc Natl Acad Sci U S A 104: 9410–9415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Liu WY, Wang Y, Qin Y, Wang YP, Zhu ZY (2007) Site-directed gene integration in transgenic zebrafish mediated by cre recombinase using a combination of mutant lox sites. Mar Biotechnol (NY) 9: 420–428. [DOI] [PubMed] [Google Scholar]
- 8. Pan X, Wan H, Chia W, Tong Y, Gong Z (2005) Demonstration of site-directed recombination in transgenic zebrafish using the Cre/loxP system. Transgenic Res 14: 217–223. [DOI] [PubMed] [Google Scholar]
- 9. Thummel R, Burket CT, Brewer JL, Sarras MP Jr, Li L, et al. (2005) Cre-mediated site-specific recombination in zebrafish embryos. Dev Dyn 233: 1366–1377. [DOI] [PubMed] [Google Scholar]
- 10. Langenau DM, Feng H, Berghmans S, Kanki JP, Kutok JL, et al. (2005) Cre/lox-regulated transgenic zebrafish model with conditional myc-induced T cell acute lymphoblastic leukemia. Proc Natl Acad Sci U S A 102: 6068–6073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Dong J, Stuart GW (2004) Transgene manipulation in zebrafish by using recombinases. Methods Cell Biol 77: 363–379. [DOI] [PubMed] [Google Scholar]
- 12. Ni TT, Lu J, Zhu M, Maddison LA, Boyd KL, et al. (2012) Conditional control of gene function by an invertible gene trap in zebrafish. Proc Natl Acad Sci U S A 109: 15389–15394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Trinh le A, Hochgreb T, Graham M, Wu D, Ruf-Zamojski F, et al. (2011) A versatile gene trap to visualize and interrogate the function of the vertebrate proteome. Genes Dev 25: 2306–2320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wong AC, Draper BW, Van Eenennaam AL (2011) FLPe functions in zebrafish embryos. Transgenic Res 20: 409–415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Boniface EJ, Lu J, Victoroff T, Zhu M, Chen W (2009) FlEx-based transgenic reporter lines for visualization of Cre and Flp activity in live zebrafish. Genesis 47: 484–491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Wang Y, Rovira M, Yusuff S, Parsons MJ (2011) Genetic inducible fate mapping in larval zebrafish reveals origins of adult insulin-producing beta-cells. Development 138: 609–617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Sauer B, McDermott J (2004) DNA recombination with a heterospecific Cre homolog identified from comparison of the pac-c1 regions of P1-related phages. Nucleic Acids Research 32: 6086–6095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Anastassiadis K, Fu J, Patsch C, Hu S, Weidlich S, et al. (2009) Dre recombinase, like Cre, is a highly efficient site-specific recombinase in E. coli, mammalian cells and mice. Dis Model Mech 2: 508–515. [DOI] [PubMed] [Google Scholar]
- 19. Sauer B, McDermott J (2004) DNA recombination with a heterospecific Cre homolog identified from comparison of the pac-c1 regions of P1-related phages. Nucleic Acids Res 32: 6086–6095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Mosimann C, Kaufman CK, Li P, Pugach EK, Tamplin OJ, et al. (2011) Ubiquitous transgene expression and Cre-based recombination driven by the ubiquitin promoter in zebrafish. Development 138: 169–177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Kawakami K (2004) Transgenesis and gene trap methods in zebrafish by using the Tol2 transposable element. Methods Cell Biol 77: 201–222. [DOI] [PubMed] [Google Scholar]
- 22. Parsons MJ, Pisharath H, Yusuff S, Moore JC, Siekmann AF, et al. (2009) Notch-responsive cells initiate the secondary transition in larval zebrafish pancreas. Mech Dev 126: 898–912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Hesselson D, Anderson RM, Stainier DY (2011) Suppression of Ptf1a activity induces acinar-to-endocrine conversion. Curr Biol 21: 712–717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Kellendonk C, Tronche Fo, Monaghan AP, Angrand P-O, Stewart F, et al. (1996) Regulation of Cre Recombinase Activity by the Synthetic Steroid RU 486. Nucleic Acids Research 24: 1404–1411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Zhang Y, Riesterer C, Ayrall A-M, Sablitzky F, Littlewood TD, et al. (1996) Inducible Site-Directed Recombination in Mouse Embryonic Stem Cells. Nucleic Acids Research 24: 543–548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Awatramani R, Soriano P, Rodriguez C, Mai JJ, Dymecki SM (2003) Cryptic boundaries in roof plate and choroid plexus identified by intersectional gene activation. Nat Genet 35: 70–75. [DOI] [PubMed] [Google Scholar]
- 27.Hirsch MR, d’Autreaux F, Dymecki SM, Brunet JF, Goridis C (2013) A Phox2bColon, two colonsFLPo transgenic mouse line suitable for intersectional genetics. Genesis. [DOI] [PMC free article] [PubMed]
- 28. Dymecki SM, Ray RS, Kim JC (2010) Mapping cell fate and function using recombinase-based intersectional strategies. Methods Enzymol 477: 183–213. [DOI] [PubMed] [Google Scholar]
- 29. Halloran MC, Sato-Maeda M, Warren JT, Su F, Lele Z, et al. (2000) Laser-induced gene expression in specific cells of transgenic zebrafish. Development 127: 1953–1960. [DOI] [PubMed] [Google Scholar]
- 30. Adam A, Bartfai R, Lele Z, Krone PH, Orban L (2000) Heat-inducible expression of a reporter gene detected by transient assay in zebrafish. Exp Cell Res 256: 282–290. [DOI] [PubMed] [Google Scholar]
- 31. Knopf F, Schnabel K, Haase C, Pfeifer K, Anastassiadis K, et al. (2010) Dually inducible TetON systems for tissue-specific conditional gene expression in zebrafish. Proc Natl Acad Sci U S A 107: 19933–19938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Huang CJ, Jou TS, Ho YL, Lee WH, Jeng YT, et al. (2005) Conditional expression of a myocardium-specific transgene in zebrafish transgenic lines. Dev Dyn 233: 1294–1303. [DOI] [PubMed] [Google Scholar]
- 33. Emelyanov A, Parinov S (2008) Mifepristone-inducible LexPR system to drive and control gene expression in transgenic zebrafish. Dev Biol 320: 113–121. [DOI] [PubMed] [Google Scholar]
- 34. Maddison LA, Lu J, Chen W (2011) Generating conditional mutations in zebrafish using gene-trap mutagenesis. Methods Cell Biol 104: 1–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Provost E, Rhee J, Leach SD (2007) Viral 2A peptides allow expression of multiple proteins from a single ORF in transgenic zebrafish embryos. Genesis 45: 625–629. [DOI] [PubMed] [Google Scholar]
- 36. Bedell VM, Wang Y, Campbell JM, Poshusta TL, Starker CG, et al. (2012) In vivo genome editing using a high-efficiency TALEN system. Nature 491: 114–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Hwang WY, Fu Y, Reyon D, Maeder ML, Tsai SQ, et al. (2013) Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat Biotechnol 31: 227–229. [DOI] [PMC free article] [PubMed] [Google Scholar]