Abstract
Previously, a peptide epitope derived from the Hsp 60 molecule of Salmonella that is presented by the major histocompatibility complex (MHC) class Ib molecule Qa-1 to CD8+ cytotoxic T cells (CTLs) was described. In the present study we investigated the Salmonella-induced processing and presentation pathway for generating this Qa-1-restricted epitope. Live bacteria and, to a lesser extent, opsonized heat-killed bacteria are able to sensitize target cells for lysis by Salmonella-specific CTL. In contrast, heat-killed bacteria cannot sensitize target cells. Presentation of the Hsp 60 epitope appears independent of bacterial internalization, because cytochalasin D does not affect presentation. Moreover, Salmonella strains defective in the InvA or InvE operon, two critical components of the type III secretion pathway, are as efficient as wild-type Salmonella enterica serovar Typhimurium in sensitizing infected targets to lysis. Collectively, these results suggest the existence of a novel antigen-processing pathway in which exogenous antigens gain access to the cytosolic MHC class I processing machinery. Considering the abundant nature of bacterial Hsp 60 and the upregulation of this protein after Salmonella infection of eukaryotic cells, this mode of antigen presentation may be particularly relevant to understanding the host defense mechanisms against gram-negative bacteria.
Class I molecules play a fundamental role in the activation of CD8+ cytotoxic T cells (CTLs) during the generation of an adaptive immune response to intracellular pathogens (43). It has been generally accepted that the first step in the endogenous class I antigen presentation pathway is the hydrolysis of cytosolic proteins into peptides by the proteasome (41). These peptides are then selectively translocated into the endoplasmic reticulum (ER) by the transporter associated with antigen processing (TAP) complex. As a result, this set of peptides is available to bind to the peptide-binding site of newly synthesized major histocompatibility complex (MHC) class I molecules in the ER. Once a stable complex of peptide, MHC class I molecule, and β-2 microglobulin is formed, the complex is transported to the plasma membrane and is available for recognition by and activation of peptide-specific CD8+ T cells. In this manner the immune system can recognize host cells that have been altered by infection, mutation, or malignant transformation.
Intracellular and extracellular bacterial pathogens can bind to and be internalized by professional antigen-presenting cells (APCs) (29). Many intracellular pathogens survive within host membrane-bound vesicles and, in some instances, replicate within these vesicles. Despite this subcellular localization, several examples have been described wherein bacterial antigens derived from intracellular bacteria gain access to the class I processing pathway for presentation by MHC class I to bacterium-specific CD8+ effector T cells (reviewed in reference 21).
The processing of bacterial antigens through the MHC class I pathway has been shown to be independent of bacterial cytoplasmic localization (47). Several models have been proposed to account for this phenomenon, including peptide regurgitation, leaky endosomal membranes, disruption of overloaded endosomes, and direct binding to cell surface class I molecules (reviewed in reference 40). However, none of these models to date has resulted in a general picture of the mechanism(s) by which endosomal antigens access the cytosol. More recently, it has been proposed that direct cytosolic transfer of selected Salmonella or Yersinia proteins occurs efficiently and is independent of host cell invasion (7). It is interesting that even noninvasive Escherichia coli strains can sensitize target cells to lysis by human bacterium-specific CTLs (24). Studies with salmonellae have shown that, once internalized, Salmonella organisms reside within a membrane-bound vacuolar compartment (4, 46). All of these observations indicate that multiple mechanisms likely exist by which a peptide(s) can be presented by class I molecules.
Previous studies have identified a Salmonella Hsp 60-derived epitope presented to CD8+ CTLs by the Qa-1 class Ib molecule (32). The presentation of this Hsp 60 epitope is both TAP and proteasome dependent. In the present study we investigated the processing and presentation pathway for the generation of Salmonella-derived Qa-1-restricted epitopes. Our results suggest the existence of a novel alternative class I antigen-processing pathway in which exogenous antigens can be processed through a cytosolic pathway under phagocytic stimuli provided by live Salmonella organisms. Considering the abundant nature of bacterial Hsp 60 and the upregulation of this protein upon infection of eukaryotic cells (5), this mode of antigen presentation may be relevant to a general understanding of the host defense against all gram-negative bacteria.
MATERIALS AND METHODS
Bacteria strains and growth conditions.
Wild-type virulent Salmonella enterica serovar Typhimurium strain C5 (C5) was utilized (51). Wild-type S. enterica serovar Typhimurium strain SR11 and the SR11-derived Salmonella pathogenicity island (SPI)-1-deficient strains SB147 (invA) and SB109 (invE) were also used (12, 14, 51). For the studies described herein, all bacterial strains were grown overnight in Luria-Bertani (LB) broth at 37°C, diluted in fresh medium, and recultured at 37°C to mid-log phase (optical density at 600 nm of 0.5). Bacteria were centrifuged and washed in saline twice before use.
In experiments with heat-killed S. enterica serovar Typhimurium (HKST), log-phase bacteria were incubated in a 72°C water bath for 1 h and the viability was assessed by plating on LB plates. In selected experiments using opsonized heat-killed bacteria, heat-killed bacteria were exposed to 50% normal homologous mouse serum in Hanks' balanced salt solution (HBSS) for 30 min and were washed twice with HBSS before use.
Cell lines and in vitro infection of tissue culture cells.
The J774 macrophage cell line, the A20 B-cell lymphoma cell line, and the EL4 T-cell lymphoma cell line were cultured in RPMI supplemented with 10% fetal calf serum (RPMI-10). Qa-1b-expressing variants of the class I-deficient murine melanoma cell line B78H1 and the L-cell fibroblast line generated by cotransfection of the plasmids pSG-37 and pSV-2neo have been described previously (27, 45). The Qa-1b-expressing B78Hi and L-cell lines were maintained in selection medium (DMEM-10 supplemented with G418 [Sigma Chemical, St. Louis, Mo.] at 600 μg/ml so as to maintain high levels of Qa-1b expression). Tissue culture cells (106) were coincubated with the bacteria at a multiplicity of infection (MOI) of 100 and were pelleted by centrifugation at 600 × g. The bacterial cell pellets were incubated at 37°C for 30 min and washed two times in medium, and the cell density was adjusted as appropriate. For CTL assays, all infected cells were incubated at 37°C for another 1 h to allow for antigen processing. The efficiency of infection was assessed 2 h after the last wash step by enumerating gentamicin-protected intracellular bacteria as described previously (34) and by staining bacteria with the Diff-quik stain set (Baxter Scientific, Edison, N.J.) per the manufacturer's instructions.
Cytotoxic T-cell assays.
In experiments using cytochalasin D (Calbiochem, La Jolla, Calif.), J774 and P815 cells were pretreated with cytochalasin D at 50 μg/ml for 1 h at 37°C. Cells were then washed thoroughly with complete medium and were infected with S. enterica serovar Typhimurium C5 as described above. In experiments using chloroquine (Calbiochem), C5-infected P815 cells were incubated with 50 μM chloroquine for 1 h and then were washed before 51Cr labeling.
Infected and uninfected target cells (2 × 106/200 μl) were labeled with 51Cr for 1 h. Labeled cells were washed with prewarmed (37°C) medium, resuspended in 2 ml of T-cell medium (TCM) (32, 33), centrifuged through 1.5 ml of warm fetal calf serum, resuspended in 5 ml of TCM, and allowed to incubate at 37°C for 1 h. Suspensions of labeled cells were then spun through fetal calf serum again and were counted (37). The density of the labeled target cells was adjusted to 5 × 104/ml. One hundred microliters (5 × 103 cells) of the labeled targets was dispensed into each well of 96-well U-bottomed plates and was mixed with an equal volume of effector cells at the indicated effector:target ratios. After 4 h of incubation, 100 μl of the supernatant was harvested and the released 51Cr was counted. The percent specific lysis was calculated as 100% × (counts per million [cpm] of experimental release − cpm of spontaneous release)/(cpm of total release in the presence of 1% Nonidet P-40 − cpm of spontaneous release).
Detection of translocated bacterial proteins in S. enterica serovar Typhimurium-infected cells.
A modification of the procedure developed by Collazo et al. (7) was utilized. Briefly, cultures of J774 cells were infected with S. enterica serovar Typhimurium strains at an MOI of 100 as described above. Following infection, cells were washed with HBSS and then were incubated for various periods of time in medium with 100 μg of gentamicin/ml to kill extracellular bacteria. Cells were harvested and either mock treated or treated with 30 μg of proteinase K/ml in HBSS for 15 min at 37°C to eliminate cell surface-associated residual bacterial proteins. After proteinase K treatment, cells were washed and solubilized in 1% sodium dodecyl sulfate (SDS). Solubilized samples were sonicated on ice and centrifuged at 15,000 × g for 10 min; protein levels were determined by using the Bio-Rad protein assay, and the samples were adjusted to 2% 2-mercaptoethanol prior to gel electrophoresis. Approximately 106 cell equivalents were applied to each lane of the gel and were analyzed by SDS-10% polyacrylamide gel electrophoresis. Lysates prepared from uninfected control targets were run in parallel. SDS-polyacrylamide gels were then transferred to nitrocellular membranes and were probed with horseradish peroxidase-coupled polyclonal rabbit anti-GroEL (dilution, 1:2,000; Sigma Chemical). This reagent does not cross-react with the mammalian Hsp 60 molecule and cross-recognizes Salmonella GroEL (see Fig. 6).
FIG. 6.
Detection of Hsp 60 in Salmonella-infected cells. J774 cells were infected at an MOI of ∼100 followed by incubation in the presence of 0.1 mg of gentamicin/ml. At 0, 1, 2, and 3 h after infection, cells were harvested, resuspended with (+) or without (−) 30 μg of proteinase K (pk)/ml, and incubated for 15 min at 37°C. Following washing, cells were solubilized in SDS and the presence of bacterial Hsp 60 was detected by Western blotting with rabbit anti-Hsp 60. The arrow designates the Salmonella Hsp 60 species detected when cells were infected with strain SR11 (wild type, top panel) or invasion-deficient strain invA (middle panel) or invE (bottom panel). Extracts from uninfected cells (lane U) were analyzed in parallel. wt, wild type.
RESULTS
Both live and opsonized heat-killed bacteria, but not untreated dead bacteria, sensitize J774 macrophages to lysis by Salmonella-specific CTL clone SalTc 1.69.
Studies of CD8+ T-cell responses to Listeria monocytogenes established that bacterial extracts, outer membrane fractions, and heat-killed bacteria are taken up by professional APCs and are presented by the MHC class Ib molecule, H2-M3, to Listeria-specific CD8+ T cells (2, 3, 31). Nonphagocytic cells, such as EL-4 cells (36), were not able to execute the same function. To determine if Salmonella-specific CD8+ T cells had similar recognition restrictions, we assessed the capability of HKST or opsonized heat-killed bacteria to sensitize targets for lysis by CTLs. In these experiments, J774 cells were exposed to HKST for 1 h (Fig. 1B) or overnight (Fig. 1A) and were tested for recognition by SalTc 1.69. SalTc 1.69 did not lyse HKST-pulsed (1 h) J774 cells and only marginally lysed J774 cells pulsed (∼6%) overnight with HKST. To enhance the phagocytosis of bacteria by J774 cells, HKST were incubated with normal mouse serum for 30 min, after which they were used to pulse J774 cells (Fig. 1B). The results show that after opsonization, SalTc 1.69 was now able to recognize HKST-pulsed J774 targets, but the levels of CTL-mediated lysis was reduced in comparison with CTL-mediated lysis in response to live-bacterium-infected J774 cells (Fig. 1). In contrast to the results observed with the gram-positive bacterium L. monocytogenes, our results indicate that infection of target cells with viable bacteria rather than killed S. enterica serovar Typhimurium is significantly more effective for the initiation of class I processing of serovar Typhimurium-derived antigens.
FIG. 1.
The Salmonella-specific Qa-1b-restricted CTL clone SalTc 1.69 recognizes opsonized heat-killed bacteria. J774 macrophages were pulsed with HKST for 1 h (B) or overnight (A) or with HKST opsonized in normal mouse serum (NMS) (B) for 30 min. At the end of coincubation, J774 cells were labeled with 51Cr and used as targets in a CTL assay employing SalTc 1.69 as the effector at the indicated effector:target ratios. The viability of the bacteria was confirmed by plating an aliquot of the heat-killed bacteria on LB plates. For comparison, live C5-infected J774 cells were included in these assays.
Both infected phagocytes and nonphagocytes are capable of presenting Salmonella epitopes to CTLs.
Previously, it has been shown that Salmonella organisms are able to infect both phagocytes and nonphagocytes (9, 10). However, the capacity of infected phagocytes and nonphagocytes to present Salmonella-specific CTL epitopes has not been clearly delineated. As shown in Fig. 2, Salmonella-infected EL-4 cells (T-cell lymphoma), Salmonella-infected A20 (B-cell lymphoma) cells, and Salmonella-infected B78Hi melanoma cells expressing Qa-1b were all lysed by SalTc 1.69. The percentage of specific lysis of these targets was comparable to that for Salmonella-infected J774 macrophages.
FIG. 2.
SalTc 1.69 recognizes infected target cells of various lineages. EL-4 (A), A20 (B), B78Hi and B78Hi-Qa-1b (C), and J774 (D) cells were infected with S. enterica serovar Typhimurium C5 and tested for recognition by SalTc 1.69. (E) The average numbers of intracellular bacteria for J774, EL-4, A20, and B78Hi-Qa-1b were 3.2, 1.2, 1.5, and 2.5, respectively. The average number of viable bacteria, determined 2 h after infection, represents the mean taken from three separate samples each of 106 cells.
In addition, we assessed the efficiency of bacterial invasion by measuring the number of intracellular bacteria. In these assays, a panel of infected target cells was incubated with the cell membrane-impermeable antibiotic gentamicin, which kills extracellular but not intracellular bacteria. After 2 h of incubation the number of intracellular bacteria was determined. As shown in Fig. 2E, J774 macrophages harbored, on average, 3.2 bacteria per cell. The nonphagocytes EL-4, A20, and B78Hi-Qa-1b contained only 1.2, 1.5, and 2.5 bacteria per cell, respectively. This result suggested that the degree of bacterial invasion may not correlate with the susceptibility of target cells to Salmonella-specific CTL lysis.
Previous studies demonstrated that SalTc 1.69 recognizes a peptide derived from the Salmonella Hsp 60 molecule (32). In the present study, we asked whether the Salmonella Hsp 60 protein can be detected in infected target cells. C5-infected J774, P815, and L-Qa-1b cells were lysed, and the lysates were probed with anti-Hsp 60 immune sera (7). The results are shown in Fig. 3 and indicate that Salmonella Hsp 60 was detected in infected cells but not in uninfected cells (Fig. 3B). In parallel, internalized bacteria were quantified by invasion assays as described above. Similar to the results shown in Fig. 2, P815 and L-Qa-1b were infected at a lower efficiency than J774 macrophages (Fig. 3A). More importantly, these experiments demonstrate that significant levels of Salmonella Hsp 60 are detected in infected target cells, despite the low level of internalized bacteria. Collectively, our results show that active uptake of bacteria by host cells does not correlate with susceptibility to CTL lysis.
FIG. 3.
Sensitization of target cells to CTL lysis does not correlate with bacterial invasion. (A) J774, P815, and L-Qa-1b cells infected with C5 were lysed with 1% Triton X-100 for quantification of intracellular bacteria 2 h following infection. The average number of viable bacteria, determined 2 h after infection, represents the mean of three separate samples, each of 106 cells. (B) Cellular lysates prepared from infected (C5) and uninfected (−) target cells were probed with immune sera against bacterial Hsp 60 as described in Materials and Methods.
Bacterial internalization is not required for sensitization of target cells.
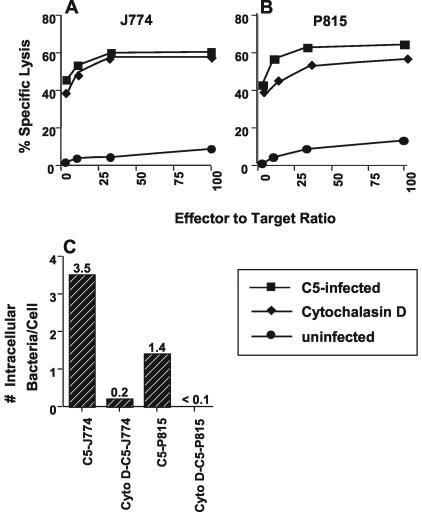
To further investigate the possible mechanism(s) for processing and presentation of bacterially expressed Hsp 60 protein, we determined whether a complete block in bacterial internalization would affect the presentation of bacterial epitopes to CTLs. Cytoskeletal rearrangement is a critical step in bacterial internalization, and previous studies have shown that disruption of the cytoskeleton by cytochalasin D abolishes bacterial invasion (7, 15, 34). Pretreatment of both J774 and P815 cells with 50 μg of cytochalasin D/ml significantly reduced the efficiency of bacterial internalization (Fig. 4C). More importantly, Fig. 4A and B show that pretreated Salmonella-infected cells were as susceptible to SalTc 1.69 lysis as untreated Salmonella-infected cells. These observations argue that bacterial internalization is not required for sensitization of host target cells to occur.
FIG. 4.
Pretreatment of target cells with cytochalasin D does not affect bacterial Hsp 60 antigen processing. J774 (A) and P815 (B) cells were left untreated (squares) or exposed to cytochalasin D at 50 μg/ml (diamonds) for 1 h at 37°C. The cells were then washed and infected with C5. (C) The average number of viable bacteria, determined 2 h after C5 infection, represents the mean taken from three separate samples, each of 106 cells.
Both host cell elements and bacterial genes contribute to effective invasion of mammalian cells by pathogenic bacteria. For example, bacterial genes that control the highly specialized type III secretion system are essential for Salmonella invasion of host cells (7, 9, 11, 20, 42). To determine the contribution of this pathway to the generation of Salmonella-specific Qa-1b-restricted CTL epitopes, J774 macrophages and P815 mastocytoma cells were infected with two S. enterica serovar Typhimurium mutants defective in essential components of this apparatus, invA− and invE− (12, 14). Interestingly, both invA− and invE− mutants, as well as the parental wild-type strain SR11, were able to sensitize both J774 and P815 cells to CTL lysis (Fig. 5). Taken together, the data in Fig. 4 and 5 demonstrate that interfering in the invasion process by blocking host cell or bacterial elements does not interfere with the generation of the Qa-1b-restricted bacterial epitopes.
FIG. 5.
Susceptibility of infected target cells does not depend on the type III protein secretion system of S. enterica serovar Typhimurium. J774 (A) and P815 (B) cells were infected with parental strain SR11 (squares), the invA− mutant (diamonds), or the invE− mutant (circles), respectively. Infected target cells were tested for lysis by clone SalTc 1.69.
Salmonella Hsp 60 gains access to the cytoplasm in the absence of bacterial internalization.
The results in the previous section suggest that the Salmonella Hsp 60 polypeptide gains access to the class I processing pathway in the absence of bacterial internalization. To determine if this conclusion is correct, we wished to establish whether Hsp 60 is detected intracellularly following infection with invasion-defective Salmonella organisms. In these studies, J774 cells were incubated with wild-type, invA− mutant, or invE− mutant Salmonella strain, and at various times thereafter cells were washed and then mock treated or treated with protease to remove any remaining cell-bound bacteria. Following protease treatment, the J774 cells were lysed in detergent and the presence of Hsp 60 in the lysates was determined by Western blotting. Cell-associated Hsp 60 can be detected early after infection with wild-type strain SR11, as well as with the invA− and invE− mutants (Fig. 6). The Hsp 60 detected at the early time points appears to be derived from extracellular bacteria, because this signal is protease sensitive. However, at later time points a significant fraction of the Hsp 60 molecules enter a protease-resistant compartment when J774 cells are infected with strain SR11 or the invA− or invE− mutant (Fig. 6). The presence of protease-resistant Hsp 60 in detergent lysates of infected cells indicates an intracellular localization of Hsp 60 (7). These results suggest that intracellular Hsp 60 is acquired through a type III-independent pathway.
Based on the above-described studies that indicated that processing and presentation of the Salmonella Hsp 60 epitope does not require internalization of whole viable bacteria and yet is Tap and proteasome dependent, we hypothesized that the host was playing a major role in the internalization of Hsp 60. Therefore, we examined whether a novel endocytic process of the host that uses the organic base chloroquine, which blocks the release of endocytosed material, could be implicated. As shown in Fig. 7, target cells treated with chloroquine were no longer efficiently recognized by peptide-specific CTLs. These results indicate that the Salmonella Hsp 60 molecule that is processed and presented gains entry into the host cell by a chloroquine-dependent endocytic pathway.
FIG. 7.
Chloroquine interferes with CTL recognition. Salmonella-infected P815 cells were treated with 50 μM chloroquine (circle) or were mock treated (square) prior to labeling with 51Cr and testing for lysis by bulk Salmonella-specific CTLs derived from the spleen of infected BALB/c mice (left panel) or the Salmonella-specific CTL clone SalTc 1.69 (right panel). Control uninfected cells are also shown (triangle).
DISCUSSION
Our studies using HKST demonstrate that bacterial viability is required for the efficient processing and presentation of the Salmonella Hsp 60 epitope to the Salmonella-specific CTL clone SalTc 1.69. Dead bacteria fail to sensitize target cells to lysis even following an overnight bacterial pulse of the J774 cells. These observations are in contrast to the H2-M3 restricted recognition of Listeria-infected targets, wherein dead bacteria or bacterial extracts have been shown to be effective sources of antigen (38). However, opsonization of heat-killed bacteria with normal mouse serum prior to pulsing of J774 cells does facilitate processing and CTL recognition. This result is consistent with uptake of the bacteria via a complement C3-mediated macropinocytosis mechanism that efficiently promotes the processing and presentation of the constitutively expressed bacterial Hsp 60.
The observation that viable bacteria are required for the elicitation of target sensitivity implies that S. enterica serovar Typhimurium is an active participant in the process that permits the bacterial Hsp 60 protein to gain access to the class I processing pathway. It is well known that when Salmonella encounters an appropriate host cell surface, the bacterium initiates a series of events that leads to internalization and residency in membrane-bound vesicles. Therefore, it seemed reasonable that these internalized bacteria could be the source of antigen that is processed and presented. However, bacterial internalization did not appear to be required, because blocking internalization with cytochalasin D or the use of Salmonella isolates that are deficient in cell invasion failed to affect the processing and presentation of the Hsp 60-derived peptide. These data argue that extracellular bacteria are an important source of Hsp 60 antigen that eventually gains access to the cytoplasm for processing. This conclusion is supported by the observation that nonphagocytic cell types, which internalize bacteria inefficiently, are also efficiently recognized by CTLs.
Interestingly, under conditions that prevent cellular invasion, bacterial Hsp 60 proteins can be detected in the cytoplasm of infected cells. This observation indicates that Hsp 60 molecules derived from extracellular bacteria need to be translocated into the eukaryotic cell. Translocation of Hsp 60 does not involve the Salmonella SPI-1-encoded type III secretion system, because intracellular Hsp 60 can be easily detected after incubation of target cells with SPI-1-deficient Salmonella invA or invE mutants. This finding suggests that while viable extracellular bacteria are the source of antigen, bacteria are not mediating the translocation of Hsp 60 into the host cell via the type III secretion pathway.
The host cell appears to be largely responsible for Hsp 60 uptake, because chloroquine disrupts the eukaryotic endocytic pathway and blocks presentation of Hsp 60 in this case. The mechanism by which bacterial Hsp 60 is bound and internalized into an endocytic pathway is unclear. Several receptors have been reported for bacterial and mammalian Hsp 60 molecules, including Toll-like receptor-2 (TLR-2) and TLR-4 as well as a TLR-independent mechanism (6, 17, 35, 50). Studies examining the role of the cell surface protein OmpA on the gram-negative pathogen Klebsiella pneumoniae demonstrated that OmpA could bind to and signal cells in a TLR-2-dependent fashion (28). Furthermore, this binding leads to OmpA internalization and subsequent access to a TAP-dependent class I processing pathway. Therefore, it is reasonable to speculate that Salmonella-derived Hsp 60 binds to an Hsp 60 cell surface receptor(s) and initiates a signaling pathway leading to endocytosis and subsequent access to the MHC class I processing pathway.
The Hsp 60 protein is largely localized in the cytoplasm of bacteria, where it functions as a chaperone (1, 49). Also, Hsp 60 does not appear to be secreted or released from Salmonella organisms, because Hsp 60 is not detected in cell-free media from Salmonella cells grown under various growth conditions (C. D. Dunn and M. J. Soloski, unpublished data). However, Hsp 60 has been reported to be associated with the bacterial surfaces of Helicobacter pylori, Legionella pneumophila, and Clostridium difficile, and this may be a common feature of bacteria that bind to mammalian cell surfaces (23, 25, 52). In the case of L. pneumophila and C. difficile, a role in adherence and invasion has been proposed. Therefore, bacterial cell surface-associated proteins, or other forms of Hsp 60 expressed by Salmonella organisms, may become available for internalization upon binding to mammalian cell surfaces.
It remains unclear how Salmonella-derived antigens gain access to the cytosol for proteasome-dependent degradation, but bacterial Hsp 60 is clearly detected in infected target cells (Fig. 3B and 6). Salmonellae infect host cells and remain predominantly, if not exclusively, within a vacuolar endosomal compartment. S. enterica serovar Typhimurium can direct the translocation of bacterial proteins into host cells through a type III protein secretion system, and this process occurs with both intracellular and extracellular bacteria (7, 8). In our studies, however, invA− and invE− mutants, which have a defective type III secretion system, sensitize target cells to lysis to the same extent as the parental wild-type SR11 strain. These observations suggest that the SPI-1 type III secretion pathway of S. enterica serovar Typhimurium is not involved in the translocation of Hsp 60 into the cytoplasm for processing and presentation. While a type I (hemolysin-based) secretion system has been described for serovar Typhimurium (9, 11) and could function as a novel antigen delivery pathway, it is also possible that a host cell-based mechanism plays a role in the movement of antigen into the cytoplasm. In this regard, a class I processing pathway has been described where antigenic proteins localized to a vacuolar compartment are transported into the cytosol via a leakage mechanism where they intersect the MHC class I pathway (22, 30, 39, 48). This pathway, also referred to as cross-presentation, has been demonstrated to operate in professional APCs active in phagocytosis (22, 40, 44). Furthermore, recent studies have indicated that the translocation of vacuolar proteins into the cytosol may involve ER-associated elements, including the Sec61 retrotranslocation machinery, thereby providing a mechanism by which vacuole-associated proteins gain access to the proteosome (16, 26). Our observations suggest the presence of a vacuole-to-cytoplasm translocation pathway that is operative in both phagocytic and nonphagocytic cell types, because both are capable of presenting the Qa-1b-restricted Salmonella epitopes and the processing is TAP and proteosome dependent (32, 33).
Previous studies with Salmonella-expressed defined antigens showed that only professional phagocytes, such as macrophages, but not epithelial cells, are capable of processing and presenting Salmonella-carried antigens for the generation of class Ia-restricted T-cell epitopes (13, 18, 19). Taken together with our data, we would argue that while class Ia molecules present relevant bacterial antigens processed only by dendritic cells and macrophages, class Ib molecules, and Qa-1 in particular, may broadly present bacterial antigens processed by host cells of multiple lineages. Our findings make feasible the possibility that class Ib molecules expressed on nonphagocytic cells play a greater role in the generation of protective immunity than heretofore observed.
Acknowledgments
This work was supported by NIH grants RO1AI42287 and RO1AI20922, by an Award from the Maryland Chapter of the National Arthritis Foundation (M.J.S.), and by NIH grants RO1AI32951 and USUHS R073-IN (E.S.M.).
Editor: F. C. Fang
REFERENCES
- 1.Ben-Zvi, A. P., and P. Goloubinoff. 2001. Review: mechanisms of disaggregation and refolding of stable protein aggregates by molecular chaperones. J. Struct. Biol. 135:84-93. [DOI] [PubMed] [Google Scholar]
- 2.Brocke, S., and H. Hahn. 1991. Heat-killed Listeria monocytogenes and L. monocytogenes-soluble antigen induce clonable CD4+ T lymphocytes with protective and chemotactic activities in vivo. Infect. Immun. 59:4531-4539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brown, M. L., P. E. Fields, and R. J. Kurlander. 1992. Metabolic requirements for macrophage presentation of Listeria monocytogenes to immune CD8 cells. J. Immunol. 148:555-561. [PubMed] [Google Scholar]
- 4.Brumell, J. H., A. J. Perrin, D. L. Goosney, and B. B. Finlay. 2002. Microbial pathogenesis: new niches for salmonella. Curr. Biol. 12:R15-R17. [DOI] [PubMed] [Google Scholar]
- 5.Buchmeier, N. A., and F. Heffron. 1990. Induction of Salmonella stress proteins upon infection of macrophages. Science 248:730-732. [DOI] [PubMed] [Google Scholar]
- 6.Bulut, Y., E. Faure, L. Thomas, H. Karahashi, K. S. Michelsen, O. Equils, S. G. Morrison, R. P. Morrison, and M. Arditi. 2002. Chlamydial heat shock protein 60 activates macrophages and endothelial cells through Toll-like receptor 4 and MD2 in a MyD88-dependent pathway. J. Immunol. 168:1435-1440. [DOI] [PubMed] [Google Scholar]
- 7.Collazo, C. M., and J. E. Galan. 1997. The invasion-associated type III system of Salmonella typhimurium directs the translocation of Sip proteins into the host cell. Mol. Microbiol. 24:747-756. [DOI] [PubMed] [Google Scholar]
- 8.Collazo, C. M., and J. E. Galan. 1997. The invasion-associated type-III protein secretion system in Salmonella—a review. Gene 192:51-59. [DOI] [PubMed] [Google Scholar]
- 9.Galan, J. E. 1996. Molecular genetic basis of Salmonella entry into host cells. Mol. Microbiol. 20:263-271. [DOI] [PubMed] [Google Scholar]
- 10.Galan, J. E. 2001. Salmonella interactions with host cells: type III secretion at work. Annu. Rev. Cell Dev. Biol. 17:53-86. [DOI] [PubMed] [Google Scholar]
- 11.Galan, J. E., and J. B. Bliska. 1996. Cross-talk between bacterial pathogens and their host cells. Annu. Rev. Cell Biol. 12:219-253. [DOI] [PubMed] [Google Scholar]
- 12.Galan, J. E., C. Ginocchio, and P. Costeas. 1992. Molecular and functional characterization of the salmonella invasion gene invA: homology of invA to members of a new protein family. J. Bacteriol. 174:4338-4349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gao, X. M., J. P. Tite, M. Lipscombe, J. S. Rowland, D. J. Ferguson, and A. J. McMichael. 1992. Recombinant Salmonella typhimurium strains that invade nonphagocytic cells are resistant to recognition by antigen-specific cytotoxic T lymphocytes. Infect. Immun. 60:3780-3789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ginocchio, C., J. Pace, and J. E. Galan. 1992. Identification and molecular characterization of a salmonella typhimurium gene involved in triggering the internalization of salmonellae into cultured epithelial cells. Proc. Natl. Acad. Sci. USA 89:5976-5980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ginocchio, C. C., S. B. Olmsted, C. L. Wells, and J. E. Galan. 1994. Contact with epithelial cells induces the formation of surface appendages on Salmonella typhimurium. Cell 76:717-724. [DOI] [PubMed] [Google Scholar]
- 16.Guermonprez, P., L. Saveanu, M. Kleijmeer, J. Davoust, P. Van Endert, and S. Amigorena. 2003. ER-phagosome fusion defines an MHC class I-presentation compartment in dendritic cells. Nature 425:397-402. [DOI] [PubMed] [Google Scholar]
- 17.Habich, C., K. Baumgart, H. Kolb, and V. Burkart. 2002. The receptor for heat shock protein 60 on macrophages is saturable, specific, and distinct from receptors for other heat shock proteins. J. Immunol. 168:569-576. [DOI] [PubMed] [Google Scholar]
- 18.Harding, C. V., and J. D. Pfeifer. 1994. Antigen expressed by Salmonella typhimurium is processed for class I major histocompatibility complex presentation by macrophages but not infected epithelial cells. Immunology 83:670-674. [PMC free article] [PubMed] [Google Scholar]
- 19.Harding, C. V., and R. Song. 1994. Phagocytic processing of exogenous particulate antigens by macrophages for presentation by class I MHC molecules. J. Immunol. 153:4925-4933. [PubMed] [Google Scholar]
- 20.Hardt, W. D., L. M. Chen, K. E. Schuebel, X. R. Bustelo, and J. E. Galan. 1998. S. typhimurium encodes an activator of Rho GTPases that induces membrane ruffling and nuclear responses in host cells. Cell 93:815-826. [DOI] [PubMed] [Google Scholar]
- 21.Harty, J. T., A. R. Tvinnereim, and D. W. White. 2000. CD8+ T cell effector mechanisms in resistance to infection. Annu. Rev. Immunol. 18:275-308. [DOI] [PubMed] [Google Scholar]
- 22.Heath, W. R., and F. R. Carbone. 2001. Cross-presentation in viral immunity and self-tolerance. Nat. Rev. Immunol. 1:126-134. [DOI] [PubMed] [Google Scholar]
- 23.Hennequin, C., F. Porcheray, A. Waligora-Dupriet, A. Collignon, M. Barc, P. Bourlioux, and T. Karjalainen. 2001. GroEL (Hsp60) of Clostridium difficile is involved in cell adherence. Microbiology 147:87-96. [DOI] [PubMed] [Google Scholar]
- 24.Hermann, E., W. J. Mayet, K. H. Meyer zum Buschenfelde, and B. Fleischer. 1992. MHC-unrestricted recognition of bacteria-infected target cells by human CD8+ cytotoxic T lymphocytes. Cell. Immunol. 143:253-260. [DOI] [PubMed] [Google Scholar]
- 25.Hoffman, P. S., and R. A. Garduno. 1999. Surface-associated heat shock proteins of Legionella pneumophila and Helicobacter pylori: roles in pathogenesis and immunity. Infect. Dis. Obstet. Gynecol. 7:58-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Houde, M., S. Bertholet, E. Gagnon, S. Brunet, G. Goyette, A. Laplante, M. F. Princiotta, P. Thibault, and D. Sacks. 2003. Phagosomes are competent organelles for antigen cross-presentation. Nature 425:402-406. [DOI] [PubMed] [Google Scholar]
- 27.Imani, F., and M. J. Soloski. 1991. Heat shock proteins can regulate expression of the Tla region-encoded class Ib molecule Qa-1. Proc. Natl. Acad. Sci. USA 88:10475-10479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jeannin, P., T. Renno, L. Goetsch, I. Miconnet, J. P. Aubry, Y. Delneste, N. Herbault, T. Baussant, G. Magistrelli, C. Soulas, P. Romero, J. C. Cerottini, and J. Y. Bonnefoy. 2000. OmpA targets dendritic cells, induces their maturation and delivers antigen into the MHC class I presentation pathway. Nat. Immunol. 1:502-509. [DOI] [PubMed] [Google Scholar]
- 29.Jones, B. D., and S. Falkow. 1996. Salmonellosis: host immune responses and bacterial virulence determinants. Annu. Rev. Immunol. 14:533-561. [DOI] [PubMed] [Google Scholar]
- 30.Kovacsovics-Bankowski, M., and K. L. Rock. 1995. A phagosome-to-cytosol pathway for exogenous antigens presented on MHC class I molecules. Science 267:243-246. [DOI] [PubMed] [Google Scholar]
- 31.Kurlander, R. J., S. M. Shawar, M. L. Brown, and R. R. Rich. 1992. Specialized role for a murine class I-b MHC molecule in prokaryotic host defenses. Science 257:678-679. [DOI] [PubMed] [Google Scholar]
- 32.Lo, W.-F., A. Woods, R. Cotter, A. DeCloux, E. S. Metcalf, and M. J. Soloski. 2000. Molecular mimicry mediated by MHC class Ib molecules following infection with gram-negative pathogens. Nat. Med. 6:215-218. [DOI] [PubMed] [Google Scholar]
- 33.Lo, W. F., H. Ong, E. S. Metcalf, and M. J. Soloski. 1999. T cell responses to gram-negative intracellular bacterial pathogens: a role for CD8+ T cells in immunity to Salmonella infection and the involvement of MHC class Ib molecules. J. Immunol. 162:5398-5406. [PubMed] [Google Scholar]
- 34.Monack, D. M., B. Raupach, A. E. Hromockyj, and S. Falkow. 1996. Salmonella typhimurium invasion induces apoptosis in infected macrophages. Proc. Natl. Acad. Sci. USA 93:9833-9838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ohashi, K., V. Burkart, S. Flohe, and H. Kolb. 2000. Cutting edge: heat shock protein 60 is a putative endogenous ligand of the toll-like receptor-4 complex. J. Immunol. 164:558-561. [DOI] [PubMed] [Google Scholar]
- 36.Pamer, E. G., C. R. Wang, L. Flaherty, K. F. Lindahl, and M. J. Bevan. 1992. H-2M3 presents a Listeria monocytogenes peptide to cytotoxic T lymphocytes. Cell 70:215-223. [DOI] [PubMed] [Google Scholar]
- 37.Pope, M., I. Kotlarski, and K. Doherty. 1994. Induction of Lyt-2+ cytotoxic T lymphocytes following primary and secondary Salmonella infection. Immunology 81:177-182. [PMC free article] [PubMed] [Google Scholar]
- 38.Reimann, J., and S. H. E. Kaufmann. 1997. Alternative antigen processing pathways in anti-infective immunity. Curr. Opin. Immunol. 9:462-469. [DOI] [PubMed] [Google Scholar]
- 39.Reis e Sousa, C., and R. N. Germain. 1995. Major histocompatibility complex class I presentation of peptides derived from soluble exogenous antigen by a subset of cells engaged in phagocytosis. J. Exp. Med. 182:841-851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rock, K. L. 1996. A new foreign policy: MHC class I molecules monitor the outside world. Immunol. Today 17:131-137. [DOI] [PubMed] [Google Scholar]
- 41.Rock, K. L., and A. L. Goldberg. 1999. Degradation of cell proteins and the generation of MHC class I-presented peptides. Annu. Rev. Immunol. 17:739-779. [DOI] [PubMed] [Google Scholar]
- 42.Russmann, H., H. Shams, F. Poblete, Y. Fu, J. E. Galan, and R. O. Donis. 1998. Delivery of epitopes by the Salmonella type III secretion system for vaccine development. Science 281:565-568. [DOI] [PubMed] [Google Scholar]
- 43.Shastri, N., S. Schwab, and T. Serwold. 2002. Producing nature's gene-chips: the generation of peptides for display by MHC class I molecules. Annu. Rev. Immunol. 20:463-493. [DOI] [PubMed] [Google Scholar]
- 44.Shen, Z., G. Reznikoff, G. Dranoff, and K. L. Rock. 1997. Cloned dendritic cells can present exogenous antigens on both MHC class I and class II molecules. J. Immunol. 158:2723-2730. [PubMed] [Google Scholar]
- 45.Speiser, D. E., M. F. Bachmann, M. J. Soloski, J. Forman, and P. S. Ohashi. 1998. Alloreactive cytotoxic T cells recognize minor transplantation antigens presented by major histocompatibility complex class Ib molecules. Transplantation 66:646-650. [DOI] [PubMed] [Google Scholar]
- 46.Steele-Mortimer, O., J. H. Brumell, L. A. Knodler, S. Meresse, A. Lopez, and B. B. Finlay. 2002. The invasion-associated type III secretion system of Salmonella enterica serovar Typhimurium is necessary for intracellular proliferation and vacuole biogenesis in epithelial cells. Cell. Microbiol. 4:43-54. [DOI] [PubMed] [Google Scholar]
- 47.Szalay, G., J. Hess, and S. H. Kaufmann. 1994. Presentation of Listeria monocytogenes antigens by major histocompatibility complex class I molecules to CD8 cytotoxic T lymphocytes independent of listeriolysin secretion and virulence. Eur. J. Immunol. 24:1471-1477. [DOI] [PubMed] [Google Scholar]
- 48.Szalay, G., C. H. Ladel, and S. H. Kaufmann. 1995. Stimulation of protective CD8+ T lymphocytes by vaccination with nonliving bacteria. Proc. Natl. Acad. Sci. USA 92:12389-12392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Thirumalai, D., and G. H. Lorimer. 2001. Chaperonin-mediated protein folding. Annu. Rev. Biophys. Biomol. Struct. 30:245-269. [DOI] [PubMed] [Google Scholar]
- 50.Vabulas, R. M., P. Ahmad-Nejad, C. da Costa, T. Miethke, C. J. Kirschning, H. Hacker, and H. Wagner. 2001. Endocytosed HSP60s use toll-like receptor 2 (TLR2) and TLR4 to activate the toll/interleukin-1 receptor signaling pathway in innate immune cells. J. Biol. Chem. 276:31332-31339. [DOI] [PubMed] [Google Scholar]
- 51.Weinstein, D. L., B. L. O'Neill, D. M. Hone, and E. S. Metcalf. 1998. Differential early interactions between Salmonella enterica serovar Typhi and two other pathogenic Salmonella serovars with intestinal epithelial cells. Infect. Immun. 66:2310-2318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yamaguchi, H., T. Osaki, H. Taguchi, T. Hanawa, T. Yamamoto, M. Fukuda, H. Kawakami, H. Hirano, and S. Kamiya. 1997. Growth inhibition of Helicobacter pylori by monoclonal antibody to heat-shock protein 60. Microbiol. Immunol. 41:909-916. [DOI] [PubMed] [Google Scholar]