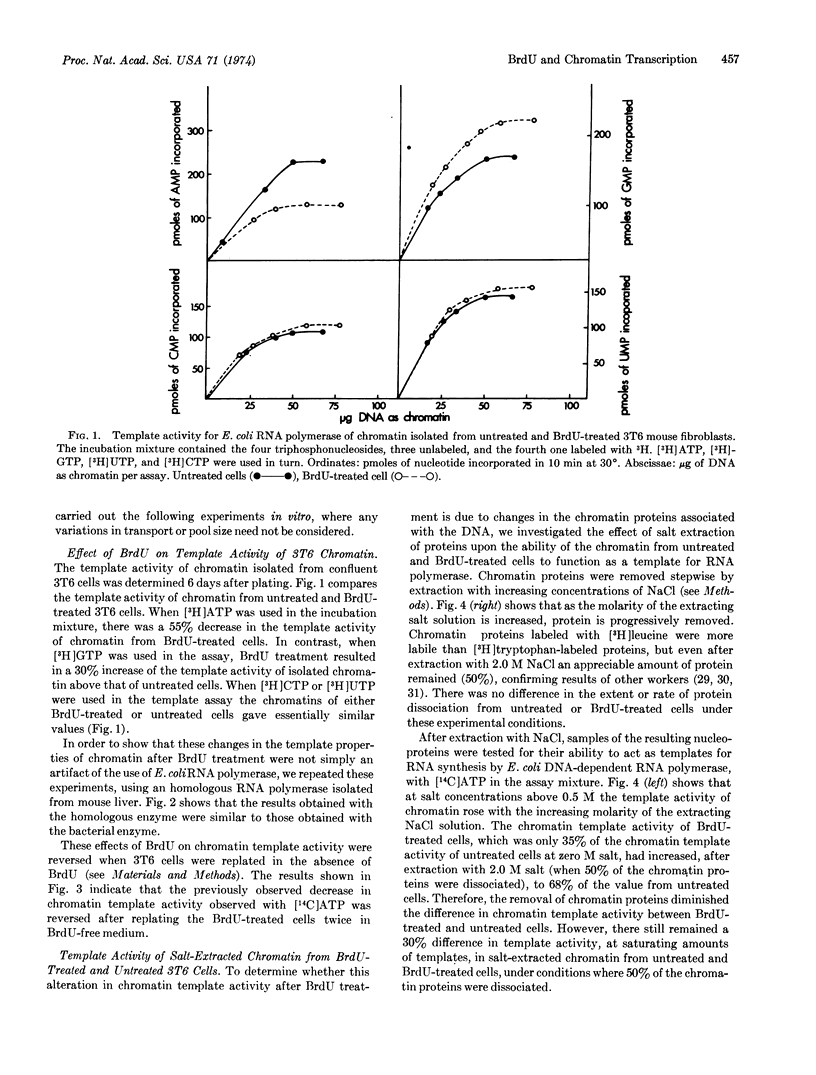
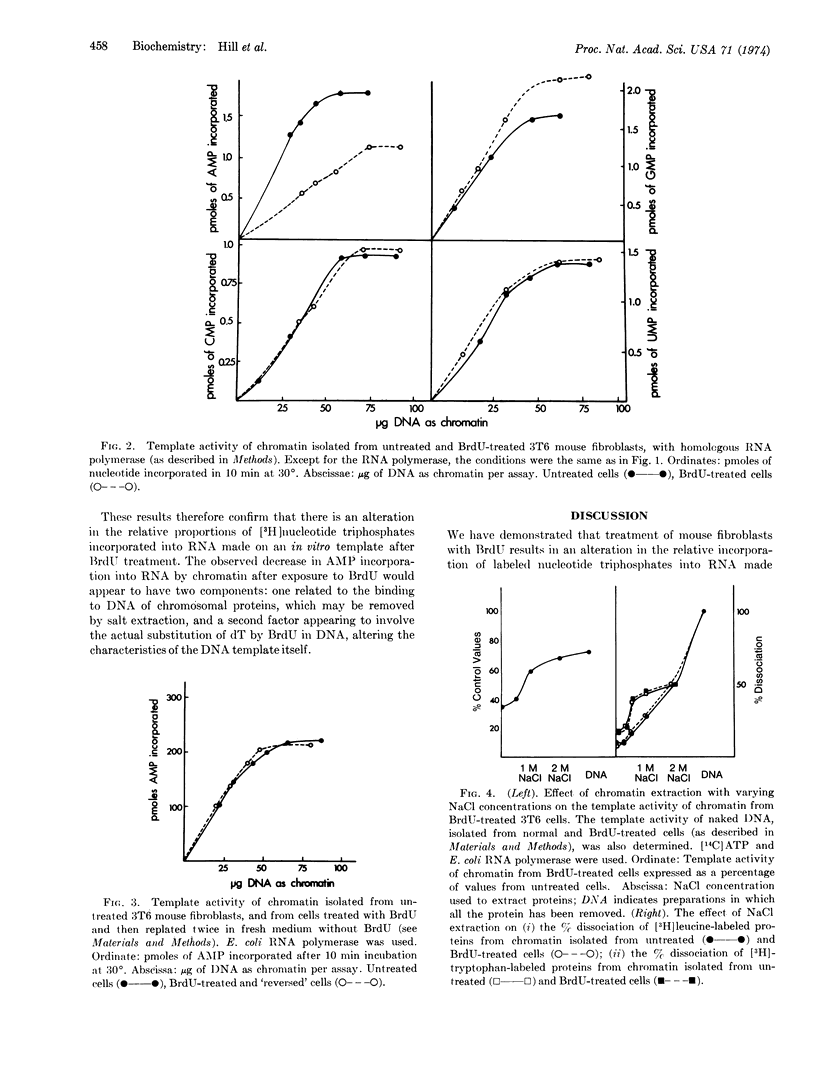
Abstract
3T6 mouse fibroblasts were grown in 5-bromodeoxyuridine (BrdU) so that approximately 20% of the thymine residues in DNA were replaced by BrdU. BrdU replacement caused an alteration in the relative incorporation of labeled nucleotide precursors into RNA. The RNA synthesized by cells grown in BrdU has a lower proportion of adenine and a higher guanine complement. This was shown for (a) nascent RNA made in vivo by confluent monolayers of cells in culture; (b) RNA synthesized in vitro on a chromatin template with either homologous or heterologous RNA polymerase; and (c) RNA synthesized in vitro on a DNA template with a highly purified RNA polymerase. The product was completely digested by RNase. The relative decrease in the incorporation of adenine into RNA was reserved when BrdU-treated cells were allowed to proliferate in BrdU-free medium.
Keywords: cell differentiation, template activity, RNA polymerase, DNA
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abbott J., Holtzer H. The loss of phenotypic traits by differentiated cells, V. The effect of 5-bromodeoxyuridine on cloned chondrocytes. Proc Natl Acad Sci U S A. 1968 Apr;59(4):1144–1151. doi: 10.1073/pnas.59.4.1144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BURTON K. A study of the conditions and mechanism of the diphenylamine reaction for the colorimetric estimation of deoxyribonucleic acid. Biochem J. 1956 Feb;62(2):315–323. doi: 10.1042/bj0620315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHAMBERLIN M., BERG P. Deoxyribo ucleic acid-directed synthesis of ribonucleic acid by an enzyme from Escherichia coli. Proc Natl Acad Sci U S A. 1962 Jan 15;48:81–94. doi: 10.1073/pnas.48.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHEONG L., RICH M. A., EIDINOFF M. L. Introduction of the 5-halogenated uracil moiety into deoxyribonucleic acid of mammalian cells in culture. J Biol Chem. 1960 May;235:1441–1447. [PubMed] [Google Scholar]
- Coleman A. W., Coleman J. R., Kankel D., Werner I. The reversible control of animal cell differentiation by the thymidine analog, 5-bromodeoxyuridine. Exp Cell Res. 1970 Feb;59(2):319–328. doi: 10.1016/0014-4827(70)90606-3. [DOI] [PubMed] [Google Scholar]
- Coleman J. R., Coleman A. W., Hartline E. J. A clonal study of the reversible inhibition of muscle differentiation by the halogenated thymidine analog 5-bromodeoxyuridine. Dev Biol. 1969 Jun;19(6):527–548. doi: 10.1016/0012-1606(69)90036-0. [DOI] [PubMed] [Google Scholar]
- Davidson R. L., Bick M. D. Bromodeoxyuridine dependence--a new mutation in mammalian cells. Proc Natl Acad Sci U S A. 1973 Jan;70(1):138–142. doi: 10.1073/pnas.70.1.138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farber J., Rovera G., Baserga R. Template activity of chromatin during stimulation of cellular proliferation in human diploid fibroblasts. Biochem J. 1971 Apr;122(2):189–195. doi: 10.1042/bj1220189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasaryan K. G., Andreeva N. B. Changes in the template activity of chromatin from pigeon erythrocytes and erythroblasts induced by partial salt deproteinization. FEBS Lett. 1972 Nov 1;27(2):263–266. doi: 10.1016/0014-5793(72)80636-7. [DOI] [PubMed] [Google Scholar]
- KAJIWARA K., MUELLER G. C. MOLECULAR EVENTS IN THE REPRODUCTION OF ANIMAL CELLS. 3. FRACTIONAL SYNTHESIS OF DEOXYRIBONUCLEIC ACID WITH 5-BROMODEOXYURIDINE AND ITS EFFECT ON CLONING EFFICIENCY. Biochim Biophys Acta. 1964 Nov 15;91:486–493. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lin S. Y., Riggs A. D. Lac operator analogues: bromodeoxyuridine substitution in the lac operator affects the rate of dissociation of the lac repressor. Proc Natl Acad Sci U S A. 1972 Sep;69(9):2574–2576. doi: 10.1073/pnas.69.9.2574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowy D. R., Rowe W. P., Teich N., Hartley J. W. Murine leukemia virus: high-frequency activation in vitro by 5-iododeoxyuridine and 5-bromodeoxyuridine. Science. 1971 Oct 8;174(4005):155–156. doi: 10.1126/science.174.4005.155. [DOI] [PubMed] [Google Scholar]
- More I. A., Paul J. Template activity and electron microscopic appearance of salt-extracted chromatin. Exp Cell Res. 1973 Jan;76(1):79–86. doi: 10.1016/0014-4827(73)90421-7. [DOI] [PubMed] [Google Scholar]
- O'Meara A. R., Herrmann R. L. A modified mouse liver chromatin preparation displaying age-related differences in salt dissociation and template ability. Biochim Biophys Acta. 1972 May 29;269(3):419–427. doi: 10.1016/0005-2787(72)90129-3. [DOI] [PubMed] [Google Scholar]
- Ohlenbusch H. H., Olivera B. M., Tuan D., Davidson N. Selective dissociation of histones from calf thymus nucleoprotein. J Mol Biol. 1967 Apr 28;25(2):299–315. doi: 10.1016/0022-2836(67)90143-x. [DOI] [PubMed] [Google Scholar]
- PATTERSON M. S., GREENE R. C. MEASUREMENT OF LOW ENERGY BETA-EMITTERS IN AQUEOUS SOLUTION BY LIQUID SCINTILLATION COUNTING OF EMULSIONS. Anal Chem. 1965 Jun;37:854–857. doi: 10.1021/ac60226a017. [DOI] [PubMed] [Google Scholar]
- Paul J., Gilmour R. S. Organ-specific restriction of transcription in mammalian chromatin. J Mol Biol. 1968 Jul 14;34(2):305–316. doi: 10.1016/0022-2836(68)90255-6. [DOI] [PubMed] [Google Scholar]
- Roeder R. G., Rutter W. J. Specific nucleolar and nucleoplasmic RNA polymerases. Proc Natl Acad Sci U S A. 1970 Mar;65(3):675–682. doi: 10.1073/pnas.65.3.675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rovera G., Baserga R. Early changes in the synthesis of acidic nuclear proteins in human diploid fibroblasts stimulated to synthesize DNA by changing the medium. J Cell Physiol. 1971 Apr;77(2):201–211. doi: 10.1002/jcp.1040770211. [DOI] [PubMed] [Google Scholar]
- SCOTT J. F., FRACCASTORO A. P., TAFT E. B. Studies in histochemistry. I. Determination of nucleic acids in microgram amounts of tissue. J Histochem Cytochem. 1956 Jan;4(1):1–10. doi: 10.1177/4.1.1. [DOI] [PubMed] [Google Scholar]
- STOCKDALE F., OKAZAKI K., NAMEROFF M., HOLTZER H. 5-BROMODEOXYURIDINE: EFFECT ON MYOGENESIS IN VITRO. Science. 1964 Oct 23;146(3643):533–535. doi: 10.1126/science.146.3643.533. [DOI] [PubMed] [Google Scholar]
- Tsuboi A., Baserga R. Effect of 5-bromo-2-deoxyuridine on transport of deoxyglucose and cycloleucine in 3T6 fibroblasts. Cancer Res. 1973 Jun;33(6):1326–1330. [PubMed] [Google Scholar]
- Tsuboi A., Baserga R. Synthesis of nuclear acidic proteins in density-inhibited fibroblasts stimulated to proliferate. J Cell Physiol. 1972 Aug;80(1):107–118. doi: 10.1002/jcp.1040800112. [DOI] [PubMed] [Google Scholar]
- Weintraub H., Campbell G. L., Holtzer H. Identification of a developmental program using bromodeoxyuridine. J Mol Biol. 1972 Sep 28;70(2):337–350. doi: 10.1016/0022-2836(72)90543-8. [DOI] [PubMed] [Google Scholar]



