Abstract
Paraquat (PQ) is one of the most widely used herbicides in the world, which has a good occupational safety record when used properly. While, it presents high mortality index after intentional exposure. Accidental deaths and suicides from PQ ingestion are relatively common in developing countries with an estimated 300,000 deaths occurring in the Asia–Pacific region alone each year, and there are no specific antidotes. Good predictors of outcome and prognosis may be plasma and urine testing within the first 24 h of intoxication. A fluorescence enhancement of approximately 30 times was seen following addition of PQ to a solution of the supramolecular compound 2MB@CB[8], which comprised two methylene blue (MB) molecules within one cucurbit[8]uril (CB[8]) host molecule. The fluorescence intensity was linearly proportional to the amount of PQ added over the concentration range 2.4 × 10−10 M–2.5 × 10−4 M. The reaction also occurred in living cells and within live mice.
Paraquat (PQ, Fig. 1) is one of the most widely used herbicides in the world. Its distinctive features — including non-selectivity; rapid action, with killing of green plant tissue on contact; plus its inactivation on contact with soil — makes PQ an essential tool in the development of no-till farming, being one of few chemical options which avoids the evolved glyphosate resistance problem1,2. Although PQ has a good occupational safety record when used properly, it presents pretty high mortality index after intentional exposure. Accidental deaths and suicides from PQ ingestion are relatively common in developing countries3,4,5,6 with an estimated 300,000 deaths occurring in the Asia–Pacific region alone each year7,8 and there are no specific antidotes. No direct interaction between PQ and plasma protein can be observed after ingestion. The PQ concentrations in plasma reach the highest level in two hours after ingestion, accompanied by rapid distribution to the kidneys, liver, lungs, muscle, thyroid, etc. following which the plasma PQ level falls very quickly9. Good predictors of outcome and prognosis may be plasma and urine testing within the first 24 h of intoxication3,9. Patients whose plasma PQ levels are less than 2.0, 0.6, 0.3, 0.16, and 0.1 mg/L at 4, 6, 10, 16, and 24 h, respectively, are likely to survive. Plasma levels exceeding 2 mg/L are likely to be fatal in most cases10. An error of an hour or two in the estimate of ingestion time can move a patient from the 30% to the 70% survival curve11. Unfortunately, it takes four hours to obtain reliable analytical results on plasma detection11, and three days12 to observe obviously lesions in lung X-ray. The dithionite urine test for confirming PQ ingestion is of doubtful prognostic value and can be misleading13. All these are quite bothering in clinical diagnosis.
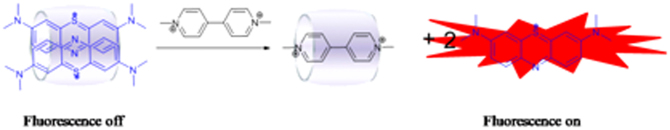
Figure 1. Schematic representation of fluorescence response of 2MB@CB[8] to PQ.
Many detection methods for PQ have been developed, such as spectroscopy14, HPLC15, liquid chromatography-mass spectrometry16 and immunoassay17. Unfortunately, most procedures are time-consuming and require trained staff. This is exacerbated by the common need for purified PQ samples, a difficult problem with such a small quaternary nitrogen salt. Development of a simple but reliable detection method for a rapid and sensitive determination of PQ, both qualitative and quantitative, remains a considerable challenge. A possible approach is to exploit the high sensitivity fluorescence enhancement phenomenon. However, to the best of our knowledge, few fluorescence enhancement methods18 for detecting of PQ have been reported.
As a relatively new class of host molecules, cucurbit[n]uril (CB[n], where n = 5–12) have attracted great attention in recent years. Among these, CB[8] has been studied extensively and applied in fields such as molecular machines, molecular switches and molecular wires19,20,21. It was noted that the self-assembly of PQ and derivatives with CB[8]22,23,24,25, yielding PQ@CB[8], occurs readily, owing to the two positive charges carried by PQ, with a binding constant of approximately 1.1 × 105 M−1. However, an organic dye such as methylene blue (MB) carries only a single positive charge, and hence must form a head to tail dimer within the CB[8] cavity to yield 2MB@CB[8]26. The fluorescence of MB was inhibited (“off ”) due to the dimerization. Consequently when PQ was added into a solution of the supramolecular compound 2MB@CB[8], MBs were replaced by PQ immediately, and MB was ejected from the CB[8] cavity. As these released MBs exist as single molecules in the solution, this results in a strong fluoresence enhancement which can be readily observed (the fluorescence is “on”), as illustrated in Fig. 1.
Results
As shown in Fig. 2a, following addition of PQ to 2MB@CB[8] in tris-HCl buffer solution (pH = 7.14), a strong fluorescence emission (up to 30-fold enhancement) which peaked at 682 nm was observed, when excited at 620 nm. The inset in Fig. 2a shows the appearance of the cuvettes when irradiated by a 625 nm laser pen. A pink line is seen in the cuvette containing 2MB@CB[8] + PQ whilst no fluorescence can be observed when only 2MB@CB[8] is present. The effect is further demonstrated by the UV-Vis spectra shown in Fig. 2b. As increasing amounts of PQ were added, the spectra of 2MB@CB[8] showed a significant hypochromic effect accompanied by the appearance of an isosbestic point at 630 nm and a new peak at 660 nm. This is the characteristic absorbance of free MB, demonstrating that 2MB@CB[8] had a very strong interaction with PQ, leading to the formation of PQ@CB[8] and free MB in solution.
Figure 2.
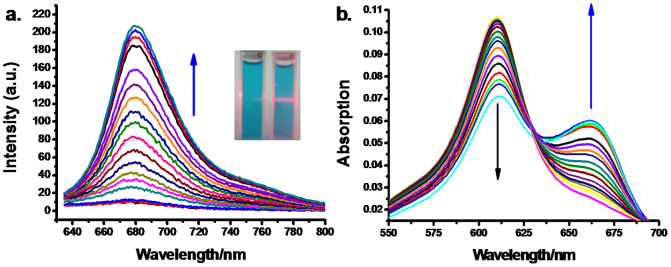
(a) Fluorescence spectral changes of 2MB@CB [8] (5 μM) upon addition of PQ (0–250 μM). Inset: Pictures taken under 625 nm laser pen irradiation, left 2MB@CB[8] + PQ, right 2MB@CB[8] alone. (b) The absorption spectra of 2MB@CB[8] (1 μM) with increasing addition of PQ (0–25 μM).
To assess the selectivity of the detection method, the probe 2MB@CB[8] was exposed to 50 equivalents of potentially competing species — such as a mixture of inorganic salts Na+, K+, Mg2+, different herbicides such as aquacide, acetochlor, (2,4-dichlorophenyloxy)acetic acid (2,4-D), dichlorovos, glyphosate, haloxyfop, methomyl, omethoate and quizalofop-p-ethyl. Most of these potentially competing species gave little fluorescence enhancement even though present in large excess (Supporting Information Fig. S1). This indicates that the method has a high selectivity for PQ detection.
It was further noted that the fluorescence intensities were linearly proportional to the amount of PQ added, over the range 2.4 × 10−10 M–2.5 × 10−4 M (Supporting Information Fig. S2), and that a relative standard deviation (RSD) of less than 2% was found for 5 repeat observations, suggesting this method can be utilized as good predictors of outcome and prognosis within the first 24 h of intoxication9,10,11. The detection limit was as sensitive as 2.4 × 10−10 M (0.06 μg/L), which is lower than the current PQ residue tolerance (no more than 0.05 mg/kg) for fruits, vegetables and drinking water in the EU and the US20,21,22. As a result, the specific interaction between 2MB@CB[8] and PQ can be employed for the quantitative detection of trace levels of PQ.
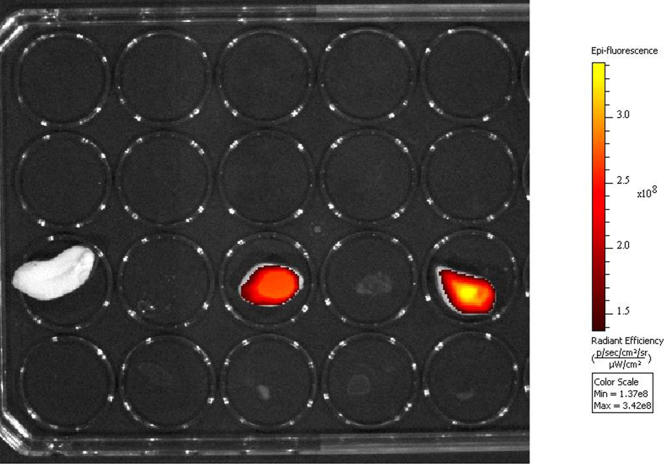
To demonstrate the practical utility of the PQ detecting method, two peanut kernels were separately immersed into 5 × 10−4 M and 5 × 10−3 M PQ solutions. As a negative control, a third peanut kernel was immersed into drinking water. After 10 min, the peanut kernels were placed into the wells of a 96-well plate. Then 2MB@CB[8] (200 μL, 5 μM) was added to each well. Any resulting fluorescence was measured using a microplate reader, with fluorescence filters providing an excitation wavelength of 640 nm and an emission wavelength of 680 nm. As shown in Fig. 3, the higher the PQ concentration to which the peanut kernels were exposed, the stronger the fluorescence exhibited, with the control showing no fluorescence.
Figure 3. Fluorescence, with background deducted, of the peanut kernels after exposure to 2MB@CB[8] (200 μL, 5 μM) observed in a 96-well plate.
Left, negative control; middle and right, after immersion into 5 × 10−4 M and 5 × 10−3 M of PQ solution, respectively.
To provide another everyday example, fresh potato (50 g) purchased from a local market was immersed in 100 mL drinking water for 30 min. The mud was filtered off, and the filtrate used to dissolve PQ. The same trend was observed on the calibration cures between the PQ samples prepared by the drinking water before and after potato washing (Fig. S3a). To check how many washings were required to remove PQ residues on polluted potatoes, 50 g potatoes were soaked in the solution of PQ (100 mL, 5 × 10−3 M) for 30 min, then the potatoes were washed with 10 mL drinking water. 10 μL of the washing water was added into the solution of 2MB@CB[8] (3 mL, 5 μM), with the corresponding fluorescence intensities being as shown (Fig. S3b). The contaminated potatoes were free of PQ after third washing. The several investigations noted above demonstrated the method can be used in various everyday situations.
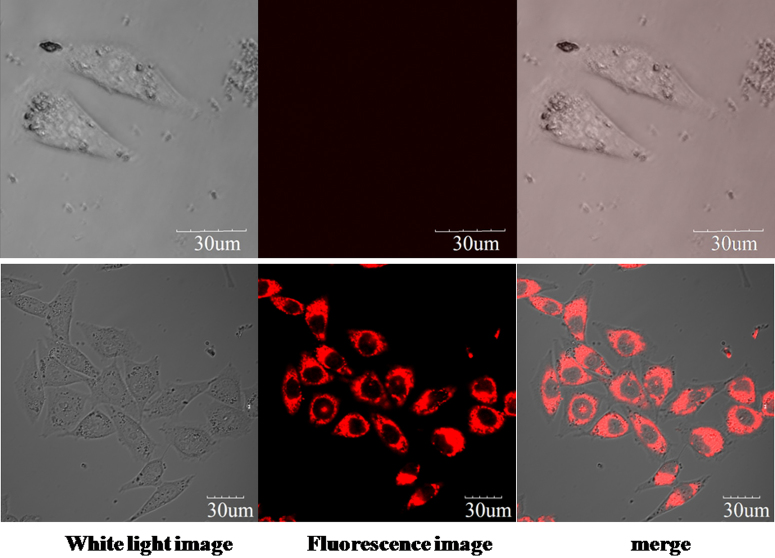
Microscopy imaging studies were carried out to examine application of the method to living cells. MCF7 cells were incubated with 10 μM 2MB@CB[8] for 30 min, and then washed with phosphate-buffered saline (PBS) three times. Following this, cells were further incubated for 90 min with 25 μM PQ. Fluorescence imaging was carried out using a spectral confocal microscope (Olympus, FV1000). As seen in Fig. 4, staining results indicate that both 2MB@CB[8] and PQ enter the cells, where they react: the difference between cells with and without PQ treatment being dramatic.
Figure 4. Confocal fluorescence imaging of MCF7 cells incubated with 10 μM 2MB@CB[8] for 30 min, then PBS washed.
Cells appeared as in the upper panel. Some cells were further incubated with 25 μM PQ for 90 min, and then appeared as in the lower panel (excited at 635 nm and collected at 665–695 nm). Scale bar represents 30 μm.
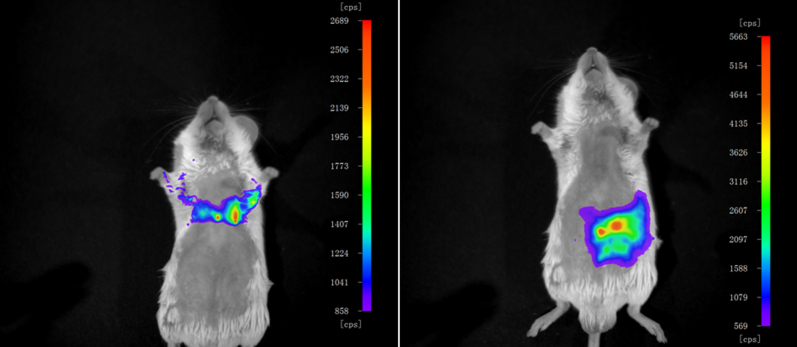
Real-time in vivo fluorescence imaging of PQ in the living mice was also carried out, using a NightOWL II LB 983 system equipped with a NC 100 CCD deep-cooled camera (Berthold Technologies, Bad Wildbad, Germany). Before the study, the mice were sedated with isoflurane (2% in air) and shaved. PQ was introduced by thoracic (Fig. 5 left) or intraperitoneal injection (Fig. 5 right). Three minutes later, 2MB@CB[8] was injected at the same sites and fluorescence images obtained with the animals in a prone position. It is apparent from Fig. 5 that PQ can be readily tracked by using 2MB@CB[8] in living mice.
Figure 5. Fluorescence imaging of PQ in living mice.
Excitation wavelengths of 580 ± 20 nm were used and the fluorescence emission was detected at 680 ± 30 nm in a Berthold chamber. Injection sites were thoracic (left) or intraperitoneal (right).
Discussion
To further confirm the above mentioned proposed reaction process, electrospray ionization mass spectrometry (ESI-MS) was carried out on the system (Supporting Information Fig. S4). The MS spectrum gave one positively charged peak at m/z 948.4 (calc. for [2MB + CB[8]]2+, 948.3) for 2MB@CB[8] alone. To avoid any influence of free CB[8] on the ESI-MS, 3 equiv. of MB were used in the detection. After addition of PQ, an additional peak at m/z 756.96 (calc. for [PQ + CB[8]-2Cl]2+, 757.2) was observed, providing strong evidence for the formation of a host–guest complex PQ@CB[8]. The supramolecular interaction of PQ with 2MB@CB[8] was further studied by1H NMR experiments (Supporting Information Fig. S5). Upon introduction of PQ into an aqueous solution of 2MB@CB[8], typical peaks of the free MB molecule appeared. This result indicates that the MB molecules were ejected from the cavity of CB[8] as illustrated in Fig. 1.
Meanwhile, the phenomenon appeared robust during MCF7 cell staining, since if MCF7 cells were first incubated with PQ (25 μM for 30 min), washed with PBS three times, then incubated with 2MB@CB[8] (10 μM for 90 min), similar results were obtained (Supporting Information Fig. S6). In particular, 2MB@CB[8] sensor showed a good selectivity in living cells towards other competitive binding analytes like aquacide and glyphosate etc (Supporting Information Fig. S7). The staining results thus demonstrated that 2MB@CB[8] could enter living cells and selectively demonstrate PQ in situ.
According to the literature28, no significant cytotoxicity was observed for CB[8] alone within its solubility range. To confirm the cytotoxicity of the 2MB@CB[8] sensor to living cells, MTT assays were carried out and low toxicity of 2MB@CB[8] in vivo was acquired for the 12 h measurement (Supporting Information Fig. S8), suggesting that this probe would function in a biological environment.
Methods
All the solvents were of analytic grade, and the water used was double distilled. Cucurbit[8]uril was synthesized by a published method27. Paraquat (PQ) and methylene blue (MB) were purchased from Sigma Aldrich.1H NMR spectra were recorded on a Varian Inova-400 spectrometer with chemical shifts reported as ppm. Mass spectrometric data were obtained on a Q-Tof MS spectrometer (Micromass, Manchester, England). Absorption spectra were measured on a Perkin-Elmer Lambda 35 UV-Vis spectrophotometer. Fluorescence measurements were performed on a Varian Cary Eclipse Fluorescence Spectrophotometer. Cell imaging measurements were obtained with a spectral confocal microscope, Olympus, FV1000. Real-time in vivo fluorescence imaging of PQ in the living mice were acquired using a NightOWL II LB 983 system equipped with a NC 100 CCD deep-cooled camera (Berthold Technologies, Bad Wildbad, Germany). Mouse experiments were performed in compliance with the relevant laws and institutional guidelines, and were approved by Dalian University of Technology.
MCF7 cells were cultured in DEME (Invitrogen) supplemented with 10% FCS (Invitrogen). Prior to staining experiments, cultured cells grown on a special confocal microscope dish were fixed by precooled methanol (−20°C) for 15 min, and washed twice with PBS for 5 min. Then the pretreated MCF7 cells were incubated with 10 μM 2MB@CB[8] for 30 min, then PBS washed. Some cells were further incubated with 25 μM PQ for 90 min before imaging. For cells without any PQ treatment were taken as a staining control.
Author Contributions
S.G.S. supervised and interpreted the research and wrote the manuscript. F.S.L. and F.Y.L. performed the measurements. J.T.W. performed cell staining. X.J.P. helped with interpreted data and wrote the manuscript. All authors discussed the results and commented on the manuscript.
Supplementary Material
Fluorescence detecting of paraquat using host-guest chemistry with cucurbit[8]uril
Acknowledgments
This work was financially supported by State Key Laboratory of Fine Chemicals, Dalian University of Technology, the Education Ministry and National Natural Science Foundation of China (Nos. 21306019, 21072024, 21272030), National Key Technology R&D Program (2011BAE07B06) and National Basic Research Program of China (2009CB724706).
References
- Beckie H. Herbicide-resistant weed-management: focus on glyphosate. Pest Manag. Sci. 67, 1037–1048 (2011). [DOI] [PubMed] [Google Scholar]
- Thomas W. et al. Glyphosate-resistant horseweed (Conyza canadensis) control using glyphosate-, paraquat-, and glufosinate-based herbicide programs. Weed Technol. 22, 16–21 (2008). [Google Scholar]
- Lin J., Leu M., Liu Y. & Chen G. A prospective clinical trial of pulse therapy with glucocorticoid and cyclophosphamide in moderate to severe paraquat-poisoned patients. Am. J. Respir. Crit. Care Med. 159, 357–360 (1999). [DOI] [PubMed] [Google Scholar]
- Yen T. et al. Spectrum of corrosive esophageal injury after intentional paraquat ingestion. Am. J. emerg. Med. 28, 728–733 (2010). [DOI] [PubMed] [Google Scholar]
- Wilks M. et al. Improvement in survival after paraquat ingestion following introduction of a new formulation in Sri Lanka. PLoS Medicine 5, 250–259 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seok S., Kim Y., Gil H., Song H. & Hong S. The time between paraquat ingestion and a negative dithionite urine test in an independent risk factor for death and organ failure in acute paraquat intoxication. J. Korean Med. Sci. 27, 993–998 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeyaratnam J. Acute pesticide poisoning: a major global health problem. World Health Stat. Q. 43, 139–144 (1990). [PubMed] [Google Scholar]
- Eddleston M. & Phillips M. Self poisoning with pesticides. BMJ. 328, 42–44 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gawarammana I. & Buckley N. Medical management of paraquat ingestion. Br. J. Clin. Pharmacol. 72, 745–757 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gil H., Kang M., Yang J., Lee E. & Hong S. Association between plasma paraquat level and outcome of paraquat poisoning in 375 paraquat poisoning patients. Clin. Toxicol. 46, 515–518 (2008). [DOI] [PubMed] [Google Scholar]
- Hart T., Nevitt A. & Whitehead A. A new statistical approach to the prognostic significance of plasma concentrations. Lancet 2, 1222–1223 (1984). [DOI] [PubMed] [Google Scholar]
- Sittipunt C. Paraquat Poisoning. Respiratory Care. 50, 383–385 (2005). [PubMed] [Google Scholar]
- Widdop B. Detection of paraquat in urine. BMJ. 4, 1135 (1976). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo T., Lin D., Liu R., Moriya F. & Hashimoto Y. Spectra interference between diquat and paraquat by second derivative spectrophotometry. Forensic Sci. Int. 121, 134–139 (2001). [DOI] [PubMed] [Google Scholar]
- Fuke C. et al. Analysis of paraquat, diquat and two diquat metabolites in biological materials by high-performance liquid chromatography. Leg. Med. (Tokyo) 4, 156–163 (2002). [DOI] [PubMed] [Google Scholar]
- Grey L., Nguyen B. & Yang P. Liquid chromatography-electrospray ionization isotope dilution mass spectrometry analysis of paraquat and diquat using conventional and multilayer solid-phase extraction cartridges. J. Chromatogr. A. 958, 25–33 (2002). [DOI] [PubMed] [Google Scholar]
- Mastichiadis C. et al. Simultaneous determination of pesticides using a four-band disposable optical capillary immunosensor. Anal. Chem. 74, 6064–6072 (2002). [DOI] [PubMed] [Google Scholar]
- Sendra B., Panadero S. & Gómez-Hens A. Selective kinetic determination of paraquat using long-wavelength fluorescence detection. J. Agric. Food Chem. 47, 3733–3737 (1999). [DOI] [PubMed] [Google Scholar]
- Sindelar V., Silvi S., Parker S., Sobransingh D. & Kaifer A. Proton and electron transfer control of the position of cucurbit[n]uril wheels in pseudorotaxanes. Adv. Funct. Mater. 17, 694–701 (2007). [Google Scholar]
- Frampton M. & Anderson H. Insulated molecular wires. Angew. Chem., Int. Ed. 46, 1028–1064 (2007). [DOI] [PubMed] [Google Scholar]
- Yui N. & Ooya T. Molecular mobility of interlocked structures exploiting new functions of advanced biomaterials. Chem. Eur. J. 12, 6730–6737 (2006). [DOI] [PubMed] [Google Scholar]
- Sun S. et al. Synthesis and DNA photocleavage study of Ru(bpy)32+-(CH2)n-MV2+ complexes. Dalton Trans. 39, 4411–4416 (2010). [DOI] [PubMed] [Google Scholar]
- Sun S. et al. Redox-induced Ru(bpy)32+-methylviologen radical formation and its dimerization in cucurbit[8]uril. Phys. Chem. Chem. Phys. 13, 570–575 (2011). [DOI] [PubMed] [Google Scholar]
- Sun S., Gao W., Liu F., Fan J. & Peng X. Study of an unusual charge-transfer inclusion complex with NIR absorption and its application for DNA photocleavage. J. Mater. Chem. 20, 5888–5892 (2010). [Google Scholar]
- Jeon W., Kim H., Lee C. & Kim K. Control of the stoichiometry in host-guest complexation by redox chemistry of guests: Inclusion of methylviologen in cucurbit[8]uril. Chem. Commun. 1828–1829 (2002). [DOI] [PubMed] [Google Scholar]
- Montes-Navajas P., Corma A. & Garcia H. Complexation and Fluorescence of Tricyclic Basic Dyes Encapsulated in Cucurbiturils. ChemPhysChem. 9, 713–720 (2008). [DOI] [PubMed] [Google Scholar]
- Kim J. et al. New Cucurbituril Homologues: Syntheses, Isolation, Characterization, and X-ray Crystal Structures of Cucurbit[n]uril (n = 5, 7, and 8). J. Am. Chem. Soc. 122, 540–541 (2000). [Google Scholar]
- Uzunova V., Cullinane C., Brix K., Nau W. & Day A. Toxicity of cucurbit[7]uril and cucurbit[8]uril: an exploratory in vitro and in vivo study. Org. Biomol. Chem. 8, 2037–2042 (2010). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Fluorescence detecting of paraquat using host-guest chemistry with cucurbit[8]uril