Abstract
Positron Emission Tomography (PET) experienced accelerated development and has become an established method for medical research and clinical routine diagnostics on patient individualized basis. Development and availability of new radiopharmaceuticals specific for particular diseases is one of the driving forces of the expansion of clinical PET. The future development of the 68Ga-radiopharmaceuticals must be put in the context of several aspects such as role of PET in nuclear medicine, unmet medical needs, identification of new biomarkers, targets and corresponding ligands, production and availability of 68Ga, automation of the radiopharmaceutical production, progress of positron emission tomography technologies and image analysis methodologies for improved quantitation accuracy, PET radiopharmaceutical regulations as well as advances in radiopharmaceutical chemistry. The review presents the prospects of the 68Ga-based radiopharmaceutical development on the basis of the current status of these aspects as well as wide range and variety of imaging agents.
Keywords: positron emission tomography, 68Ga, radiochemistry, coordination chemistry, conjugation chemistry.
Introduction
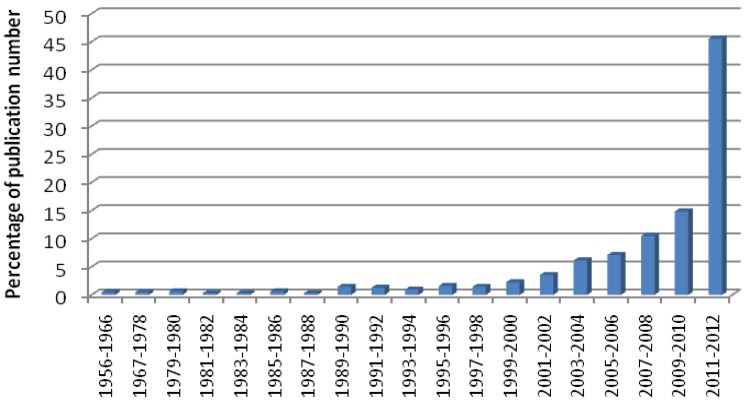
The number of publications devoted to 68Ga-radiopharmaceutical basic and clinical research has increased drastically during last two years. Rough estimation demonstrates that the number of 68Ga-related scientific articles published during 2011-2012 stands for over 45% of all publications since 1956 (Figure 1). Rather crucial changes and progress occurred during this short period and they influenced the basic perceptions and thus speculation about the nearest 5-10 year future of the field. Nuclear medicine applications and in particular positron emission tomography (PET) have experienced accelerated development.
Fig 1.
The fraction (%) of 68Ga-related publications per two successive years (except for the periods of 1956-1966 and 1967-1978) appeared from 1956 through the end of 2012. PubMed, Scopus, SciFinder Scholar, Web of Science, and Beilstein databases as well as references from published articles were used for the literature search and assay.
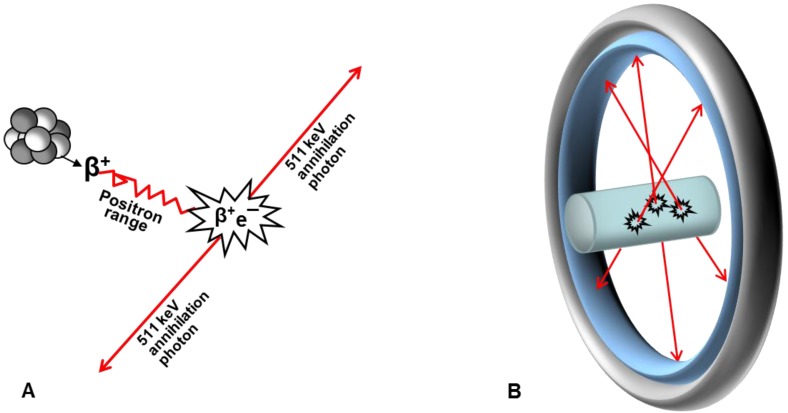
The major advantage of PET is that it is not only enables in vivo visualization of physiological processes on molecular level in real time, but it also quantifies them by measuring regional concentration of the radiation source. PET employs imaging agents comprising positron-emitting radionuclides (Figure 2A), and scanners detecting radiation (Figure 2B). Positron scan registration is based on the 180±0.25° correlation of the 511 keV photons arising from the annihilation of positrons with electrons and detection by means of two opposing counters recording only coincident events 1. The registered events are reconstructed into images representing spatial distribution of the radioactivity in a subject. Positron emission is an attribute of neutron deficient nuclides requiring artificial production generally by cyclotrons. However 68Ga is obtained from a 68Ge/68Ga generator system which is simple in use and relatively inexpensive.
Fig 2.
Schematic representation of PET principle. (A) After travelling in tissue (positron range) positron looses energy and annihilates with electron resulting in two 511 keV annihilation photons travelling in opposite directions; (B) Annihilation photons are registered externally by radiodetectors consisting of scintillation crystals and photomultiplier tubes and assembled in a ring. Only photons that are registered in coincidence are used for image reconstruction.
PET has become an established method for medical research and clinical routine diagnostics. Development and availability of new radiopharmaceuticals specific for particular diseases is one of the driving forces of the expansion of clinical nuclear medicine providing early personalized diagnosis and efficient therapy. The future development of 68Ga-radiopharmaceuticals must be put in the context of several aspects such as role of PET in nuclear medicine, unmet medical needs, identification of new biomarkers, targets and corresponding ligands, production and availability of 68Ga, automation of the radiopharmaceutical production, progress of PET technologies and image analysis methodologies for improved quantitation accuracy, PET radiopharmaceutical regulations as well as radiopharmaceutical chemistry advances. This review presents the prospects of the 68Ga-based radiopharmaceutical development on the basis of the current status of these aspects. 68Ga has demonstrated its applicability for the labelling of small compounds, biological macromolecules as well as nano- and micro-particles promoting the growth of PET field 2. The major application domain is oncology; however, potential has been demonstrated for imaging of myocardial perfusion, pulmonary perfusion and ventilation as well as inflammation and infection. Imaging of general biologic properties and processes such as proliferation, apoptosis, hypoxia, glycolysis, and angiogenesis have also been investigated. These prerequisites may trigger an explosive progress and introduction of new 68Ga-radiopharmaceuticals into clinics in the nearest future of 5-10 years.
The focus of this critical review resides on the publications from year 2008-2012 reflecting latest achievements as well as selected earlier references that give the background and support the foundation for the future development of basic research and clinical applications. The achievements in gallium radiopharmaceutical chemistry and imaging agent development from 1956 through January 2011 were addressed in a comprehensive and exhaustive review published earlier 2 and references therein. The explosive growth of publications reflecting the success of 68Ga applications is remarkable. Another strong indication of the worldwide growth of 68Ga studies is the “First World Congress on Ga-68 and peptide receptor radionuclide therapy (PRRT)” that took place in 2011 and attracted participants from all continents 3.
Role of PET in nuclear medicine
Nuclear medicine is a quickly expanding field for the diagnostics and therapy on cellular and molecular level. Molecular imaging techniques such as PET and Single Photon Emission Computed Tomography (SPECT) utilize, respectively, positron and gamma emitting radionuclides for the generation of the signal that results in a whole-body scan in a single examination. They provide fast and non-invasive evaluation of physiology and pathology, and together with external and internal radiotherapy merge into theranostics resulting in personalized medicine 4-7. Imaging diagnostics enables early detection, staging, therapy selection, planning, and monitoring treatment response thus considerably improving cancer therapy. The most pronounced example is the selection of oncological patients for PRRT.
Radionuclidic properties and detection techniques determine the advantages of PET over SPECT in terms of 100-1000 fold higher sensitivity, higher speed, accurate quantitation, and dynamic image reconstruction. However gamma and SPECT scanners are more accessible and have lower cost. Moreover, a wider range of registered gamma emitting radiopharmaceuticals is available, though their price has enhanced currently due to the reduction of public subsidy 8. On the other hand, the shorter scanning time and thus higher throughput of PET decreases the cost per patient examination. Nuclear medicine practice is dominated by 99mTc/SPECT standing for 85% of all clinical examinations in the USA with 50% in cardiology 9. Dual-tracer imaging is one more advantage of SPECT, however recent research has demonstrated possibility of multiple PET tracer imaging based on the signal separation according to the differences in radionuclide half-life as well as tracer kinetics and distribution 1.
Although PET was introduced as early as in the 1970s, only recently it has been recognized clinically relevant, to some extent due to the introduction of hybrid PET-CT scanners that are spreading worldwide with acceleration and have become the gold standard of PET imaging. Rather radical question has been raised if there is a role in oncology for SPECT considering current advantages of PET and respective radiopharmaceuticals 8. The authors suggest that PET provides more accurate and quantitative diagnosis enabling individualized cancer therapy planning resulting in the efficient and cost saving treatment. The propagation of PET technique is greatly stimulated by the widespread use of [18F]-fluorodeoxyglucose ([18F]FDG/PET-CT) for many indications in oncology as well as centralized production and distribution of [18F]FDG to satellite clinical centres. Another factor influencing the expansion of PET is the need for imaging agents with disease specific action, and 68Ga has a significant contribution to make. The reports on such category of radiopharmaceuticals from molecular imaging and contrast agent database (MICAD) state that PET and SPECT imaging agents stand respectively for 42% and 31% 10. However, PET still has to overcome the major hurdles such as regulatory barriers for introducing new imaging agents and restriction of reimbursement. The substitution of SPECT with PET might be just a part of evolutionary process until another more advanced technology appears. Just like rectilinear scanners were substituted with gamma cameras.
PET provides more accurate staging than other conventional diagnostic means and thus it is rational to use it independently and as the first diagnostic choice 11. Dramatic impact of PET-CT on patient management has been recognized by Medicare and Medicaid Services 12. This can be illustrated by the evidence that the course of considerable number of patient treatments (50-60 %) was changed or adjusted on the basis of 68Ga/PET-CT examinations using somatostatin (SST) ligand analogues in diagnosis and staging of neuroendocrine tumours (NETs) 2, 13. Somatostatin receptor scintigraphy will most likely be replaced with 68Ga/PET-CT in the nearest future 14.
The widespread opinion is that PET is a costly technique, but in fact the total cost of the otherwise required examinations, hospitalization and the risk associated with biopsy as well as false diagnosis leading to futile surgery cost and patient distress greatly overweighs. In particular, the accuracy of diagnostic methods related to the heterogeneity of primary tumour and between a primary tumour and metastases should be addressed. The biopsy results might be misleading since the tumour diagnosis and staging is conducted ex vivo on invasively collected tissue. The sampled tissue is commonly heterogenic and thus might not be representative, while PET may provide more detailed and accurate information on heterogeneity of the whole tumour non-invasively in a single examination. The cost of [68Ga]Ga-DOTA-TOC/PET-CT including material and personnel was found lower as compared to that of 111In-DTPA-octreotide/SPECT 15. Moreover, when using the former fewer additional examinations were required. In addition, the cost-effectiveness and cost-saving of PET-CT was demonstrated by [18F]FDG/PET-CT used for staging of lung cancer 8, 16, 17. Currently PET stands for only ~1.5% of Medicare (USA) cancer care expenses, nevertheless the total imaging costs are increasing fast 18, and the reimbursement of [18F]FDG/PET-CT of lung cancer by public health care system might become reality in the nearest future in industrialized countries, and also will propagate into other cancer groups. This in turn will stimulate the investments and development of new imaging agents. The dramatic growth in PET-CT during the last decade has already been accompanied with reducing costs and introduction of new specific PET tracers.
Unmet medical needs
Early diagnosis is important in order to identify functional abnormalities which precede morphological changes, and in particular for cancer, molecular imaging may contribute to the reduction of morbidity and mortality 19. Over 90% of clinical PET investigations are nowadays performed with [18F]FDG, however interest in specific molecular probes for, e.g. cancer and inflammation/infection is getting stronger among scientists and clinicians 20, 21. The reason for this is that although [18F]FDG has been successfully used in many cancers as a biomarker of glucose transport, it fails in diagnosis of slowly growing tumours and in differentiation between tumour and such processes as inflammation, infection, reactive lymph nodes, tuberculosis and sarcoidosis 22, 23. Moreover, the high uptake in normal organs, particularly in brain and gut, results in poor contrast in those areas and potential failure in lesion detection. Thus alternative imaging agents with specific binding capability to e.g. receptors, antigens, enzymes are of strong interest. Specific imaging agents providing information on the molecular and cellular background of various diseases and in particular cancer would allow improvement in patient management and outcome. Generator produced 68Ga may not only enable PET examinations at remote hospitals distant from [18F]FDG distribution, but would also enrich the radiopharmaceutical arsenal at the medical centers both with and without accelerators.
Various 68Ga-based imaging agents have already been tested in humans 7. The basic research and development of new 68Ga-based agents for targeted imaging of specific protein expression products, pre-targeted imaging as well as non-targeted imaging of pulmonary and myocardial perfusion and pulmonary ventilation is expanding steadily. Imaging of cell proliferation, hypoxia, glycolysis, angiogenes as well as inflammation and infection has also been considered 2. Radiochemistry plays a central role in the development and 68Ga in particular has demonstrated powerful potential. The prevalent area of 68Ga clinical applications in the nearest future will most probably be oncology. More research is required for the development of agents for cardiology. On the other hand, neurology will still be dominated by 11C- and 18F-based imaging agents since common requirements to specific binding, small size, lipophilicity and charge might be difficult to meet when using bulky radiometal-chelator complex to be attached to a small biologically active molecule.
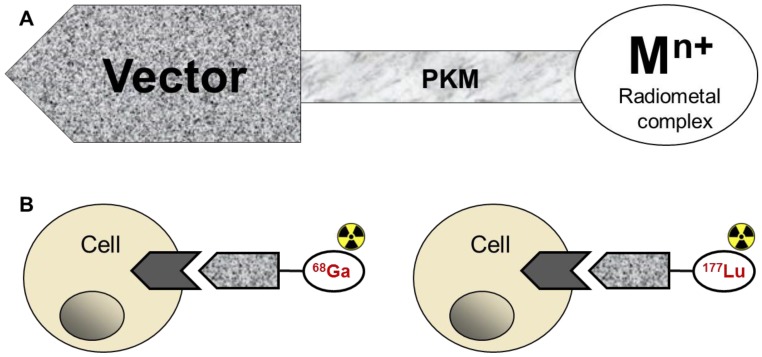
Theranostics in nuclear medicine has manifested in pre-therapeutic diagnosis for PRRT. In this context the diagnosis and radiotherapy are conducted with the same ligand labelled respectively with imaging radionuclide, e.g. 68Ga and therapeutic one, e.g. 177Lu (Figure 3B). The radiopharmaceuticals bind to tumour-type specific receptors enabling personalized approach of accurate quantitative diagnosis and staging for subsequent selection and planning of therapeutic means as well as monitoring response to the treatment. The dosimetry estimation prior to PRRT is one of the most important prerequisites of successful treatment, and the higher spatial resolution and inherent quantitative accuracy of 68Ga/PET are of paramount importance for the dosimetry precision and treatment response evaluation. The common perception is that 68Ga is not applicable for the pre-therapeutic dosimetry due to the short half-life of 68Ga (68 min) mismatching that of therapeutic radionuclides 90Y (64 h) and 117Lu (6.71 d), and consequently resulting in different time windows of the pharmacokinetics. However, e.g. in the case of somatostatin analogues their fast pharmacokinetics may enable the utilization of kinetic analysis of 68Ga-labelled peptide for the prediction of radiation doses during PRRT with the 177Lu-labelled analogue. This might be accomplished by the combination of the respective agents, namely by either co-injection or separate use of 68Ga- and 177Lu-labelled peptides. The high accuracy at early time points could be achieved by 68Ga/PET quantitation with an error of less than 5% since the tracer accumulation in the organs at risk reaches its maximum within 2-3 half-lives of 68Ga 24. The kinetics at later time points could be obtained by either blood sampling during the radiotherapy with 177Lu-labelled analogue or by imaging gammas emitted by 177Lu. The dual-tracer examination is possible since the gamma energy of 177Lu is outside the PET detection window and thus would not interfere with 68Ga/PET-CT quantitation. Such combination of the tracers would provide highly improved internal dosimetry.
Fig 3.
A) Depiction of the basic components of an imaging agent comprising a vector for specific binding, pharmacokinetic modifier, and the complex of the chelator moiety with a radiometal; B) Drawing of the interaction of the agent, either imaging if labelled with 68Ga (left) or radiotherapetic if labelled with 177Lu (right), with the cell receptor.
Specific radioactivity (SRA) of an imaging agent is another parameter influencing the organ distribution of the agent and subsequent dosimetry. In the case of macromolecular agents, e.g. based on peptides, SRA is calculated as radioactivity per total amount of the agent. The targeted and pre-targeted imaging might require relatively high SRA in order to avoid saturation of binding sites by non-labeled counterpart and possible pharmacological side effects. In this context the total amount of the injected agent, e.g. peptide, should be optimized prior to the radiotherapy providing the highest possible uptake in the lesions and lowest possible uptake in the healthy tissue and organs with physiological expression of the target 2, 24, 25.
Development of scanner hardware and software technologies
The technology developed from PET scanners to hybrid PET-CT and PET-MRI 26, 27. The morphologic information from CT and MRI is crucial for the accurate determination of location of the lesions. New hardware, detector, and image reconstruction algorithm technologies (e.g. time-of-flight (ToF-PET), point-spread-function (PSF-PET)) continuously improve image contrast, spatial and temporal resolution as well as quantitation accuracy 1, 28. Moreover, dual-tracer imaging might be possible distinguishing signals on the basis of differences in radionuclide half-lives, tracer kinetics and distribution, however dynamic scanning and multivariate software tools such as principle component analysis would be required 1. PET-MRI combining the high soft tissue contrast of PET and high spatial resolution of MRI has become reality although it is used at present mainly for research in neurology, cardiology, and oncology. It considerably decreases the radiation dose to the patient as compared to PET-CT. The special resolution of PET might also be improved since the positron range can be shortened by the influence of strong magnetic field. PET-MRI with [68Ga]Ga-DOTA-TOC was found superior to MRI alone or PET-CT in terms of detection rate and specificity of liver lesions in patients with NETs 29. It has also been found to improve the accuracy of target volume delineation in intensity modulated radiotherapy (IMRT) treatment planning and delivery 30. Moreover, it reduced the number of pretreatment imaging sessions for meningioma patients.
The spatial and temporal resolution of instrumentation, radionuclide decay mode and energy as well as organ movement and size influence the accuracy of PET quantitation which is especially crucial when monitoring the treatment response with marginal changes. Scanning technology and data reconstruction technique have been improved during last 10-15 years in order to meet the requirements. These advances will definitely stimulate development of 68Ga-based imaging agents, empowered by the relation to the therapy selection and planning. It should be stressed that the full potential of the imaging technology can only be realized if new imaging agents for specific indications emerge. Improved clinical outcomes of molecular imaging-guided radiation therapy have already been demonstrated 23. PET radiopharmaceuticals induce adverse reactions extremely seldom with no serious or life-threatening events 31. While hybrid imaging, PET-CT and PET-MRI, may require high dose of CT and MRI contrast agents thus enhancing the probability of adverse events.
Overview of common clinical imaging radionuclides and 68Ga
The most common radionuclides relevant for the labelling synthesis of radiopharmaceuticals for PET, SPECT, and radiotherapy are presented in Table 1. The majority are metals typically involved in coordination labelling chemistry. Only few of the radionuclides are produced in generator systems. Such factors as production mode as well as physical and chemical characteristics determine the choice of a radionuclide.
Table 1.
Selected radionuclides used in clinical PET, SPECT and radiotherapy, their production mode and decay properties. Radiometals are marked in grey.
| Radionuclide | Half-life | Emax (keV) | Radiation | Production |
|---|---|---|---|---|
| Positron emitters | ||||
| 11C | 20.3 min | 961 | β+ (100%) | Cyclotron |
| 18F | 110 min | 634 | β+ (97%) | Cyclotron |
| 64Cu | 12.8 h | 656 | β+ (19%) | Cyclotron |
| 66Ga | 9.5 h | 4153 | β+ (56%) | Cyclotron |
| 68Ga | 67.6 min | 1899, 770 | β+ (89%) | Generator |
| 89Zr | 78.4 h | 900 | β+ (23%) | Cyclotron |
| 124I | 4.17 d | 2100 | β+ (23%) | Cyclotron |
| Gamma emitters | ||||
| 67Ga | 78.26 h | 91, 93, 185, 296, 388 | γ | Cyclotron |
| 99mTc | 6.0 h | 141 | γ | Generator |
| 111In | 67.9 h | 245, 172 | γ | Cyclotron |
| Therapeutic radionuclides | ||||
| 90Y | 64.0 h | 2270 | β- | Generator |
| 125I | 60 d | 350 | Auger electrons | Reactor |
| 131I | 8.0 d | 1810 | β- | Fission |
| 177Lu | 6.71 d | 500 | β- | Reactor |
Two gallium isotopes, 66Ga (t1/2 = 9.5 h) and 68Ga (t1/2 = 68 min), decay by β+-emission and are therefore suitable for PET imaging, 67Ga (t1/2 = 78 h) decays by electron capture with concomitant γ-emission and is used for SPECT imaging, examples of clinically used radionuclides include 11C, 18F, 64Cu, 89Zr, 99mTc, 111In, 124I (Table 1). MICAD has reported that ~41%, 31% and 28% of PET agents is labelled, respectively with 18F, 11C, and 64Cu, 68Ga, 89Zr, 124I taken together 10. Amongst SPECT agents the leading position belongs to 99mTc (42%) followed by 111In (29%). The major advantage of 99mTc and 68Ga is that they are obtained from generators that are cost-effective alternative to reactors and cyclotrons. They are also cheaper as compared to commercial radionuclides, for example 111In and 124I. 68Ga/PET-CT provides advantage over 99mTc/SPECT not only due to the inherent benefits of the technology, but also due to the resemblance of coordination chemistry of 68Ga, 90Y and 177Lu and, thus possibility to use the same vector molecule for the subsequent radiotherapy. As a result, the diagnostic agent is directed to the same molecular target as radiotherapeutic one allowing the prediction of the treatment efficacy and selection of patients within the frame of theranostics.
With regard to the decay mode, the advantage of positron emitting radionuclides over gamma emitting ones is inherent and related to the benefits of PET technique such as higher sensitivity, resolution, quantitation, and dynamic scanning. The energy and the content of the emitted particles influence the resolution. 68Ga with rather high positron energy could be expected to perform with lower resolution as compared to 18F, however both computational analysis and experimental measurements demonstrated equally high quality images for these two radionuclides assuming the scanner detector resolution of 3 mm 2. The disadvantage with 64Cu, 89Zr, and 124I as compared to 68Ga include low positron abundance (64Cu, 89Zr), higher positron energy (124I), as well as simultaneous gamma emission (124I) resulting in poorer image quality, longer scanning duration, and additional radiation dose. However, their longer half-lives allow development of agents based on vectors with slow pharmacokinetics such as, for example antibodies that require 2-4 days post injection for blood clearance and optimal image contrast. Though, it is worth mentioning that the relatively short half-life of 68Ga allows repetitive examinations on the same day 24.
One of the most rapidly expanding areas is the development of peptide-based agents for targeted imaging. Metal and halogen radionuclides such as 64Cu, 89Zr, 99mTc, 111In, 18F, and 124I are most commonly used for the design of the agents. Typically, the metal ion coordination chemistry is straightforward and mild. However, it requires attachment of a rather bulky chelator to the vector molecule what may deteriorate the biological activity. Simple direct iodination has been used for decades introducing minor modification at tyrosine amino acid residue though demonstrating poor residualizing of the radioactive iodine catabolites in the cell. On the contrary hydrophilic complexes of radiometals stay trapped in the cell after the degradation of the internalized imaging agent. The incorporation of 18F can be accomplished by the conventional nucleophilic or electrophilic addition, or via prosthetic groups 32. However, the synthesis might be rather complex with harsh conditions and high macromolecule concentration. A novel method utilizing similarity of Al18F2+ with metal cations and thus possibility of coordination labelling chemistry has potential for kit type production like radiometals 33. The relatively long half-life (110 min) of 18F enables centralized production and distribution of either the radionuclide or prepared imaging agents to the satellite clinical centres. Most of the PET agents used in clinical studies are 18F-based. Nevertheless, the simpler labelling chemistry of 68Ga and its availability from the generator system which can be used even at distant and isolated medical centres make 68Ga more preferable.
Only 11C amongst over mentioned radionuclides provides true tracers since it is an endogenous element and the radioactive compounds are chemically identical to the stable counterparts. It is perfect radionuclide for the labelling of small bioactive organic molecules. This field is rather challenging for metal radionuclides including 68Ga, and 11C together with 18F will most probably dominate in the field of neurology and metabolic tracers in oncology employing small molecules 34.
As mentioned earlier the advantages of 68Ga over 111In are multiple and have already been demonstrated in patients affected by NETs in terms of diagnostic output and logistics, superior sensitivity and accuracy, higher detection rate and specificity, faster acquisition and shorter examination time, lower radiation exposure as well as cost, and 68Ga-labelled somatostatin analogues are predicted to replace [111In]In -DTPA-octreotide (Octreoscan®) in the nearest future 14, 15, 35, 36.
Another advantage of 68Ga over 18F, 99mTc, and 111In is comparable or lower level of effective dose (Table 2) 37, 38. The latter was calculated considering administration of commonly used levels of radioactivity of 100, 200, 370, and 570 MBq respectively for [68Ga]Ga-DOTA-TOC, [111In]In-DTPA-octreotide, [18F]FDG, and [99mTc]-MDP. It is worth mentioning that the examination time is considerably shorter for 68Ga-agents as compared to SPECT agents and somewhat shorter compared to [18F] FDG.
Table 2.
Effective doses for some PET and SPECT imaging agents.
| Agent | Examination time | Effective dose, [mSv] |
|---|---|---|
| [111In]In-DTPA-octreotide/SPECT | 24-48 h | 10.8 |
| [68Ga]Ga-DOTA-TOC/PET | 30-60 min | 2.3 |
| [18F]FDG/PET | 60-120 min | 5.6 |
| [99mTc]-BPAMD/SPECT | 2-6 h | 6 |
| [99mTc]-MDP/SPECT | 2-6 h | 3-4 |
| [68Ga]Ga-BPAMD/PET | 30-60 min | 3-4 |
The benefits of 68Ga can be summarized as follows. It is produced from a long shelf-life and cost-effective generator. The half-life of 68Ga permits production and application of resultant agents, and the labelling synthesis is amenable to automation and kit type preparation. It provides sufficient levels of radioactivity for high quality images, short scanning time (fast patient examination) while minimizing the radiation dose to the patient and personnel, and allows fast dischargement of the patient. It also allows repetitive examinations within the same day 24. The majority of the therapeutic radionuclides is also metals and might allow for the theranostic development.
In comparison with the other radionuclides mentioned above there are a number of advantages and disadvantages, and they all complement each other to make PET imaging applicable to a broad range of diseases. 99mTc/SPECT has been holding the leading position in nuclear medicine for decades due to such factors as favorable gamma energy, availability from a generator system and kit type radiopharmaceutical production. The future of 68Ga is predicted as PET analogue of 99mTc/SPECT with added value of higher sensitivity, resolution as well as accurate quantitation and personalized medicine. However, even if the vision of 68Ga becoming as immense as 99mTc will not get fulfilled, 68Ga has definitely strong contribution to make in the improvement of personalized patient management and there is a niche for 68Ga-based agents in nuclear medicine. Moreover, 68Ga-agents have the prerequisites to substitute also 111In-based radiopharmaceuticals.
Biomarkers, targets, and ligands
Development of agents for imaging and radiotherapy involves identification of biological process and target underlying the pathology as well as respective lead compound. Then the radioactive lead compound counterpart must be designed, chemically characterized as well as preclinically and clinically validated. In particular, the specific targeting and pre-targeted imaging require information on biomarkers and is dependent on their discovery. Thus, advances in biological research and biotechnology are crucial. Proteomics and genomics considerably contribute to the expansion due to the increasing knowledge and access to the vectors and targets such as receptors, enzymes, antigens as well as their ligands and substrates. Proteins demonstrate remarkable capability of molecular recognition. Advances in genetic and biochemical techniques resulted in a large number of antibody radioimmunotherapeutics. That in turn triggered further development with the objective to overcome the drawbacks related to antibody high molecular weight, slow pharmacokinetics and clearance that cause high radiation dose to normal tissue and poor image contrast. Thus, large libraries of high affinity small proteins have been created using combinatorial engineering and phage display techniques that provide efficient screening of ligands as well as identification and selection of antibodies and receptors for drug discovery and therapy. A number of non-immunoglobulin scaffolds has also been studied that resulted in libraries as, for example Affibody® molecules originating from Z domain scaffold of staphylococcal protein A. Thirteen surface-exposed residues of the scaffold were randomized resulting in a library (3x109 members) of high-affinity binders to various targets 39. Radiolabelled Affibody molecules have extensively been investigated preclinically, and 68Ga- and 111In-labelled analogues with high affinity to HER2 receptors up-regulated in breast cancer has also been studied in patients 40. This is a strong evidence for the future fruitful development of engineered high affinity proteins and abundant source of ligands to various receptors expressed in diseased tissues.
G-protein and G-protein coupled receptor discoveries, both awarded with Nobel Prize, respectively in 1994 and 2012, provided basis for the important and most explored class of imaging agents comprising small regulatory peptides that are involved in many metabolic processes in almost all organs 41-43. It should be mentioned that one of the factors that tremendously contributed to the progress of 68Ga applications was the advent of agonist somatostatin ligand analogue (DOTA-D-Phe1-Tyr3-Octreotide, (DOTA-TOC)). The current development is directed to pansomatostatin and antagonist analogues 44. Other examples of peptide receptor targets in various cancer types are: gastrin-releasing peptide (GRP) receptors in prostate, ovarian and urinary tract cancers; neuropeptide Y receptors in breast cancers; glucagon-like peptide 1 receptors (GLP-1) in benign insulinomas and islet cells; vasoactive intestinal peptide receptor (VPAC-1) in breast, prostate cancer; cholecystokinin 2 receptors (CCK) in medullary thyroid cancers; neurokinin-1 receptor in glioblastoma, breast cancer; melanocortin-1 receptor in melanomas; chemokine receptor 4 (CXCR4) in lung, breast, prostate cancer; neurotensin (NT) receptor in SCLC, colon, breast, prostate cancer. Corresponding peptide ligands such as bombesin, CCK/gastrin, exendin, α-MSH, neurotensin, substance P labelled with 18F, 68Ga, 99mTc or 111In have been studied clinically 45-50. Even though just few of them were labelled with 68Ga, it is a solid background and springboard for 68Ga-based analogues to come in the nearest future. Many more studies have been conducted preclinically and it is worth mentioning that the total critical mass of basic knowledge and experience gained until now will lead to the explosively growing clinical studies with 68Ga. The design of an imaging agent is a complex process, and although the major factors that influence the biological function of a probe such as peptide/protein sequence and 3D structure, pharmacokinetic modifiers, chelators, metal cations are known, it is not that straightforward to predict the pharmacokinetics and pharmacodynamics of the candidate agent 51.
Thus fundamental research in biology of surface receptors and antigens, enzyme activity, transport systems, proliferation, apoptosis, hypoxia, glycolysis, and angiogenesis provides invaluable information and lays a basis for imaging agent development.
PET radiopharmaceutical regulations
The absence of legislation and regulations specific to PET radiopharmaceuticals made it difficult to conduct clinical trials and introduce new imaging agents into clinical routine however the situation has improved during last years. It required considerable effort from academic, clinical and patient communities and societies, and the hard work has started giving results. Recent developments indicate possibility of regulatory specific solutions that may allow the clinical use of small scale preparation radiopharmaceuticals without obligation to apply for manufacturing authorization or clinical trial 52.
One of the major difficulties was that regulatory bodies evaluated therapeutic and imaging agents by the same process and put forward the same requirements, however there has been an improvement such as the recognition of the microdosing concept 53-55 by EMEA and FDA, and introduction of the Exploratory Investigational New Drug (eIND) guidelines that reduces the demand on toxicity studies and respective cost burden 56, 57. This is possible because of the high sensitivity of PET and consequently use of nonpharmacological radiopharmaceutical doses of picomoles (nanograms-micrograms). It should also be mentioned that commonly the adverse reactions to PET radiopharmaceuticals are extremely rare and with no serious or life-threatening events 31.
The recommendations on compliance with regulatory requirements for radiopharmaceutical production in clinical trials have been discussed for Europe and the USA 58, 59. A comprehensive overview of relevant current EU documentation concerning requirements to the quality of starting materials and final drug products/radiopharmaceuticals as well as to the preparation procedure has been published with references to specific directives and regulations 52, 58, 60. Another guidance is more specific and covers Part B of the EANM “Guidelines on Good Radiopharmacy Practice (GRPP)” for small-scale “in house” production of radiopharmaceuticals that are not intended for industrial production, sale or distribution 61. European pharmacopoeia monograph on compounding of radiopharmaceuticals that includes chapter dealing with standards of the extemporaneous preparation of radiopharmaceuticals has been prepared and submitted to the authorities.
Recommendations on the patient examination protocols, interpretation and reporting of the results have been summarized as guidelines for the assistance to nuclear medicine physicians 62, 63. Two European Pharmacopeia monographs are in the preparation and publication, “Gallium (68Ga) edotreotide injection” 64 and “Gallium (68Ga) chloride solution for radiolabelling”. One of the critical quality parameters is the breakthrough from the 68Ge/68Ga generator of long-lived parent radionuclide, 68Ge (t½=270.8 d) and its content in the radiopharmaceutical preparation of not more than 0.001% may restricts the use of the generators and limit clinical applications of 68Ga specifically with regard to kit type preparation. The limit defined in the European Pharmacopeia monographs was based on a hypothetical assumption of total accumulation of 68Ge(IV) radioactivity in the bone marrow with an infinite retention. However, 68Ge(IV) biodistribution studies conducted currently in rats with extrapolation to the human organ and whole-body radiation dosimetry demonstrated that the limit defined in the monograph could be increased at least 100 times without compromising patient safety 65. If the 68Ge limit is increased, then together with the labelling methods of high selectivity towards 68Ga at room temperature, it will open possibility for kit type production at radiopharmacy practice and consequently even wider use of 68Ga-based agents. There are no labelling kits for the preparation of 68Ga-based radiopharmaceuticals with marketing authorization available currently, and it is a matter of years before they appear on the market.
One more crucial reason postponing the clinical introduction of 68Ga-agents was the scarcity of generators, their quality, and absence of corresponding regulations. However currently at least four different generators are commercially available and more are under development. Some of the manufacturers (Eckert & Ziegler; iThemba Labs) hold license for good manufacturing practice (GMP) production and the generators of pharmaceutical grade are on the way (Eckert & Ziegler). The draft monograph, “Gallium (68Ga) chloride solution for radiolabelling”, regulates the requirements to the quality and safety.
In order to accelerate the use of PET for assessing treatment responses, standardization and consolidation of image acquisition, interpretation, and quantitation criteria that have been considered in Europe 52, 66, 67 and the USA 68 are required to be introduced into practice 18. International Atomic Energy Agency conducted survey of the status of nuclear medicine in developing countries and localized the areas requiring international cooperation and help in order to improve the level of use and the quality as well as promote global standardization, growth, and dissemination 69.
There is an ongoing dispute on the benefits of PET-CT in patient management conducted by national authorities. The validity of such review judgments of health technology assessment (HTA) agencies was questioned 70. The authors investigated HTA reviews (1996-2010) and found them misleading and that in many countries they unfortunately discredited evidence of PET and PET-CT capability to improve patient outcomes. However, original conclusions of clinicians have proven the opposite, and access to this technology though was delayed it could not be prevented.
In summary, the regulation and legislation of the manufacturing of PET radiopharmaceuticals and in particularly 68Ga-related documentation has been defined during current years reflecting the acceptance of obvious benefits of 68Ga/PET-CT to the patient management. Interestingly the analysis of various reasons causing performance inefficiency of nuclear medicine centres in developing countries revealed that regulatory hinder was the major problem only at 5% of the centres 69. The USA had a 10 y delay in implementing human studies with 68Ga-labelled somatostatin analogues as compared to Europe due to mainly poor availability of generators, uncertainty about intellectual property of the ligands and regulation of manufacturing; however regulatory uncertainty and generators are less of a problem currently in the USA 71. There are at least three sites, University of Iowa, Vanderbilt University, and Excel Diagnostics & Nuclear Oncology Center holding approval from FDA for the use of 68Ga-labelled somatostatin analogues in the management of patients with NETs. Peptide based precursors of GMP grade for labelling with 68Ga are becoming available commercially which will most likely accelerate the introduction of respective imaging agents into clinical environment. Nevertheless currently the facilitation of the entry of novel radiopharmaceuticals into clinical practice relies mostly on magisterial preparation/prescription and compassionate use under responsibility of the prescribing physician.
Automation of the labelling procedures
Automated synthesis reduces the radiation exposure to the operator, improves robustness of the production as well as provides on-line documentation of the manufacturing process thus improving GMP compliance. Considerable number of semi-automated and fully automated devices with either stationary tubing system or disposable cassettes for the automated production of radioactive probes is available on the market as well as built “in house” 72-85. They offer application of three most common methods: fractionation and preconcentration/prepurification of the generator eluate using either cation or anion exchange resins. Synthesizers based on the disposable sterile cassettes and devoted to the production of 68Ga-comprising imaging agents are most preferred in routine clinical setup even though the cost in higher. They exclude the risk of cross-contamination, improve the reproducibility and reduce the radiation burden upon the operator. Both hardware and software should comply with GMP and good automated manufacturing practice 5 (GAMP5). The automation may provide possibility for the harmonized and standardized multicentre clinical studies that in turn would accelerate the introduction of new radiopharmaceuticals as well as their regulatory approval. This in turn will motivate investments into the research and development of novel 68Ga-based radiopharmaceuticals.
Chemistry of 68Ga-based imaging agents
Above mentioned interrelated aspects are critical and prepare soil for the justification and stimulation of the development of new imaging agents which is an expensive and time consuming process. However, the influence is two way and the development of these aspects is in turn motivated by the advent of new agents. Chemistry is a driving force of the development of nuclear medicine and hundreds of various imaging agents have been synthesized and preclinically evaluated.
The labelling with radiometals can be direct or chelator-mediated (tagged). The direct 68Ga-labelling of macromolecules is limited and applies to proteins (e.g. lactoferrin, transferrin, ferritin) designed by nature for iron binding thus utilizing chemistry similarity of Ga(III) and Fe(III) 86. The direct 68Ga-labelling and formation of low molecular weight complexes is commonly employed for the development of imaging agents for perfusion or for imaging of biological processes where the agent uptake is defined by its charge, lipophilicity, and size. Particulate agents can also be produced by the direct 68Ga-labelling either by co-precipitation (e.g. macroaggregated albumin) or by co-condensation (e.g. 68Ga-carbon nanoparticles) 87-92. The chelator mediated 68Ga-labelling, requiring first synthesis of a bioconjugate comprising vector molecule and chelate moiety for the coordination of the radiometal ion, is the most common pathway of imaging agent design. The principle components of such agents are targeting vector, chelator, and radionuclide (Figure 3A). The modulation of pharmacokinetics, biodistribution, and stability can be achieved by the introduction of pharmacokinetic modifiers (PKM) such as hydrocarbon chain, polyethylene glycol (PEG), carbohydrate, and polypeptide chain. PKM may also serve as linker/spacer between the bulky chelate moiety and the active site of the vector molecule. Thermodynamic and kinetic stability, geometry and lipophilicity of a chelator-metal ion complex are important parameters in the development of radiometal based radiopharmaceuticals.
These design principles are valid for the 68Ga-labelling of small biologically active organic molecules, biological macromolecules, complexes of variable charge and lipophilicity as well as particles. Thus the chemistry considered in the development of 68Ga-based imaging agents has several cornerstones: aqueous chemistry of 68Ga, coordination chemistry of Ga(III), design of mono- and bifunctional chelators, and bioconjugate chemistry. As mentioned above most of 68Ga imaging agents known by now are tagged which means that their biodistribution is defined by the vector molecule and 68Ga may or may not influence biodistribution or affinity to the target. Such are agents for targeting and pre-targeted imaging of receptors, enzymes, antigens, and transporters.
Production, availability and quality of 68Ga
68Ga is artificially produced one of twenty nine isotopes of gallium. It is a product of 68Ge decay (t1/2 = 270.8 d) and is obtained from a 68Ge/68Ga generator system where 68Ge is absorbed on a chromatographic column with either inorganic or organic matrix 2, 93. The parent radionuclide decays to a shorter-lived daughter 68Ga that can be simply eluted from the column. It is not only a reliable source of 68Ga at clinical centres remote from the cyclotron and distribution sites but it also enhances imaging agent assortment at centres possessing cyclotrons. Although first generators were introduced in late 1950s early 1960s and 68Ga-based imaging agents were available already from 1970s 2, the development of the respective radiopharmaceuticals was precluded by the absence of reliable sources of the generators as well as their properties and quality. The first generators produced 68Ga in a complex with EDTA that could be directly used for the clinical patient imaging of blood flow of brain tumours. However, [68Ga]Ga-EDTA complex prevented the development of other radiopharmaceuticals since the chemistry was complicated by prior need of the complex decomposition. The situation in the recent years has changed considerably. Several different generators manufactured in the USA (IGG100, Eckert & Ziegler), Germany (ITG Isotope Technologies), Russia (Obninsk, Cyclotron Co, Eckert & Ziegler) and South Africa (iThemba Labs) are commercially available and more are under development in Europe, North America and Asia 76, 94-97. The generators are eluted with hydrochloric acid of various molarity (0.05M to 5.5M, 5-7 ml) offering 68Ga in cationic form suitable for the subsequent labelling. The generators have been thoroughly characterized by various research groups and methods for the efficient and GMP compatible labelling synthesis have been developed 2, 98-101. The generators can be eluted repeatedly during the day. Fifty percent of the maximum available 68Ga-radioactivity can be obtained every consecutive hour. Four hour accumulation provides respectively over 94%. The long half-life of 68Ge (271 d) allows 1-2 year shelf-life in the clinical setting dependent on the loaded radioactivity amount. The amount of 68Ga and the shelf-life of the generators can be increased by the elution of several sequentially coupled generators.
The long shelf-life of the generator triggered concerns regarding microbiological safety even though the acidic eluent (e.g. 0.1M HCl) is highly unfavourable environment for the microbial growth. However it was demonstrated in a study with a 68Ge/68Ga generator column intentionally loaded with various bacteria and fungi in exhaustive amounts that the risk of incidental microbial contamination was very low 102. Another concern was the breakthrough of parent long-lived 68Ge and its content in the 68Ga eluate. However, germanium is not pharmacologically active and in vivo it acts as a non-toxic foreign material which is readily eliminated and thus as a chemical presents low risk to man 65. Thus with regard to radioactive 68Ge(IV) where the amounts of the element are negligible, the safety issue is reduced to ionizing radiation and, in particular the buildup of 68Ga(III) at the sites of deposition of the 68Ge(IV). However, 68Ge(IV) biodistribution study in rat demonstrated fast excretion (t1/2=0.6 h) and no deposition in any organ 65. Dosimetry calculations revealed that as much as 645 MBq (female) and 935 MBq (male) could be administered before reaching effective dose of 10 mSv which is a general limit to healthy volunteers to which most authorities adhere. Moreover, it was shown that 68Ge(IV) was not chelated by DOTA-TOC and thus accumulation in the sites of DOTA-based imaging agents was also excluded.
Three major methods for the quality improvement of the generator eluate such as fractionation, cation exchange chromatography, and anion exchange chromatography are currently in common use 2. In particular, reduction of the generator eluate volume as well as purification from metallic impurities (Al(III), In(III), Ti(IV), Zn(II), Ti(IV)) and long-lived parent 68Ge(IV) were achieved to certain extend. However, it should be mentioned that the reduction of the concentration of contaminant metal ions including Zn(II) being product of 68Ga decay in the generator eluate can be achieved simply by regular elution of the generator 99. The shorter elution time interval is the less contaminant cations are accumulated. Unfortunately, none of these methods fully eliminates Fe(III) which is the strongest competitor to 68Ga(III) since its chemistry is very similar to that of Ga(III). The anion exchange prepurification and preconcentration method was further modified in order to remove the excess of [H+] and ensure high reproducibility of pH which is crucial for the successful labelling 103. The stabilization of pH and omission of strong sodium hydroxide use was achieved by the addition of a cartridge washing step using 5 M sodium chloride solution. The content and type of gallium chemical species under this conditions was investigated and shown to be prevailed by [GaCl]2+ and Ga3+, respectively before and after the buffering (pH 3) 104. Another advantage of the anion exchange method is that the highly concentrated 68Ga in small volume can directly be used for production of 68Ga-carbon nanoparticle aerosol for the examination of acute pulmonary emboli 91, 105. Furthermore the anion exchange method is applicable to any generator type eluted with HCl independent on the molarity of the latter. It can also be used for the preconcentration of several generator eluates independent on the total volume. High amount of radioactivity would provide high SRA however it should be taken into consideration that it also may induce radiolysis of sensitive active substances. In such case radical scavengers, e.g. ethanol or ascorbic acid can be added directly to the reaction mixture. In combination with microwave heating 68Ga preconcentration method allowed production of agents with enhanced specific radioactivity 99, and was successfully used for the labelling of DOTA conjugates with macromolecules and small molecules 2. The method was automated 84.
The cationic method for the preconcentration and purification of the generator eluate using cation exchange resins and resulting in 400 µL solution of acetone/HCl mixture 101 has also been improved. It offers possibility for the labelling in non-aqueous solution via 68Ga(acac)3 precursor formation 106. However, precautions for the storage of acetone/HCl mixture should be taken since acetone in the acidic pH under light forms mesityloxide 107. Methods to avoid usage of the acetone/HCl mixture has been developed based on sodium chloride 108.
In addition the number of patient administration doses from one production would depend on the initially available radioactivity (strength and age of the generator), labelling duration and efficiency as well as the number of scanners for examinations. The maximized utilization would require simultaneous multiple patient examinations immediately after the tracer production since the short half-life of the 68Ga limits the consumption duration. However, this drawback is compensated by the fact that the injected dose can be as low as 100-150 MBq still resulting in high quality images. Another advantage is that the generator can be eluted repeatedly providing imaging agent production with 1-2 h (50-70% of maximum eluted 68Ga) interval.
The 99mTc-based radiopharmaceuticals have globally been used for decades, partly due to the readily availability from a generator system. However, production of the parent 99Mo was drastically decreased due to the closure of the reactor in Canada, 99Mo waste and security issues related to the weapon grade 235U in the nuclear reactor target all together rendering increase in generator price and limited availability 109. Consequently, the number of clinical examinations using 99mTc-radiopharmaceuticals decreased during last years. Even though parent 68Ge is produced in high energy accelerators the latter are more available and affordable than reactors 93. The shortage of 99Mo has also motivated development of 68Ga-based alternatives to 99mTc-radiopharmaceuticals. The authors of 110 have wisely noticed that the solution of the supply shortage is not only in the investment and extension of 99Mo production, but in creating diversity of radiopharmaceuticals by introducing alternative medically relevant isotopes with 68Ga as an obvious candidate.
The current development of the generator market, research on the quality of eluted 68Ga as well as establishment of regulations indicate future diversity and high quality of generators with low 68Ge breakthrough, narrow elution profile, and low metal ion contamination.
Coordination chemistry and design of chelators
The hard acid Ga(III) can form four-, five-, and six-coordinated complexes. The latter are the most stable with octahedral coordination sphere. Oxygen, nitrogen, and sulfur donor atoms form stable coordinate bonds with Ga(III). The examples of the most common functional groups are amine, carboxylate, hydroxamate, phenolate 2. The coordination reaction requires buffering for two reasons: first, to ensure right pH for the deprotonation of electron donor atoms; second, to weekly coordinate and maintain Ga(III) in solution that might otherwise form Ga(OH)3 and precipitate at pH 3-7 2. Various buffers such as sodium acetate, 4-(2-Hydroxyethyl) piperazine-1-ethanesulfonic acid (HEPES) buffer, succinate, formate, tris, glutamate, lactate, oxalate and tartrate have been investigated 75, 99. Although HEPES is biologically compatible buffer 24, 25 and provides high radioactivity incorporation (>95%) and specific radioactivity 75, 99, 111, it is not listed in pharmacopoeia and thus prior to clinical use the radiopharmaceutical should be purified from it and additional quality control must be conducted to ensure that HEPES does not exceed the limit. The use of sodium acetate has the advantage of being eligible for human use possibly simplifying the regulatory approval of the radiopharmaceutical as well as feasibility to omit additional quality control tests 75.
As mentioned above coordination chemistry is one of the cornerstones of the development of 68Ga-based imaging agents and thus advances in mono- and bifunctional chelator design is of paramount importance. The topic has recently been reviewed elsewhere 2, 112 and here only the fundamental and recent advances that shape the future of the field are discussed. The basic structures that have been most thoroughly studied are polyaminopolycarboxylate, hydroxyaromatic, macrocyclic and amine-thiol type ligands. The principle requirements are that they should form stable complexes with Ga(III) favourably of octahedral geometry in order to yield stable complexes. The association kinetics must be fast and reaction desirably taking place at room temperature while dissociation kinetics must be very slow 2. Charge and lipophilicity can be adjusted dependent of the application. Two classes of chelators namely open chain and macrocyclic have been considered. Although the former commonly formed insufficiently stable complexes the interest towards them still remains with the desire for fast complexation kinetics at room temperature that would like NOTA 113 provide possibility for kit type preparation of radioparmaceuticals and labelling of temperature labile molecules.
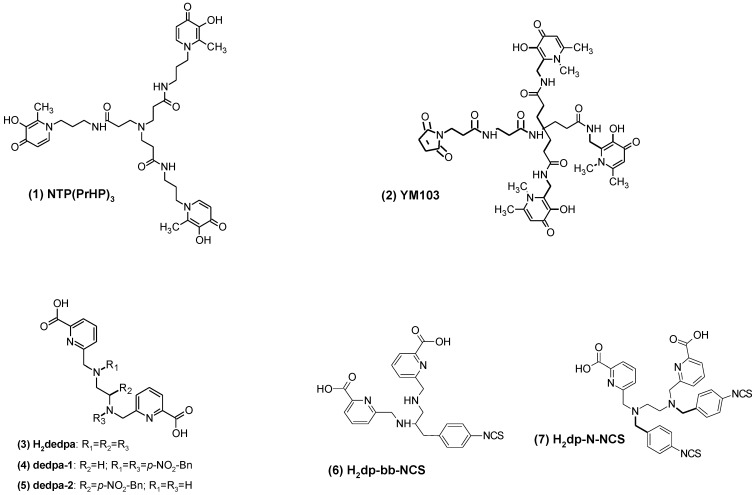
Several open-chain chelators such as hydroxyaromatic diamines like N,N'-di(2-hydroxybenzyl)ethylenediamine-N,N'-diacetic acid (HBED), pyridoxylethylenediamines such as N,N'-dipyridoxyethylenediamine-N,N'-diacetic acid (PLED), mono- and bifunctional open chain chelators based on [tris(aminomethyl)ethane] (TAME) formed complexes with improved stability 2. However, neither of them or four-, five- and six-coordinate amino-thiol (N2S2, S3N, 4SS, 5SS, 6SS, N3S3) based open chain chelators 2 have found further application. There have been just few publications reporting on open-chain chelators and not all of them demonstrated desired properties. 67Ga-labelled tripodal 3-hydroxy-4-pyridinone (1, NTP (PrHP)3, Figure 4) showed in vivo stability and fast renal excretion in healthy rats 114. Similar chelator, tripodaltris(hydroxypyridinone) (YM103), was functionalized with maleimide for the conjugation to proteins via Cys 115. The efficient and stable 68Ga-labelling occurred at room temperature. H2dedpa (N4O2), and its bifunctional derivatives containing amine, pyridine and carboxyl moieties (H2dedpa (3), dedpa-1 (4), dedpa-2 (5), Figure 4) were stably labelled with 67/68Ga at room temperature (SRA~360 MBq/nmol) using 0.1 µM chelate 116. While HBED possessing hydroxybenzyl and amine groups 117 showed low labelling efficiency and slow blood clearance. The isothiocyanato derivatives of the H2dedpa (H2dp-bb-NCS (6), H2dp-N-NCS (7)) have been synthesized and conjugated to c(RGDyK) resulting in monomer and dimer 118. The uptake of monomer was higher than that of dimer in RAG2M xenografts. However, very slow clearance from blood requires further improvement of pharmacokinetic properties.
Fig 4.
Basic structures of open chain mono- and bifunctional chelators.
An exhaustive number of triazacyclononane (TACN (8)) and tetraazacyclododecane (TACD (9)) (Figure 5) derivatives have been synthesized. The backbone and pendant arms were functionalized for the conjugation to vector molecules and in order to modulate complexation kinetics, charge, lipophilicity and stability of the complex as well as biodistribution, pharmacokinetics, excretion pathways and blood clearance rate. The pendant arm modifications include such functional groups as carboxylic acid, phosphinic acid, α-haloacetyl, alkoxy, alkyl- and arylamine, alkyl- and aryl sulphide, phenol, hydroxamate. A number of TACN based molecules functionalized with 3-hydroxy-4-pyrone pendant arms (H3NOKA (10)), with carboxylic pendant arms (NOTA (11)) and its various derivatives, namely, 1,4,7-tris(2-mercaptoethyl)-1,4,7-triazacyclononane (TACN-TM (12)), 1,4,7-triazacyclononane-1-succinic acid-4,7-deacetic acid (NODASA (13)), 1,4,7-triazacyclononane-N,N′,N′′-tris(methylenephosphonic) acid (NOTP (14)), 1,4,7-triazacyclononane-N,N′,N′′-tris(methylenephosphonate-monoethylester) (NOTPME (15)) (Figure 5) demonstrated similar plasma and in vivo stability. NOTA and its bioconjugates showed efficient chelation (>95%) of 68Ga at pH 3.5 and room temperature within 10 min 113, 119, 120. Mechanistic studies of the unexpectedly fast complexation kinetics at such low pH suggested that the transchelation step from the buffer to NOTA involved protonation of the buffer and decoordination that lead to the final Ga-NOTA product 121. The room temperature is advantageous for the labelling of fragile molecules as well as “shake and shoot” type kit production. Triazacyclononane with either hydroxybenzyl or hydroxypyridyl pendant arms at the nitrogens (TACN-meHP (16), TACN-TX (17), TACN-HP (18), TACN-HB (19), TACN-TM-Bn (20) Figure 5) were synthesized in order to increase the lipophilicity of gallium complexes and enable the blood brain barrier penetration 2. The resultant complexes were stable however did not serve the purpose.
Fig 5.
Examples of TACN and TACD based mono- and bifunctional chelators.
The most promising and thoroughly investigated group of chelators is based on TACN and functionalized with phosphinic acid pendant arms. In particular, chelates with basic structure of N,N′,N′′-trisubstitutedtriazacyclononane with methyl(2-carboxyethyl)phosphinic acid pendant arms (PrP9 or TRAP-Pr (21)) were synthesized for the fast complexation with 68Ga 122. The labelling was possible at ambient temperature and as extreme pH as 1. The complexes were thermodynamically stable (logK=26.24) and kinetically inert. TRAP-Pr was activated with propargylamine for the further conjugation with cyclo(RGDfK) at each pendant arm of the chelate resulting in RGD-trimer 123. The in vivo performance of [68Ga]Ga-TRAP-(RGD)3 was evaluated in athymic nude mice bearing M21 (αvβ3-positive) and M21L (low expression of αvβ3) human melanoma xenografts. The uptake could be fully blocked by excess of unlabelled precursor. Another derivative of TRAP possessing just one site for bioconjugation (NOPO) was conjugated with cyclo(RGDfK) and NOC and labelled with 68Ga with as high SRA as 5.6 GBq/nmol 124. The uptake of [68Ga]Ga-NOPO-RGD and [68Ga]Ga-NOPO-NOC respectively in M21 and AR42J mouse xenografts was specific. NOPO demonstrated highly selective complexation with Ga(III) as compared to Fe(III) and Zn(II) 124. The higher complexation selectivity of TACD derivatives for Ga(III) as compared to In(III) and Al(III) has also been demonstrated using DOTA-TOC 25. A comprehensive and thorough investigation has been conducted on the influence of Zn(II), Cu(II), Fe(III), Al(II), Ti(IV), and Sn(IV) on the incorporation of 68Ga(III) into NOTA, DOTA (22), TRAP, TRAP-Pr, and NOPO as well as their peptide conjugates 125. The selectivity of TRAP, TRAP-Pr, and NOPO towards Ga(III) was considerably improved as compared to NOTA and DOTA. This is particularly very critical with regard to Fe(III) and Zn(II). The structural investigation of the underlying mechanism of the coordination revealed formation of various complex diastereoisomers 126. The presence of phosphinic acid pendant arms improved the ligand coordination ability and resulted in fast complexation in acidic media. The complexes were kinetically inert in acidic as well as basic solutions. The broad range of pH at which the complexation reaction can take place would allow avoiding the pH 3-7 when the Ga(III) is insoluble and thus omit the use of buffers that contribute to cation contamination. This might improve the complexation efficiency and thus SRA. In addition the stability constant for [68Ga]Ga-DOTA was recently revised and demonstrated to be even higher (logK = 26) than previously known (logK = 21.33) 127.
Four bifunctional macrocyclic chelators have been investigated with the objective to compare their labelling chemistry and in vivo stability and clearance 128. Nine and twelve member rings were considered, namely 1,4,7-triazacyclononane-1,4,7-triacetic acid (p-NO2-Bn-NOTA (23)), 1-oxa-4,7,10-triazacyclododecane-4,7,10-triacetic acid (p-NO2-Bn-Oxo (24)), 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (p-NO2-Bn-DOTA (25)) and 3,6,9,15-tetraazabicyclo [9.3.1]pentadeca-1(15), 11,13-triene-3,6,9-triacetic acid (p-NO2-Bn-PCTA (26)) (Figure 5). The labelling was more efficient for p-NO2-Bn-NOTA, p-NO2-Bn-PCTA, and p-NO2-Bn-Oxo as compared to p-NO2-Bn-DOTA. However, p-NO2-Bn-Oxo was unstable in vivo. p-NO2-Bn-NOTA cleared rapidly from the blood and muscle but had 5-fold higher uptake in kidneys.
A novel approach providing a chelator, protoporphyrinIX (PPIX), that can be used both for in vitro fluorescence microscopy and in vivo PET or SPECT imaging has an advantage of using exactly the same molecule 129. PPIX was conjugated to RGD peptide sequence and 68Ga-labelled under microwave heating. The resulting probe was evaluated in MDA-MB-435 cancer cell line expressing integrin receptors, and demonstrated binding specificity. Porphyrins specifically accumulate in tumour tissue and five various 68Ga-labelled analogues were suggested as imaging agents 130. Preliminary results indicated accumulation in DS sarcoma tumour in rat, however the mechanism of the uptake requires further investigation 130.
Another class of macrocyclic chelators is bridged or macrobicyclic 131. The structure of [Ga-(1-NH3-8-NH2-sar)]4+ (where sar =3,6,10,13,16,19-hexaazabicyclo[6.6.6.]icosane) was determined by X-ray crystallography as distorted octahedral with six nitrogen atoms. The chelator was coupled to two RGDfK moieties. The resulting conjugate was labelled with 68Ga at elevated temperature (85 °C). The specific uptake was observed in vivo in 66c14β3 xenografts in mice.
In summary, even though open chain chelators serve the aim of rapid chelation at room temperature they have it difficult to compete with macrocyclic chelators based on TACN and TACD that provide stability, selectivity, fast association kinetics and extremely slow dissociation kinetics as well as high thermodynamic stability. It should also be mentioned that the advent of macrocyclic chelators was one of the critical factors influencing the broader demand for 68Ga and its versatile applications. The current improvement in macrocyclic design is substantial, and mechanistic as well as structural investigation of coordination chemistry provides knowledge for more efficient and diverse imaging agent development and radiopharmaceutical routine production. The development of mono- and bifunctional chelators will direct towards fast complexation and room temperature, broad working pH range and high SRA. Although TACN derived chelators offer an advantage over TACD with respect to fast complexation kinetics at room temperature and higher selectivity there are more aspects to take into consideration such as theranostics applications where it is desirable that both diagnostic and therapeutic radionuclides can be coordinated by the same chelator moiety.
Conjugation chemistry
The most common and commercially available BFC comprise esters (p-nitrophenyl, pentafluorophenyl, N-hydroxysuccinimide, sulfo-N-hydroxysuccinimide), isothiocyanates, maleimides, hydrazides, α-haloamides for the reaction with nucleophilic functional groups (-NH2, -SH, -OH) of vector molecules and formation of amide, urea, thiourea, Schiff-base, or thioester bond 2. Methods have been developed for the conjugation with peptides by solid-phase peptide synthesis (SPPS) resulting in defined position and number of chelate moieties. The outcome of the conjugation in solution wherein peptides and proteins comprise several reactive sites is very often a mixture of bioconjugate molecules with various content of the chelator. Such heterogeneity may cause the interpretation ambiguity of the performance of such imaging agents. Regioselective conjugation to antibodies was achieved by enzymatic reaction with lysine and glutamine residues using bacterial and human tissue transglutaminase as catalysts 132.
Milder conditions, faster reaction, and lower concentration of macromolecules were achieved by 6π-Azaelectrocyclization using unsaturated (E)-ester aldehydes with 1,2-dihydropyridines 133 and CuI-catalyzed azide-alkyne cycloaddition 134-136. The “click chemistry“ using N-ε-azido derivatives of the peptides (mono-, di-, tetramericcyclo(Arg-Gly-Asp-D-Phe-Lys)) allowed efficient production of the respective bioconjugates 135. The alkyne derivative of DOTA-tris-tert-butyl ester was coupled to folate -γ-(4-azido)butane amide by similar chemistry 137. A versatile two stage click ligation method based on CuI-catalyzed 1,3 dipolar addition and a thio acid/sulfonylazideamidation (“sulfo-click”) was developed for an efficient systhesis of dimeric peptide agents with various spacer lengths between the peptide units 138. A novel Cu-free click chemistry method has been developed for the conjugation of chelators functionalised with monofluorocyclooctyne to azide-modified peptide in aqueous solution at room temperature 139. Divalent bifunctional chelators based on DO2A and functionalized with 4-aminobenzyl, 4-isothiocyanabenzyl and 4-maleimidobenzyl groups were developed for the synthesis of bivalent imaging agents 140.
In summary, a new technique, so called “click chemistry”, has been developed for the synthesis of precursors for chelator-mediated radiometallabelling. The reactions are fast, regioselective, require small concentration of reagents and take place under mild conditions often in aqueous solution at room temperature.
Current status and directions of 68Ga-based imaging agent development
68Ga-based imaging agents comprising small molecules, large biomolecules, and particles have been explored for the imaging and quantitation of various physiological disorders and biological functions. The molecular imaging of oncological diseases have been investigated most extensively targeting receptors (e.g. G-protein coupled receptor family and human epidermal growth factor receptor (HER) family, folate and urokinase receptors), enzymes, antigenes as well as visualizing downstream biological processes such as angiogenesis, hypoxia, proliferation, apoptosis, glycolysis. Probes for non-targeted imaging of pulmonary and myocardial perfusion and ventilation as well as targeting imaging of inflammation, infection, and mRNA have also been considered and fundamental exploration is ongoing.
Oncological application
Various approaches have been developed for the imaging of certain biological processes involved in cancer diseases with receptor imaging being most thoroughly investigated. There are currently few 68Ga-based imaging agents in routine clinical practice and clinical studies however the number of applications is increasing with acceleration. Somatostatin analogues are already established in clinical routine. Imaging feasibility of GRPR, GLP-1R, MSH, HER2 as well as prostate-specific membrane antigen, osteoblastic bone metastases, amino acid uptake, glucose transport, angiogenesis has been clinically demonstrated in patients 7, 40, 141-145. Extensive basic research is conducted on the development of 68Ga-based probes for the imaging of CCK, CXCR, MSH, gonadotropin releasing hormone (GnRH), NT, folate, integrin, vascular endothelial growth factor (VEGF), urokinase receptors as well as enzymes, antigens, and multidrug resistance (MDR1) P-glycoprotein (Pgp). Imaging probes for visualizing such processes in tumours as proliferation, apoptosis, hypoxia, glycolysis, and angiogenesis as well as bone metastases have also been investigated. A number of particulate agents have been developed for the delivery of imaging reporters or therapeutic agents to the tumour target.
Imaging of G-protein coupled receptor family
Somatostatin receptor imaging
The expression of SSTRs has been found in neuroendocrine tumours, small cell lung cancer, renal cell carcinoma, malignant lymphoma, breast cancer, and prostate cancer. PET-CT using somatostatin ligand analogues labelled with 68Ga has become a new golden standard in imaging of NETs with specificity and sensitivity well above 90% and advantages over conventional radiologic and scintigraphic imaging 14, 146-149. It is the most pronounced example of theranostics 6, 7, 150.
Extensive basic research has been conducted on the development and biological validation of analogues varied in peptide sequence, size and number of peptide rings, chelator (DFO, DTPA, DOTA, NOTA, and their derivatives) and radiometal (Ga, Y, Tc, In, Lu) type. Their receptor binding affinity, internalization and biodistribution have been shown to be dependent on the chemical modifications 2. Fast tumour localization, blood clearance, and renal excretion are typical characteristics of clinically used [68Ga]Ga-DOTA-TATE, [68Ga]Ga-DOTA-TOC, [68Ga]Ga-DOTA-1-Nal3-octreotide ([68Ga]Ga-DOTA-NOC). Structure activity relation studies allowed fine tuning for the agent properties such as receptor affinity, in vivo stability, biodistribution, pharmacokinetics, excretion pathway, and kidney uptake, and pharmacological activity 138, 151-155.
Consistently higher uptake of antagonist SSTR ligand as compared to agonist counterpart was found in forty-eight SST2-positive human tumour frozen sections in vitro 156. Similar peptide sequences, p-Cl-Phe-cyclo(D-Cys-Tyr-D-Aph(Cbm)-Lys-Thr-Cys)D-Tyr-NH2 (where d-Aph(Cbm) is d-4-amino-carbamoyl-phenylalanine) (LM3), p-NO2-Phe-cyclo(D-Cys-Tyr-D-Aph(Cbm)-Lys-Thr-Cys)D-Tyr-NH2 (JR10), and Cpa-cyclo(D-Cys-Tyr-D-Aph(Cbm)-Lys-Thr-Cys)D-Tyr-NH2 were conjugated to DOTA and NODAGA 157, 158. The agents demonstrated antagonistic properties with higher affinity of NODAGA counterparts. Clinical case report with 111In-lablled SST analogues demonstrated higher detection rate for the antagonist counterpart 159. This fact opens possibility for the use of SST analogues not only for the diagnosis of neuroendocrine tumours but also breast carcinomas, renal cell carcinomas, non-Hodgkin lymphomas that express SSTR2 to lesser extent.
The feasibility of quantitation of SSTR density has been demonstrated both preclinically 25, 160 and clinically 24 using [68Ga]Ga-DOTA-TOC. Although a ten-fold higher affinity for the SSTR2 had been demonstrated for DOTA-TATE as compared to DOTA-TOC in vitro in transfected cell cultures 161, no statistically significant difference between [68Ga]Ga-DOTA-TOC and [68Ga]Ga-DOTA-TATE uptake could be observed in vitro in monkey brain tissue sections or in vivo in rat organs expressing SSTRs ( pituitary, adrenal, pancreas) 103. Moreover, clinical study involving 40 patients did not verify the 10-fold higher affinity for the SSTR2 of [68Ga]Ga-DOTA-TATE, on the contrary, standardized uptake value (SUVmax) of [68Ga]Ga-DOTA-TOC tented to be higher 162. Another aspect investigated preclinically is the influence of treatment with octreotide and interferon-a (IFNa) used for biotherapy of NETs on the uptake of [68Ga]Ga-DOTA-TATE. The exposure of the animals to the cold octreotide did not enhance the uptake of the tracer while IFNa did, however the mechanism of the observation was not clear 163, 164. Gene therapy protocols were optimized on the basis of in vivo imaging ([68Ga]Ga-DOTA-TATE) of gene expression and quantitative monitoring of gene transfer 165.
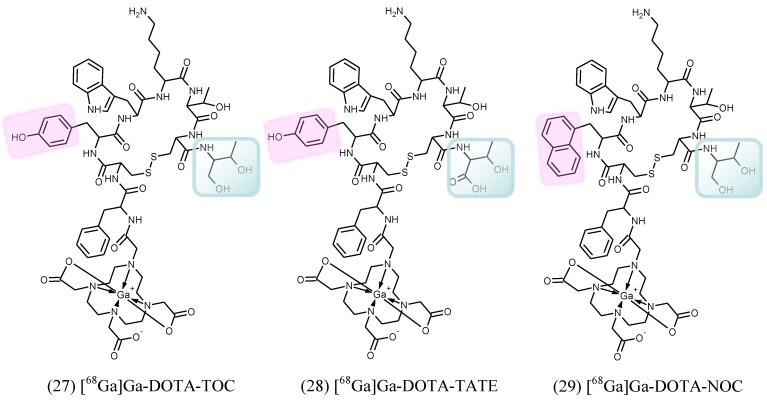
[68Ga]Ga-DOTA-TOC (27), [68Ga]Ga-DOTA-TATE (28) and [68Ga]Ga-DOTA-NOC (29) (Figure 6) are the most commonly used analogues in clinical studies 2, 166, 167. Their pharmacokinetics, blood clearance and target localization rate are compatible with half-life of 68Ga. Renal excretion, short scanning time, high sensitivity and resolution assure high contrast and quality images over organs of interest as well as accurate quantitation. Relatively low radiation dose is one more advantage that should be mentioned. They served for diagnosis, staging, prognosis, therapy selection and response monitoring of NETs and other types of cancers and diseases. [68Ga]Ga-DOTA-TATE was compared with [68Ga]Ga-DOTA-NOC in 20 patients in terms of detection rate and SUVs 168. The agents had comparable diagnostic accuracy with higher SUVmax for the former. One more analogue, [68Ga]Ga-DOTA-2-Nal, Tyr3, ThrNH28-octreotide (DOTA-lanreotide, DOTA-LAN) was successfully used for lung and thyroid tumour detection 169.
Fig 6.
Structural formulae of the clinically used somatostatin analogue imaging agents. TOC stands for D-Phe-Cys-Tyr-D-Trp-Lys-Thr-Cys-Thr(OH); TATE stands for D-Phe-Cys-Tyr-D-Trp-Lys-Thr-Cys-Thr; and NOC stands for D-Phe-Cys-Nal-D-Trp-Lys-Thr-Cys-Thr(OH). The differences in structures are highlighted.
The individualized diagnosis has been practiced in the selection of patients for PRRT, target definition for fractionated stereotactic radiotherapy (FSRT) planning, target volume delineation for intensity modulated radiotherapy 170-174. The diagnosis on the cellular and molecular level and determination of the disease associated biomarkers provides basis for the treatment optimization and efficacy for a particular patient 175. The personalized therapy planning necessity was demonstrated in retrospective study of ten patients examined with [68Ga]Ga-DOTA-TATE 176. It was concluded that the radiotherapeutical dose should be determined by the tumour burden since the latter influences the radioactivity distribution to the healthy organs, and in particular the higher burden decreases the radiation accumulation in the kidney. The starting point of PRRT after preceding cold octreotide therapy in patients with NETs could be determined 177. These imaging agents improved the detection rate and diagnostic accuracy 178, 179. They were used for therapy planning and monitoring response to treatment 180, as well as influenced and changed the therapeutic course 181-184.
The majority of the clinical studies have been conducted in the field of GEP NETs however considerable number of other indications has been reported demonstrating wider application possibilities. [68Ga]Ga-DOTA-NOC was successfully used for the assessment of NETs 185, well-differentiated medullary thyroid carcinoma (MTC) 186, bronchial carcinoid (BC) and Von-HippelLindau (VHL) disease 187, alveolar rhabdomyosarcoma with neuroendocrine differentiation 188, carotid body chemodectomas (CBCs) 189, idiopathic pulmonary fibrosis 190. [68Ga]Ga-DOTA-TOC was found valuable for therapy planning and accurate diagnosis of multiple endocrine neoplasia 184, sudden onset of vision problems 183, duodenopancreatic NET 182, autoimmune thyroid disease like Graves' disease and Hashimoto's disease 191. Patients affected by prostate cancer with bone metastases were examined with [68Ga]Ga-DOTA-TOC 192. However, due to the low expression of SSTR2 and SSTR5 the uptake in the lesions was low and the signal was weak. Even broader areas were covered by [68Ga]Ga-DOTA-TATE. Benign and malignant thrombi were distinguished in a patient affected by pancreatic NET tumour with hepatic and regional lymph node metastases 193, 194. It was found relevant for the diagnosis and therapy planning for meningiomas 195, recurrent medullary carcinoma 196, plaque imaging and characterization in the coronary arteries 197. In addition, the usefulness of 68Ga-labelled somatostatin analogues has been discussed in the context of other available imaging techniques for staging of lung neuroendocrine cancer and therapy selection 147 as well as for the evaluation of Hurthle cell thyroid carcinoma 198, and neuroectodermaltumours 167.
The large number of clinical applications of somatostatin ligand analogues triggers investigations that refine the examination protocols and interpretation of results. Somatostatin receptors are expressed also physiologically in healthy organs and the awareness of that is very crucial for correct interpretation of examination results, and possible causes for false negative or positive findings should be kept in mind. Distribution of [68Ga]Ga-DOTA-TATE and organ uptake (SUV) in healthy volunteers has been studied and compared with in vitro findings 199. The organs with physiological expression of SSTR2 as well as metabolic and excretion sites (pituitary, adrenals, pancreas, spleen, thyroid, stomach wall, liver, salivary glands, bowel, pelvicalyceal system, kidneys, urinary bladder) demonstrated distinguished uptake. Forty-three patients with NETs were examined with [68Ga]Ga-DOTA-TOC in order to distinguish pathological and physiological uptake of the head of pancreas 200. The uptake that was similar to that of liver was considered physiological. The findings stressed importance of accurate quantitation of the uptake for the avoidance of false-positive diagnosis. The uptake in the head of pancreas was also considered physiological in the retrospective study of 100 patients 201 and specifically designed study of 96 patients 202 since the accumulation was stable over time regardless of the uptake intensity or shape. [68Ga]Ga-DOTA-TOC scans of 165 patients with various thyroid pathologies were analyzed 203 and in 8 cases of normal thyroids with increased uptake, follow-up examination after 6-14 months did not show any thyroid pathology. Another case study using [68Ga]Ga-DOTA-TOC reported on the uptake in wandering spleen which could mistakenly be interpreted as malignant process instead of physiological expression of SSTR2 in spleen in an unusual position 204.
The option to quantify the disease underlying processes and provide personalized diagnosis and therapy is one of the crucial advantages of PET-CT. Various methodologies and approaches are being considered with the aim to assure accuracy and unambiguity of the quantitation. The uptake of [68Ga]Ga-DOTA-NOC measured in SUVmax correlated with pathologic features and was suggested as a prognostic tool characterizing stable disease with SUVmax cutoff value ranging from 17.9 to 19.3 205. The quantitation of diagnostic procedures is getting refined. The correlation between [68Ga]Ga-DOTA-TOC (SUV) and SST2 mRNA was investigated in 120 patients and a novel normative database was created for the accurate quantitative evaluation. It may improve diagnostics, treatment monitoring and therapy of SST expressing tumours or inflammation on a molecular basis 206. In another study the evaluation of the response to the radiotherapeutic treatment was conducted using tumour-to-spleen SUV ratio which was found more accurate measure than solely SUVmax 180. Treatment with cold somatostatin analogue did not reduce the binding of [68Ga]Ga-DOTA-TATE, but improved the tumour-to-background ratio 207. The detection rate of pre-therapeutic [68Ga]Ga-DOTA-TATE/PET-CT was found higher as compared to sequential planar scans with [177Lu]Lu-DOTA-TATE (9% of the lesions not detected) 208. Most importantly the highest detection concordance was observed at later time point (72h) thus indicating the potential of [68Ga]Ga-DOTA-TATE for pre-therapeutic dosimetry and dose planning.
The superiority of 68Ga-labelled somatostatin analogues in terms of specificity, sensitivity, staging accuracy, detection rate, quantitation, acquisition time to [18F]-FDG 210, [18F]F-DOPA 211, 123I-metaiodobenzylguanidine (123I-MIBG) 212, 213, and [111In]In-octreotide (Octreoscan®) (Figure 7) 209, [18F]-NaF and [99mTc]-dicarboxypropanediphosphonate 214 has been demonstrated. Advantage of 68Ga/PET-CT in terms of detection rate over ultrasonography, CT, MRI, and skeletal scintigraphy was confirmed in over 5800 clinical cases 7.
Fig 7.
A 69-y-old man with low grade metastatic midgut NET. (A) Arterial-phase CT shows multiple arterially enhancing and low-attenuation liver metastases. (B and C) Anterior and posterior whole body 111In-DTPA-octreotide scintigraphy shows low grade (Krenning score), 1) mesenteric metastases (arrow) but no liver metastases. (D and E) Axial 111In-DTPA-octreotide SPECT at level of spleen shows heterogeneous liver uptake with no discernable liver deposits. (F) Maximum-intensity-projection [68Ga]Ga-DOTA-TATE PET shows multiple deposits in liver and mesentery. (G) Coronal [68Ga]Ga-DOTA-TATE PET anterior to kidney shows multiple liver metastases. (H) [68Ga]Ga-DOTA-TATE PET of G. (I) Axial [68Ga]Ga-DOTATATE PET at level of spleen shows multiple liver metastases. (J) Axial [68Ga]Ga-DOTA-TATE PET at level of spleen shows multiple liver metastases. Reproduced by permission of SNMMI from 209.
Further unique application is the use of hand-held gamma probe in combination with either [68Ga]Ga-DOTA-TATE or [68Ga]Ga-DOTA-NOC intraoperatively in order to localize metastases and primary tumours 215. The surgery was conducted 30 min after agent injection. One of the most significant advantages of the radioguided surgery (RGS) was the differentiation of scar tissue from small tumour metastases. RGS resulted in change in the operative procedure in 56%. In addition, overexpression of SSTR has been found in patients with inflammatory bowel diseases, pulmonary diseases, rheumatoid arthritis, Sjogren's syndrome, Grave's disease 43, 216.
Imaging gastrin releasing peptide receptors
Breast, prostate, gastrointestinal, and small cell lung cancers overexpress bombesin receptors, specifically gastrin releasing peptide (GRP) receptors. An efficient diagnostic tool for the staging of prostate cancer specifically metastasis spreading to soft tissue is required in order to avoid the understaging or overtreatment of prostate cancer. Around 300 studies on the development of corresponding ligands labelled with 68Ga 74, 217-219, 18F, 64Cu, 90Y, 99mTc, 111In, 125I, 185/187Re, 177Lu have been conducted preclinically as well as clinically in patients. Twelve clinical studies several of which utilizing 68Ga were performed using analogues with agonistic function 143. [68Ga]Ga-DOTA-PEG2-[D-Tyr6,βAla11,Thi13,Nle14]bombesin (6-14) ([68Ga]Ga-BZH3) demonstrated high in vivo stability and uptake in a pancreatic carcinoma model 217, 220 and gastrointestinal stromal tumour (GIST) 142 in patients, however detection rate was higher for [18F]-FDG. Nonetheless the quantitation of [68Ga]Ga-BZH3/PET-CT uptake in patients with recurrent gliomas allowed differentiation between low- and high-grade gliomas thus demonstrating the advantage over [18F]-FDG 221. Although tumours with smallest ones of 5 mm were detected in patients affected by prostate cancer using [68Ga]Ga-DOTA-BOM already 15-25 minutes post injection, the nonspecific radioactivity accumulation in the upper abdomen presumably in pancreas deteriorated the tumour detection in that area 222. Comparative study of [68Ga]Ga-DOTA-CHCO-Gly-4-aminobenzyl-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH ([68Ga]Ga-AMBA) and [18F]FCH in PC xenograft-bearing mice revealed superior performance of the former in terms of tumour uptake and image contrast 223.
Although bombesin-based tracer agonists have demonstrated high binding affinity to GRPRs and good radioligand internalization, they have strong potency and mitogenicity even in trace amount as well as high and maintained uptake in the normal GRPR positive organs that results in poor contrast of images and radiation dose to normal tissue. In order to exclude the agonistic function and related side effects, a bombesin antagonist was developed and showed higher uptake compared to the agonist counterpart 224. The biodistribution in PC-3 and LNCap tumour-bearing nude mice of 68Ga-labelled DOTA-4-amino-1-carboxymethyl-piperidine-D-Phe-Gln-Trp-Ala-Val-Gly-His-Sta-Leu-NH2,was studied in order to investigate the difference between androgen-dependent and androgen-independent tumours. The pharmacokinetics was similar however the uptake in PC-3 tumour was higher. In the further development DOTA-4-amino-1-carboxymethyl-piperidine was substituted with NODAGA and 68Ga-labelled counterpart demonstrated high contrast tumour accumulation in PC3 tumour-bearing nude mice, however uptake in abdomen organs was also high at 1 h time point 225. The excretion pathway of these series of analogues was modulated and shifted from the liver by introducing NOTA bifunctional chelator via PEG2 linker 226. One carboxylic arm of NOTA was used for the conjugation thus resulting in positively charged [68Ga]-NOTA complex moiety. The modification resulted in improved biodistribution pattern and image contrast. Heterodimeric peptide ligand for imaging of breast cancer comprising RGD and bombesin motifs (RGD-BBN) was designed in order to compensate for the variable GRPR and αvβ3 receptor density in estrogen dependent and estrogen independent breast cancers and demonstrated higher tumour uptake in mice than monomeric BBN did 227. In another report the performance of the heterodimeric [68Ga]Ga-NOTA-RGD-BBN was compare to both [68Ga]Ga-NOTA-RGD and [68Ga]Ga-NOTA-BBN in a PC3 tumour model revealing higher tumour uptake 228.
Bombesin receptor ligand analogues have been used in clinical studies however the high potency of agonists brings up challenges requiring synthesis with high specific radioactivity in order to exclude pharmacological effects. Antagonists even though they do not internalize have shown improved imaging capabilities. Given recent achievements in the design of antagonists and experience with SST analogues demonstrating advantages of antagonists, the future development will most likely move in the direction of bombesin antagonists.
Imaging GLP-1R, CCK, CXCR, MSH, GnRHR, NT receptors
The development of new peptide based agents is expanding with novel targets of interest. High incidence and density expression of GLP-1R has been found in insulinomas and pancreatic beta cells. Metabolically stable agonists of GLP-1R, exendin-3 and exendin-4, have been labelled with 68Ga via DOTA moiety and demonstrated specific uptake in mouse tumour models as well as in pancreatic beta cells is healthy non-human primate 229-231. Moreover, examination of a patient affected by insulinoma revealed not only multiple small liver metastases but also paraaortal lymph node lesions that could not be conclusively detected by computed tomography, ultrasound, [18F]FDG/PET-CT or [11C]HTP/PET-CT (unpublished data). The CCK-2 is upregulated in NETs and could complement the diagnostic means of NETs with low expression of SSTRs such as stromal tumours, medullary thyroid carcinoma (MTC), and neuroendocrine gut tumours. Linear and cyclic peptide analogues were designed and preclinically evaluated 232, 233. Several cyclic monomeric and dimeric peptide ligands targeting chemokine receptor subtype CXCR4 which is up-regulated in human solid tumours as, for example mammary cancer, prostate cancer, melanoma, glioma, lymphoma, cervical adenocarcinoma demonstrated specific binding 234-237. A number of 68Ga-labelled α-MSH analogues have been constructed and preclinically evaluated for melanoma imaging and staging. The content, lipophilicity and charge were varied in order to optimize the retention in tumour and kidneys 139, 238-241. Non-peptidic probe comprising benzamide derivative that specifically binds to melanin demonstrated uptake in B16F10 tumour model 242. GnRHR expressed on the reproductive system 243, 244 and NT receptor subtype 1 involved in neoplastic processes and up-regulated lung, prostate, colon, pancreatic and breast cancers 245 were targeted by 68Ga-based peptide and peptoid-peptide hybrid agents.
Human epidermal growth factor receptor (HER) family
HER family receptors are overexpressed in head-and-neck, lung, breast, colorectal, ovary, and urothelial cancer cells. A number of imaging agents labelled with 68Ga has been preclinically evaluated for the visualization of the receptors: natural peptide EGFR ligand (DOTA-hEGF) 246, Affibody® molecules 247-249, 2-helix Affibody derivative 250, F(ab')2 fragment of trastuzumab 251, as well as tyrosine kinase inhibitor 252. An innovative approach where various affibody molecules were pre-administered and allowed saturation of physiological EGFR uptake prior to the injection of [67/68Ga]Ga-DOTA-hEGF was successfully proved with the aim to improve the image contrast 253. Fast target localization, blood clearance and normal tissue wash out providing high image contrast as well as high kidney uptake are characteristic to various Affibody molecule based agents. A clinical imaging of metastatic breast cancer using [68Ga]Ga-ABY-002 proved to be an efficient method for the non-invasive determination of HER2 status of metastases not amenable to biopsy 40. Two-helix Affibody derivative ([68Ga]Ga-DOTA-MUT-DS) 250, Herceptin fragment ([68Ga]Ga-DOTA-F(ab')2) 254, camelid heavy-chain antibody variable domain based ligand ([68Ga]Ga-Df-Bz-NCS-7D12) 255 were developed from a larger molecules in order to accelerate pharmacokinetics and demonstrated compatibility with 68Ga half-life time frame.
Pre-targeted imaging
Although the number of antibody based radioimmunotherapeutic agents is considerable, their use is limited by the slow pharmacokinetics and blood clearance that cause high radiation dose to the normal organs 256. In the case of imaging the achievement of optimal image contrast might require as long as 2-4 d. In order to improve the tissue penetration and facilitate clearance and excretion, the size of an antibody was reduced to antibody fragments (F(ab')2, Fab'), medium-sized proteins and small peptides with high affinity for the same target 2, 257. Another method that has attracted strong interest is pre-targeting. It uses bispecific antibody (bsmAb) with two binding sites for the interaction with a cell target and a hapten molecule carrying radionuclide. First bsmAb is administered and after its localization at the target, clearance from the blood, and washout from the normal tissue, the radiolabelled hapten molecule is administered. The latter has fast pharmacokinetics so that high contrast imaging is possible within short time. Hapten molecules comprising histamine-succinyl-glycine (HSG) motif and chelate moiety (30) (Figure 8) 258-261 have been used for the imaging of carcinoembryonic antigen (CEA) pre-targeted with anti-CEA bsmAb 262-264.
Fig 8.
The principle of pre-targeting: A) An example structure of peptide hapten molecule containing histamine-succinyl-glycine residues and coupled to NOTA chelate moiety complexed with 68Ga (***); B) Bispecific antibody with two antigen binding F(ab')2 arms (*) and one anti-hapten binding F(ab) arm (**) interacts with the target antigene (CEA). 68Ga-labelled hapten molecule (***) binds to the bispecific antibody (**) on the next step.
Transplantation of islets of Langerhans is a promising treatment for type 1 diabetes mellitus. Avidin-covered agarose resin microbeads were used as a model for islet transplantation in mice 265, 266. After intraportal transplantation they were visualized in hepatic volume by [68Ga]Ga-DOTA-(PEG)2-biotin (31) (Figure 9).
Fig 9.
Left) Structure of [68Ga]Ga-DOTA-(PEG)2-biotin (31) analogue used for the imaging of avidin-covered agarose resins transplanted in mice. Right) Coronal views showing total tracer uptake in left kidney (black arrows) and liver (red arrows) in transplanted (top left) and non-transplanted mice (top right). Pretreatment with unlabelled biotin displaced a majority of the tracer uptake in both transplanted (bottom left) and non-transplanted mice (bottom right). The color bar indicates SUV ranging from 0 to 6. All images are summations of the initial 20 minutes following tracer administration. Reproduced from 265.
Imaging angiogenesis
Growth of cancer cells and healing of ischemic injuries require formation of new capillaries which is known as angiogenesis process. Monitoring of angiogenetic activity would allow validation of antiangiogenic drug efficacy on early stage and on personalized basis. Molecular targets such as integrin receptors, vascular endothelial growth factor (VEGF) receptors, and matrix metalloproteinases 9 (MM9) have been identified and the respective imaging agents were developed 2.
The most widely studied is the imaging of integrins using radiolabelled peptide ligands comprising arginine-glycine-aspartic acid (RGD) sequence for binding to αvβ3 integrin receptors 267. The influence of the major components of the agent structure such as DOTA and NOTA chelator moiety, PEG linker as well as multivalency on the pharmacokinetics has been studied preclinically in mice with various cancer xenografts or ischemic or atherosclerotic models 80, 99, 120, 268-274. The enhancement of the valency from mono- to di- to tetrameric cyclic RGD commonly results in increased uptake, however the mechanism is unclear 275-278. Further optimization is required in order to find fine balance between the tumour uptake on one hand and blood clearance and kidney accumulation on the other hand. The performance of 68Ga-based agents has been compared with clinically used [18F]Galacto-RGD and demonstrated comparable or better pharmacokinetics. Another advantage of 68Ga-based agents is more efficient and cost-effective as well as simpler production chemistry 80.
Single chain VEGF (scVEGF, MW ≈ 28 kDa) which is a functionally active single-chain version of VEGF 279, 280 was modified with PEG linkers in order to modulate pharmacokinetics. 68Ga-labelling was accomplished via HBED or NOTA chelate moiety 117, 281. The agents accumulated in the tumour xenografts in mice, and the further development is focused on the improvement of tumour-to-background ratio and addressing high kidney uptake.
The first studies in the development of peptide-based ligands to MM9 proved the concept however further fundamental research is required 282. A peptide (HWGF) probe developed for dual-labelling with 68Ga and IRDye 800CW, respectively for PET and near infrared imaging, demonstrated specific binding to the metalloproteinases-2 and -9 (MMP-2/-9) 283. The dual-labelling strategy provides the advantage of using identical agent for both optical and nuclear imaging.
Imaging hypoxia, bone metastases, proliferation, glycolysis
Small biologically active molecules have also been tagged with 68Ga for the visualization of hypoxia, glucose transport, cell proliferation, and bone metastases. Although most of them are currently on preclinical level however these examples demonstrate potential of 68Ga for the diversity of imaging agents like 99mTc-based radiopharmaceuticals. Biphosphonates 144, 284-287 and 68Ga-labelled ethylene diamino-N,N,N',N'-tetrakis-methylene phosphoric acid 288, 289 were found useful for early diagnosis of bone metastases. An encouraging illustration is 68Ga-labelled bisphosphonate probe (BPAMD (32), Figure 10) that successfully proved the concept in a clinical examination of a patient with prostate cancer. Osteoblastic bone metastases were visualized with high contrast and detection rate (Figure 11) 144. Analogues comprising nitroimidazole, mercaptobenzynamine or 5-nitroimidazole derivatives coupled to DOTA have been investigated for the imaging of hypoxia which is an important parameter in tumour and myocardial ischemia physiology 290-293. The monitoring of hypoxia could contribute to improved diagnosis, prognosis, treatment planning and validation of response to therapy. DOTA- and NOTA-coupled alanine, lysine and tyrosine analogues targeting transporters for the cell proliferation visualization showed uptake in animal tumour models 294-296. [68Ga]Ga-DOTA-2-deoxy-D-glucosamine demonstrated higher tumour-to-organ ratio as compared to [18F]FDG 297.
Fig 10.
Low molecular weight vectors for the labelling with 68Ga and subsequent imaging of bone metastases (32), folate receptors (33), MDR1 Pgp (34-35), myocardial perfusion (36-39), and necrosis (40).
Fig 11.
[68Ga]BPAMD was injected i.v. into a patient with known extensive bone metastases of prostate cancer, and revealed intense accumulation in multiple osteoblastic lesions in the central skeleton, ribs and proximal extremities: a) coronal PET; b) sagittal PET/CT; c) 18F-fluoride PET (sagittal). Reproduced from 144.
Imaging of tumour with particulate agents
Nano- and microparticles are used as vectors to deliver imaging reporters or therapeutic agents to the target. Radiolabelled nanoparticles such as 68Ga-labelled albumin nanoparticles can be used for the imaging and localization of sentinel lymph nodes in breast and melanoma cancer not only for diagnosis and prognosis but also for therapy and surgery planning 298. Commercially available albumin nanoparticle kit (99mTc Nanocoll) was labelled with 68Ga, however, the biodistribution of the particles in rat revealed unexpected accumulation in lung 88. A novel mannosylated human serum albumin (MSA) was synthesized by coupling α-D-mannopyranosylphenylisothiocyanate to HSA and then to NOTA active ester (NOTA-MSA) 299. High uptake was observed in liver, spleen and femur with RES content after intravenous injection to mice, and after subcutaneous injection into mice footpads it migrated to lymph nodes. Novel particles amenable to functionalization and labelling with 68Ga, e.g. organic CdSe/ZnS quantum dots (QDs) incapsulated by polysorbate 60 300, nano-graphene oxide sheets (GOs) 301, cobalt-ferrite 302, and bifunctional zeolite Y particles 303, demonstrated promising results in mouse tumour models. GOs was chemically modified and coupled to NOTA for 66Ga-labelling. The imaging agent showed specific uptake in tumour vasculature CD105 301. QDs functionalized with such amphiphiles as RGD-C18, NOTA-C18, mannose-C18, lactose-C18, and labelled with 68Ga were specifically accumulated in U87MG human glioma xenogratfs in mice 300. Micelle agents were formed by the aggregation of amphiphilic ligands comprising NOTA or DOTA coupled to an α-alkyl chain via acetate pendant arms 304, 305. Direct proportionality was found between the agent accumulation in rat liver and the length of the pendant α-alkyl chain and thus lipophilicity of the ligands 305.
Miscellaneous imaging targets in oncology
Breast, cervical, ovarian, colorectal, nasopharyngeal, renal, and endometrial cancers express folate receptors (FR). [111In]In-DTPA-folate is clinically used and motivates development of an imaging agent comprising 68Ga. Folic acid has been conjugated to NOTA and DOTA (Folate (33), Figure 10) based chelators and resultant analogues were validated in mouse tumour models 306, 307. The improvement of tumour-to-kidney ratio and protection of kidneys remains a challenging task.
An agent comprising 9 amino acid residues, DOTA and 68Ga was developed for the detection of the receptors of urokinase-type plasminogen activator (uPAR) upregulated in gastric, colorectal, and breast cancers 308. Its uptake in human glioblastoma cancer xenograft mouse model could be observed, though tumour-to-background ratio was low.
Tria- and diabody derivatives of an antibody to epithelial cell adhesion molecule (EpCAM) expressed in tumours were constructed with the goal to accelerate the blood clearance and improve the image contrast 257. The diaboby variant labelled with 68Ga via HBED-CC showed faster blood clearance and normal tissue washout as well as rapid tumour uptake in mice A-431 xenograft.
The response to therapy can be evaluated by monitoring expression of multidrug resistance (MDR1) P-glycoprotein (Pgp). 68Ga-labelled hexadentate Schiff base 309 (MFL6.MZ (34), Figure 10) and [68Ga]Ga-(bis(3-ethoxy-2-hydroxy-benzylidene)-N,N'-bis(2,2-dimethyl-3-propyl)ethylenediamine) [68Ga]Ga-ENBDMPI 310 (3-ethoxy-ENBDMPI (35), Figure 10) were used for the imaging of Pgp activity dependent on the extracellular acidosis. [68Ga]Ga-ENBDMPI demonstrated better balance between uptake and inhibition in Pgp-positive tumour cells 310.
Annexin A5 was used for the development of a probe for the imaging and quantitation of apoptosis in the evaluation of therapy response 311. Regiospecific coupling of the protein to DOTA maleimide was achieved via Cys2 and Cys165 amino acid residues thus maintaining the active binding sites intact. The resultant bioconjugate labelled with 68Ga demonstrated statistically higher uptake after cancer therapy in mouse model of hepatic apoptosis.
Rapid progress has taken place in imaging of prostate cancer reaching clinical studies within short period of time. The urea-based inhibitor of the prostate-specific membrane antigen (PSMA) 312 was modified in order to adjust all components required for the strong interaction with PSMA. For one of the analogues HBED-CC was added as a lipophilic side chain to the hydrophilic pharmacophore Glu-NH-CO-NH-Lys for the interaction with the active binding site of PSMA. This analogue demonstrated enhanced internalization in LNCaP cells in vitro and high accumulation in mice LNCaP tumour xenografts. Thorough study on structure-activity relation has been performed by 313. [68Ga]Ga-HBED-CC-Lys-NH-CO-NH-Glu was taken further to the clinical study and compared with [18F]FECH 145. Prostate carcinoma relapses could be clearly visualized only by 68Ga-agent. The biodistribution in 37 patients demonstrated uptake in kidneys, salivary glands, lacrimal glands, liver, spleen, and bowel (Figure 12) 314. The lesions were detected with excellent contrast already at 1 h p.i. even at low PSMA levels.
Fig 12.
68Ga-PSMA PET/CT of patient 22 who received the lowest dose of radiotracer (52 MBq). Red arrows point to several small lymph nodes with clearly visible pathological tracer uptake. A1 CT, A2 PET, B1 fusion of PET and CT, B2 MIP. Reproduced from 314.
68Ga-based agents for imaging of thrombosis, atherosclerosis, intracoronary radiation therapy, imaging agent for the diagnosis of hepatobiliary and renal disorders, mitochondrial-targeting imaging of cancer were suggested earlier 2. However, to my best knowledge no related studies have been published during last five years.
Antisense oligonucleotide based tracers
A number of 68Ga-labelled antisense oligonucleotide agents were developed for in vivo imaging of gene expression and tumours that express ras oncogene point mutations 2, 315. The oligonucleotides varied in size (15-30 mer), sequence and basic structure: phosphodiester, phosphorothioate, 2'-O-methyl phosphodiester, locked nucleic acid (LNA), LNA-DNA mixmer (LNA and DNA nucleotides in alternation along the sequence), and peptide nucleic acid 316-320. The uptake of the 68Ga-labelled analogues in non-hybridization specific tissue was most likely mediated by scavenger receptors. The rat distribution pattern of 68Ga-labelled oligonucleotides varied and was influenced by sequence and length rather than chelator type 315, 321. The mechanism of the oligonucleotide agent uptake is complex and not understood, and more fundamental research is required in order to elucidate the applicability of this category of vectors for imaging purposes.
Myocardial perfusion, necrosis, and lung ventilation
Imaging agents for myocardial perfusion were constructed using either small lipophilic chelators or particles. Lipophilic and cationic gallium complexes with bis- and tris(salicylaldimine) and bisaminothiolate (36, 37) (Figure 10) analogues have shown myocardial uptake and retention in rodents. However, the optimization of the lipophilicity of the agents so that the higher uptake in the heart is not accompanied by the high uptake in the liver is very challenging. Highly lipophilic [67Ga]Ga-bisaminothiolate complex was accumulated during the first-pass extraction with high uptake in the healthy heart, but not in the permanently legated coronary artery of infarct Sprague-Dawley rat heart 322. Still the imaging quality was deteriorated by high liver uptake. Tris(4,6-dimethoxysalicylaldimin-N,N'-bis(3-aminopropyl) -N,N'-ethylenediamine (BAPEN (38), Figure 10) 323, hexadentate bis(salicylaldimine) ligands (BAPDMEN (39), Figure 10) 324 with variable aromatic ring substituents for lipophilicity modulation have also demonstrated hepatobiliary excretion. The distribution in rat varied dependent on the imaging agent and animal species. Four analogues of BAPEN with varied substituents on the salicyl ring and a number of salicyl rings that were labelled with 68Ga and tested in pigs at rest and under adenosine stress did not reveal correlation with myocardial perfusion measured with [15O]water and their myocardial accumulation was not determined by perfusion 325.
Various human serum albumin (HSA) based nano- and microparticles have been developed for the lung function monitoring by measuring pulmonary and myocardial perfusion as well as vascular permeability 2. The first studies on 68Ga-labelled HSA microspheres were conducted in the 1970s but nowadays the interest is revitalized. The labelling with 68Ga was accomplished either by co-precipitation with the commercially available macroaggregated albumin (MAA) for 99mTc-labelling 88, 89 or by complexation with DOTA chelator attached to HSAM via surface lysine amino groups and p-SCN-Bn-DOTA 326, 327. Simultaneous formation of MMA and its 68Ga-labelling under microwave heating resulted in stable agent that was successfully tested in a piglet model of pulmonary embolism 87. [68Ga]Ga-MAA has been used to quantify spatial differences in pulmonary blood flow in rats in different postures 90. The visualization of pulmonary thromboembolism and perfusion was conducted prior selective intra-arterial therapy using [68Ga]Ga-MMA 7.
N,N'-bis(diethylenetriaminepentaacetic acid)-pamoic acid bis-hydrazide (bis-DTPA-PA (40), Figure 10) was labelled with 68Ga for the visualization of necrosis that occurs in acute myocardial infarction, stroke, chronic heart failure, neurodegenerative disorders, allograft rejection, and inflammation 328.
Acute pulmonary emboli requires fast evaluation of lung ventilation distribution for accurate diagnosis. PET provides necessary prerequisites such as high resolution and quantitation. 68Ga-labelled carbon nanoparticles in the form of inhalation aerosol ([68Ga]Ga-GallGas) were tested in piglets with lobar obstruction or diffuse airway obstruction 91. The same animals were examined with [99mTc]-Technegas aerosol for comparison (Figure 13). The heterogeneity of the airway response in the case of bronchoconstrictive challenge was more distinguishable in the investigation with [68Ga]Ga-GallGas. The distribution of the aerosol in the lung, alveolar space of healthy volunteers was homogeneous and without bronchial deposition 105. In an intraindividual comparative study ten patients with suspected pulmonary embolism underwent conventional ventilation-perfusion (V/Q) scintigraphy and PET-CT V/Q imaging after inhalation of 68Ga-carbon nanoparticles (“Galligas”) and administration of 68Ga-macrosggregated albumin 92. PET-CT provided images of higher resolution and possibility of lung function quantitation, and thus more accurate diagnosis. The short half-life of 68Ga enabled more flexible acquisition protocols.
Fig 13.
Lung ventilation imaging using carbon particles behaving as a “pseudo gas” for diagnosis of acute pulmonary emboli. Dual study using [68Ga]Ga-GallGas (left) and [99mTc]-Technegas (right) in healthy piglets demonstrating similarity in ventilation distribution (A), in piglets with lower lobar obstruction (B), and in the piglets with diffuse airway obstruction induced by infusion of methacholine (C).
Imaging inflammation and infection
The development of 68Ga-based imaging agents of various action mechanisms for the differentiation and diagnosis of inflammation and infection started during last five years 77, 86, 282, 329-338. Several peptide analogues were designed for the visualization of vascular adhesion protein-1(VAP-1) expressed on endothelial surface in inflammation and infection. A ligand to VAP-1, nine amino acid residue linear peptide conjugated with DOTA, was labelled and studied in rats with diffuse Staphylococcus aureus tibial osteomyelitis (infection), in rats with healing cortical bone defect (inflammation) demonstrating specific binding and capability to differentiate between inflammation and infection 329, 332. In order to prolong the in vivo stability, peptide was coupled to PEG4 spacer 333. The resulting agent demonstrated higher stability and target-to-background ratio. The renal excretion was modulated also by PEG modification introduced to another ligand to VAP-1 (cyclic 17 amino acid residue peptide; CARLSLSWRGLTLPSK) 331, 334. The accumulation in the site of inflammation induced in rats was clearly observed in the whole body coronal PET image. The important function of such agent must be the differentiation between tumour and inflammation/infection. However, this novel tracer demonstrated also uptake in the melanoma xenografts in mice.
Siderophores that are iron transporters in bacteria, fungi, and some plants were used for the development of imaging agent for invasive pulmonary aspergillosis (IPA) caused by Aspergillusfumigatus 335. Desferri-triacetylfusarinine C (TAFC) and desferri-ferricrocin (FC) siderophores were labelled with 68Ga. [68Ga]Ga-TAFC demonstrated specific binding, fast blood clearance and high stability in a rat model of A. fumigatus infection while [68Ga]Ga-FC showed prolonged retention in blood and rapid metabolism. TAFC and ferrioxamine E (FOXE) 336 were selected from a group of 7 various siderophores as best candidates for Aspergillus infection imaging in terms of 68Ga complex stability, uptake specificity, rapid clearance and elimination. Both agents demonstrated lung focal uptake in invasive aspergillosis rat model correlated with the severity of disease and high image contrast 337.
[68Ga]Ga-citrate was found effective in clinical examination of bone infection such as osteomyelitis and diskitis. The overall accuracy of diagnosis was of 90% 77, 338. Apo-transferrin was labelled with 68Ga and subsequently used for the bacterial infection imaging in rat model with Staphyococcusaureus 86. Infection site could be clearly visualized within 1 h after intravenous injection.
Future perspective
First generators were introduced in the late 1950s early 1960s and 68Ga-based imaging agents were available already from the 1970s but further progress was precluded by the absence of reliable sources of the generators as well as their properties and quality. During last decade PET has been recognized as a clinically relevant and valuable diagnostic technique providing quantitative information on the physiological status of a disease and possibility to perform a whole-body scan in a single examination. There are many interrelated factors that contributed to the clinical acceptance of PET in general and blossom of 68Ga/PET in particular. Technological aspects include achievements in improved sensitivity, resolution, accurate quantitation, and introduction of hybrid systems, PET-CT and PET-MRI; availability and increasing assortment of commercial 68Ge/68Ga generators; advances in biological research and biotechnology providing identification of biomarkers; accessibility of automated synthesizers for GMP compliant production of imaging agents. The automation may also provide possibility for the harmonized and standardized multicentre clinical studies that in turn will accelerate the introduction of new radiopharmaceuticals as well as their regulatory approval. Steady progress is being observed in PET radiopharmaceutical regulations and multicenter trials. The plausible correction and increase of the parent 68Ge breakthrough limit in the radiopharmaceutical preparation (European Pharmacopoeia) may together with the labelling methods of high selectivity towards 68Ga(III) at room temperature allow for kit type production at radiopharmacy practice and consequently even wider allocation and use of 68Ga-radiopharmaceuticals. The feasibility of PET-CT being cost-effective and cost-saving has been demonstrated, especially in combination with cheaper, as compared to cyclotron, generator produced 68Ga radionuclide. Further technological development might render cost reduction and consequently improve profitability and affordability. This in turn may stimulate investments in radiopharmaceutical research and development.
The above mentioned factors have played prominent roles in accelerating the growth of 68Ga-radiopharmaceutical field and they will stimulate the investments into development of new imaging agents. The growth has been reflected in the explosive increase of related publication number and more 68Ga-agents reported at an ever-increasing rate. However the central role in the expansion of nuclear medicine belongs to identification of unmet medical needs as well as development and introduction of new specific imaging agents and thus achievements in chemistry.
Methods for the enhancement of 68Ga quality have been developed and automated however their further simplification is required. Mono- and bifunctional chelators for fast, efficient and selective complexation with 68Ga are established and further development will be directed towards radiopharmaceuticals comprising chelators that are highly selective for Ga(III), form efficiently and rapidly complexes at room temperature, broad pH range and low concentration. Conjugation chemistry will further develop towards fast and regioselective coupling reactions at mild conditions and low concentrations.
The 68Ga-applications in oncology will continue growing in harmony with the introduction of new receptor specific peptides and high affinity proteins for tumour-type specific diagnosis and treatment that would allow personalized approach of accurate quantitative diagnosis and staging for subsequent selection and planning of therapeutic means as well as monitoring response to the treatment. The imaging of receptors with peptidic agents is the most explored field. The high clinical value of 68Ga-labelled SST ligand analogues is recognized and clinical routine practice and regulations are getting established. Imaging feasibility of GRPR, GLP-1R, MSH, HER2 as well as prostate-specific membrane antigen, osteoblastic bone metastases, amino acid uptake, glucose transport, angiogenesis using 68Ga-agents has been clinically demonstrated in patients. The experience with a number of ligands specific to G-protein coupled receptors (GRP, CCK, GLP-1, MSH, neuropeptide Y, melanocortin-1, VPAC-1, NT, CXCR4, GnRHR) labelled with 99mTc, 111In, 18F, 64Cu, 90Y, 177Lu and used in clinical studies provide solid basis and will facilitate advent of 68Ga-labelled analogues in the nearest future. Obviously, oncology will be the major arena for 68Ga-agents, especially those based on regulatory small peptides specific to disease type. This is greatly supported by the robustness, favourable pharmacokinetics and accessibility of the peptides. Immuno-PET with 68Ga will also expand due to the pre-targeted imaging technique. The basic research for the development of agents for myocardial perfusion imaging will most probably persist in order to meet the need for quantitative diagnostic means as well as to compensate for 99mTc shortage. Strong clinical need for the specific and quantitative imaging of inflammation and infection will stimulate the development of the corresponding 68Ga-agents, and the vast experience of 99mTc-radiopharmaceuticals provides valuable basis 216.
The future of 68Ga is predicted as PET analogue of 99mTc with added value of higher sensitivity, resolution, quantitation, dynamic scanning, and personalized medicine. The production of 68Ga-based imaging agents can be accomplished either under GMP or radiopharmacy practice, and it is a cost-effective complement to cyclotron-based tracers. The kit type preparation similar to that of 99mTc is feasible. Most importantly it will enable PET-CT investigations globally in remote hospitals without access to accelerators and radiopharmaceutical distribution centres thus promoting worldwide PET technique for earlier better diagnostics and further for individualized medicine. The input of 68Ga into oncology and development of theranostics bridging quantitative diagnosis with subsequent therapeutic management has already been proven and will continue to expand. Moreover, it is simply a matter of time when the imaging of bone, sentinel lymph node, lung ventilation/perfusion will be taken over by 68Ga/PET-CT. However, even if the vision of 68Ga becoming as immense as 99mTc will not get fulfilled in the nearest future, 68Ga definitely has strong contribution to make in the improvement of personalized patient management and there is a niche for 68Ga-based agents in nuclear medicine. Moreover, 68Ga-agents have the prerequisites to substitute also 111In-based radiopharmaceuticals.
References
- 1.Rahmim A, Zaidi H. PET versus SPECT: strengths, limitations and challenges. Nucl Med Commun. 2008;29:193–207. doi: 10.1097/MNM.0b013e3282f3a515. [DOI] [PubMed] [Google Scholar]
- 2.Velikyan I. Positron emitting [68Ga]Ga-based imaging agents: chemistry and diversity. Med Chem. 2011;7:338–72. doi: 10.2174/157340611796799195. [DOI] [PubMed] [Google Scholar]
- 3.Baum RP, Roesch F. Theranostics, Gallium-68, and other radionuclides: Springer; 2013.
- 4.Velikyan I. Molecular imaging and radiotherapy: theranostics for personalized patient management. Theranostics. 2012;2:424–6. doi: 10.7150/thno.4428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rosch F, Baum RP. Generator-based PET radiopharmaceuticals for molecular imaging of tumours: on the way to THERANOSTICS. Dalton Trans. 2011;40:6104–11. doi: 10.1039/c0dt01504k. [DOI] [PubMed] [Google Scholar]
- 6.Baum RP, Kulkarni HR, Carreras C. Peptides and receptors in image-guided therapy: theranostics for neuroendocrine neoplasms. Semin Nucl Med. 2012;42:190–207. doi: 10.1053/j.semnuclmed.2012.01.002. [DOI] [PubMed] [Google Scholar]
- 7.Baum RP, Kulkarni HR. Theranostics: from molecular imaging using Ga-68 labeled tracers and PET/CT to personalized radionuclide therapy - the Bad Berka experience. Theranostics. 2012;2:437–47. doi: 10.7150/thno.3645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hicks RJ, Hofman MS. Is there still a role for SPECT-CT in oncology in the PET-CT era? NAT REV CLIN ONCOL. 2012;9:712–20. doi: 10.1038/nrclinonc.2012.188. [DOI] [PubMed] [Google Scholar]
- 9.Delbeke D, Segall GM. Status of and Trends in Nuclear Medicine in the United States. J NUCL MED. 2011;52:24S–8S. doi: 10.2967/jnumed.110.085688. [DOI] [PubMed] [Google Scholar]
- 10.Chopra A, Shan L, Eckelman WC, Leung K, Latterner M, Bryant SH. et al. Molecular imaging and contrast agent database (MICAD): Evolution and progress. Mol Imaging Biol. 2012;14:4–13. doi: 10.1007/s11307-011-0521-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hicks RJ. Should positron emission tomography/computed tomography be the first rather than the last test performed in the assessment of cancer? Cancer Imaging. 2012;12:315–23. doi: 10.1102/1470-7330.2012.9005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Czernin J, Weber WA. Issues and controversies in nuclear medicine. Introduction. J Nucl Med. 2011;52(Suppl 2):1S–2S. doi: 10.2967/jnumed.111.097071. [DOI] [PubMed] [Google Scholar]
- 13.Treglia G, Castaldi P, Rindi G, Giordano A, Rufini V. Diagnostic performance of Gallium-68 somatostatin receptor PET and PET/CT in patients with thoracic and gastroenteropancreatic neuroendocrine tumours: A meta-analysis. Endocrine. 2012;42:80–7. doi: 10.1007/s12020-012-9631-1. [DOI] [PubMed] [Google Scholar]
- 14.Öberg K. Gallium-68 somatostatin receptor PET/CT: Is it time to replace 111Indium DTPA octreotide for patients with neuroendocrine tumors? Endocrine. 2012;42:3–4. doi: 10.1007/s12020-012-9681-4. [DOI] [PubMed] [Google Scholar]
- 15.Schreiter NF, Brenner W, Nogami M, Buchert R, Huppertz A, Pape UF. et al. Cost comparison of 111In-DTPA-octreotide scintigraphy and 68Ga-DOTATOC PET/CT for staging enteropancreatic neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2012;39:72–82. doi: 10.1007/s00259-011-1935-5. [DOI] [PubMed] [Google Scholar]
- 16.Buck AK, Herrmann K, Schreyogg J. PET/CT for staging lung cancer: costly or cost-saving? Eur J Nucl Med Mol Imaging. 2011;38:799–801. doi: 10.1007/s00259-011-1803-3. [DOI] [PubMed] [Google Scholar]
- 17.Manus MPM, Hicks RJ. How can we tell if PET imaging for cancer is cost effective? The Lancet Oncology. 2010;11:711–2. doi: 10.1016/S1470-2045(10)70164-9. [DOI] [PubMed] [Google Scholar]
- 18.Yang Y, Czernin J. Contribution of Imaging to Cancer Care Costs. J Nucl Med. 2011;52:86S–92S. doi: 10.2967/jnumed.110.085621. [DOI] [PubMed] [Google Scholar]
- 19.Higgins LJ, Pomper MG. The evolution of imaging in cancer: current state and future challenges. Seminars in oncology. 2011;38:3–15. doi: 10.1053/j.seminoncol.2010.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gemmel F, Van den Wyngaert H, Love C, Welling MM, Gemmel P, Palestro CJ. Prosthetic joint infections: radionuclide state-of-the-art imaging. Eur J Nucl Med Mol Imaging. 2012;39:892–909. doi: 10.1007/s00259-012-2062-7. [DOI] [PubMed] [Google Scholar]
- 21.Sorensen J. How Does the Patient Benefit from Clinical PET? Theranostics. 2012;2:427–36. doi: 10.7150/thno.3794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rice SL, Roney CA, Daumar P, Lewis JS. The next generation of positron emission tomography radiopharmaceuticals in oncology. Semin Nucl Med. 2011;41:265–82. doi: 10.1053/j.semnuclmed.2011.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wahl RL, Herman JM, Ford E. The Promise and Pitfalls of Positron Emission Tomography and Single-Photon Emission Computed Tomography Molecular Imaging-Guided Radiation Therapy. Semin Radiat Oncol. 2011;21:88–100. doi: 10.1016/j.semradonc.2010.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Velikyan I, Sundin A, Eriksson B, Lundqvist H, Sorensen J, Bergstrom M. et al. In vivo binding of [68Ga]-DOTATOC to somatostatin receptors in neuroendocrine tumours--impact of peptide mass. Nucl Med Biol. 2010;37:265–75. doi: 10.1016/j.nucmedbio.2009.11.008. [DOI] [PubMed] [Google Scholar]
- 25.Velikyan I, Beyer GJ, Bergstrom-Pettermann E, Johansen P, Bergstrom M, Langstrom B. The importance of high specific radioactivity in the performance of (68)Ga-labeled peptide. Nucl Med Biol. 2008;35:529–36. doi: 10.1016/j.nucmedbio.2008.03.002. [DOI] [PubMed] [Google Scholar]
- 26.Sauter AW, Wehrl HF, Kolb A, Judenhofer MS, Pichler BJ. Combined PET/MRI: one step further in multimodality imaging. Trends in molecular medicine. 2010;16:508–15. doi: 10.1016/j.molmed.2010.08.003. [DOI] [PubMed] [Google Scholar]
- 27.Wehrl HF, Sauter AW, Judenhofer MS, Pichler BJ. Combined PET/MR imaging--technology and applications. Tech Canc Res Treat. 2010;9:5–20. doi: 10.1177/153303461000900102. [DOI] [PubMed] [Google Scholar]
- 28.Prieto E, Domínguez-Prado I, García-Velloso MJ, Peñuelas I, Richter JA, Martí-Climent JM. Impact of time-of-flight and point-spread-function in SUV quantification for oncological PET. Clin Nuc Med. 2013;38:103–9. doi: 10.1097/RLU.0b013e318279b9df. [DOI] [PubMed] [Google Scholar]
- 29.Schreiter NF, Nogami M, Steffen I, Pape UF, Hamm B, Brenner W. et al. Evaluation of the potential of PET-MRI fusion for detection of liver metastases in patients with neuroendocrine tumours. European Radiology. 2012;22:458–67. doi: 10.1007/s00330-011-2266-4. [DOI] [PubMed] [Google Scholar]
- 30.Thorwarth D, Henke G, Muller AC, Reimold M, Beyer T, Boss A. et al. Simultaneous 68Ga-DOTATOC-PET/MRI for IMRT treatment planning for meningioma: first experience. Int J Radiat Oncol Biol Phys. 2011;81:277–83. doi: 10.1016/j.ijrobp.2010.10.078. [DOI] [PubMed] [Google Scholar]
- 31.Hesse B, Vinberg N, Berthelsen AK, Ballinger JR. Adverse events in nuclear medicine - cause for concern? Eur J Nucl Med Mol Imaging. 2012;39:782–5. doi: 10.1007/s00259-012-2071-6. [DOI] [PubMed] [Google Scholar]
- 32.Coenen HH, Elsinga PH, Iwata R, Kilbourn MR, Pillai MRA, Rajan MGR. et al. Fluorine-18 radiopharmaceuticals beyond [18F]FDG for use in oncology and neurosciences. Nucl Med Biol. 2010;37:727–40. doi: 10.1016/j.nucmedbio.2010.04.185. [DOI] [PubMed] [Google Scholar]
- 33.McBride WJ, D'Souza CA, Sharkey RM, Karacay H, Rossi EA, Chang CH. et al. Improved 18F labeling of peptides with a fluoride-aluminum-chelate complex. Bioconjug Chem. 2010;21:1331–40. doi: 10.1021/bc100137x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lodi F, Malizia C, Castellucci P, Cicoria G, Fanti S, Boschi S. Synthesis of oncological [ 11C]radiopharmaceuticals for clinical PET. Nucl Med Biol. 2012;39:447–60. doi: 10.1016/j.nucmedbio.2011.10.016. [DOI] [PubMed] [Google Scholar]
- 35.Hofman MS, Kong G, Neels OC, Eu P, Hong E, Hicks RJ. High management impact of Ga-68 DOTATATE (GaTate) PET/CT for imaging neuroendocrine and other somatostatin expressing tumours. Journal of medical imaging and radiation oncology. 2012;56:40–7. doi: 10.1111/j.1754-9485.2011.02327.x. [DOI] [PubMed] [Google Scholar]
- 36.Jacobsson H, Larsson P, Jonsson C, Jussing E, Gryback P. Normal uptake of 68Ga-DOTA-TOC by the pancreas uncinate process mimicking malignancy at somatostatin receptor PET. Clin Nucl Med. 2012;37:362–5. doi: 10.1097/RLU.0b013e3182485110. [DOI] [PubMed] [Google Scholar]
- 37.Eberlein U, Lassmann M. Dosimetry of [(68)Ga]-labeled compounds. Appl Radiat Isot. 2012;76:70–4. doi: 10.1016/j.apradiso.2012.06.033. [DOI] [PubMed] [Google Scholar]
- 38.Pettinato C, Sarnelli A, Di Donna M, Civollani S, Nanni C, Montini G. et al. (68)Ga-DOTANOC: biodistribution and dosimetry in patients affected by neuroendocrine tumors. Eur J Nucl Med Mol Imaging. 2008;35:72–9. doi: 10.1007/s00259-007-0587-y. [DOI] [PubMed] [Google Scholar]
- 39.Friedman M, Ståhl S. Engineered affinity proteins for tumour-targeting applications. Biotechnol Appl Biochem. 2009;53:1–29. doi: 10.1042/BA20080287. [DOI] [PubMed] [Google Scholar]
- 40.Baum RP, Prasad V, Muller D, Schuchardt C, Orlova A, Wennborg A. et al. Molecular Imaging of HER2-Expressing Malignant Tumors in Breast Cancer Patients Using Synthetic 111In- or 68Ga-Labeled Affibody Molecules. J Nucl Med. 2010;51:892–7. doi: 10.2967/jnumed.109.073239. [DOI] [PubMed] [Google Scholar]
- 41.Al-Nahhas A, Fanti S. Radiolabelled peptides in diagnosis and therapy: an introduction. Eur J Nucl Med Mol Imaging. 2012;39(Suppl 1):S1–3. doi: 10.1007/s00259-012-2064-5. [DOI] [PubMed] [Google Scholar]
- 42.Decristoforo C, Pickett RD, Verbruggen A. Feasibility and availability of (6)(8)Ga-labelled peptides. Eur J Nucl Med Mol Imaging. 2012;39(Suppl 1):S31–40. doi: 10.1007/s00259-011-1988-5. [DOI] [PubMed] [Google Scholar]
- 43.Koopmans KP, Glaudemans AW. Rationale for the use of radiolabelled peptides in diagnosis and therapy. Eur J Nucl Med Mol Imaging. 2012;39(Suppl 1):S4–10. doi: 10.1007/s00259-011-2038-z. [DOI] [PubMed] [Google Scholar]
- 44.Maecke HR, Reubi JC. Somatostatin receptors as targets for nuclear medicine imaging and radionuclide treatment. J Nucl Med. 2011;52:841–4. doi: 10.2967/jnumed.110.084236. [DOI] [PubMed] [Google Scholar]
- 45.Schottelius M, Wester HJ. Molecular imaging targeting peptide receptors. Methods. 2009;48:161–77. doi: 10.1016/j.ymeth.2009.03.012. [DOI] [PubMed] [Google Scholar]
- 46.Reubi JC. Old and new peptide receptor targets in cancer: future directions. Recent Results Cancer Res. 2013;194:567–76. doi: 10.1007/978-3-642-27994-2_34. [DOI] [PubMed] [Google Scholar]
- 47.Laverman P, Sosabowski JK, Boerman OC, Oyen WJ. Radiolabelled peptides for oncological diagnosis. Eur J Nucl Med Mol Imaging. 2012;39(Suppl 1):S78–92. doi: 10.1007/s00259-011-2014-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Aloj L, Aurilio M, Rinaldi V, D'Ambrosio L, Tesauro D, Peitl PK. et al. Comparison of the binding and internalization properties of 12 DOTA-coupled and (1)(1)(1)In-labelled CCK2/gastrin receptor binding peptides: a collaborative project under COST Action BM0607. Eur J Nucl Med Mol Imaging. 2011;38:1417–25. doi: 10.1007/s00259-011-1816-y. [DOI] [PubMed] [Google Scholar]
- 49.Ambrosini V, Fani M, Fanti S, Forrer F, Maecke HR. Radiopeptide imaging and therapy in Europe. J Nucl Med. 2011;52(Suppl 2):42S–55S. doi: 10.2967/jnumed.110.085753. [DOI] [PubMed] [Google Scholar]
- 50.Roosenburg S, Laverman P, van Delft FL, Boerman OC. Radiolabeled CCK/gastrin peptides for imaging and therapy of CCK2 receptor-expressing tumors. Amino Acids. 2011;41:1049–58. doi: 10.1007/s00726-010-0501-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Erchegyi J, Cescato R, Waser B, Rivier JE, Reubi JC. N-imidazolebenzyl-histidine substitution in somatostatin and in its octapeptide analogue modulates receptor selectivity and function. J Med Chem. 2011;54:5981–7. doi: 10.1021/jm200307v. [DOI] [PubMed] [Google Scholar]
- 52.Decristoforo C, Peñuelas I. Towards a harmonized radiopharmaceutical regulatory framework in Europe? Quarterly Journal of Nuclear Medicine and Molecular Imaging. 2009;53:394–401. [PubMed] [Google Scholar]
- 53.Lappin G, Kuhnz W, Jochemsen R, Kneer J, Chaudhary A, Oosterhuis B. et al. Use of microdosing to predict pharmacokinetics at the therapeutic dose: Experience with 5 drugs. Clinical Pharmacology & Therapeutics. 2006;80:203–15. doi: 10.1016/j.clpt.2006.05.008. [DOI] [PubMed] [Google Scholar]
- 54.Garner RC, Lappin G. The phase 0 microdosing concept. British Journal of Clinical Pharmacology. 2006;61:367–70. doi: 10.1111/j.1365-2125.2006.02575.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bergstrom M, Grahnen A, Langstrom B. Positron emission tomography microdosing: a new concept with application in tracer and early clinical drug development. Eur J Clin Pharmacol. 2003;59:357–66. doi: 10.1007/s00228-003-0643-x. [DOI] [PubMed] [Google Scholar]
- 56.Marchetti S, Schellens JHM. The impact of FDA and EMEA guidelines on drug development in relation to Phase 0 trials. British Journal of Cancer. 2007;97:577–81. doi: 10.1038/sj.bjc.6603925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mills G. The exploratory IND. Journal of nuclear medicine: official publication, Society of Nuclear Medicine. 2008;49:45N–7N. [PubMed] [Google Scholar]
- 58.Verbruggen A, Coenen HH, Deverre JR, Guilloteau D, Langstrom B, Salvadori PA. et al. Guideline to regulations for radiopharmaceuticals in early phase clinical trials in the EU. European Journal of Nuclear Medicine and Molecular Imaging. 2008;35:2144–51. doi: 10.1007/s00259-008-0853-7. [DOI] [PubMed] [Google Scholar]
- 59.Norenberg JP, Petry NA, Schwarz S. Operation of a radiopharmacy for a clinical trial. Seminars in Nuclear Medicine. 2010;40:347–56. doi: 10.1053/j.semnuclmed.2010.06.002. [DOI] [PubMed] [Google Scholar]
- 60.European pharmacopoeia commission. Production and compounding of radiopharmaceutical preparations. Strasbourg. 2011.
- 61.Elsinga P, Todde S, Penuelas I, Meyer G, Farstad B, Faivre-Chauvet A. et al. Guidance on current good radiopharmacy practice (cGRPP) for the small-scale preparation of radiopharmaceuticals. European Journal of Nuclear Medicine and Molecular Imaging. 2010;37:1049–62. doi: 10.1007/s00259-010-1407-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Virgolini I, Ambrosini V, Bomanji JB, Baum RP, Fanti S, Gabriel M. et al. Procedure guidelines for PET/CT tumour imaging with 68Ga-DOTA-conjugated peptides: 68Ga-DOTA-TOC, 68Ga-DOTA-NOC, 68Ga-DOTA-TATE. Eur J Nucl Med Mol Imaging. 2010;37:2004–10. doi: 10.1007/s00259-010-1512-3. [DOI] [PubMed] [Google Scholar]
- 63.Janson ET, Sorbye H, Welin S, Federspiel B, Gronbaek H, Hellman P. et al. Nordic Guidelines 2010 for diagnosis and treatment of gastroenteropancreatic neuroendocrine tumours. Acta Oncologica. 2010;49:740–56. doi: 10.3109/0284186X.2010.492791. [DOI] [PubMed] [Google Scholar]
- 64.European Directorate for the Quality of Medicines Pharmeuropa. Gallium (68Ga)edotreotide injection. European Directorate for the Quality of Medicines Pharmeuropa. 2011;23:310–23. [Google Scholar]
- 65.Velikyan I, Gunnar A, Sörensen J, Estrada S. Organ biodistribution of Germanium-68 in rat in the presence and absence of [68Ga]Ga-DOTA-TOC for the extrapolation to the human organ and whole-body radiation dosimetry. American journal of nuclear medicine and molecular imaging. 2013;3:154. [PMC free article] [PubMed] [Google Scholar]
- 66.Boellaard R, O'Doherty MJ, Chiti A, Krause BJ. Reply to: FDG PET and PET/CT: EANM procedure guideline for tumour PET imaging. European Journal of Nuclear Medicine and Molecular Imaging. 2010;37:1432–3. doi: 10.1007/s00259-009-1297-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Boellaard R, O'Doherty MJ, Weber WA, Mottaghy FM, Lonsdale MN, Stroobants SG. et al. FDG PET and PET/CT: EANM procedure guidelines for tumour PET imaging: version 1.0. European Journal of Nuclear Medicine and Molecular Imaging. 2010;37:181–200. doi: 10.1007/s00259-009-1297-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: Evolving considerations for PET response criteria in solid tumors. Journal of Nuclear Medicine. 2009;50:122S–50S. doi: 10.2967/jnumed.108.057307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Dondi M, Kashyap R, Paez D, Pascual T, Zaknun J, Bastos FM. et al. Trends in Nuclear Medicine in Developing Countries. Journal of Nuclear Medicine. 2011;52:16S–23S. doi: 10.2967/jnumed.111.089193. [DOI] [PubMed] [Google Scholar]
- 70.Ware RE, Hicks RJ. Doing more harm than good? Do systematic reviews of PET by health technology assessment agencies provide an appraisal of the evidence that is closer to the truth than the primary data supporting its use? Journal of Nuclear Medicine. 2011;52:64S–73S. doi: 10.2967/jnumed.110.086611. [DOI] [PubMed] [Google Scholar]
- 71.Graham MM, Menda Y. Radiopeptide Imaging and Therapy in the United States. Journal of Nuclear Medicine. 2011;52:56S–63S. doi: 10.2967/jnumed.110.085746. [DOI] [PubMed] [Google Scholar]
- 72.Decristoforo C, Knopp R, von Guggenberg E, Rupprich M, Dreger T, Hess A. et al. A fully automated synthesis for the preparation of 68Ga-labelled peptides. Nucl Med Commun. 2007;28:870–5. doi: 10.1097/MNM.0b013e3282f1753d. [DOI] [PubMed] [Google Scholar]
- 73.Azhdarinia A, Yang DJ, Chao C, Mourtada F. Infrared-based module for the synthesis of 68Ga-labeled radiotracers. Nucl Med Biol. 2007;34:121–7. doi: 10.1016/j.nucmedbio.2006.10.011. [DOI] [PubMed] [Google Scholar]
- 74.Cagnolini A, Chen J, Ramos K, Skedzielewski TM, Lantry LE, Nunn AD. et al. Automated synthesis, characterization and biological evaluation of [(68)Ga]Ga-AMBA, and the synthesis and characterization of (nat)Ga-AMBA and [(67)Ga]Ga-AMBA. Appl Radiat Isot. 2010;68:2285–92. doi: 10.1016/j.apradiso.2010.06.023. [DOI] [PubMed] [Google Scholar]
- 75.Bauwens M, Chekol R, Vanbilloen H, Bormans G, Verbruggen A. Optimal buffer choice of the radiosynthesis of Ga-68-Dotatoc for clinical application. Nuclear Medicine Communications. 2010;31:753–8. doi: 10.1097/MNM.0b013e32833acb99. [DOI] [PubMed] [Google Scholar]
- 76.Meyer GJ, Macke H, Schuhmacher J, Knapp WH, Hofmann M. 68Ga-labelled DOTA-derivatised peptide ligands. Eur J Nucl Med Mol Imaging. 2004;31:1097–104. doi: 10.1007/s00259-004-1486-0. [DOI] [PubMed] [Google Scholar]
- 77.Rizzello A, Di Pierro D, Lodi F, Trespidi S, Cicoria G, Pancaldi D. et al. Synthesis and quality control of Ga-68 citrate for routine clinical PET. Nuclear Medicine Communications. 2009;30:542–5. doi: 10.1097/MNM.0b013e32832b9ac8. [DOI] [PubMed] [Google Scholar]
- 78.Di Pierro D, Rizzello A, Cicoria G, Lodi F, Marengo M, Pancaldi D. et al. Radiolabelling, quality control and radiochemical purity assessment of the Octreotide analogue 68Ga DOTA NOC. Applied radiation and isotopes: including data, instrumentation and methods for use in agriculture, industry and medicine. 2008;66:1091–6. doi: 10.1016/j.apradiso.2007.12.001. [DOI] [PubMed] [Google Scholar]
- 79.Ocak M, Antretter M, Knopp R, Kunkel F, Petrik M, Bergisadi N. et al. Full automation of (68)Ga labelling of DOTA-peptides including cation exchange prepurification. Applied radiation and isotopes: including data, instrumentation and methods for use in agriculture, industry and medicine. 2010;68:297–302. doi: 10.1016/j.apradiso.2009.10.006. [DOI] [PubMed] [Google Scholar]
- 80.Pohle K, Notni J, Bussemer J, Kessler H, Schwaiger M, Beer AJ. 68Ga-NODAGA-RGD is a suitable substitute for (18)F-Galacto-RGD and can be produced with high specific activity in a cGMP/GRP compliant automated process. Nucl Med Biol. 2012;39:777–84. doi: 10.1016/j.nucmedbio.2012.02.006. [DOI] [PubMed] [Google Scholar]
- 81.Boschi S, Malizia C, Lodi F. Overview and perspectives on automation strategies in 68Ga radiopharmaceutical preparations. Recent Results Cancer Res. 2012;194:17–31. doi: 10.1007/978-3-642-27994-2_2. [DOI] [PubMed] [Google Scholar]
- 82.Lin M, Ranganathan D, Mori T, Hagooly A, Rossin R, Welch MJ. et al. Long-term evaluation of TiO2-based 68Ge/68Ga generators and optimized automation of [68Ga]DOTATOC radiosynthesis. Appl Radiat Isot. 2012;70:2539–44. doi: 10.1016/j.apradiso.2012.05.009. [DOI] [PubMed] [Google Scholar]
- 83.Petrik M, Knetsch PA, Knopp R, Imperato G, Ocak M, von Guggenberg E. et al. Radiolabelling of peptides for PET, SPECT and therapeutic applications using a fully automated disposable cassette system. Nucl Med Commun. 2011;32:887–95. doi: 10.1097/MNM.0b013e3283497188. [DOI] [PubMed] [Google Scholar]
- 84.Gebhardt P, Opfermann T, Saluz HP. Computer controlled Ga-68 milking and concentration system. Applied Radiation and Isotopes. 2010;68:1057–9. doi: 10.1016/j.apradiso.2010.01.024. [DOI] [PubMed] [Google Scholar]
- 85.De Decker M, Turner JH. Automated module radiolabeling of peptides and antibodies with gallium-68, lutetium-177 and iodine-131. Cancer Biotherapy & Radiopharmaceuticals. 2012;27:72–6. doi: 10.1089/cbr.2011.1073. [DOI] [PubMed] [Google Scholar]
- 86.Kumar V, Boddeti DK, Evans SG, Roesch F, Howman-Giles R. Potential use of Ga-68-apo-transferrin as a PET imaging agent for detecting Staphylococcus aureus infection. Nuclear Medicine and Biology. 2011;38:393–8. doi: 10.1016/j.nucmedbio.2010.09.011. [DOI] [PubMed] [Google Scholar]
- 87.Velikyan I, Berhane T, Khan T, Borges J, Sorensen J. One pot preparation of Ga-68-labelled macroaggregated albumin for PET-imaging of acute pulmonary embolism and myocardial perfusion quantification. European Journal of Nuclear Medicine and Molecular Imaging. 2012;39:S410–S1. [Google Scholar]
- 88.Maus S, Buchholz H-G, Ament S, Brochhausen C, Bausbacher N, Schreckenberger M. Labelling of commercially available human serum albumin kits with 68Ga as surrogates for 99mTc-MAA microspheres. Appl Radiat Isot. 2011;69:171–5. doi: 10.1016/j.apradiso.2010.09.008. [DOI] [PubMed] [Google Scholar]
- 89.Mathias CJ, Green MA. A convenient route to Ga-68 Ga-MAA for use as a particulate PET perfusion tracer. Applied Radiation and Isotopes. 2008;66:1910–2. doi: 10.1016/j.apradiso.2008.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Richter T, Bergmann R, Pietzsch J, Kozle I, Hofheinz F, Schiller E. et al. Effects of posture on regional pulmonary blood flow in rats as measured by PET. Journal of Applied Physiology. 2010;108:422–9. doi: 10.1152/japplphysiol.91257.2008. [DOI] [PubMed] [Google Scholar]
- 91.Borges JB, Velikyan I, Långström B, Sörensen J, Ulin J, Maripuu E. et al. Ventilation distribution studies comparing Technegas and Gallgas using 68GaCl3 as the label. The journal of nuclear medicine. 2011;52:206–9. doi: 10.2967/jnumed.110.083881. [DOI] [PubMed] [Google Scholar]
- 92.Hofman MS, Beauregard JM, Barber TW, Neels OC, Eu P, Hicks RJ. 68Ga PET/CT ventilation-perfusion imaging for pulmonary embolism: a pilot study with comparison to conventional scintigraphy. Journal of nuclear medicine: official publication, Society of Nuclear Medicine. 2011;52:1513–9. doi: 10.2967/jnumed.111.093344. [DOI] [PubMed] [Google Scholar]
- 93.Roesch F, Filosofov DV. Production, radiochemical processing and quality evaluation of Ge-68 suitable for production of a 68Ge/68Ga generator. Int. Atomic Energy Agency. 2010.
- 94.Chakravarty R, Shukla R, Ram R, Tyagi AK, Dash A, Venkatesh M. Development of a nano-zirconia based 68Ge/68Ga generator for biomedical applications. Nucl Med Biol. 2011;38:575–83. doi: 10.1016/j.nucmedbio.2010.10.007. [DOI] [PubMed] [Google Scholar]
- 95.Le VS. 68Ga generator integrated system: Elution-purification- concentration integration. 2013;:43-75. [DOI] [PubMed]
- 96.Rösch F. 68Ge/ 68Ga Generators: Past, present, and future. 2013;: 3-16. [DOI] [PubMed]
- 97.Shehata MM, Scholten B, Spahn I, Qaim SM, Coenen HH. Radiochemical studies relevant to the separation of 68Ga and 68Ge. Journal of Radioanalytical and Nuclear Chemistry. 2011;288:887–93. [Google Scholar]
- 98.de Blois E, Chan HS, Naidoo C, Prince D, Krenning EP, Breeman WAP. Characteristics of SnO2-based Ge-68/Ga-68 generator and aspects of radiolabelling DOTA-peptides. Applied Radiation and Isotopes. 2011;69:308–15. doi: 10.1016/j.apradiso.2010.11.015. [DOI] [PubMed] [Google Scholar]
- 99.Velikyan I, Beyer GJ, Langstrom B. Microwave-supported preparation of 68Ga-bioconjugates with high specific radioactivity. Bioconjugate Chem. 2004;15:554–60. doi: 10.1021/bc030078f. [DOI] [PubMed] [Google Scholar]
- 100.Velikyan I. The Diversity of 68Ga-Based Imaging Agents. Recent Results Cancer Res. 2013;194:101–31. doi: 10.1007/978-3-642-27994-2_7. [DOI] [PubMed] [Google Scholar]
- 101.Zhernosekov KP, Filosofov DV, Baum RP, Aschoff P, Bihl H, Razbash AA. et al. Processing of Generator-Produced 68Ga for Medical Application. Journal of Nuclear Medicine. 2007;48:1741–8. doi: 10.2967/jnumed.107.040378. [DOI] [PubMed] [Google Scholar]
- 102.Petrik M, Schuessele A, Perkhofer S, Lass-Florl C, Becker D, Decristoforo C. Microbial challenge tests on nonradioactive TiO2-based 68Ge/68Ga generator columns. Nuclear Medicine Communications. 2012;33:819–23. doi: 10.1097/MNM.0b013e3283543323. [DOI] [PubMed] [Google Scholar]
- 103.Velikyan I, Xu H, Nair M, Hall H. Robust labeling and comparative preclinical characterization of DOTA-TOC and DOTA-TATE. Nucl Med Biol. 2012;39:628–59. doi: 10.1016/j.nucmedbio.2011.12.010. [DOI] [PubMed] [Google Scholar]
- 104.Sen K, Wolterbeek HT, Breeman WAP. Speciation of no-carrier-added 68Ga prior to its labeling for PET imaging. Journal of Radioanalytical and Nuclear Chemistry. 2012;291:683–7. [Google Scholar]
- 105.Kotzerke J, Andreeff M, Wunderlich G. PET aerosol lung scintigraphy using Galligas. Eur J Nucl Med Mol Imaging. 2010;37:175–7. doi: 10.1007/s00259-009-1304-9. [DOI] [PubMed] [Google Scholar]
- 106.Zoller F, Riss PJ, Montforts FP, Rosch F. Efficient post-processing of aqueous generator eluates facilitates Ga-68-labelling under anhydrous conditions. Radiochimica Acta. 2010;98:157–60. [Google Scholar]
- 107.Petrik M, Ocak M, Rupprich M, Decristoforo C. Impurity in (68)Ga-peptide preparation using processed generator eluate. Journal of nuclear medicine: official publication, Society of Nuclear Medicine. 2010;51:495. doi: 10.2967/jnumed.109.070953. author reply -6. [DOI] [PubMed] [Google Scholar]
- 108.Schultz MK, Mueller D, Baum RP, Leonard Watkins G, Breeman WAP. A new automated NaCl based robust method for routine production of gallium-68 labeled peptides. Applied Radiation and Isotopes. 2012. [DOI] [PMC free article] [PubMed]
- 109.Ballinger JR. Short- and long-term responses to molybdenum-99 shortages in nuclear medicine. Br J Radiol. 2010;83:899–901. doi: 10.1259/bjr/17139152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Bartholoma MD, Louie AS, Valliant JF, Zubieta J. Technetium and gallium derived radiopharmaceuticals: comparing and contrasting the chemistry of two important radiometals for the molecular imaging era. Chem Rev. 2010;110:2903–20. doi: 10.1021/cr1000755. [DOI] [PubMed] [Google Scholar]
- 111.Breeman WA, de Jong M, de Blois E, Bernard BF, Konijnenberg M, Krenning EP. Radiolabelling DOTA-peptides with (68)Ga. Eur J Nucl Med Mol Imaging. 2005;32:478–85. doi: 10.1007/s00259-004-1702-y. [DOI] [PubMed] [Google Scholar]
- 112.Bartholomä MD. Recent developments in the design of bifunctional chelators for metal-based radiopharmaceuticals used in Positron Emission Tomography. Inorganica Chimica Acta. 2012;389:36–51. [Google Scholar]
- 113.Velikyan I, Maecke H, Langstrom B. Convenient preparation of 68Ga-based PET-radiopharmaceuticals at room temperature. Bioconjug Chem. 2008;19:569–73. doi: 10.1021/bc700341x. [DOI] [PubMed] [Google Scholar]
- 114.Chaves S, Mendonca AC, Marques SM, Prata MI, Santos AC, Martins AF. et al. A gallium complex with a new tripodal tris-hydroxypyridinone for potential nuclear diagnostic imaging: solution and in vivo studies of 67Ga-labeled species. J Inorg Biochem. 2011;105:31–8. doi: 10.1016/j.jinorgbio.2010.09.012. [DOI] [PubMed] [Google Scholar]
- 115.Berry DJ, Ma Y, Ballinger JR, Tavare R, Koers A, Sunassee K. et al. Efficient bifunctional gallium-68 chelators for positron emission tomography: tris(hydroxypyridinone) ligands. Chem Commun (Camb) 2011;47:7068–70. doi: 10.1039/c1cc12123e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Boros E, Ferreira CL, Cawthray JF, Price EW, Patrick BO, Wester DW. et al. Acyclic Chelate with Ideal Properties for Ga-68 PET Imaging Agent Elaboration. Journal of the American Chemical Society. 2010;132:15726–33. doi: 10.1021/ja106399h. [DOI] [PubMed] [Google Scholar]
- 117.Eder M, Krivoshein AV, Backer M, Backer JM, Haberkorn U, Eisenhut M. ScVEGF-PEG-HBED-CC and scVEGF-PEG-NOTA conjugates: comparison of easy-to-label recombinant proteins for [68Ga]PET imaging of VEGF receptors in angiogenic vasculature. Nucl Med Biol. 2010;37:405–12. doi: 10.1016/j.nucmedbio.2010.02.001. [DOI] [PubMed] [Google Scholar]
- 118.Boros E, Ferreira CL, Yapp DT, Gill RK, Price EW, Adam MJ. et al. RGD conjugates of the H2dedpa scaffold: synthesis, labeling and imaging with 68Ga. Nucl Med Biol. 2012;39:785–94. doi: 10.1016/j.nucmedbio.2012.01.003. [DOI] [PubMed] [Google Scholar]
- 119.Riss PJ, Kroll C, Nagel V, Rosch F. NODAPA-OH and NODAPA-(NCS)n: synthesis, 68Ga-radiolabelling and in vitro characterisation of novel versatile bifunctional chelators for molecular imaging. Bioorg Med Chem Lett. 2008;18:5364–7. doi: 10.1016/j.bmcl.2008.09.054. [DOI] [PubMed] [Google Scholar]
- 120.Jeong JM, Hong MK, Chang YS, Lee YS, Kim YJ, Cheon GJ. et al. Preparation of a promising angiogenesis PET imaging agent: Ga-68-labeled c(RGDyK)-isothiocyanatobenzyl-1,4,7-triazacyclononane-1,4,7-triacetic acid and feasibility studies in mice. J Nucl Med. 2008;49:830–6. doi: 10.2967/jnumed.107.047423. [DOI] [PubMed] [Google Scholar]
- 121.Morfin JF, Toth E. Kinetics of Ga(NOTA) Formation from Weak Ga-Citrate Complexes. Inorganic Chemistry. 2011;50:10371–8. doi: 10.1021/ic201445e. [DOI] [PubMed] [Google Scholar]
- 122.Notni J, Hermann P, Havlickova J, Kotek J, Kubicek V, Plutnar J. et al. A triazacyclononane-based bifunctional phosphinate ligand for the preparation of multimeric 68Ga tracers for positron emission tomography. Chemistry. 2010;16:7174–85. doi: 10.1002/chem.200903281. [DOI] [PubMed] [Google Scholar]
- 123.Notni J, Simecek J, Hermann P, Wester HJ. TRAP, a Powerful and Versatile Framework for Gallium-68 Radiopharmaceuticals. Chemistry-a European Journal. 2011;17:14718–22. doi: 10.1002/chem.201103503. [DOI] [PubMed] [Google Scholar]
- 124.Simecek J, Zemek O, Hermann P, Wester HJ, Notni J. A monoreactive bifunctional triazacyclononane phosphinate chelator with high selectivity for gallium-68. ChemMedChem. 2012;7:1375–8. doi: 10.1002/cmdc.201200261. [DOI] [PubMed] [Google Scholar]
- 125.Simecek J, Hermann P, Wester HJ, Notni J. How is (68) Ga Labeling of Macrocyclic Chelators Influenced by Metal Ion Contaminants in (68) Ge/(68) Ga Generator Eluates? ChemMedChem. 2012. [DOI] [PubMed]
- 126.Simecek J, Schulz M, Notni J, Plutnar J, Kubicek V, Havlickova J. et al. Complexation of metal ions with TRAP (1,4,7-triazacyclononane phosphinic acid) ligands and 1,4,7-triazacyclononane-1,4,7-triacetic acid: phosphinate-containing ligands as unique chelators for trivalent gallium. Inorg Chem. 2012;51:577–90. doi: 10.1021/ic202103v. [DOI] [PubMed] [Google Scholar]
- 127.Kubíček V, Havlíčková J, Kotek J, Tircsó G, Hermann P, Tóth E. et al. Gallium(III) complexes of DOTA and DOTA-Monoamide: Kinetic and thermodynamic studies. Inorganic Chemistry. 2010;49:10960–9. doi: 10.1021/ic101378s. [DOI] [PubMed] [Google Scholar]
- 128.Ferreira CL, Lamsa E, Woods M, Duan Y, Fernando P, Bensimon C. et al. Evaluation of Bifunctional Chelates for the Development of Gallium-Based Radiopharmaceuticals. Bioconjugate Chemistry. 2010;21:531–6. doi: 10.1021/bc900443a. [DOI] [PubMed] [Google Scholar]
- 129.Azad BB, Cho CF, Lewis JD, Luyt LG. Synthesis, radiometal labeling and in vitro evaluation of a targeted PPIX derivative. Appl Radiat Isot. 2012;70:505–11. doi: 10.1016/j.apradiso.2011.11.054. [DOI] [PubMed] [Google Scholar]
- 130.Zoller F, Riss PJ, Montforts FP, Kelleher DK, Eppard E, Rosch F. Radiolabelling and preliminary evaluation of (68)Ga-tetrapyrrole derivatives as potential tracers for PET. Nuclear Medicine and Biology. 2012. [DOI] [PubMed]
- 131.Ma MT, Neels OC, Denoyer D, Roselt P, Karas JA, Scanlon DB. et al. Gallium-68 complex of a macrobicyclic cage amine chelator tethered to two integrin-targeting peptides for diagnostic tumor imaging. Bioconjug Chem. 2011;22:2093–103. doi: 10.1021/bc200319q. [DOI] [PubMed] [Google Scholar]
- 132.Mindt TL, Jungi V, Wyss S, Friedli A, Pla G, Novak-Hofer I. et al. Modification of different IgG1 antibodies via glutamine and lysine using bacterial and human tissue transglutaminase. Bioconjugate Chemistry. 2008;19:271–8. doi: 10.1021/bc700306n. [DOI] [PubMed] [Google Scholar]
- 133.Tanaka K, Masuyama T, Hasegawa K, Tahara T, Mizuma H, Wada Y. et al. A submicrogram-scale protocol for biomolecule-based PET imaging by rapid 6 pi-azaelectrocyclization: Visualization of sialic acid dependent circulatory residence of glycoproteins. Angewandte Chemie-International Edition. 2008;47:102–5. doi: 10.1002/anie.200702989. [DOI] [PubMed] [Google Scholar]
- 134.Knor S, Modlinger A, Poethko T, Schottelius M, Wester HJ, Kessler H. Synthesis of novel 1,4,7,10-tetraazacyclodecane-1,4,7,10-tetraacetic acid (DOTA) derivatives for chemoselective attachment to unprotected polyfunctionalized compounds. Chemistry-a European Journal. 2007;13:6082–90. doi: 10.1002/chem.200700231. [DOI] [PubMed] [Google Scholar]
- 135.Dijkgraaf I, Rijnders AY, Soede A, Dechesne AC, van Esse GW, Brouwer AJ. et al. Synthesis of DOTA-conjugated multivalent cyclic-RGD peptide dendrimers via 1,3-dipolar cycloaddition and their biological evaluation: implications for tumor targeting and tumor imaging purposes. Org Biomol Chem. 2007;5:935–44. doi: 10.1039/b615940k. [DOI] [PubMed] [Google Scholar]
- 136.Tanaka K, Fukase K, Katsumura S. Exploring a unique reactivity of 6-azaelectrocyclization to enzyme inhibition, natural products synthesis, and molecular imaging: An approach to chemical biology by synthetic chemists. Synlett. 2011. pp. 2115–39.
- 137.Mindt TL, Müller C, Stuker F, Salazar JF, Hohn A, Mueggler T. et al. A "click chemistry" approach to the efficient synthesis of multiple imaging probes derived from a single precursor. Bioconjugate Chemistry. 2009;20:1940–9. doi: 10.1021/bc900276b. [DOI] [PubMed] [Google Scholar]
- 138.Yim CB, van der Wildt B, Dijkgraaf I, Joosten L, Eek A, Versluis C. et al. Spacer effects on in vivo properties of DOTA-conjugated dimeric [Tyr3]octreotate peptides synthesized by a "Cu(I)-click" and "sulfo-click" ligation method. Chembiochem. 2011;12:750–60. doi: 10.1002/cbic.201000639. [DOI] [PubMed] [Google Scholar]
- 139.Baumhover NJ, Martin ME, Parameswarappa SG, Kloepping KC, O'Dorisio MS, Pigge FC. et al. Improved synthesis and biological evaluation of chelator-modified alpha-MSH analogs prepared by copper-free click chemistry. Bioorg Med Chem Lett. 2011;21:5757–61. doi: 10.1016/j.bmcl.2011.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Riss PJ, Burchardt C, Zimny MJ, Peters J, Roesch F. A concise synthesis procedure to furnish multi-gram amounts of hexadentate, bivalent DO2A-based chelators. RSC Advances. 2012;2:7156–60. [Google Scholar]
- 141.Dimitrakopoulou-Strauss A, Georgoulias V, Eisenhut M, Herth F, Koukouraki S, Macke HR. et al. Quantitative assessment of SSTR2 expression in patients with non-small cell lung cancer using Ga-68-DOTATOC PET and comparison with F-18-FDG PET. Eur J Nucl Med Mol Imaging. 2006;33:823–30. doi: 10.1007/s00259-005-0063-5. [DOI] [PubMed] [Google Scholar]
- 142.Dimitrakopoulou-Strauss A, Hohenberger P, Haberkorn U, Macke HR, Eisenhut M, Strauss LG. Ga-68-Labeled bombesin studies in patients with gastrointestinal stromal tumors: Comparison with F-18-FDG. J Nucl Med. 2007;48:1245–50. doi: 10.2967/jnumed.106.038091. [DOI] [PubMed] [Google Scholar]
- 143.Sancho V, Di Florio A, Moody TW, Jensen RT. Bombesin receptor-mediated imaging and cytotoxicity: review and current status. Current drug delivery. 2011;8:79–134. doi: 10.2174/156720111793663624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Fellner M, Baum RP, Kubicek V, Hermann P, Lukes I, Prasad V. et al. PET/CT imaging of osteoblastic bone metastases with Ga-68-bisphosphonates: first human study. Eur J Nucl Med Mol Imaging. 2010;37:834. doi: 10.1007/s00259-009-1355-y. [DOI] [PubMed] [Google Scholar]
- 145.Afshar-Oromieh A, Haberkorn U, Eder M, Eisenhut M, Zechmann CM. [68Ga]Gallium-labelled PSMA ligand as superior PET tracer for the diagnosis of prostate cancer: comparison with 18F-FECH. Eur J Nucl Med Mol Imaging. 2012;39:1085–6. doi: 10.1007/s00259-012-2069-0. [DOI] [PubMed] [Google Scholar]
- 146.Al-Nahhas A. Nuclear medicine imaging of neuroendocrine tumours. Clin Med. 2012;12:377–80. doi: 10.7861/clinmedicine.12-4-377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Ambrosini V, Nicolini S, Caroli P, Nanni C, Massaro A, Marzola MC. et al. PET/CT imaging in different types of lung cancer: an overview. European journal of radiology. 2012;81:988–1001. doi: 10.1016/j.ejrad.2011.03.020. [DOI] [PubMed] [Google Scholar]
- 148.Ambrosini V, Campana D, Tomassetti P, Fanti S. (6)(8)Ga-labelled peptides for diagnosis of gastroenteropancreatic NET. Eur J Nucl Med Mol Imaging. 2012;39(Suppl 1):S52–60. doi: 10.1007/s00259-011-1989-4. [DOI] [PubMed] [Google Scholar]
- 149.Oberg KE, Reubi JC, Kwekkeboom DJ, Krenning EP. Role of somatostatins in gastroenteropancreatic neuroendocrine tumor development and therapy. Gastroenterology. 2010;139:742–53. doi: 10.1053/j.gastro.2010.07.002. 53 e1. [DOI] [PubMed] [Google Scholar]
- 150.Oberg K. Molecular Imaging Radiotherapy: Theranostics for Personalized Patient Management of Neuroendocrine Tumors (NETs) Theranostics. 2012;2:448–58. doi: 10.7150/thno.3931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Erchegyi J, Grace CR, Samant M, Cescato R, Piccand V, Riek R. et al. Ring size of somatostatin analogues (ODT-8) modulates receptor selectivity and binding affinity. J Med Chem. 2008;51:2668–75. doi: 10.1021/jm701444y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Grace CR, Erchegyi J, Samant M, Cescato R, Piccand V, Riek R. et al. Ring size in octreotide amide modulates differently agonist versus antagonist binding affinity and selectivity. J Med Chem. 2008;51:2676–81. doi: 10.1021/jm701445q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Grace CR, Erchegyi J, Reubi JC, Rivier JE, Riek R. Three-dimensional consensus structure of sst2-selective somatostatin (SRIF) antagonists by NMR. Biopolymers. 2008;89:1077–87. doi: 10.1002/bip.21060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Cescato R, Erchegyi J, Waser B, Piccand V, Maecke HR, Rivier JE. et al. Design and in vitro characterization of highly sst2-selective somatostatin antagonists suitable for radiotargeting. J Med Chem. 2008;51:4030–7. doi: 10.1021/jm701618q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Fani M, Mueller A, Tamma ML, Nicolas G, Rink HR, Cescato R. et al. Radiolabeled bicyclic somatostatin-based analogs: a novel class of potential radiotracers for SPECT/PET of neuroendocrine tumors. J Nucl Med. 2010;51:1771–9. doi: 10.2967/jnumed.110.076695. [DOI] [PubMed] [Google Scholar]
- 156.Cescato R, Waser B, Fani M, Reubi JC. Evaluation of 177Lu-DOTA-sst2 antagonist versus 177Lu-DOTA-sst2 agonist binding in human cancers in vitro. J Nucl Med. 2011;52:1886–90. doi: 10.2967/jnumed.111.095778. [DOI] [PubMed] [Google Scholar]
- 157.Fani M, Del Pozzo L, Abiraj K, Mansi R, Tamma ML, Cescato R. et al. PET of somatostatin receptor-positive tumors using 64Cu- and 68Ga-somatostatin antagonists: the chelate makes the difference. J Nucl Med. 2011;52:1110–8. doi: 10.2967/jnumed.111.087999. [DOI] [PubMed] [Google Scholar]
- 158.Fani M, Braun F, Waser B, Beetschen K, Cescato R, Erchegyi J. et al. Unexpected sensitivity of sst2 antagonists to N-terminal radiometal modifications. J Nucl Med. 2012;53:1481–9. doi: 10.2967/jnumed.112.102764. [DOI] [PubMed] [Google Scholar]
- 159.Wild D, Fani M, Behe M, Brink I, Rivier JE, Reubi JC. et al. First clinical evidence that imaging with somatostatin receptor antagonists is feasible. J Nucl Med. 2011;52:1412–7. doi: 10.2967/jnumed.111.088922. [DOI] [PubMed] [Google Scholar]
- 160.Zhang H, Moroz MA, Serganova I, Ku T, Huang R, Vider J. et al. Imaging expression of the human somatostatin receptor subtype-2 reporter gene with 68Ga-DOTATOC. J Nucl Med. 2011;52:123–31. doi: 10.2967/jnumed.110.079004. [DOI] [PubMed] [Google Scholar]
- 161.Reubi JC, Schär J-C, Waser B, Wenger S, Heppeler A, Schmitt JS. et al. Affinity profiles for human somatostatin receptor subtypes SST1-SST5 of somatostatin radiotracers selected for scintigraphic and radiotherapeutic use. Eur J Nucl Med Mol Imaging. 2000;27:273–82. doi: 10.1007/s002590050034. [DOI] [PubMed] [Google Scholar]
- 162.Poeppel TD, Binse I, Petersenn S, Lahner H, Schott M, Antoch G. et al. 68Ga-DOTATOC versus 68Ga-DOTATATE PET/CT in functional imaging of neuroendocrine tumors. J Nucl Med. 2011;52:1864–70. doi: 10.2967/jnumed.111.091165. [DOI] [PubMed] [Google Scholar]
- 163.Hanin FX, Pauwels S, Bol A, Breeman W, de Jong M, Jamar F. Tumor uptake of 68Ga-DOTA-Tyr3-octreotate: animal PET studies of tumor flow and acute somatostatin receptor modulation in the CA20948 rat model. Nucl Med Biol. 2010;37:157–65. doi: 10.1016/j.nucmedbio.2009.09.006. [DOI] [PubMed] [Google Scholar]
- 164.Hanin FX, Pauwels S, Bol A, Melis M, Breeman W, De Jong M. et al. Effect of interferon-a treatment on [68Ga-DOTA,Tyr3,Thre8] octreotide uptake in CA20948 tumors: A small-animal PET study. Journal of Nuclear Medicine. 2011;52:580–5. doi: 10.2967/jnumed.110.084152. [DOI] [PubMed] [Google Scholar]
- 165.Cotugno G, Aurilio M, Annunziata P, Capalbo A, Faella A, Rinaldi V, Noninvasive Repetitive Imaging of Somatostatin Receptor 2 Gene Transfer with Positron Emission Tomography. Hum Gene Ther. 2011. [DOI] [PMC free article] [PubMed]
- 166.Tan EH, Goh SW. Exploring new frontiers in molecular imaging: Emergence of Ga PET/CT. World J Radiol. 2010;2:55–67. doi: 10.4329/wjr.v2.i2.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Naji M, A AL-N. (6)(8)Ga-labelled peptides in the management of neuroectodermal tumours. Eur J Nucl Med Mol Imaging. 2012;39(Suppl 1):S61–7. doi: 10.1007/s00259-011-1990-y. [DOI] [PubMed] [Google Scholar]
- 168.Kabasakal L, Demirci E, Ocak M, Decristoforo C, Araman A, Ozsoy Y, Comparison of (68)Ga-DOTATATE and (68)Ga-DOTANOC PET/CT imaging in the same patient group with neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2012. [DOI] [PubMed]
- 169.Traub-Weidinger T, Von Guggenberg E, Dobrozemsky G, Kendler D, Eisterer W, Bale R. et al. Preliminary experience with Ga-68-DOTA-lanreotide positron emission tomography. Q J Nucl Med Mol Imaging. 2010;54:52–60. [PubMed] [Google Scholar]
- 170.Gehler B, Paulsen F, Oksuz MO, Hauser TK, Eschmann SM, Bares R. et al. [68Ga]-DOTATOC-PET/CT for meningioma IMRT treatment planning. Radiat Oncol. 2009;4:56. doi: 10.1186/1748-717X-4-56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Milker-Zabel S, Zabel-du Bois A, Henze M, Huber P, Schulz-Ertner D, Hoess A. et al. Improved target volume definition for fractionated stereotactic radiotherapy in patients with intracranial meningiomas by correlation of CT, MRI, and [68Ga]-DOTATOC-PET. Int J Radiat Oncol Biol Phys. 2006;65:222–7. doi: 10.1016/j.ijrobp.2005.12.006. [DOI] [PubMed] [Google Scholar]
- 172.Gabriel M, Andergassen U, Putzer D, Kroiss A, Waitz D, Von Guggenberg E. et al. Individualized peptide-related-radionuclide-therapy concept using different radiolabelled somatostatin analogs in advanced cancer patients. Q J Nucl Med Mol Imaging. 2010;54:92–9. [PubMed] [Google Scholar]
- 173.Putzer D, Gabriel M, Kendler D, Henninger B, Knoflach M, Kroiss A. et al. Comparison of (68)Ga-DOTA-Tyr(3)-octreotide and (18)F-fluoro-L-dihydroxyphenylalanine positron emission tomography in neuroendocrine tumor patients. Q J Nucl Med Mol Imaging. 2010;54:68–75. [PubMed] [Google Scholar]
- 174.Win Z, Al-Nahhas A, Towey D, Todd JF, Rubello D, Lewington V. et al. 68Ga-DOTATATE PET in neuroectodermal tumours: first experience. Nucl Med Commun. 2007;28:359–63. doi: 10.1097/MNM.0b013e32808ea0b0. [DOI] [PubMed] [Google Scholar]
- 175.Modlin IM, Oberg K, Chung DC, Jensen RT, de Herder WW, Thakker RV. et al. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008;9:61–72. doi: 10.1016/S1470-2045(07)70410-2. [DOI] [PubMed] [Google Scholar]
- 176.Beauregard JM, Hofman MS, Kong G, Hicks RJ. The tumour sink effect on the biodistribution of 68Ga-DOTA-octreotate: implications for peptide receptor radionuclide therapy. Eur J Nucl Med Mol Imaging. 2012;39:50–6. doi: 10.1007/s00259-011-1937-3. [DOI] [PubMed] [Google Scholar]
- 177.Maecke HR, Hofmann M, Haberkorn U. (68)Ga-labeled peptides in tumor imaging. J Nucl Med. 2005;46(Suppl 1):172S–8S. [PubMed] [Google Scholar]
- 178.Prasad V, Baum RP. Biodistribution of the Ga-68 labeled somatostatin analogue DOTA-NOC in patients with neuroendocrine tumors: characterization of uptake in normal organs and tumor lesions. Q J Nucl Med Mol Imaging. 2010;54:61–7. [PubMed] [Google Scholar]
- 179.Ambrosini V, Nanni C, Zompatori M, Campana D, Tomassetti P, Castellucci P. et al. Ga-68-DOTA-NOC PET/CT in comparison with CT for the detection of bone metastasis in patients with neuroendocrine tumours. European Journal of Nuclear Medicine and Molecular Imaging. 2010;37:722–7. doi: 10.1007/s00259-009-1349-9. [DOI] [PubMed] [Google Scholar]
- 180.Haug AR, Auernhammer CJ, Wangler B, Schmidt GP, Uebleis C, Goke B. et al. 68Ga-DOTATATE PET/CT for the early prediction of response to somatostatin receptor-mediated radionuclide therapy in patients with well-differentiated neuroendocrine tumors. Journal of nuclear medicine. 2010;51:1349–56. doi: 10.2967/jnumed.110.075002. [DOI] [PubMed] [Google Scholar]
- 181.Frilling A, Sotiropoulos GC, Radtke A, Malago M, Bockisch A, Kuehl H. et al. The impact of 68Ga-DOTATOC positron emission tomography/computed tomography on the multimodal management of patients with neuroendocrine tumors. Ann Surg. 2010;252:850–6. doi: 10.1097/SLA.0b013e3181fd37e8. [DOI] [PubMed] [Google Scholar]
- 182.Versari A, Camellini L, Carlinfante G, Frasoldati A, Nicoli F, Grassi E. et al. Ga-68 DOTATOC PET, endoscopic ultrasonography, and multidetector CT in the diagnosis of duodenopancreatic neuroendocrine tumors: a single-centre retrospective study. Clin Nucl Med. 2010;35:321–8. doi: 10.1097/RLU.0b013e3181d6677c. [DOI] [PubMed] [Google Scholar]
- 183.Van Riet J, Rattat D, Verbruggen A, Mortelmans L, Mottaghy FM. Ga-68 DOTATOC PET/CT changed the therapeutic course of a patient with the sudden onset of vision problems. Clin Nucl Med. 2009;34:27–8. doi: 10.1097/RLU.0b013e31818f4588. [DOI] [PubMed] [Google Scholar]
- 184.Froeling V, Elgeti F, Maurer MH, Scheurig-Muenkler C, Beck A, Kroencke TJ. et al. Impact of Ga-68 DOTATOC PET/CT on the diagnosis and treatment of patients with multiple endocrine neoplasia. Ann Nucl Med. 2012;26:738–43. doi: 10.1007/s12149-012-0634-z. [DOI] [PubMed] [Google Scholar]
- 185.Ambrosini V, Campana D, Nanni C, Cambioli S, Tomassetti P, Rubello D. et al. Is (6)(8)Ga-DOTA-NOC PET/CT indicated in patients with clinical, biochemical or radiological suspicion of neuroendocrine tumour? Eur J Nucl Med Mol Imaging. 2012;39:1278–83. doi: 10.1007/s00259-012-2146-4. [DOI] [PubMed] [Google Scholar]
- 186.Nicolini S, Ambrosini V, Fantini L, Fanti S. 68Ga DOTANOC PET/CT detects medullary thyroid cancer relapse at bone level. Clin Nucl Med. 2012;37:591–2. doi: 10.1097/RLU.0b013e31824c5eab. [DOI] [PubMed] [Google Scholar]
- 187.Ambrosini V, Campana D, Allegri V, Opocher G, Fanti S. 68Ga-DOTA-NOC PET/CT detects somatostatin receptors expression in von hippel-lindau cerebellar disease. Clin Nucl Med. 2011;36:64–5. doi: 10.1097/RLU.0b013e3181fef14a. [DOI] [PubMed] [Google Scholar]
- 188.Zanoni L, Lopci E, Ambrosini V, Boschi S, Fanti S. Alveolar rhabdomyosarcoma with neuroendocrine differentiation detected by Ga-68 DOTA-NOC PET/CT: a case report. Clin Nucl Med. 2011;36:915–8. doi: 10.1097/RLU.0b013e31821a2691. [DOI] [PubMed] [Google Scholar]
- 189.Naswa N, Kumar A, Sharma P, Bal C, Malhotra A, Kumar R. Imaging carotid body chemodectomas with (6)(8)Ga-DOTA-NOC PET-CT. Br J Radiol. 2012;85:1140–5. doi: 10.1259/bjr/17448792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Ambrosini V, Zompatori M, De Luca F, Antonia DE, Allegri V, Nanni C. et al. 68Ga-DOTANOC PET/CT Allows Somatostatin Receptor Imaging in Idiopathic Pulmonary Fibrosis: Preliminary Results. J Nucl Med. 2010;51:1950–5. doi: 10.2967/jnumed.110.079962. [DOI] [PubMed] [Google Scholar]
- 191.Lincke T, Singer J, Kluge R, Sabri O, Paschke R. Relative quantification of indium-111 pentetreotide and gallium-68 DOTATOC uptake in the thyroid gland and association with thyroid pathologies. Thyroid. 2009;19:381–9. doi: 10.1089/thy.2008.0389. [DOI] [PubMed] [Google Scholar]
- 192.Luboldt W, Zophel K, Wunderlich G, Abramyuk A, Luboldt HJ, Kotzerke J. Visualization of somatostatin receptors in prostate cancer and its bone metastases with Ga-68-DOTATOC PET/CT. Mol Imaging Biol. 2010;12:78–84. doi: 10.1007/s11307-009-0230-3. [DOI] [PubMed] [Google Scholar]
- 193.Sainz-Esteban A, Prasad V, Baum RP. Pancreatic Neuroendocrine Tumor With Involvement of the Inferior Mesenteric Vein Diagnosed by Ga-68 DOTA-TATE PET/CT. Clin Nucl Med. 2010;35:40–1. doi: 10.1097/RLU.0b013e3181c3b74a. [DOI] [PubMed] [Google Scholar]
- 194.Lim TC, Tan EH, Zaheer S. Use of Ga-68 DOTATATE PET/CT to Confirm Portal Vein Tumor Thrombosis in a Patient With Pancreatic Neuroendocrine Tumor. Clin Nucl Med. 2011;36:498–9. doi: 10.1097/RLU.0b013e318217398b. [DOI] [PubMed] [Google Scholar]
- 195.Boss A, Bisdas S, Kolb A, Hofmann M, Ernemann U, Claussen CD. et al. Hybrid PET/MRI of Intracranial Masses: Initial Experiences and Comparison to PET/CT. J Nucl Med. 2010;51:1198–205. doi: 10.2967/jnumed.110.074773. [DOI] [PubMed] [Google Scholar]
- 196.Conry BG, Papathanasiou ND, Prakash V, Kayani I, Caplin M, Mahmood S. et al. Comparison of Ga-68-DOTATATE and F-18-fluorodeoxyglucose PET/CT in the detection of recurrent medullary thyroid carcinoma. Eur J Nucl Med Mol Imaging. 2010;37:49–57. doi: 10.1007/s00259-009-1204-z. [DOI] [PubMed] [Google Scholar]
- 197.Rominger A, Saam T, Vogl E, Ubleis C, la Fougere C, Forster S. et al. In Vivo Imaging of Macrophage Activity in the Coronary Arteries Using 68Ga-DOTATATE PET/CT: Correlation with Coronary Calcium Burden and Risk Factors. J Nucl Med. 2010;51:193–7. doi: 10.2967/jnumed.109.070672. [DOI] [PubMed] [Google Scholar]
- 198.Gambini JP, Quagliata A, Finozzi R, Serra P, Lago G, Gaudiano J. et al. Tc-99m- and Ga-68-labeled somatostatin analogues in the evaluation of Hurthle cell thyroid cancer. Clin Nucl Med. 2011;36:803–4. doi: 10.1097/RLU.0b013e318219b15c. [DOI] [PubMed] [Google Scholar]
- 199.Shastry M, Kayani I, Wild D, Caplin M, Visvikis D, Gacinovic S. et al. Distribution pattern of Ga-68-DOTATATE in disease-free patients. Nucl Med Comm. 2010;31:1025–32. doi: 10.1097/MNM.0b013e32833f635e. [DOI] [PubMed] [Google Scholar]
- 200.Al-Ibraheem A, Bundschuh RA, Notni J, Buck A, Winter A, Wester HJ. et al. Focal uptake of 68Ga-DOTATOC in the pancreas: pathological or physiological correlate in patients with neuroendocrine tumours? Eur J Nucl Med Mol Imaging. 2011;38:2005–13. doi: 10.1007/s00259-011-1875-0. [DOI] [PubMed] [Google Scholar]
- 201.Castellucci P, Pou Ucha J, Fuccio C, Rubello D, Ambrosini V, Montini GC. et al. Incidence of increased 68Ga-DOTANOC uptake in the pancreatic head in a large series of extrapancreatic NET patients studied with sequential PET/CT. J Nucl Med. 2011;52:886–90. doi: 10.2967/jnumed.111.088328. [DOI] [PubMed] [Google Scholar]
- 202.Krausz Y, Rubinstein R, Appelbaum L, Mishani E, Orevi M, Fraenkel M. et al. Ga-68 DOTA-NOC uptake in the pancreas: pathological and physiological patterns. Clin Nucl Med. 2012;37:57–62. doi: 10.1097/RLU.0b013e3182393404. [DOI] [PubMed] [Google Scholar]
- 203.Lincke T, Orschekowski G, Singer J, Sabri O, Paschke R. Increased Gallium-68 DOTATOC Uptake in Normal Thyroid Glands. Hormone and Metabolic Research. 2011;43:282–6. doi: 10.1055/s-0030-1270522. [DOI] [PubMed] [Google Scholar]
- 204.Lips N, Deroose CM, Bielen D, Bossuyt P, Mortelmans L. Wandering spleen on a 68Ga-DOTATOC-PET/CT scan. Eur J Nucl Med Mol Imaging. 2011;38:982. doi: 10.1007/s00259-010-1714-8. [DOI] [PubMed] [Google Scholar]
- 205.Campana D, Ambrosini V, Pezzilli R, Fanti S, Labate AMM, Santini D. et al. Standardized uptake values of 68Ga-DOTANOC PET: a promising prognostic tool in neuroendocrine tumors. J Nucl Med. 2010;51:353–9. doi: 10.2967/jnumed.109.066662. [DOI] [PubMed] [Google Scholar]
- 206.Boy C, Heusner TA, Poeppel TD, Redmann-Bischofs A, Unger N, Jentzen W. et al. 68Ga-DOTATOC PET/CT and somatostatin receptor (sst1-sst5) expression in normal human tissue: correlation of sst2 mRNA and SUVmax. Eur J Nucl Med Mol Imaging. 2011;38:1224–36. doi: 10.1007/s00259-011-1760-x. [DOI] [PubMed] [Google Scholar]
- 207.Haug AR, Rominger A, Mustafa M, Auernhammer C, Goke B, Schmidt GP. et al. Treatment with octreotide does not reduce tumor uptake of (68)Ga-DOTATATE as measured by PET/CT in patients with neuroendocrine tumors. J Nucl Med. 2011;52:1679–83. doi: 10.2967/jnumed.111.089276. [DOI] [PubMed] [Google Scholar]
- 208.Sainz-Esteban A, Prasad V, Schuchardt C, Zachert C, Carril JM, Baum RP. Comparison of sequential planar 177Lu-DOTA-TATE dosimetry scans with 68Ga-DOTA-TATE PET/CT images in patients with metastasized neuroendocrine tumours undergoing peptide receptor radionuclide therapy. Eur J Nucl Med Mol Imaging. 2012;39:501–11. doi: 10.1007/s00259-011-2003-x. [DOI] [PubMed] [Google Scholar]
- 209.Srirajaskanthan R, Kayani I, Quigley AM, Soh J, Caplin ME, Bomanji J. The role of 68Ga-DOTATATE PET in patients with neuroendocrine tumors and negative or equivocal findings on 111In-DTPA-Octreotide scintigraphy. J Nucl Med. 2010;51:875–82. doi: 10.2967/jnumed.109.066134. [DOI] [PubMed] [Google Scholar]
- 210.Kayani I, Bomanji JB, Groves A, Conway G, Gacinovic S, Win T. et al. Functional imaging of neuroendocrine tumors with combined PET/CT using 68Ga-DOTATATE (DOTA-DPhe1,Tyr3-octreotate) and 18F-FDG. Cancer. 2008;112:2447–55. doi: 10.1002/cncr.23469. [DOI] [PubMed] [Google Scholar]
- 211.Ambrosini V, Tomassetti P, Castellucci P, Campana D, Montini G, Rubello D. et al. Comparison between 68Ga-DOTA-NOC and 18F-DOPA PET for the detection of gastro-entero-pancreatic and lung neuro-endocrine tumours. Eur J Nucl Med Mol Imaging. 2008;35:1431–8. doi: 10.1007/s00259-008-0769-2. [DOI] [PubMed] [Google Scholar]
- 212.Kroiss A, Putzer D, Uprimny C, Decristoforo C, Gabriel M, Santner W. et al. Functional imaging in phaeochromocytoma and neuroblastoma with 68Ga-DOTA-Tyr 3-octreotide positron emission tomography and 123I-metaiodobenzylguanidine. Eur J Nucl Med Mol Imaging. 2011;38:865–73. doi: 10.1007/s00259-010-1720-x. [DOI] [PubMed] [Google Scholar]
- 213.Naji M, Zhao C, Welsh SJ, Meades R, Win Z, Ferrarese A. et al. 68Ga-DOTA-TATE PET vs. 123I-MIBG in identifying malignant neural crest tumours. Mol Imaging Biol. 2011;13:769–75. doi: 10.1007/s11307-010-0396-8. [DOI] [PubMed] [Google Scholar]
- 214.Putzer D, Gabriel M, Henninger B, Kendler D, Uprimny C, Dobrozemsky G. et al. Bone metastases in patients with neuroendocrine tumor: 68Ga-DOTA-Tyr3-Octreotide PET in comparison to CT and bone scintigraphy. J Nucl Med. 2009;50:1214–21. doi: 10.2967/jnumed.108.060236. [DOI] [PubMed] [Google Scholar]
- 215.Kaemmerer D, Prasad V, Daffner W, Haugvik SP, Senftleben S, Baum RP. et al. Radioguided surgery in neuroendocrine tumors using Ga-68-labeled somatostatin analogs: a pilot study. Clin Nucl Med. 2012;37:142–7. doi: 10.1097/RLU.0b013e3182291de8. [DOI] [PubMed] [Google Scholar]
- 216.Signore A, Glaudemans AWJM. The molecular imaging approach to image infections and inflammation by nuclear medicine techniques. Annals of Nuclear Medicine. 2011;25:681–700. doi: 10.1007/s12149-011-0521-z. [DOI] [PubMed] [Google Scholar]
- 217.Zhang HW, Schuhmacher J, Waser B, Wild D, Eisenhut M, Reubi JC. et al. DOTA-PESIN, a DOTA-conjugated bombesin derivative designed for the imaging and targeted radionuclide treatment of bombesin receptor-positive tumours. Eur J Nucl Med Mol Imaging. 2007;34:1198–208. doi: 10.1007/s00259-006-0347-4. [DOI] [PubMed] [Google Scholar]
- 218.Baum RP, Prasad V, Frischknecht M, Maecke H, Reubi J. Bombesin receptor imaging in various tumors: First results of Ga-68 AMBA PET/CT. Eur J Nucl Med Mol Imaging. 2007;34:S193–S. [Google Scholar]
- 219.Koumarianou E, Loktionova NS, Fellner M, Roesch F, Thews O, Pawlak D. et al. Sc-44-DOTA-BN 2-14 NH2 in comparison to Ga-68-DOTA-BN 2-14 NH2 in pre-clinical investigation. Is Sc-44 a potential radionuclide for PET? Appl Radiat Isot. 2012;70:2669–76. doi: 10.1016/j.apradiso.2012.08.004. [DOI] [PubMed] [Google Scholar]
- 220.Schuhmacher J, Zhang H, Doll J, Macke HR, Matys R, Hauser H. et al. GRP Receptor-Targeted PET of a Rat Pancreas Carcinoma Xenograft in Nude Mice with a 68Ga-Labeled Bombesin(6-14) Analog. J Nucl Med. 2005;46:691–9. [PubMed] [Google Scholar]
- 221.Dimitrakopoulou-Strauss A, Seiz M, Tuettenberg J, Schmieder K, Eisenhut M, Haberkorn U. et al. Pharmacokinetic studies of 68Ga-labeled bombesin ( 68Ga-BZH3) and F-18 FDG PET in patients with recurrent gliomas and comparison to grading: Preliminary results. Clin Nucl Med. 2011;36:101–8. doi: 10.1097/RLU.0b013e318203bb24. [DOI] [PubMed] [Google Scholar]
- 222.Hofmann M, Machtens S, Stief C, Langer F, Boerner AR, Maecke H. et al. Feasibility of Ga-68-DOTABOM PET in prostate carcinoma patients. Eur J Nucl Med Mol Imaging. 2004;31:S253–S. [Google Scholar]
- 223.Schroeder RP, van Weerden WM, Krenning EP, Bangma CH, Berndsen S, Grievink-de Ligt CH. et al. Gastrin-releasing peptide receptor-based targeting using bombesin analogues is superior to metabolism-based targeting using choline for in vivo imaging of human prostate cancer xenografts. European Journal of Nuclear Medicine and Molecular Imaging. 2011;38:1257–66. doi: 10.1007/s00259-011-1775-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Mansi R, Wang X, Forrer F, Waser B, Cescato R, Graham K. et al. Development of a potent DOTA-conjugated bombesin antagonist for targeting GRPr-positive tumours. Eur J Nucl Med Mol Imaging. 2011;38:97–107. doi: 10.1007/s00259-010-1596-9. [DOI] [PubMed] [Google Scholar]
- 225.Abiraj K, Mansi R, Tamma ML, Fani M, Forrer F, Nicolas G. et al. Bombesin antagonist-based radioligands for translational nuclear imaging of gastrin-releasing peptide receptor-positive tumors. J Nucl Med. 2011;52:1970–8. doi: 10.2967/jnumed.111.094375. [DOI] [PubMed] [Google Scholar]
- 226.Varasteh Z, Velikyan I, Lindegerg G, Sorensen J, Larhed M, Tolmachev V. et al. Synthesis and characterization of a high affinity NOTA-conjugated bombesin antagonist for GRPR-targeted tumor imaging. European Journal of Nuclear Medicine and Molecular Imaging. 2012;39:S240–S. doi: 10.1021/bc300659k. [DOI] [PubMed] [Google Scholar]
- 227.Liu ZF, Yan YJ, Liu SL, Wang F, Chen XY. F-18, Cu-64, and Ga-68 Labeled RGD-Bombesin Heterodimeric Peptides for PET Imaging of Breast Cancer. Bioconjugate Chemistry. 2009;20:1016–25. doi: 10.1021/bc9000245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Liu Z, Niu G, Wang F, Chen X. (68)Ga-labeled NOTA-RGD-BBN peptide for dual integrin and GRPR-targeted tumor imaging. Eur J Nucl Med Mol Imaging. 2009;36:1483–94. doi: 10.1007/s00259-009-1123-z. [DOI] [PubMed] [Google Scholar]
- 229.Brom M, Oyen WJ, Joosten L, Gotthardt M, Boerman OC. 68Ga-labelled exendin-3, a new agent for the detection of insulinomas with PET. Eur J Nucl Med Mol Imaging. 2010;37:1345–55. doi: 10.1007/s00259-009-1363-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Wild D, Wicki A, Mansi R, Behe M, Keil B, Bernhardt P. et al. Exendin-4-Based Radiopharmaceuticals for Glucagonlike Peptide-1 Receptor PET/CT and SPECT/CT. J Nucl Med. 2010;51:1059–67. doi: 10.2967/jnumed.110.074914. [DOI] [PubMed] [Google Scholar]
- 231.Selvaraju R, Velikyan I, Johansson L, Wu Z, Todorov I, Shively J, In vivo imaging of the Glucagon Like Peptide-1 receptor in pancreas by 68Ga-DO3A-exendin-4. Journal of Nuclear Medicine. 2013. [DOI] [PubMed]
- 232.Brom M, Joosten L, Laverman P, Oyen WJG, Béhé M, Gotthardt M. et al. Preclinical evaluation of 68Ga-DOTA-minigastrin for the detection of cholecystokinin-2/gastrin receptor-positive tumors. Molecular Imaging. 2011;10:144–52. [PMC free article] [PubMed] [Google Scholar]
- 233.von Guggenberg E, Rangger C, Sosabowski J, Laverman P, Reubi JC, Virgolini IJ. et al. Preclinical evaluation of radiolabeled DOTA-derivatized cyclic minigastrin analogs for targeting cholecystokinin receptor expressing malignancies. Mol Imaging Biol. 2012;14:366–75. doi: 10.1007/s11307-011-0506-2. [DOI] [PubMed] [Google Scholar]
- 234.Gourni E, Demmer O, Schottelius M, D'Alessandria C, Schulz S, Dijkgraaf I. et al. PET of CXCR4 expression by a (68)Ga-labeled highly specific targeted contrast agent. J Nucl Med. 2011;52:1803–10. doi: 10.2967/jnumed.111.098798. [DOI] [PubMed] [Google Scholar]
- 235.Demmer O, Dijkgraaf I, Schumacher U, Marinelli L, Cosconati S, Gourni E. et al. Design, synthesis, and functionalization of dimeric peptides targeting chemokine receptor CXCR4. J Med Chem. 2011;54:7648–62. doi: 10.1021/jm2009716. [DOI] [PubMed] [Google Scholar]
- 236.Demmer O, Gourni E, Schumacher U, Kessler H, Wester HJ. PET imaging of CXCR4 receptors in cancer by a new optimized ligand. ChemMedChem. 2011;6:1789–91. doi: 10.1002/cmdc.201100320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Hennrich U, Seyler L, Schafer M, Bauder-Wust U, Eisenhut M, Semmler W. et al. Synthesis and in vitro evaluation of 68Ga-DOTA-4-FBn-TN14003, a novel tracer for the imaging of CXCR4 expression. Bioorg Med Chem. 2012;20:1502–10. doi: 10.1016/j.bmc.2011.12.052. [DOI] [PubMed] [Google Scholar]
- 238.Froidevaux S, Calame-Christe M, Schuhmacher J, Tanner H, Saffrich R, Henze M. et al. A Gallium-Labeled DOTA-{alpha}-Melanocyte- Stimulating Hormone Analog for PET Imaging of Melanoma Metastases. J Nucl Med. 2004;45:116–23. [PubMed] [Google Scholar]
- 239.Cantorias MV, Figueroa SD, Quinn TP, Lever JR, Hoffman TJ, Watkinson LD. et al. Development of high-specific-activity Ga-68-labeled DOTA-rhenium-cyclized alpha-MSH peptide analog to target MC1 receptors overexpressed by melanoma tumors. Nucl Med Biol. 2009;36:505–13. doi: 10.1016/j.nucmedbio.2009.01.017. [DOI] [PubMed] [Google Scholar]
- 240.Wei L, Miao Y, Gallazzi F, Quinn TP, Welch MJ, Vavere AL. et al. Gallium-68-labeled DOTA-rhenium-cyclized alpha-melanocyte-stimulating hormone analog for imaging of malignant melanoma. Nucl Med Biol. 2007;34:945–53. doi: 10.1016/j.nucmedbio.2007.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Guo H, Gallazzi F, Miao Y. Gallium-67-labeled lactam bridge-cyclized alpha-MSH peptides with enhanced melanoma uptake and reduced renal uptake. Bioconjugate Chem. 2012;23:1341–8. doi: 10.1021/bc300191z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Kim HJ, Kim DY, Park JH, Yang SD, Hur MG, Min JJ. et al. Synthesis and evaluation of a novel 68Ga-labeled DOTA-benzamide derivative for malignant melanoma imaging. Bioorganic & Medicinal Chemistry Letters. 2012;22:5288–92. doi: 10.1016/j.bmcl.2012.06.043. [DOI] [PubMed] [Google Scholar]
- 243.Schottelius M, Berger S, Poethko T, Schwaiger M, Wester HJ. Development of novel Ga-68- and F-18-labeled GnRH-I analogues with high GnRHR-targeting efficiency. Bioconjugate Chem. 2008;19:1256–68. doi: 10.1021/bc800058k. [DOI] [PubMed] [Google Scholar]
- 244.Demmer O, Dijkgraaf I, Schottelius M, Wester HJ, Kessler H. Introduction of functional groups into peptides via N-alkylation. Organic Letters. 2008;10:2015–8. doi: 10.1021/ol800654n. [DOI] [PubMed] [Google Scholar]
- 245.Maschauer S, Einsiedel J, Hocke C, Hubner H, Kuwert T, Gmeiner P. et al. Synthesis of a Ga-68-Labeled Peptoid-Peptide Hybrid for Imaging of Neurotensin Receptor Expression in Vivo. Acs Medicinal Chemistry Letters. 2010;1:224–8. doi: 10.1021/ml1000728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Velikyan I, Sundberg AL, Lindhe O, Hoglund AU, Eriksson O, Werner E. et al. Preparation and evaluation of (68)Ga-DOTA-hEGF for visualization of EGFR expression in malignant tumors. J Nucl Med. 2005;46:1881–8. [PubMed] [Google Scholar]
- 247.Tolmachev V, Velikyan I, Sandstrom M, Orlova A. A HER2-binding Affibody molecule labelled with 68Ga for PET imaging: direct in vivo comparison with the 111In-labelled analogue. Eur J Nucl Med Mol Imaging. 2010;37:1356–67. doi: 10.1007/s00259-009-1367-7. [DOI] [PubMed] [Google Scholar]
- 248.Velikyan I. Synthesis, Characterization and Application of 68Ga-Labelled Macromolecules; http://urn.kb.se/resolve?urn=urn:nbn:se:uu:diva-5876 [Doctorate thesis] Uppsala: Uppsala University; 2005. [Google Scholar]
- 249.Kramer-Marek G, Shenoy N, Seidel J, Griffiths GL, Choyke P, Capala J. 68Ga-DOTA-affibody molecule for in vivo assessment of HER2/neu expression with PET. Eur J Nucl Med Mol Imaging. 2011;38:1967–76. doi: 10.1007/s00259-011-1810-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Ren G, Zhang R, Liu Z, Webster JM, Miao Z, Gambhir SS. et al. A 2-Helix Small Protein Labeled with 68Ga for PET Imaging of HER2 Expression. J Nucl Med. 2009;50:1492–9. doi: 10.2967/jnumed.109.064287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Smith-Jones PM, Solit DB, Akhurst T, Afroze F, Rosen N, Larson SM. Imaging the pharmacodynamics of HER2 degradation in response to Hsp90 inhibitors. Nat Biotechnol. 2004;22:701–6. doi: 10.1038/nbt968. [DOI] [PubMed] [Google Scholar]
- 252.Theeraladanon C, Takahashi N, Shiina M, Hamada K, Takada Y, Endo H. et al. Rational Approach to the Synthesis, Evaluation, and 68Ga Labeling of a Novel 4-Anilinoquinoline Epidermal Growth Factor Receptor Inhibitor as a New Imaging Agent That Selectively Targets the Epidermal Growth Factor Receptor Tyrosine Kinase. Cancer Biother Radio. 2010;25:479–85. doi: 10.1089/cbr.2009.0614. [DOI] [PubMed] [Google Scholar]
- 253.Sandstrom K, Haylock AK, Velikyan I, Spiegelberg D, Kareem H, Tolmachev V. et al. Improved Tumor-to-Organ Ratios of a Novel (67)Ga-Human Epidermal Growth Factor Radionuclide Conjugate with Preadministered Antiepidermal Growth Factor Receptor Affibody Molecules. Cancer Biother Radio. 2011;26:593–601. doi: 10.1089/cbr.2011.0981. [DOI] [PubMed] [Google Scholar]
- 254.Smith-Jones PM, Solit D, Afroze F, Rosen N, Larson SM. Early Tumor Response to Hsp90 Therapy Using HER2 PET: Comparison with 18F-FDG PET. J Nucl Med. 2006;47:793–6. [PMC free article] [PubMed] [Google Scholar]
- 255.Vosjan MJ, Perk LR, Roovers RC, Visser GW, Stigter-van Walsum M, van Bergen En Henegouwen PM, Facile labelling of an anti-epidermal growth factor receptor Nanobody with (68)Ga via a novel bifunctional desferal chelate for immuno-PET. Eur J Nucl Med Mol Imaging. 2011. [DOI] [PMC free article] [PubMed]
- 256.Hong H, Sun J, Cai W. Radionuclide-based cancer imaging targeting the carcinoembryonic antigen. Biomark Insights. 2008;3:435–51. doi: 10.4137/bmi.s1124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Eder M, Knackmuss S, Le Gall F, Reusch U, Rybin V, Little M. et al. 68Ga-labelled recombinant antibody variants for immuno-PET imaging of solid tumours. Eur J Nucl Med Mol Imaging. 2010;37:1397–407. doi: 10.1007/s00259-010-1392-6. [DOI] [PubMed] [Google Scholar]
- 258.Sharkey RM, Cardillo TM, Rossi EA, Chang CH, Karacay H, McBride WJ. et al. Signal amplification in molecular imaging by pretargeting a multivalent, bispecific antibody. Nature medicine. 2005;11:1250–5. doi: 10.1038/nm1322. [DOI] [PubMed] [Google Scholar]
- 259.Goldenberg DM, Sharkey RM. Novel radiolabeled antibody conjugates. Oncogene. 2007;26:3734–44. doi: 10.1038/sj.onc.1210373. [DOI] [PubMed] [Google Scholar]
- 260.Goldenberg DM, Rossi EA, Sharkey RM, McBride WJ, Chang CH. Multifunctional antibodies by the Dock-and-Lock method for improved cancer imaging and therapy by pretargeting. J Nucl Med. 2008;49:158–63. doi: 10.2967/jnumed.107.046185. [DOI] [PubMed] [Google Scholar]
- 261.Goldenberg DM, Chang CH, Rossi EA. et al. Pretargeted molecular imaging and radioimmunotherapy. Theranostics. 2012;2:523–40. doi: 10.7150/thno.3582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Schoffelen R, Sharkey RM, Goldenberg DM, Franssen G, McBride WJ, Rossi EA. et al. Pretargeted immuno-positron emission tomography imaging of carcinoembryonic antigen-expressing tumors with a bispecific antibody and a 68Ga- and 18F-labeled hapten peptide in mice with human tumor xenografts. Mol Cancer Ther. 2010;9:1019–27. doi: 10.1158/1535-7163.MCT-09-0862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Karacay H, Sharkey RM, McBride WJ, Rossi EA, Chang CH, Goldenberg DM. Optimization of hapten-peptide labeling for pretargeted immunoPET of bispecific antibody using generator-produced 68Ga. Journal of nuclear medicine. 2011;52:555–9. doi: 10.2967/jnumed.110.083568. [DOI] [PubMed] [Google Scholar]
- 264.Hall H, Velikyan I, Blom E, Ulin J, Monazzam A, Pahlman L. et al. In vitro autoradiography of carcinoembryonic antigen in tissue from patients with colorectal cancer using multifunctional antibody TF2 and (67/68Ga)-labeled haptens by pretargeting. Am J Nucl Med Mol Imaging. 2012;2:141–50. [PMC free article] [PubMed] [Google Scholar]
- 265.Eriksson O, Carlsson F, Blom E, Sundin A, Langstrom B, Korsgren O. et al. Preclinical evaluation of a 68Ga-labeled biotin analogue for applications in islet transplantation. Nucl Med Biol. 2012;39:415–21. doi: 10.1016/j.nucmedbio.2011.09.009. [DOI] [PubMed] [Google Scholar]
- 266.Blom E, Langstrom B, Velikyan I. (68)Ga-Labeling of Biotin Analogues and their Characterization. Bioconjug Chem. 2009;20:1146–51. doi: 10.1021/bc800538s. [DOI] [PubMed] [Google Scholar]
- 267.Dijkgraaf I, Boerman OC. Radionuclide imaging of tumor angiogenesis. Cancer Biother Radiopharm. 2009;24:637–47. doi: 10.1089/cbr.2009.0694. [DOI] [PubMed] [Google Scholar]
- 268.Velikyan I, Långström B, Bergström M, Lindhe Ö. 68Ga-labeled peptide-based radiopharmaceuticals. USA: WO/2008/026040; 2008. [Google Scholar]
- 269.Decristoforo C, Hernandez Gonzalez I, Carlsen J, Rupprich M, Huisman M, Virgolini I. et al. 68Ga- and 111In-labelled DOTA-RGD peptides for imaging of alphavbeta3 integrin expression. Eur J Nucl Med Mol Imaging. 2008;35:1507–15. doi: 10.1007/s00259-008-0757-6. [DOI] [PubMed] [Google Scholar]
- 270.Haukkala J, Laitinen I, Luoto P, Iveson P, Wilson I, Karlsen H. et al. (68)Ga-DOTA-RGD peptide: biodistribution and binding into atherosclerotic plaques in mice. Eur J Nucl Med Mol Imaging. 2009;36:2058. doi: 10.1007/s00259-009-1220-z. [DOI] [PubMed] [Google Scholar]
- 271.Dumont RA, Deininger F, Haubner R, Maecke HR, Weber WA, Fani M. Novel (64)Cu- and (68)Ga-labeled RGD conjugates show improved PET imaging of alpha(nu)beta(3) integrin expression and facile radiosynthesis. J Nucl Med. 2011;52:1276–84. doi: 10.2967/jnumed.111.087700. [DOI] [PubMed] [Google Scholar]
- 272.Knetsch PA, Petrik M, Griessinger CM, Rangger C, Fani M, Kesenheimer C. et al. [68Ga]NODAGA-RGD for imaging alphavbeta3 integrin expression. Eur J Nucl Med Mol Imaging. 2011;38:1303–12. doi: 10.1007/s00259-011-1778-0. [DOI] [PubMed] [Google Scholar]
- 273.Ferreira CL, Yapp DT, Mandel D, Gill RK, Boros E, Wong MQ. et al. (68)Ga Small Peptide Imaging: Comparison of NOTA and PCTA. Bioconjug Chem. 2012;23:2239–46. doi: 10.1021/bc300348d. [DOI] [PubMed] [Google Scholar]
- 274.Blom E, Velikyan I, Estrada S, Hall H, Muhammad T, Ding C. et al. (68)Ga-Labeling of RGD peptides and biodistribution. Int J Clin Exp Med. 2012;5:165–72. [PMC free article] [PubMed] [Google Scholar]
- 275.Li ZB, Chen K, Chen X. (68)Ga-labeled multimeric RGD peptides for microPET imaging of integrin alpha(v)beta (3) expression. Eur J Nucl Med Mol Imaging. 2008;35:1100–8. doi: 10.1007/s00259-007-0692-y. [DOI] [PubMed] [Google Scholar]
- 276.Dijkgraaf I, Yim CB, Franssen GM, Schuit RC, Luurtsema G, Liu S. et al. PET imaging of alphavbeta integrin expression in tumours with Ga-labelled mono-, di- and tetrameric RGD peptides. Eur J Nucl Med Mol Imaging. 2011;38:128–37. doi: 10.1007/s00259-010-1615-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Singh AN, Liu W, Hao GY, Kumar A, Gupta A, Oz OK. et al. Multivalent Bifunctional Chelator Scaffolds for Gallium-68 Based Positron Emission Tomography Imaging Probe Design: Signal Amplification via Multivalency. Bioconjugate Chem. 2011;22:1650–62. doi: 10.1021/bc200227d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Liu Z, Niu G, Shi J, Liu S, Wang F, Chen X. (68)Ga-labeled cyclic RGD dimers with Gly3 and PEG4 linkers: promising agents for tumor integrin alphavbeta3 PET imaging. Eur J Nucl Med Mol Imaging. 2009;36:947–57. doi: 10.1007/s00259-008-1045-1. [DOI] [PubMed] [Google Scholar]
- 279.Backer MV, Levashova Z, Levenson R, Blankenberg FG, Backer JM. Cysteine-containing fusion tag for site-specific conjugation of therapeutic and imaging agents to targeting proteins. Methods in molecular biology (Clifton, NJ. 2008;494:275–94. doi: 10.1007/978-1-59745-419-3_16. [DOI] [PubMed] [Google Scholar]
- 280.Backer MV, Backer JM. Imaging key biomarkers of tumor angiogenesis. Theranostics. 2012;2:502–15. doi: 10.7150/thno.3623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Blom E, Velikyan I, Monazzam A, Razifar P, Nair M, Razifar P. et al. Synthesis and characterization of scVEGF-PEG-[68Ga]NOTA and scVEGF-PEG-[68Ga]DOTA PET tracers. J Labelled Comp Radiopharm. 2011;54:685–92. [Google Scholar]
- 282.Ujula T, Huttunen M, Luoto P, Perakyla H, Simpura I, Wilson I. et al. Matrix Metalloproteinase 9 Targeting Peptides: Syntheses, Ga-68-labeling, and Preliminary Evaluation in a Rat Melanoma Xenograft Model. Bioconjugate Chem. 2010;21:1612–21. doi: 10.1021/bc1000643. [DOI] [PubMed] [Google Scholar]
- 283.Azhdarinia A, Wilganowski N, Robinson H, Ghosh P, Kwon S, Lazard ZW. et al. Characterization of chemical, radiochemical and optical properties of a dual-labeled MMP-9 targeting peptide. Bioorg Med Chem. 2011;19:3769–76. doi: 10.1016/j.bmc.2011.04.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Suzuki K, Satake M, Suwada J, Oshikiri S, Ashino H, Dozono H. et al. Synthesis and evaluation of a novel (68)Ga-chelate-conjugated bisphosphonate as a bone-seeking agent for PET imaging. Nuclear Medicine and Biology. 2011;38:1011–8. doi: 10.1016/j.nucmedbio.2011.02.015. [DOI] [PubMed] [Google Scholar]
- 285.Ogawa K, Takai K, Kanbara H, Kiwada T, Kitamura Y, Shiba K. et al. Preparation and evaluation of a radiogallium complex-conjugated bisphosphonate as a bone scintigraphy agent. Nuclear Medicine and Biology. 2011;38:631–6. doi: 10.1016/j.nucmedbio.2010.12.004. [DOI] [PubMed] [Google Scholar]
- 286.Fellner M, Biesalski B, Bausbacher N, Kubícek V, Hermann P, Rösch F. et al. 68Ga-BPAMD: PET-imaging of bone metastases with a generator based positron emitter. Nuclear Medicine and Biology. 2012;39:993–9. doi: 10.1016/j.nucmedbio.2012.04.007. [DOI] [PubMed] [Google Scholar]
- 287.Fellner M, Riss P, Loktionova N, Zhernosekov K, Thews O, Geraldes CFGC. et al. Comparison of different phosphorus-containing ligands complexing 68Ga for PET-imaging of bone metabolism. Radiochimica Acta. 2011;99:43–51. [Google Scholar]
- 288.Toegel S, Wadsak W, Mien LK, Viernstein H, Kluger R, Eidherr H. et al. Preparation and pre-vivo evaluation of no-carrier-added, carrier-added and cross-complexed Ga-68 -EDTMP formulations. European Journal of Pharmaceutics and Biopharmaceutics. 2008;68:406–12. doi: 10.1016/j.ejpb.2007.05.015. [DOI] [PubMed] [Google Scholar]
- 289.Mitterhauser M, Toegel S, Wadsak W, Lanzenberger RR, Mien LK, Kuntner C. et al. Pre vivo, ex vivo and in vivo evaluations of Ga-68 -EDTMP. Nuclear Medicine and Biology. 2007;34:391–7. doi: 10.1016/j.nucmedbio.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 290.Hoigebazar L, Jeong JM, Hong MK, Young JK, Lee JY, Shetty D. et al. Synthesis of Ga-68-labeled DOTA-nitroimidazole derivatives and their feasibilities as hypoxia imaging PET tracers. Bioorg Med Chem. 2011;19:2176–81. doi: 10.1016/j.bmc.2011.02.041. [DOI] [PubMed] [Google Scholar]
- 291.Hoigebazar L, Jeong JM, Choi SY, Choi JY, Shetty D, Lee YS. et al. Synthesis and characterization of nitroimidazole derivatives for 68Ga-labeling and testing in tumor xenografted mice. J Med Chem. 2010;53:6378–85. doi: 10.1021/jm100545a. [DOI] [PubMed] [Google Scholar]
- 292.Mukai T, Suwada J, Sano K, Okada M, Yamamoto F, Maeda M. Design of Ga-DOTA-based bifunctional radiopharmaceuticals: Two functional moieties can be conjugated to radiogallium-DOTA without reducing the complex stability. Bioorg Med Chem. 2009;17:4285–9. doi: 10.1016/j.bmc.2009.05.041. [DOI] [PubMed] [Google Scholar]
- 293.Fernandez S, Dematteis S, Giglio J, Cerecetto H, Rey A. Synthesis, in vitro and in vivo characterization of two novel (68)Ga-labelled 5-nitroimidazole derivatives as potential agents for imaging hypoxia. Nucl Med Biol. 2012. [DOI] [PubMed]
- 294.Shetty D, Jeong JM, Ju CH, Lee YS, Jeong SY, Choi JY. et al. Synthesis of novel 68Ga-labeled amino acid derivatives for positron emission tomography of cancer cells. Nucl Med Biol. 2010;37:893–902. doi: 10.1016/j.nucmedbio.2010.06.003. [DOI] [PubMed] [Google Scholar]
- 295.Shetty D, Jeong JM, Ju CH, Kim YJ, Lee JY, Lee YS. et al. Synthesis and evaluation of macrocyclic amino acid derivatives for tumor imaging by gallium-68 positron emission tomography. Bioorganic & Medicinal Chemistry. 2010;18:7338–47. doi: 10.1016/j.bmc.2010.09.022. [DOI] [PubMed] [Google Scholar]
- 296.Burchardt C, Riss PJ, Zoller F, Maschauer S, Prante O, Kuwert T. et al. Ga-68 Ga-DO(2)A-(OBu-L-tyr)(2): Synthesis, Ga-68-radiolabeling and in vitro studies of a novel Ga-68-DO(2)A-tyrosine conjugate as potential tumor tracer for PET. Bioorganic & Medicinal Chemistry Letters. 2009;19:3498–501. doi: 10.1016/j.bmcl.2009.05.001. [DOI] [PubMed] [Google Scholar]
- 297.Yang Z, Xiong C, Zhang R, Zhu H, Li C. Synthesis and evaluation of (68)Ga-labeled DOTA-2-deoxy-D-glucosamine as a potential radiotracer in muPET imaging. Am J Nucl Med Mol Imaging. 2012;2:499–507. [PMC free article] [PubMed] [Google Scholar]
- 298.Jain R, Dandekar P, Patravale V. Diagnostic nanocarriers for sentinel lymph node imaging. J Control Release. 2009;138:90–102. doi: 10.1016/j.jconrel.2009.05.010. [DOI] [PubMed] [Google Scholar]
- 299.Choi JY, Jeong JM, Yoo BC, Kim K, Kim Y, Yang BY. et al. Development of Ga-68-labeled mannosylated human serum albumin (MSA) as a lymph node imaging agent for positron emission tomography. Nucl Med Biol. 2011;38:371–9. doi: 10.1016/j.nucmedbio.2010.09.010. [DOI] [PubMed] [Google Scholar]
- 300.Lee YK, Jeong JM, Hoigebazar L, Yang BY, Lee YS, Lee BC. et al. Nanoparticles modified by encapsulation of ligands with a long alkyl chain to affect multispecific and multimodal imaging. J Nucl Med. 2012;53:1462–70. doi: 10.2967/jnumed.111.092759. [DOI] [PubMed] [Google Scholar]
- 301.Hong H, Zhang Y, Engle JW, Nayak TR, Theuer CP, Nickles RJ. et al. In vivo targeting and positron emission tomography imaging of tumor vasculature with (66)Ga-labeled nano-graphene. Biomaterials. 2012;33:4147–56. doi: 10.1016/j.biomaterials.2012.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Hwang DW, Ko HY, Lee JH, Kang H, Ryu SH, Song IC. et al. A Nucleolin-Targeted Multimodal Nanoparticle Imaging Probe for Tracking Cancer Cells Using an Aptamer. J Nucl Med. 2010;51:98–105. doi: 10.2967/jnumed.109.069880. [DOI] [PubMed] [Google Scholar]
- 303.Ndiege N, Raidoo R, Schultz MK, Larsen S. Preparation of a Versatile Bifunctional Zeolite for Targeted Imaging Applications. Langmuir: the ACS journal of surfaces and colloids; 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.de Sa A, Prata MIM, Geraldes C, Andre JP. Triaza-based amphiphilic chelators: Synthetic route, in vitro characterization and in vivo studies of their Ga(III) and Al(III) chelates. Journal of Inorganic Biochemistry. 2010;104:1051–62. doi: 10.1016/j.jinorgbio.2010.06.002. [DOI] [PubMed] [Google Scholar]
- 305.Fontes A, Prata MIM, Geraldes C, Andre JP. Ga(III) chelates of amphiphilic DOTA-based ligands: synthetic route and in vitro and in vivo studies. Nucl Med Biol. 2011;38:363–70. doi: 10.1016/j.nucmedbio.2010.10.003. [DOI] [PubMed] [Google Scholar]
- 306.Fani M, Wang X, Nicolas G, Medina C, Raynal I, Port M. et al. Development of new folate-based PET radiotracers: preclinical evaluation of Ga-DOTA-folate conjugates. Eur J Nucl Med Mol Imaging. 2011;38:108–19. doi: 10.1007/s00259-010-1597-8. [DOI] [PubMed] [Google Scholar]
- 307.Fani M, Tamma ML, Nicolas GP, Lasri E, Medina C, Raynal I. et al. In vivo imaging of folate receptor positive tumor xenografts using novel 68Ga-NODAGA-folate conjugates. Molecular pharmaceutics. 2012;9:1136–45. doi: 10.1021/mp200418f. [DOI] [PubMed] [Google Scholar]
- 308.Persson M, Madsen J, Ostergaard S, Ploug M, Kjaer A. 68Ga-labeling and in vivo evaluation of a uPAR binding DOTA- and NODAGA-conjugated peptide for PET imaging of invasive cancers. Nucl Med Biol. 2012;39:560–9. doi: 10.1016/j.nucmedbio.2011.10.011. [DOI] [PubMed] [Google Scholar]
- 309.Thews O, Dillenburg W, Fellner M, Buchholz HG, Bausbacher N, Schreckenberger M. et al. Activation of P-glycoprotein (Pgp)-mediated drug efflux by extracellular acidosis: in vivo imaging with Ga-68-labelled PET tracer. Eur J Nucl Med Mol Imaging. 2010;37:1935–42. doi: 10.1007/s00259-010-1504-3. [DOI] [PubMed] [Google Scholar]
- 310.Fellner M, Dillenburg W, Buchholz HG, Bausbacher N, Schreckenberger M, Renz F. et al. Assessing p-glycoprotein (Pgp) activity in vivo utilizing 68Ga-Schiff base complexes. Mol Imaging Biol. 2011;13:985–94. doi: 10.1007/s11307-010-0410-1. [DOI] [PubMed] [Google Scholar]
- 311.Bauwens M, De Saint-Hubert M, Devos E, Deckers N, Reutelingsperger C, Mortelmans L. et al. Site-specific Ga-68-labeled Annexin A5 as a PET imaging agent for apoptosis. Nucl Med Biol. 2011;38:381–92. doi: 10.1016/j.nucmedbio.2010.09.008. [DOI] [PubMed] [Google Scholar]
- 312.Banerjee SR, Pullambhatla M, Byun Y, Nimmagadda S, Green G, Fox JJ. et al. Ga-68-Labeled Inhibitors of Prostate-Specific Membrane Antigen (PSMA) for Imaging Prostate Cancer. J Med Chem. 2010;53:5333–41. doi: 10.1021/jm100623e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Eder M, Schafer M, Bauder-Wust U, Hull WE, Wangler C, Mier W. et al. 68Ga-complex lipophilicity and the targeting property of a urea-based PSMA inhibitor for PET imaging. Bioconjug Chem. 2012;23:688–97. doi: 10.1021/bc200279b. [DOI] [PubMed] [Google Scholar]
- 314.Afshar-Oromieh A, Malcher A, Eder M, Eisenhut M, Linhart HG, Hadaschik BA. et al. PET imaging with a [(68)Ga]gallium-labelled PSMA ligand for the diagnosis of prostate cancer: biodistribution in humans and first evaluation of tumour lesions. Eur J Nucl Med Mol Imaging. 2012;39:1085–6. doi: 10.1007/s00259-012-2298-2. [DOI] [PubMed] [Google Scholar]
- 315.Lendvai G, Estrada S, Bergstrom M. Radiolabelled oligonucleotides for imaging of gene expression with PET. Curr Med Chem. 2009;16:4445–61. doi: 10.2174/092986709789712844. [DOI] [PubMed] [Google Scholar]
- 316.Lendvai G, Monazzam A, Velikyan I, Eriksson B, Josephsson R, Langstrom B. et al. Non-Hybridization Saturable Mechanisms Play a Role in the Uptake of (68)Ga-Labeled LNA-DNA Mixmer Antisense Oligonucleotides in Rats. Oligonucleotides. 2009;19:223–32. doi: 10.1089/oli.2009.0192. [DOI] [PubMed] [Google Scholar]
- 317.Lendvai G, Velikyan I, Bergstrom M, Estrada S, Laryea D, Valila M. et al. Biodistribution of 68Ga-labelled phosphodiester, phosphorothioate, and 2'-O-methyl phosphodiester oligonucleotides in normal rats. Eur J Pharm Sci. 2005;26:26–38. doi: 10.1016/j.ejps.2005.04.017. [DOI] [PubMed] [Google Scholar]
- 318.Lendvai G, Velikyan I, Estrada S, Eriksson B, Langstrom B, Bergstrom M. Biodistribution of (68)Ga-Labeled LNA-DNA Mixmer Antisense Oligonucleotides for Rat Chromogranin-A. Oligonucleotides. 2008;18:33–49. doi: 10.1089/oli.2007.0104. [DOI] [PubMed] [Google Scholar]
- 319.Roivainen A, Tolvanen T, Salomaki S, Lendvai G, Velikyan I, Numminen P. et al. 68Ga-labeled oligonucleotides for in vivo imaging with PET. J Nucl Med. 2004;45:347–55. [PubMed] [Google Scholar]
- 320.Velikyan I, Lendvai G, Valila M, Roivainen A, Yngve U, Bergstrom M. et al. Microwave accelerated 68Ga-labelling of oligonucleotides. Journal of Labelled Compounds & Radiopharmaceuticals. 2004;47:79–89. [Google Scholar]
- 321.Kiviniemi A, Makela J, Makila J, Saanijoki T, Liljenback H, Poijarvi-Virta P. et al. Solid-supported NOTA and DOTA chelators useful for the synthesis of 3'-radiometalated oligonucleotides. Bioconjugate Chemistry. 2012;23:1981–8. doi: 10.1021/bc300253t. [DOI] [PubMed] [Google Scholar]
- 322.Plossl K, Chandra R, Qua WC, Lieberman BP, Kung MP, Zhou R. et al. A novel gallium bisaminothiolate complex as a myocardial perfusion imaging agent. Nuclear Medicine and Biology. 2008;35:83–90. doi: 10.1016/j.nucmedbio.2007.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Yang BY, Jeong JM, Kim YJ, Choi JY, Lee YS, Lee DS. et al. Formulation of 68Ga BAPEN kit for myocardial positron emission tomography imaging and biodistribution study. Nucl Med Biol. 2010;37:149–55. doi: 10.1016/j.nucmedbio.2009.10.010. [DOI] [PubMed] [Google Scholar]
- 324.Hsiao YM, Mathias CJ, Wey SP, Fanwick PE, Green MA. Synthesis and biodistribution of lipophilic and monocationic gallium radiopharmaceuticals derived from N,N '-bis(3-aminopropyl)-N,N '-dimethylethylenediamine: potential agents for PET myocardial imaging with Ga-68. Nuclear Medicine and Biology. 2009;36:39–45. doi: 10.1016/j.nucmedbio.2008.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Tarkia M, Saraste A, Saanijoki T, Oikonen V, Vahasilta T, Strandberg M. et al. Evaluation of 68Ga-labeled tracers for PET imaging of myocardial perfusion in pigs. Nucl Med Biol. 2012;39:715–23. doi: 10.1016/j.nucmedbio.2011.11.007. [DOI] [PubMed] [Google Scholar]
- 326.Wunderlich G, Schiller E, Bergmann R, Pietzsch H-J. Comparison of the stability of Y-90-, Lu-177- and Ga-68- labeled human serum albumin microspheres (DOTA-HSAM) Nuclear Medicine and Biology. 2010;37:861–7. doi: 10.1016/j.nucmedbio.2010.05.004. [DOI] [PubMed] [Google Scholar]
- 327.Schiller E, Bergmann R, Pietzsch J, Noll B, Sterger A, Johannsen B. et al. Yttrium-86-labelled human serum albumin microspheres: relation of surface structure with in vivo stability. Nucl Med Biol. 2008;35:227–32. doi: 10.1016/j.nucmedbio.2007.10.008. [DOI] [PubMed] [Google Scholar]
- 328.Prinsen K, Li JJ, Vanbilloen H, Vermaelen P, Devos E, Mortelmans L. et al. Development and evaluation of a Ga-68 labeled pamoic acid derivative for in vivo visualization of necrosis using positron emission tomography. Bioorganic & Medicinal Chemistry. 2010;18:5274–81. doi: 10.1016/j.bmc.2010.05.050. [DOI] [PubMed] [Google Scholar]
- 329.Lankinen P, Maekinen TJ, Poeyhoenen TA, Virsu P, Salomaeki S, Hakanen AJ. et al. Ga-68-DOTAVAP-P1 PET imaging capable of demonstrating the phase of inflammation in healing bones and the progress of infection in osteomyelitic bones. European Journal of Nuclear Medicine and Molecular Imaging. 2008;35:352–64. doi: 10.1007/s00259-007-0637-5. [DOI] [PubMed] [Google Scholar]
- 330.Autio A, Ujula T, Luoto P, Salomaki S, Jalkanen S, Roivainen A. PET imaging of inflammation and adenocarcinoma xenografts using vascular adhesion protein 1 targeting peptide 68Ga-DOTAVAP-P1: comparison with 18F-FDG. Eur J Nucl Med Mol Imaging. 2010;37:1918–25. doi: 10.1007/s00259-010-1497-y. [DOI] [PubMed] [Google Scholar]
- 331.Silvola J, Autio A, Luoto P, Jalkanen S, Roivainen A. Preliminary evaluation of novel 68Ga-DOTAVAP-PEG-P2 peptide targeting vascular adhesion protein-1. Clin Physiol Funct Imaging. 2010;30:75–8. doi: 10.1111/j.1475-097X.2009.00907.x. [DOI] [PubMed] [Google Scholar]
- 332.Ujula T, Salomaki S, Virsu P, Lankinen P, Makinen TJ, Autio A. et al. Synthesis, Ga-68 labeling and preliminary evaluation of DOTA peptide binding vascular adhesion protein-1: a potential PET imaging agent for diagnosing osteomyelitis. Nuclear Medicine and Biology. 2009;36:631–41. doi: 10.1016/j.nucmedbio.2009.04.008. [DOI] [PubMed] [Google Scholar]
- 333.Autio A, Henttinen T, Sipila HJ, Jalkanen S, Roivainen A. Mini-PEG spacering of VAP-1-targeting 68Ga-DOTAVAP-P1 peptide improves PET imaging of inflammation. EJNMMI Res. 2011;1:10. doi: 10.1186/2191-219X-1-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 334.Aalto K, Autio A, Kiss EA, Elima K, Nymalm Y, Veres TZ. et al. Siglec-9 is a novel leukocyte ligand for vascular adhesion protein-1 and can be used in PET imaging of inflammation and cancer. Blood. 2011;118:3725–33. doi: 10.1182/blood-2010-09-311076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Petrik M, Haas H, Dobrozemsky G, Lass-Florl C, Helbok A, Blatzer M. et al. 68Ga-Siderophores for PET Imaging of Invasive Pulmonary Aspergillosis: Proof of Principle. J Nucl Med. 2010;51:639–45. doi: 10.2967/jnumed.109.072462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 336.Petrik M, Haas H, Schrettl M, Helbok A, Blatzer M, Decristoforo C. In vitro and in vivo evaluation of selected 68Ga-siderophores for infection imaging. Nucl Med Biol. 2012;39:361–9. doi: 10.1016/j.nucmedbio.2011.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Petrik M, Franssen GM, Haas H, Laverman P, Hortnagl C, Schrettl M. et al. Preclinical evaluation of two 68Ga-siderophores as potential radiopharmaceuticals for Aspergillus fumigatus infection imaging. Eur J Nucl Med Mol Imaging. 2012;39:1175–83. doi: 10.1007/s00259-012-2110-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 338.Nanni C, Errani C, Boriani L, Fantini L, Ambrosini V, Boschi S. et al. 68Ga-Citrate PET/CT for Evaluating Patients with Infections of the Bone: Preliminary Results. J Nucl Med. 2010;51:1932–6. doi: 10.2967/jnumed.110.080184. [DOI] [PubMed] [Google Scholar]