Abstract
The reduction of N2 to NH3 is a requisite transformation for life.1 While it is widely appreciated that the iron-rich cofactors of nitrogenase enzymes facilitate this transformation,2-5 how they do so remains poorly understood. A central element of debate has been the exact site(s) of nitrogen coordination and reduction.6,7 The synthetic inorganic community placed an early emphasis on Mo8, because Mo was thought to be an essential element of nitrogenases3 and because pioneering work by Chatt and coworkers established that well-defined Mo model complexes could mediate the stoichiometric conversion of N2 to NH3.9 This chemical transformation can be performed in a catalytic fashion by two well-defined molecular systems that feature Mo centres.10,11 However, it is now thought that Fe is the only transition metal essential to all nitrogenases,3 and recent biochemical and spectroscopic data has implicated Fe instead of Mo as the site of N2 binding in the FeMo-cofactor.12 In this work, we describe a tris(phosphine)borane-supported Fe complex that catalyzes the reduction of N2 to NH3 under mild conditions, wherein >40% of the H+/e- equivalents are delivered to N2. Our results indicate that a single Fe site may be capable of stabilizing the various NxHy intermediates generated en route to catalytic NH3 formation. Geometric tunability at Fe imparted by a flexible Fe-B interaction in our model system appears to be important for efficient catalysis.13-15 We propose that the interstitial light C-atom recently assigned in the nitrogenase cofactor may play a similar role,16,17 perhaps by enabling a single Fe site to mediate the enzymatic catalysis via a flexible Fe-C interaction.18
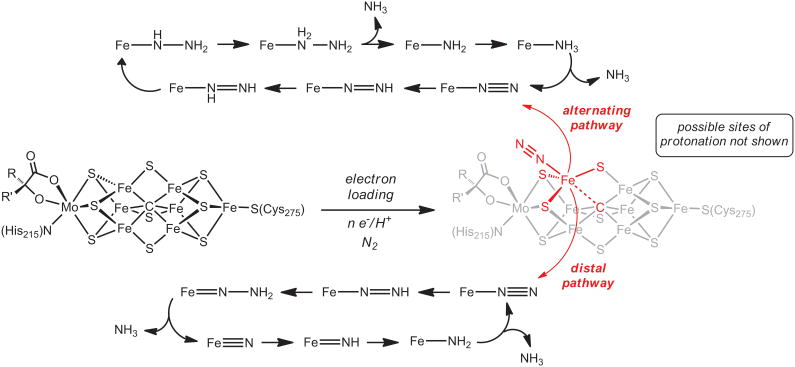
Nitrogen is fixed on a staggering scale by the industrial Haber-Bosch process using a solid-state Fe catalyst at very high temperatures and pressures, and in Nature by nitrogenase enzymes under ambient conditions.1 These enzymes feature active site cofactors rich in S and Fe (FeFecofactor), and can additionally contain a Mo (FeMo-cofactor; Figure 1) or V (FeV-cofactor) site.2,3
Fig. 1. Chemical line representations of the FeMo-cofactor of nitrogenase.
A schematic depiction of postulated N2 binding and reduction at an Fe site via limiting alternating (top) and distal (bottom) mechanisms. The drawing emphasizes a possible hemi-labile role for the interstitial C-atom with respect to an Fe-N2 binding site.
The intimate mechanism of biological nitrogen fixation is a topic that has fascinated chemists, biochemists, and biologists alike.4,5,6,7 Synthetic chemists have searched for decades for well-defined complexes that can catalyze N2 reduction to NH3.19,20,21,22 Chatt's early work with low-valent Mo complexes provided a mechanistic outline for approaching this problem now commonly called the “Chatt” or “distal” mechanism, wherein a terminal nitride intermediate is generated upon liberation of the first NH3 equivalent (Figure 1, bottom).9 Other mechanisms, including an “alternating” scenario (Figure 1, top), have also received attention.6 To date, even modest catalysis of NH3 production directly from N2 by a well-defined model complex is still limited to the original tri(amido)amine Mo systems of Schrock and coworkers, and the more recently discovered phosphine-pincer Mo system of Nishibayashi and coworkers.10,11 Earlier work by Pickett had established the electrochemical feasibility of an NH3 production cycle with W.19
Synthetic efforts to establish whether one or more Fe sites can catalyze N2 reduction to NH3 in a well-defined model complex have progressed more slowly. For example, previous Fe-N2 model complexes have not afforded more than ca. 10% of NH3 per Fe equivalent upon treatment with proton sources.7,23,24 Despite significant advances,24 which have most recently included reductive N2 cleavage at iron22 and the catalytic reductive silylation of N2 facilitated by unknown Fe species derived from Fe precursors such as ferrocene and iron pentacarbonyl,25 the delivery of protons and electrons to N2 to catalytically generate NH3 at a synthetic Fe complex has remained an unsolved challenge. Here we show that a recently reported mononuclear Fe complex, [(TPB)Fe(N2)][Na(12-crown-4)2] (TPB = tris(phosphine)borane; see Figure 2A),13,14 can catalyze the reduction of N2 to NH3 by protons and electrons in solution at -78 °C under one atmosphere of N2.
Fig. 2. Stoichiometric (TPB)Fe-N2model reactions.
(a) N2 binding to [(TPB)Fe][BArF4] under electron-loading to generate [(TPB)Fe(N2)][Na(12-crown-4)2]. (b) Reductive protonolysis of (TPB)Fe(NH2) to release NH3 with concomitant N2 uptake. (c) Generation of [(TPB)Fe(NH3)][BArF4] and other (TPB)Fe-species upon addition of acid to [(TPB)Fe(N2)][Na(12-crown-4)2] at low temperature, followed by warming and then addition of base. 12-C-4 is an abbreviation for 12-crown-4. Note: TPB = tris(phosphine)borane.
The Fe center of the “(TPB)Fe” fragment readily binds dinitrogen as evidenced by the featured 5-coordinate complex [(TPB)Fe(N2)][Na(12-crown-4)2] and the previously reported neutral N2 adduct, (TPB)Fe(N2).14 The same scaffold also accommodates a variety of other nitrogenous ligands relevant to NH3 generation, including terminally bonded NH2, NH3, and N2H4 ligands.15 Both the substrate-free complex, [(TPB)Fe][BArF4] (where [BArF4]- represents the weakly coordinating anion [B(3,5-(CF3)2-C6H3)4]-),15 and (TPB)Fe(N2) may be reduced to [(TPB)Fe(N2)][Na(12-crown-4)2] by Na/Hg reduction under 1 atm N2 followed by the addition of two equivalents of 12-crown-4 to encapsulate the sodium cation (Figure 2A). Model reactions with silyl electrophiles have also shown that the β-N of the coordinated N2 ligand can be mono- or difunctionalized with concomitant lengthening of the Fe-B distance.13 Furthermore, starting from (TPB)Fe(NH2), a reductive protonation sequence has been established (Figure 2B) that liberates NH3 and affords (TPB)Fe(N2).15 This reaction sequence demonstrates the synthetic viability of reductive release of NH3 from an Fe-NH2 intermediate with simultaneous uptake of N2. In sum, the rich reaction chemistry of the (TPB)Fe scaffold with nitrogenous ligands motivated us to more closely examine the possibility that it might catalyze N2 fixation.
The addition of excess acid to [(TPB)Fe(N2)][Na(12-crown-4)2] at -78 °C was investigated. When [(TPB)Fe(N2)][Na(12-crown-4)2] was dissolved in THF, cooled to -78 °C, and exposed to six equivalents of H+ in the form of HBArF4·2 Et2O, a previously unobserved yellow solution resulted that, upon warming followed by addition of proton sponge (1,8-bis(dimethylamino)naphthalene), was shown by 1H NMR analysis to contain the previously characterized complex [(TPB)Fe(NH3)][BArF4] (ca. 30-35% of the total Fe),15 along with resonances consistent with [(TPB)Fe][BArF4] (ca. 40-45% of the total Fe) and two other minor and as yet unidentified paramagnetic (TPB)Fe-species (see SI). An independent EPR study of this low temperature protonation reaction in 2-methyltetrahydrofuran revealed a new rhombic S = ½ signal (see SI) that we speculate may be (TPB)Fe(=N-NH2)+ or an alternative structural isomer such as (TPB)Fe(NH=NH)+. Spin quantification of this species shows it to represent >85% of the Fe species in solution, and its rhombic EPR spectrum is highly similar to the rhombic EPR signature that is obtained upon oxidation of (TPB)Fe=N(p-tolyl) to generate (TPB)Fe=N(p-tolyl)+ (SI). Subsequent low temperature reduction of a similarly prepared mixture regenerates [(TPB)FeN2][Na(12-crown-4)2], as determined by IR spectroscopy, suggesting the possibility of cycling protonation and reduction with this Fe system.
To explore the possibility of N2 reduction catalysis using this (TPB)Fe system, we canvassed several reductants (e.g., Na[naphthalenide], Mg(THF)3(anthracene), Na/Hg, KC8) and acids (e.g., HCl, trifluoromethanesulfonic acid, HBArF4·2 Et2O) in a variety of solvents and solvent mixtures (e.g., tetrahydrofuran, dimethoxyethane, diethyl ether, toluene). When carried out at -78 °C numerous reaction conditions showed unusually high yields of NH3 relative to the number of Fe equivalents in the reaction vessel, and the combination of KC8, HBArF4·2 Et2O, and Et2O solvent enabled the catalytic generation of NH3.
In a representative catalytic run, red [(TPB)Fe(N2)][Na(12-crown-4)2] was suspended in diethyl ether in a reaction vessel at -78 °C, followed by the sequential addition of excess acid and then excess reductant. Ammonia analysis followed the indophenol protocol (see Methods and SI) and the independent identification of ammonium salts by 1H NMR spectroscopy in DMSO by comparison with an authentic sample of [NH4][Cl] (Figure 3A). An experiment performed using the [(TPB)Fe(14N2)][Na(12-crown-4)2] catalyst under an 15N2 atmosphere, followed by 1H NMR analysis (Figure 3A) of the volatile reaction products, confirmed the production of [15NH4][Cl], as expected, with only trace [14NH4][Cl]. This overall procedure has been repeated many times, and Table 1 includes data averaged from 16 independent runs (entry 1) wherein yields were, on average, 7.0 equiv NH3 per Fe equiv. Using 7.0 equiv NH3 as the product stoichiometry, 44% of the added protons are reliably delivered to N2 to produce NH3. Individual runs have in our hands reached a maximum of 8.5 equiv NH3 per Fe equiv under these standard conditions. [(TPB)Fe][BArF4] is also an effective catalyst and afforded 6.2 ± 0.7 equiv NH3 per added Fe equivalent (Table 1, entry 2). For comparison, the Mo systems of Schrock and Nishibayashi have afforded between 7-12 equiv NH3 per Mo equiv.10,11 The current Fe system appears to be active at an unusually low temperature (-78 °C) but benefits from a strong reductant (KC8). We do not yet know if conditions can be found that will tolerate a milder reductant, for example by circumventing the need to generate the (TPB)Fe(N2)- anion during catalysis.
Fig. 3. Spectral data for ammonia analysis, and catalyst poisoning.
(a) 1H NMR spectrum (300 MHz) of [14NH4][Cl] in DMSO-d6 produced from a catalytic run under 14N2 (top) and of [15NH4][Cl] in DMSO-d6 produced from an independent catalytic run under 1 atm 15N2. (b) Representative optical data for NH3 product analysis using the indophenol method from catalytic runs using the standard conditions with the precursors indicated. (c) H2 binds to (TPB)Fe(N2) to generate (TPB)(μ-H)Fe(N2)(H), which itself is ineffective for the catalytic generation of NH3 from N2 under the standard conditions. Note: TPB = tris(phosphine)borane; DMSO = dimethylsulfoxide.
Table 1. Catalytic ammonia generation from N2 mediated by Fe precursors.

| ||
|---|---|---|
|
| ||
| Using standard catalytic conditions as described in the text | ||
| Entry | Fe precursor | NH3 equiv/Fea,b,c |
| 1 | [(TPB)Fe(N2)][Na(12-crown-4)2] | 7.0 ± 1d |
| 2 | [(TPB)Fe][BArF4] | 6.2 |
| 3 | [(SiPiPr3)Fe(N2)][Na(12-crown-4)2] | 0.7 |
| 4 | (TPB)(μ-H)Fe(N2)(H) | 0.5 |
| 5 | FeCl2·1.5 THF | <0.1 |
| 6 | FeCl3 | <0.1 |
| 7 | Cp2Fe | <0.2 |
| 8 | Fe(CO)5 | <0.1 |
| 9 | none | <0.1 |
|
| ||
| Variations on standard conditions using [(TPB)FeN2][Na(12-crown-4)2] | ||
| Entry | Variation | NH3 equiv/Fea,b,c |
|
| ||
| 10 | HOTf as acide | 0.4 |
| 11 | [Lutidinium][BArF4] as acid | <0.1 |
| 12 | HCl as acid | <0.1 |
| 13 | Cp*2Co as reductant | 0.6 |
| 14 | Cp*2Cr as reductant | <0.2 |
| 15 | K metal as reductant | 0.4 |
NH3 was collected by vacuum transfer of the reaction volatiles into HCl in diethyl ether. A dimethoxyethane solution of [Na][OtBu] (20 equiv relative to Fe) was added to the reaction vessel residue, followed by an additional vacuum transfer, to ensure complete liberation of all NH3. The [NH4][Cl] precipitate formed in the acidic Et2O collection vessel was reconstituted in deionized H2O, from which an aliquot was taken for indophenol quantification. Analysis of the [NH4][Cl] formed by 1H NMR spectroscopy in DMSO, compared with an authentic sample, provided independent confirmation of NH3 generation.
Data for individual experimental runs, and additional runs using potential precatalysts that are not presented in this table, are provided in the SI.
Unless noted otherwise, all yields are reported as an average of 4 runs.
Average of 16 runs.
HOTf = trifluoromethanesulfonic acid.
Table 1 lists several sets of conditions (entries 10-15) other than the standard conditions described above that were canvassed. Several of these alternative conditions showed NH3 generation, though not in catalytic or even in high yields. N2H4 was not detected (SI) as an additional product when using the standard catalytic protocol for NH3 generation with [(TPB)Fe(N2)][Na(12-crown-4)2] (SI). If two equivalents N2H4 (per Fe) are added to [(TPB)Fe(N2)][Na(12-crown-4)2] in diethyl ether, followed by subjecting the mixture to the standard catalytic conditions and work-up, only trace N2H4 remains (SI). This result suggests that if N2H4 is generated as an intermediate during catalysis it would not likely be detectable upon work-up and analysis. Worth noting is that HBArF4·2 Et2O and KC8 reacts in the absence of an Fe precursor, under the standard catalytic conditions at -78 °C, to generate H2 but not NH3 (>75% yield of H2 after 40 minutes). That H2 generation is kinetically feasible without the addition of an Fe precursor, and yet NH3 can nonetheless be generated upon the addition of [(TPB)Fe(N2)][Na(12-crown-4)2] or [(TPB)Fe][BArF4], underscores the facility by which this Fe system mediates overall H-atom delivery to N2.
To further explore whether a (TPB)Fe containing precursor is needed to facilitate the overall catalysis, beyond the stoichiometric model reactions summarized above, we canvassed several Fe complexes under analogous conditions. Of most interest is the complex [(SiPiPr3)Fe(N2)][Na(12-crown-4)2], which is isostructural to [(TPB)Fe(N2)][Na(12-crown-4)2] but replaces the B atom of TPB by a Si atom.26 A central difference between (TPB)Fe and (SiPiPr3)Fe complexes is the far great flexibility of the Fe-B versus the Fe-Si bond that is positioned trans to the apical ligand.13,14,15,26 While some NH3 generation was observed for [(SiPiPr3)Fe(N2)][Na(12-crown-4)2] when subjected to the standard catalytic reaction conditions described above, sub-stoichiometric yields of NH3 relative to Fe were obtained (0.7 ± 0.5 equiv NH3 per Fe equiv; entry 3). We also conducted additional control experiments under the standard catalytic conditions with FeCl2·1.5 THF, FeCl3, Cp2Fe,25 and Fe(CO)525 (entries 5-8) and found that only trace amounts of NH3 (<0.2 equiv in all cases on average; 4 runs) were produced by these Fe precursors (SI).27 The known phosphine-supported Fe(0)-N2 complex Fe(depe)2(N2)28 was also subjected to the standard conditions and afforded sub-stoichiometric yields of NH3 per Fe equivalent (see SI).
In separate work, the addition of an atmosphere of H2 to (TPB)Fe(N2) was shown to generate (TPB)(μ-H)Fe(N2)(H) as a stable product (Figure 3C).29 We hence suspected that catalyst poisoning might occur in part via the formation of (TPB)(μ-H)Fe(N2)(H) under the catalytic reaction conditions. In accord with this idea, when [(TPB)Fe(N2)][Na(12-crown-4)2] was exposed to 10 equiv HBArF4·2 Et2O and 12 equiv KC8 at low temperature, IR and 31P NMR analysis of the resulting mixture showed the presence of (TPB)(μ-H)Fe(N2)(H) via its signature spectroscopic features (30% of total Fe by 31P NMR integration; SI).29 (TPB)(μ-H)Fe(N2)(H) is stable for short periods to both HBArF4·2 Et2O and also KC8 in Et2O at room temperature, and when subjected to the standard catalytic conditions for NH3 production liberates only 0.5 ± 0.1 equiv NH3 per Fe equiv (Table 1 entry 4).
The general absence of a functional, catalytic Fe model system over the past few decades has often led to an emphasis on Mo30 as a plausible site of N2 uptake and reduction at the most widely studied FeMo-cofactor. While this may yet prove to be true, recent spectroscopic and biochemical evidence has sharpened the focus on an Fe center as the N2 binding site.12 The results reported here establish that it is possible to catalyze the conversion of N2 to NH3 by protons and electrons using a well-defined mononuclear Fe-N2 complex, and suggests the possibility that a single Fe-binding site of the cofactor could in principle mediate N2 reduction catalysis.18 To achieve this catalytic behavior, geometric flexibility at the Fe-N2 binding site would be beneficial as it would stabilize NxHy intermediates of different electronic demands. Such geometric and redox flexibility, under the local three-fold symmetry presented by an Fe center, its three neighboring sulfides, and the interstitial light atom of the FeMo-co,16,17 may at least in part be achieved by attributing a hemi-labile role to the interstitial C-atom (Figure 1). Such a role could serve to expose an initial Fe-N2 binding site by Fe-C elongation. Subsequent modulation of the Fe-C interaction and hence the local Fe geometry as a function of the N2 reduction state would enable the Fe center to stabilize the various NxHy intermediates along a pathway to NH3. While inherently speculative, this hypothesis is rooted in the functional (TPB)Fe catalysis discussed herein, along with the types of (TPB)Fe complexes and stoichiometric transformations described previously for this scaffold.13,14,15
The mechanistic questions that arise from the present Fe catalyst system are rich. While all of the model complexes relevant to the (TPB)Fe-NxHy system are mononuclear, we cannot exclude the possibility of bimolecular reaction intermediates. We more generally don't know whether the N2 reduction catalysis proceeds along a distal pathway via a terminal nitride intermediate, such as (TPB)Fe(N) or (TPB)Fe(N)+, via intermediates along an alternating pathway, such as (TPB)Fe-NH-NH2 or (TPB)Fe-NH2-NH2+, or via some hybrid pathway. The tentatively assigned (TPB)Fe=N-NH2+ species that can be observed by EPR spectroscopy provides us a useful starting point for such studies. In light of the identification of C as the interstitial light atom of the cofactor, it is also of interest to develop and compare synthetic model systems that feature a C-atom in the ligand backbone instead of a B-atom.
Methods Summary
General considerations
All complexes and reagents were prepared according to literature procedures referenced in the Supplemental Information (SI) unless otherwise noted. Manipulations were carried out under an N2 atmosphere utilizing standard dry glove-box or Schlenk-line techniques. All solvents used were deoxygenated and dried by an argon sparge followed by passage through an activated alumina column.
Spectroscopy
NMR measurements were obtained on Varian 300 MHz spectrometers. Deuterated solvents for these measurements were obtained from Cambridge Isotope Laboratories and were dried and degassed prior to use. All 1H NMR spectra were referenced to residual solvent peaks. EPR X-band spectra were obtained on a Bruker EMX spectrometer with the aid of the Bruker Win-EPR software suite version 3.0. The EPR spectrometer was equipped with a rectangular cavity that operated in the TE102 mode. Temperature control was achieved with a liquid-N2-filled quartz-dewar in which the sample was submerged during data collection. UV-Vis spectra were acquired on a Cary 50 spectrometer from 1100 nm to 200 nm in the fast scan mode. IR spectra were obtained via KBr pellets on a Bio-Rad Excalibur FTS 3000 spectrometer using Varian Resolutions Pro software set at 4 cm-1 resolution.
Catalysis and ammonia collection and quantification
The standard catalysis protocol involved the addition first of acid, followed by reductant, to a suspension of the precatalyst in diethyl ether at -78 °C in a closed vessel under 1 atm N2. Ammonia produced during each catalytic run was collected by vacuum transfer of the reaction volatiles onto anhydrous HCl in diethyl ether. The resulting slurry was dried and extracted into water and aliquots were then tested for the presence of ammonia via the indophenol method.
A Methods section, associated references, and a detailed Supplemental Information are available in the online version of this paper.
Methods
General Considerations
[(TPB)Fe(N2)][Na(12-crown-4)2],14 [(TPB)Fe][BArF4],15 (TPB)(μ-H)Fe(H)(N2),29 [Lutidinium][BArF4],31 HBArF4 · 2 Et2O,32 [(SiPiPr3)Fe(N2)][Na(12-crown-4)2],26 FeCl2·(THF)1.5,33 KC8,34 [(TPB)Fe(NH3)][BArF4],15 [(TPB)Fe(N2H4)][BArF4],15 and Fe(depe)2N228 were prepared according to literature procedures. Note: [Lutidinium] = 2,6-dimethylpyridinium; [BArF4] = [B(3,5-(CF3)2-C6H3)4]-. Labeled 15N2 (98% purity) was obtained from Cambridge Isotope Laboratories. Solvents used for catalytic runs were additionally stirred for more than 2 hours over Na/K alloy and then filtered prior to use, in addition to standard sparging (Ar gas) and passage through an activated alumina column.
Ammonia Quantification
A Schlenk tube was charged with HCl (3 mL of a 2.0 M solution in Et2O, 6 mmol). Reaction mixtures were vacuum transferred into this collection flask. Residual solid in the reaction vessel was treated with a solution of [Na][O-t-Bu] (40 mg, 0.4 mmol) in 1,2-dimethoxyethane (1 mL) and sealed. The resulting suspension was allowed to stir for 10 minutes before all volatiles were again vacuum transferred into the collection flask. After completion of the vacuum transfer, the flask was sealed and warmed to room temperature. Solvent was removed in vacuo and the remaining residue was dissolved in H2O (1 mL). An aliquot of this solution (20 or 40 μL) was then analyzed for the presence of NH3 (trapped as [NH4][Cl]) via the indophenol method.35 Quantification was performed with UV-Vis spectroscopy by analyzing absorbance at 635 nm. The tables shown in the SI indicate the raw data for the runs. Runs with small absorbance levels (< 0.02 absorbance units) suffer from a large degree of error due to a small signal-to-noise ratio. Catalytic runs that used a 40 μL aliquot are denoted with an asterisk, accounting for larger relative absorbances.
Standard Catalytic Protocol
[(TPB)Fe(N2)][Na(12-crown-4)2] (2 mg, 0.002 mmol) was suspended in Et2O (0.5 mL) in a 20 mL scintillation vial equipped with a stir bar. This suspension was vigorously stirred and cooled to -78 °C in a cold well inside of the glove box. A similarly cooled solution of HBArF4 · 2 Et2O (93 mg, 0.092 mmol) in Et2O (1.5 mL) was added to the suspension in one portion with rapid stirring. Any remaining acid was dissolved in cold Et2O (0.25 mL) and added subsequently. The reaction mixture turned light yellow-orange and homogeneous upon addition of acid and the resulting solution was allowed to stir for 5 minutes before being transferred into a pre-cooled Schlenk tube equipped with a stirbar. The original reaction vial was washed with cold Et2O (0.25 mL) and was subsequently transferred to the Schlenk tube. Solid KC8 (15 mg, 0.100 mmol) was suspended in cold Et2O (0.75 mL) and added dropwise to the rapidly stirred solution in the Schlenk tube and was then tightly sealed. The reaction was allowed to stir for 40 minutes at -78 °C before being warmed to room temperature and stirred for 15 minutes.
Detailed experimental procedures and data for all experiments can be found in the Supplemental Information.
Supplementary Material
Acknowledgments
This work was supported by the NIH (GM 070757) and the Gordon and Betty Moore Foundation. Dr. Ayumi Takaoka is thanked for developing the calibration curves used for ammonia and hydrazine quantification. Professors Douglas Rees and Dianne Newman are acknowledged for many insightful discussions.
Footnotes
Supplementary Information is linked to the online version of the paper at www.nature.com/nature.
Author Contributions JSA, JR, and JCP designed the study. JSA and JR conducted the experiments. JSA, JR, and JCP interpreted the data. JSA, JR, and JCP wrote the manuscript.
Author information Reprints and permissions information is available at www.nature.com/reprints. Correspondence and requests for materials should be addressed to JCP (jpeters@caltech.edu).
References
- 1.Smil V. Enriching the Earth. Cambridge, MA: MIT Press; 2001. [Google Scholar]
- 2.Burgess BK, Lowe DJ. Mechanism of molybdenum nitrogenase. Chem Rev. 1996;96:2983–3011. doi: 10.1021/cr950055x. [DOI] [PubMed] [Google Scholar]
- 3.Eady RR. Structure-function relationships of alternative nitrogenases. Chem Rev. 1996;96:3013–3030. doi: 10.1021/cr950057h. [DOI] [PubMed] [Google Scholar]
- 4.Howard JB, Rees DC. How many metals does it take to fix N2? A mechanistic overview of biological nitrogen fixation. Proc Natl Acad Sci U S A. 2006;103:17088–17093. doi: 10.1073/pnas.0603978103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schwarz G, Mendel RR, Ribbe MW. Molybdenum cofactors, enzymes and pathways. Nature. 2009;460:839–847. doi: 10.1038/nature08302. [DOI] [PubMed] [Google Scholar]
- 6.Seefeldt L, Hoffman BM, Dean DR. Mechanism of Mo-dependent nitrogenase. Annu Rev Biochem. 2009;78:701–722. doi: 10.1146/annurev.biochem.78.070907.103812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Peters JC, Mehn MP. In: Bio-organometallic Approaches to Nitrogen Fixation Chemistry Activation of Small Molecules. Tolman WB, editor. Wiley-VCH; 2006. pp. 81–119. [Google Scholar]
- 8.Shilov AE, et al. New nitrogenase model for reduction of molecular nitrogen in protonic media. Nature. 1971;231:460–461. doi: 10.1038/231460a0. [DOI] [PubMed] [Google Scholar]
- 9.Chatt J, Dilworth JR, Richards RL. Recent advances in chemistry of nitrogen-fixation. Chem Rev. 1978;78:589–625. [Google Scholar]
- 10.Yandulov DV, Schrock RR. Catalytic reduction of dinitrogen to ammonia at a single molybdenum center. Science. 2003;301:76–78. doi: 10.1126/science.1085326. [DOI] [PubMed] [Google Scholar]
- 11.Arashiba A, Miyake Y, Nishibayashi Y. A molybdenum complex bearing PNP-type pincer ligands leads to the catalytic reduction of dinitrogen into ammonia. Nature Chem. 2011;3:120–125. doi: 10.1038/nchem.906. [DOI] [PubMed] [Google Scholar]
- 12.Hoffman BM, Dean DR, Seefeldt LC. Climbing nitrogenase: toward a mechanism of enzymatic nitrogen fixation. Acc Chem Res. 2009;42:609–619. doi: 10.1021/ar8002128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Moret ME, Peters JC. N2 functionalization at iron metallaboratranes. J Am Chem Soc. 2011;133:18118–18121. doi: 10.1021/ja208675p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Moret ME, Peters JC. Terminal iron dinitrogen and iron imide complexes supported by a tris(phosphino)borane ligand. Angew Chem Int Ed. 2011;50:2063–2067. doi: 10.1002/anie.201006918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Anderson JA, Moret ME, Peters JC. Conversion of Fe–NH2 to Fe–N2 with release of NH3. J Am Chem Soc. 2013;135:534–537. doi: 10.1021/ja307714m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lancaster KM, et al. X-ray emission spectroscopy evidences a central carbon in the nitrogenase iron-molybdenum cofactor. Science. 2011;334:974–977. doi: 10.1126/science.1206445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Spatzal TM, et al. Evidence for interstitial carbon in nitrogenase FeMo cofactor. Science. 2011;334:940–940. doi: 10.1126/science.1214025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hendrich MP, et al. On the feasibility of N2fixation via a single-site FeI/FeIVcycle: Spectroscopic studies of FeI(N2)FeI, FeIV≡N, and related species. Proc Natl Acad Sci U S A. 2006;103:17107–17112. doi: 10.1073/pnas.0604402103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pickett CJ, Talarmin J. Electrosynthesis of ammonia. Nature. 1985;317:652–653. [Google Scholar]
- 20.Fryzuk MD, Love JB, Rettig SJ, Young VG. Transformation of coordinated dinitrogen by reaction with dihydrogen and primary silanes. Science. 1997;275:1445–1447. [Google Scholar]
- 21.Pool JA, Lobkovsky E, Chirik PJ. Hydrogenation and cleavage of dinitrogen to ammonia with a zirconium complex. Nature. 2004;427:527–530. doi: 10.1038/nature02274. [DOI] [PubMed] [Google Scholar]
- 22.Rodriguez MM, Bill E, Brennessel WW, Holland PL. N2 reduction and hydrogenation to ammonia by a molecular iron-potassium complex. Science. 2011;334:780–783. doi: 10.1126/science.1211906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hills A, et al. Bis[1,2-bis(dimethylphosphino)ethane]dihydrogenhydridoiron(II) tetraphenylborate as a model for the function of nitrogenases. J Chem Soc Dalton Trans. 1993;25:3041–3049. [Google Scholar]
- 24.Crossland JL, Tyler DR. Iron-dinitrogen coordination chemistry: Dinitrogen activation and reactivity. Coord Chem Rev. 2011;255:949–974. [Google Scholar]
- 25.Yuki M, Tanaka H, Miyake Y, Yoshizawa K, Nishibayashi Y. Iron-catalysed transformation of molecular dinitrogen into silylamine under ambient conditions. Nature Comm. 2012;3:1254. doi: 10.1038/ncomms2264. [DOI] [PubMed] [Google Scholar]
- 26.Lee YH, Mankad NP, Peters JC. Triggering N2 uptake via redox-induced expulsion of coordinated NH3 and N2 silylation at trigonal bipyramidal iron. Nature Chem. 2010;2:558–565. doi: 10.1038/nchem.660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bell LG, Brintzinger HH. Stoichiometric reduction of molecular nitrogen by iron complexes. J Am Chem Soc. 1970;92:4464–4465. [Google Scholar]
- 28.Hall DA, Leigh GJ. J Chem Soc Dalton. 1996:3539–3541. [Google Scholar]
- 29.Fong H, Moret ME, Lee YH, Peters JC. Heterolytic H2 cleavage and catalytic hydrogenation by an iron metallaboratrane. Organometallics. 2013;32:3053–3062. doi: 10.1021/om400281v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schrock RR. Nitrogen reduction: molybdenum does it again. Nature Chem. 2011;3:95–96. doi: 10.1038/nchem.977. [DOI] [PubMed] [Google Scholar]
- 31.Yandulov DV, Schrock RR. Reduction of dinitrogen to ammonia at a well-protected reaction site in a molybdenum triamidoamine complex. J Am Chem Soc. 2002;124:6252–6253. doi: 10.1021/ja020186x. [DOI] [PubMed] [Google Scholar]
- 32.Brookhart M, Grant B, Volpe AF., Jr [(3,5-(CF3)2C6H3)4B]-[H(OEt2)2]+: a convenient reagent for generation and stabilization of cationic, highly electrophilic organometallic complexes. Organometallics. 1992;11:3920–3922. [Google Scholar]
- 33.Kern RJ. Tetrahydrofuran complexes of transition metal chlorides. J Inorg Nucl Chem. 1962;24:1105–1109. [Google Scholar]
- 34.Wietz IS, Rabinovitz M. The application of C8K for organic synthesis: reduction of substituted naphthalenes. J Chem Soc Perkin Trans. 1993;1:117–120. [Google Scholar]
- 35.Weatherburn MW. Phenol-hypochlorite reaction for determination of ammonia. Anal Chem. 1967;39:971. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.