Abstract
Bengkoang (Pachyrhizus erosus (L.) Urban) is one of the most popular edible root vegetables in Indonesia. Bengkoang contains fairly large amounts of carbohydrates and crude fiber. The purpose of this research is to evaluate the immunomodulatory effect of the bengkoang fiber extract (BFE) in vitro and in vivo. BFE was prepared by heating the powder of bengkoang fiber suspended in distilled water at 121 °C for 20 min. BFE facilitated IgM production by the human hybridoma cell line HB4C5 cells. In addition, production of IgM, IgG, and IgA by mouse primary splenocytes was facilitated by BFE in a dose-dependent manner. BFE also significantly facilitated production of both interleukin-5 and interleukin-10 by splenocytes. Immunoglobulin production by lymphocytes from the spleen, Peyer’s patch, and mesenteric lymph node were significantly activated by oral administration of BFE to mice for 14 days. The serum immunoglobulin levels of IgG, IgM, and IgA were also significantly enhanced. Furthermore, cytokine production by lymphocytes from the spleen, Peyer’s patch, and mesenteric lymph node were also facilitated by oral administration of BFE. These results suggest that BFE has positive effects on the immune system in vitro and in vivo.
Keywords: Bengkoang, Immunoglobulin, Cytokine, Immunomodulatory activity, Lymphocyte
Introduction
It is well known that the immune function is vitally important for prevention and recovery from infectious diseases. There are many evidences about modulation and activation of the immune function and immune response by food intake. Many foodstuffs that can activate the immune system have been screened out (Miyazaki et al. 2005; Cherng et al. 2007; Lin et al. 2005; Chen et al. 2012, Thakur et al. 2012). Activation of the immune response contributes to reduction of the risks of chronic diseases. The immune response can be divided into two categories such as the innate immune response and adaptive immune response. Although the adaptive immune response takes a long time after infection, it provides highly specific protection against pathogens. Lymphocytes play an essential role in the adaptive immune response. B cells give an important contribution to the humoral immune response by production of immunoglobulins (Igs). On the other hand, T cells also play an important role in the cell-mediated immunity. We have focused on the foodstuffs that activate the immune system for development of functional foods (Sugahara et al. 2005; Nishi et al. 2011, 2012; Morishige et al. 2011; Daifuku et al. 2012).
Bengkoang (Pachyrhizus erosus (L.) Urban) is an underutilized crop of family Leguminosae, subfamily Papilionoidea. The underground starchy root of bengkoang is one of the most popular edible root vegetables that grow in many areas of tropical and sub-tropical regions, especially in Indonesia. Bengkoang tuber is brown-skinned, white-fleshed, crispy, and juicy with an irregularly globular shape. Both cooked and uncooked bengkoang can be eaten in many kinds of dishes. The edible part of bengkoang consists of 82.0 % of water, 14.9 % of carbohydrates, 1.2 % of proteins, 0.1 % of lipids, and 1.4 % of crude fiber (Noman et al. 2007). The tuber also contains a large amount of ascorbic acid. Sweet flavour of bengkoang comes from the oligofructose inulin. In addition, flavonoids, thiamine, riboflavin, pyridoxine, adenine, choline, saponine, niacin, phytoestrogen, and folic acid are also detected (Noman et al. 2007; Nurrochmad et al. 2010). Non-digestible carbohydrates have a beneficial impact on modulation of the immune system by their fermented products that associate with the gut-associated lymphoid tissue (GALT). Dietary fiber has resistant properties in the small intestine of mammals and is fermentable in the large intestine partially or completely. Dietary fiber components are usually grouped into two major types depending on their solubility in the intestine; soluble fiber such as pectins, mucilage, loosely bound hemicelluloses, β-glucans, and non-digestible oligosaccharides including inulin, and insoluble fiber such as cellulose, lignin, and tightly bound celluloses (Rodríguez et al. 2006; Álvarez and Peña-Valdivia 2009). Dietary fiber has beneficial effects on the gastrointestinal track, and their digested products are associated with GALT that can modulate various properties of the immune system. GALT is composed of aggregated tissues in the form of Peyer’s patches (PPs), solitary lymphoid follicles, non-aggregated cells in the lamina propia, and intraepithelial regions of the intestine as well as mesenteric lymph nodes (MLNs) (Langkamp-Henken et al. 1992). Bengkoang has a larger amount of crude fiber than potato or sweet potato. However, the potency of crude fiber from bengkoang in our body has yet to be reported. An evidence of the immunomodulatory activity of bengkoang fiber is important and valuable for improving the bengkoang fiber as functional foods with the potency to modulate the immune system. In this study, we focused on the immunomodulatory effect of the bengkoang fiber extract (BFE) in vitro and in vivo.
Materials and methods
Preparation of BFE
Bengkoang tubers were peeled, grated, and suspended in distilled water. The suspension was settled down overnight to separate fiber from starch. The supernatant was collected as the bengkoang crude fiber. The crude fiber was steamed for 30 min, soaked in 80 % ethanol at 60 °C for 20 min, filtrated, and squeezed to collect the concentrate. The fiber concentrate was oven-dried and ground into powder. A BFE solution was prepared by suspending the bengkoang fiber powder in distilled water at 10 g/100 mL. The suspension was heated at 121 °C for 20 min or left at 25 °C for 2 h. Both suspensions were centrifuged at 15,600×g for 20 min to remove insoluble materials. Each supernatant was dialyzed against distilled water using a dialysis membrane with molecular weight cut off of 14 kDa (Wako Pure Chemical Industries, Osaka, Japan) and sterilized by filtration.
Experimental animals
Five-week-old female BALB/c mice were purchased from Japan SLC (Shizuoka, Japan). The mice were kept in a specific pathogen-free facility and acclimated to their housing environment for 1 week prior to experiment. They were given free access to standard laboratory rodent chow (Rodent LabDiet EQ 5L37; Nutrition International, Brentwood, MO, USA) and water. Animal room was maintained under controlled conditions of temperature at 25 ± 1 °C, humidity at 55 ± 5 %, and 12-h light/12-h dark cycle. All animal experiments described herein were carried out in accordance with the protocol approved by the Laboratory Animal Care Committee of Ehime University. Mice were maintained in accordance with the Guidelines for the Care and Use of Laboratory Animals of Ehime University.
Immunomodulatory effect of BFE on HB4C5 cells
The Ig production-stimulating activity of BFE was examined using the human–human hybridoma HB4C5 cells that were a fusion product of a human B lymphocyte from a lung cancer patient with a human fusion partner NAT-30 (Murakami et al. 1985). HB4C5 cells were cultured in ERDF medium (Kyokuto Pharmaceutical, Tokyo, Japan) containing 10 μg/mL of insulin (Sigma, St. Louis, MO, USA), 20 μg/mL of transferrin (Sigma), 20 μM ethanolamine (Sigma), and 25 nM sodium selenite (Sigma) (ITES-ERDF medium) at 37 °C under humidified 5 % CO2–95 % air (Murakami et al. 1982). HB4C5 cells were inoculated at 5.0 × 104 cells/mL in ITES-ERDF medium containing various concentrations of BFE into a 96-well culture plate. The amount of IgM in culture medium 6 h after inoculation was measured by an in-house-developed enzyme-linked immunosorbent assay (ELISA) as described previously (Sugahara et al. 1994). The assays were carried out in triplicate. The limit of detection (LOD) for human-IgM was 1 ng/mL.
Immunomodulatory effect of BFE on mouse splenocytes in vitro
Following 1 week acclimation, 6-week-old female BALB/c mice were sacrificed by cervical dislocation, and splenocytes were prepared as described previously (Kumalasari et al. 2012). Splenocytes were suspended in RPMI 1640 medium (Sigma) supplemented with 100 U/mL of penicillin (Sigma), 100 μg/mL of streptomycin (Sigma), and 5 % of fetal bovine serum (FBS; SAFC Biosciences, Lenexa, KS, USA). Splenocytes were then inoculated into a 48-well culture plate at 1.0 × 106 cells/mL in 5 % FBS-RPMI 1640 medium containing BFE at various concentrations and cultured in a CO2 incubator at 37 °C for 24 h. The amounts of IgM, IgA, and IgG secreted into culture medium were measured by ELISA as described previously (Nishimoto et al. 2009). The LOD of each assay was 0.39 ng/mL for mouse-IgM, 1.56 ng/mL for mouse-IgG, and 0.39 ng/mL for mouse-IgA. To examine the effect of BFE on cytokine production, splenocytes were inoculated into a 48-well culture plate at 2.0 × 106 cells/mL in 5 % FBS-RPMI 1640 medium containing 5 μg/mL of concanavalin A (Con A; Seikagaku, Tokyo, Japan) and cultured for 48 h. The amounts of interleukin (IL)-4, IL-5, IL-6, IL-10, tumor necrosis factor (TNF)-α, and interferon (IFN)-γ in culture medium were measured by commercially available ELISA kits (eBioscience, San Diego, CA, USA). The assays were performed in triplicate. The LOD of each assay was 4 pg/mL for IL-4, 4 pg/mL for IL-5, 4 pg/mL for IL-6, 15 pg/mL for IL-10, 0.75 pg/mL for TNF-α, and 15 pg/mL for IFN-γ.
Immunomodulatory effect of BFE in vivo
Following 1 week acclimation, 6-week-old female BALB/c mice were divided into 3 groups (6 mice per group). They were orally administered with BFE at 6.75 or 27 mg/kg body weight/day for consecutive 14 days or with distilled water for the control group. One day after the oral administration finished, the blood was collected and centrifuged at 18,000×g for 30 min at 4 °C to collect the serum (LeBlanc et al. 2004). The spleen, PP, and MLN were collected from each mouse. Lymphocytes from the PP and MLN were prepared with the same procedure as for the preparation of splenocytes and suspended in 5 % FBS-RPMI 1640 medium. For determination of the Ig production-stimulating activity, lymphocytes were inoculated into 48-well culture plates at 1.0 × 106 cells/mL and cultured for 24 h. For determination of the cytokine production-modulating activity, lymphocytes were inoculated into 48-well culture plates at 2.0 × 106 cells/mL in 5 % FBS-RPMI 1640 medium containing 5 μg/mL of Con A and cultured for 48 h. The amounts of IgA, IgG, and IgM secreted into culture medium were measured by ELISA as described previously (Nishimoto et al. 2009) and the amounts of IL-4, IL-6, IL-10, IFN-γ, TNF-α, and transforming growth factor (TGF)-β secreted into the culture medium were measured using ELISA kits (eBioscience). The assays were carried out in triplicate. The LOD of the TGF-β assay was 60 pg/mL. The cultured cells were also used for real-time RT-PCR as described below.
Real-time RT-PCR analysis
The cells used for evaluation of the Ig or cytokine production-modulating activity were also used for real-time RT-PCR analysis. Total RNA was isolated from the cells using Sepasol-RNA I Super G (Nacalai Tesque, Kyoto, Japan) according to the manufacturer’s instructions. Total RNA was used as a template for cDNA synthesis performed using MMLV-reverse transcriptase (Promega, Madison, WI, USA) and oligo-(dT)20 primer. The real-time PCR mixture, with a final volume of 20 μL, consisted of Fast SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA), 1 μM of a forward primer, 1 μM of a reverse primer, and 0.1 μg of a cDNA sample. The thermal cycling conditions were: 20 s at 95 °C, and 40 cycles of 3 s at 95 °C and 30 s at 60 °C. PCR products were measured with a StepOnePlus Real-time PCR System (Applied Biosystems), and the relative gene expression was calculated based on the comparative CT method using a StepOne Software v2.1 (Applied Biosystems). The expression of β-actin was used as an endogenous control. Specific oligonucleotide sequences for each gene are listed in Table 1.
Table 1.
Sequences of primers for real-time RT-PCR
| Primer | Sequence (5′–3′) |
|---|---|
| Mouse β-actin | 5′-CATCCGTAAAGACCTCTATGCCAAC-3′ (sense) 5′-ATGGAGCCACCGATCCACA-3′ (antisense) |
| Mouse IgM | 5′-GGGGCACTGGTCACATACTT-3′ (sense) 5′-CAGCTCGTGAGCAACTGAAC-3′ (antisense) |
| Mouse IgG1 | 5′-TATGGACTACTGGGGTCAAG-3′ (sense) 5′-CCTGGGCACAATTTTCTTGT-3′ (antisense) |
| Mouse IgA | 5′-TGCA CAGCTTTCTTCTGCAC-3′ (sence) 5′-TGCCAGCCTCACATGTACTC-3′ (antisense) |
Statistical analysis
Each result is expressed as the mean ± standard deviation (SD). One way ANOVA followed by Tukey’s test was used to assess the statistical significance of the difference. Values with *p < 0.05, **p < 0.01, or ***p < 0.001 were considered to be statistically significant.
Results and discussion
Effect of BFE on IgM production by HB4C5 cells
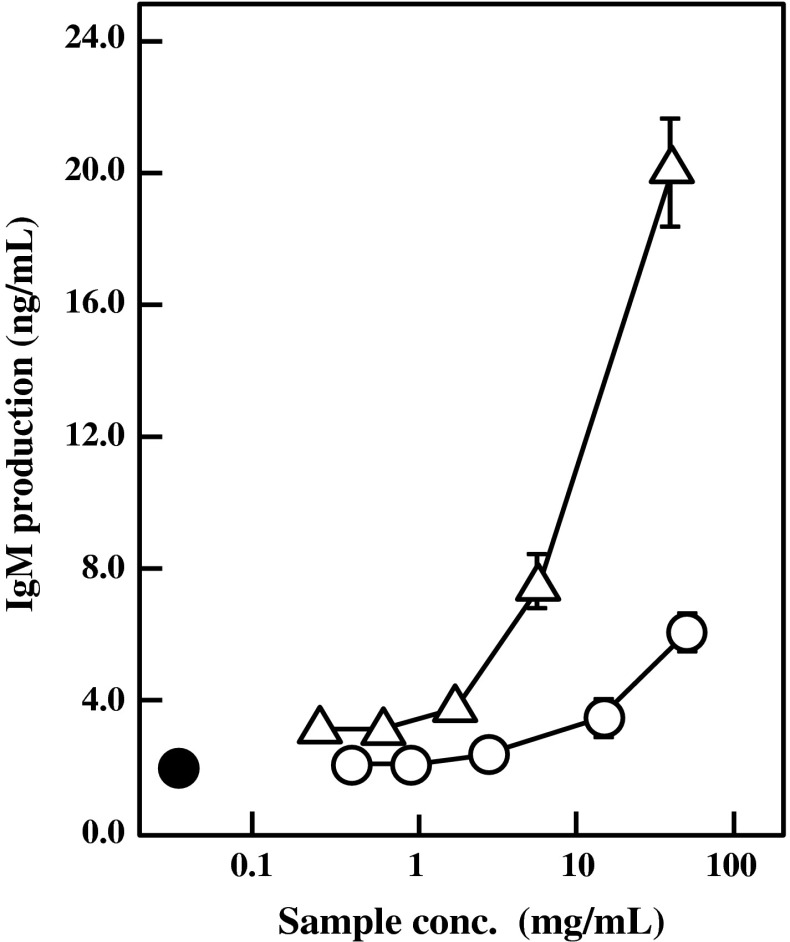
At first, two kinds of BFE were obtained from different extraction methods. One BFE was prepared by heat-treatment of the bengkoang fiber powder at 121 °C for 20 min in distilled water. The other BFE was prepared by stirring the powder at 25 °C for 2 h in distilled water. Both of the BFE were dialyzed against distillated water with a dialysis membrane that cuts off molecules under the molecular weight of 14 kDa. The IgM production-stimulatory activities of both BFE were compared to determine the suitable extraction method. The HB4C5 cells were inoculated in ITES-ERDF medium supplemented with various concentrations of either BFE and cultured for 6 h. As shown in Fig. 1, the IgM production by HB4C5 cells was enhanced by both BFE preparations in a dose-dependent manner. The specific activity of heat-extracted BFE was much higher than that of non-heated BFE, indicating that a heating process during extraction from the bengkoang fiber powder would effectively solubilize the active substance. This finding also suggests that the active substance in BFE is heat-stable. Because BFE was dialyzed using a dialysis membrane with molecular weight cut off of 14 kDa, the active substance seems to be macromolecules. In addition, the Ig production-stimulatory effect of the heat-extracted BFE was completely inactivated on HB4C5 cells by treating with either amylase or trypsin (data not shown). It is supposed from these findings that the active substance in BFE may be a complex of carbohydrates and proteins.
Fig. 1.
Effect of BFE on IgM production by HB4C5 cells. Bengkoang fiber powder suspended in distilled water was heat-extracted at 121 °C for 20 min (triangle) or extracted at 25 °C for 2 h without heating (open circle). Both BFEs were then dialyzed against distillated water. HB4C5 cells were inoculated in ITES-ERDF medium with various concentrations of BFE and cultured for 6 h. Distilled water was supplemented instead of BFE as control (closed circle). Results are represented as the mean ± SD of three independent measurements
Immunomodulatory effects of BFE on Ig and cytokine production by splenocytes in vitro
Igs are the major secretory products of the adaptive immune system and they are expressed and secreted by activated B lymphocytes. Next, the effects of BFE on the Ig and cytokine production by primary splenocytes from female BALB/c mice were examined in vitro. Splenocytes were inoculated in 5 % FBS-RPMI 1640 medium containing various concentrations of the heat-extracted BFE. The amounts of Igs and cytokines in culture medium were determined by ELISA 24 h and 48 h after inoculation, respectively. As shown in Fig. 2, the production levels of IgM, IgG, and IgA by splenocytes were significantly enhanced by the heat-extracted BFE in a dose-dependent manner. These facts suggested that BFE activates Ig production not only by a specific cell line like HB4C5 cells but also by primary Ig producers. The Ig production-stimulatory activity of the heat-extracted BFE was much higher than that of non-heated BFE (data not shown). Therefore, the heat-extracted BFE was used for further experiments.
Fig. 2.
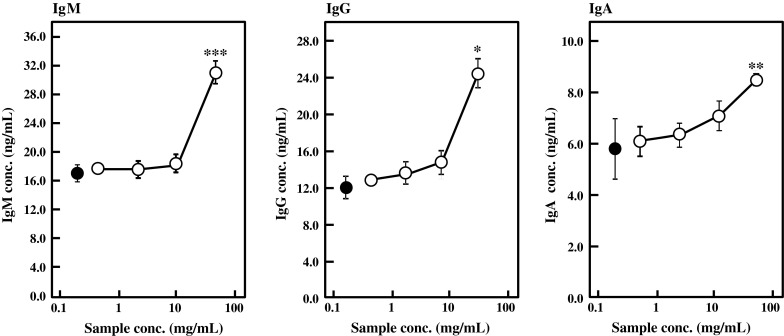
Effect of BFE on Ig production by mouse primary splenocytes in vitro. Splenocytes were inoculated in 5 % FBS-RPMI 1640 medium containing various concentrations of heat-extracted BFE and cultured for 24 h. The amounts of IgA, IgG, and IgM in culture medium were measured by ELISA. Control (closed circle) was supplemented with distilled water instead of BFE. Results are represented as the mean ± SD of three independent measurements. Significant differences against control are represented as *p < 0.05, **p < 0.01, or ***p < 0.001
The effects of BFE on cytokine production by primary splenocytes were also evaluated. As summarized in Fig. 3, BFE enhanced the cytokine production by splenocytes treated with 5 μg/mL of Con A. Significant differences were observed in the production levels of IL-5 and IL-10 against the control. Production of IL-4, IL-6, TNF-α, and IFN-γ was also stimulated by BFE. BFE activated Ig and cytokine production by B cells and T cells, respectively. T cells produce cytokines that induce growth and differentiation of B cells, giving rise to the humoral immune response. It is supposed from these results that BFE may stimulate both T-helper 1 and 2 cells. T-helper 1 cells characterized by production of IFN-γ and TNF-α are essential for defense against the intracellular pathogens such as viruses, bacteria, protozoa, and fungi (Nishimura et al. 2000; Szabo et al. 2003; Wills-karp 1999), while T-helper 2 cells characterized by secretion of IL-4, IL-5, IL-6, and IL-10 are desirable for antibody production. Both T-helper 1 and 2 cells activate and stimulate proliferation and differentiation of B lymphocyte to differentiate into plasma cells that produce antibodies.
Fig. 3.
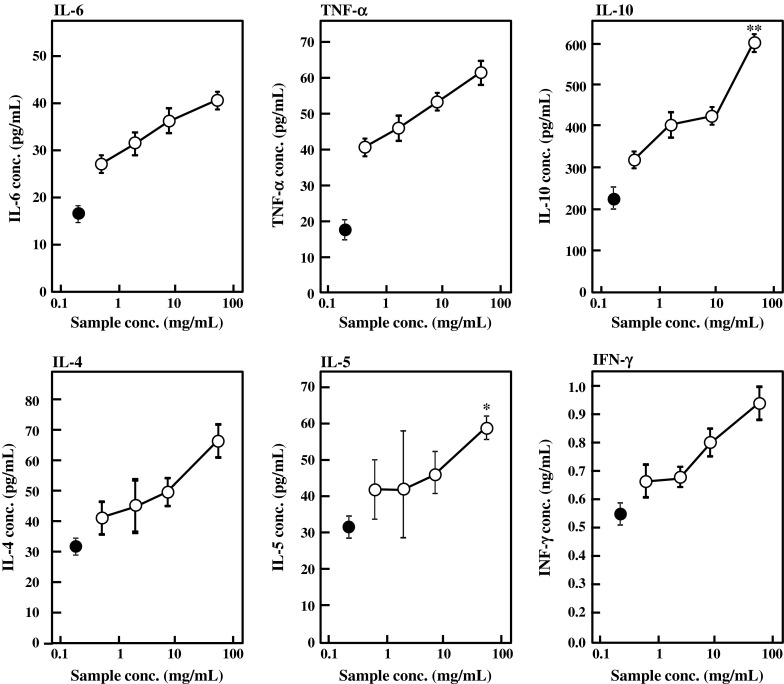
Effect of BFE on cytokine production by mouse primary splenocytes in vitro. Mouse splenocytes were inoculated in 5 % FBS-RPMI 1640 medium containing 5 μg/mL of Con A and various concentrations of BFE and cultured for 48 h. Control (closed circle) was supplemented with distilled water instead of BFE. Results are represented as the mean ± SD of three independent measurements. Significant differences against control are represented as *p < 0.05 or **p < 0.01
Effect of oral administration of BFE on Ig levels in serum
The immunomodulatory activity of BFE was examined in vivo. Following 1 week acclimation, 6-week-old female BALB/c mice were divided into 3 groups (n = 6 in each group). The low- and high-dose groups were administered with BFE at 6.75 and 27 mg/kg body weight/day for 14 days, respectively. The control group was administered with distilled water as vehicle control. As shown in Fig. 4, the IgA, IgG, and IgM levels in serum were significantly enhanced by oral administration of BFE. Since the serum Ig level represents the Ig production by plasma cells, it is conceivable that intake of BFE might affect the plasma cells to enhance Ig production in vivo.
Fig. 4.
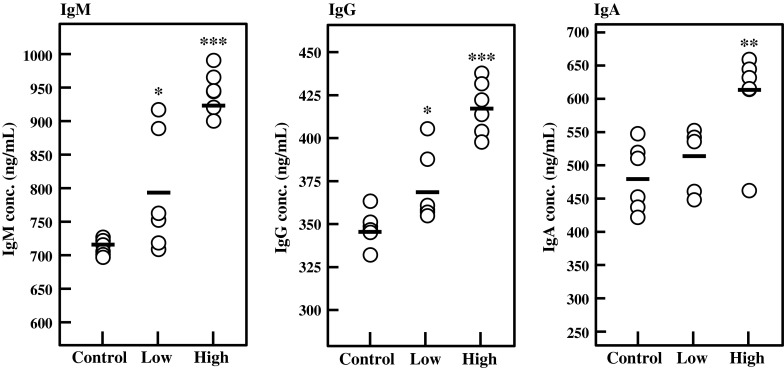
Effect of oral administration of BFE on serum Ig levels. Six-week-old female BALB/c mice (6 mice per group) were orally administered with 6.75 (low-dose) or 27 mg/kg body weight/day (high-dose) of BFE for 14 days. Control group was administered with distilled water as vehicle control. The amounts of Igs in serum were measured using ELISA. Each circle represents the serum Ig level from individual mice. Bars represent an average value of each group (n = 6). Significant differences against control group are represented as *p < 0.05, **p < 0.01, or ***p < 0.001
Ig production-stimulatory activity of BFE in mice
Lymphocytes from the spleen, MLN, and PP were collected from the BFE-administered mice and cultured for 24 h, and the amounts of IgA, IgG, and IgM in culture medium were measured by ELISA. As shown in Fig. 5, intake of BFE significantly enhanced the production of IgM (p < 0.01), IgG (p < 0.01), and IgA (p < 0.05) by splenocytes in the high-dose group against the control. Ig production by MLN lymphocytes was also enhanced by BFE intake (Fig. 6). In the high-dose group, IgA and IgG production by MLN was significantly activated by BFE administration (p < 0.05). IgM production was also stimulated, although there was no significant difference against the control. As shown in Fig. 7, BFE significantly enhanced the Ig production activity of PP lymphocytes. Especially, the IgA production activity of PP lymphocytes was strongly enhanced in both low- and high-dose groups. IgA is the most important class of Igs in the intestinal immune response. The main function of IgA in gut lumen is to prevent the attachment of intestinal pathogens (Kagnoff 1993). B cells living in GALT mainly proliferate and differentiate into IgA-producing plasma cells. The majority of B cells in the lymphoid tissues of normal individuals are IgM+ or IgM+ IgD+. The IgM+ B cells undergo class switch recombination (CSR) and differentiate to IgA+ B cells and eventually to IgA-producing plasma cells, then migrate from the inductive site of PP to the effector site of the gut lamina propria via blood circulation (Chen and Cerutti 2010; Lamm and Phillips-Quagliata 2002; Mora and von Andrian 2008). Thus, it is possible that BFE stimulates IgA CSR in PP and that class-switched IgA+ B cells circulate in the whole body, leading to increases in the IgA level in serum and in IgA production by lymphocytes from the PP, MLN, and spleen especially in the high-dose group. These results suggest that the intake of BFE activates the intestinal immune system.
Fig. 5.
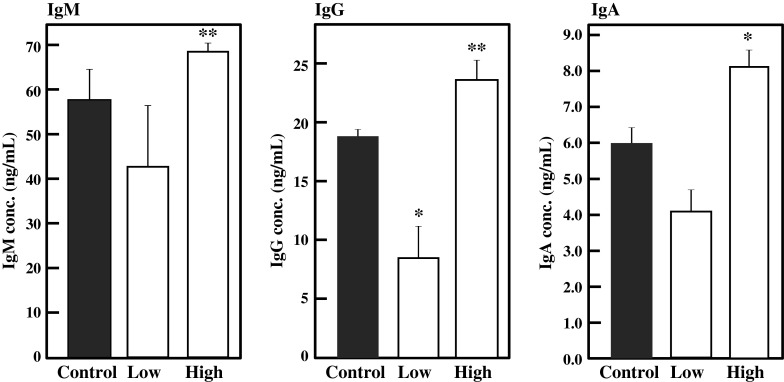
Effect of oral administration of BFE on Ig production activity of splenocytes. Splenocytes collected from BFE-administered mice were inoculated in 5 % FBS-RPMI 1640 medium and cultured for 24 h. The amounts of Igs were measured by ELISA. Results are represented as the mean ± SD (n = 6). Significant differences against control group are represented as *p < 0.05 or **p < 0.01
Fig. 6.
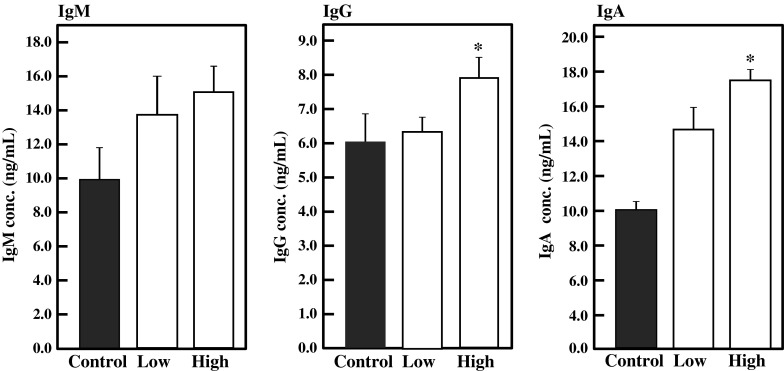
Effect of oral administration of BFE on Ig production activity of MLN lymphocytes. MLN lymphocytes collected from BFE-administered mice were inoculated in 5 % FBS-RPMI 1640 medium and cultured for 24 h. The amounts of Igs were measured by ELISA. Results are represented as the mean ± SD (n = 6). Significant differences against control group are represented as *p < 0.05
Fig. 7.
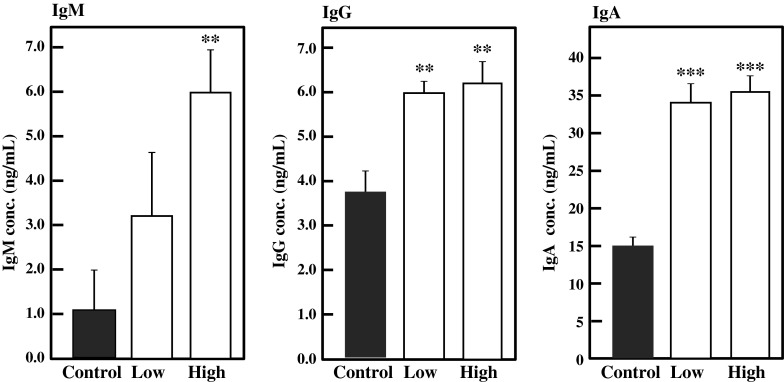
Effect of oral administration of BFE on Ig production activity of PP lymphocytes. PP lymphocytes collected from BFE-administered mice were inoculated in 5 % FBS-RPMI 1640 medium and cultured for 24 h. The amounts of Igs were measured by ELISA. Results are represented as the mean ± SD (n = 6). Significant differences against control group are represented as **p < 0.01 or ***p < 0.001
Transcriptional levels of IgM, IgG1, and IgA were evaluated in splenocytes and PP lymphocytes. As indicated in Fig. 8, oral administration of BFE enhanced gene expression levels of IgM, IgG1, and IgA in both the spleen and PP. Gene expression levels in PP lymphocytes were obviously stimulated by oral administration of BFE. These facts suggest that accelerated Ig production activities result from the enhanced gene expression levels.
Fig. 8.
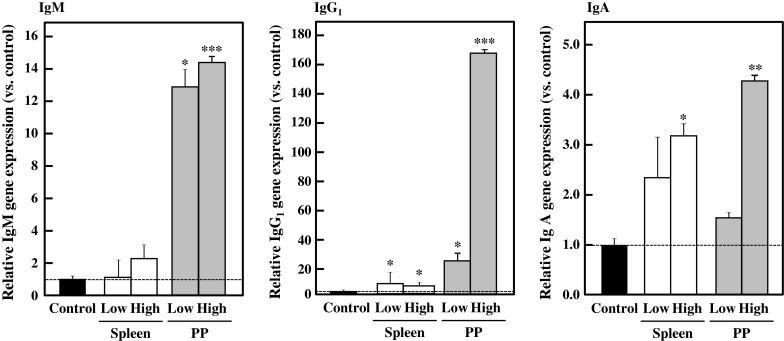
Effect of oral administration of BFE on gene expression levels in lymphocytes. Splenocytes and PP lymphocytes from BFE-administered mice were inoculated in 5 % FBS-RPMI 1640 medium containing 5 μg/mL of Con A and cultured for 48 h. After cultivation, cells were harvested and cDNAs were synthesized using total RNAs from the cells. Each gene expression level was normalized by β-actin as an internal control. Results are represented as the mean ± SD (n = 6). Significant differences against vehicle control group are represented as *p < 0.05, **p < 0.01, or ***p < 0.001
Effect of oral administration of BFE on cytokine production by mouse lymphocytes
To examine the effect of BFE administration on the cytokine production activity of lymphocytes from the spleen, MLN, and PP, these lymphocytes were cultured in 5 % FBS-RPMI 1640 medium supplemented with Con A at 37 °C for 48 h. Con A is a mitogen that selectively promotes T cells (Shih et al. 2009). As summarized in Table 2, the production of IL-6, IL-10, TNF-α, TGF-β, and IFN-γ by splenocytes was significantly stimulated by BFE intake in the high-dose group. BFE intake activated IL-4, IL-6, IL-10, TNF-α, and TGF-β production by MLN lymphocytes. On the other hand, the effect of oral administration of BFE on the cytokine production activity of PP lymphocytes was moderate compared with those of splenocytes or MLN lymphocytes. In addition, mouse CD4+ helper T cell clones can indeed be divided into subsets that secrete either some cytokines such as IL-2 and INF-γ stimulating the cell-mediated immunity or others such as IL-4, IL-5, IL-6, and IL-10 which are particularly efficient helpers for antibody production. Especially IL-4 and IL-10 relate to increase in IgA production by promoting expansion of IgA class-switched B cells and their differentiation into IgA-secreting plasma cells (Abbas et al. 1991; Chen and Cerutti 2010; Lamm and Phillips-Quagliata 2002; Mora and von Andrian 2008). Cytokines also have profound inhibitory effects that serve to regulate the immune responses. IgG production by splenocytes from the mice administered with low-dose of BFE was significantly suppressed (Fig. 5). In addition, production of both IgM and IgA was also decreased, although there was no significant difference. However, the gene expression level of IgG1 was significantly increased (Fig. 8). These might be caused by increased production of TGF-β by splenocytes from the mice administered with low-dose of BFE compared with the control (Table 2), because TGF-β is known as a potent immunosuppressive factor (Abbas et al. 1991). In mice, IgG isotype is divided into IgG1, IgG2a, IgG2b, and IgG3. IL-4 induces IgG1 production and reduces IgG2a production. Production of IgG2a in mice is enhanced by IFN-γ that is also secreted by T cells (Abbas et al. 1991). Production of IL-4 by splenocytes from the mice administered with low-dose of BFE was higher than in the control and high-dose group. IFN-γ was lower in the low-dose group than in the control, although there was no significant difference. This might be the reason why the IgG1 gene expression was facilitated, although IgG production was suppressed by splenocytes from the low-dose group. Production of IgG2a, IgG2b, and IgG3 might be suppressed despite increased production of IgG1. Overall results suggested that BFE intake stimulates the cytokine production activity of lymphocytes and that Ig production was significantly accelerated by intake of high-dose of BFE.
Table 2.
Effect of oral administration of BFE on cytokine production activity of lymphocytes from the spleen, MLN, and PP
| Cytokine (pg/mL) | Spleen | MLN | PP | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Control | Low | High | Control | Low | High | Control | Low | High | |
| IL-4 | 23.4 ± 4.9 | 31.4 ± 10.5 | 27.7 ± 4.6 | 10.3 ± 2.5 | 16.3 ± 5.2* | 37.7 ± 3.8*** | 8.1 ± 0.6 | 7.8 ± 0.6 | 10.4 ± 0.0* |
| IL-6 | 7.7 ± 1.3 | 9.7 ± 3.5 | 12.0 ± 3.7* | 12.3 ± 0.7 | 16.4 ± 3.8* | 36.5 ± 6.1*** | 11.8 ± 2.9 | 11.5 ± 2.8 | 13.9 ± 2.2 |
| IL-10 | 122 ± 43 | 183 ± 74 | 305 ± 101* | 283 ± 27 | 301 ± 55 | 337 ± 53* | 140 ± 31 | 247 ± 25 | 304 ± 227 |
| TNF-α | 11.8 ± 2.8 | 7.5 ± 5.1 | 30.8 ± 16.0* | 16.1 ± 6.5 | 17.5 ± 6.6 | 43.7 ± 5.0*** | 10.1 ± 1.9 | 8.9 ± 0.9 | 12.2 ± 2.3 |
| TGF-β | 602 ± 42 | 700 ± 69* | 696 ± 71* | 423 ± 27 | 492 ± 67 | 858 ± 201* | 587 ± 52 | 528 ± 71 | 686 ± 256 |
| IFN-γ | 67.8 ± 4.2 | 55.1 ± 6.0 | 82.7 ± 10.1** | 632 ± 77 | 602 ± 58 | 504 ± 45 | 628 ± 54 | 569 ± 180 | 815 ± 153* |
Six-week-old female BALB/c mice in three groups (6 mice per group) were orally administered with BFE at 6.75 (low-dose) or 27 mg/kg body weight/day (high-dose) or with distilled water as vehicle control for 14 days. Lymphocytes from the spleen, MLN, and PP were cultured in 5 % FBS-RPMI 1640 medium containing Con A for 48 h. The amounts of cytokines were measured using ELISA. Each result is represented as the mean ± SD (n = 6). Significant differences against the vehicle control group are represented as *p < 0.05, **p < 0.01, or ***p < 0.001
In conclusion, BFE stimulated the Ig production by HB4C5 cells and mouse primary splenocyte in vitro. Oral administration of BFE also enhanced the serum Ig levels and activated production of Igs and cytokines (IL-6, IL-10, TNF-α, TGF-β, and IFN-γ) by lymphocytes from the spleen, MLN, and PP in mice. BFE may activate the adaptive immune responses by enhancing the production of Igs and cytokines in vivo. Hence, it would be possible to consider that bengkoang has a potentially beneficial effect on human health and on prevention of the immune system-related diseases.
References
- Abbas AK, Lichtman AH, Pober JS. Cellular and molecular immunology. Philadelphia: W.B. Saunders; 1991. [Google Scholar]
- Álvarez AR, Peña-Valdivia CB. Structural polysaccharides in xoconostle (Opuntia matudae) fruits with different ripening stages. J PACD. 2009;11:26–44. [Google Scholar]
- Chen K, Cerutti A. Vaccination strategies to promote mucosal antibody responses. J Immun. 2010;33:479–491. doi: 10.1016/j.immuni.2010.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X, Nie W, Yu G, Li Y, Hu Y, Lu J, Jin L. Antitumor and immunomodulatory activity of polysaccharides from Sargassum fusiforme. Food Chem Toxycol. 2012;50:695–700. doi: 10.1016/j.fct.2011.11.015. [DOI] [PubMed] [Google Scholar]
- Cherng JM, Chiang W, Chiang LC. Immunomodulatory activities of edible beans and related constituents from soybean. J Food Chem. 2007;104:613–618. doi: 10.1016/j.foodchem.2006.12.011. [DOI] [Google Scholar]
- Daifuku M, Nishi K, Okamoto T, Nakano H, Nishimoto S, Sugahara T. Immunostimulatory effects of water extract from bulbus arteriosus in tuna in vitro. J Func Food. 2012;4:263–270. doi: 10.1016/j.jff.2011.12.002. [DOI] [Google Scholar]
- Kagnoff MF. Immunology of the intestinal tract. Gastroenterology. 1993;105:1275–1280. doi: 10.1016/0016-5085(93)90128-Y. [DOI] [PubMed] [Google Scholar]
- Kumalasari ID, Harmayani E, Lestari LA, Raharjo S, Asmara W, Nishi K, Sugahara T. Evaluation of immunostimulatory effect of the arrowroot (Maranta arundinacea. L) in vitro and in vivo. Cytotechnology. 2012;64:131–137. doi: 10.1007/s10616-011-9403-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamm ME, Phillips-Quagliata JM. Origin and homing of intestinal IgA antibody-secreting cells. J Exp Med. 2002;195:F5–F8. doi: 10.1084/jem.20011910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langkamp-Henken B, Glezer JA, Kudsk KA. Immunologic structure and function of the gastrointestinal tract. Nutr Clin Pract. 1992;7:100–108. doi: 10.1177/0115426592007003100. [DOI] [PubMed] [Google Scholar]
- LeBlanc JG, Silvestroni A, Connes C, Juillard V, de Giori GS, Piard JC, Sesma F. Reduction of non-digestible oligosaccharides in soymilk: application of engineered lactic acid bacteria that produce α-galactosidase. Gen Mol Res. 2004;3:432–440. [PubMed] [Google Scholar]
- Lin BF, Chiang BL, Lin JY. Amaranthus spinosus water extract directly stimulates proliferation of B lymphocytes in vitro. Int Immunopharmacol. 2005;5:711–722. doi: 10.1016/j.intimp.2004.12.001. [DOI] [PubMed] [Google Scholar]
- Miyazaki Y, Kusano S, Doi H, Aki O. Effects on immune response of antidiabetic ingredients from white-skinned sweet potato (Ipomoea batatas L.) Nutrition. 2005;21:358–362. doi: 10.1016/j.nut.2004.11.001. [DOI] [PubMed] [Google Scholar]
- Mora JR, von Andrian UH. Differentiation and homing of IgA-secreting cells. Mucosal Immunol. 2008;1:96–109. doi: 10.1038/mi.2007.14. [DOI] [PubMed] [Google Scholar]
- Morishige H, Sugahara T, Nishimoto S, Muranaka A, Ohno F, Shiraishi R, Doi M. Immunostimulatory effects of collagen from jellyfish in vivo. Cytotechnology. 2011;63:481–492. doi: 10.1007/s10616-011-9371-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murakami H, Masui H, Sato GH, Sueoka N, Chow TP, Kono-Sueoka T. Growth of hybridoma cells in serum-free medium: ethanolamine is an essential component. Proc Natl Acad Sci USA. 1982;79:1158–1162. doi: 10.1073/pnas.79.4.1158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murakami H, Hashizume S, Ohashi H, Shinohara K, Yasumoto K, Nomoto K, Omura H. Human-human hybridomas secreting antibodies specific to human lung carcinoma. In Vitro Cell Dev Biol. 1985;21:593–596. doi: 10.1007/BF02620891. [DOI] [PubMed] [Google Scholar]
- Nishi K, Kondo A, Okamoto T, Nakano H, Daifuku M, Nishimoto S, Ochi K, Takaoka T, Sugahara T. Immunostimulatory in vitro and in vivo effects of a water-soluble extract from kale. Biosci Biotech Biochem. 2011;75:40–46. doi: 10.1271/bbb.100490. [DOI] [PubMed] [Google Scholar]
- Nishi K, Muranaka A, Nishimoto S, Kadota A, Sugahara T. Immunostimulatory effect of β-cryptoxanthin in vitro and in vivo. J Func Food. 2012;4:618–625. doi: 10.1016/j.jff.2012.04.001. [DOI] [Google Scholar]
- Nishimoto S, Kanda K, Yamawaki M, Okabe M, Akiyama K, Kakinuma Y, Sugahara T. Heavy oil fractions induce negative influences on mouse immune system. J Toxicol Sci. 2009;34:459–468. doi: 10.2131/jts.34.459. [DOI] [PubMed] [Google Scholar]
- Nishimura T, Nakui M, Sato M, Iwakabe K, Kitamura H, Sekimoto M, Ohta A, Koda T, Nishimura S. The critical role of Th1-dominant immunity in tumor immunology. Cancer Chemother Pharmacol. 2000;46:S52–S61. doi: 10.1007/PL00014051. [DOI] [PubMed] [Google Scholar]
- Noman ASM, Hoque MA, Haque MM, Pervin F, Karim MR. Nutritional and anti-nutritional components in Pachyrhizus erosus L. tuber. Food Chem. 2007;102:1112–1118. doi: 10.1016/j.foodchem.2006.06.055. [DOI] [Google Scholar]
- Nurrochmad A, Leviana F, Wulancarsari CG, Lukitaningsih E. Phytoestrogens of Pachyrhizus erosus prevent bone loss in an ovariectomized rat model of osteoporosis. Int J Phytomed. 2010;2:363–372. [Google Scholar]
- Rodríguez R, Jiménez A, Fernández-Bolaños J, Guillén R, Heredia A. Dietary fibre from vegetable products as source of functional ingredients. Trends Food Sci Tech. 2006;17:3–15. doi: 10.1016/j.tifs.2005.10.002. [DOI] [Google Scholar]
- Shih MC, Kuo CC, Chiang W. Effects of drying and extrusion on colour, chemical composition, antioxidant activities and mitogenic response of spleen lymphocytes of sweet potatoes. Food Chem. 2009;117:114–121. doi: 10.1016/j.foodchem.2009.03.084. [DOI] [Google Scholar]
- Sugahara T, Sasaki T, Murakami H. Enhancement of immunoglobulin productivity of human–human hybridoma HB4C5 cells by basic proteins and poly-basic amino acids. Biosci Biotech Biochem. 1994;58:2212–2214. doi: 10.1271/bbb.58.2212. [DOI] [PubMed] [Google Scholar]
- Sugahara T, Onda H, Shinohara Y, Horii M, Akiyama K, Nakamoto K, Hara K. Immunostimulation effects of proteose-peptone component 3 fragment on human hybridomas and peripheral blood lymphocytes. Biochim Biophys Acta. 2005;1725:233–240. doi: 10.1016/j.bbagen.2005.05.008. [DOI] [PubMed] [Google Scholar]
- Szabo SJ, Sullivan BM, Peng SL, Glimcher LH. Molecular mechanisms regulating Th1 immune responses. Annu Rev Immunol. 2003;21:713–758. doi: 10.1146/annurev.immunol.21.120601.140942. [DOI] [PubMed] [Google Scholar]
- Thakur M, Conellan P, Deseo MA, Morris C, Praznik W, Loeppert R, Dixit VK. Characterization and in vitro immunomodulatory screening of fructo-oligosaccharides of Asparagus racemosus Willd. Int J Biol Macromol. 2012;50:77–81. doi: 10.1016/j.ijbiomac.2011.09.027. [DOI] [PubMed] [Google Scholar]
- Wills-Karp M. Immunologic basis of antigen-induced airway hyperresponsiveness. Annu Rev Immunol. 1999;17:255–281. doi: 10.1146/annurev.immunol.17.1.255. [DOI] [PubMed] [Google Scholar]