Abstract
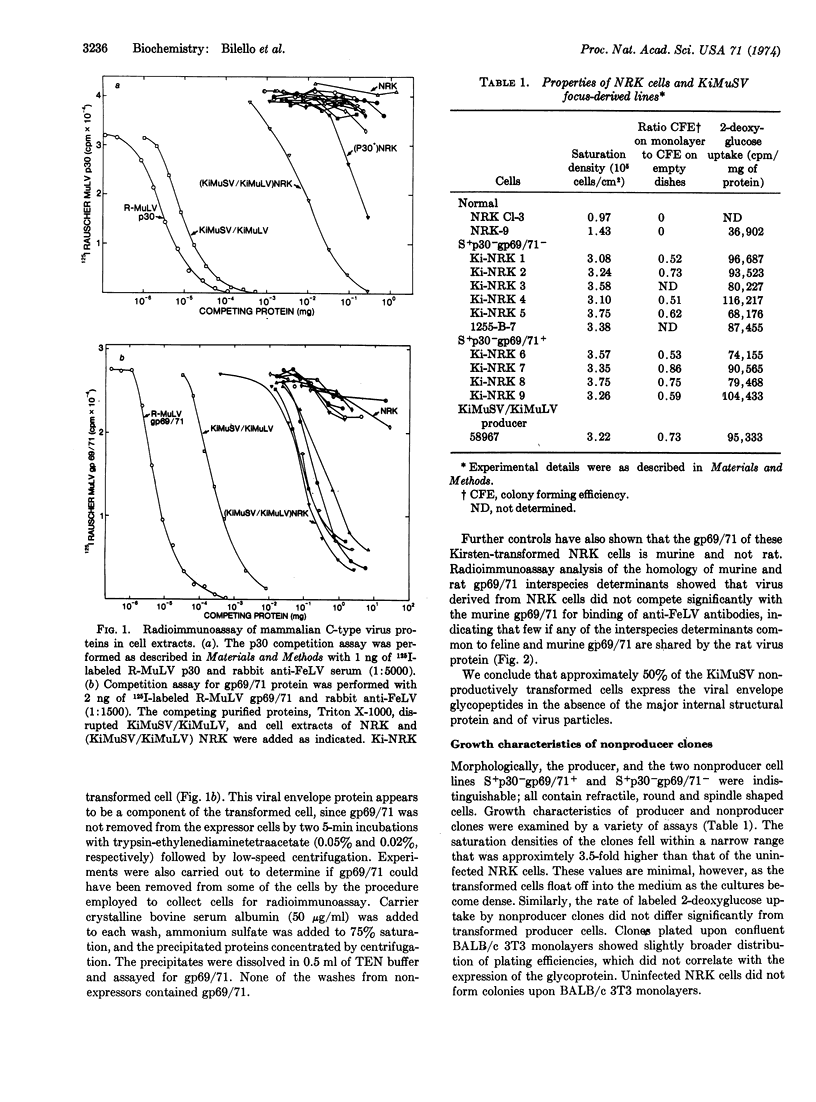
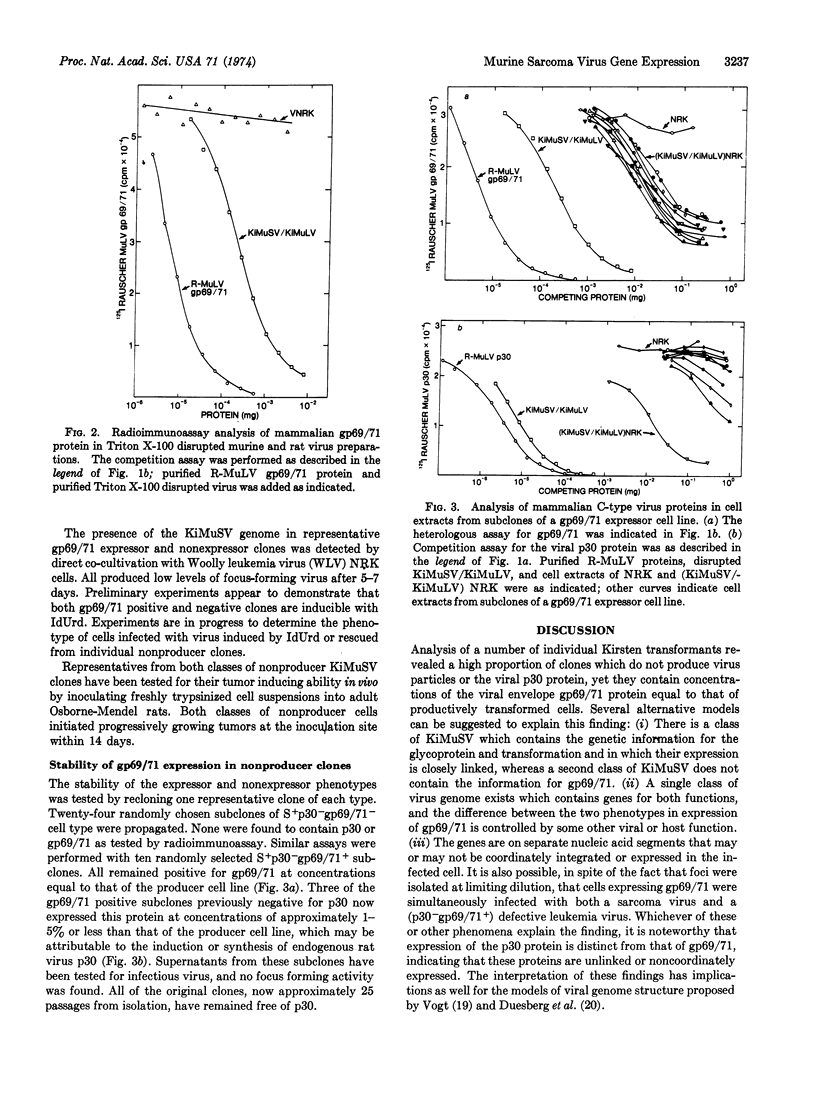
Expression of the major internal protein and the envelope glycoprotein of murine C-type viruses in focus-derived lines of normal rat kidney cells infected with Kirsten murine sarcoma virus was measured by radioimmunoassay. Of the clones selected, which do not produce virus particles or the major viral structural protein, approximately half express the viral envelope glycoprotein at concentrations found in productively infected cells. Expression of the envelope glycoprotein did not appear to alter significantly the properties of the transformed cells in culture.
Keywords: Kirsten murine sarcoma virus
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aaronson S. A., Bassin R. H., Weaver C. Comparison of murine sarcoma viruses in nonproducer and S + L - -transformed cells. J Virol. 1972 Apr;9(4):701–704. doi: 10.1128/jvi.9.4.701-704.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aaronson S. A., Todaro G. J. Basis for the acquisition of malignant potential by mouse cells cultivated in vitro. Science. 1968 Nov 29;162(3857):1024–1026. doi: 10.1126/science.162.3857.1024. [DOI] [PubMed] [Google Scholar]
- Aaronson S. A., Weaver C. A. Characterization of murine sarcoma virus (Kirsten) transformation of mouse and human cells. J Gen Virol. 1971 Nov;13(2):245–252. doi: 10.1099/0022-1317-13-2-245. [DOI] [PubMed] [Google Scholar]
- Ball J., McCarter J. A., Sunderland S. M. Evidence for helper independent murine sarcoma virus. I. Segregation of replication-defective and transformation-defective viruses. Virology. 1973 Nov;56(1):268–284. doi: 10.1016/0042-6822(73)90305-x. [DOI] [PubMed] [Google Scholar]
- Bassin R. H., Phillips L. A., Kramer M. J., Haapala D. K., Peebles P. T., Nomura S., Fischinger P. J. Transformation of mouse 3T3 cells by murine sarcoma virus: release of virus-like particles in the absence of replicating murine leukemia helper virus. Proc Natl Acad Sci U S A. 1971 Jul;68(7):1520–1524. doi: 10.1073/pnas.68.7.1520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benveniste R. E., Scolnick E. M. RNA in mammalian sarcoma virus transformed nonproducer cells homologous to murine leukemia virus RNA. Virology. 1973 Feb;51(2):370–382. doi: 10.1016/0042-6822(73)90436-4. [DOI] [PubMed] [Google Scholar]
- Benveniste R. E., Todaro G. J., Scolnick E. M., Parks W. P. Partial transcription of murine type C viral genomes in BALB c cell lines. J Virol. 1973 Oct;12(4):711–720. doi: 10.1128/jvi.12.4.711-720.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gazdar A. F., Phillips L. A., Sarma P. S., Peebles P. T., Chopra H. C. Presence of sarcoma genome in a "non-infectious" mammalian virus. Nat New Biol. 1971 Nov 17;234(46):69–72. doi: 10.1038/newbio234069a0. [DOI] [PubMed] [Google Scholar]
- Gazdar A. F., Stull H. B., Chopra H. C., Ikawa Y. Properties of flat variants of murine sarcoma virus transformed non-producer cells isolated after high-temperature passage. Int J Cancer. 1974 Feb 15;13(2):219–226. doi: 10.1002/ijc.2910130209. [DOI] [PubMed] [Google Scholar]
- Greenberger J. S., Aaronson S. A. Morphologic revertants of murine sarcoma virus transformed nonproducer BALB-3T3: selective techniques for isolation and biologic properties in vitro and in vivo. Virology. 1974 Feb;57(2):339–346. doi: 10.1016/0042-6822(74)90173-1. [DOI] [PubMed] [Google Scholar]
- HANAFUSA H. ANALYSIS OF THE DEFECTIVENESS OF ROUS SARCOMA VIRUS. 3. DETERMINING INFLUENCE OF A NEW HELPER VIRUS ON THE HOST RANGE AND SUSCEPTIBILITY TO INTERFERENCE OF RSV. Virology. 1965 Feb;25:248–255. doi: 10.1016/0042-6822(65)90203-5. [DOI] [PubMed] [Google Scholar]
- HANAFUSA H., HANAFUSA T., RUBIN H. ANALYSIS OF THE DEFECTIVENESS OF ROUS SARCOMA VIRUS, II. SPECIFICATION OF RSV ANTIGENICITY BY HELPER VIRUS. Proc Natl Acad Sci U S A. 1964 Jan;51:41–48. doi: 10.1073/pnas.51.1.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huebner R. J., Hartley J. W., Rowe W. P., Lane W. T., Capps W. I. Rescue of the defective genome of Moloney sarcoma virus from a noninfectious hamster tumor and the production of pseudotype sarcoma viruses with various murine leukemia viruses. Proc Natl Acad Sci U S A. 1966 Oct;56(4):1164–1169. doi: 10.1073/pnas.56.4.1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klement V., Freedman M. H., McAllister R. M., Nelson-Rees W. A., Heubner R. J. Differences in susceptibility of human cells to mouse sarcoma virus. J Natl Cancer Inst. 1971 Jul;47(1):65–73. [PubMed] [Google Scholar]
- Klement V., Nicolson M. O., Huebner R. J. Rescue of the genome of focus forming virus from rat non-productive lines by 5'-bromodeoxyruidine. Nat New Biol. 1971 Nov 3;234(44):12–14. doi: 10.1038/newbio234012a0. [DOI] [PubMed] [Google Scholar]
- Law L. W., Chang K. S., Nakata K. Heterogeneity of virus production and antigenicity detected in cell clones derived from a "nonproducer" neoplasm induced by Moloney murine sarcoma virus. J Natl Cancer Inst. 1974 Feb;52(2):437–443. doi: 10.1093/jnci/52.2.437. [DOI] [PubMed] [Google Scholar]
- Lieber M. M., Benveniste R. E., Livingston D. M., Todaro G. J. Mammalian cells in culture frequently release type C viruses. Science. 1973 Oct 5;182(4107):56–59. doi: 10.1126/science.182.4107.56. [DOI] [PubMed] [Google Scholar]
- McCoy J. L., Ting R. C., Morton D. L., Law L. W. Immunologic and virologic studies of a nonproducer tumor induced by murine sarcoma virus (Harvey). J Natl Cancer Inst. 1972 Feb;48(2):383–391. [PubMed] [Google Scholar]
- Nomura S., Fischinger P. J., Mattern C. F., Peebles P. T., Bassin R. H., Friedman G. P. Revertants of mouse cells transformed by murine sarcoma virus. I. Characterization of flat and transformed sublines without a rescuable murine sarcoma virus. Virology. 1972 Oct;50(1):51–64. doi: 10.1016/0042-6822(72)90345-5. [DOI] [PubMed] [Google Scholar]
- Peebles P. T., Bassin R. H., Haapala D. K., Phillips L. A., Nomura S., Fischinger P. J. Rescue of murine sarcoma virus from a sarcoma-positive leukemia-negative cell line: requirement for replicating leukemia virus. J Virol. 1971 Nov;8(5):690–694. doi: 10.1128/jvi.8.5.690-694.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peebles P. T., Haapala D. K., Gazdar A. F. Deficiency of viral ribonucleic acid-dependent deoxyribonucleic acid polymerase in noninfectious virus-like particles released from murine sarcoma virus-transformed hamster cells. J Virol. 1972 Mar;9(3):488–493. doi: 10.1128/jvi.9.3.488-493.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Risser R., Pollack R. A nonselective analysis of SV40 transformation of mouse 3T3 cells. Virology. 1974 Jun;59(2):477–489. doi: 10.1016/0042-6822(74)90457-7. [DOI] [PubMed] [Google Scholar]
- Scheele C. M., Hanafusa H. Proteins of helper-dependent RSV. Virology. 1971 Aug;45(2):401–410. doi: 10.1016/0042-6822(71)90341-2. [DOI] [PubMed] [Google Scholar]
- Scolnick E. M., Parks W. P. Isolation and characterization of a primate sarcoma virus: mechanism of rescue. Int J Cancer. 1973 Jul 15;12(1):138–147. doi: 10.1002/ijc.2910120115. [DOI] [PubMed] [Google Scholar]
- Scolnick E. M., Rands E., Williams D., Parks W. P. Studies on the nucleic acid sequences of Kirsten sarcoma virus: a model for formation of a mammalian RNA-containing sarcoma virus. J Virol. 1973 Sep;12(3):458–463. doi: 10.1128/jvi.12.3.458-463.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steeves R. A., Strand M., August J. T. Structural proteins of mammalian oncogenic RNA viruses: murine leukemia virus neutralization by antisera prepared against purified envelope glycoprotein. J Virol. 1974 Jul;14(1):187–189. doi: 10.1128/jvi.14.1.187-189.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strand M., August J. T. Structural proteins of mammalian oncogenic RNA viruses: multiple antigenic determinants of the major internal protein and envelope glycoprotein. J Virol. 1974 Jan;13(1):171–180. doi: 10.1128/jvi.13.1.171-180.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strand M., August J. T. Structural proteins of oncogenic ribonucleic acid viruses. Interspec II, a new interspecies antigen. J Biol Chem. 1973 Aug 25;248(16):5627–5633. [PubMed] [Google Scholar]
- Ting R. C., Yang S. S., Gallo R. C. Reverse transcriptase, RNA tumour virus transformation and derivatives of rifamycin SV. Nat New Biol. 1972 Apr 12;236(67):163–166. doi: 10.1038/newbio236163a0. [DOI] [PubMed] [Google Scholar]
- Tronick S. R., Stephenson J. R., Aaronson S. A. Immunological characterization of a low molecular weight polypeptide of murine leukemia virus. Virology. 1973 Jul;54(1):199–206. doi: 10.1016/0042-6822(73)90129-3. [DOI] [PubMed] [Google Scholar]
- Tsuchida N., Robin M. S., Green M. Viral RNA subunits in cells transformed by RNA tumor viruses. Science. 1972 Jun 30;176(4042):1418–1420. doi: 10.1126/science.176.4042.1418. [DOI] [PubMed] [Google Scholar]



