Abstract
Mitochondria supply cellular energy and also perform a role in the adaptation to metabolic stress. In mammals, the ataxia-telangiectasia mutated (ATM) kinase acts as a redox sensor controlling mitochondrial function. Subsequently, transcriptomic and genetic studies were utilized to elucidate the role played by a fungal ATM homolog during carbon starvation. In Aspergillus nidulans, AtmA was shown to control mitochondrial function and glucose uptake. Carbon starvation responses that are regulated by target of rapamycin (TOR) were shown to be AtmA-dependent, including autophagy and hydrolytic enzyme secretion. AtmA also regulated a p53-like transcription factor, XprG, inhibiting starvation-induced XprG-dependent protease secretion and cell death. Thus, AtmA possibly represents a direct or indirect link between mitochondrial stress, metabolism, and growth through the influence of TOR and XprG function. The coordination of cell growth and division with nutrient availability is crucial for all microorganisms to successfully proliferate in a heterogeneous environment. Mitochondria supply cellular energy but also perform a role in the adaptation to metabolic stress and the cross-talk between prosurvival and prodeath pathways. The present study of Aspergillus nidulans demonstrated that AtmA also controlled mitochondrial mass, function, and oxidative phosphorylation, which directly or indirectly influenced glucose uptake. Carbon starvation responses, including autophagy, shifting metabolism to the glyoxylate cycle, and the secretion of carbon scavenging enzymes were AtmA-dependent. Transcriptomic profiling of the carbon starvation response demonstrated how TOR signaling and the retrograde response, which signals mitochondrial dysfunction, were directly or indirectly influenced by AtmA. The AtmA kinase was also shown to influence a p53-like transcription factor, inhibiting starvation-induced XprG-dependent protease secretion and cell death. Therefore, in response to metabolic stress, AtmA appears to perform a role in the regulation of TOR signaling, involving the retrograde and SnfA pathways. Thus, AtmA may represent a link between mitochondrial function and cell cycle or growth, possibly through the influence of the TOR and XprG function.
Keywords: ATM kinase, glucose starvation, cell death, autophagy
The coordination of cell growth and division with nutrient availability is crucial for all microorganisms to successfully proliferate in a heterogeneous environment. The extracellular detection of nutrient availability and the intracellular sensing of energetic status induce a complex network of interlinked signal transduction pathways that subsequently regulate the appropriate cellular responses, coordinating metabolism and growth. The current understanding of how cell growth is controlled in response to nutrient availability is largely based on the study of mammalian systems in which several central protein kinases, including the AMPK (AMP-activated kinase) and TOR, were identified (Oldham and Hafen 2003; Fingar and Blenis 2004). Saccharomyces cerevisiae is a powerful model system for the study of nutrient sensing (Dechant and Peter 2008) and has provided a detailed understanding of nutrient availability signaling pathways. The mammalian kinases and signaling pathways implicated in the involvement in the control of cell growth are well-conserved in S. cerevisiae (Wilson and Roach 2002; de Virgilio and Loewith 2006; Busti et al. 2010; Rubio-Texeira et al. 2010; Santos et al. 2012).
The cAMP-dependent protein kinase A (PKA) and TOR pathways are essential for the promotion of S. cerevisiae cell growth and proliferation under nutrient-rich conditions. The cAMP–PKA pathway influences cell growth and sporulation via the activation of the Kss1/Fus3 mitogen-activated protein kinase (MAPK) cascades (Dechant and Peter 2008). The TOR kinases are activated by glucose and nitrogen sources (Barbet et al. 1996; Rolland et al. 2000) and during nutrient deprivation become inactive, resulting in a downregulation of cell growth and protein synthesis, while activating autophagy (Dechant and Peter 2008). Inactivation of either the cAMP–PKA pathway or the TOR pathway results in G1 arrest (Matsumoto et al. 1982; Barbet et al. 1986) and the activation of starvation responses (Gray et al. 2004), suggesting that PKA and TOR regulate cell growth by promoting G1 progression (Dechant and Peter 2008).
ATM is a serine/threonine protein kinase and a member of the phosphoinositide 3-kinase–related protein kinase family (Derheimer and Kastan 2010). Ataxia-telangiectasia is a rare autosomal-recessive disorder that causes progressive cerebellar ataxia, neurodegeneration, radio sensitivity, cell-cycle checkpoint defects, genome instability, and a predisposition for cancer (Boder and Sedgwick 1958; Kastan and Lim 2000; Lavin and Shiloh 1997). ATM plays a central role in coordinating the molecular events involved in DNA double-strand break signaling and repair (Langerak and Russell 2011; Stracker et al. 2013). Extensive evidence demonstrates how ATM is involved in the regulation of mitochondrial function, glucose homeostasis, serum starvation, and autophagy (Eaton et al. 2007; Halaby et al., 2008; Alexander et al. 2010; Ching et al. 2010; Ditch and Paull 2012; Vazquez-Martin et al. 2011; Patel et al. 2011; Yang et al. 2011; Valentin-Vega and Kastan 2012; Valentin-Vega et al. 2012). In A. nidulans, the ATM homolog, AtmA, has an additional function in the regulation of polarized hyphal growth and the ΔatmA mutant has been shown to possess an accelerated rate of proliferation and increased nuclear kinetics (Malavazi et al. 2006, 2007). Interestingly, AtmA was recently also shown to be involved in the regulation of hydrolytic enzyme secretion (Brown et al. 2013). An interconnected network of activation between ATM, AMPK and TOR, in response to nutritional cues has been elucidated in mammals (Ditch and Paull 2012). Therefore, AtmA may also play a central role in the sensing of cellular energetic status.
Autophagy and apoptosis represent two distinct forms of programmed cell death (PCD) (Kourtis and Tavernarakis 2009). Autophagy forms part of a starvation response that is controlled by the highly conserved autophagy-related genes (ATGs). However, the function of autophagy is not restricted to nutrient recycling and is also involved in the removal of damaged proteins and/or organelles (Bursch et al. 2008). In mammals and S. cerevisiae, autophagic death can occur upon severe glucose deprivation and is regulated by the atg1 kinase and TOR (Wullschleger et al. 2006; Levine and Kroemer 2008; Mizushima et al. 2008; Kourtis and Tavernarakis 2009). In S. cerevisiae and filamentous fungi, apoptotic-like cell death occurs during aging, reproduction, and after exposure to antifungal compounds (de Castro et al. 2011; Ramsdale 2008; Sharon et al. 2009). In Aspergilli, cell death is induced by carbon starvation (Nitsche et al. 2012; Szilágyi et al. 2013). Initially, autophagy is observed as an early starvation response (Nitsche et al. 2012; Szilágyi et al. 2013). Later, as a consequence of an apoptotic-like process and the degradation of cell wall biopolymers, hyphal fragmentation and autolysis occur (Emri et al. 2004, 2008).
The A. nidulans XprG transcription factor belongs to the p53 superfamily involved in gene regulation during starvation and subsequently influences cell death (Katz et al. 2006; Katz et al. 2009). The xprG gene was first identified in a genetic screen for mutants with elevated proteases secretion (Katz et al. 1996). The XprG homolog in Neurospora crassa, Vib-1, has also been shown to be involved in cell death during heterokaryon incompatibility (Dementhon et al. 2006). In mammalian cells, different residues of p53 protein are phosphorylated by ATM on exposure to distinct stimuli (Bensimon et al. 2011; Ditch and Paull 2012). In response to DNA damage activation and during the regulation of glucose homeostasis, ATM phosphorylates p53, resulting in its activation and an increase in protein stability (Shiloh 2006; Armata et al. 2010). In addition, ATM phosphorylates many other downstream protein kinases, with more than 700 proteins being shown to possess ATM phosphorylation motifs (Matsuoka et al. 2007). The reason for multiple ATM phosphorylation targets within each ATM-dependent pathway demonstrates how ATM functions to fine-tune various pathways influencing the same process.
The present study aimed to generate a better understanding of the involvement of AtmA in the regulation of mitochondrial function, glucose uptake, and autophagy during carbon starvation in A. nidulans. The loss of AtmA led to a reduction in glucose uptake, decreased respiratory capacity, and the elevated production of reactive oxygen species (ROS) representative of mitochondrial dysfunction. The role performed by AtmA during carbon starvation was investigated by transcriptional profiling. AtmA was shown to be involved in the control of extracellular hydrolase production and glucose uptake during carbon starvation. In addition, AtmA was shown to be involved in the regulation of XprG-dependent processes, including carbon starvation–induced protease secretion and cell death.
Materials and Methods
Strains and growth conditions
All A. nidulans strains used in this study are listed in Table 1. Complete media YG (2% w/v glucose, 0.5% w/v yeast extract, trace elements) and minimal medium MM (1% w/v glucose, nitrate salts, trace elements, pH 6.5) were used. The compositions of the trace elements, vitamins, and nitrate salts were described by Kafer (1977). According to strain auxotrophy, the media was supplemented with 1.2 g/liter of uracil and uridine (for pyrG auxotrophs), 1 g/liter of aminobenzoate (for pabaA auxotrophs), or 0.5 g/liter of pyridoxine HCl (for pyroA auxotrophs). Solid derivatives of YG and MM were prepared via the addition of agar (2% w/v). Before starvation, 1×107 conidia were incubated in 50 ml liquid MM at 37° in a rotatory shaker (180 rpm) for 24 hr. Subsequently, mycelia were washed with distilled water and then incubated in glucose-free MM at 37° for 12, 24, 48, 96, or 192 hr.
Table 1. Aspergillus nidulans strains used in this study.
| Strain | Genotypea | Reference |
|---|---|---|
| MK85 | biA1; cho1; xprG1; niiA4 | Katz et al. (1996) |
| MK414 | pabaA1; yA2; argB2; xprGΔ1(xprG:: argB) | Katz et al. (2006) |
| GFPatg8 | pyrG89; argB2; argB::ΔnkuA; pyroA4 atgH::GFP pyrG | Mark Marten’s laboratory |
| alcA::xprG | cho1; pyroA4 pyrG89; argB2; argB::ΔnkuA; aclA xprG::pyrG89 | This study |
| ACS16 alcA::xprG | wA3; ΔatmA; alcA xprG | This study |
| R21 | yA1 pabaA1 | Fantes and Roberts (1973) |
| ACS16 | pyrG89; pyroA1; wA3; ΔatmA::pyrG(FOA) | Malavazi et al. (2006) |
| MK85ACS16 | wA3; xprG1; ΔatmA | This study |
| MK414ACS16 | wA3; pyrG89; xprGΔ1, ΔatmA | This study |
| ACS16 GFPatg8 | wa3; ΔatmA; atgH::GFP | This study |
| Δatg1 | pyrG89; wA3; argB2; ΔnkuAku70::argB pyroA4; sE15 nirA14 chaA1fwA1; Δatg1::pyrG89 | FGSCb |
| Δatg1 atg8::GFP | cho1; Δatg1; atgH::GFP | This study |
For the meanings of gene symbols, see Clutterbuck (1993).
Fungal Genetics Stock Center (http://www.fgsc.net).
Oxygen uptake measurements
Wild-type and ΔatmA germlings were obtained by incubating a total of 1×108 conidia in 50 ml MM for 16 hr at 37° (250 rpm). The germlings were harvested by centrifugation and washed twice with cold distilled water. Precipitated germlings were incubated for 5 hr (90 rpm) in a standard A. nidulans protoplasting solution (Novozyme 234 from Novo Nordisk was used as a lytic enzyme) (Osmani et al. 1987) at 30° to partially lysis the cell wall to facilitate respiratory substrate and inhibitors entry. After incubation, germlings were washed three times with 0.7 mM sorbitol, 10 mM HEPES–KOH, pH 7.2, and were subsequently kept on ice. Oxygen uptake was measured with a Clark-type electrode fitted to a Gilson oxygraph (Gilson Medical Electronics, Middleton, WI) in 1.8 ml of medium containing 0.7 mM sorbitol, 10 mM HEPES–KOH, pH 7.2, 5 mM MgCl2, and 0.5 mM EGTA at 30° (Dinamarco et al. 2010). The initial solubility of oxygen in the reaction buffer was considered to be 445 ng atoms of oxygen per ml. Further additions are represented in Table 2.
Table 2. Oxygen consumption in the wild-type and ΔatmA mutant strain.
| Treatmentsa | Wild-type | ΔatmA |
|---|---|---|
| Respiration medium | 43.90 ± 3.2 | 21.40 ± 4.06 |
| Antimycin Ab | 30.10 ± 1.2 | 14.03 ± 3.45 |
| SHAMc | 3.25 ± 0.3 | 2.10 ± 1.52 |
| TMPD/Ascorbated | 28.66 ± 0.9 | 11.85 ± 1.50 |
| KCNe | 2.20 ± 0.65 | 3.80 ± 0.80 |
Mean ± SD of three independent experiments. The values represent the rate of oxygen uptake and were expressed as ng atoms O/min/3 ×107 cells.ml−1.
0.5 µg Antimycin A.
2.5 mM SHAM (salicylhydroxamic acid).
150 µM TMPD (N.N.N.N-tetramethyl-p-phenylenediamine) and 0.5 mM ascorbate (ASC).
1.0 mM KCN.
Mitochondrial mass measurements
Conidia from wild-type and ΔatmA strains (1×108 ml−1) were incubated in 50 ml liquid MM at 37° on a reciprocal shaker for 6 hr. Subsequently, conidia were washed with phosphate-buffered saline (PBS; 140 mM NaCl, 2 mM KCl, 10 mM NaHPO4, 1.8 mM KH2PO4, pH 7.4) and incubated with 8 nM MitoTracker Green FM (Invitrogen) or 5 nM Nonyl Acridine Orange (NAO; Invitrogen) diluted in PBS plus 5% fetal bovine serum (FBS) for 10 min at 37°. Stained conidia were washed with PBS, resuspended in PBS plus 5% FBS, and then analyzed by flow cytometry. Propidium iodide (0.5 µM) staining of the nucleus was utilized to exclude dead cells. Flow cytometry was analyzed by guava Easycity 8 HH (Millipore) using 10,000 acquisitions for each analysis.
For cytochrome c measurements, 2×108 conidia/ml of wild-type and ΔatmA strains were inoculated in MM for 16 hr at 37°. After this period, the cultures were filtered and washed with 100 ml H2O, immediately frozen in liquid nitrogen and macerated. For every 0.1 g of dry weight, 1.3 ml HB buffer (150 mM NaCl, 30 mM KCl, 10 mM Na2HPO4, pH 7.0) was added. The suspension was centrifuged at 21,000 rpm at 4° for 30 min. The supernatant was removed and centrifuged again for 10 min under the same conditions. After determining protein concentration, samples were prepared for SDS-PAGE by the addition of 1× sample buffer (62.5 mM Tris-HCl pH 6.8, 2% SDS, 10% glycerol, 5% β-mercaptoethanol, and 5% bromophenol blue) and heating at 100° for 3 min. Thirty micrograms of total protein from each sample were loaded into each lane of a 15% SDS-PAGE gel. After separation of the proteins, the gel was blotted onto a pure nitrocellulose membrane (0.2 μm; Bio-Rad) and after being blocked in 5% dried milk in TBS-T buffer (10 mM Tris-HCl, 150 mM NaCl, pH 8.0, and 0.05% Tween 20), the membrane was probed with the rabbit anti-cyc1 antibody (CNAT against native cytochrome c from yeast; Sigma) at a 1:200 dilution in TBS-T buffer for 1 hr at room temperature. The membrane was washed four times with TBS-T buffer for 5 min and then incubated with a 1:5000 dilution of goat anti-rabbit IgG peroxidase-labeled (KPL) antibody for 1 hr. After being washed, the blot was developed by use of the SuperSignal Ultra chemiluminescence detection system (Pierce) and recorded by the use of Hyperfilm ECL (Amersham Biosciences).
Glucose uptake assay
Glucose uptake rates were measured by assessing the incorporation of D-[U-14C] glucose [250–360 mCi (9.25–13.33 GBq)/mmol; Perkin Elmer Life Sciences] into germinating conidia. Briefly, 1.2×109 conidia was inoculated into 300 ml MM containing 2% glucose (w/v) as a carbon source, and incubated for 6 hr at 37° in an orbital shaker (180 rpm). Germinating conidia were harvest by centrifugation (3000 rpm) for 5 min and washed four times with ice-cold MM without a carbon source to eliminate traces of glucose. Washed conidia were gently resuspended. For the glucose transport analysis, aliquots of 250 µl (of 2.5×107 germinating conidia) containing D-glucose (0.1–20 mM) were dispensed into 2-ml tubes plus 1 µl radiolabeled glucose (0.2 µCi). Incubation times ranged from 30 to 180 sec at 37° with shaking. To quench the reaction, 1 ml ice-cold MM without carbon was added. The germinating conidia were washed twice with 1 ml ice-cold MM without carbon and transferred to 8 ml ScintiSafe Econo1 scintillation liquid (Fisher Scientific). The D-[U-14C] glucose taken-up by cells was measured using Tri-Carb 2100TR Liquid Scintillation Counter.
ROS detection
Intracellular ROS levels were monitored with the oxidant-sensitive probe 5-(and 6)-chloromethyl-2′,7′-dichlorofluorescin diacetate CM-H2DCFDA (Invitrogen). Liquid YUU medium (50 ml) was inoculated with 1×107 conidia and incubated on a rotatory shaker (180 rpm) for 6 hr at 37°. The nonstarved cultures were centrifuged at 4000 rpm and the pellet was resuspended in 2 ml YUU. The starved culture was centrifuged at 4000 rpm, the pellets were washed in MM without any carbon source, centrifuged again, resuspended in MM without a carbon source and incubated for another 1 hr to starve the culture. The starved cultures were centrifuged at 4000 rpm and the pellet was resuspended in 2 ml MM without a carbon source. After starvation, the ROS assay was prepared from 20 µl of either culture (starved or nonstarved) plus 180 µl fresh YUU or MM without a carbon source and 2.9 µg/ml CM-H2DCFDA in a 96-well imaging plates (BD Falcon). The assay plate was incubated at 37° for 30 min under shaking. The arbitrary fluorescence units (AFUs) were measured at 503 nm of excitation and 529 nm of emission in the fluorimeter Synergy (Biotek) using the Gen5 software.
RNA isolation, cDNA synthesis and quantitative PCRs
Mycelia were harvested by filtration, washed with sterile water, immediately frozen in liquid nitrogen and ground into a powder while frozen. Total RNA was extracted with Trizol (Life Technologies) and RNAse-free DNAse treated as described by Semighini et al. (2002) and then purified using the RNeasy Plant Mini Kit (Qiagen). RNA integrity was confirmed on a bioanalyzer (Agilent). The synthesis of cDNA was performed with approximately 10 µg of RNA, 150 pmol oligo (d)T, and 200 units Superscript II (Invitrogen) according to manufacturer’s instructions. Quantitative PCRs were performed as previously described by Semighini et al. 2002. Primers used are list in Table S1. Expression of the tubulin gene TubC (AN6838) was used as an endogenous control for XprG normalisation.
Microarray slide construction and gene expression methods
A. nidulans genes transcriptionally regulated after exposure to carbon starvation for 12 hr and 24 hr were identified using Agilent custom-designed oligonucleotides arrays (4× 45K microarray). The two strains used in this study were the wild-type and ΔatmA. The 0 hr starvation time point (growth in glucose 2% for 24 hr) was used as a hybridization reference for each strain. To construct the microarray slides, the Agilent E-array software tool was used (available at https://earray.chem.agilent.com/earray/). Briefly, we uploaded gene sequences representing the whole A. nidulans A4 gene sequences. The ORF number was carefully validated by comparing the sequences deposited in three databanks [CADRE (The Central Aspergillus Resource), AspGD (Aspergillus Genome Database), and BROAD Institute] aiming to identify and validate the sequences for probe design, which resulted in 11,251 ORFs being submitted to Agilent E-array. Based on some quality parameter implemented in Aglilent E-array (such as sequences with high scores for cross-hybridization potential throughout the genome and sequences that no appropriate regions could be found as targets to be represented in the slides), 11,143 probes were designed from the uploaded sequence of the A4 strain. These probes were represented three to four times in the microarray slides and the annotation based on [1, 2] was used to generate the annotation file used in the analysis. Therefore, the microarray slides comprised 45,220 features including 1417 Agilent internal controls and 800 internal controls that represented 80 randomly chosen A. nidulans ORFs (each 10-times replicated).
The gene expression analysis used in this work was performed using custom-designed oligonucleotides slides (4× 44K microarray) from Agilent Technologies based on A. nidulans genome annotation publicly available. After RNA isolation and purification, the samples were labeled with Cy-3 or Cy-5-dUTP using the two-color microarray-based gene expression analysis (Quick Amp Labeling Kit; Agilent Technologies) following the manufacturer’s protocol. Initially, 200 ng total RNA was incubated with the Agilent RNA Spike-In control probes (RNA Spike A or B mix). Before labeling, synthesis of cRNA was performed by incubating the samples with 1.2 µl T7 promoter primer and nuclease-free water in an appropriate volume. The template and primer were denatured by incubating the reaction at 65° in a circulating water bath for 10 min, and after the reactions were placed on ice for 5 min. cRNA Master Mix (4 µl 5× First Strand Buffer, 2 µL 0.1 M DTT, 1 µl 10 mM dNTP mix, 1 µl MMLV-RT, and 0.5 µl RNaseOut) were added to the samples, and the mixture was incubated at 40° in a circulating water bath for 2 hr. After, the samples were moved to a 65° circulating water bath and incubated for 15 min. cRNA amplification and labeling were performed by adding the Agilent Transcription Master Mix (20 µl 4× transcription buffer, 6 µl 0.1 M DTT, 8 µl NTP mix, 6.4 µl 50% PEG, 0.5 µL RNase OUT, 0.6 µl inorganic pyrophosphatase, 0.8 µl T7 RNA polymerase, 2.4 µl Cyanine 3-CTP to control samples, or cyanine 5-CTP to treated samples, and 15.3 µl nuclease-free water) to the samples and incubating the mixtures in a circulating water bath at 40° for 2 hr. The labeled cRNA was purified using RNeasy Mini Kit (Qiagen) and then quantified in the NanoDrop 2000 Thermo Scientific (Uniscience).
For the hybridization, 825 ng of each labeled cRNA was mixed with Agilent Fragmentation Mix (11 µl 10× blocking agent, 2.2 µl 25× fragmentation buffer, and nuclease-free water to bring the volume to 52.8 µl) and incubated at 60° for exactly 30 min to fragment RNA. The fragmentation was interrupted by adding 55 µl of 2× GE Hybridization Buffer HI-RPM. Finally, 100 µL sample was placed onto the microarray slide, which was mounted in the Agilent Microarray Hybridization Chamber Kit. The hybridization was performed in an Agilent G2545A Hybridization Oven set to 65° for 17 hr. After, microarray slides were washed according to Agilent’s instructions and scanned using GenePix 4000B microarray scanner (Molecular Devices).
Gene expression analysis
The extraction of data from TIFF images generated through scanning of microarray slides was performed by using Agilent Feature Extraction (FE) software version 9.5.3.1 (Agilent Technologies) using Linear Lowess algorithm to obtain background subtracted and normalized intensity values. The dye-normalized values generated in the FE data files were uploaded into the software Express Converter (version 2.1; TM4 platform available at http://www.tm4.org/utilities.html), which converts the Agilent file format to a mev (multi-experiment viewer) file format compatible with the TM4 software for microarray analysis (available at http://www.tm4.org/). The mev files were then uploaded in the MIDAS software (TM4 platform), where the resulting data were averaged from replicated genes on each array from three biological replicates of each treatment. The generated mev files were finally analyzed by using TIGR MeV (TM4 platform, multi-experiment viewer; available at http://www.tigr.org/software/microarray.shtml), whereby differentially expressed genes were statistically identified using one-class t test (P > 0.01). Significantly different genes were those whose mean log2 expression ratios was statistically different from 0, which indicates the absence of gene modulation. The full dataset was deposited in the Gene Expression Omnibus (GEO) from the National Center of Biotechnology Information (NCBI) with the number http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE42732.
Staining and microscopy
Sterile coverslips were overlaid with 5 ml liquid YUU medium containing approximately 1×106 conidia and incubated at 25° for 12 hr before starvation. Starvation was induced by replacing YUU with MM without carbon and incubating at 25° for different time periods. Germlings starved of carbon for 12-hr and 24-hr were fixed (3.7% formaldehyde; 50 mM Pipes, pH 6.7; 25 mM EGTA, pH 7.8; 5% dimethyl sulfoxide) for 15 min and washed three times for 5 min in PEM (50 mM Pipes, 25 mM EGTA, 5 mM MgS04). Cell walls were partially permeabilized with a digestion solution [50 mg/ml lysing enzymes (Lallzyme Mmx; Lallemand), 6 mg/ml BSA (Sigma) solubilized in PEM] for 1 hr at 30°. After cell wall permeabilization, germlings were washed for 10 min in PEM and then PEM plus BSA (1% w/v). Finally, the germlings were stained with TUNEL (In Situ Cell Death Detection Kit; Roche Diagnostics) for 1 hr at 37°, with 100 ng/ml Hoescht 33258 (Molecular Probes) for 2 min and subsequently washed three times with PEM plus BSA. Propidium iodide (3.75 µg/ml) and Hoescht double staining was performed at room temperature for 2 min and then washed three times for 5 min with PBS. The samples were examined using a Zeiss epifluorescence microscope with excitations of 359, 498, 536, and 563 nm and emissions of 461, 516, 617, and 582 nm for Hoescht, GFP, PI, and TUNEL, respectively. The phase contrast bright field and fluorescent images were captured with AxioCam camera (Carl Zeiss) and processed using the AxioVision software version 3.1.
To determine mitochondrial mass, the strains were grown on coverslips overlaid with MM containing 1% glucose. MitoTracker Green FM (8 nM) and NAO (5 nM) staining were performed at 37° for 10 min. The germlings were examined using a Zeiss epifluorescence microscope with MitoTracker Green FM excitations of 470/20 and emissions of 525/50 and NAO excitations of 572/25 nm and emissions of 626/62 nm. The phase contrast bright field and fluorescent images were captured with AxioCam camera (Carl Zeiss) and processed using the AxioVision software version 3.1.
Construction of the alcA::xprG mutant
The strategy to overexpress the xprG gene was based on the replacement of its promoter with the alcA promoter (Flipphi et al. 2002). First, a 1087-bp DNA fragment from the 5′ unstranslated region (UTR) from the xprG gene was PCR-amplified from A. nidulans wild-type genomic DNA by using the 5′ UTR XprG pRS426 forward and 5′ UTR XprG pyrG reverse primers. Then, a 1900-bp DNA fragment of the A. fumigatus pyrG marker gene was PCR-amplified from the pCDA21 vector by using the pyrG alcA forward and pyrG alcA reverse primers. Subsequently, the alcA minimal promoter (307-bp) was PCR-amplified from the pMCB17apx vector with pyrG alcA forward and pMCB17 alcA reverse primers. Finally, to allow homologous integration at the 3′-end, a 1159-bp DNA fragment of the xprG ORF was PCR-amplified with the primers alcA orf XprG and pRS426 orf XprG reverse. Because each DNA fragment has approximately 20-bp overlapping sequences, it was possible to assemble them by mixing equal proportions of the four fragments described and PCR-amplifying them by using the external primers 5′ UTR XprG pRS426 forward and pRS426 orf XprG reverse. The PCR-mediated assembled DNA fragment was transformed into the A. nidulans TNO2A3 (ΔnkuA) strain (Nayak et al. 2006) according to standard protocols (Osmani et al. 1987). Primer sequences are described in the Supporting Information, Table S1.
Protease activity
Two assays were used to test protease activity. For the semi-quantitative assay, the different strains were grown for 48 hr at 37° on solid MM plus 1% milk powder as a sole carbon source. Plates containing the alcA::xprG and ΔatmA alcA::xprG mutant strains were supplemented with 10 mM cyclopentanone. The formation of a halo of protein degradation, representative of protease secretion, was evaluated and the clearance index calculated (diameter of clearance zone / diameter of colony). The quantitative method used the fluorescence resonance energy transfer (FRET) peptide library Abz GXXXXXQ-EDDnp (Oliveira et al. 2012); 100 µl supernatant from 48-hr carbon-starved cultures was mixed with 100 µl buffer (sodium acetate 100 mM, pH 4.5; sodium phosphate 250 mM, pH 7.0; or tris-HCl 100 mM, pH 8.5) and 1 µl of the Abz GXXXXXQ-EDDnp peptide library (1 mg/ml of DMSO) was added. The FRET peptide library is composed of Abz (or MCA)-GXXXXXQ-EDDnp and Abz (or MCA)-GXXZXXQ-EDDnp, where X stands for an equimolar mixture of all amino acids, the Z position is fixed with one of the proteinogenic amino acids (cysteine was excluded), Abz (ortho-aminobenzoic acid) is the fluorescence donor, and Q-EDDnp (glutamine-[N-(2,4-dinitrophenyl)-ethylenediamine]) is the fluorescence acceptor (Oliveira et al. 2012). Reactions were performed within 96-well imaging plates (BD Falcon) at 30° for 30 min under shaking. The AFUs were measured in a fluorimeter Synergy (Biotek) using the Gen5 software (excitation 320 nm, emission 420 nm). Finally, the results were expressed as AFU/mg of mycelia dry weight.
Results
AtmA influences mitochondrial function plus glucose uptake and/or consumption
The ΔatmA strain demonstrated elevated ROS accumulation in both nonstarved and starved cells (Figure 7), which could have reflected mitochondrial or respiratory dysfunction. Initially, the speed of oxygen consumption was determined. The absence of atmA was shown to impact aerobic respiration efficiencies and resulted in a 50% reduction in oxygen consumption when compared to the wild-type strain (Table 2). Mitochondrial mass in the wild-type and ΔatmA strains was subsequently evaluated. Fluorescent microscopy and flow cytometric analyses (FCA) of the mitochondrial stains, Mitotracker Green and NAO, revealed the fluorescence emitted by the ΔatmA strain was 3.0-fold and 2.0-fold higher than the wild-type strain using the two respective stains (Figure 1, A and B). One possible explanation for the different mitochondrial staining patterns is that the overall mitochondrial content might be higher in the ΔatmA mutant than in the wild-type strain. However, ΔatmA germlings have a larger volume, which could contribute to some degree for the higher fluorescent signal. To discard this possibility and test this hypothesis, we quantified the levels of cytochrome c in both the wild-type and ΔatmA mutant strains by Western blot via densitometry by using the ImageJ software (http://rsbweb.nih.gov/ij/download.html) (Figure 1C). The ΔatmA mutant showed approximately two-fold more cytochrome c than the wild-type strain (Figure 1C). The reduction in oxygen consumption in the ΔatmA strain was therefore not attributed to a reduction in mitochondrial mass. Subsequently, to identify the phase of the oxidative phosphorylation process that was affected by the loss of AtmA function, phase-specific inhibitors were sequentially added and oxygen consumption determined. To evaluate complex IV activity (cytochrome c oxidase), complex III and the alternative oxidase were inhibited by the addition of Antimycin A and SHAM, respectively (Table 2). Inhibition of complex III of the respiratory chain, via the addition of antimycin A, resulted in a 31.5% and 34.5% reduction in the wild-type and ΔatmA strains, respectively. The remaining capacity to consume oxygen was lost via the addition of the alternative oxidase inhibitor SHAM (these results were similar for both strains) (Table 2). Subsequently, the addition of complex IV substrates, TMPD and ascorbate, were supplied exogenously, increasing oxygen consumption 8.8-fold and 5.6-fold (Table 2) in the wild-type and ΔatmA, respectively. The reduced recovery in oxygen consumption in the ΔatmA strain reflects a 40% reduction in complex IV cytochrome c oxidase activity.
Figure 7.
ROS production by ΔatmA and ΔxprG mutant strains. Intracellular ROS levels after 1-hr carbon starvation determined via the oxidant-sensitive probe 5-(and 6)-chloromethyl-2′,7′-dichlorofluorescin diacetate CM-H2DCFDA. AFU, arbitrary fluorescence units.
Figure 1.
The ΔatmA strain has increased mitochondrial mass. (A) Fluorescent microscopy for both wild-type and ΔatmA mitochondria stained with Mito Tracker Green and Nonyl Acridine Orange. (B) Flow cytometric analyses (FCA) for both dyes are shown. The results are expressed as mean ± SD and were considered statistically different (*), with P < 0.05 determined by Student t test using GraphPad Prism software version 5 (GraphPad Software). FAU, fluorescent arbitrary units. (C) Western blot (upper panel) of the cytochrome c of the total proteins (lower panel) extracted from the wild-type and ΔatmA strains grown for 16 hr in MM.
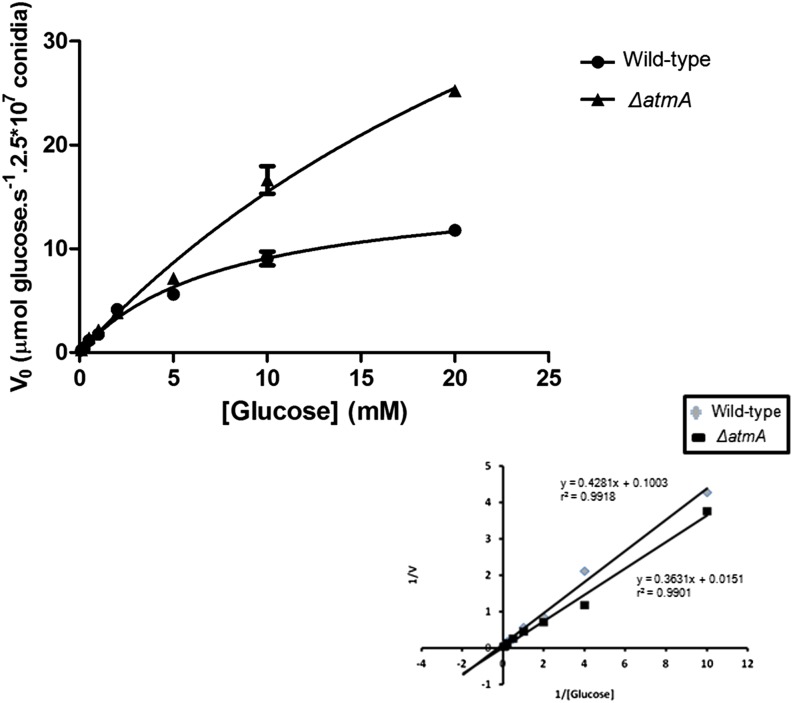
Mitochondrial dysfunction could result in a reduced ability to uptake and utilize glucose. The absence of atmA was subsequently revealed to impact glucose uptake. Typical Michaelis-Menten saturation kinetics for glucose uptake was observed for both the wild-type and ΔatmA strains (Figure 2). The wild-type strain showed a Km of 7.83 µM and Vmax of 14.96 ± 1.25 µM glucose s−1, whereas the ΔatmA strain demonstrated Km values of 36.76 µM, and Vmax value of 60.87 ± 11.48 µM µM glucose s−1, per 2.5×107 conidia. The higher Km and Vmax for the ΔatmA strain indicated a reduced ability to uptake glucose. Collectively, these results strongly indicate that AtmA is important for mitochondrial function and influences aerobic respiration via the cytochrome c oxidase and glucose uptake and/consumption.
Figure 2.
Glucose uptake is impaired in the ΔatmA mutant strain. Km values for glucose in the A. nidulans wild-type and ΔatmA mutant strains. Uptake rates for [14C] glucose germinating conidia of the wild-type and ΔatmA mutant strains were determined at the indicated substrate concentrations at pH 7.0. (n = 3; ±SD).
Transcriptional profiling of carbon-starved and nonstarved cultures elucidated a role for AtmA during starvation
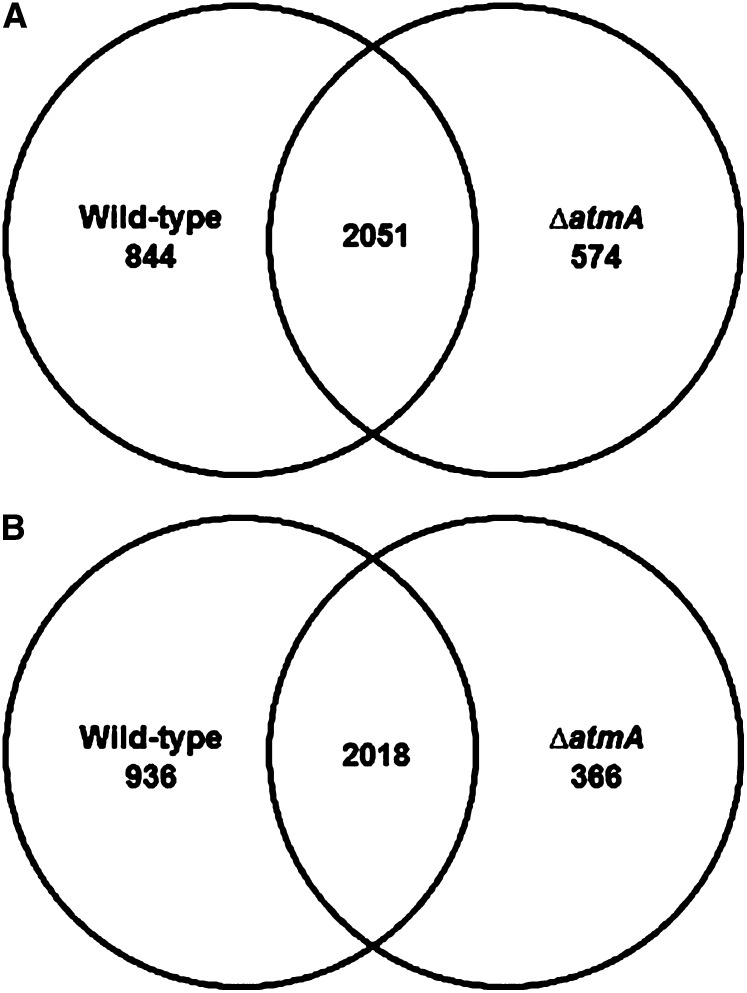
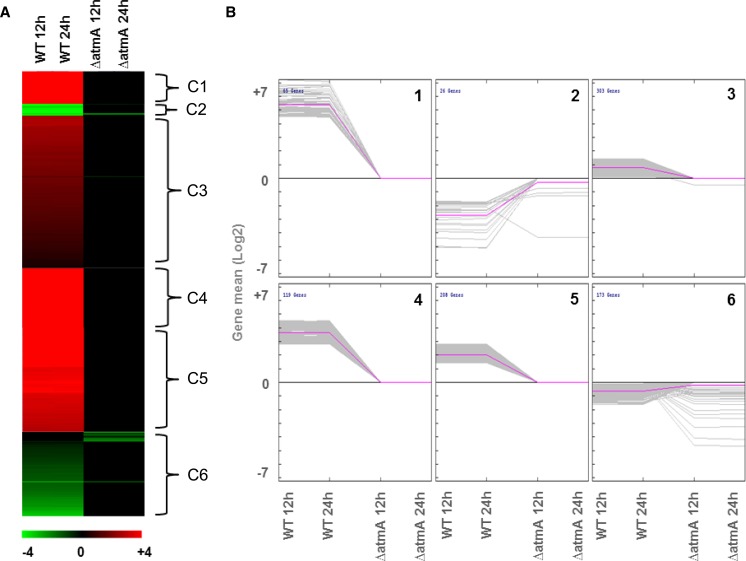
Genome-wide transcriptional profiling of the ΔatmA and wild-type strains in the presence and absence of a carbon source was undertaken to investigate the role of AtmA during aerobic respiration and the carbon starvation response. The two strains were grown for 24 hr in MM plus 2% glucose and then transferred to MM without any carbon source for 12 hr and 24 hr. The data set was deposited at http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE42732. A very high genome-wide correlation in gene expression was observed between the two time points (R > 0.95) within the individual strains. Subsequently, the time points were combined for further analyses. In the wild-type strain, 2895 and 2954 genes were upregulated or downregulated, whereas 2625 and 2384 genes were upregulated or downregulated in the ΔatmA strain, after starvation (Figure S1). The genes specifically modulated in an individual strain were identified (Table S2). The majority of the carbon starvation transcriptional responses were conserved between the two strains (Figure 3). The majority of significant differences between the two strains were represented by an absence of a transcriptional response to carbon starvation in the ΔatmA strain, predominantly gene induction (Figure 4 and Table S2).
Figure 3.
Venn diagrams of the wild-type and ΔatmA transcriptomes. The overlap of genes exhibiting a statistically significant (P < 0.001) increase (A) or decrease (B) in expression after carbon starvation compared to the equivalent transcriptomes when grown on glucose containing media.
Figure 4.
Expression pattern of the genes differentially expressed between the wild-type and ΔatmA strains after carbon starvation. (A) Heat map of the six gene clusters (C1 to C6) identified via KMC analysis as demonstrating a similar expression pattern. (B) Expression profile (mean log2 fold change) of the genes within the six clusters.
All the biological processes, cellular components, and molecular functions (GO terms) overrepresented in the transcriptional response to carbon starvation in the wild-type and ΔatmA strains were identified (Table S3 and Table S4). In both strains, carbon starvation resulted in a downregulation of mitochondrial functions and aerobic respiration, whereas an upregulation of autophagy, vesicle transport, and protein targeting to the vacuole. However, strain-specific responses represented the focus of this study. Wild-type strain–specific responses included an upregulation of TOR signaling, the actin cytoskeleton, and asexual reproduction (Table 3A and Figure 4). Apart from TOR, the other signaling components identified included the S. cerevisiae Sit4, Tap42, and Lst8 homologs (AN0120, AN0504, AN1335) and the heat-shock transcription factor Hsf1 homolog (AN8035). Conversely, there was a wild-type–specific downregulation of the microtubule cytoskeleton, ion transport, ribosomal biogenesis, and RNA processing (Table 3B and Figure 4).
Table 3. The GO terms specifically overrepresented in the list of genes significantly upregulated or downregulated in the wild-type strain after carbon starvation.
| GO Term | Description | No. Genes | P | |
|---|---|---|---|---|
| A | 0019954 | Asexual reproduction | 37 | 0.0016 |
| 0031929 | TOR signaling cascade | 7 | 0.0015 | |
| 0030036 | Actin cytoskeleton organization | 24 | 0.0033 | |
| 0043687 | Posttranslational protein modification | 71 | 0.0006 | |
| 0043938 | Positive regulation of sporulation | 12 | 0.0034 | |
| 0045461 | Sterigmatocystin biosynthetic process | 15 | 1.6e-06 | |
| 0030427 | Site of polarized growth | 48 | 0.0016 | |
| 0000145 | Exocyst | 5 | 0.0051 | |
| 0016787 | Hydrolase activity | 183 | 8.3e-05 | |
| 0044264 | Polysaccharide metabolic process | 29 | 0.0031 | |
| B | 0006879 | Cellular iron ion homeostasis | 15 | 0.0062 |
| 0000226 | Microtubule cytoskeleton organization | 25 | 0.0066 | |
| 0007017 | Microtubule-based process | 31 | 0.0045 | |
| 0033753 | Establishment of ribosome localization | 17 | 0.0034 | |
| 0000054 | Ribosomal subunit export from nucleus | 17 | 0.0034 | |
| 0042254 | Ribosome biogenesis | 124 | 7.1e-14 | |
| 0006396 | RNA processing | 138 | 5e-08 | |
| 0032543 | Mitochondrial translation | 18 | 7e-06 | |
| 0006412 | Translation | 70 | 1.3e-05 | |
| 0015101 | Cation membrane transporter activity | 7 | 0.0010 |
The ΔatmA strain–specific responses to carbon starvation included an upregulation of genes involved in secretion, the CVT pathway, and ER-to-golgi transport [Table 4A, Figure 4 (clusters C2 and C6), Table S2, and Table S3]. However, existence of a CVT pathway in filamentous fungi is still a controversial topic (Kershaw and Talbot 2009; Talbot and Kershaw 2009; Yanagisawa et al. 2013). In addition, there was an upregulation of pre-autophagosomal structures, G1/S transition, the amino acid starvation response, and fatty acid oxidation [Table 4A;, Figure 4 (clusters 2 and C6), Table S2, and Table S3). The ΔatmA-specific downregulation of glucose catabolic processes was also observed in addition to the downregulation of multiple mitochondrial components, such as the mitochondrial proton transporting ATP synthase, NADH dehydrogenase/oxidoreductase, respiratory chain complex subunits, heme biosynthetic processes, and the pyruvate dehydrogenase complex [Table 4B (cluster C1, C3 to C5), Table S2, and Table S3]. The described set of functions comprehensively demonstrated the downregulation of glucose utilization and mitochondrial oxidative phosphorylation in the absence of AtmA in concordance with the reduced ability of the ΔatmA strain to uptake glucose and consume oxygen.
Table 4. The GO terms specifically overrepresented in the list of genes significantly upregulated or down regulated in the ΔatmA strain after carbon starvation.
| GO Term | Description | No. Genes | P | |
|---|---|---|---|---|
| A | 0046903 | Secretion | 13 | 0.0010 |
| 0034198 | Cellular response to amino acid starvation | 6 | 0.0012 | |
| 0032258 | CVT pathway | 10 | 0.0001 | |
| 0000082 | G1/S transition of mitotic cell cycle | 11 | 0.0019 | |
| 0019395 | Fatty acid oxidation | 7 | 0.0003 | |
| 0006887 | Exocytosis | 12 | 0.0014 | |
| 0009063 | Cellular amino acid catabolic process | 21 | 0.0007 | |
| 0030134 | ER to Golgi transport vesicle | 15 | 0.0003 | |
| 0005773 | Vacuole | 43 | 0.0007 | |
| 0031982 | Vesicle | 29 | 0.0010 | |
| 0042175 | Nuclear outer membrane–endoplasmic reticulum membrane network | 31 | 0.0015 | |
| 0005789 | Endoplasmic reticulum membrane | 29 | 0.0027 | |
| 0000407 | Pre-autophagosomal structure | 7 | 0.0031 | |
| 0000139 | Golgi membrane | 15 | 0.0068 | |
| B | 0006122 | Mitochondrial electron transport, ubiquinol to cytochrome c | 6 | 0.0042 |
| 0006783 | Heme biosynthetic process | 9 | 0.0019 | |
| 0015986 | ATP synthesis coupled proton transport | 10 | 0.0019 | |
| 0006007 | Glucose catabolic process | 17 | 0.0026 | |
| 0019674 | NAD metabolic process | 9 | 0.0046 | |
| 0033615 | Mitochondrial proton-transporting ATP synthase complex assembly | 5 | 0.0027 | |
| 0045039 | Protein import into mitochondrial inner membrane | 6 | 0.0042 | |
| 0042719 | Mitochondrial intermembrane space protein transporter complex | 4 | 0.0087 | |
| 0005750 | Mitochondrial respiratory chain complex III | 6 | 0.0042 | |
| 0045275 | Respiratory chain complex III | 6 | 0.0042 | |
| 0000275 | Mitochondrial proton-transporting ATP synthase complex, catalytic core F(1) | 4 | 0.0087 | |
| 0005751 | Mitochondrial respiratory chain complex IV | 6 | 0.0042 | |
| 0045254 | Pyruvate dehydrogenase complex | 4 | 0.0086 | |
| 0045277 | Respiratory chain complex IV | 6 | 0.0042 | |
| 0045259 | Proton-transporting ATP synthase complex | 10 | 0.0019 | |
| 0016651 | Oxidoreductase activity, acting on NADH or NADPH | 18 | 0.0001 | |
| 0050136 | NADH dehydrogenase (quinone) activity | 9 | 0.0007 | |
| 0016655 | Oxidoreductase activity, acting on NADH or NADPH, quinone as acceptor | 10 | 0.0002 | |
| 0008137 | NADH dehydrogenase (ubiquinone) activity | 9 | 0.0007 | |
| 0016491 | Oxidoreductase activity | 140 | 1.9e-07 | |
| 0046961 | Proton-transporting ATPase activity, rotational mechanism | 10 | 0.0002 | |
| 0003954 | NADH dehydrogenase activity | 9 | 0.0007 | |
| 0008121 | Ubiquinol–cytochrome-c reductase activity | 7 | 0.0014 | |
| 0016681 | Oxidoreductase activity, acting on diphenol, cytochrome as acceptor | 7 | 0.0014 |
The reduced ability of the ΔatmA strain to utilize glucose was accompanied by an absence of wild-type upregulation in alternative carbon utilization, such as polysaccharides and fatty acids (Table 3, A and B, and Figure 4). The absence of starvation-induced hydrolases transcription, particularly xylosidases, in the ΔatmA strain demonstrated the role played by AtmA in the induction of hydrolase secretion, possibly scavenging for any available carbon source (Figure S2).
Collectively, the transcriptomic analysis demonstrated the differential sensing of nutrition/cellular energetic status, plus a reduction in glucose utilization and respiration. A role for AtmA in the carbon starvation–induced transcription of hydrolases also was identified.
Autophagy is decreased in ΔatmA mutant strain
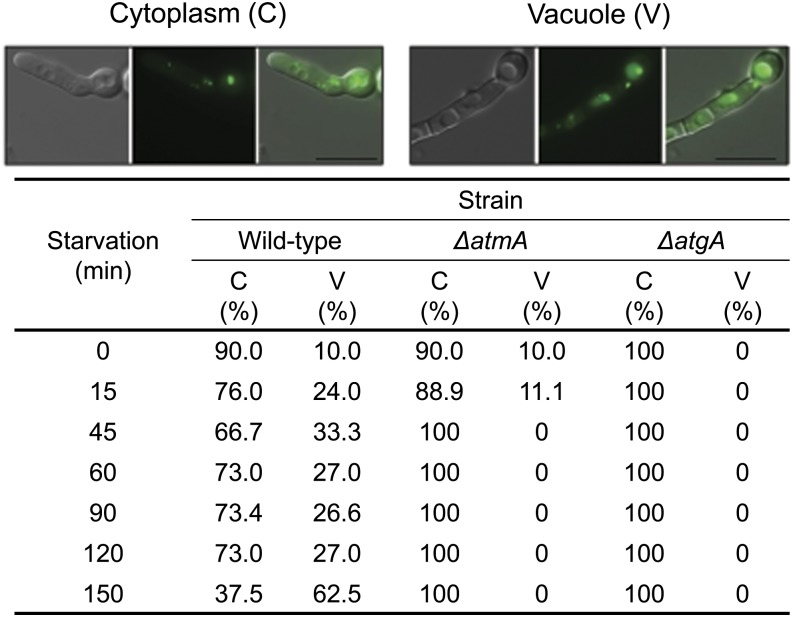
Autophagy forms an integral part of the carbon starvation response in fungi. Transcriptomic analyses showed that autophagy was significantly induced in both the wild-type and ΔatmA strains after carbon starvation. However, multiple autophagic processes, such as TOR signaling, the CVT pathway, and pre-autophagosomal structures, were differentially expressed in a strain-specific manner. Subsequently, the influence of AtmA on carbon starvation–induced autophagy in A. nidulans was evaluated. The cellular localization of AtgHATG8::GFP, which is recruited to autophagosomes and transported to the vacuole in response to starvation, was monitored in the wild-type and ΔatmA strains. A ΔatgAATG1 AtgHATG8::GFP double mutant, which also lacked the protein kinase required for vesicle formation during autophagy and the CVT pathway, was utilized as a negative control.
Initially, to confirm that the atg1 homolog in A. nidulans, AtgA, was essential for autophagy, the translocation of AtgH::GFP to the vacuole upon carbon starvation in the ΔatgAATG1 atgHATG8::GFP double mutant was assessed. The atgHATG8::GFP strain was used as a positive control. Germlings were grown for 12 hr in 2% glucose and then transferred to MM without any carbon source for 15 to 150 min (Figure 5). After 12 hr of growth in the presence of glucose, AtgH::GFP localized to pre-autophagosomal-like structures and in the cytoplasm (Figure 5). After 150 min of starvation, approximately 60% of germlings contained punctuate fluorescent spots that localize to the vacuole (Figure 5). In the ΔatgAATG1 atgHATG8::GFP strain, the AtgH::GFP signal did not localize to the vacuoles after 150 min (Figure 5). These results indicate that in A. nidulans AtgH translocation to the vacuole on carbon starvation is AtgA-dependent.
Figure 5.
The atmA positively controls autophagy. A. nidulans AtgH::GFP germlings previously grown for 12 hr in MM plus 2% glucose were transferred to MM without any carbon source for 15, 30, and 60 min. Bars: 10 µm. The AtgH::GFP, ΔatmA AtgH::GFP, and ΔatgA AtgH::GFP grown for 12 hr in MM plus 2% glucose (time zero) and transferred to MM without any carbon source for 15, 45, 60, 90, 120, and 150 min. Bars: 10 µm.
Subsequently, the translocation of AtgH::GFP to the vacuole upon carbon starvation in the ΔatmA background was evaluated. AtgH::GFP translocation to the vacuoles during starvation was dramatically reduced in the absence of AtmA (Figure 5). Interestingly, before starvation, a low level of AtgH::GFP localized in the vacuole similar to that of the wild-type strain. However, during the prolonged periods of starvation vacuolar localization was lost, reminiscent of the ΔatgAATG1 strain, suggesting that under nonstarving conditions AtmA does not control AtgH localization. Therefore, under carbon starvation, autophagy appears to be regulated by AtgAATG1 and AtmA.
A. nidulans AtmA interacts genetically with XprG on carbon starvation
A. nidulans secretes proteases in an XprG-dependent manner during carbon starvation irrespective of the presence of proteins (Katz et al. 1996). Protease secretion was assessed via determining the size of a clearance zone surrounding the fungal colony on agar plates that contained milk as a sole carbon source (Figure 6A) and the clearance index (CI; clearance zone diameter / colony diameter) was calculated. The single ΔxprG and double ΔatmA ΔxprG mutant strains were confirmed to have a diminished clearance zone (CIs undetermined). The ΔatmA strain, however, demonstrated increased secretion or protease activity (CI, 1.58) compared to the wild-type strain (CI, 1.06), despite the fact that as a class of proteins overall proteolytic enzyme transcription was not comparatively increased in the ΔatmA strain. However, difference in individual protease transcription could have been sufficiently contributed to the observed phenotype. Similarly, the xprG1 gain-of-function mutation demonstrated increased protease activity(CI,1.28). Interestingly, the double ΔatmA xprG1 mutant produced a larger clearance zone (CI, 2.10) than that of either the ΔatmA or xprG1 single mutants.
Figure 6.
AtmA genetically interacts with XprG. (A) The semi-quantitative determination of protease secretion after growth on MM plus milk as the sole carbon source for the wild-type, ΔatmA, ΔxprG, xprG1, ΔatmA ΔxprG, and ΔatmA xprG1 mutant strains. (B) Quantitative evaluation of protease activity of 48-hr carbon-starved cultures for the wild-type, ΔatmA, ΔxprG, xprG1, ΔatmA ΔxprG, and ΔatmA xprG1 mutant strains. (C) Left panel shows real-time RT-PCR for the xprG gene. The wild-type and alcA::xprG mutant strains were grown in liquid MM containing either 2% glycerol and 100 mM threonine or 4% glucose for 24 hr at 37°C. Right panel shows the semi-quantitative determination of protease secretion after growth on MM plus milk as the sole carbon source supplemented with 10 mM cyclopentanone for the wild-type, alcA::atmA, and ΔatmA alcA::xprG mutant strains. The numbers on the left bottom side of each panel in (A) and in (B) represent the clearance index (clearance zone diameter/ colony diameter).
To clarify this genetic relationship, we utilized a more quantitative method to assess protease activity. Because the specificity of the extracellular proteases was not known, we used a synthetic FRET peptide library Abz (or MCA)-GXXXXXQ-EDDnp and Abz (or MCA)-GXXZXXQ-EDDnp. For all strains, no significant differences were seen in the extracellular protease activity in three different pH conditions tested (pH 4.0, 7.0, and 8.5) (Figure 6B). Both the ΔxprG and ΔatmA ΔxprG mutant strains demonstrated comparable protease activity to the wild-type strain, whereas both the ΔatmA and xprG1 demonstrated approximately twice the protease activity (Figure 6B). The protease activity was six-fold to seven-fold higher in the ΔatmA xprG1 double mutant (Figure 6B). Therefore, the FRET assay corroborated the previous result observed using the CI.
We also constructed an alcA::xprG conditional mutant for xprG by replacing the xprG promoter with the alcA promoter. The alcA promoter is repressed by glucose, derepressed by glycerol, and induced to high levels by ethanol, L-threonine, or cyclopentanone (Flipphi et al. 2002). Transformants were selected that accumulated approximately 13-fold higher xprG mRNA when transferred to glycerol 2% plus threonine than when transferred to glucose 4% (Figure 6C, left panel). An increased clearance zone around the alcA::xprG mutant colony was observed when grown on agar plates containing milk supplemented with 10 mM cyclopentanone (CI, 1.26) (Figure 6C, right panel). Again, to examine the possible genetic interactions between atmA and xprG, a double ΔatmA alcA::xprG mutant was constructed. The clearance zone of the ΔatmA alcA::xprG strain (CI, 1.90) (Figure 6C, right panel) was larger than that produced by the single alcA::xprG mutant. These results imply that AtmA performs a role in the inhibition of XprG.
The accumulation of ROS in the ΔatmA strain, when grown in the presence of glucose or after carbon starvation, was twice that observed for the wild-type strain (Figure 7). Accumulation of ROS in strains lacking xprG was moderately less than that of the wild-type strain, whereas the gain-of-function xprG1 strain demonstrated ROS levels comparable to the ΔatmA strain. Similar to the protease results previously presented, the combination of the ΔatmA and xprG1 mutations in a single strain demonstrated an additive effect and increased ROS accumulation to the highest levels. These results suggest that under these conditions, XprG is epistatic and is needed for the increased production of ROS caused by the loss of AtmA.
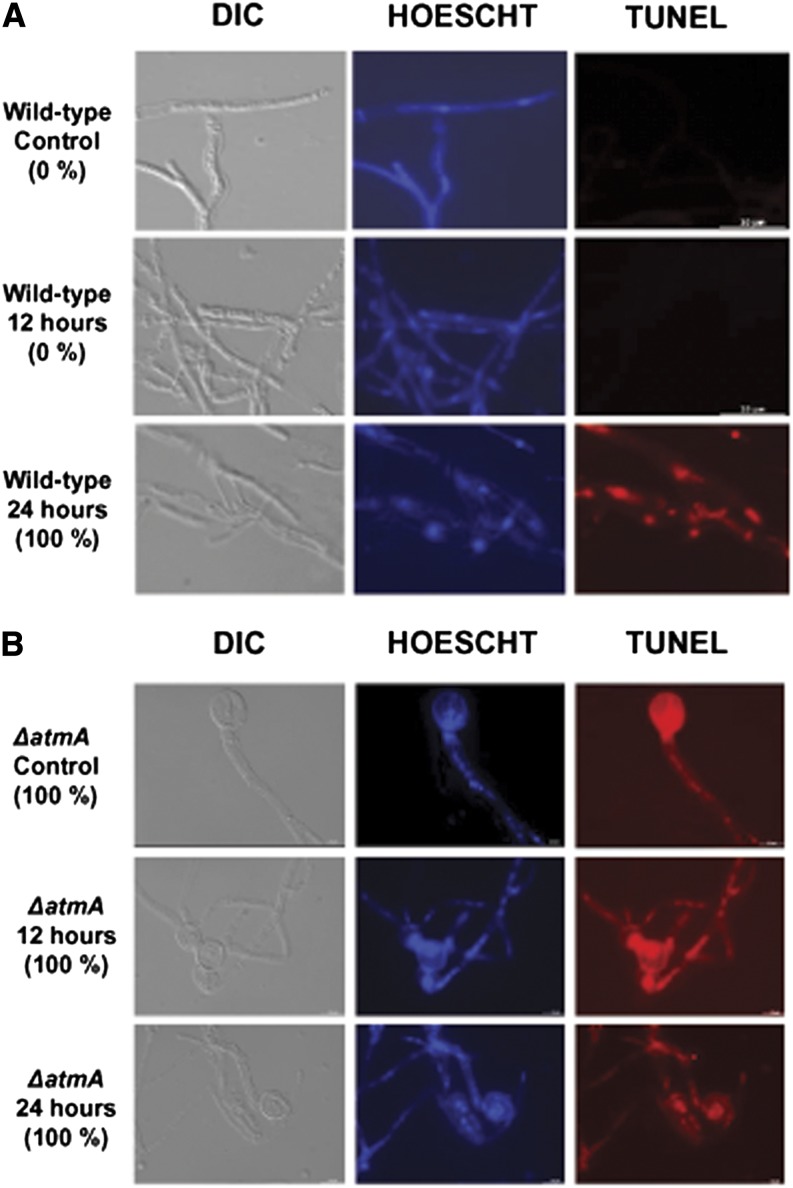
Although the ΔatmA mutant is viable, it shows nuclei with considerable DNA fragmentation (Malavazi et al. 2006, 2008). The AtmAATM loss-of-function caused synthetic lethality when combined with mutation in UvsBATR, suggesting that AtmA and UvsB interact and are probably partially redundant in terms of DNA damage sensing or repairing and polar growth (Malavazi et al. 2008). This functional redundancy could help to explain why ΔatmA mutant is viable despite all the DNA fragmentation. Thus, a combination of TUNEL and propidium iodide (PI) staining, which respectively identified DNA fragmentation or a loss of cell membrane integrity, was utilized to monitor apoptosis and necrosis upon carbon starvation (Figure 8 and Figure 9, respectively). Before 24-hr carbon starvation, the wild-type strain showed no signs of apoptosis or necrosis. However, after 24-hr starvation, 100% and 70% of wild-type hyphae demonstrated positive TUNEL and PI staining, respectively. In contrast, all nonstarved or starved ΔatmA hyphae stained positively with TUNEL, whereas 80% and 100% of hyphae stained positively with PI under nonstarved and starved conditions, respectively. The ΔxprG strain showed no TUNEL or PI staining in nonstarved or starved hyphae. The ΔatmA ΔxprG double mutant demonstrated no staining for the nonstarved culture and approximately 50% TUNEL and PI nuclear staining after starvation. Collectively, these data demonstrate that under starvation, AtmA inhibits XprG-dependent and XprG-independent forms of inducing cell death.
Figure 8.
TUNEL assay to detect DNA fragmentation in the wild-type and ΔatmA strains evaluating the influence of 0, 12, and 24 hr of starvation. Nuclei were visualized by Hoescht staining. (A) Wild-type and (B) ΔatmA mutant strains. Bars: 10 µm.
Figure 9.
Detection of necrosis using propidium iodide (PI) staining for wild-type, ΔatmA, and ΔxprG strains after 0, 12, and 24 hr of starvation. Nuclei were visualized by Hoescht staining. (A) Wild-type, (B) ΔatmA, and (C) ΔxprG. Bars: 10 µm.
Discussion
Mitochondria supply cellular energy but also perform a role in the adaptation to stress and the cross-talk between prosurvival and prodeath pathways. ATM phosphorylates more than 700 proteins, including many downstream kinases (Matsuoka et al. 2007) demonstrating a broad range of functions. ATM is known for its role in the DNA damage response and cell cycle in mammalian and fungal cells (Ditch and Paull 2012; Malavazi et al. 2006, 2007). ATM also has recently been shown to be the redox sensor that controls mitochondrial and metabolic function through the action of the downstream kinases AMPK and mTOR (Ditch and Paull 2012). Additionally, Atm-deficient mice cells revealed increased mitochondrial mass, altered morphology, and deficiencies in mitochondrial electron transport chain activity correlating to impaired ATP levels and elevated mitochondrial ROS (Valentin-Vega and Kastan 2012). In the presented study of A. nidulans, the fungal ATM homolog was shown to control mitochondrial mass and function, which directly or indirectly influenced glucose uptake while also influencing cell death in response to carbon starvation. Moreover, the increased mitochondrial mass in ΔatmA cells may not result from changes in mitochondrial biogenesis, but rather from defects in the selective clearance of damaged mitochondria because autophagy is also affected in this strain, in agreement with the work of Valentin-Vega and Kastan (2012). Subsequently, transcriptomic and genetic studies were utilized to elucidate the role played by AtmA during starvation.
The retrograde response signals mitochondrial dysfunction to the nucleus, reconfiguring expression, including an induction of gluconeogenesis, the glyoxylate cycle, and glutamate biosynthesis (Jazwinski 2013). The intrinsic induction of cell death, caused by energetic failure, oxidative stress, and lipid metabolism abnormalities, is mainly regulated by the mitochondria and involves the stress-mediated release of cytochrome C (Zdralevic et al. 2012). Inhibition of TOR can induce autophagy and the retrograde response (Zdralevic et al. 2012). Increased glyoxylate cycle activity occurs in S. cerevisiae during severe nutrient limitation (Wang et al. 2010). Fatty acid β-oxidation, which is required to generate acetate to fuel the glyoxylate cycle, was shown to be regulated by AtmA during starvation, suggesting that AtmA performs a role in the retrograde response. In addition, during starvation, TOR and the S. cerevisiae Sit4, Tap42, and Lst8 homologs (AN0120, AN0504, AN1335) were shown to be directly or indirectly influenced by AtmA. In S. cerevisiae Sit4 and Tap42, which associate with the protein phosphatase 2A, are regulated by TOR and control G1/S cell-cycle transition, whereas Lst8 is a known retrograde response regulator (Jazwinski 2013; Zdralevic et al. 2012). The homolog of the Hsf1 heat-shock transcription factor (AN8035), which is hyperphosphorylated by Snf1 (fungal AMPK homolog) in carbon-starved S. cerevisiae cells (Hahn and Thiele 2004), was also influenced by AtmA. Therefore, in response to metabolic stress, AtmA may perform a role in the regulation of TOR signaling that involves the retrograde response and the SnfA pathways. This hypothesis is supported by the observation that the ΔatmA strain does not successfully undergo autophagy during carbon starvation, a process known to be controlled by TOR. This suggests that a link between AtmA, SnfA, and TOR may also exist in A. nidulans, as observed in mammalian cells.
Recently, AtmA has been demonstrated to perform a role in the regulation of hydrolytic enzyme production (Brown et al. 2013). However, nuclear localization of the carbon catabolite repressor, CreA, in the ΔatmA strain was similar to the parental strains (Brown et al. 2013). On carbon starvation, A. nidulans induces the transcription of alternative carbon source usage genes, including carbohydrate active enzymes (CAZy), a response that is dependent on the SnfA and SchA (Brown et al. 2013). The absence of AtmA resulted in a loss of starvation-induced CAZy enzyme induction. Therefore, AtmA forms part of a starvation response that influences hydrolytic enzyme transcription and secretion while not being involved in CreA derepression. This supports the hypothesis that other pathways for SnfA and SchA induction exist.
Autophagy is a highly conserved process in eukaryotic cells and occurs in response to a diverse array of conditions, including nutrient deprivation (Martinet et al. 2006; Mizushima and Klionsky 2007). Carbon-starved A. nidulans downregulated mitochondrial functions and aerobic respiration while upregulating autophagy, vesicle transport, and protein targeting to the vacuole. A dramatic reduction in autophagy was observed in the ΔatmA strain, strongly suggesting that AtmA positively regulates autophagy. Autophagy forms part of a starvation-induced prosurvival strategy, which is inhibited by the action of TOR, PKA, and Sch9 on the Atg1 kinase in S. cerevisiae (Kamada et al. 2000; Yorimitsu et al. 2007). In A. nidulans, AtgAATG1 was confirmed to be essential for the induction of autophagy, whereas the absence of AtmA resulted in the loss of AtgHATG8 vacuole localization and impaired autophagy. Therefore, the reduction in autophagy in the ΔatmA strain could have contributed to the faster rate of starvation-induced cell death.
In mammalian cells, p53 is a multi-functional protein that simultaneously regulates distinct downstream pathways controlling antioxidant defense, cell-cycle progression, mitochondrial respiration, glucose homeostasis, and apoptosis (Ko and Prives 1996; Levine 1997; Armata et al. 2010). Different residues of the p53 protein are phosphorylated by ATM on exposure to distinct stimuli (Bensimon et al. 2011; Ditch and Paull 2012). In response to DNA damage and during the regulation of glucose homeostasis, ATM phosphorylates p53, resulting in its activation and an increase in protein stability (Shiloh 2006; Armata et al. 2010). In mammalian cells, p53-induced autophagy has been reported (Inoki et al. 2003; Zhao and Klionsky 2011). In A. nidulans, autolysis and starvation-induced protease secretion are controlled by the p53-like transcription factor XprG (Katz et al. 2006, 2009). The present study suggests that AtmA regulated the XprG-dependent starvation-induced protease secretion and cell death.
ROS act as signaling molecules and cellular toxicants. Mitochondrial dysfunction or the impairment of oxidative phosphorylation results in ROS accumulation (Barros et al. 2004). A shift in the balance between oxidants/antioxidants in the direction of oxidation contributes to the induction of apoptosis (Liu et al. 2008; Giorgio et al. 2007). In mammalian cells, p53 mediates antioxidant pathways and thereby protects against endogenous ROS. After stress, high levels of p53 lead to a shift in oxidant/antioxidant balance, which can result in apoptotic cell death (Kang et al. 2013). Atm-deficient mammalian cells demonstrate increased ROS levels and oxidative stress (Barlow et al. 1999; Gatei et al. 2001; Kamsler et al. 2001), with both conditions resulting in the phosphorylation of p53 at the ATM site (Armata et al. 2010). In A. nidulans, AtmA negatively regulated XprG-dependent ROS accumulation. Therefore, the absence of AtmA or XprG would result in faster and slower rates in ROS-mediated cell death in the respective single-gene deletion strains. This suggests that AtmA negatively regulates XprG, as demonstrated by the increase in XprG-dependent secretion of proteases, accumulation of ROS, and cell death. This concept is supported by the observed higher level of protease secretion and ROS accumulation in both the ΔatmA xprG1 (gain-of-function) and the ΔatmA alcA::XprG overexpression double mutant strains. The interaction between ATM and p53 has been extensively studied in mammalian systems, in which the interaction between ATM and p53 regulates the G1/S cell-cycle checkpoint, apoptosis, or senescence through p53 phosphorylation at Ser15 residue (Barlow et al. 1997; Banin et al. 1998; Bartkova et al. 2006; Canman et al. 1998; Zhan et al. 2010). The mammalian p53 has four ATM-dependent phosphorylation sites (Saito et al. 2002; Armata et al, 2010; File S1). The A. nidulans XprG only has two serine residues at positions 18 and 46 (File S1). The functionality of these residues in XprG and if AtmA phosphorylates them remains to be determined.
Mitochondria are crucial to metabolism, cell-cycle progression, signaling, and apoptosis. One of the striking aspects of the ATM-dependent pathology is that the mitochondria leads to inefficient respiration and energy metabolism plus the increased generation of free radicals that are able to create life-threatening DNA lesions (Ambrose and Gatti 2013). We have observed an apparent discrepancy between the increased mitochondrial copy number in the A. nidulans ΔatmA mutant and defects in glucose uptake and oxygen consumption in this mutant. Valentin-Vega et al. (2012) have shown that Atm loss also leads to an elevated mitochondrial mass. These authors investigated the dynamics of this organelle and suggested that the increase in mitochondrial mass in Atm-null cells does not result from changes in mitochondrial biogenesis, but rather from defects in the selective clearance of damaged mitochondria by autophagy (mitophagy). We observed decreased autophagy in the A. nidulans ΔatmA mutant, therefore it is possible the increased mitochondrial mass in this mutant would reflect a decreased mitophagy as observed in mammalian Atm cells. In response to severe carbon limitation, the retrograde response communicates mitochondrial dysfunction to the cell nucleus, resulting in stress adaptations. The mammalian ATM kinase acts as a redox sensor, coordinating such stress responses via the mTOR, AMPK, and p53 pathways. In A. nidulans, AtmA also regulated glucose utilization and mitochondrial oxidative phosphorylation. Carbon starvation responses, including autophagy, shifting metabolism to the glyoxylate cycle and the secretion of carbon scavenging enzymes were AtmA-dependent. Thus, AtmA may represent a link between mitochondrial function and metabolic output with cell cycle and growth, possibly through the influence of the TOR and XprG signaling.
Supplementary Material
Acknowledgments
We acknowledge Dr. Margaret Katz (University of New England, Australia) for providing the A. nidulans ΔxprG and xprG1 mutant strains, Dr. Mark Marten (University of Maryland, Baltimore County, Baltimore, MD) for providing the A. nidulans AtgH::GFP mutant, and the anonymous reviewers for their comments and suggestions. We thank FAPESP (Fundação para Amparo a Pesquisa do Estado de São Paulo) and CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) for financial support.
Footnotes
Communicating editor: J. C. Dunlap
Literature Cited
- Alexander A., Kim J., Walker C. L., 2010a ATM engages the TSC2/mTORC1 signaling node to regulate autophagy. Autophagy 6: 672–673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ambrose M., Gatti R. A., 2013. Pathogenesis of ataxia-telangiectasia: the next generation of ATM functions. Blood 121: 4036–4045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armata H. L., Golebiowski D., Jung D. Y., Ko H. J., Kim J. K., et al. , 2010. Requirement of the ATM/p53 tumor suppressor pathway for glucose homeostasis. Mol. Cell. Biol. 30: 5787–5794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banin S., Shieh S., Taya Y., Anderson C. W., Chessa L., et al. , 1998. Enhanced phosphorylation of p53 by ATM in response to DNA damage. Science 281: 1674–1677. [DOI] [PubMed] [Google Scholar]
- Barbet N. C., Schneider U., Helliwell S. B., Stansfield I., Tuite M. F., et al. , 1996. TOR controls translation initiation and early G1 progression in yeast. Mol. Biol. Cell 7: 25–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barlow C., Brown K. D., Deng C.-X., Tagle D. A., Wynshaw-Boris A., 1997. Atm selectively regulates distinct p53-dependent cell-cycle checkpoint and apoptotic pathways. Nat. Genet. 17: 453–456. [DOI] [PubMed] [Google Scholar]
- Barlow C., Dennery P. A., Shigenaga M. K., Smith M. A., Morrow J. D., et al. , 1999. Loss of the ataxiatelangiectasia gene product causes oxidative damage in target organs. Proc. Natl. Acad. Sci. USA 96: 9915–9919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barros M. H., Bandy B., Tahara E. B., Kowaltowski A. J., 2004. Higher respiratory activity decreases mitochondrial reactive oxygen release and increases lifespan in Saccharomyces cerevisiae. J. Biol. Chem. 279: 49883–49888. [DOI] [PubMed] [Google Scholar]
- Bartkova J., Rezaei N., Liontos M., Karakaidos P., Kletsas D., et al. , 2006. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature 444: 633–637. [DOI] [PubMed] [Google Scholar]
- Bensimon A., Aebersold R., Shiloh Y., 2011. Beyond ATM: The protein kinase landscape of the DNA damage response. FEBS Lett. 585: 1625–1639. [DOI] [PubMed] [Google Scholar]
- Boder E., Sedgwick R. P., 1958. Ataxia-telangiectasia: a familial syndrome of progressive cerebellar ataxia, oculocutaneous telangiectasia and frequent pulmonary infection. Pediatrics 21: 526–554. [PubMed] [Google Scholar]
- Brown NA, P. F. de Gouve, N. G. Krohn, M. Savoldi, and G. H. Goldman, 2013. Functional characterisation of the non-essential protein kinases and phosphatases regulating Aspergillus nidulans hydrolytic enzyme production. Biotechnol. Biofuels 6: 91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bursch W., Karwan A., Mayer M., Dornetshuber J., Fröhwein U., et al. , 2008. Cell death and autophagy: cytokines, drugs, and nutritional factors. Toxicology 254: 147–157. [DOI] [PubMed] [Google Scholar]
- Busti S., Coccetti P., Alberghina L., Vanoni M., 2010. Glucose signaling-mediated coordination of cell growth and cell cycle in Saccharomyces cerevisiae. Sensors (Basel Switzerland) 10: 6195–6240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Canman C. E., Lim D. S., Cimprich K. A., Taya Y., Tamai K., et al. , 1998. Activation of the ATM kinase by ionizing radiation and phosphorylation of p53. Science 281: 1677–1679. [DOI] [PubMed] [Google Scholar]
- Ching J. K., Rajguru P., Marupudi N., Banerjee S., Fisher J. S., 2010. A role for AMPK in increased insulin action after serum starvation. Am. J. Physiol. Cell Physiol. 299: C1171–C1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clutterbuck, A. J., 1993 Aspergillus nidulans (nuclear genes), pp. 3.71–3.84 in Locus Maps of Complex Genomes: Lower Eukaryotes, Vol. 3, edited by O’Brien, S. J., Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York. [Google Scholar]
- de Castro P. A., Savoldi M., Bonatto D., Barros M. H., Goldman M. H. S., et al. , 2011. Molecular characterization of propolis-induced cell death in Saccharomyces cerevisiae. Eukaryot. Cell 10: 398–411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dechant R., Peter M., 2008. Nutrient signals driving cell growth. Curr. Opin. Cell Biol. 20: 678–687. [DOI] [PubMed] [Google Scholar]
- Dementhon K., Iyer G., Glass N. L., 2006. VIB-1 is required for expression of genes necessary for programmed cell death in Neurospora. Eukaryot. Cell 5: 2161–2173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derheimer F. A., Kastan M. B., 2010. Multiple roles of ATM in monitoring and maintaining DNA integrity. FEBS Lett. 584: 3675–3681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Virgilio C., Loewith R., 2006. Cell growth control: little eukaryotes make big contributions. Oncogene 25: 6392–6415. [DOI] [PubMed] [Google Scholar]
- Dinamarco T. M., Pimentel Figueiredo B. C., Savoldi M., Malavazi I., Soriani F. M., et al. , 2010. The roles played by Aspergillus nidulans apoptosis-inducing factor (AIF)-like mitochondrial oxidoreductase (AifA) and NADH-ubiquinone oxidoreductases (NdeA-B and NdiA) in farnesol resistance. Fungal Genet. Biol. 12: 1055–1069. [DOI] [PubMed] [Google Scholar]
- Ditch S., Paull T. T., 2012. ATM protein kinase and cellular redox signaling: beyond the DNA damage response. Trends Biochem. Sci. 37: 15–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eaton J. S., Lin Z. P., Sartorelli A. C., Bonawitz N. D., Shadel G. S., 2007. Ataxia-telangiectasia mutated kinase regulates ribonucleotide reductase and mitochondrial homeostasis. J. Clin. Invest. 117: 2723–2734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emri T., Molnár Z., Pusztahelyi T., Pócsi I., 2004. Physiological and morphological changes in autolysing Aspergillus nidulans cultures. Folia Microbiol. (Praha) 49: 277–284. [DOI] [PubMed] [Google Scholar]
- Emri T., Molnár Z., Szilágyi M., Pócsi I., 2008. Regulation of autolysis in Aspergillus nidulans. Appl. Biochem. Biotechnol. 151: 211–220. [DOI] [PubMed] [Google Scholar]
- Fantes P. A., Roberts C. F., 1973. Beta-galactosidase activity and lactose utilization in Aspergillus nidulans. J. Gen. Microbiol. 77: 417–486. [DOI] [PubMed] [Google Scholar]
- Fingar D. C., Blenis J., 2004. Target of rapamycin (TOR): an integrator of nutrient and growth factor signals and coordinator of cell growth and cell cycle progression. Oncogene 23: 3151–3171. [DOI] [PubMed] [Google Scholar]
- Flipphi M., Kocialkowska J., Felenbok B., 2002. Characteristics of physiological inducers of the ethanol utilization (alc) pathway in Aspergillus nidulans. Biochem. J. 15: 25–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gatei M., Shkedy D., Khanna K. K., Uziel T., Shiloh Y., et al. , 2001. Ataxia-telangiectasia: chronic activation of damage-responsive functions is reduced by alpha-lipoic acid. Oncogene 20: 289–294. [DOI] [PubMed] [Google Scholar]
- Giorgio M., Trinei M., Migliaccio E., Pelicci P. G., 2007. Hydrogen peroxide: a metabolic by-product or a common mediator of ageing signals? Nat. Rev. Mol. Cell Biol. 8: 722–728. [DOI] [PubMed] [Google Scholar]
- Gray J. V., Petsko G. A., Johnston G. C., Ringe D., Singer R. A., et al. , 2004. ‘Sleeping beauty’: quiescence in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 68: 187–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halaby M. J., Hibma J. C., He J., Yang A. N. D. D. Q., 2008. ATM protein kinase mediates full activation of Akt and regulates glucose transporter 4 translocation by insulin in muscle cells. Cell. Signal. 20: 1555–1563. [DOI] [PubMed] [Google Scholar]
- Hahn J. S., Thiele D. J., 2004. Activation of the Saccharomyces cerevisiae heat shock transcription factor under glucose starvation conditions by Snf1 protein kinase. J. Biol. Chem. 279: 5169–5176. [DOI] [PubMed] [Google Scholar]
- Inoki K., Zhu T., Guan K. L., 2003. TSC2 mediates cellular energy response to control cell growth and survival. Cell 115: 577–590. [DOI] [PubMed] [Google Scholar]
- Jazwinski S. M., 2013. The retrograde response: When mitochondrial quality control is not enough. Biochim. Biophys. Acta 1833: 400–409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kafer E., 1977. Meiotic and mitotic recombination in Aspergilllus and its chromosomal aberrations. Adv. Genet. 19: 33–131. [DOI] [PubMed] [Google Scholar]
- Kamada Y., Funakoshi T., Shintani T., Nagano K., Ohsumi M., et al. , 2000. Tor-mediated induction of autophagy via an Apg1 protein kinase complex. J. Cell Biol. 150: 1507–1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamsler A., Daily D., Hochman A., Stern N., Shiloh Y., et al. , 2001. Increased oxidative stress in ataxia telangiectasia evidenced by alterations in redox state of brains from Atm-deficient mice. Cancer Res. 61: 1849–1854. [PubMed] [Google Scholar]
- Kang M. Y., Kim H.-B., Piao C., Lee K. H., Hyun J. W., et al. , 2013. The critical role of catalase in prooxidant and antioxidant function of p53. Cell Death Differ. 20: 117–129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kastan M. B., Lim D.-S., 2000. The many substrates and functions of ATM. Nat. Rev. Mol. Cell Biol. 1: 179–186. [DOI] [PubMed] [Google Scholar]
- Katz M. E., Flynn P. K., vanKuyk P. A., Cheetham B. F., 1996. Mutations affecting extracellular protease production in the filamentous fungus Aspergillus nidulans. Mol. Gen. Genet. 250: 715–724. [DOI] [PubMed] [Google Scholar]
- Katz M. E., Gray K.-A., Cheetham B. F., 2006. The Aspergillus nidulans xprG (phoG) gene encodes a putative transcriptional activator involved in the response to nutrient limitation. Fungal Genet. Biol. 43: 190–199. [DOI] [PubMed] [Google Scholar]
- Katz M. E., Evans C. J., Heagney E. E., van Kuyk P. A., Kelly J. M., et al. , 2009. Mutations in genes encoding sorting nexins alter production of intracellular and extracellular proteases in Aspergillus nidulans. Genetics 181: 1239–1247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kershaw M. J., Talbot N. J., 2009. Genome-wide functional analysis reveals that infection-associated fungal autophagy is necessary for rice blast disease. Proc. Natl. Acad. Sci. USA 106: 15967–15972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ko L. J., Prives C., 1996. p53: puzzle and paradigm. Genes Dev. 10: 1054–1072. [DOI] [PubMed] [Google Scholar]
- Kourtis N., Tavernarakis N., 2009. Autophagy and cell death in model organisms. Cell Death Differ. 16: 21–30. [DOI] [PubMed] [Google Scholar]
- Langerak P., Russell P., 2011. Regulatory networks integrating cell cycle control with DNA damage checkpoints and double-strand break repair. Philos. Trans. R. Soc. Lond. B Biol. Sci. 366: 3562–3571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavin M. F., Shiloh Y., 1997. The genetic defect in ataxia-telangiectasia. Annu. Rev. Immunol. 15: 177–202. [DOI] [PubMed] [Google Scholar]
- Levine A. J., 1997. p53, the cellular gatekeeper for growth and division. Cell 88: 323–331. [DOI] [PubMed] [Google Scholar]
- Levine B., Kroemer G., 2008. Autophagy in the pathogenesis of disease. Cell 132: 27–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu B., Chen Y., St Clair D. K., 2008. ROS and p53: a versatile partnership. Free Radic. Biol. Med. 44: 1529–1535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malavazi I., Semighini C. P., Kress M. R., Harris S. D., Goldman G. H., 2006. Regulation of hyphal morphogenesis and the DNA damage response by the Aspergillus nidulans ATM homolog AtmA. Genetics 173: 99–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malavazi I., Savoldi M., da Silva Ferreira M. E., Soriani F. M., Bonato P. S., et al. , 2007. Transcriptome analysis of the Aspergillus nidulans AtmA (ATM, ataxia-telangiectasia) null mutant. Mol. Microbiol. 66: 74–99. [DOI] [PubMed] [Google Scholar]
- Malavazi I., Lima J. F., de Castro P. A., Savoldi M., de Souza W., et al. , 2008. Genetic interactions of the Aspergillus nidulans atmAATM homolog with different components of the DNA damage response pathway. Genetics 178: 675–691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinet W., De Meyer G. R., Andries L., Herman A. G., Kockx M. M., 2006. In situ detection of starvation-induced autophagy. J. Histochem. Cytochem. 54: 85–96. [DOI] [PubMed] [Google Scholar]
- Matsumoto K., Uno I., Oshima Y., Ishikawa T., 1982. Isolation and characterization of yeast mutants deficient in adenylate cyclase and cAMP-dependent protein kinase. Proc. Natl. Acad. Sci. USA 79: 2355–2359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuoka S., Ballif B. A., Smogorzewska A., McDonald E. R., III, Hurov K. E., et al. , 2007. ATM and ATR substrate analysis reveals extensive protein networks responsive to DNA damage. Science 316: 1160–1166. [DOI] [PubMed] [Google Scholar]
- Mizushima N., Klionsky D. J., 2007. Protein turnover via autophagy: implications for metabolism. Annu. Rev. Nutr. 27: 19–40. [DOI] [PubMed] [Google Scholar]
- Mizushima N., Levine B., Cuervo A. M., Klionsky D. J., 2008. Autophagy fights disease through cellular self-digestion. Nature 451: 1069–1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nayak T., Szewczyk E., Oakley C. E., Osmani A., Ukil L., et al. , 2006. A versatile and efficient gene targeting system for Aspergillus nidulans. Genetics 172: 1557–1566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nitsche B. M., Jørgensen T. R., Akeroyd M., Meyer V., Ram A. F., 2012. The carbon starvation response of Aspergillus niger during submerged cultivation: insights from the transcriptome and secretome. BMC Genomics 13: 380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldham S., Hafen E., 2003. Insulin/IGF and target of rapamycin signaling: a TOR de force in growth control. Trends Cell Biol. 13: 79–85. [DOI] [PubMed] [Google Scholar]
- Oliveira L. C. G., Silva V. O., Okamoto D. N., Kondo M. Y., Santos S. M. B., et al. , 2012. Internally quenched fluorescent peptide libraries with randomized sequences designed to detect endopeptidases. Anal. Biochem. 421: 299–307. [DOI] [PubMed] [Google Scholar]
- Osmani S. A., May G. S., Morris N. R., 1987. Regulation of the mRNA levels of nimA, a gene required for the G2-M transition in Aspergillus nidulans. J. Cell Biol. 104: 1495–1504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel A. Y., McDonald T. M., Spears L. D., Ching J. K., Fisher J. S., 2011. Ataxia telangiectasia mutated influences cytochrome c oxidase activity. Biochem. Biophys. Res. Commun. 405: 599–603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramsdale M., 2008. Programmed cell death in pathogenic fungi. Biochim. Biophys. Acta 1783: 1369–1380. [DOI] [PubMed] [Google Scholar]
- Rolland F., de Winde J. H., Lemaire K., Boles E., Thevelein J. M., et al. , 2000. Glucose-induced cAMP signalling in yeast requires both a G-protein coupled receptor system for extracellular glucose detection and a separable hexose kinase-dependent sensing process. Mol. Microbiol. 38: 348–358. [DOI] [PubMed] [Google Scholar]
- Rubio-Texeira M., Van Zeebroeck G., Voordeckers K., Thevelein J. M., 2010. Saccharomyces cerevisiae plasma membrane nutrient sensors and their role in PKA signaling. FEMS Yeast Res. 10: 134–149. [DOI] [PubMed] [Google Scholar]
- Saito S., Goodarzi A. A., Higashimoto Y., Noda Y., Lees-Miller S. P., et al. , 2002. ATM mediates phosphorylation at multiple p53 sites, including Ser46, in response to ionizing radiation. J. Biol. Chem. 277: 12491–12494. [DOI] [PubMed] [Google Scholar]
- Santos, J., C. Leão, and M. J. Sousa, 2012 Growth culture conditions and nutrient signaling modulating yeast chronological longevity. Oxid. Med. Cell Longev. 2012: 680604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semighini C. P., Marins M., Goldman M. H. S., Goldman G. H., 2002. Quantitative analysis of the relative transcript levels of ABC transporter Atr genes in Aspergillus nidulans by real-time reverse transcription-PCR assay. Appl. Environ. Microbiol. 68: 1351–1357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharon A., Finkelstein A., Shlezinger N., Hatam I., 2009. Fungal apoptosis: function, genes and gene function. FEMS Microbiol. Rev. 33: 833–854. [DOI] [PubMed] [Google Scholar]
- Shiloh Y., 2006. The ATM-mediated DNA-damage response: taking shape. Trends Biochem. Sci. 31: 402–410. [DOI] [PubMed] [Google Scholar]
- Stracker T. H., Roig I., Knobel P. A., Marjanović M., 2013. The ATM signaling network in development and disease. Front Genet. 4: 37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szilágyi M., Miskei M., Karányi Z., Lenkey B., Pócsi I., et al. , 2013. Transcriptome changes initiated by carbon starvation in Aspergillus nidulans. Microbiology 159: 176–190. [DOI] [PubMed] [Google Scholar]
- Talbot N. J., Kershaw M. J., 2009. The emerging role of autophagy in plant pathogen attack and host defense. Curr. Opin. Plant Biol. 12: 444–450. [DOI] [PubMed] [Google Scholar]
- Valentin-Vega Y. A., Kastan M. B., 2012. A new role for ATM: regulating mitochondrial function and mitophagy. Autophagy 8: 840–841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valentin-Vega Y. A., Maclean K. H., Tait-Mulder J., Milasta S., Steeves M., et al. , 2012. Mitochondrial dysfunction in ataxia-telangiectasia. Blood 119: 1490–1500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vazquez-Martin A., Oliveras-Ferraros C., Cufí S., Martin-Castillo B., Menendez J. A., 2011. Metformin activates an ataxia telangiectasia mutated (ATM)/Chk2-regulated DNA damage-like response. Cell Cycle 10: 1499–1501. [DOI] [PubMed] [Google Scholar]
- Wang J., Jiang J. C., Jazwinski S. M., 2010. Gene regulation in yeast during life extension by nutrient limitation. Exp. Gerontol. 45: 621–631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson W. A., Roach P. J., 2002. Nutrient-regulated protein kinases in budding yeast. Cell 111: 155–158. [DOI] [PubMed] [Google Scholar]
- Wullschleger S., Loewith R., Hall M., 2006. TOR signaling in growth and metabolism. Cell 124: 471–484. [DOI] [PubMed] [Google Scholar]
- Yanagisawa S., Kikuma T., Kitamoto K., 2013. Functional analysis of Aoatg1 and detection of the Cvt pathway in Aspergillus oryzae. FEMS Microbiol. Lett. 338: 168–176. [DOI] [PubMed] [Google Scholar]
- Yorimitsu T., Zaman S., Broach J. R., Klionsky D. J., 2007. Protein kinase A and Sch9 cooperatively regulate induction of autophagy in Saccharomyces cerevisiae. Mol. Biol. Cell. 18(10): 4180–4189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zdralevic M., Guaragnella N., Antonacci E. M., Giannattasio S., 2012. Yeast as a tool to study signaling pathways in mitochondrial stress response and cytoprotection. Sci. World J. 2012: 912147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhan H., Suzuki T., Aizawa K., Miyagawa K., Nagai R., et al. , 2010. Ataxia telangiectasia mutated (ATM)-mediated DNA damage response in oxidative stress-induced vascular endothelial cell senescence. J. Biol. Chem. 285: 29662–29670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao M., Klionsky D. J., 2011. AMPK-dependent phosphorylation of ULK1 induces autophagy. Cell Metab. 13: 119–120. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.