Abstract
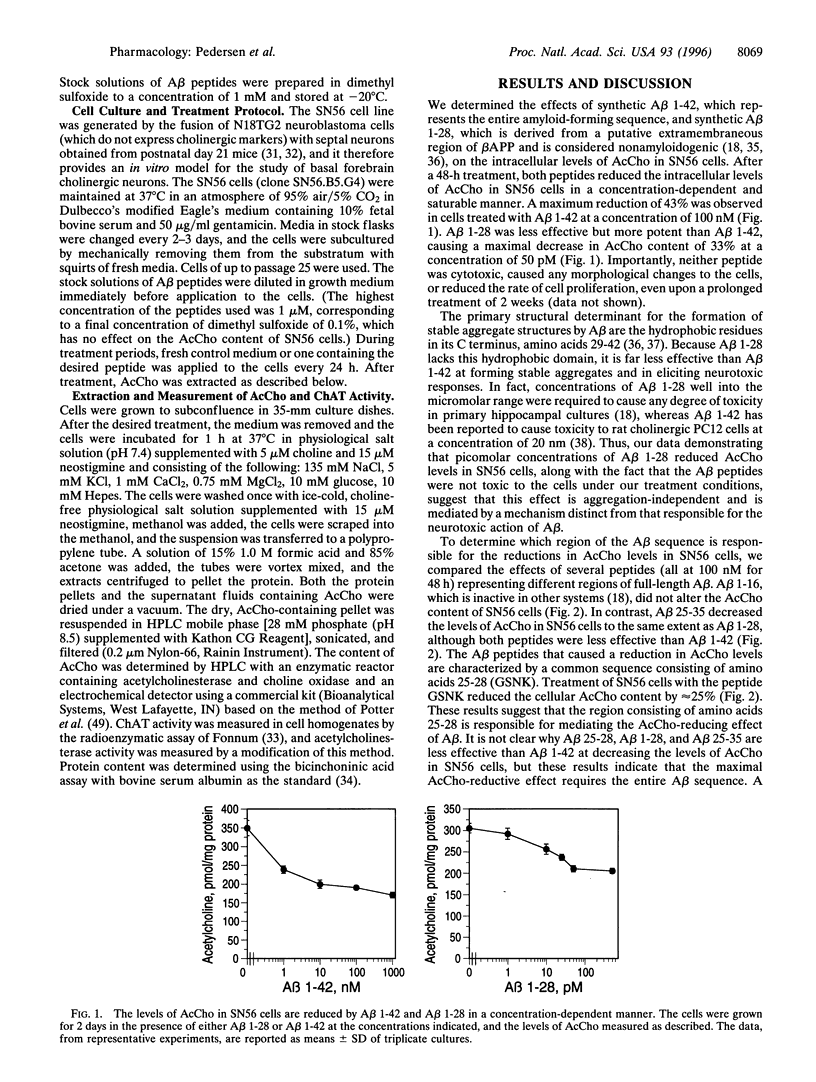
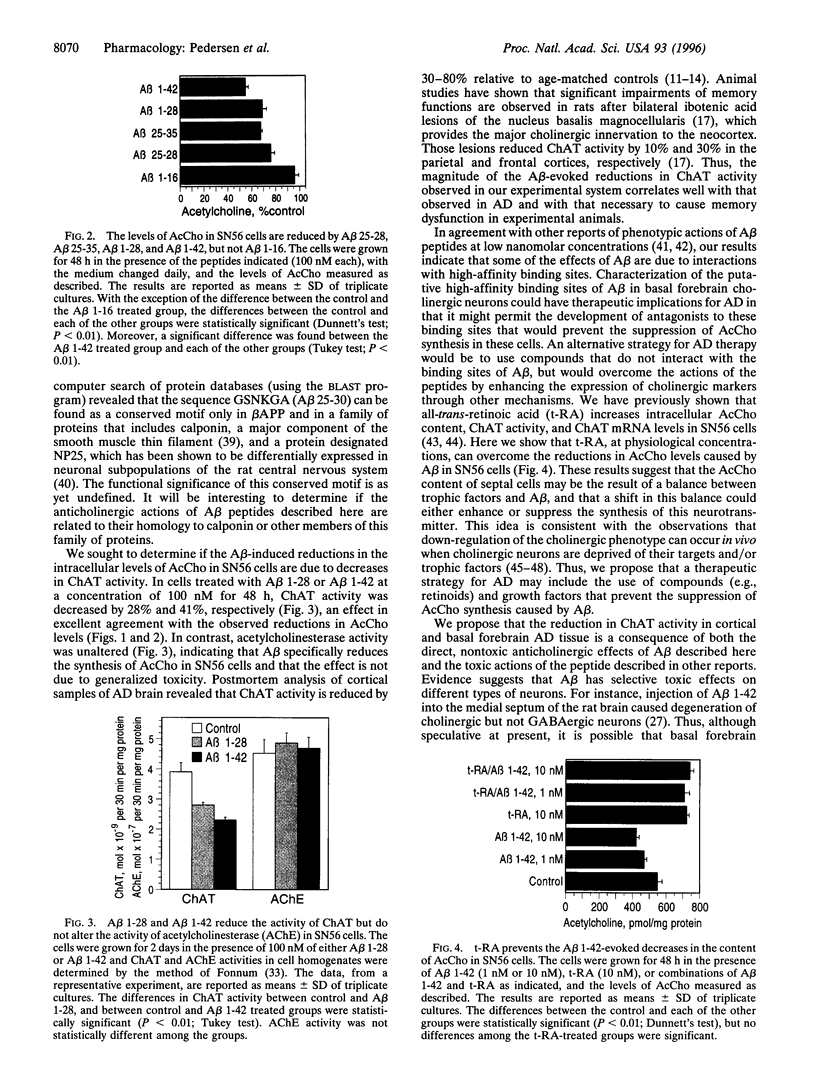
The characteristic features of a brain with Alzheimer disease (AD) include the presence of neuritic plaques composed of amyloid beta-protein (Abeta) and reductions in the levels of cholinergic markers. Neurotoxic responses to Abeta have been reported in vivo and in vitro, suggesting that the cholinergic deficit in AD brain may be secondary to the degeneration of cholinergic neurons caused by Abeta. However, it remains to be determined if Abeta contributes to the cholinergic deficit in AD brain by nontoxic effects. We examined the effects of synthetic Abeta peptides on the cholinergic properties of a mouse cell line, SN56, derived from basal forebrain cholinergic neurons. Abeta 1-42 and Abeta 1-28 reduced the acetylcholine (AcCho) content of the cells in a concentration-dependent fashion, whereas Abeta 1-16 was inactive. Maximal reductions of 43% and 33% were observed after a 48-h treatment with 100 nM of Abeta 1-42 and 50 pM of Abeta 1-28, respectively. Neither Abeta 1-28 nor Abeta 1-42 at a concentration of 100 nM and a treatment period of 2 weeks was toxic to the cells. Treatment of the cells with Abeta 25-28 (48 h; 100 nM) significantly decreased AcCho levels, suggesting that the sequence GSNK (aa 25-28) is responsible for the AcCho-reducing effect of Abeta. The reductions in AcCho levels caused by Abeta 1-42 and Abeta 1-28 were accompanied by proportional decreases in choline acetyltransferase activity. In contrast, acetylcholinesterase activity was unaltered, indicating that Abeta specifically reduces the synthesis of AcCho in SN56 cells. The reductions in AcCho content caused by Abeta 1-42 could be prevented by a cotreatment with all-trans-retinoic acid (10 nM), a compound previously shown to increase choline acetyltransferase mRNA expression in SN56 cells. These results demonstrate a nontoxic, suppressive effect of Abeta on AcCho synthesis, an action that may contribute to the cholinergic deficit in AD brain.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abe E., Casamenti F., Giovannelli L., Scali C., Pepeu G. Administration of amyloid beta-peptides into the medial septum of rats decreases acetylcholine release from hippocampus in vivo. Brain Res. 1994 Feb 4;636(1):162–164. doi: 10.1016/0006-8993(94)90193-7. [DOI] [PubMed] [Google Scholar]
- Behl C., Davis J., Cole G. M., Schubert D. Vitamin E protects nerve cells from amyloid beta protein toxicity. Biochem Biophys Res Commun. 1992 Jul 31;186(2):944–950. doi: 10.1016/0006-291x(92)90837-b. [DOI] [PubMed] [Google Scholar]
- Berse B., Blusztajn J. K. Coordinated up-regulation of choline acetyltransferase and vesicular acetylcholine transporter gene expression by the retinoic acid receptor alpha, cAMP, and leukemia inhibitory factor/ciliary neurotrophic factor signaling pathways in a murine septal cell line. J Biol Chem. 1995 Sep 22;270(38):22101–22104. doi: 10.1074/jbc.270.38.22101. [DOI] [PubMed] [Google Scholar]
- Bierer L. M., Haroutunian V., Gabriel S., Knott P. J., Carlin L. S., Purohit D. P., Perl D. P., Schmeidler J., Kanof P., Davis K. L. Neurochemical correlates of dementia severity in Alzheimer's disease: relative importance of the cholinergic deficits. J Neurochem. 1995 Feb;64(2):749–760. doi: 10.1046/j.1471-4159.1995.64020749.x. [DOI] [PubMed] [Google Scholar]
- Bowen D. M., Smith C. B., White P., Davison A. N. Neurotransmitter-related enzymes and indices of hypoxia in senile dementia and other abiotrophies. Brain. 1976 Sep;99(3):459–496. doi: 10.1093/brain/99.3.459. [DOI] [PubMed] [Google Scholar]
- Burdick D., Soreghan B., Kwon M., Kosmoski J., Knauer M., Henschen A., Yates J., Cotman C., Glabe C. Assembly and aggregation properties of synthetic Alzheimer's A4/beta amyloid peptide analogs. J Biol Chem. 1992 Jan 5;267(1):546–554. [PubMed] [Google Scholar]
- Cai X. D., Golde T. E., Younkin S. G. Release of excess amyloid beta protein from a mutant amyloid beta protein precursor. Science. 1993 Jan 22;259(5094):514–516. doi: 10.1126/science.8424174. [DOI] [PubMed] [Google Scholar]
- Chartier-Harlin M. C., Crawford F., Houlden H., Warren A., Hughes D., Fidani L., Goate A., Rossor M., Roques P., Hardy J. Early-onset Alzheimer's disease caused by mutations at codon 717 of the beta-amyloid precursor protein gene. Nature. 1991 Oct 31;353(6347):844–846. doi: 10.1038/353844a0. [DOI] [PubMed] [Google Scholar]
- Citron M., Oltersdorf T., Haass C., McConlogue L., Hung A. Y., Seubert P., Vigo-Pelfrey C., Lieberburg I., Selkoe D. J. Mutation of the beta-amyloid precursor protein in familial Alzheimer's disease increases beta-protein production. Nature. 1992 Dec 17;360(6405):672–674. doi: 10.1038/360672a0. [DOI] [PubMed] [Google Scholar]
- Citron M., Vigo-Pelfrey C., Teplow D. B., Miller C., Schenk D., Johnston J., Winblad B., Venizelos N., Lannfelt L., Selkoe D. J. Excessive production of amyloid beta-protein by peripheral cells of symptomatic and presymptomatic patients carrying the Swedish familial Alzheimer disease mutation. Proc Natl Acad Sci U S A. 1994 Dec 6;91(25):11993–11997. doi: 10.1073/pnas.91.25.11993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dutar P., Bassant M. H., Senut M. C., Lamour Y. The septohippocampal pathway: structure and function of a central cholinergic system. Physiol Rev. 1995 Apr;75(2):393–427. doi: 10.1152/physrev.1995.75.2.393. [DOI] [PubMed] [Google Scholar]
- Etcheberrigaray R., Ito E., Kim C. S., Alkon D. L. Soluble beta-amyloid induction of Alzheimer's phenotype for human fibroblast K+ channels. Science. 1994 Apr 8;264(5156):276–279. doi: 10.1126/science.8146663. [DOI] [PubMed] [Google Scholar]
- Fonnum F. A rapid radiochemical method for the determination of choline acetyltransferase. J Neurochem. 1975 Feb;24(2):407–409. doi: 10.1111/j.1471-4159.1975.tb11895.x. [DOI] [PubMed] [Google Scholar]
- Games D., Adams D., Alessandrini R., Barbour R., Berthelette P., Blackwell C., Carr T., Clemens J., Donaldson T., Gillespie F. Alzheimer-type neuropathology in transgenic mice overexpressing V717F beta-amyloid precursor protein. Nature. 1995 Feb 9;373(6514):523–527. doi: 10.1038/373523a0. [DOI] [PubMed] [Google Scholar]
- Giordano T., Pan J. B., Monteggia L. M., Holzman T. F., Snyder S. W., Krafft G., Ghanbari H., Kowall N. W. Similarities between beta amyloid peptides 1-40 and 40-1: effects on aggregation, toxicity in vitro, and injection in young and aged rats. Exp Neurol. 1994 Feb;125(2):175–182. doi: 10.1006/exnr.1994.1022. [DOI] [PubMed] [Google Scholar]
- Giovannelli L., Casamenti F., Scali C., Bartolini L., Pepeu G. Differential effects of amyloid peptides beta-(1-40) and beta-(25-35) injections into the rat nucleus basalis. Neuroscience. 1995 Jun;66(4):781–792. doi: 10.1016/0306-4522(94)00610-h. [DOI] [PubMed] [Google Scholar]
- Goate A., Chartier-Harlin M. C., Mullan M., Brown J., Crawford F., Fidani L., Giuffra L., Haynes A., Irving N., James L. Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer's disease. Nature. 1991 Feb 21;349(6311):704–706. doi: 10.1038/349704a0. [DOI] [PubMed] [Google Scholar]
- Hammond D. N., Wainer B. H., Tonsgard J. H., Heller A. Neuronal properties of clonal hybrid cell lines derived from central cholinergic neurons. Science. 1986 Dec 5;234(4781):1237–1240. doi: 10.1126/science.3775382. [DOI] [PubMed] [Google Scholar]
- Harkany T., De Jong G. I., Soós K., Penke B., Luiten P. G., Gulya K. Beta-amyloid (1-42) affects cholinergic but not parvalbumin-containing neurons in the septal complex of the rat. Brain Res. 1995 Nov 6;698(1-2):270–274. doi: 10.1016/0006-8993(95)01013-l. [DOI] [PubMed] [Google Scholar]
- Harkany T., Lengyel Z., Soós K., Penke B., Luiten P. G., Gulya K. Cholinotoxic effects of beta-amyloid (1-42) peptide on cortical projections of the rat nucleus basalis magnocellularis. Brain Res. 1995 Oct 9;695(1):71–75. doi: 10.1016/0006-8993(95)00823-9. [DOI] [PubMed] [Google Scholar]
- Ichimiya Y., Arai H., Kosaka K., Iizuka R. Morphological and biochemical changes in the cholinergic and monoaminergic systems in Alzheimer-type dementia. Acta Neuropathol. 1986;70(2):112–116. doi: 10.1007/BF00691428. [DOI] [PubMed] [Google Scholar]
- Kim C. S., Han Y. F., Etcheberrigaray R., Nelson T. J., Olds J. L., Yoshioka T., Alkon D. L. Alzheimer and beta-amyloid-treated fibroblasts demonstrate a decrease in a memory-associated GTP-binding protein, Cp20. Proc Natl Acad Sci U S A. 1995 Mar 28;92(7):3060–3064. doi: 10.1073/pnas.92.7.3060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kowall N. W., Beal M. F., Busciglio J., Duffy L. K., Yankner B. A. An in vivo model for the neurodegenerative effects of beta amyloid and protection by substance P. Proc Natl Acad Sci U S A. 1991 Aug 15;88(16):7247–7251. doi: 10.1073/pnas.88.16.7247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambert M. P., Stevens G., Sabo S., Barber K., Wang G., Wade W., Krafft G., Snyder S., Holzman T. F., Klein W. L. Beta/A4-evoked degeneration of differentiated SH-SY5Y human neuroblastoma cells. J Neurosci Res. 1994 Nov 1;39(4):377–385. doi: 10.1002/jnr.490390404. [DOI] [PubMed] [Google Scholar]
- Lee H. J., Hammond D. N., Large T. H., Wainer B. H. Immortalized young adult neurons from the septal region: generation and characterization. Brain Res Dev Brain Res. 1990 Mar 1;52(1-2):219–228. doi: 10.1016/0165-3806(90)90238-t. [DOI] [PubMed] [Google Scholar]
- Mann D. M., Jones D., Prinja D., Purkiss M. S. The prevalence of amyloid (A4) protein deposits within the cerebral and cerebellar cortex in Down's syndrome and Alzheimer's disease. Acta Neuropathol. 1990;80(3):318–327. doi: 10.1007/BF00294651. [DOI] [PubMed] [Google Scholar]
- Mullan M., Crawford F. Genetic and molecular advances in Alzheimer's disease. Trends Neurosci. 1993 Oct;16(10):398–403. doi: 10.1016/0166-2236(93)90007-9. [DOI] [PubMed] [Google Scholar]
- Naumann T., Kermer P., Frotscher M. Fine structure of rat septohippocampal neurons. III. Recovery of choline acetyltransferase immunoreactivity after fimbria-fornix transection. J Comp Neurol. 1994 Dec 8;350(2):161–170. doi: 10.1002/cne.903500202. [DOI] [PubMed] [Google Scholar]
- Pedersen W. A., Berse B., Schüler U., Wainer B. H., Blusztajn J. K. All-trans- and 9-cis-retinoic acid enhance the cholinergic properties of a murine septal cell line: evidence that the effects are mediated by activation of retinoic acid receptor-alpha. J Neurochem. 1995 Jul;65(1):50–58. doi: 10.1046/j.1471-4159.1995.65010050.x. [DOI] [PubMed] [Google Scholar]
- Perry E. K., Tomlinson B. E., Blessed G., Bergmann K., Gibson P. H., Perry R. H. Correlation of cholinergic abnormalities with senile plaques and mental test scores in senile dementia. Br Med J. 1978 Nov 25;2(6150):1457–1459. doi: 10.1136/bmj.2.6150.1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pike C. J., Burdick D., Walencewicz A. J., Glabe C. G., Cotman C. W. Neurodegeneration induced by beta-amyloid peptides in vitro: the role of peptide assembly state. J Neurosci. 1993 Apr;13(4):1676–1687. doi: 10.1523/JNEUROSCI.13-04-01676.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pike C. J., Overman M. J., Cotman C. W. Amino-terminal deletions enhance aggregation of beta-amyloid peptides in vitro. J Biol Chem. 1995 Oct 13;270(41):23895–23898. doi: 10.1074/jbc.270.41.23895. [DOI] [PubMed] [Google Scholar]
- Pike C. J., Walencewicz-Wasserman A. J., Kosmoski J., Cribbs D. H., Glabe C. G., Cotman C. W. Structure-activity analyses of beta-amyloid peptides: contributions of the beta 25-35 region to aggregation and neurotoxicity. J Neurochem. 1995 Jan;64(1):253–265. doi: 10.1046/j.1471-4159.1995.64010253.x. [DOI] [PubMed] [Google Scholar]
- Pike C. J., Walencewicz A. J., Glabe C. G., Cotman C. W. In vitro aging of beta-amyloid protein causes peptide aggregation and neurotoxicity. Brain Res. 1991 Nov 1;563(1-2):311–314. doi: 10.1016/0006-8993(91)91553-d. [DOI] [PubMed] [Google Scholar]
- Potter P. E., Meek J. L., Neff N. H. Acetylcholine and choline in neuronal tissue measured by HPLC with electrochemical detection. J Neurochem. 1983 Jul;41(1):188–194. doi: 10.1111/j.1471-4159.1983.tb13668.x. [DOI] [PubMed] [Google Scholar]
- Ren W. Z., Ng G. Y., Wang R. X., Wu P. H., O'Dowd B. F., Osmond D. H., George S. R., Liew C. C. The identification of NP25: a novel protein that is differentially expressed by neuronal subpopulations. Brain Res Mol Brain Res. 1994 Mar;22(1-4):173–185. doi: 10.1016/0169-328x(94)90045-0. [DOI] [PubMed] [Google Scholar]
- Selkoe D. J. Normal and abnormal biology of the beta-amyloid precursor protein. Annu Rev Neurosci. 1994;17:489–517. doi: 10.1146/annurev.ne.17.030194.002421. [DOI] [PubMed] [Google Scholar]
- Smith P. K., Krohn R. I., Hermanson G. T., Mallia A. K., Gartner F. H., Provenzano M. D., Fujimoto E. K., Goeke N. M., Olson B. J., Klenk D. C. Measurement of protein using bicinchoninic acid. Anal Biochem. 1985 Oct;150(1):76–85. doi: 10.1016/0003-2697(85)90442-7. [DOI] [PubMed] [Google Scholar]
- Sofroniew M. V., Cooper J. D., Svendsen C. N., Crossman P., Ip N. Y., Lindsay R. M., Zafra F., Lindholm D. Atrophy but not death of adult septal cholinergic neurons after ablation of target capacity to produce mRNAs for NGF, BDNF, and NT3. J Neurosci. 1993 Dec;13(12):5263–5276. doi: 10.1523/JNEUROSCI.13-12-05263.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sofroniew M. V., Galletly N. P., Isacson O., Svendsen C. N. Survival of adult basal forebrain cholinergic neurons after loss of target neurons. Science. 1990 Jan 19;247(4940):338–342. doi: 10.1126/science.1688664. [DOI] [PubMed] [Google Scholar]
- Suzuki N., Cheung T. T., Cai X. D., Odaka A., Otvos L., Jr, Eckman C., Golde T. E., Younkin S. G. An increased percentage of long amyloid beta protein secreted by familial amyloid beta protein precursor (beta APP717) mutants. Science. 1994 May 27;264(5163):1336–1340. doi: 10.1126/science.8191290. [DOI] [PubMed] [Google Scholar]
- Svendsen C. N., Cooper J. D., Sofroniew M. V. Trophic factor effects on septal cholinergic neurons. Ann N Y Acad Sci. 1991;640:91–94. doi: 10.1111/j.1749-6632.1991.tb00197.x. [DOI] [PubMed] [Google Scholar]
- Ueda K., Fukui Y., Kageyama H. Amyloid beta protein-induced neuronal cell death: neurotoxic properties of aggregated amyloid beta protein. Brain Res. 1994 Mar 14;639(2):240–244. doi: 10.1016/0006-8993(94)91736-1. [DOI] [PubMed] [Google Scholar]
- Whitehouse P. J., Price D. L., Struble R. G., Clark A. W., Coyle J. T., Delon M. R. Alzheimer's disease and senile dementia: loss of neurons in the basal forebrain. Science. 1982 Mar 5;215(4537):1237–1239. doi: 10.1126/science.7058341. [DOI] [PubMed] [Google Scholar]
- Winder S. J., Walsh M. P. Calponin: thin filament-linked regulation of smooth muscle contraction. Cell Signal. 1993 Nov;5(6):677–686. doi: 10.1016/0898-6568(93)90029-l. [DOI] [PubMed] [Google Scholar]
- Winkler J., Suhr S. T., Gage F. H., Thal L. J., Fisher L. J. Essential role of neocortical acetylcholine in spatial memory. Nature. 1995 Jun 8;375(6531):484–487. doi: 10.1038/375484a0. [DOI] [PubMed] [Google Scholar]
- Yankner B. A., Duffy L. K., Kirschner D. A. Neurotrophic and neurotoxic effects of amyloid beta protein: reversal by tachykinin neuropeptides. Science. 1990 Oct 12;250(4978):279–282. doi: 10.1126/science.2218531. [DOI] [PubMed] [Google Scholar]
- Zhang Z., Drzewiecki G. J., Hom J. T., May P. C., Hyslop P. A. Human cortical neuronal (HCN) cell lines: a model for amyloid beta neurotoxicity. Neurosci Lett. 1994 Aug 15;177(1-2):162–164. doi: 10.1016/0304-3940(94)90892-3. [DOI] [PubMed] [Google Scholar]