Patients who received the latest generation venoarterial extracorporeal membrane oxygenation (ECMO) for refractory cardiogenic shock still had a high risk of developing nosocomial infections, particularly ventilator-associated pneumonia. Rates of nosocomial infections increased with longer ECMO support, and infection severity was independently associated with in–intensive care unit death.
Abstract
Background. Incidence and impact on adult patients’ outcomes of nosocomial infections (NIs) occurring during venoarterial extracorporeal membrane oxygenation (VA-ECMO) support for refractory cardiogenic shock have rarely been described.
Methods. We retrospectively reviewed the charts of a large series of patients who received VA-ECMO in our intensive care unit (ICU) from January 2003 through December 2009. Incidence, types, risk factors, and impact on outcomes of NIs occurring during ECMO support were analyzed.
Results. Among 220 patients (49 ± 16 years old, simplified acute physiology score (SAPS) II 61 ± 20) who underwent ECMO support for >48 hours for a total of 2942 ECMO days, 142 (64%) developed NIs. Ventilator-associated pneumonia (VAP), bloodstream infections, cannula infections, and mediastinitis infections occurred in 55%, 18%, 10% and 11% of the patients, respectively. More critical condition at ICU admission, but not antibiotics at the time of ECMO cannulation, was associated with subsequently developing NIs (hazard ratio, 0.73; 95% confidence interval [CI], .50–1.05; P = .09). Infected patients had longer durations of mechanical ventilation, ECMO support, and hospital stays. Independent predictors of death were infection with severe sepsis or septic shock (odds ratio, 1.93; 95% CI, 1.26–2.94; P = .002) and SAPS II, whereas immunosuppression and myocarditis as the reason for ECMO support were associated with better outcomes.
Conclusions. Cardiogenic shock patients who received the latest generation VA-ECMO still had a high risk of developing NIs, particularly VAP. Strategies aimed at preventing these infections may improve the outcomes of these critically ill patients.
Venoarterial extracorporeal membrane oxygenation (VA-ECMO) is an easily applicable and widely accepted treatment option for temporary circulatory assistance in patients with cardiogenic shock refractory to conventional medical therapies [1–3]. This procedure provides prolonged cardiac and respiratory life support, allowing myocardial recovery or bridging to cardiac transplantation or the implantation of a left ventricle assist device (LVAD). Despite major advances in device technology and the intensive care of these critically ill patients, high short-term mortality rates persist [3–5]. Although reasons for this high mortality are many, nosocomial infections (NIs) occurring while on ECMO may have dreadful consequences. Indeed, risks of developing infections are markedly increased in these very sick patients with disease-induced compromised immune systems and many indwelling medical devices (ie, large ECMO cannulas, endotracheal tube, intra-aortic balloon pump, and central venous catheter).
To date, only a few studies have carefully evaluated the NI incidence and impact on outcomes of patients receiving ECMO [6–15], and most of them were conducted on children [6–8, 13–15] or on a majority of patients receiving venovenous ECMO for respiratory failure [9] or were conducted before the diffusion of the newest generation of ECMO system driven by miniaturized centrifugal pumps and comprising biocompatible and heparin-coated circuits and membrane oxygenators [6–9, 11, 13]. Thus, the objective of this study was to analyze the NI incidence, types, risk factors, and impact on outcomes in a homogeneous group of adults who received the latest generation VA-ECMO devices for refractory cardiogenic shock.
PATIENTS AND METHODS
Setting
This study was conducted in an 18-bed medical-surgical intensive care unit (ICU) at La Pitié-Salpêtrière University Hospital, Paris, France. Its protocol was in accordance with the ethical standards of our institution's Committee for the Protection of Human Research Subjects. No informed consent was obtained because this epidemiologic study did not modify existing diagnostic or therapeutic strategies.
Patients
We retrospectively analyzed the charts of the 271 consecutive patients who received VA-ECMO for refractory cardiogenic shock in our ICU from January 2003 through December 2009. Every patient had the following signs of acute refractory cardiogenic shock before ECMO institution: evidence of tissue hypoxia concomitant with adequate intravascular volume and sustained hypotension and reduced cardiac index (<2.2 L/minutes/m2) despite infusion of high-dose catecholamines (epinephrine >0.2 µg/kg/minutes or dobutamine >20 µg/kg/minutes ± norepinephrine >0.2 µg/kg/minutes). Venoarterial extracorporeal membrane oxygenation support was initiated under the following 4 circumstances: (1) acute refractory cardiogenic shock complicating acute myocardial infarction, end-stage dilated cardiomyopathy, or fulminant myocarditis; (2) postcardiotomy cardiogenic shock; (3) immediate post-transplant cardiac graft failure, with elevated pulmonary pressures and right and/or left ventricular dysfunction; (4) miscellaneous conditions (eg, cardiotoxic drug overdose, acute cardiac allograft rejection, traumatic cardiac tamponade with persistent cardiac arrest or myocardial contusion following ballistic chest trauma). Patients receiving venovenous ECMO and the 51 patients who had ECMO for ≤48 hours and were not exposed to ECMO long enough to identify ECMO-related infections were excluded from the study.
ECMO Support
The detailed surgical procedure for ECMO placement has been described elsewhere [5, 16]. Briefly, cannulation for VA-ECMO support was either percutaneous at the femoral site or central (ie, intrathoracic) with right atrial and aortic lines. Peripheral ECMO was switched to central ECMO when acute pulmonary edema due to insufficient left ventricular unloading or acute leg ischemia occurred. Prophylactic antibiotic therapy with second-generation cephalosporin or glycopeptides as a single intravenous injection and based on patients’ risk factors for methicillin-resistant cocci carriage was used only for intrathoracic ECMO cannula implantation when patients were not receiving antibiotics at that time. When ECMO weaning was impossible, bridging to an LVAD or transplantation was considered.
Standard Care Procedure for Patients on ECMO
Standard care for these patients included at least 1 peripheral intravenous line, orotracheal intubation and tracheostomy when mechanical ventilation (MV) duration exceeded 10 days, nasogastric tube insertion, and urethral catheterization. Extracorporeal membrane oxygenation cannula insertion sites, central venous and arterial catheters, chest tube, and sternum wound were observed daily. Transparent dressings at the insertion sites were changed every 2 days after disinfection with a chlorhexidine solution. A ventilator-associated pneumonia (VAP) prevention care bundle was instituted in our ICU many years ago, which involves hand hygiene with alcohol-based sanitizer; use of gloves and gowns; keeping patients in a semirecumbent (30°– 45°) rather than supine position to prevent aspiration; monitoring to maintain endotracheal tube cuff pressure >20 cm water, which lowers the risk of bacterial pathogen leakage around the cuff into the lower respiratory tract; and oral chlorhexidine mouth decontamination performed at least 6 times per day. Compliance with these infection prevention measures is regularly evaluated by internal audits and remains high (>70%). If compliance with 1 of the measures of the bundle decreases to <50%, an educational program is reinstituted. Standardized prophylactic or selective decontamination antibiotic regimens were not used; stress ulcer prophylaxis was not systematic, and, when needed, proton pump inhibitor was preferred. Tracheal aspirates, cannula or catheter insertion sites, mediastinal wound, and blood were not routinely cultured. However, extreme vigilance for the onset of infectious complications was maintained throughout hospitalization. When VAP was suspected based on clinical, biological, or radiological signs, quantitative culture of bronchoalveolar lavage fluid collected under fiberoptic bronchoscopy was performed before the introduction of new antibiotics. Subcutaneous needle aspirates at the ECMO cannula insertion site or from the mediastinal wound were obtained when cannula-related infection was suspected. When catheter-related infection was suspected, blood cultures were performed, and the catheter was removed and cultured. Blood cultures were also performed when body temperature was >38°C or <36°C or when clinical or biological signs of sepsis occurred. Empiric antibiotic therapy was initiated as soon as a serious infection was suspected. To reduce unnecessary antibiotic prescriptions and decrease antibiotic selection pressure and emergence of antibiotic-resistant bacteria, antibiotic therapy was adjusted or stopped according to the results of bacterial cultures. Duration of antibiotic therapy was based on a procalcitonin-guided strategy [17], except for mediastinitis, endocarditis, or cannula-associated cellulitis, when removal of ECMO cannula was deemed impossible.
Definitions of NI
Nosocomial infection definitions agreed with those of the Centers for Disease Control and Prevention National Nosocomial Infections Surveillance System [18]. Only infections occurring >24 hours after ECMO initiation and within 48 hours after ECMO discontinuation were defined as ECMO-associated. Pathogens and NI type and severity (ie, sepsis, severe sepsis, or septic shock) were recorded [19]. Ventilator-associated pneumonia was suspected when a new and persistent radiographic infiltrate was accompanied by purulent secretions, fever ≥38.3°C, or leukocyte count >109/L or when 1 of these occurred in patients with baseline diffuse, dense infiltrates. Ventilator-associated pneumonia was diagnosed before antibiotics by quantitative distal bronchoalveolar lavage cultures growing ≥104 colony-forming units/mL [20, 21]. Poststernotomy mediastinitis was defined as a deep wound infection associated with sternal osteomyelitis, with or without retrosternal space involvement, that required surgical debridement [22, 23]. Diagnosis of cannula infections required local signs of infection at the access site with positive culture of subcutaneous needle aspirate from the cannula site. Criteria for catheter-related infection included culture of the intravascular tip of a central venous or arterial catheter yielding at least 103 colony-forming units/mL associated with catheter exit-site inflammation. Bloodstream infections were classified as catheter related when the same organism(s) were recovered from blood and intravascular tip of the catheter cultures; >2 positive blood cultures were required for coagulase-negative staphylococci. Urinary tract infections were not surveyed because all patients had urethral catheters. Three physicians (M. S., S. H., and A. C.) analyzed causes of death, and their relationships with infection were determined.
Data Collection
Each patient's hospital chart included the following data recorded at ICU admission: age, sex, body mass index, underlying medical conditions and their severity stratified according to the criteria of McCabe and Jackson [24], simplified acute physiology score II (SAPS) [25], sequential organ failure assessment (SOFA) at ICU admission [26], immunocompromised status, reason for ECMO initiation, ICU and hospital days before ECMO, location of ECMO cannulation (ie, operation room, ICU, catheterization laboratory, another hospital calling upon the mobile ECMO team), site of ECMO cannulation (ie, extrathoracic cannulation only, extrathoracic cannulation then switched to intrathoracic cannulation, intrathoracic cannulation only), antibiotics administered for any reason within the 24 hours preceding ECMO, and intra-aortic balloon pump use. Outcomes following ECMO initiation included NI, MV duration, number and duration of each ECMO episode, switches from femoral to intrathoracic ECMO, ICU and hospital lengths of stay, heart transplantation or LVAD insertion, and overall survival.
Statistical Analyses
Continuous variables are expressed as means ± standard deviations and were compared with Student's t test or the Mann−Whitney U test, as appropriate. Categorical variables, expressed as percentages, were compared with a χ2 or Fisher's exact test, as appropriate. Kaplan−Meier survival analysis was used to estimate the probability of being infection-free. Multivariate analysis by Cox regression was used to identify independent risk factors for first NI and in-ICU death. All subjects were included in the models, and follow-up began at the time of ECMO initiation. Variables achieving P ≤ .10 in univariable analyses were entered in the multivariable models, and antibiotics at the time of ECMO initiation was forced in the final models [27]. Variables with association among each other were not included in the multivariate models. Nosocomial infection with severe sepsis or septic shock was entered in the model predicting ICU death as a time-dependent covariate. Statistical significance was defined as P < .05. Analyses were performed using SPSS 11.0 (SPSS Inc) software.
RESULTS
Study Population
Among 220 patients (aged 48.9 ± 15.8 years; 67% male) who underwent ECMO for >48 hours and for a total of 2942 ECMO days, 142 (64%) developed 222 NIs, corresponding to a rate of 75.5 infectious episodes per 1000 ECMO days. Mean times to the first NI, first VAP, mediastinitis, and infection at the femoral cannula insertion site were 8 ± 11, 7 ± 12, 16 ± 8, and 12 ± 6 days, respectively, among those who had the corresponding outcome. At the time of ECMO initiation, 84 (38%) patients were receiving antibiotics. Clinical and demographic characteristics of infected and uninfected patients were comparable except that nonimmunocompromised patients and those who were first cannulated in the ICU or received any extrathoracic ECMO were less likely to have infection (Table 1). All patients received MV during course of ECMO, but at ECMO initiation, 7 patients were not mechanically ventilated (5 in the NI group).
Table 1.
Demographic and Clinical Characteristics of Extracorporeal Membrane Oxygenation Patients With and Without Nosocomial Infections
| Variable | Uninfected Patients, n = 78 | Infected Patients, n = 142 | P Value |
|---|---|---|---|
| Age, years | 47.9 (15.1) | 49.5 (16.9) | .46 |
| Male sex | 57 (73) | 90 (63) | .14 |
| SAPS IIa | 60.4 (19.1) | 61.3 (20.4) | .76 |
| SOFA scorea | 11.6 (4.5) | 12.6 (4.5) | .17 |
| Performance status ≥2 | 23 (30) | 46 (33) | .65 |
| Body mass index, kg/cm² | 25.2 (5.6) | 25.3 (4.8) | .85 |
| McCabe score ≥2 | 37 (47) | 57 (40) | .29 |
| Underlying conditionb | |||
| Diabetes mellitus | 9 (12) | 26 (18) | .19 |
| Renal insufficiency | 1 (1) | 5 (4) | .33 |
| COPD | 2 (3) | 6 (4) | .53 |
| Previous cardiac surgery | 12 (15) | 19 (13) | .68 |
| Pregnancy or postpartum | 3 (4) | 1 (1) | .09 |
| Immunocompromised statusc | 15 (19) | 49 (35) | .02 |
| Reason for ECMOd | |||
| Dilated cardiomyopathy | 16 (21) | 18 (13) | .58 |
| Myocarditis | 14 (18) | 16 (11) | .68 |
| Acute myocardial infarction | 11 (14) | 31 (22) | .04 |
| Cardiac arrest | 11 (14) | 17 (12) | .66 |
| Postcardiotomy | 23 (29) | 55 (39) | .16 |
| CABG | 6 (8) | 12 (8) | .32 |
| Valve procedure | 7 (9) | 7 (5) | .73 |
| CABG and valve procedure | 0 | 2 (1) | .18 |
| Heart transplantation | 6 (8) | 25 (18) | .007 |
| Miscellaneous | 4 (6) | 9 (6) | .28 |
| Other reasons | 3 (4) | 5 (4) | |
| Site of ECMO cannulatione | |||
| Extrathoracic only | 45 (58) | 54 (38) | .005 |
| Extrathoracic then intrathoracic | 11 (14) | 47 (33) | .002 |
| Intrathoracic only | 22 (28) | 41 (29) | .71 |
| Location of the first ECMO cannulationf | |||
| Operating room | 33 (42) | 73 (51) | .20 |
| Catheterization lab | 1 (1) | 11 (8) | .04 |
| ICU | 32 (41) | 33 (23) | .006 |
| Mobile ECMO team | 12 (15) | 24 (17) | .96 |
| ICU days before ECMO | 1.4 (3.7) | 2.2 (7.5) | .35 |
| Hospital days before ECMO | 7.7 (14.5) | 6.2 (11.8) | .41 |
| Antibiotics at the time of ECMO cannulationg | 32 (41) | 52 (37) | .52 |
Data are No. (%) of patients or mean value (± standard deviation).
Abbreviations: CABG, coronary artery bypass graft; COPD, chronic obstructive airway disease; ECMO, extracorporeal membrane oxygenation; ICU, intensive care unit; SAPS II, simplified acute physiology score; SOFA, sequential organ-failure assessment.
a Calculated at ICU admission.
b Global P value for underlying condition, P = .35.
c Includes patients with AIDs, solid organ transplantation, or hematological malignancy and those receiving chemotherapy, immunosuppressive agents, or long-term corticosteroid therapy.
d Global P value for reason for ECMO, P = .25.
e Global P value for site of ECMO cannulation, P = .004.
f Global P value for location of the first ECMO cannulation, P = .02.
g Includes patients who received prophylactic antibiotic therapy for central ECMO placement and those treated with antibiotics for an infection at the time of ECMO cannulation.
Infected Sites and Causative Organisms
The most frequently infected site was the lung, with 163 VAP episodes occurring in 120 patients, corresponding to a rate of 55.4 episodes per 1000 ECMO days. Poststernotomy mediastinitis, cannula-related NIs, and catheter-related NIs, numbered 23 (20 patients; 7.8 episodes per 1000 ECMO days), 21 (20 patients; 7.1 episodes per 1000 ECMO days), and 10 (10 patients; 3.4 episodes per 1000 ECMO days), respectively. Among the 47 bloodstream infections (39 patients; 16 episodes per 1000 ECMO days), 23 were associated with a VAP, 10 were associated with mediastinitis, 5 were associated with cannula infection, 4 were associated with catheter infection, and 5 were isolated. Rates of septic shock were higher for VAP (36%), mediastinitis (35%), and bloodstream infections (43%) than for cannula- (19%) and catheter-related (20%) infections.
Micro-organisms associated with NIs are detailed in Table 2. Pseudomonas aeruginosa was the most frequently grown bacterium from pulmonary samples (26%). Of the 47 bloodstream infection episodes, 21% were due to Pseudomonas aeruginosa, 15% were due to Enterococcus species, and 13% were due to Escherichia coli. Eight cases of Candida and 7 cases of Staphylococcus epidermidis posternotomy mediastinitis were also recorded. Pathogens most frequently associated with cannula infection were Escherichia coli (24%), Enterococcus species (19%), and coagulase-negative staphylococci (19%).
Table 2.
Microorganisms Associated With Various Nosocomial Infections in 142 Extracorporeal Membrane Oxygenation Patients
| Ventilator-Associated Pneumonia, n = 163a |
Cannula Infection, n = 21b |
Poststernotomy Mediastinitis, n = 23c |
Bloodstream Infection, n = 47d |
||||
|---|---|---|---|---|---|---|---|
| Organism | No. (%) | Organism | No. (%) | Organism | No. (%) | Organism | No. (%) |
| Pseudomonas aeruginosa | 43 (26) | Escherichia coli | 5 (24) | Candida spp. | 8 (35) | P. aeruginosa | 10 (21) |
| Polymicrobiale | 19 (12) | Enterococcus spp. | 4 (19) | Staphylococcus epidermidis | 7 (30) | Enterococcus spp. | 7 (15) |
| Staphylococcus aureus | 16 (10) | S. epidermidis | 4 (19) | P. aeruginosa | 2 (9) | Escherichia coli | 6 (13) |
| Enterobacter sp. | 16 (10) | Polymicrobialf | 3 (14) | S. aureus | 2 (9 | S. epidermidis | 5 (10) |
| Escherichia coli | 14 (9) | S. aureus | 2 (10) | Escherichia coli | 2 (9) | S. aureus | 4 (9) |
| Haemophilus influenzae | 14 (9) | P. aeruginosa | 2 (10) | Enterobacter spp. | 1 (4) | Streptococcus spp. | 3 (6) |
| Klebsiella spp. | 10 (6) | Proteus mirabilis | 1 (5) | Neisseria sp. | 1 (4) | Enterobacter spp. | 3 (6) |
| Neisseria spp. | 5 (3) | Candida spp. | 3 (6) | ||||
| Proteus mirabilis | 5 (3) | Anaerobes spp.g | 3 (6) | ||||
| Streptococcus spp. | 4 (2) | Citrobacter sp. | 1 (2) | ||||
| Hafnia alvei | 3 (2) | Proteus mirabilis | 1 (2) | ||||
| Enterococcus spp. | 3 (2) | Polymicrobial | 1 (2) | ||||
| Serratia marcescens | 3 (2) | ||||||
| Citrobacter spp. | 2 (1) | ||||||
| Candida spp. | 2 (1) | ||||||
| S. epidermidis | 1 (1) | ||||||
| Aspergillus | 1 (1) | ||||||
| Acinetobacter baumannii | 1 (1) | ||||||
| Anaerobes | 1 (1) | ||||||
a One hundred sixty-three ventilator-associated pneumonia episodes occurred in 120 patients.
b Twenty-one cannula infections occurred in 20 patients.
c Twenty-three poststernotomy mediastinitis infections occurred in 20 patients.
d Forty-seven bloodstream infections occurred in 39 patients.
e Includes ≥2 oropharyngeal pathogens.
f Includes ≥2 pathogens.
g Anaerobes include 1 Clostridium and 2 Bacteroides.
Outcomes
Extracorporeal membrane oxygenation support (8 ± 5 vs 16 ± 17 days; P < .0001), MV (14 ± 18 vs 26 ± 32 days; P < .0001) durations, and ICU (19 ± 21 vs 32 ± 25 days; P = .0004) and hospital (26 ± 29 vs 36 ± 30 days; P = .01) length of stays were significantly longer for the infected group. The cumulative probability of being infection-free was 17% (95% confidence interval [CI], 8%–28%) after 20 days of ECMO support (Figure 1). Only the SOFA score at ICU admission was independently associated with NI occurrence under ECMO by Cox regression analysis (Table 3); antibiotics at the time of ECMO cannulation was not (hazard ratio, 0.73; 95% CI, .50–1.05; P = .09).
Figure 1.
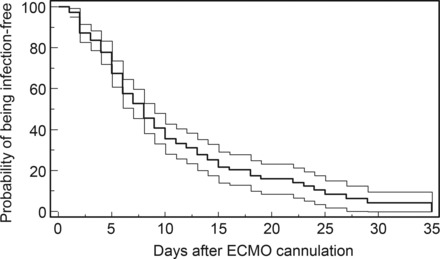
Kaplan−Meier estimates of the unadjusted cumulative probability of being free of infection (bold line) for patients with venoarterial extracorporeal membrane oxygenation. Upper and lower bands represent 95% confidence interval of the cumulative probability. Abbreviation: ECMO, extracorporeal membrane oxygenation.
Table 3.
Cox Regression Analyses of Factors Associated With First Nosocomial Infection Under Extracorporeal Membrane Oxygenation Patients
| Univariate Analysis |
Multivariate Analysis |
|||
|---|---|---|---|---|
| Factor | HR (95% CI) | P Value | HR (95% CI) | P Value |
| SAPS II scorea | 1.01 (.99–1.02) | .09 | ||
| SOFA scorea | 1.05 (1.01–1.09) | .03 | 1.04 (1.00–1.08) | .05 |
| Immunosuppressionb | 1.14 (.81–1.62) | .46 | ||
| Reason for ECMO | .38 | |||
| Miscellaneousc | 1 | |||
| Myocarditis | 0.84 (.49–1.44) | |||
| Post–cardiac surgery | 1.20 (.84–1.71) | |||
| Location of ECMO cannulation | .09 | .07 | ||
| Mobile unit | 1 | 1 | ||
| Operating room | 1.10 (.70–1.74) | 1.16 (.71–1.89) | ||
| ICU | 0.59 (.35–1.01) | 0.64 (.37–1.10) | ||
| Site of ECMO cannulation | .85 | |||
| Intrathoracic only | 1 | |||
| Extrathoracic only | 0.90 (.60–1.36) | |||
| Extrathoracic then intrathoracic | 0.99 (.65–1.51) | |||
| Antibiotics at the time of ECMOd | 1.07 (.76–1.51) | .68 | 0.73 (.50–1.05) | .09 |
Abbreviations: CI, confidence interval; ECMO, extracorporeal membrane oxygenation; HR, hazard ratio; ICU, intensive care unit; SAPS II, simplified acute physiology score; SOFA, sequential organ-failure assessment.
a Scores calculated at ICU admission. SAPS II and SOFA scores are correlated (r = 0.57; P < .0001)
b Includes patients with AIDS, solid organ transplantation, or hematological malignancy and those receiving chemotherapy, immunosuppressive agents, or long-term corticosteroid therapy.
c Includes dilated cardiomyopathy, acute myocardial infarction, cardiac arrest, and other medical indications.
d Includes patients who received prophylactic antibiotic therapy for central ECMO placement and those treated with antibiotics for an infection at the time of ECMO cannulation.
Overall survival rates for patients with NIs or without were comparable (51% vs 62%; P = .15). Survival without transplantation and/or LVAD tended to be lower for infected patients than for uninfected patients (34% vs 46%; P = .07). Twenty-seven deaths were directly attributable to NIs during ECMO support and 12 were directly attributable to NIs after ECMO withdrawal. Independent predictors of death using Cox regression analysis were higher SAPS II at ICU admission and nosocomial infection with severe sepsis or septic shock, whereas myocarditis as indication for ECMO support and immunosuppression were associated with better outcomes (Table 4).
Table 4.
Cox Regression Analyses of Factors Associated With Death in the Intensive Care Unit
| Univariate Analysis |
Multivariate Analysis |
|||
|---|---|---|---|---|
| Factor | HR (95% CI) | P Value | HR (95% CI) | P Value |
| SAPS II scorea | 1.02 (1.01–1.03) | .0002 | 1.02 (1.01–1.03) | .0001 |
| SOFA scorea | 1.06 (1.01–1.11) | .02 | ||
| Immunosuppression | 0.55 (.35–.88) | .01 | 0.54 (.33–.88) | .01 |
| Reason for ECMO | .004 | .01 | ||
| Miscellaneousb | 1 | 1.0 | ||
| Myocarditis | 0.21 (.08–.58) | 0.21 (.08–.61) | ||
| Post–cardiac surgery | 0.67 (.44–1.01) | 0.87 (.54–1.40) | ||
| Location of ECMO cannulation | .20 | |||
| Mobile unit | 1 | |||
| Operating room | 0.67 (.36–1.12) | |||
| ICU | 0.86 (.47–1.56) | |||
| Site of ECMO cannulation | .77 | |||
| Intrathoracic only | 1 | |||
| Extrathoracic only | 1.20 (.73–1.97) | |||
| Extrathoracic then intrathoracic | 1.12 (.68–1.86) | |||
| Nosocomial infection with severe sepsis or septic shockc | 1.86 (1.23–2.83) | .003 | 1.93 (1.26–2.94) | .002 |
| Antibiotics at the time of ECMOd | 0.77 (.51–1.17) | .23 | 0.68 (.43–1.08) | .10 |
Abbreviations: CI, confidence interval; ECMO, extracorporeal membrane oxygenation; HR, hazard ratio; ICU, intensive care unit; SAPS II, simplified acute physiology score; SOFA, sequential organ-failure assessment.
a Scores calculated at ICU admission. SAPS II and SOFA scores are correlated (r = 0.57; P < .0001).
b Includes dilated cardiomyopathy, acute myocardial infarction, cardiac arrest, and other medical indications.
c Nosocomial infection with severe sepsis or septic shock as a time-dependent covariate.
d Includes patients who received prophylactic antibiotic therapy for central ECMO placement and those treated with antibiotics for an infection at the time of ECMO cannulation.
DISCUSSION
We described the outcomes and infectious complications of 220 patients who received VA-ECMO for refractory cardiogenic shock. Our results indicate that 64% of these patients developed an NI while on ECMO. Ventilator-associated pneumonia, bloodstream infections, mediastinitis, and cannula infections occurred in 55%, 18%, 11%, and 10% of patients, respectively. The probability of remaining NI-free decreased after prolonged ECMO support and in case of more severe disease. As previously reported [28], higher SAPS II and severe sepsis or septic shock were independent factors associated with mortality in this patient population, whereas immunosuppression and myocarditis [16] as the reason for ECMO support were associated with better outcomes.
To date, only a few studies [9–12] reported infectious complications in adults receiving ECMO, and, in most of them, indications for ECMO support were both respiratory and cardiac failures. Indeed, for a large series from the Extracorporeal Life Support Organization (ELSO) registry, it was recently shown that infection rates were higher for adults than newborns (30.6 vs 10.1 per 1000 ECMO days) and that rates were even higher (37 per 1000 ECMO days) for adults receiving ECMO for cardiac reasons [11]. However, detailed descriptions of types of infections were not provided. Despite a rigorous program to prevent NIs in our ICU, the infection rate reported herein (75.5 per 1000 ECMO days) was higher than that of the ELSO registry [11] and other studies from single centers (12–57 episodes per 1000 ECMO days) [9, 10, 12]. Our cannula, catheter-related, and bloodstream infections and mediastinitis rates were comparable with theirs [9, 10, 12], but we had a higher NI rate, which is mainly explained by the high VAP incidence (55 episodes per 1000 ECMO days; 55% of all ECMO patients; as opposed to 4 episodes each among their 114 patients [10] and 334 patients [12]). However, the latter group acknowledged that the frequency of respiratory tract infections might have been underestimated due to their surveillance system. Indeed, our VAP rate is within the range of that previously reported for patients undergoing prolonged MV in the ICU [29–31]. In a recent randomized trial evaluating early tracheostomy after heart surgery, VAP frequency was >45% of 216 patients who underwent MV for 18 days on average [31]. The 28-day VAP rate was even higher (>60%) in another randomized study of early tracheostomy in medical patients [29] whose median MV duration was 15 days, as herein. Furthermore, despite high compliance to a multifaceted program for VAP prevention, Bouadma et al [32] have recently shown that VAP rates remain substantially high (50%–75% after 30 days of MV). However, it should be noted that despite our patients’ extreme critical status, attested by SAPS II and SOFA score, and the high NI rate, their overall survival (55%) was higher than for Burket et al [9] (51%), Sun et al [12] (32%) and Hsu et al [10] (25%) patients.
In agreement with previous reports [7, 8, 10–12, 33], rates of NIs increased after prolonged ECMO support and in case of more severe disease. Specifically, although the SOFA score at ICU admission did not differ between patients who ultimately did or did not develop infections, the time to first nosocomial infection was significantly shorter with increasing number of organ failures, suggesting reduced immune response to bacterial invasion in this setting. It should also be noted that, although no systematic antibiotic prophylaxis was administered to our patients receiving peripheral VA-ECMO, risks of developing NIs were not significantly increased for patients not receiving antibiotics at the time of ECMO cannulation. Indeed, antibiotic prophylaxis to prevent NIs in ECMO patients remains highly controversial. To decrease VAP rates, a strategy of selective digestive decontamination might be applied [34]. However, this strategy might be viewed as a double-edged sword. In patients with very prolonged MV, it might just delay the first VAP episode and select bacteria highly resistant to antibiotics. Implementation of widespread selective digestive decontamination is costly and has the potential for causing serious harm to patients, not only because of the emergence of resistance to antibiotics but also because of Clostridium difficile–associated colitis [35]. In a recent survey of 132 ELSO centers, the majority of respondents declared that antibiotic prophylaxis was administered at their center and that it was frequently prescribed for the duration of ECMO support. Although Hsu et al [10] used no standardized prophylactic antibiotic regimen, >75% of their patients received antibiotics with glycopeptides or anti-Pseudomonas agents at the time of ECMO insertion. Nonetheless, antibiotic therapy was not retained in the multivariable model of risk factors of NIs during ECMO use, and, as herein, most infections at the cannula insertion site occurred >10 days following ECMO initiation. To resolve this controversial issue, a randomized trial testing administration of a single dose of intravenous antibiotics at the time of ECMO cannulation to prevent surgical site infection against no prophylaxis for patients who receive peripheral ECMO might be designed.
It has been advocated that daily cultures of endotracheal aspirates, urine, skin and/or wound swabs, and blood samples might provide an alternative to antibiotic prophylaxis for ECMO patients [36]. Of the 132 ELSO centers recently surveyed, 50% reported performing routine surveillance cultures of blood (100%), sputum (48%), urine (44%) and others (9%; ie, wound, cannulation site, circuit cultures, throat, and rectal swabs). The periodicity of surveillance cultures was every 24–72 hours for >80% of the cases. However, this approach is costly and resource consuming, and to date, no randomized trial of routine surveillance cultures has demonstrated decreased infection-associated morbitity, compared with targeted bacterial cultures of sites of suspected infections.
We are aware of several limitations of this study. First, although this report uses one of the largest populations of VA-ECMO published to date, this is a retrospective study performed in a single canter caring for patients with acute refractory cardiogenic shock. Therefore, our results might not be applicable to centers with different case mixes. Second, we studied a mixed population of acute refractory cardiogenic shock (ie, medical, postcardiotomy, or postheart transplantation) patients who had received peripheral and/or central ECMO support, and detailed analysis of NIs in each patient subgroup was not performed. Third, we cannot exclude that the results of our multivariable analyses might have been biased by residual confounding not accounted for in this study.
In conclusion, our findings indicate that patients who received the latest generation VA-ECMO support for refractory cardiogenic shock still had a high risk of developing NIs, particularly VAP. Rates of NIs increased with longer ECMO support, and NI severity was independently associated with in-ICU death. Antibiotic prophylaxis at the time of cannula insertion or the use of chlorhexidine gluconate–impregnated sponges in cannula dressings [37] should be tested as potentially effective means of reducing cannula-related infections in this setting.
Note
Potential conflicts of interest. A. C. has received consulting fees from MAQUET. P. C. has received institutional grant support from MAQUET. All other authors report no potential conflicts.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Chen YS, Chao A, Yu HY, et al. Analysis and results of prolonged resuscitation in cardiac arrest patients rescued by extracorporeal membrane oxygenation. J Am Coll Cardiol. 2003;41:197–203. doi: 10.1016/s0735-1097(02)02716-x. [DOI] [PubMed] [Google Scholar]
- 2.Magovern GJ, Jr., Simpson KA. Extracorporeal membrane oxygenation for adult cardiac support: the Allegheny experience. Ann Thorac Surg. 1999;68:655–61. doi: 10.1016/s0003-4975(99)00581-0. [DOI] [PubMed] [Google Scholar]
- 3.Smedira NG, Moazami N, Golding CM, et al. Clinical experience with 202 adults receiving extracorporeal membrane oxygenation for cardiac failure: survival at five years. J Thorac Cardiovasc Surg. 2001;122:92–102. doi: 10.1067/mtc.2001.114351. [DOI] [PubMed] [Google Scholar]
- 4.Chen JS, Ko WJ, Yu HY, et al. Analysis of the outcome for patients experiencing myocardial infarction and cardiopulmonary resuscitation refractory to conventional therapies necessitating extracorporeal life support rescue. Crit Care Med. 2006;34:950–7. doi: 10.1097/01.CCM.0000206103.35460.1F. [DOI] [PubMed] [Google Scholar]
- 5.Combes A, Leprince P, Luyt CE, et al. Outcomes and long-term quality-of-life of patients supported by extracorporeal membrane oxygenation for refractory cardiogenic shock. Crit Care Med. 2008;36:1404–11. doi: 10.1097/CCM.0b013e31816f7cf7. [DOI] [PubMed] [Google Scholar]
- 6.Schutze GE, Heulitt MJ. Infections during extracorporeal life support. J Pediatr Surg. 1995;30:809–12. doi: 10.1016/0022-3468(95)90753-x. [DOI] [PubMed] [Google Scholar]
- 7.Steiner CK, Stewart DL, Bond SJ, Hornung CA, McKay VJ. Predictors of acquiring a nosocomial bloodstream infection on extracorporeal membrane oxygenation. J Pediatr Surg. 2001;36:487–92. doi: 10.1053/jpsu.2001.21609. [DOI] [PubMed] [Google Scholar]
- 8.Coffin SE, Bell LM, Manning M, Polin R. Nosocomial infections in neonates receiving extracorporeal membrane oxygenation. Infect Control Hosp Epidemiol. 1997;18:93–6. doi: 10.1086/647561. [DOI] [PubMed] [Google Scholar]
- 9.Burket JS, Bartlett RH, Vander Hyde K, Chenoweth CE. Nosocomial infections in adult patients undergoing extracorporeal membrane oxygenation. Clin Infect Dis. 1999;28:828–33. doi: 10.1086/515200. [DOI] [PubMed] [Google Scholar]
- 10.Hsu MS, Chiu KM, Huang YT, Kao KL, Chu SH, Liao CH. Risk factors for nosocomial infection during extracorporeal membrane oxygenation. J Hosp Infect. 2009;73:210–6. doi: 10.1016/j.jhin.2009.07.016. [DOI] [PubMed] [Google Scholar]
- 11.Bizzarro MJ, Conrad SA, Kaufman DA, Rycus P. Infections acquired during extracorporeal membrane oxygenation in neonates, children, and adults. Pediatr Crit Care Med. 2011;12:277–81. doi: 10.1097/PCC.0b013e3181e28894. [DOI] [PubMed] [Google Scholar]
- 12.Sun HY, Ko WJ, Tsai PR, et al. Infections occurring during extracorporeal membrane oxygenation use in adult patients. J Thorac Cardiovasc Surg. 2010;140:1125–32. doi: 10.1016/j.jtcvs.2010.07.017. e2. [DOI] [PubMed] [Google Scholar]
- 13.Montgomery VL, Strotman JM, Ross MP. Impact of multiple organ system dysfunction and nosocomial infections on survival of children treated with extracorporeal membrane oxygenation after heart surgery. Crit Care Med. 2000;28:526–31. doi: 10.1097/00003246-200002000-00040. [DOI] [PubMed] [Google Scholar]
- 14.Gardner AH, Prodhan P, Stovall SH, et al. Fungal infections and antifungal prophylaxis in pediatric cardiac extracorporeal life support. J Thorac Cardiovasc Surg. 2012;143:689–95. doi: 10.1016/j.jtcvs.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 15.Brown KL, Ridout DA, Shaw M, et al. Healthcare-associated infection in pediatric patients on extracorporeal life support: the role of multidisciplinary surveillance. Pediatr Crit Care Med. 2006;7:546–50. doi: 10.1097/01.PCC.0000243748.74264.CE. [DOI] [PubMed] [Google Scholar]
- 16.Mirabel M, Luyt CE, Leprince P, et al. Outcomes, long-term quality of life, and psychologic assessment of fulminant myocarditis patients rescued by mechanical circulatory support. Crit Care Med. 2011;39:1029–35. doi: 10.1097/CCM.0b013e31820ead45. [DOI] [PubMed] [Google Scholar]
- 17.Bouadma L, Luyt CE, Tubach F, et al. Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet. 2010;375:463–74. doi: 10.1016/S0140-6736(09)61879-1. [DOI] [PubMed] [Google Scholar]
- 18.Garner JS, Jarvis WR, Emori TG, Horan TC, Hughes JM. CDC definitions for nosocomial infections, 1988. Am J Infect Control. 1988;16:128–40. doi: 10.1016/0196-6553(88)90053-3. [DOI] [PubMed] [Google Scholar]
- 19.Bone RC, Balk RA, Cerra FB, et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest. 1992;101:1644–55. doi: 10.1378/chest.101.6.1644. [DOI] [PubMed] [Google Scholar]
- 20.Chastre J, Fagon JY. Ventilator-associated pneumonia. Am J Respir Crit Care Med. 2002;165:867–903. doi: 10.1164/ajrccm.165.7.2105078. [DOI] [PubMed] [Google Scholar]
- 21.Chastre J, Wolff M, Fagon JY, et al. Comparison of 8 vs 15 days of antibiotic therapy for ventilator-associated pneumonia in adults: a randomized trial. JAMA. 2003;290:2588–98. doi: 10.1001/jama.290.19.2588. [DOI] [PubMed] [Google Scholar]
- 22.Horan TC, Gaynes RP, Martone WJ, Jarvis WR, Emori TG. CDC definitions of nosocomial surgical site infections, 1992: a modification of CDC definitions of surgical wound infections. Infect Control Hosp Epidemiol. 1992;13:606–8. [PubMed] [Google Scholar]
- 23.Combes A, Trouillet JL, Joly-Guillou ML, Chastre J, Gibert C. The impact of methicillin resistance on the outcome of poststernotomy mediastinitis due to Staphylococcus aureus. Clin Infect Dis. 2004;38:822–9. doi: 10.1086/381890. [DOI] [PubMed] [Google Scholar]
- 24.Jackson GG, Arana-Sialer JA, Andersen Br Grieble HG, Mc CW. Profiles of pyelonephritis. Arch Intern Med. 1962;110:63–75. [PubMed] [Google Scholar]
- 25.Le Gall JR, Lemeshow S, Saulnier F. A new simplified acute physiology score (SAPS II) based on a European/North American multicenter study. JAMA. 1993;270:2957–63. doi: 10.1001/jama.270.24.2957. [DOI] [PubMed] [Google Scholar]
- 26.Vincent JL, Moreno R, Takala J, et al. The SOFA (sepsis-related organ failure assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the European society of intensive care medicine. Intensive Care Med. 1996;22:707–10. doi: 10.1007/BF01709751. [DOI] [PubMed] [Google Scholar]
- 27.Katz MA. Mutivariate analysis. New York: Cambridge University Press; 1999. [Google Scholar]
- 28.Vincent JL, Rello J, Marshall J, et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA. 2009;302:2323–9. doi: 10.1001/jama.2009.1754. [DOI] [PubMed] [Google Scholar]
- 29.Blot F, Similowski T, Trouillet JL, et al. Early tracheotomy versus prolonged endotracheal intubation in unselected severely ill ICU patients. Intensive Care Med. 2008;34:1779–87. doi: 10.1007/s00134-008-1195-4. [DOI] [PubMed] [Google Scholar]
- 30.Combes A, Costa MA, Trouillet JL, et al. Morbidity, mortality, and quality-of-life outcomes of patients requiring >14 days of mechanical ventilation. Crit Care Med. 2003;31:1373–81. doi: 10.1097/01.CCM.0000065188.87029.C3. [DOI] [PubMed] [Google Scholar]
- 31.Trouillet JL, Luyt CE, Guiguet M, et al. Early percutaneous tracheotomy versus prolonged intubation of mechanically ventilated patients after cardiac surgery: a randomized trial. Ann Intern Med. 2011;154:373–83. doi: 10.7326/0003-4819-154-6-201103150-00002. [DOI] [PubMed] [Google Scholar]
- 32.Bouadma L, Deslandes E, Lolom I, et al. Long-term impact of a multifaceted prevention program on ventilator-associated pneumonia in a medical intensive care unit. Clin Infect Dis. 2010;51:1115–22. doi: 10.1086/656737. [DOI] [PubMed] [Google Scholar]
- 33.O'Neill JM, Schutze GE, Heulitt MJ, Simpson PM, Taylor BJ. Nosocomial infections during extracorporeal membrane oxygenation. Intensive Care Med. 2001;27:1247–53. doi: 10.1007/s001340101029. [DOI] [PubMed] [Google Scholar]
- 34.de Smet AM, Kluytmans JA, Cooper BS, et al. Decontamination of the digestive tract and oropharynx in ICU patients. N Engl J Med. 2009;360:20–31. doi: 10.1056/NEJMoa0800394. [DOI] [PubMed] [Google Scholar]
- 35.Paterson DL. Collateral damage” from cephalosporin or quinolone antibiotic therapy. Clin Infect Dis. 2004;38(Suppl 4):S341–5. doi: 10.1086/382690. [DOI] [PubMed] [Google Scholar]
- 36.Kaczala GW, Paulus SC, Al-Dajani N, et al. Bloodstream infections in pediatric ECLS: usefulness of daily blood culture monitoring and predictive value of biological markers. The British Columbia experience. Pediatr Surg Int. 2009;25:169–73. doi: 10.1007/s00383-008-2299-1. [DOI] [PubMed] [Google Scholar]
- 37.Timsit JF, Schwebel C, Bouadma L, et al. Chlorhexidine-impregnated sponges and less frequent dressing changes for prevention of catheter-related infections in critically ill adults: a randomized controlled trial. JAMA. 2009;301:1231–41. doi: 10.1001/jama.2009.376. [DOI] [PubMed] [Google Scholar]


