Abstract
While there are broad differential diagnoses for either the clinical finding of hemichorea-hemiballism or the imaging finding of lateralizing/asymmetric basal ganglia lesions (hyperdense on computed tomography, hyperintense on T1 magnetic resonance imaging), the presence of both findings is highly suggestive of nonketotic hyperglycemia. We present an unusual case of a patient with vague stroke-like complaints and imaging findings notable for lateralizing basal ganglia lesions. Laboratory analysis revealed nonketotic hyperglycemia and neurologic exam failed to elicit any findings of movement disorder. As far as we know, this is the first published case of a patient with typical neuroimaging manifestations of nonketotic hyperglycemia without associated hemichorea-hemiballism (a disorder of abnormal movements comprised of more proximal, higher amplitude movements-ballismus and lower amplitude, more distal movements-chorea). This finding suggests that radiologists should be alert to the possibility of nonketotic hyperglycemia in patients with asymmetric/lateralizing basal ganglia lesions even in the absence of a movement disorder.
Keywords: Nonketotic hyperglycemia, T1 hyperintense basal ganglia lesion, hemichorea-hemiballismus
CASE REPORT
A 42 year-old African American male presented to the emergency department with a 1 week history of progressively worsening generalized weakness, lightheadedness and “cloudiness,” as well as diffuse “achy” shoulder and back pain. Additionally, the patient reported a history of his legs feeling “wobbly” and falling once during the week prior to presentation. He eventually presented to the emergency room out of concern he was suffering a stroke. Pertinent past medical history included type II diabetes, hypertension and residual left sided weakness and gait problems from a prior right pontine infarct which occurred approximately 10 months prior to the current presentation. The patient had prescriptions for nifedipine, subcutaneous insulin, folate, and vibramycin. He did not take neuroleptic medication.
The patient’s neurologic exam revealed mild right sided deficits which were new, including flattening of the nasolabial fold on the right and a slight right pronator drift, although significant right sided weakness could not be elicited on formal strength testing. In addition, there was residual weakness and hyperreflexia on the left side from his prior infarct. The remainder of the neurological exam was unremarkable. Specifically, there was no evidence of hemichorea-hemiballism (HC-HB), a disorder of abnormal movements comprised of more proximal, higher amplitude movements-ballismus; and lower amplitude, more distal movements-chorea.
Due to concern for acute ischemia, a non-contrast head computed tomography = (CT) exam was performed in the emergency department which demonstrated hyperdensity within the caudate nuclei and left lentiform nucleus which was new from a study performed ten months earlier (Figure 1). Contrast enhanced magnetic resonance imaging (MRI) of the brain was subsequently obtained within 12 hours of the initial CT (Figures 2–6) and demonstrated corresponding intrinsic T1 shortening in the left globus pallidus (GP) with contrast enhancement in the lentiform and caudate nuclei (left much greater than right) (Figures 4,6). There was also faint fluid attenuated inversion recovery (FLAIR) signal abnormality in the GP (left greater than right) and left putamen (Figure 5). Diffusion weighted imaging was unremarkable apart for some very faint left putamen restricted diffusion without correlative FLAIR hyperintensity (FLAIR hyperintensity in the left putamen did not quite correspond to the site of apparent diffusion restriction), and there was no abnormality on susceptibility-weighted imaging (SWI) to suggest hemorrhage/blood products (Figures 3,2). Consistent with the patient’s history of prior pontine infarct, the MR demonstrated the sequela of an old stroke involving the right corticospinal and corticobulbar tracts from the cerebral peduncle superiorly through the medulla inferiorly. The basal ganglia (BG) imaging findings were regarded as highly suggestive of nonketotic hyperglycemia, even though no movement disorder was detected on clinical exam. Given the CT findings of hyperdensity without evidence of hemorrhage on MRI, subacute infarction with petechial hemorrhage was discarded as a possible etiology.
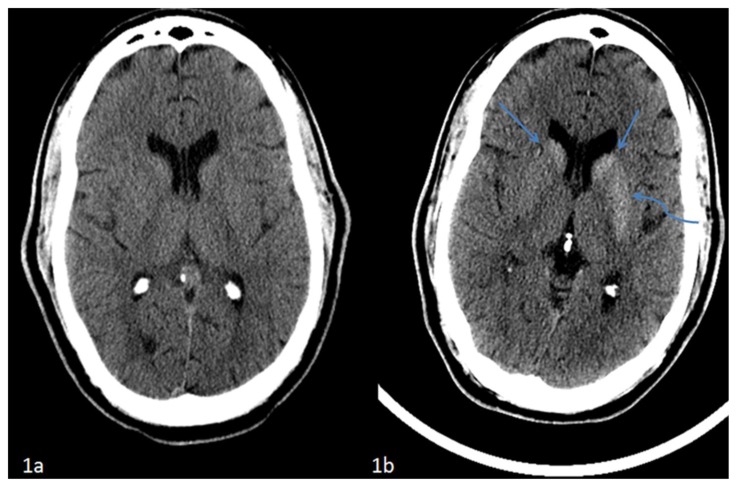
Figure 1.
42 year-old male who presented with vague right sided stroke-like symptoms and was found to have nonketotic hyperglycemia. FINDINGS: (1a) Axial non-contrast computed tomography of the head performed at ten months prior to current presentation demonstrates no abnormalities at the level of the basal ganglia. TECHNIQUE: Philips 64-slice CT scanner, 501mAs, 120 kvp, 5mm slice thickness. FINDINGS: (1b) Axial non-contrast computed tomography of the head performed at time of current presentation demonstrates interval development of hyperdensity within the caudate nuclei (straight arrows) and left lentiform nucleus (curved arrow) which respects the neuroanatomic boundaries of the basal ganglia without associated edema. TECHNIQUE: Philips 64-slice CT scanner, 350mAs, 120 kvp, 5mm slice thickness.
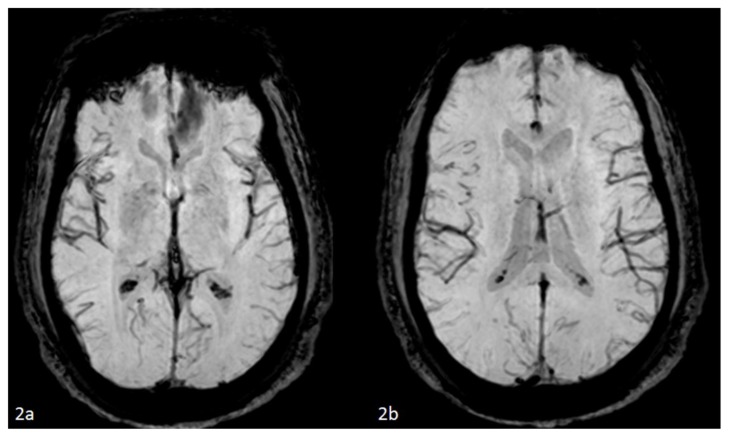
Figure 2.
42 year-old male who presented with vague right sided stroke-like symptoms and was found to have nonketotic hyperglycemia. FINDINGS: Axial SWI MR images of the brain (2a) at the level of the basal ganglia and (2b) slightly superior at the level of the atria of the lateral ventricles performed at time of current presentation are unremarkable without basal ganglia or caudate nuclei blooming to suggest blood products.. TECHNIQUE: 1.5T Philips Intera MR scanner, susceptibility-weighted imaging (VEN_BOLD_HR SENSE) sequence, TR=23.60, TE=33.60, no contrast.
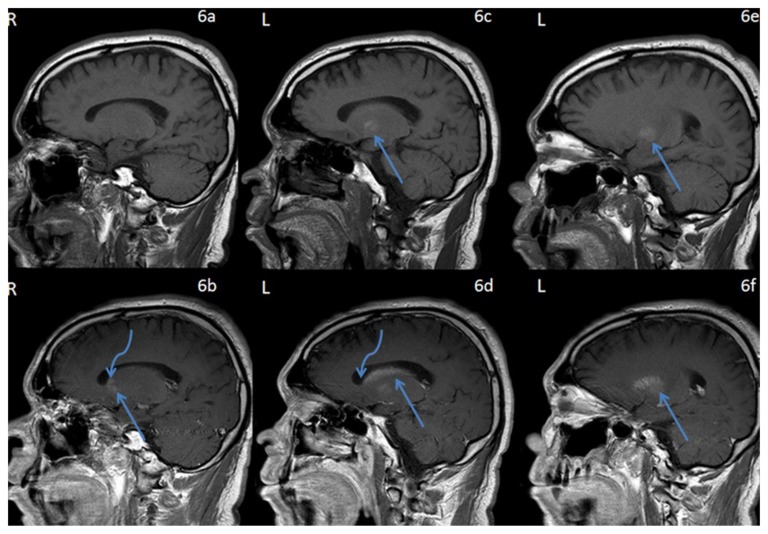
Figure 6.
42 year-old male who presented with vague right sided stroke-like symptoms and was found to have nonketotic hyperglycemia. FINDINGS: Sagittal T1-weighted pre-contrast MR images of the brain through the basal ganglia and caudate nuclei (6a) right and (6c,6e) left demonstrates intrinsic T1 shortening confined to the left globus pallidus (arrows). TECHNIQUE: 1.5T Philips Intera MR scanner, T1-weighted imaging, TR=541.53, TE=12, pre-contrast. Sagittal T1-weighted post-contrast MR images of the brain through the basal ganglia and caudate nuclei, (6b) corresponding to (6a) on the right and (6d,f) corresponding to (6c,e) respectively on the left demonstrate contrast enhancement which respects the neuroanatomic boundaries of the lentiform (straight arrows) and caudate nuclei (curved arrows) (left much greater than right). TECHNIQUE: 1.5T Philips Intera MR scanner, T1-weighted imaging, TR=541.53, TE=12, with 18ml of gadodiamide (Omniscan, GE health care, Princeton, NJ) injection.
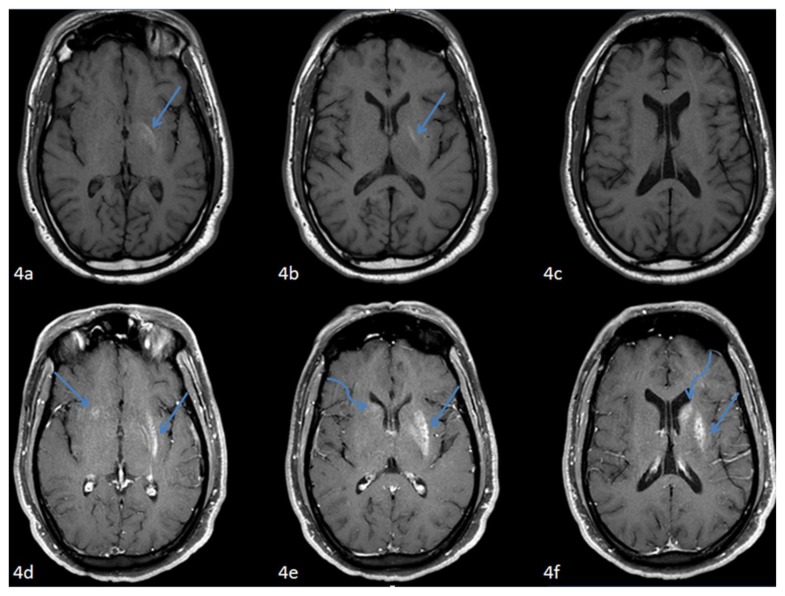
Figure 4.
42 year-old male who presented with vague right sided stroke-like symptoms and was found to have nonketotic hyperglycemia. FINDINGS: Axial pre-contrastT1-weighted MR images of the brain through the basal ganglia and caudate nuclei from caudal to cranial (4a–c) demonstrate intrinsic T1 shortening confined to the left globus pallidus (arrows), corresponding to the hyperdensity seen on presenting CT. TECHNIQUE: 1.5T Philips Intera MR scanner, T1-weighted imaging, TR=586.65, TE=12, pre-contrast. FINDINGS: Axial post-contrast T1-weighted MR images of the brain through the basal ganglia and caudate nuclei from caudal to cranial (4d–f) corresponding to (4a–c) respectively demonstrate contrast enhancement respecting the neuroanatomic boundaries of the lentiform (straight arrows) and caudate nuclei (curved arrows) (left much greater than right). TECHNIQUE: 1.5T Philips Intera MR scanner, fat saturated T1-weighted imaging, TR=595.10, TE=12, with 18ml of gadodiamide (Omniscan, GE health care, Princeton, NJ) injection.
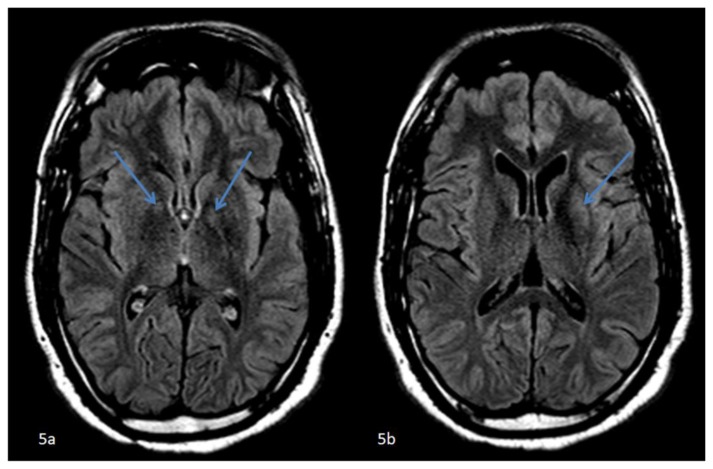
Figure 5.
42 year-old male who presented with vague right sided stroke-like symptoms and was found to have nonketotic hyperglycemia. . FINDINGS: Axial FLAIR MR images of the brain at the level of the basal ganglia (5a) slightly caudal to (5b) demonstrate subtle hyperintense signal abnormalities in the globus pallidi (left greater than right) (arrows). (5b) FLAIR hyperintense signal abnormality confined to the left putamen (arrow). TECHNIQUE: 1.5T Philips Intera MR scanner, fluid attenuated inversion recovery imaging (FLAIR), TR=11000, TE=140, no contrast.
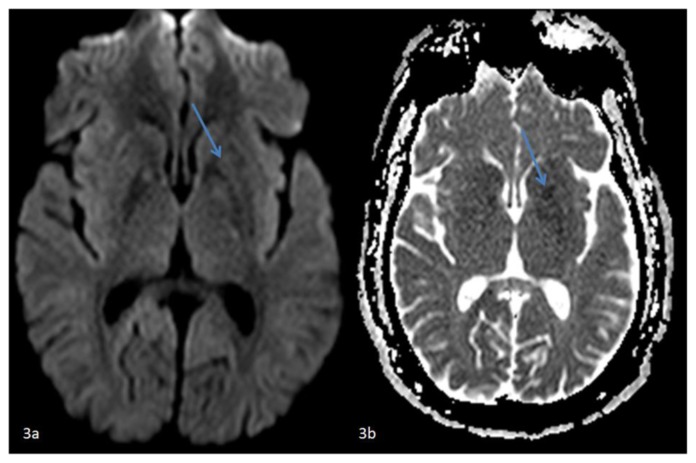
Figure 3.
42 year-old male who presented with vague right sided stroke-like symptoms and was found to have nonketotic hyperglycemia. FINDINGS: (3a) Axial DWI MR image of the brain at the level of the basal ganglia and (3b) axial ADC map correlate demonstrate faint restricted diffusion confined to the left putamen. TECHNIQUE: (3a) 1.5T Philips Intera MR scanner, diffusion-weighted imaging (DWI), TR=3873.32, TE=73.10, no contrast and (3b)1.5T Philips Intera MR scanner, diffusion-weighted ADC map, TR=3873.32, TE=73.10, no contrast.
Laboratory evaluation was notable for serum glucose of 323 mg/dL and a hemoglobin A1c concentration of 13.1% (normal values are 60–109 mg/dL and 3.9–6.1% respectively). The urine specimen was negative for ketones. Calcium level was 8.5 mg/dL (normal 8.4–10.2) and magnesium was 1.6 mg/dL (normal 1.6–2.5). Supported by the imaging findings, these laboratory tests were interpreted as consistent with nonketotic hyperglycemia in the context of poorly controlled diabetes. The patient later admitted to non-compliance with his insulin regimen. His symptoms improved and his neurologic exam returned to baseline with insulin administration and the normalization of his serum glucose level. Therefore, the presumed etiology for his initial neurologic symptoms was a global neurologic dysfunction secondary to hyperglycemia. After a four day hospitalization, the patient was discharged in his usual state of health and instructed to follow-up as an outpatient.
DISCUSSION
Nonketotic hyperglycemia affects patients with poorly controlled diabetes mellitus who classically present with the clinical finding of HC-HB [1]. This is a rare condition with the largest published case series consisting of only 53 patients. The majority of reported cases have involved Asians, with a female to male ratio of 1.76. This condition is thought to occur most frequently in the elderly, most commonly in the seventh decade [2].
In the radiology literature, nonketotic hyperglycemia has been associated with a characteristic appearance of unilateral or asymmetric lesions of the BG which are typically contralateral to the side of the patient’s presenting symptoms. The most frequently described finding associated with nonketotic hyperglycemia on non-contrast head CT examination is a hyperdense putamen and/or caudate nucleus contralateral to the side of the patient’s symptoms [1]. The most commonly described MRI findings associated with nonketotic hyperglycemia include high signal intensity BG lesions on T1-weighted brain MRI on the side contralateral to patient symptoms. The putamen is almost always involved with variable additional associated BG lesions. There have been no reports of isolated involvement of the GP or caudate nucleus. The T2-weighted findings are much more variable with the majority of BG lesions described as either hypo-intense or iso-intense to background normal BG[2]. Relatively little has been published regarding nonketotic hyperglycemia and findings on DWI and SWI. A small case series and case report have demonstrated mild to moderate restricted diffusion corresponding to the T1-weighted hyperintense BG lesions [3–5]. On SWI, results have been mixed with some of these cases demonstrating susceptibility while others have not [3–4]. Most of the published reports did not use contrast, and those that did use contrast generally indicate no appreciable enhancement [5]. However, at least one case report describes BG enhancement with resolution once the serum glucose level was corrected [6]. On follow-up MRI, Oh et al reported the resolution of T1 signal abnormalities in 19 of 22 patients presenting with nonketotic hyperglycemia, suggesting that the neuroimaging findings of nonketotic hyperglycemia are usually reversible with appropriate treatment [2]. When performed, nuclear scintigraphy has demonstrated hypoperfusion in the BG with abnormal MRI/CT signal characteristics, though in some cases after transient hyperperfusion [1–2,5].
The etiology of the hyperintense signal observed on T1-weighted imaging has been the subject of controversy with several competing hypotheses proposed to account for these findings. The most widely promoted hypothesis ascribes the T1 hyperintensity to the protein hydration layer in the cytoplasm of swollen gemistocytes (reactive astrocytes) [3,5]. Supporting this hypothesis, Shan et al. reported that a biopsy of a hyperintense putaminal lesion demonstrated abundant gemistocytes in a fragment of brain tissue [7]. The most common alternate hypothesis is that the signal abnormality represents putaminal petechial hemorrhage [7]. However, blood products are felt to be a less likely etiology given the manner in which the T1 hyperintensity respects neuroanatomic boundaries, lack of consistent positive SWI findings and lack of expected methemoglobin T2-weighted hyperintense evolution [3]. Nagai et al. has suggested that demyelination similar to that seen in diabetic peripheral neuropathy leads to exchange of myelin-bound and axonal water with resultant T1 shortening [8]. Finally, some have speculated that the T1 shortening reflects transient ischemic changes or desiccation seen as part of localized Wallerian degeneration [2,4].
It is important to remember that T1-weighted hyperintensity involving the lentiform nucleus does have a differential diagnosis, particularly if symmetric/bilateral, and must be interpreted in the context of the patient’s past medical history, presentation and laboratory values to arrive at the correct diagnosis. Similar T1-weighted hyperintense findings on neuroimaging have been observed with manganese toxicity in long-term parenteral nutrition, chronic liver disease, hypoxic-ischemic changes, disorders of calcium metabolism (i.e. hypo- or hyperparathyroidism, etc), Fahr disease, lupus, neurofibromatosis and Wilson disease [5,9,10]. While the majority of the aforementioned metabolic and systemic conditions typically present with bilateral, symmetric T1 hyperintense BG lesions, as opposed to the classic unilateral BG findings in nonketotic hyperglycemia, it is important to remember that nonketotic hyperglycemia can also present with bilateral T1 hyperintense BG lesions. Differential considerations for asymmetric/unilateral BG hyperattenuation on non-contrast head CT and intrinsic T1 hyperintensity on MRI include hemorrhage/blood products and asymmetric calcification/mineralization including that associated with underlying lesions such as developmental venous anomalies [11].
The typical imaging manifestations of nonketotic hyperglycemia are closely associated with HC-HB. In the largest published case series of nonketotic hyperglycemia and BG lesions, all 53 patients demonstrated either unilateral or bilateral ballismus-chorea [2]. Mechanistically, this association with movement disorders is unsurprising as the BG(GP, putamen, caudate, subthalamic nucleus and substantia nigra) comprise a functional circuit within the deep grey matter structures of the cerebral hemispheres which controls movement through its influence on thalamocortical tracts. Dysfunction within this circuit will therefore lead to a myriad of different movement disorders including ballismus (more proximal, higher amplitude movements) and chorea (lower amplitude, more distal movements), which often occur together clinically. While HC-HB, has been classically thought to most commonly reflect an underlying subthalamic nucleus lesion on the contralateral side, imaging evidence suggests that lesions elsewhere in the BG are actually more common in HC-HB [12–13]. Other well documented etiologies for HC-HB include ischemic or hemorrhagic stroke and tumors [12].
Nonketotic hyperglycemia is speculated to cause HC-HB because it forces cerebral metabolism to shift to the anaerobic pathway abandoning the Krebs cycle. The brain responds by using an alternative source of energy which includes the metabolization of the inhibitory neurotransmitter GABA into succinic acid. In nonketotic hyperglycemia, GABA and acetate are rapidly depleted and not readily resynthesized, leading to a reduction of acetylcholine synthesis as well. It is hypothesized that limited availability of both acetylcholine and GABA in the BG, coupled with metabolic acidosis and lack of energy production, may result in BG dysfunction presenting clinically as chorea [2]. The reason for selective vulnerability of one side’s BG remains unclear.
Our case of nonketotic hyperglycemia demonstrated the classic finding of CT hyperdensity and T1-weighted hyperintensity in the left BG, referable to the side of the patient’s presenting symptoms of mild weakness on the right. In this case, there was also left side predominant FLAIR signal increase and enhancement of the BG. On DWI, there was only faint restricted diffusion in the left putamen. On SWI, the BG demonstrated no convincing abnormalities. As with prior cases of nonketotic hyperglycemia, the imaging findings in the BG conformed to the neuroanatomic boundaries of the caudate nuclei and left lentiform nucleus without associated mass effect or edema. Along with the SWI data, this pattern argues against parenchymal hemorrhage or infarction (infarctions and hemorrhage typically extend outside the BG into the internal capsule and incite some mass effect). Follow-up imaging for our patient was not available to evaluate for residual enhancement after normalization of serum glucose. However, the imaging findings in this case are typical of those previously reported for nonketotic hyperglycemia, and not consistent with other entities in the differential diagnosis (see table 2).
Table 2.
Differential diagnosis table for basal ganglia signal abnormalities
| Entity | Imaging Findings: MRI | Imaging Findings: CT | Clinical Findings |
|---|---|---|---|
| Physiologic calcification, normal variant [15] |
|
CT: Calcifications in same distribution as MR |
|
| Manganese toxicity in long term total parenteral nutrition [9] |
|
CT: Unremarkable |
|
| Disorders of calcium metabolism [16–17] |
|
CT: Bilateral BG calcifications |
|
| Chronic liver disease [18–19] |
|
|
|
| Wilson disease [20] |
|
MR: T2 “face of giant panda sign” normal red nucleus on background of hyperintense tegmentum
|
|
| Fahr disease [21–22] |
|
CT: Extensive symmetric BG calcifications (especially GP), thalami, cerebellum (dentate nuclei) |
|
| Hypoxic brain changes [23–24] |
|
CT: If acute, normal
|
|
| Neurofibromatosis type one [25–26] |
|
CT?: BG unremarkable; optic or parenchymal gliomas may be present; sphenoid wing dysplasia |
|
What makes this case unusual is the absence of chorea or another movement disorder in association with this pathognomonic imaging appearance of the BG. Instead, the patient had a subjective sensation of generalized weakness and mild right sided weakness on neurologic exam, initially concerning for a new ischemic insult. There has been a case report of nonketotic hyperglycemia presenting acutely with stroke-like symptoms, but even in this report the clinical presentation included chorea [4]. To the best of our knowledge, our case is the first in the literature to describe the classic neuroimaging findings of nonketotic hyperglycemia in a patient who did not clinically have HC-HB or any other findings suggestive of a movement disorder. Therefore, this case suggests that nonketotic hyperglycemia may have a broader spectrum of clinical presentation than is currently appreciated even when it has characteristic involvement of the BG. This observation is concordant with the well-described phenomenon in neurology that hyperglycemia (as well as hypoglycemia) can present with global neurologic dysfunction (encephalopathy and coma) as well as focal neurologic deficits (hemiplegia, aphasia and seizures) [14].
TEACHING POINT
Nonketotic hyperglycemia should be a diagnostic consideration even without hemichorea-hemiballismus when the typical neuroimaging findings are present. This is an important diagnosis to make as early as possible because the clinical outcome is excellent with the rapid institution of appropriate insulin therapy and the radiologist may be the first to appreciate the importance of the patient’s hyperglycemia to their clinical presentation given the characteristic imaging appearance.
Table 1.
Summary table for nonketotic hyperglycemia
| Etiology: | Controversial, leading hypothesis states hyperintense T1-weighted signal is secondary to unwrapping of myelin sheath and swollen gemistocytes [3,5] |
| Incidence: | Rare; largest published case series consists of 53 patients, majority of reported cases involve Asians [2] |
| Gender ratio: | Of the 53 cases with gender available, 30 females and 17 males (ratio of F to M: 1.76) [2] |
| Age predilection: | Range: 22–92 years, mean age of 71.1 years [2] |
| Risk factors: | History of poorly controlled diabetes mellitus [2] |
| Treatment: | Normalization of blood glucose levels with appropriate insulin therapy [2] |
| Prognosis: | Excellent with timely instillation of insulin therapy [2] |
| Findings on imaging: | MR: unilateral or asymmetric T1-weighted hyperintense BG lesion, T2-variable, susceptibility and diffusion - variable, often enhances [2] |
ABBREVIATIONS
- BG
basal ganglia
- CT
computed tomography
- DWI
diffusion-weighted imaging
- FLAIR
fluid attenuated inversion recovery imaging
- GP
globus pallidus
- HC-HB
hemichorea-hemiballism
- MRI
magnetic resonance imaging
- SWI
susceptibility-weighted imaging
REFERENCES
- 1.Lai PH, Tien RD, Chang MH, et al. Chorea-ballismus with nonketotic hyperglycemia in primary diabetes mellitus. AJNR Am J Neuroradiol. 1996;17:1057–64. [PMC free article] [PubMed] [Google Scholar]
- 2.Oh SH, Lee KY, Im JH, Lee MS. Chorea associated with non-ketotic hyperglycemia and hyperintensity basal ganglia lesion on T1-weighted brain MRI study: a meta-analysis of 53 cases including four present cases. J Neurol Sci. 2002;200:57–62. doi: 10.1016/s0022-510x(02)00133-8. [DOI] [PubMed] [Google Scholar]
- 3.Cherian A, Thomas B, Baheti NN, Chemmanam T, Kesavadas C. Concepts and controversies in nonketotic hyperglycemia-induced hemichorea: further evidence from susceptibility-weighted MR imaging. J Magn Reson Imaging. 2009;29:699–703. doi: 10.1002/jmri.21672. [DOI] [PubMed] [Google Scholar]
- 4.Wintermark M, Fischbein NJ, Mukherjee P, Yuh EL, Dillon WP. Unilateral putaminal CT, MR, and diffusion abnormalities secondary to nonketotic hyperglycemia in the setting of acute neurologic symptoms mimicking stroke. AJNR Am J Neuroradiol. 2004;25:975–76. [PMC free article] [PubMed] [Google Scholar]
- 5.Lee EJ, Choi JY, Lee SH, Song SY, Lee YS. Hemichorea-hemiballism in primary diabetic patients: MR Correlation. J Comput Assist Tomogr. 2002;26:905–11. doi: 10.1097/00004728-200211000-00009. [DOI] [PubMed] [Google Scholar]
- 6.Iwata A, Koike F, Arasaki K, Tamaki M. Blood brain barrier destruction in hyperglycemic chorea in a patient with poorly controlled diabetes. J Neurol Sci. 1999;163:90–93. doi: 10.1016/s0022-510x(98)00325-6. [DOI] [PubMed] [Google Scholar]
- 7.Shan DE, Ho DM, Chang C, et al. Hemichorea-hemiballism: an explanation for MR signal changes. AJNR Am J Neuroradiol. 1998;19:863–70. [PMC free article] [PubMed] [Google Scholar]
- 8.Nagai C, Kato T, Katagiri T, Sasaki H. Hyperintense putamen on T1-weighted MR images in a case of chorea with hyperglycemia. AJNR Am J Neuroradiol. 1995;16:1243–46. [PMC free article] [PubMed] [Google Scholar]
- 9.Lai PH, Chen C, Liang HL, Pan HB. Hyperintense basal ganglia on T1-weighted MR imaging. AJR Am J Roentgenol. 1999;172:1109–15. doi: 10.2214/ajr.172.4.10587157. [DOI] [PubMed] [Google Scholar]
- 10.Kashihara K, Nakashima S, Kohira I, Shohomori T, Fujiwara Y, Kuroda S. Hyperintense basal ganglia on T1- weighted MR images in a patient with central nervous system lupus and chorea. AJNR Am J Neuroradiol. 1998;19:284–86. [PMC free article] [PubMed] [Google Scholar]
- 11.Dehkharghani S, Dillon WP, Bryant SO, Fischbein NJ. Unilateral calcifications of the caudate and putamen: association with underlying development venous anomaly. AJNR Am J Neuroradiol. 2010;31:1848–52. doi: 10.3174/ajnr.A2199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Daroff RB. Bradley’s neurology in clinical practice. 6th edition [Google Scholar]; Jankovic J, Lang AE. Chapter 21 Movement disorders: diagnosis and assessment. p. 250.p. 271. [Google Scholar]; Jankovic J. Chapter 71 Movement disorders. 2012. pp. 1762–63. An Imprint of Elsevier. [Google Scholar]
- 13.Postuma RB, Lang AE. Hemiballism: revisiting a classic disorder. Lancet Neurol. 2003;2:661–68. doi: 10.1016/s1474-4422(03)00554-4. [DOI] [PubMed] [Google Scholar]
- 14.Arieff A, Guisado R. Neurologic manifestations of diabetic comas: correlation with biochemical alterations in the brain. J of Metabolism. 1975;24:665–79. doi: 10.1016/0026-0495(75)90146-8. [DOI] [PubMed] [Google Scholar]
- 15.Milton WJ, Atlas SW, Lexa FJ, Mozley PD, Gur RE. Deep gray matter hypointensity with aging in healthy adults: MR imaging at 1.5 T. Radiology. 1999;181:715–19. doi: 10.1148/radiology.181.3.1947087. [DOI] [PubMed] [Google Scholar]
- 16.You JS, Kim HJ, Chung SP, Park S. Intracranial bilateral symmetric calcifications in hypoparathyroidism. Emerg Med J. 2008;25:162. doi: 10.1136/emj.2007.051052. [DOI] [PubMed] [Google Scholar]
- 17.Dorenbeck U, Leingartner T, Bretschneider T, Kramer BK, Feuerbach S. Tentorial and dural calcification with tertiary hyperparathyroidism: a rare entity in chronic renal failure. Eur Radiol. 2002;12(Suppl 3):S11–3. doi: 10.1007/s00330-002-1406-2. [DOI] [PubMed] [Google Scholar]
- 18.Rovira A, Alonso J, Cordoba J. MR imaging findings in hepatic encephalopathy. AJNR Am J Neuroradiol. 2008;29:1612–21. doi: 10.3174/ajnr.A1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pujol A, Pujol J, Graus F, et al. Hyperintense globus pallidus on T1-weighted MRI in cirrhotic patients is associated with severity of liver failure. Neurology. 1993;43:65–69. doi: 10.1212/wnl.43.1_part_1.65. [DOI] [PubMed] [Google Scholar]
- 20.Kim TJ, Kim IO, Kim WS, et al. MR imaging of the brain in Wilson disease of childhood: findings before and after treatment with clinical correlation. AJNR Am J Neuroradiol. 2006;27:1373–80. [PMC free article] [PubMed] [Google Scholar]
- 21.Smith AB, Smirniotopoulos JG, Rushing EJ, Goldstein SJ. Bilateral thalamic lesions. AJR Am J Roentgenol. 2009;192:53–62. doi: 10.2214/AJR.08.1585. [DOI] [PubMed] [Google Scholar]
- 22.Hegde AN, Mohan S, Lath N, Lim CC. Differential diagnosis for bilateral abnormalities of the basal ganglia and thalamus. Radiographics. 2011;31:5–30. doi: 10.1148/rg.311105041. [DOI] [PubMed] [Google Scholar]
- 23.Huang BY, Castillo M. Hypoxic-ischemic brain injury: imaging findings from birth to adulthood. Radiographics. 2008;28:417–39. doi: 10.1148/rg.282075066. [DOI] [PubMed] [Google Scholar]
- 24.Kjos BO, Brant-Zawadzki M, Young RG. Early CT findings of global central nervous system hypoperfusion. AJR Am J Roentgenol. 1983;141:1227–32. doi: 10.2214/ajr.141.6.1227. [DOI] [PubMed] [Google Scholar]
- 25.DiPaolo DP, Zimmerman RA, Rorke LB, Zackai EH, Bilaniuk LT, Yachnis AT. Neurofibromatosis type 1: pathologic substrate of high-signal intensity foci in the brain. Radiology. 1995;195:721–24. doi: 10.1148/radiology.195.3.7754001. [DOI] [PubMed] [Google Scholar]
- 26.Cairns AG, North KN. Cerebrovascular dysplasia in neurofibromatosis type 1. J Neurol Neurosurg Psychiatry. 2008;79:1165–70. doi: 10.1136/jnnp.2007.136457. [DOI] [PubMed] [Google Scholar]