Abstract
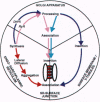
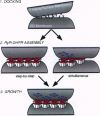
During excitation-contraction (e-c) coupling of striated muscle, depolarization of the surface membrane is converted into Ca2+ release from internal stores. This process occurs at intracellular junctions characterized by a specialized composition and structural organization of membrane proteins. The coordinated arrangement of the two key junctional components--the dihydropyridine receptor (DHPR) in the surface membrane and the ryanodine receptor (RyR) in the sarcoplasmic reticulum--is essential for their normal, tissue-specific function in e-c coupling. The mechanisms involved in the formation of the junctions and a potential participation of DHPRs and RyRs in this process have been subject of intensive studies over the past 5 years. In this review we discuss recent advances in understanding the organization of these molecules in skeletal and cardiac muscle, as well as their concurrent and independent assembly during development of normal and mutant muscle. From this information we derive a model for the assembly of the junctions and the establishment of the precise structural relationship between DHPRs and RyRs that underlies their interaction in e-c coupling.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams B. A., Beam K. G. A novel calcium current in dysgenic skeletal muscle. J Gen Physiol. 1989 Sep;94(3):429–444. doi: 10.1085/jgp.94.3.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adams B. A., Beam K. G. Muscular dysgenesis in mice: a model system for studying excitation-contraction coupling. FASEB J. 1990 Jul;4(10):2809–2816. doi: 10.1096/fasebj.4.10.2165014. [DOI] [PubMed] [Google Scholar]
- Adams B. A., Tanabe T., Mikami A., Numa S., Beam K. G. Intramembrane charge movement restored in dysgenic skeletal muscle by injection of dihydropyridine receptor cDNAs. Nature. 1990 Aug 9;346(6284):569–572. doi: 10.1038/346569a0. [DOI] [PubMed] [Google Scholar]
- Beam K. G., Knudson C. M., Powell J. A. A lethal mutation in mice eliminates the slow calcium current in skeletal muscle cells. Nature. 1986 Mar 13;320(6058):168–170. doi: 10.1038/320168a0. [DOI] [PubMed] [Google Scholar]
- Block B. A., Imagawa T., Campbell K. P., Franzini-Armstrong C. Structural evidence for direct interaction between the molecular components of the transverse tubule/sarcoplasmic reticulum junction in skeletal muscle. J Cell Biol. 1988 Dec;107(6 Pt 2):2587–2600. doi: 10.1083/jcb.107.6.2587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bossen E. H., Sommer J. R., Waugh R. A. Comparative stereology of the mouse and finch left ventricle. Tissue Cell. 1978;10(4):773–784. doi: 10.1016/0040-8166(78)90062-9. [DOI] [PubMed] [Google Scholar]
- Brandt N. R., Caswell A. H., Wen S. R., Talvenheimo J. A. Molecular interactions of the junctional foot protein and dihydropyridine receptor in skeletal muscle triads. J Membr Biol. 1990 Feb;113(3):237–251. doi: 10.1007/BF01870075. [DOI] [PubMed] [Google Scholar]
- Cannell M. B., Cheng H., Lederer W. J. The control of calcium release in heart muscle. Science. 1995 May 19;268(5213):1045–1049. doi: 10.1126/science.7754384. [DOI] [PubMed] [Google Scholar]
- Carl S. L., Felix K., Caswell A. H., Brandt N. R., Ball W. J., Jr, Vaghy P. L., Meissner G., Ferguson D. G. Immunolocalization of sarcolemmal dihydropyridine receptor and sarcoplasmic reticular triadin and ryanodine receptor in rabbit ventricle and atrium. J Cell Biol. 1995 May;129(3):673–682. doi: 10.1083/jcb.129.3.673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caswell A. H., Brandt N. R., Brunschwig J. P., Purkerson S. Localization and partial characterization of the oligomeric disulfide-linked molecular weight 95,000 protein (triadin) which binds the ryanodine and dihydropyridine receptors in skeletal muscle triadic vesicles. Biochemistry. 1991 Jul 30;30(30):7507–7513. doi: 10.1021/bi00244a020. [DOI] [PubMed] [Google Scholar]
- Caswell A. H., Lau Y. H., Garcia M., Brunschwig J. P. Recognition and junction formation by isolated transverse tubules and terminal cisternae of skeletal muscle. J Biol Chem. 1979 Jan 10;254(1):202–208. [PubMed] [Google Scholar]
- Chaudhari N. A single nucleotide deletion in the skeletal muscle-specific calcium channel transcript of muscular dysgenesis (mdg) mice. J Biol Chem. 1992 Dec 25;267(36):25636–25639. [PubMed] [Google Scholar]
- Chaudhari N., Beam K. G. mRNA for cardiac calcium channel is expressed during development of skeletal muscle. Dev Biol. 1993 Feb;155(2):507–515. doi: 10.1006/dbio.1993.1048. [DOI] [PubMed] [Google Scholar]
- Corbett A. M., Caswell A. H., Brandt N. R., Brunschwig J. P. Determinants of triad junction reformation: identification and isolation of an endogenous promotor for junction reformation in skeletal muscle. J Membr Biol. 1985;86(3):267–276. doi: 10.1007/BF01870606. [DOI] [PubMed] [Google Scholar]
- Courbin P., Koenig J., Ressouches A., Beam K. G., Powell J. A. Rescue of excitation-contraction coupling in dysgenic muscle by addition of fibroblasts in vitro. Neuron. 1989 Apr;2(4):1341–1350. doi: 10.1016/0896-6273(89)90072-x. [DOI] [PubMed] [Google Scholar]
- Dolber P. C., Sommer J. R. Corbular sarcoplasmic reticulum of rabbit cardiac muscle. J Ultrastruct Res. 1984 May;87(2):190–196. doi: 10.1016/s0022-5320(84)80078-7. [DOI] [PubMed] [Google Scholar]
- Edge M. B. Development of apposed sarcoplasmic reticulum at the T system and sarcolemma and the change in orientation of triads in rat skeletal muscle. Dev Biol. 1970 Dec;23(4):634–650. doi: 10.1016/0012-1606(70)90144-2. [DOI] [PubMed] [Google Scholar]
- Fabiato A. Simulated calcium current can both cause calcium loading in and trigger calcium release from the sarcoplasmic reticulum of a skinned canine cardiac Purkinje cell. J Gen Physiol. 1985 Feb;85(2):291–320. doi: 10.1085/jgp.85.2.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferguson D. G., Schwartz H. W., Franzini-Armstrong C. Subunit structure of junctional feet in triads of skeletal muscle: a freeze-drying, rotary-shadowing study. J Cell Biol. 1984 Nov;99(5):1735–1742. doi: 10.1083/jcb.99.5.1735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flucher B. E., Andrews S. B., Daniels M. P. Molecular organization of transverse tubule/sarcoplasmic reticulum junctions during development of excitation-contraction coupling in skeletal muscle. Mol Biol Cell. 1994 Oct;5(10):1105–1118. doi: 10.1091/mbc.5.10.1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flucher B. E., Andrews S. B., Fleischer S., Marks A. R., Caswell A., Powell J. A. Triad formation: organization and function of the sarcoplasmic reticulum calcium release channel and triadin in normal and dysgenic muscle in vitro. J Cell Biol. 1993 Dec;123(5):1161–1174. doi: 10.1083/jcb.123.5.1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flucher B. E., Morton M. E., Froehner S. C., Daniels M. P. Localization of the alpha 1 and alpha 2 subunits of the dihydropyridine receptor and ankyrin in skeletal muscle triads. Neuron. 1990 Sep;5(3):339–351. doi: 10.1016/0896-6273(90)90170-k. [DOI] [PubMed] [Google Scholar]
- Flucher B. E., Phillips J. L., Powell J. A., Andrews S. B., Daniels M. P. Coordinated development of myofibrils, sarcoplasmic reticulum and transverse tubules in normal and dysgenic mouse skeletal muscle, in vivo and in vitro. Dev Biol. 1992 Apr;150(2):266–280. doi: 10.1016/0012-1606(92)90241-8. [DOI] [PubMed] [Google Scholar]
- Flucher B. E., Phillips J. L., Powell J. A. Dihydropyridine receptor alpha subunits in normal and dysgenic muscle in vitro: expression of alpha 1 is required for proper targeting and distribution of alpha 2. J Cell Biol. 1991 Dec;115(5):1345–1356. doi: 10.1083/jcb.115.5.1345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flucher B. E. Structural analysis of muscle development: transverse tubules, sarcoplasmic reticulum, and the triad. Dev Biol. 1992 Dec;154(2):245–260. doi: 10.1016/0012-1606(92)90065-o. [DOI] [PubMed] [Google Scholar]
- Flucher B. E., Takekura H., Franzini-Armstrong C. Development of the excitation-contraction coupling apparatus in skeletal muscle: association of sarcoplasmic reticulum and transverse tubules with myofibrils. Dev Biol. 1993 Nov;160(1):135–147. doi: 10.1006/dbio.1993.1292. [DOI] [PubMed] [Google Scholar]
- Flucher B. E., Terasaki M., Chin H. M., Beeler T. J., Daniels M. P. Biogenesis of transverse tubules in skeletal muscle in vitro. Dev Biol. 1991 May;145(1):77–90. doi: 10.1016/0012-1606(91)90214-n. [DOI] [PubMed] [Google Scholar]
- Franzini-Armstrong C., Pincon-Raymond M., Rieger F. Muscle fibers from dysgenic mouse in vivo lack a surface component of peripheral couplings. Dev Biol. 1991 Aug;146(2):364–376. doi: 10.1016/0012-1606(91)90238-x. [DOI] [PubMed] [Google Scholar]
- Franzini-Armstrong C. STUDIES OF THE TRIAD : I. Structure of the Junction in Frog Twitch Fibers. J Cell Biol. 1970 Nov 1;47(2):488–499. doi: 10.1083/jcb.47.2.488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jewett P. H., Sommer J. R., Johnson E. A. Cardiac muscle. Its ultrastructure in the finch and hummingbird with special reference to the sarcoplasmic reticulum. J Cell Biol. 1971 Apr;49(1):50–65. doi: 10.1083/jcb.49.1.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jorgensen A. O., Shen A. C., Arnold W., Leung A. T., Campbell K. P. Subcellular distribution of the 1,4-dihydropyridine receptor in rabbit skeletal muscle in situ: an immunofluorescence and immunocolloidal gold-labeling study. J Cell Biol. 1989 Jul;109(1):135–147. doi: 10.1083/jcb.109.1.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jorgensen A. O., Shen A. C., Arnold W., McPherson P. S., Campbell K. P. The Ca2+-release channel/ryanodine receptor is localized in junctional and corbular sarcoplasmic reticulum in cardiac muscle. J Cell Biol. 1993 Feb;120(4):969–980. doi: 10.1083/jcb.120.4.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Junker J., Sommer J. R., Sar M., Meissner G. Extended junctional sarcoplasmic reticulum of avian cardiac muscle contains functional ryanodine receptors. J Biol Chem. 1994 Jan 21;269(3):1627–1634. [PubMed] [Google Scholar]
- Kim K. C., Caswell A. H., Brunschwig J. P., Brandt N. R. Identification of a new subpopulation of triad junctions isolated from skeletal muscle; morphological correlations with intact muscle. J Membr Biol. 1990 Feb;113(3):221–235. doi: 10.1007/BF01870074. [DOI] [PubMed] [Google Scholar]
- Kim K. C., Caswell A. H., Talvenheimo J. A., Brandt N. R. Isolation of a terminal cisterna protein which may link the dihydropyridine receptor to the junctional foot protein in skeletal muscle. Biochemistry. 1990 Oct 2;29(39):9281–9289. doi: 10.1021/bi00491a025. [DOI] [PubMed] [Google Scholar]
- Knudson C. M., Chaudhari N., Sharp A. H., Powell J. A., Beam K. G., Campbell K. P. Specific absence of the alpha 1 subunit of the dihydropyridine receptor in mice with muscular dysgenesis. J Biol Chem. 1989 Jan 25;264(3):1345–1348. [PubMed] [Google Scholar]
- Knudson C. M., Stang K. K., Moomaw C. R., Slaughter C. A., Campbell K. P. Primary structure and topological analysis of a skeletal muscle-specific junctional sarcoplasmic reticulum glycoprotein (triadin). J Biol Chem. 1993 Jun 15;268(17):12646–12654. [PubMed] [Google Scholar]
- Lau Y. H., Caswell A. H., Brunschwig J. P. Isolation of transverse tubules by fractionation of triad junctions of skeletal muscle. J Biol Chem. 1977 Aug 10;252(15):5565–5574. [PubMed] [Google Scholar]
- Lu X., Xu L., Meissner G. Activation of the skeletal muscle calcium release channel by a cytoplasmic loop of the dihydropyridine receptor. J Biol Chem. 1994 Mar 4;269(9):6511–6516. [PubMed] [Google Scholar]
- Marks A. R., Taubman M. B., Saito A., Dai Y., Fleischer S. The ryanodine receptor/junctional channel complex is regulated by growth factors in a myogenic cell line. J Cell Biol. 1991 Jul;114(2):303–312. doi: 10.1083/jcb.114.2.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meissner G. Ryanodine receptor/Ca2+ release channels and their regulation by endogenous effectors. Annu Rev Physiol. 1994;56:485–508. doi: 10.1146/annurev.ph.56.030194.002413. [DOI] [PubMed] [Google Scholar]
- Melzer W., Herrmann-Frank A., Lüttgau H. C. The role of Ca2+ ions in excitation-contraction coupling of skeletal muscle fibres. Biochim Biophys Acta. 1995 May 8;1241(1):59–116. doi: 10.1016/0304-4157(94)00014-5. [DOI] [PubMed] [Google Scholar]
- Nakai J., Dirksen R. T., Nguyen H. T., Pessah I. N., Beam K. G., Allen P. D. Enhanced dihydropyridine receptor channel activity in the presence of ryanodine receptor. Nature. 1996 Mar 7;380(6569):72–75. doi: 10.1038/380072a0. [DOI] [PubMed] [Google Scholar]
- Näbauer M., Callewaert G., Cleemann L., Morad M. Regulation of calcium release is gated by calcium current, not gating charge, in cardiac myocytes. Science. 1989 May 19;244(4906):800–803. doi: 10.1126/science.2543067. [DOI] [PubMed] [Google Scholar]
- Pape P. C., Jong D. S., Chandler W. K. Calcium release and its voltage dependence in frog cut muscle fibers equilibrated with 20 mM EGTA. J Gen Physiol. 1995 Aug;106(2):259–336. doi: 10.1085/jgp.106.2.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pizarro G., Csernoch L., Uribe I., Rodríguez M., Ríos E. The relationship between Q gamma and Ca release from the sarcoplasmic reticulum in skeletal muscle. J Gen Physiol. 1991 May;97(5):913–947. doi: 10.1085/jgp.97.5.913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Protasi F., Sun X. H., Franzini-Armstrong C. Formation and maturation of the calcium release apparatus in developing and adult avian myocardium. Dev Biol. 1996 Jan 10;173(1):265–278. doi: 10.1006/dbio.1996.0022. [DOI] [PubMed] [Google Scholar]
- Radermacher M., Rao V., Grassucci R., Frank J., Timerman A. P., Fleischer S., Wagenknecht T. Cryo-electron microscopy and three-dimensional reconstruction of the calcium release channel/ryanodine receptor from skeletal muscle. J Cell Biol. 1994 Oct;127(2):411–423. doi: 10.1083/jcb.127.2.411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rieger F., Pinçon-Raymond M., Tassin A. M., Garcia L., Romey G., Fosset M., Lazdunski M. Excitation-contraction uncoupling in the developing skeletal muscle of the muscular dysgenesis mouse embryo. Biochimie. 1987 Apr;69(4):411–417. doi: 10.1016/0300-9084(87)90033-2. [DOI] [PubMed] [Google Scholar]
- Rios E., Brum G. Involvement of dihydropyridine receptors in excitation-contraction coupling in skeletal muscle. Nature. 1987 Feb 19;325(6106):717–720. doi: 10.1038/325717a0. [DOI] [PubMed] [Google Scholar]
- Romey G., Garcia L., Dimitriadou V., Pincon-Raymond M., Rieger F., Lazdunski M. Ontogenesis and localization of Ca2+ channels in mammalian skeletal muscle in culture and role in excitation-contraction coupling. Proc Natl Acad Sci U S A. 1989 Apr;86(8):2933–2937. doi: 10.1073/pnas.86.8.2933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider M. F., Chandler W. K. Voltage dependent charge movement of skeletal muscle: a possible step in excitation-contraction coupling. Nature. 1973 Mar 23;242(5395):244–246. doi: 10.1038/242244a0. [DOI] [PubMed] [Google Scholar]
- Schneider M. F. Membrane charge movement and depolarization-contraction coupling. Annu Rev Physiol. 1981;43:507–517. doi: 10.1146/annurev.ph.43.030181.002451. [DOI] [PubMed] [Google Scholar]
- Serysheva I. I., Orlova E. V., Chiu W., Sherman M. B., Hamilton S. L., van Heel M. Electron cryomicroscopy and angular reconstitution used to visualize the skeletal muscle calcium release channel. Nat Struct Biol. 1995 Jan;2(1):18–24. doi: 10.1038/nsb0195-18. [DOI] [PubMed] [Google Scholar]
- Stern M. D., Lakatta E. G. Excitation-contraction coupling in the heart: the state of the question. FASEB J. 1992 Sep;6(12):3092–3100. doi: 10.1096/fasebj.6.12.1325933. [DOI] [PubMed] [Google Scholar]
- Sun X. H., Protasi F., Takahashi M., Takeshima H., Ferguson D. G., Franzini-Armstrong C. Molecular architecture of membranes involved in excitation-contraction coupling of cardiac muscle. J Cell Biol. 1995 May;129(3):659–671. doi: 10.1083/jcb.129.3.659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takekura H., Bennett L., Tanabe T., Beam K. G., Franzini-Armstrong C. Restoration of junctional tetrads in dysgenic myotubes by dihydropyridine receptor cDNA. Biophys J. 1994 Aug;67(2):793–803. doi: 10.1016/S0006-3495(94)80539-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takekura H., Nishi M., Noda T., Takeshima H., Franzini-Armstrong C. Abnormal junctions between surface membrane and sarcoplasmic reticulum in skeletal muscle with a mutation targeted to the ryanodine receptor. Proc Natl Acad Sci U S A. 1995 Apr 11;92(8):3381–3385. doi: 10.1073/pnas.92.8.3381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takekura H., Sun X., Franzini-Armstrong C. Development of the excitation-contraction coupling apparatus in skeletal muscle: peripheral and internal calcium release units are formed sequentially. J Muscle Res Cell Motil. 1994 Apr;15(2):102–118. doi: 10.1007/BF00130422. [DOI] [PubMed] [Google Scholar]
- Takekura H., Takeshima H., Nishimura S., Takahashi M., Tanabe T., Flockerzi V., Hofmann F., Franzini-Armstrong C. Co-expression in CHO cells of two muscle proteins involved in excitation-contraction coupling. J Muscle Res Cell Motil. 1995 Oct;16(5):465–480. doi: 10.1007/BF00126431. [DOI] [PubMed] [Google Scholar]
- Takeshima H., Iino M., Takekura H., Nishi M., Kuno J., Minowa O., Takano H., Noda T. Excitation-contraction uncoupling and muscular degeneration in mice lacking functional skeletal muscle ryanodine-receptor gene. Nature. 1994 Jun 16;369(6481):556–559. doi: 10.1038/369556a0. [DOI] [PubMed] [Google Scholar]
- Takeshima H., Yamazawa T., Ikemoto T., Takekura H., Nishi M., Noda T., Iino M. Ca(2+)-induced Ca2+ release in myocytes from dyspedic mice lacking the type-1 ryanodine receptor. EMBO J. 1995 Jul 3;14(13):2999–3006. doi: 10.1002/j.1460-2075.1995.tb07302.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanabe T., Beam K. G., Adams B. A., Niidome T., Numa S. Regions of the skeletal muscle dihydropyridine receptor critical for excitation-contraction coupling. Nature. 1990 Aug 9;346(6284):567–569. doi: 10.1038/346567a0. [DOI] [PubMed] [Google Scholar]
- Tanabe T., Beam K. G., Powell J. A., Numa S. Restoration of excitation-contraction coupling and slow calcium current in dysgenic muscle by dihydropyridine receptor complementary DNA. Nature. 1988 Nov 10;336(6195):134–139. doi: 10.1038/336134a0. [DOI] [PubMed] [Google Scholar]
- Tanabe T., Mikami A., Numa S., Beam K. G. Cardiac-type excitation-contraction coupling in dysgenic skeletal muscle injected with cardiac dihydropyridine receptor cDNA. Nature. 1990 Mar 29;344(6265):451–453. doi: 10.1038/344451a0. [DOI] [PubMed] [Google Scholar]
- Varadi G., Mikala G., Lory P., Varadi M., Drouet B., Pinçon-Raymond M., Schwartz A. Endogenous cardiac Ca2+ channels do not overcome the E-C coupling defect in immortalized dysgenic muscle cells: evidence for a missing link. FEBS Lett. 1995 Jul 24;368(3):405–410. doi: 10.1016/0014-5793(95)00697-8. [DOI] [PubMed] [Google Scholar]
- Wagenknecht T., Grassucci R., Frank J., Saito A., Inui M., Fleischer S. Three-dimensional architecture of the calcium channel/foot structure of sarcoplasmic reticulum. Nature. 1989 Mar 9;338(6211):167–170. doi: 10.1038/338167a0. [DOI] [PubMed] [Google Scholar]
- Yuan S. H., Arnold W., Jorgensen A. O. Biogenesis of transverse tubules and triads: immunolocalization of the 1,4-dihydropyridine receptor, TS28, and the ryanodine receptor in rabbit skeletal muscle developing in situ. J Cell Biol. 1991 Jan;112(2):289–301. doi: 10.1083/jcb.112.2.289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- el-Hayek R., Antoniu B., Wang J., Hamilton S. L., Ikemoto N. Identification of calcium release-triggering and blocking regions of the II-III loop of the skeletal muscle dihydropyridine receptor. J Biol Chem. 1995 Sep 22;270(38):22116–22118. doi: 10.1074/jbc.270.38.22116. [DOI] [PubMed] [Google Scholar]