Abstract
Background & Aims
Diet has major effects on the intestinal microbiota, but the exact mechanisms that alter complex microbial communities have been difficult to elucidate. In addition to the direct influence that diet exerts on microbes, changes in microbiota composition and function can alter host functions such as gastrointestinal (GI) transit time, which in turn can further affect the microbiota.
Methods
We investigated the relationships among diet, GI motility, and the intestinal microbiota using mice that are germ-free (GF) or humanized (ex-GF mice colonized with human fecal microbiota).
Results
Analysis of gut motility revealed that humanized mice fed a standard polysaccharide-rich diet had faster GI transit and increased colonic contractility compared with GF mice. Humanized mice with faster transit due to administration of polyethylene glycol or a nonfermentable cellulose-based diet had similar changes in gut microbiota composition, indicating that diet can modify GI transit, which then affects the composition of the microbial community. However, altered transit in mice fed a diet of fermentable fructooligosaccharide indicates that diet can change gut microbial function, which can affect GI transit.
Conclusions
Based on studies in humanized mice, diet can affect GI transit through microbiota-dependent or microbiota-independent pathways, depending on the type of dietary change. The effect of the microbiota on transit largely depends on the amount and type (fermentable vs non-fermentable) of polysaccharides present in the diet. These results have implications for disorders that affect GI transit and gut microbial communities, including irritable bowel syndrome and inflammatory bowel disease.
Keywords: Dietary Carbohydrates, Microbiome, Metabolomics, Serotonin
The human gut is a diverse ecosystem harboring trillions of microbial cells and hundreds of microbial species.1 Like other ecosystems, it responds to changes in environment, including nutrient availability (eg, diet),2,3 antibiotic use,4 and inflammation.5 Human gut microbes are involved in several vital processes that benefit the host, such as vitamin biosynthesis,6 development of the host immune system,7 and fermentation of undigested carbohydrates.8,9 Gut resident microbes and their interactions with the host can benefit the host but have also been implicated in diseases including inflammatory bowel disease, irritable bowel syndrome (IBS), and antibiotic-associated diarrhea.10 The complex signaling between microbes and their human hosts, and the resulting effect on normal physiologic functions, is still poorly understood. The key question in this regard is the directionality of influence between the gut microbiota and host physiology when one or the other is perturbed: does the gut microbiota affect host physiology, does host physiology affect the gut microbiota, or both?
Gastrointestinal (GI) motility and the gut microbiota are clearly associated. Early experiments in germ-free (GF) mice suggest that gut microbes alter development of small bowel motility patterns.11,12 Small bowel bacterial overgrowth, a clinical syndrome often seen with impaired GI motility, suggests that changes in GI motility can also modify the resident microbial population.13 Because microbial ecosystems, including the gut microbiota, respond to changes in environmental factors, motility likely shapes microbiota composition and function. For example, some microbial taxa benefit from increases in motility, relative to other species adapted to conditions associated with slower motility. This concept is consistent with ecological principles of r/K selection in response to environmental disturbance.14 As GI transit time decreases, such as with diarrhea, species better adapted to grow rapidly during reduced competition (r-selected) will dominate the gut. Conversely, as GI transit time increases, the community will be dominated by species that tend to grow more slowly in unrestricted conditions but are better adapted to persist in competitive environments (K-selected); these species include metabolically economical taxa. Because the community is interconnected metabolically, direct effects of motility on specific groups within the microbiota could cascade into broad ecosystem changes. The question of how motility affects the microbiota is increasingly important because dysbioses, or deviations from the normal microbiota, have been linked to diseases associated with significant alterations in GI transit time such as inflammatory bowel disease, IBS, and Clostridium difficile– associated disease.9
Environmental factors, including diet, can modify the relationship between transit time and the gut microbiota. Diet may affect motility directly via changes in the bulk flow of material through the gut or indirectly through an effect on the microbiota. Dietary polysaccharides are a major fuel for the distal gut community, and accordingly gut microbiomes have a large repertoire of glycan-degrading genes.6 Dietary fermentable polysaccharides can alter microbiota composition, in part because community members differ in their ability to metabolize them.15 Additionally, different taxa produce different fermentation end products when metabolizing a specific carbohydrate.16 Changes in production of short-chain fatty acids, such as butyrate, influence diverse aspects of host physiology, including motility.17
The role of soluble dietary fiber in mitigating constipation suggests that dietary carbohydrates can modulate GI transit.18 However, most studies describing the effect of diet on gut microbes do not address the potential feedback between changes in microbiota composition and motility. Here we used humanized mice (ex-GF mice colonized with a human fecal microbiota) to study the effects of human-relevant microbes on host physiology in a controlled experimental setting. We used custom diets that differ in the content of fermentable polysaccharides, nondietary modulators of motility, pyrosequencing-based enumeration of the microbiota, and quantitative measures of motility to investigate the interplay among diet, gut motility, and the intestinal microbiota. Our results show that changes in GI transit time can alter microbiota composition and that diet can affect GI transit times in a microbiota-dependent and -independent manner.
Materials and Methods
Animals
All animal experiments were in accordance with A-PLAC, the Stanford Institutional Animal Care and Use Committee, as described in Supplementary Materials and Methods. Diets are described in Supplementary Table 1.
Pyrosequencing/Data Analysis
Fecal DNA samples were sequenced at Duke Institute for Genome Sciences & Policy, and data were processed using QIIME 1.4.0.19
GI Transit Time
Whole gut transit time was determined using the carmine red method as previously described.20
Colonic Contractility Recording
Intracolonic pressure recording of the descending colon in conscious mice and data analysis were as previously described.21
Metabolomics
Fecal samples were extracted using Oasis solid-phase extraction cartridges (Waters Corporation, Milford, MA) and run on ultra-performance liquid chromatography mass spectrometry. Data analysis was performed with MetaboAnalyst.
Measurement of Short-Chain Fatty Acid Concentration
Short-chain fatty acid concentration was measured in feces using a modification of Moreau et al22 (see Supplementary Materials and Methods).
Results
Colonization of GF Mice with Human Microbiota Decreases GI Transit Time
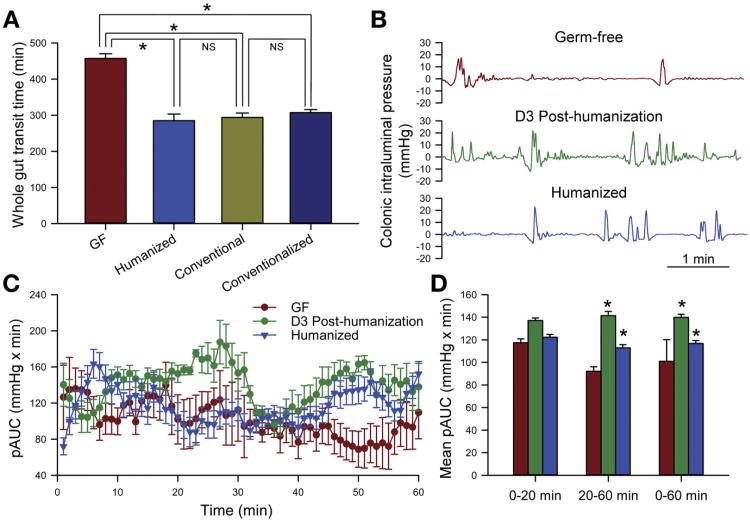
GF mice were humanized by colonization with feces obtained from a single anonymous healthy human donor. Mice were used 4 to 8 weeks after humanization, which is a sufficient interval for the microbial community to stabilize.23 We first examined whether GI transit time was influenced by the presence of a complex intestinal microbiota when mice were fed a standard polysaccharide-rich diet. GI transit time was significantly shorter in humanized mice than GF controls (285 ± 18 vs 457 ± 13 minutes; n = 4; P < .05; Figure 1A) but not significantly different from conventionally raised mice (285 ± 18 vs 294 ± 12 minutes; n = 4; P > .05; Figure 1A) or conventionalized mice (285 ± 18 vs 307.5 ± 15 minutes; n = 4; P > .05; Figure 1A).
Figure 1.
Humanization of GF mice alters GI transit time. (A) Whole gut transit time measured by the carmine red dye method in GF, humanized, conventional, or conventionalized mice. (B) Distal colonic intraluminal pressure changes recorded using a pressure transducer catheter in conscious GF mice or mice that were humanized 3 days (D3 Post-humanization) or 1 month (Humanized) before measurement. Data are representative of 3 groups of mice for each condition and smoothed with a 2-second time constant to remove breathing artifact and abdominal contractions. (C) Motility index for distal colonic contractility in humanized mice (age matched with GF mice and humanized for 1 month) and ex-GF mice 3 days after humanization as compared with GF mice. Data represent the phasic component of area under the respective curves (pAUC). (D) Mean pAUC for the first 20 minutes (stress period) versus the subsequent 40 minutes and the entire 60 minutes of recording. ANOVA, *P < .05. NS, not significant.
The highest density of microbes in the GI tract is in the distal gut; therefore, colonic contractility was measured in the distal colon approximately 4 cm above the anal verge using a miniaturized pressure transducer in vivo.21,24 Measurements were made in 3 conscious GF mice and then repeated 3 days after humanization in the same 3 mice to reduce the effects of between-mouse variability (Figure 1B). Colonic contractility was also measured independently in 4 humanized mice that were age and sex matched to the GF mice. These mice had been humanized for 1 month, serving as an agematched control with a longer period of microbial colonization. The index of colonic contractility, as measured by the area under the curve over the entire duration of recording (60 minutes), was significantly higher in both sets of humanized mice (3 days after humanization, and age-/sex-matched mice humanized for 1 month) when compared with GF mice (P < .05; oneway analysis of variance with Bonferroni correction; Figure 1C and D). Colonic contractility was not significantly different in the 3 groups of mice over the first 20 minutes, which represents the initial stress period when the mice are first restrained.21,24 Overall, these data show that both the human and murine intestinal microbiota increase GI transit in mice, partially via changes in distal colonic motility, and this effect can be seen as early as 3 days after colonization. Based on these data, we used GI transit time as a measure of GI motility in this study.
Alterations in GI Transit Time Influence the Microbial Composition of the Distal Gut
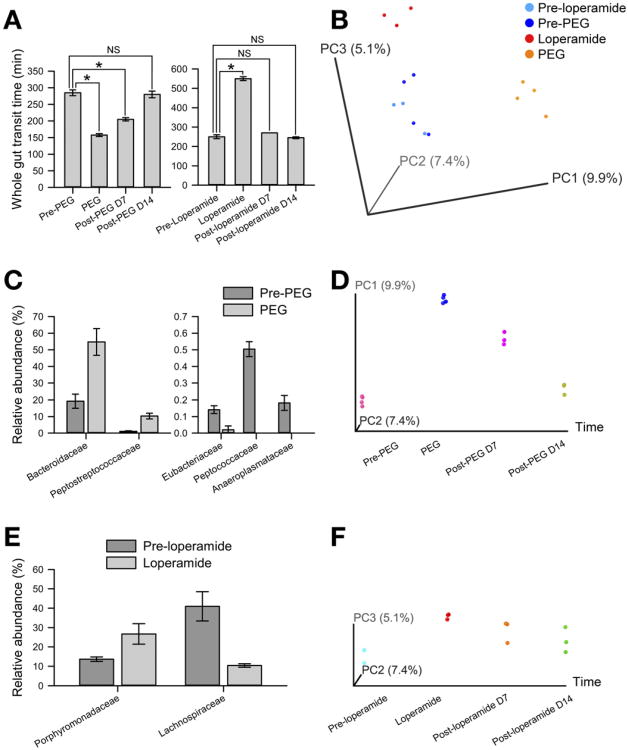
To examine whether nondietary alterations in GI transit affect the composition of the intestinal microbiota, humanized mice were treated with polyethylene glycol 3350 (PEG) for 10 days. This treatment significantly shortened GI transit time (285 ± 9 vs 157 ± 4 minutes; n = 4; P < .05). GI transit time returned to baseline within 14 days of discontinuing treatment with PEG (Figure 2A). Microbial community composition in fecal samples was determined using barcoded 454 Titanium pyrosequencing of PEG-treated fecal samples collected before treatment, on the last day of treatment (day 10), and 7 and 14 days after stopping treatment with PEG. The overall composition of the distal gut microbiota determined using principal coordinate analysis (PCoA) of the unweighted UniFrac25 distance matrix (β diversity) showed that the microbial community changed significantly as a result of treatment with PEG (Figure 2B). A similar pattern was noted with PCoA analysis of the weighted UniFrac distance matrix (Supplementary Figure 1A). Treatment with PEG resulted in a significant decrease in the relative abundance of family Peptococcaceae, Eubacteriaceae, and Anaeroplasmataceae and an increase in family Bacteroidaceae and Peptostreptococcaceae when compared with samples before treatment (Figure 2C and Supplementary Table 2). The alterations in overall microbial community composition were reversible; the gut microbial community structure resembled the original community within 14 days after discontinuing treatment with PEG (Figure 2D), and except for Peptococcaceae there were no significant differences in relative abundance of families in fecal samples collected before treatment with PEG and 14 days after stopping treatment with PEG.
Figure 2.
Alterations in GI transit time influence the microbial composition of the distal gut. (A) Whole gut transit time measured by the carmine dye method in humanized mice before, during, and 7 or 14 days after treatment with PEG or loperamide. (B) Unweighted UniFrac-based PCoA plot (2-dimensional representation of 3-dimensional plot) of gut microbial communities in humanized mice shows that pretreatment samples cluster together and posttreatment samples cluster based on treatment with PEG or loperamide. (C and E) Significant family-level differences in gut microbial communities of humanized mice treated with (C) PEG or (E) loperamide compared with pretreatment controls. (D and F) Unweighted Uni-Frac-based PCoA plots (2-dimensional representation of 3-dimensional plots with x-axis fixed as time) of gut microbial communities in humanized mice before, during, and 7 or 14 days after treatment with (D) PEG or (F) loperamide show change in microbial community structure upon treatment, which are reversible after discontinuation of treatment. *P < .05. NS, not significant.
Humanized mice were treated with loperamide hydrochloride for 10 days to determine whether experimentally increasing GI transit time would also alter intestinal microbiota composition. Treatment with loperamide significantly increased GI transit time (550 ± 10 vs 250 ± 10 minutes; n = 3; P < .05). GI transit time returned to normal within 14 days of discontinuing the drug (Figure 2A). PCoA of the unweighted UniFrac distance matrix showed that treatment with loperamide also altered the overall composition of the distal gut microbiota (Figure 2B); however, the microbial community appears distinct from the community after treatment with PEG. A similar but less pronounced effect was seen on PCoA of weighted UniFrac distance matrix (Supplementary Figure 1A). Increasing GI transit time with loperamide resulted in changes at multiple taxonomic levels quite distinct from those seen with PEG (Supplementary Table 3). These loperamide-induced changes include an increase in the Firmicutes to Bacteroidetes ratio and a significant decrease in relative abundance of the family Lachnospiraceae (Figure 2E). Mirroring the community restoration after discontinuation of treatment with PEG, the community structure closely resembled the baseline community 14 days after discontinuing treatment with loperamide (Figure 2F). There were no significant differences at the family level in samples collected before treatment with loperamide and 14 days after stopping treatment with loperamide. Changes in α diversity following treatment with PEG and loperamide are described in Supplementary Results (Supplementary Figure 2). These results highlight the close interrelationship between microbes and host GI motor function (Appendix panel A).
Carbohydrate Content of Diet Alters GI Transit Time
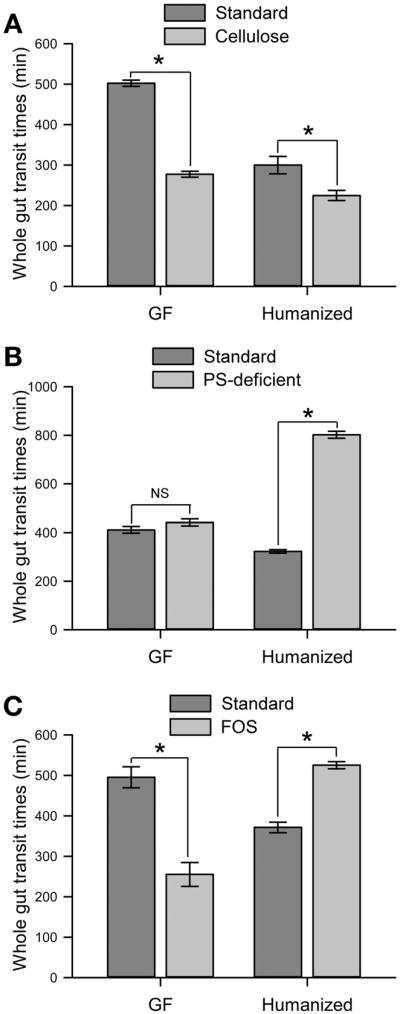
To determine the effect of diet on GI transit and whether this effect is mediated by the gut microbiota, GF and humanized mice were fed diets differing in carbohydrate content and GI transit time was measured. Mice were primarily kept on a standard diet and switched to custom diets for 10 days to determine the effect of diet on GI transit. We hypothesized that high levels of nonfermentable polysaccharides would increase motility due to the increased bulk of material consumed and transiting the gut (Supplementary Figure 3) and that this effect would be independent of microbial activity. Dietary cellulose shortened GI transit time, and this effect was independent of the presence of gut microbes; GI transit time was significantly shorter both in GF (277 ± 7 vs 502 ± 7 minutes; n = 4; P < .05; Figure 3A) and humanized mice (225 ± 12 vs 300 ± 21 minutes; n = 4; P < .05; Figure 3A). Although the cellulose-enriched diet accelerated GI transit time of both GF and humanized mice, the 2 groups maintained the same relative difference in GI transit time as observed on the standard diet, with humanized mice exhibiting more rapid transit (225 ± 12 vs 270 ± 7 minutes; n = 4; P < .05) (Figure 3A). To determine whether the change in transit time was due to the presence of nonfermentable fiber, mice were fed a custom diet that lacked polysaccharides (polysaccharide-deficient [PSD] diet). The GI transit time in the humanized mice was significantly increased on the PSD diet relative to the standard diet (802 ± 14 vs 322 ± 7 minutes; n = 4; P < .05; Figure 3B), but there was no significant change in transit time in GF mice relative to the standard diet (442 ± 14 vs 412 ± 14 minutes; n = 4; P > .05; Figure 3B). These data show that the effect of the microbiota on transit is dependent on the diet. In contrast to the polysaccharide-rich diet (Figure 1A), in the PSD dietary condition the microbiota actually exerts a cost on transit (ie, slows transit); however, that effect can be countered with the nonfermentable polysaccharide cellulose (compare Figure 3A with 3B for humanized mice). These findings support that the effect of a cellulose-enriched diet on increased GI motility is due to the presence of insoluble, nonfermentable fiber rather than simply a nonspecific effect of the custom diet.
Figure 3.
Alterations in carbohydrate content of diet alter GI transit time. (A) Whole gut transit time in GF or humanized mice fed (A) a cellulose-enriched diet, (B) a PSD diet, or (C) an FOS-enriched diet compared with standard diet controls. *P < .05.
The increase in transit time observed in humanized mice fed the PSD diet led us to hypothesize that GI transit time is affected by microbe-mediated carbohydrate fermentation. To answer this question, GF and humanized mice were fed a custom diet including a small amount of fermentable fructooligosaccharide (FOS; 10% wt/vol), a carbohydrate that can be metabolized by gut microbes but not by the mammalian host. Humanized mice had shorter GI transit time on the FOS-enriched diet relative to mice fed the PSD diet (525 ± 9 vs 802 ± 14 minutes; P < .05; Figure 3B and C) and longer transit time relative to their transit on the standard diet (525 ± 9 vs 371 ± 13 minutes; n = 3; P < .05; Figure 3B and C). In GF mice, the FOS-enriched diet resulted in diarrhea with a shortening of GI transit time when compared with their transit on the standard diet (255 ± 30 vs 495 ± 26 minutes; n = 3; P < .05; Figure 3C). Similar effects of diet on GI transit were seen in conventional mice (Supplementary Figure 4). Humanized mice decrease GI transit time as the quantity and complexity of carbohydrates increases from the PSD diet to the FOS-enriched diet to the standard diet to the cellulose diet. For cellulose, the nonfermented carbohydrates do not differ between GF and humanized mice, so the effect of diet on transit is not dependent on colonization state. Alternatively, FOS is fermented by the microbiota, so the effect of FOS depends on colonization status.
Carbohydrate Content of Diet Alters the Microbial Composition of the Distal Gut
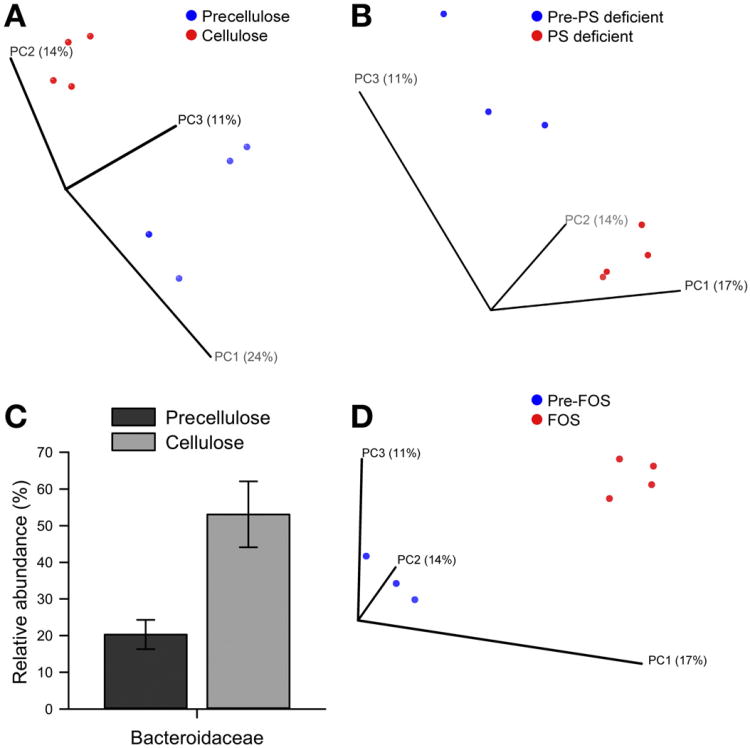
Previous studies have shown a significant correlation between dietary carbohydrate content and gut microbial composition.3 However, the effect of diet on motility, whether direct or modulated by the microbiota, as a contributor to microbiota compositional changes has not been well studied. To better understand how diet-induced changes in host GI transit time affect the gut microbiota, the microbial community composition of humanized mice fed different diets was determined using 16S ribosomal RNA gene pyrosequencing. Humanized mice were fed a standard diet after humanization for 4 to 8 weeks and then switched to a custom diet for 10 days. The cellulose-enriched diet resulted in altered microbiota relative to that observed on the standard diet (Figure 4A). To determine whether the cellulose-enriched diet-induced change in microbiota composition was simply due to the loss of fermentable carbohydrates in the standard diet, we analyzed fecal samples obtained from humanized mice on the standard diet and then switched to a PSD diet, which, unlike the cellulose-enriched diet, results in increased GI transit time. While the PSD diet resulted in significant changes in microbiota composition relative to mice fed the standard diet, these changes were distinct from those induced by the cellulose diet (Figure 4B). These results indicate that the effect of cellulose on GI transit time contributes to microbiota compositional changes given that cellulose is resistant to microbial fermentation. Fecal samples collected from mice on the cellulose diet had significant changes at multiple taxonomic levels (Supplementary Table 4), including higher relative abundance of the family Bacteroidaceae relative to the standard diet (Figure 4C). However, fecal samples from mice on the PSD diet had a significantly decreased relative abundance of families Rikenellaceae and Lactobacillaceae and several operational taxonomic units (OTUs; defined as sequences with 97% similarity) within the family Lachnospiraceae relative to the standard diet. Together, these data suggest that the differential effect on microbiota composition of the PSD diet versus the cellulose-enriched diet, which both lack fermentable carbohydrates, may be attributed in part to the opposing effect of these 2 diets on transit, although other effects of the diets may also contribute.
Figure 4.
Polysaccharide content of diet affects distal gut microbial composition. (A, B, and D) Unweighted UniFrac-based PCoA plot (2-dimensional representation of 3-dimensional plot) of gut microbial communities in humanized mice before and during administration of (A) a cellulose- enriched diet, (B) a PSD diet, or (D) FOS-enriched diet shows that samples cluster based on diet. (C) Significant family-level differences in gut microbial communities of humanized mice fed a cellulose-enriched diet compared with standard diet (Pre-cellulose).
Next we investigated how microbiota composition is affected by the presence of FOS. Unweighted UniFrac analysis revealed that the FOS diet microbiota clustered separately from the microbiota of standard diet-fed mice (Figure 4D). The changes in community structure were different from those seen in mice fed a cellulose or PSD diet, with significantly lower relative abundance of several OTUs in the family Lachnospiraceae but no significant change in Rikenellaceae, Lactobacillaceae, or Bacteroidaceae relative to the standard diet. PCoA based on weighted UniFrac distance matrix also showed similar compositional changes in humanized mice fed a cellulose-enriched, PSD, or FOS-enriched diet (Supplementary Figure 1B-D). Changes in α diversity associated with different diets are described in Supplementary Results (Supplementary Figure 5).
Together, these results support the hypothesis that a diet-induced change in GI transit alters the composition and diversity of the gut microbial community (Appendix panel B).
Changes in GI Transit Time Alter the Microbial Composition of the Distal Gut
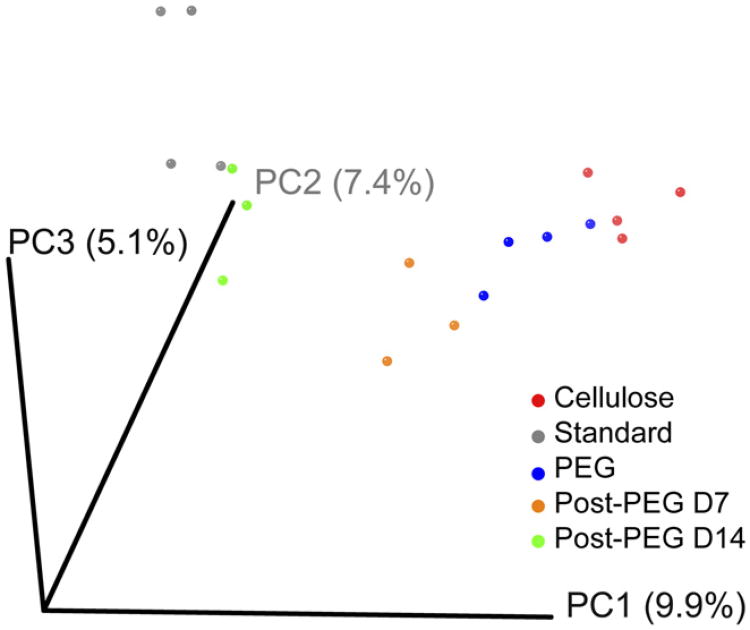
We next set out to determine whether different methods of manipulating GI transit time (ie, dietary vs nondietary) had the same effects on microbial composition. Two conditions that result in shorter GI transit time, humanized mice fed the standard diet treated with PEG and humanized mice fed the cellulose-rich diet, were compared. The distal gut microbial community from mice fed a cellulose-enriched diet clustered closely with the community from mice treated with PEG (day 10) and the community from mice treated with PEG 7 days after stopping PEG as determined by unweighted UniFrac-based analysis (Figure 5). All 3 groups with shorter transit time clustered together relative to the standard diet alone and exhibited a higher relative abundance of the family Bacteroidaceae and specifically the genus Bacteroides relative to untreated mice on the standard diet. These data suggest that the effect of cellulose on the distal gut microbial community is likely to be a result, in part, of its effect on GI transit time; treatment with PEG, an inert agent that increases GI transit time and bears no chemical resemblance to cellulose, mimics the effect of a cellulose-induced increase in transit on the microbiota. These data do not completely exclude off-target effects of PEG or cellulose, but there was no growth of Bacteroides isolates in PEG- or cellulose-supplemented culture medium (data not shown), consistent with a recent study on carbohydrate utilization by Bacteroides.8 The change in the gut microbial community on cellulose feeding and PEG treatment is consistent with transit time being a determinant of community membership. Similarly, mice fed FOS diet, which increases GI transit time similar to mice treated with loperamide, had a decrease in several members of the Lachnospiraceae family (7 OTUs, FOS diet; 4 OTUs, loperamide). However, unlike cellulose, FOS can be metabolized by gut microbes; therefore, both motility and a direct nutritional effect on the microbiota likely play a role in shaping the community under this condition.
Figure 5.
Effect of diet on distal gut microbial composition may be mediated in part by changes in GI transit time. Unweighted UniFracbased PCoA plot of gut microbial communities from humanized mice before (Standard), during (PEG), and 7 (post-PEG D7) or 14 (post-PEG D14) days after treatment with PEG compared with mice fed a celluloseenriched diet shows clustering of microbial communities with accelerated transit (cellulose, PEG, and Post-PEG D7).
FOS Diet–Induced Change in GI Transit Is Coincident With a Change in Fecal Metabolome
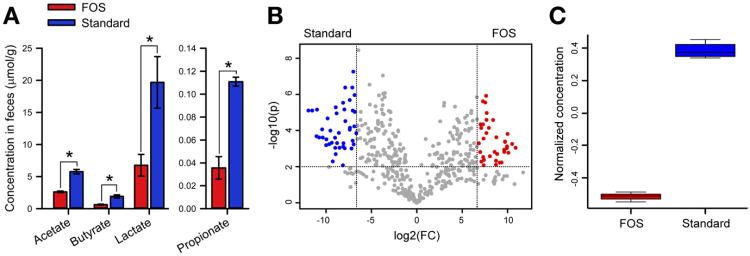
We hypothesized that the differences in GI transit observed in humanized mice fed FOS diet or a standard diet may partially result from differences in microbial community function of the respective microbiotas (Appendix panel C). Both primary (eg, the fermentation end product butyrate) and secondary (eg, derivatives of aromatic amino acids) metabolites can alter transit.17 To determine if the FOS diet results in a change in primary metabolites from bacterial fermentation, we analyzed short-chain fatty acid content in the feces of humanized mice fed the FOS or standard diet using gas chromatography. The FOS diet-fed mice had lower levels of acetate (2.6 ± 0.8 vs 5.8 ± 0.3 μmol/g feces), butyrate (0.5 ± 0.3 vs 1.9 ± 0.2 μmol/g feces), propionate (0.035 ± 0.009 vs 0.110 ±0.003 μmol/g feces), and lactate (6.8 ± 1.7 vs 19.7 ± 4.0 μmol/g feces) versus standard diet controls (Figure 6A). The role of butyrate in altering GI transit has been well described.26 To determine if the FOS diet produces distinct secondary microbial metabolites, fecal samples from humanized mice fed FOS diet or a standard diet were collected and metabolites were measured using ultra-performance liquid chromatography mass spectrometry-based nontargeted metabolite profiling. Using univariate statistical analysis (unpaired t test), 79 significantly different features (Supplementary Table 5) between the 2 dietary states were identified (defined as > 100-fold change with a P value <.01; Figure 6B). Interestingly, one feature that decreased in mice on FOS diet relative to control mice on a standard diet has the exact mass of 5-hydroxyindoleacetic acid (5-HIAA), a metabolite of the gut neurotransmitter serotonin, suggesting that FOS diet-induced changes in GI transit time may be related to alteration in the serotonin signaling pathway (Figure 6C and Supplementary Figure 6).
Figure 6.
Diet induces changes in microbiota function. (A) Short-chain fatty acid levels in the feces of mice fed FOS-enriched diet compared with a standard diet. No succinate was detected in either condition. (B) Volcano plot showing metabolites that are significantly higher (>100-fold change and P<.01) in feces of mice fed a standard diet (blue) or FOS-enriched diet (red). (C) Increase in normalized concentration of a metabolite with a mass corresponding to 5-HIAA, 191.07. *P < .05.
Discussion
Variation in GI transit time is commonly seen in healthy human subjects and also results from disease states including GI infections with rotavirus, cholera or Clostridium difficile colitis, inflammatory bowel disease, microscopic colitis, and IBS with constipation or diarrhea. Alterations in the microbiota have been described in several of these disease states and have often been implicated in their pathogenesis.10 This study illuminates the complex interactions among diet, GI transit, and gut microbial composition.
The Microbiota and Gut Transit Time Are Highly Interrelated
Here we showed that introduction into GF mice of a complex microbial community composed of human-relevant gut microbes significantly shortened GI transit time when the mice were fed a polysaccharide-rich standard diet. Notably, the magnitude and directionality of this microbiota-induced change in host physiologic function varied considerably under different dietary conditions after introduction of these human relevant microbes. Thus, microbiota functionality may act downstream of diet but upstream of gut motility. Conversely, the gut microbial ecosystem was sensitive to perturbations resulting from changes in host GI motility. Increasing or decreasing GI transit time using PEG or loperamide, respectively, led to alterations in the gut microbial community that were reversible on return to a normal gut transit time. These observations are consistent with the r/K selection theory of microbial ecology.14 Numerous examples have been reported showing that perturbation of the gut ecosystem (induced by antibiotics, diet, inflammation, and so on) is followed by dynamic changes in which species well adapted to either perturbation in general27 or the changed environment28 flourish. Alterations in transit time change many parameters of the gut ecosystem, including the flux of nutrients, and therefore are likely to be accompanied by selection for species adapted to either (1) slow but efficient growth in a more competitive environment (ie, corresponding to slower transit and limited resources; K-selected) or (2) faster growth in a less competitive environment (corresponding to faster transit and excess resources; r-selected).
In our study, changes in GI transit time led to changes in families within the order Bacteroidales, including an increase in Bacteroidaceae in PEG-treated mice (accelerated transit) and an increase in Porphyromonadaceae in loperamide-treated mice (delayed transit), suggesting that these families have distinct adaptations that differentially influence their success relative to gut transit.
These experiments showed how host GI transit time influences gut microbial composition and, conversely, how altering microbial composition influences GI transit time. The interplay between the microbiota and gut motility is relevant to experimental paradigms used to study several diseases associated with the gut microbial composition, such as inflammatory bowel disease and IBS, where GI transit is also altered.
Two Independent Mechanisms of Increased Transit Time Result in Similar Changes in Microbiota Community Composition
We investigated how diet modulates gut microbial composition and GI transit time. The insoluble dietary fiber cellulose led to significantly accelerated GI transit, an effect that was independent of the presence of a microbial community in the gut. Although it is possible that the decrease in transit time in both GF mice and humanized mice is a result of different mechanisms, the similar effect in both groups of mice is likely due to the water-holding capacity of cellulose, adding bulk to the stool.29 Cellulose cannot be used appreciably by human gut microbes but still significantly alters gut microbial composition. Interestingly, changes in microbial composition seen in mice on the cellulose diet were similar to those seen in mice on the standard diet drinking PEG-supplemented water. Specifically, an increase in Bacteroides was observed under both conditions, suggesting that shortened GI transit time plays a role in Bacteroides fitness. This novel finding suggests that changes seen in the gut microbiota as a result of dietary changes may in part be due to the microbiota-independent effect of diet on host GI transit time.
The Dietary Oligosaccharide FOS Has a Differential Effect on GI Transit Time Based on the Presence or Absence of Microbiota
In our study, we showed that a diet composed primarily of FOS leads to a marked increase in GI transit time when compared with a standard diet in humanized mice (the opposite of the effect seen in GF mice). The interaction of diet with microbiota resulting in alteration of host GI transit time may have important implications for patients with functional bowel disorders such as IBS, who often have symptom flares with certain dietary components. Several studies have focused on dietary intervention for management of symptoms in patients with IBS, and recent reports suggest that reduction of fermentable oligosaccharides (such as FOS, which are part of fermentable oligosaccharides, disaccharides, monosaccharides, and polyols) in the diet leads to improvement of bloating in patients with IBS.30 Here we showed that humanized mice fed FOS diet have an increased gut transit time and an altered microbiota relative to a standard diet. These findings, which highlight the effect of FOS-enriched diet on GI physiology and the gut microbiota, help explain why a diet high in oligosaccharides, such as FOS, can worsen symptoms in patients with IBS.
Diet-Induced Changes in Fecal Metabolome May Be Responsible for Changes in GI Transit Time
We showed here how diet leads to alterations in microbial community functionality either as a result of change in community structure or change in function of individual members. In humanized mice fed FOS diet, the fecal short-chain fatty acid profile showed lower amounts of all detected short-chain fatty acids compared with standard diet–fed controls. The colon wall is directly exposed to the end products of microbial fermentation (such as short-chain fatty acids), so colonic contractile patterns are likely affected by these microbial end products. Additionally, the fecal metabolite profile measured using ultra-performance liquid chromatography mass spectrometry in the FOS diet differed from that observed in the standard diet. We found significantly lower levels of a compound with the same mass as the serotonin metabolite 5-HIAA in the feces of mice on the FOS diet compared with standard diet controls. Approximately 90% of the serotonin in the body is produced in the gut, and serotonin is an important paracrine messenger and neurotransmitter involved in modulating GI motility.31 In infectious diarrhea, such as rotavirus and cholera, the role of serotonin signaling in the pathogenesis of diarrhea has been well described.32 This study implicates the possible involvement of the serotonin pathway in the diet-induced alterations in GI transit time, a result of signaling by commensal gut bacteria. However, given the close interrelationship of diet, transit time, and microbiota, further studies that leverage a variety of experimental tools, including genetic models of host and resident microbes, will be required to better understand the role of specific pathways and molecules (eg, serotonin) in mediating these interactions.
Conclusions
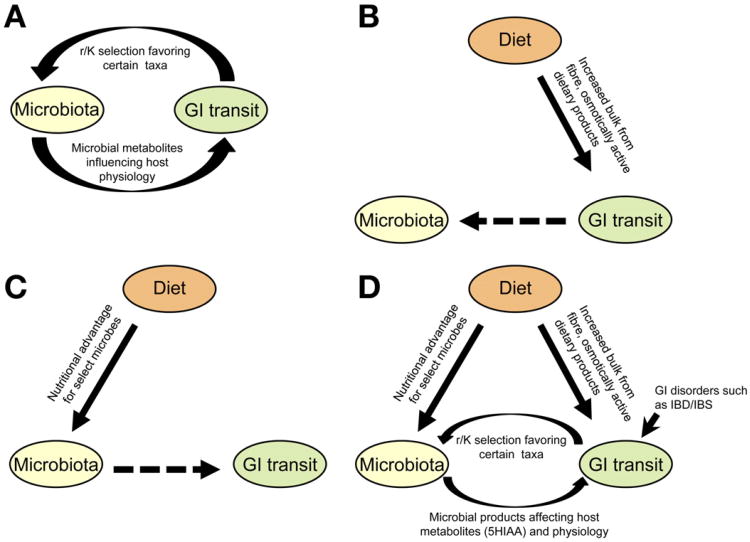
Based on the data presented here, we propose the following model for the interactions between diet, gut microbiota, and GI transit time in the host. GI transit time and gut microbiota are interrelated (Appendix panel A). Diet can independently affect both GI transit time and gut microbial composition and function (as determined by metabolite profiles). However, diet-induced changes in microbial composition may be mediated in part by changes in GI transit time (Appendix panel B), and the effect of diet on GI transit time may be a result of altered functionality of the gut microbial community caused by the dietary change (Appendix panel C). The effect of diet, transit time, and microbiota composition creates a highly interdependent and interactive environment (Appendix panel D). Here we used a highly controlled experimental model as a first step in investigating these complex interrelationships that are likely to offer new potential targets and strategies for manipulation of human GI transit.
Supplementary Material
Acknowledgments
The authors thank Peter Strege for help with preparation of figures and Sara Fisher for administrative help.
Funding: Supported by National Institutes of Health grant R01DK085025 (to J.L.S.), Digestive Diseases Center grant DK-41301 (Animal Models Core; to Y.T., M.M.) and K01 DK088937 (M.L.).
Abbreviations used in this paper
- FOS
fructo-oligosaccharide
- GF
germ-free
- GI
gastrointestinal
- 5-HIAA
5-hydroxyindoleacetic acid
- IBS
irritable bowel syndrome
- OTU
operational taxonomic unit
- PCoA
principal coordinate analysis
- PEG
polyethylene glycol
- PSD
polysaccharide-deficient
Appendix.
Model for interactions between diet, gut microbiota, and GI transit time. (A) GI motility and gut microbiota are closely interrelated and can significantly affect one another. (B) Diet-induced changes in GI transit may partially mediate the effect of diet on microbial composition. (C) Diet-induced changes in microbiota composition and function may alter GI transit time. (D) Together, these factors result in a complex interplay between diet, the gut microbiota, and GI transit.
Footnotes
Conflicts of interest: The authors disclose no conflicts.
Supplementary Material: Note: To access the supplementary material accompanying this article, visit the online version of Gastroenterology at www.gastrojournal.org, and at http://dx.doi:10.1053/j.gastro.2013.01.047.
References
- 1.Savage DC. Microbial ecology of the gastrointestinal tract. Annu Rev Microbiol. 1977;31:107–133. doi: 10.1146/annurev.mi.31.100177.000543. [DOI] [PubMed] [Google Scholar]
- 2.Sonnenburg JL, Xu J, Leip DD, et al. Glycan foraging in vivo by an intestine-adapted bacterial symbiont. Science. 2005;307:1955–1959. doi: 10.1126/science.1109051. [DOI] [PubMed] [Google Scholar]
- 3.Wu GD, Chen J, Hoffmann C, et al. Linking long-term dietary patterns with gut microbial enterotypes. Science. 2011;334:105–108. doi: 10.1126/science.1208344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dethlefsen L, Huse S, Sogin ML, et al. The pervasive effects of an antibiotic on the human gut microbiota, as revealed by deep 16S rRNA sequencing. PLoS Biol. 2008;6:e280. doi: 10.1371/journal.pbio.0060280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hakansson A, Molin G. Gut microbiota and inflammation. Nutrients. 2011;3:637–682. doi: 10.3390/nu3060637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Backhed F, Ley RE, Sonnenburg JL, et al. Host-bacterial mutualism in the human intestine. Science. 2005;307:1915–1920. doi: 10.1126/science.1104816. [DOI] [PubMed] [Google Scholar]
- 7.Mazmanian SK, Liu CH, Tzianabos AO, et al. An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell. 2005;122:107–118. doi: 10.1016/j.cell.2005.05.007. [DOI] [PubMed] [Google Scholar]
- 8.Martens EC, Lowe EC, Chiang H, et al. Recognition and degradation of plant cell wall polysaccharides by two human gut symbionts. PLoS Biol. 2011;9:e1001221. doi: 10.1371/journal.pbio.1001221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sekirov I, Russell SL, Antunes LC, et al. Gut microbiota in health and disease. Physiol Rev. 2010;90:859–904. doi: 10.1152/physrev.00045.2009. [DOI] [PubMed] [Google Scholar]
- 10.DuPont AW, DuPont HL. The intestinal microbiota and chronic disorders of the gut. Nat Rev Gastroenterol Hepatol. 2011;8:523–531. doi: 10.1038/nrgastro.2011.133. [DOI] [PubMed] [Google Scholar]
- 11.Barbara G, Stanghellini V, Brandi G, et al. Interactions between commensal bacteria and gut sensorimotor function in health and disease. Am J Gastroenterol. 2005;100:2560–2568. doi: 10.1111/j.1572-0241.2005.00230.x. [DOI] [PubMed] [Google Scholar]
- 12.Husebye E, Hellstrom PM, Sundler F, et al. Influence of microbial species on small intestinal myoelectric activity and transit in germ-free rats. Am J Physiol Gastrointest Liver Physiol. 2001;280:G368–G380. doi: 10.1152/ajpgi.2001.280.3.G368. [DOI] [PubMed] [Google Scholar]
- 13.Toskes PP. Bacterial overgrowth of the gastrointestinal tract. Adv Intern Med. 1993;38:387–407. [PubMed] [Google Scholar]
- 14.Pianka ER. On r and K selection. Am Naturalist. 1970;104:592–597. [Google Scholar]
- 15.Sonnenburg ED, Zheng H, Joglekar P, et al. Specificity of polysaccharide use in intestinal bacteroides species determines diet-induced microbiota alterations. Cell. 2010;141:1241–1252. doi: 10.1016/j.cell.2010.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Louis P, Scott KP, Duncan SH, et al. Understanding the effects of diet on bacterial metabolism in the large intestine. J Appl Microbiol. 2007;102:1197–1208. doi: 10.1111/j.1365-2672.2007.03322.x. [DOI] [PubMed] [Google Scholar]
- 17.Havenaar R. Intestinal health functions of colonic microbial metabolites: a review. Benef Microbes. 2011;2:103–114. doi: 10.3920/BM2011.0003. [DOI] [PubMed] [Google Scholar]
- 18.Gray DS. The clinical uses of dietary fiber. Am Fam Physician. 1995;51:419–426. [PubMed] [Google Scholar]
- 19.Caporaso JG, Kuczynski J, Stombaugh J, et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods. 2010;7:335–336. doi: 10.1038/nmeth.f.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li Z, Chalazonitis A, Huang YY, et al. Essential roles of enteric neuronal serotonin in gastrointestinal motility and the development/survival of enteric dopaminergic neurons. J Neurosci. 2011;31:8998–9009. doi: 10.1523/JNEUROSCI.6684-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gourcerol G, Wu SV, Yuan PQ, et al. Activation of corticotropin-releasing factor receptor 2 mediates the colonic motor coping response to acute stress in rodents. Gastroenterology. 2011;140:1586–1596.e6. doi: 10.1053/j.gastro.2011.01.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Moreau NM, Goupry SM, Antignac JP, et al. Simultaneous measurement of plasma concentrations and 13C-enrichment of short-chain fatty acids, lactic acid and ketone bodies by gas chromatography coupled to mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2003;784:395–403. doi: 10.1016/s1570-0232(02)00827-9. [DOI] [PubMed] [Google Scholar]
- 23.Turnbaugh PJ, Ridaura VK, Faith JJ, et al. The effect of diet on the human gut microbiome: a metagenomic analysis in humanized gnotobiotic mice. Sci Transl Med. 2009;1:6ra14. doi: 10.1126/scitranslmed.3000322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gourcerol G, Wang L, Adelson DW, et al. Cholinergic giant migrating contractions in conscious mouse colon assessed by using a novel noninvasive solid-state manometry method: modulation by stressors. Am J Physiol Gastrointest Liver Physiol. 2009;296:G992–G1002. doi: 10.1152/ajpgi.90436.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lozupone C, Hamady M, Knight R. UniFrac—an online tool for comparing microbial community diversity in a phylogenetic context. BMC Bioinformatics. 2006;7:371. doi: 10.1186/1471-2105-7-371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Soret R, Chevalier J, De Coppet P, et al. Short-chain fatty acids regulate the enteric neurons and control gastrointestinal motility in rats. Gastroenterology. 2010;138:1772–1782. doi: 10.1053/j.gastro.2010.01.053. [DOI] [PubMed] [Google Scholar]
- 27.Lozupone C, Faust K, Raes J, et al. Identifying genomic and metabolic features that can underlie early successional and opportunistic lifestyles of human gut symbionts. Genome Res. 2012;22:1974–1984. doi: 10.1101/gr.138198.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Winter SE, Thiennimitr P, Winter MG, et al. Gut inflammation provides a respiratory electron acceptor for Salmonella. Nature. 2010;467:426–429. doi: 10.1038/nature09415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cummings JH, Stephen AM. The role of dietary fibre in the human colon. Can Med Assoc J. 1980;123:1109–1114. [PMC free article] [PubMed] [Google Scholar]
- 30.Shepherd SJ, Parker FC, Muir JG, et al. Dietary triggers of abdominal symptoms in patients with irritable bowel syndrome: randomized placebo-controlled evidence. Clin Gastroenterol Hepatol. 2008;6:765–771. doi: 10.1016/j.cgh.2008.02.058. [DOI] [PubMed] [Google Scholar]
- 31.Gershon MD, Tack J. The serotonin signaling system: from basic understanding to drug development for functional GI disorders. Gastroenterology. 2007;132:397–414. doi: 10.1053/j.gastro.2006.11.002. [DOI] [PubMed] [Google Scholar]
- 32.Camilleri M, Nullens S, Nelsen T. Enteroendocrine and neuronal mechanisms in pathophysiology of acute infectious diarrhea. Dig Dis Sci. 2012;57:19–27. doi: 10.1007/s10620-011-1939-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.