Abstract
Microorganisms such as bacteria and fungi produce a variety of specialized metabolites that are invaluable for agriculture, biological research, and drug discovery. However, the screening of microbial metabolic output is usually a time intensive task. Here we utilize a liquid micro-junction surface sampling probe for electrospray ionization mass spectrometry to extract and ionize metabolite mixtures directly from living microbial colonies grown on soft nutrient agar in Petri-dishes without any sample pre-treatment. To demonstrate the method is robust, this technique was applied to observe the metabolic output of more than 30 microorganisms, including yeast, filamentous fungi, pathogens, and marine-derived bacteria, that were collected worldwide. Diverse natural products produced from different microbes, including Streptomyces coelicolor, Bacillus subtilis, and Pseudomonas aeruginosa are further characterized.
Microorganisms produce a wide array of specialized metabolites that phenotypically affect cell populations in their surroundings, a process known as metabolic exchange. 1 These exchange factors have a diverse array of functions: some act as signaling molecules that control metabolism and morphology, some are virulence factors or even cannibalistic factors, some serve as nutrients, and others function as antibiotic agents that promote the survival of the species. 1 Many of these specialized metabolites, such as penicillin and vancomycin, have been found to be of great clinical importance. 1 The ubiquity and variety of microbes generating compounds of such high potential utility presents a consequent challenge and opportunity. There are more than 105 formally named bacteria species in pure cultures and approximately twice as many uncultured species. 2,3 The total richness is estimated at 107to 109 bacteria species worldwide. 3,4 Microbes also often co-exist with hosts such as humans: some of these microbial communities have been characterized at the nucleotide level. 5 Human microbiota play physiological roles and are thought to be associated with multiple disease states. 6 A recent study indicates that the gut microbiome can drive hormonal regulation of human autoimmunity. 7 The ability to characterize metabolic exchange factors from agar grown microbial colonies facilitates the understanding of the roles of such specialized metabolites and their roles in virulence, symbiosis, and mutualism, yet there are very few tools available that can capture the chemistry of microbes in such a direct manner.
One of the most challenging aspects of investigating the chemistry of microbes in conventional mass spectrometric workflows is the time required for techniques such as liquid chromatography mass spectrometry (LC-MS), to isolate individual molecular species. Matrix-assisted laser desorption ionization (MALDI) mass spectrometry can be applied to directly study microbial metabolic exchange eliminating the need for compound isolation. 8-10 However, the requisite crystalline organic matrix deposition and dehydration procedures involved in sample preparation inevitably deactivates microbial cellular activity. Recently, in order to investigate microbial metabolic profiles in real-time, an angled capillary liquid bridge based technique called nanospray desorption electrospray ionization (nanoDESI) was applied to the analysis of living microbial colonies. 11,12 This technique allows spatiotemporal profiling of metabolite output directly on Petri-dishes in ambient environments without a labor intensive purification process. Although nanoDESI works well for many microbiology applications, it is susceptible to loss of solvent flow through the liquid bridge due to the hydrophilicity of the extracellular matrix (ECM) of the microbes themselves and we have observed that high salinity media alters the electrospray behavior. Because nanoDESI has low flow rates and is self-aspirating, it makes it more sensitive to loss of flow by these factors. Clogging in the transfer capillary tube due to surface roughness is also more common during nanoDESI sampling. When this happens, the user will need to replace the capillary tube. Frequent capillary replacements and subsequent setup realignment makes the implementation of nanoDESI to untrained users more difficult at this time. For these reasons, a more effective operational approach to molecular information from microbes could, through increased robustness and automation, better deal with the demand for the analysis of the plethora of specialized metabolites produced by the diverse array of microbes that inhabit our planet. Additional methods that complement nanoDESI to directly characterize the molecules of microbes grown on Petri-dishes need to be developed.
Toward this end, we herein utilize a continuous flowprobe system, based on the coaxial tube geometry liquid microjunction surface sampling probe (LMJ-SSP), for continuous in situ extraction of an assortment of analytes, as it has previously been demonstrated for various qualitative and quantitative analysis of sample surfaces including glass slides, TLC plates, skin and tissues. 13-17 LMJ-SSP differs from nanoDESI in key operational aspects. LMJ-SSP mainly uses a pneumatic nebulizer instead of the self-aspirated nanospray capillary to extract analytes from samples (Figure 1a). LMJ-SSP has a co-axial geometry enabling analysis near the edges of the Petri-dishes. When clogging occurs in LMJ-SSP, this often can be remedied by a temporarily closing the nebulizer valve, resulting in a reversal of the solvent and nebulizer gas to flush the transfer tubes. Finally, LMJ-SSP uses higher flow rates than nanoDESI. As shown in Figure 1a, cultured microbial colony plates were sequentially placed onto an x-y stage (Figure 1b) below a z-axis translated probe body. As with all liquid extraction methods, the classes of molecules observed depends on the solvent used. In order to extract a broad range of microbially-secreted compounds, a 65/35 acetonitrile/0.1% formic acid aqueous solution (vol/vol) was used for all of the experiments, pumped at a rate of 5 μL/min through the probe to the surface and continuously aspirated away by the nebulizer (80 psi) operating at a high voltage (2 kV) for electrospray at a Thermo LTQ-FTICR MS inlet.
Figure 1.
An overview of flowprobe based mass spectrometric analysis of living microorganisms on Petri dish. (a) Schematic diagram of MS analysis of microbial colonies using a coaxial LMJ-SSP method. Solvent is continuously pumped to the surface via the outer capillary tube to extract and mobilize the molecules on the colony and aspirated into the inner capillary tube by the Venturi force generated at the nebulizer where ions are electrosprayed at the MS inlet. (b) Photograph of the flowprobe prototype platform including the x-y stage. (c) Snapshot of the flowprobe desorbing metabolites from a colony of S. coelicolor A3(2).
Most microbes form biofilms (Figure 1c) making colony surfaces more heterogeneous than flat, evenly-deposited glass slides. Consequently computer controlled x-y, and z axes, respectively, were adjusted to move various colonies into the path of extraction, and maintain sufficient distance between the probe and surface at various plate locations, preserving colony morphologies. Supplemental Movie S1 demonstrates the operation of the flowprobe when it contacts a Petri dish bearing colonies of the soil bacterium Streptomyces coelicolor A3 (2) (Movie S1, bottom screen), and the mass spectrometer readout displays the instantaneous instrumental response. As shown in Movie S1 (top screen) and Figure S1, the red-pigmented antibiotics prodiginines (m/z 392 and 394, cyclic and open forms) are detected upon flowprobe liquid junction contact with the colony and are further confirmed by MS/MS (supporting information). 11
Specialized metabolites can be crucial for the survival of microorganisms subjected to harsh environmental conditions. 18 One such molecule is the nonribosomal peptide synthetase-derived surfactin, a lipopeptide antibiotic produced in 20 laboratory characterized strains of B. subtilis and found to possess antibacterial, antifungal, antiviral, and antimycoplasmic activity. 19,20 We cultured two B. subtilis strains: 3610 and PY79. B. subtilis PY79 is a laboratory domesticated strain of B. subtilis 3610 and most of the polyketide synthases and nonribosomal peptide synthetases have been silenced,21 and interrogated them using the flowprobe prototype. Surfactin analogs with different lipid chain lengths were identified from the colony surface of B. subtilis 3610 as shown in Figure S2 but, as expected, were not observed on B. subtilis PY79. These metabolomic results are consistent with reports in which mass spectrometry was used to highlight differences at the nucleotide level. 8,9,22,23 In addition to surfactin, other cyclic peptides such as plipastatin24 and subtilosin were observed from B. subtilis 3610 but not B. subtilis PY79. At different stages during the growth of a B. subtilis colony, bacterial cells differentiate in order to carry out multiple functions that enable adaptation to environmental changes. Individual cell fate in B. subtilis is coordinated by many biosynthesized chemical agents. Recently, a sporulation killing factor (SKF) that determines cannibalistic behavior was identified on the colonies of B. subtilis strains using MALDI-TOF-IMS. 9 These ribosomal peptides were also observed in the 2+ and 3+ charge states on both B. subtilis analyzed strains, as shown in Figure S2. Polysaccharide derived signals, possibly from degraded agarose, were also abundant on both colonies. The comparison of MS performance using LMJ-SSP and nanoDESI are also shown in supporting information (Fig. S13).
Metabolic exchange is not unique to microbial communities and is also observed in the context of commensal, mutualistic, competitive, and antagonistic microbe-eukaryote systems and microbe-host interactions. Alteration of metabolic signaling agents has been demonstrated to correspond with disease progression. The opportunistic pathogen Pseudomonas aeruginosa,25 which is commonly isolated from the lungs of cystic fibrosis (CF) patients,26 secretes several virulence factors including pyocyanine (PYO). 27 Furthermore, the capacity for fungal inhibition by P. aeruginosa is also associated with its secondary metabolites. To further illustrate the utility of this technique in pathogen analysis, we describe the metabolomic profiling of a cultured P. aeruginosa PA14 colony. As shown in Figure S3, both virulence factor ions (PYO with H+ and Na+adducts) as well as its biosynthetic precursor phenazine-1-carboxylic acid (PCA) were identified. The elongated conjugated system of the PYO cation stabilizes the ESI product and explains the higher PYO intensity relative to PCA signal at m/z 225. 28 Within the same fingerprint, in the range of m/z 240-320, a diverse array of quinolone molecules, including Pseudomonas quinolone signaling molecule 2-heptyl-3-hydroxy-4-quinolone (PQS) and related molecules with different carbon chain lengths were obtained. These molecules play a crucial role as signaling molecules in the quorum sensing network of Pseudomonas aeruginosa. 29,30 Above m/z 500, extensive rhamnolipid production was identified, exemplified by the sodiated mono-and di-rhamnolipid congeners, seen at m/z 527 and 673, respectively. These glycolipidic biosurfactants assist in the biodegradation of insoluble media; are associated with biofilm formation and swarming behavior; and act as virulence factors against a variety of microorganisms, such as the fungus Candida albicans. 31,32
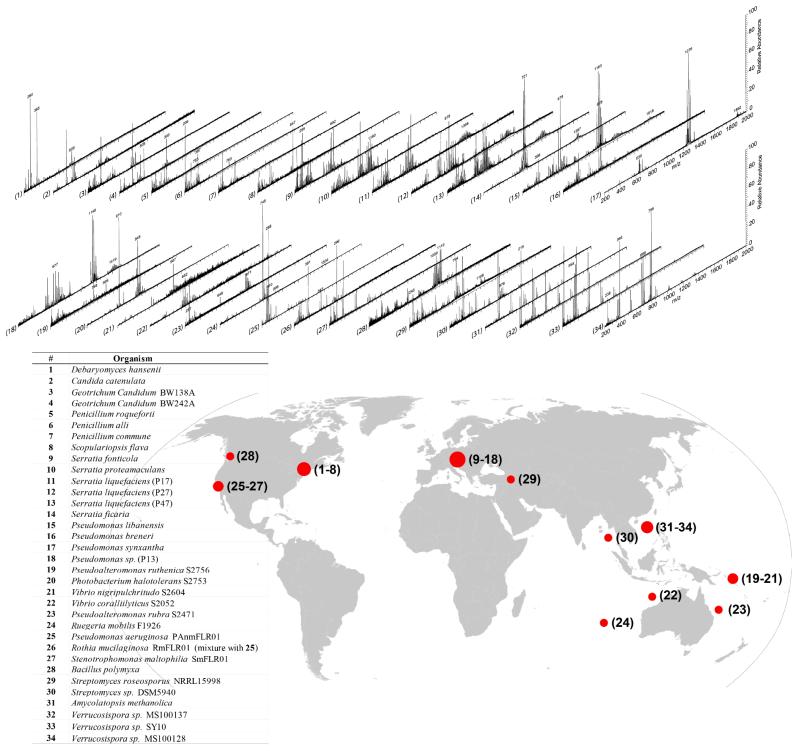
Utilizing the flowprobe system for direct colony extraction and ionization, diverse classes of natural products, ranging from small redox compounds,33 quorum sensing signaling molecules,29,30 glycolipidic antifungal31,32 and red-pigmented anticancer agents,34 to nonribosomal peptidic antibiotics19,20 and ribosomally-encoded peptidic cell differentiation inducers,9,35,36 were identified. Early success with various bacterial genera encouraged exploring a wider scope of microbial diversity. Thirty four bacterial and fungal strains isolated from worldwide sources including CF patients (San Diego, USA), bats (caves and abysses in the Czech Republic),37 cheese,38 marine habitats (distinct geographical areas from Solomon Islands to South China Sea),39-41 and symbiotic lichens (Canada) were cultured and investigated using this approach. As elaborated in Figure 2, flowprobe mass spectra from each strain were acquired up to m/z 2000 showing multiplex spectral features that illustrate sharp diversity of metabolomic profiles. Polysaccharide ions (with characteristic 162 Da repeating units) are commonly found among some of these colonies as well as B. subtilis (Figure S2), possibly as a result of agar biodegradation. These media-derived molecules contribute to the cross-genera background (mutual) ions. However, molecules detected exclusively within the same genera, such as ion clusters at m/z 564, 692 and 1400 in Serratia, enable discernment of spectral patterns of genetically related species. The resulting MS fingerprints from this initial multi-species dataset generated by the flowprobe suggest the feasibility of the conceptual microbiology scheme for rapid identification of microbial species based on chemical output similarity. This workflow has been nicely implemented on a small scale, mostly with pathogenic microbes, via MALDI or DESI-MS using individual algorithms. 42-45
Figure 2.
Real-time mass spectra acquired on the surfaces of diverse microbial colonies. Bottom: List of microorganisms and original locations of the strain isolations. Note that the numbers in the parentheses of strain #11-13 and 18 are referential numbers for mass spectrometry analysis not formal strain names.
Of utmost interest, is the high efficiency real-time microbial metabolite screening afforded by the development of the technique. Previously, Watrous et al. demonstrated a new method of viewing secreted microbial products by deconvoluting the mass spectrometry fragmentation features of parent ions mobilized from the colony surface. 11 Via this approach, structurally related molecules, presented as nodes, are mapped based on corresponding spectral similarity. This powerful tool provides system-wide molecular information and represents a new frontier in metabolomic methodology. As more extensive MS/MS datasets are collected and mapped, a novel molecule or metabolite with a characterized fragmentation pattern can be located in the “pseudospace”; of a broader molecular network that is progressively increasing in strain coverage and scale. However, one of the biggest obstacles of the molecular identification using tandem MS-based databases is instrument-dependent fragmentation mismatching. Variations between each laboratory and ionization method create imprecision in MS databases and subsequently, ambiguity during spectral comparisons. Identical molecules could possibly have very different fragmentation spectra in terms of product ions and intensities simply due to a minor change in operating parameters. 46-48 Establishing a universal standard protocol for all natural product MS analysis will improve instrument-dependent data ambiguity and, more importantly, will make spectral comparison processing more efficient and convincing. The ease of the flowprobe system provides a solution to standardize the MS acquisition step on microbial colonies directly from Petri-dishes. Most importantly, the capability of rapid surface profiling in real-time circumvents the need for labor-intensive and time-consuming procedures to improve the speed with which microbial samples from the clinic or biotechnological applications can be screened, perhaps for genus identification, fundamental biofilm studies but certainly for the discovery of specialized metabolites.
Supplementary Material
ACKNOWLEDGMENTS
Research in the P.C.D laboratory for the development of real-time MS was supported by National Institute of Health (NIH) Grant GM094802. L.M. Sanchez is a San Diego IRACDA Fellow (NIH grant GM 68524). Funding from the Programme Committee for Food, Health and Welfare under the Danish Strategic Research Council (grant #2101-07-0012) and the Danish Council for Independent Research in Technology and Production Sciences (grant #116262) for collection of marine bacteria (#19-24) is acknowledged. Isolation of bacterial strains from bats was supported by the Grant Agency of the Czech Republic (grant No. P506/12/1064). Donation of B. polymyxa from Davies group (University of British Columbia) is greatly appreciated.
Footnotes
ASSOCIATED CONTENT
(1) MS spectra acquired on the living microbial colonies and molecular structures of specialized metabolites; (2) MS/MS of all annotated molecules; (3) Preparation procedures of microbial colonies. The flowprobe prototype demonstration video. This material is available free of charge via the internet at http://pubs.acs.org.
REFERENCES
- (1).Phelan VV, Liu W-T, Pogliano K, Dorrestein PC. Nat. Chem. Biol. 2012;8:26–35. doi: 10.1038/nchembio.739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (2).Rappe MS, Giovannoni SJ. Annu. Rev. Microbiol. 2003;57:369–394. doi: 10.1146/annurev.micro.57.030502.090759. [DOI] [PubMed] [Google Scholar]
- (3).Schloss PD, Handelsman J. Microbiol. Mol. Biol. Rev. 2004;68:686–691. doi: 10.1128/MMBR.68.4.686-691.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (4).Curtis TP, Sloan WT, Scannell JW. Proc. Natl. Acad. Sci. U.S.A. 2002;99:10494–10499. doi: 10.1073/pnas.142680199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (5).Human Microbiome Project Consortium Nature. 2012;486:207–214. doi: 10.1038/nature11234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Sekirov I, Russell SL, Antunes LCM, Finlay BB. Physiol. Rev. 2010;90:859–904. doi: 10.1152/physrev.00045.2009. [DOI] [PubMed] [Google Scholar]
- (7).Markle JGM, Frank DN, Mortin-Toth S, Robertson CE, Feazel LM, Rolle-Kampczyk U, von Bergen M, McCoy KD, Macpherson AJ, Danska JS. Science. 2013;339:1084–1088. doi: 10.1126/science.1233521. [DOI] [PubMed] [Google Scholar]
- (8).Yang Y-L, Xu Y, Straight P, Dorrestein PC. Nat. Chem. Bio. 2009;5:885–887. doi: 10.1038/nchembio.252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (9).Liu W-T, Yang Y-L, Xu Y, Lamsa A, Haste NM, Yang JY, Ng J, Gonzalez D, Ellermeier CD, Straight PD, Pevzner PA, Pogliano J, Nizet V, Pogliano K, Dorrestein PC. Proc. Natl. Acad. Sci. U.S.A. 2010;107:16286–16290. doi: 10.1073/pnas.1008368107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (10).Moree WJ, Phelan VV, Wu CH, Bandeira N, Cornett DS, Duggan BM, Dorrestein PC. Proc. Natl. Acad. Sci. U.S.A. 2012;109:13811–13816. doi: 10.1073/pnas.1206855109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (11).Watrous J, Roach P, Alexandrov T, Heath BS, Yang JY, Kersten RD, van der Voort M, Pogliano K, Gross H, Raaijmakers JM, Moore BS, Laskin J, Bandeira N, Dorrestein PC. Proc. Natl. Acad. Sci. U.S.A. 2012;109:E1743–E752. doi: 10.1073/pnas.1203689109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (12).Roach PJ, Laskin J, Laskin A. Analyst. 2010;135:2233–2236. doi: 10.1039/c0an00312c. [DOI] [PubMed] [Google Scholar]
- (13).ElNaggar MS, Barbier C, Van Berkel GJ. J. Am. Soc. Mass Spectrom. 2011;22:1157–1166. doi: 10.1007/s13361-011-0145-5. [DOI] [PubMed] [Google Scholar]
- (14).Kertesz V, Van Berkel GJ. Anal. Chem. 2010;82:5917–5921. doi: 10.1021/ac100954p. [DOI] [PubMed] [Google Scholar]
- (15).Van Berkel GJ, Sanchez AD, Quirke JME. Anal. Chem. 2002;74:6216–6223. doi: 10.1021/ac020540+. [DOI] [PubMed] [Google Scholar]
- (16).Walworth MJ, ElNaggar MS, Stankovich JJ, Witkowski C, Norris JL, Van Berkel GJ. Rapid Commun. Mass Spectrom. 2011;25:2389–2396. doi: 10.1002/rcm.5132. [DOI] [PubMed] [Google Scholar]
- (17).ElNaggar MS, Van Berkel GJ. J. Am. Soc. Mass. Spectrom. 2011;22:1737–1743. doi: 10.1007/s13361-011-0209-6. [DOI] [PubMed] [Google Scholar]
- (18).Vlamakis H, Chai Y, Beauregard P, Losick R, Kolter R. Nat. Rev. Microbiol. 2013;11:157–168. doi: 10.1038/nrmicro2960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (19).Arima K, Kakinuma A, Tamura G. Bioch. Biophys. Res. Comm. 1968;31:488–494. doi: 10.1016/0006-291x(68)90503-2. [DOI] [PubMed] [Google Scholar]
- (20).Peypoux F, Bonmatin JM, Wallach J. Appl. Microbiol. Biotechnol. 1999;51:553–563. doi: 10.1007/s002530051432. [DOI] [PubMed] [Google Scholar]
- (21).Zeigler DR, Prágai Z, Rodriguez S, Chevreux B, Muffler A, Albert T, Bai R, Wyss M, Perkins JB. J. Bacteriol. 2008;190:6983–6995. doi: 10.1128/JB.00722-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (22).Song Y, Talaty N, Datsenko K, Wanner BL, Cooks RG. Analyst. 2009;134:838–841. doi: 10.1039/b900069k. [DOI] [PubMed] [Google Scholar]
- (23).Watrous J, Hendricks N, Meehan M, Dorrestein PC. Anal. Chem. 2010;82:1598–1600. doi: 10.1021/ac9027388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (24).Tsuge K, Matsui K, Itaya M. J. Biotechnol. 2007;129:592–603. doi: 10.1016/j.jbiotec.2007.01.033. [DOI] [PubMed] [Google Scholar]
- (25).Rahme LG, Stevens EJ, Wolfort SF, Shao J, Tompkins RG, Ausubel FM. Science. 1995;268:1899–1902. doi: 10.1126/science.7604262. [DOI] [PubMed] [Google Scholar]
- (26).Conrad D, Haynes M, Salamon P, Rainey PB, Youle M, Rohwer F. Am. J. Respir. Cell Mol. Biol. 2013;48:150–156. doi: 10.1165/rcmb.2012-0059PS. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (27).Dietrich LEP, Price-Whelan A, Petersen A, Whiteley M, Newman DK. Mol. Microbiol. 2006;61:1308–1321. doi: 10.1111/j.1365-2958.2006.05306.x. [DOI] [PubMed] [Google Scholar]
- (28).Lau GW, Hassett DJ, Ran HM, Kong FS. Trends Mol. Med. 2004;10:599–606. doi: 10.1016/j.molmed.2004.10.002. [DOI] [PubMed] [Google Scholar]
- (29).Venturi V. FEMS Microbiol. Rev. 2006;30:274–291. doi: 10.1111/j.1574-6976.2005.00012.x. [DOI] [PubMed] [Google Scholar]
- (30).Heeb S, Fletcher MP, Chhabra SR, Diggle SP, Williams P, Camara M. FEMS Microbiol. Rev. 2011;35:247–274. doi: 10.1111/j.1574-6976.2010.00247.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (31).Soberón-Chávez G, Lépine F, Déziel E. Appl. Microbiol. Biotechnol. 2005;68:718–725. doi: 10.1007/s00253-005-0150-3. [DOI] [PubMed] [Google Scholar]
- (32).Watrous JD, Phelan VV, Hsu C-C, Moree WJ, Duggan BM, Alexandrov T, Dorrestein PC. ISME J. 2013;7:770–780. doi: 10.1038/ismej.2012.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (33).Hassan HM, Fridovich I. J. Bacteriol. 1980;141:156–163. doi: 10.1128/jb.141.1.156-163.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (34).Williamson NR, Fineran PC, Leeper FJ, Salmond GPC. Nat. Rev. Microbiol. 2006;4:887–899. doi: 10.1038/nrmicro1531. [DOI] [PubMed] [Google Scholar]
- (35).González-Pastor JE, Hobbs EC, Losick R. Science. 2003;301:510–513. doi: 10.1126/science.1086462. [DOI] [PubMed] [Google Scholar]
- (36).Ellermeier CD, Hobbs EC, González-Pastor JE, Losick R. Cell. 2006;124:549–559. doi: 10.1016/j.cell.2005.11.041. [DOI] [PubMed] [Google Scholar]
- (37).Pikula J, Bandouchova1 H, Novotný L, Meteyer CU, Zukal J, Irwin NR, Zima J, Martínková N. J. Wildl. Dis. 2012;48:207–211. doi: 10.7589/0090-3558-48.1.207. [DOI] [PubMed] [Google Scholar]
- (38).Button JE, Dutton RJ. Curr. Bio. 2012;22:R587–589. doi: 10.1016/j.cub.2012.06.014. [DOI] [PubMed] [Google Scholar]
- (39).Gram L, Melchiorsen J, Bruhn JB. Mar. Biotechnol. 2010;12:439–451. doi: 10.1007/s10126-009-9233-y. [DOI] [PubMed] [Google Scholar]
- (40).Wang Q, Song F, Xiao X, Huang P, Li L, Monte A, Abdel-Mageed WM, Wang J, Guo H, He W, Xie F, Dai H, Liu M, Chen C, Xu H, Liu M, Piggott AM, Liu X, Capon RJ, Zhang L. Angew. Chem. Int. Ed. 2012;51:1–5. [Google Scholar]
- (41).Nielsen A, Månsson M, Wietz M, Varming AN, Phipps RK, Larsen TO, Gram L, Ingmer H. Mar. Drugs. 2012;10:2584–2595. doi: 10.3390/md10112584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (42).Zhang JI, Talaty N, Costa AB, Xia Y, Tao WA, Bell R, Callahan JH, Cooks RG. Int. J. Mass Spectrom. 2011;301:37–44. [Google Scholar]
- (43).Song Y, Talaty N, Tao WA, Pan Z, Cooks RG. Chem. Commun. 2007:61–63. doi: 10.1039/b615724f. [DOI] [PubMed] [Google Scholar]
- (44).Dubois D, Leyssene D, Chacornac JP, Kostrzewa M, Schmit PO, Talon R, Bonnet R, Delmas J. J. Clin. Microbiol. 2010;48:941–945. doi: 10.1128/JCM.00413-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (45).Benagli C, Rossi V, Dolina M, Tonolla M, Petrini O. Plos One. 2011;6:e16424. doi: 10.1371/journal.pone.0016424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (46).Weinmann W, Wiedemann A, Eppinger B, Renz M, Svoboda M. J. Am. Soc. Mass Spectrom. 1999;10:1028–1037. doi: 10.1016/S1044-0305(99)00070-7. [DOI] [PubMed] [Google Scholar]
- (47).Campbell JM, Collings BA, Douglas DJ. Rapid Commun. Mass Spectrom. 1998;12:1463–1474. doi: 10.1002/(SICI)1097-0231(19980815)12:15<1003::AID-RCM275>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- (48).Schneider BS, Douglas DJ, Chan DDY. Rapid Commun. Mass Spectrom. 2001;15:249–257. doi: 10.1002/rcm.221. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.