A revolution in our understanding of brain development, adult brain function, senescence, and disease has emerged from the recognition of structural and functional plasticity within the mammalian brain (1). For example, the role of brain-derived neurotrophic factor (BDNF) and other hormonal factors that mediate neurogenesis, as well as synaptic and dendritic plasticity in psychiatric disorders (2), has revealed unforeseen neuronal regulatory mechanisms. These build on a growing dynamic conception of brain architecture that complements the understanding of synaptic transmission. This, we are also discovering, is a plastic event altered by both structural and functional remodeling of the synapse. However, little is known about the role of mitochondria in regulating synaptic transmission, and less yet about their implications for cognitive function and memory decline in the aging brain (3). The study by Hara et al. (4) in PNAS synergizes with recent discoveries, revealing a key role of mitochondria regulating synaptic transmission, brain function, and cognition in aging.
Mitochondria populate the cytoplasm of mammalian cells, including neurons, which rely on mitochondrial energy production for survival. These organelles contain their own genome—the mitochondrial DNA (mtDNA)—which encodes essential subunits of the respiratory chain where electrons are combined with oxygen to enable the flow of energy through mitochondria. Energized mitochondria can then synthesize ATP that fuels energy-dependent intracellular reactions (such as endocytosis, ion transport, and neurotransmitter biosynthesis) and sustain other critical mitochondrial functions [Ca2+ handling, reactive oxygen species (ROS) production, and others], contributing to intracellular signaling (5). Equally important is the relatively recent discovery that mitochondria dynamically undergo shape changes through regulated processes of fusion and fission (making longer or shorter organelles, respectively) and actively traffic between cell compartments such as the soma, axon, and presynaptic boutons (6).
Extracellular signals are known to directly affect mitochondria. For example, both glucocorticoid and estrogen receptors translocate into mitochondria and regulate Ca2+ reuptake and ROS production (7, 8). Mitochondria also respond acutely to the metabolic environment by undergoing morphological and functional changes that may influence the cellular aging process (9). Interestingly, although mitochondrial dysfunction resulting from mtDNA mutations often cause severe multisystemic disease, the brain appears most vulnerable to mitochondrial defects, suggesting that neurons are particularly sensitive to bioenergetic fluctuations, and consequently, that mitochondria regulate fundamental aspects of brain function.
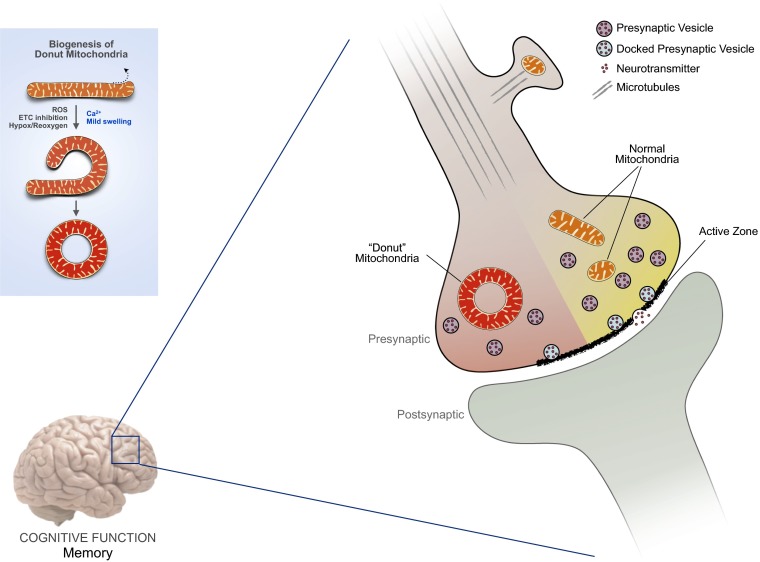
In PNAS, Hara et al. reach across levels of knowledge to demonstrate a relationship between mitochondrial shape and age-related cognitive function. By performing 3D electron microscopy reconstructions of the brain area associated with working memory (dorsolateral prefrontal cortex) in monkeys, the authors characterized mitochondrial morphology at a high level of resolution within presynaptic boutons. In young monkeys, all mitochondria had normal morphology ranging from spheroid to elongated tubule. In contrast, presynaptic boutons in brains of older monkeys contained a significant number of donut-shaped (i.e., toroid) mitochondria (Fig. 1). Donut mitochondria are a hallmark of mitochondrial stress, inducible in controlled conditions (in vitro) by respiratory chain poisons and involving oxidative stress (10, 11). In older monkeys, Hara et al. find that the number of donut mitochondria correlated with worsening of working memory. In contrast, monkeys with boutons containing more normal mitochondria had better memory irrespective of age. So, could the dysfunctional-shaped mitochondria alter synaptic transmission and thus cognition?
Fig. 1.
Schematic representation of presynaptic mitochondria in the mammalian brain and their effect on synaptic remodeling. Donut-shaped mitochondria are negative neuromodulators associated with smaller active zones and fewer docked vesicles in the dorsolateral prefrontal cortex (dlPFC) of monkeys and correlated with poor delayed response memory. (Inset) Donut mitochondria form by curvature and self-fusion of preexisting mitochondrial tubules (10, 11).
In subsequent analyses, the authors find that the presence of donut mitochondria was associated with diminished synapses, as indicated by smaller active zone sizes. Furthermore, donut-containing presynaptic terminals had fewer total docked vesicles, representing fewer readily releasable neurotransmitter-containing synaptic vesicles (Fig. 1). Given that mitochondrial volume density (i.e., content) within synaptosomes strongly correlates with total synaptic vesicle content and release (12), these new data by Hara et al. suggest that the morphological and thus functional state of mitochondria at the synapse influence brain function, possibly by modulating synaptic strength.
Another important modulator of mitochondrial function and brain plasticity is the hormone estradiol (13). Estradiol binds to estrogen receptors (particularly ERβ) that localize to mitochondria where they impact mtDNA gene expression, oxidative capacity, mitochondrial antioxidant defenses, and Ca2+ buffering capacity (7, 13). In the study by Hara et al., although natural reduction of estrogens in postmenopausal monkeys did not significantly impact presynaptic mitochondria, abrupt and complete removal of estrogens in ovariectomized monkeys markedly increased the number of synapses with donut mitochondria and dramatically impaired working memory. This was normalized by estradiol treatment, and the authors note that donut-shaped mitochondria are induced by the mitochondrial respiratory chain inhibitor rotenone and reversed by antioxidant pretreatment (11), suggesting that free radical production is a factor in age-related cognitive dysfunction. Thus, estrogen or other antioxidant treatment might be advisable to mitigate cognitive decline, particularly in the aftermath of surgical menopause.
Importantly, Hara et al. report that donut-containing synapses have smaller active zones and reduced docked vesicles regardless of estrogen status, implying that intrinsic aspects of mitochondrial function directly regulate presynaptic function (14). To this end, Sun et al. (15) used high-resolution fluorescence confocal microscopy to show in real time that the presence, absence, or movement of mitochondria through presynaptic boutons impacts neurotransmitter release by presynaptic terminals. Not only did synapses with mitochondria sustain repeated cycles of neurotransmitter release without signs of depression, but the simple movement of a mitochondrion into, or out of, a presynaptic bouton dynamically modulated presynaptic strength (15). Further to modulating synaptic function in the adult brain, the arrest of mitochondria within axons of the developing embryonic mouse brain (“presynaptic mitochondrial capture”) appears key to determining the position and establishing axon terminals (16). Mitochondria therefore influence both mammalian brain structure and function.
Finally, studies of mitochondrial genetics have also contributed additional evidence that mitochondrial function impact brain function and behavior (17). When mice were generated to contain an equal mixture of two different types of mtDNA (termed heteroplasmy, uncommon in nature), neurons got confused. Given that this mixture of otherwise normal mtDNAs had undetectable consequences on energy production, it was striking to observe that these heteroplasmic mice exhibited impaired memory retention capacity (17). Thus, subtle changes in mitochondrial bioenergetics can have broad effects on mammalian brain function.
In summary, these recent discoveries paint a rather cogent argument that mitochondrial function, positioning, and shape impact brain function and cognition, at least in part by influencing synaptic transmission. These findings in primates increase the likelihood that such neuromodulation extends, both in nature and magnitude, to humans. Whether dysmorphic mitochondria accumulate in synapses of the aging or diseased human brain and the potential of antioxidants to prevent this phenomenon remain to be tested.
Interestingly, the major categories of drugs to treat affective spectrum disorders have been demonstrated to affect mitochondrial function, and new-generation antidepressive therapeutics have preferential action on mitochondrial metabolism (18). Likewise, we know that the single most effective intervention for treating depression, preventing neurodegenerative diseases, and offsetting the age-related cognitive decline is the regular practice of physical activity and maintenance of a healthy metabolic state (19). It is then informative to consider that physical activity and exercise may influence mitochondrial dynamics (9, 20) and induce mitochondrial biogenesis (i.e., the formation of new mitochondria) (21) in various tissues including the brain. Our repertoire of approaches to promote brain adaptability and preserve brain function with aging should benefit from a bioenergetic perspective of how the brain works and dynamically responds to its environment.
Acknowledgments
We thank Meagan McManus for assistance in preparing the manuscript. Work of the authors is supported by a Canadian Institute of Health Research Fellowship to M.P. and National Institute of Health Grants R01 MH41256 and 5 P01 AG16765 to B.S.M.
Footnotes
The authors declare no conflict of interest.
See companion article on page 486.
References
- 1.McEwen BS. Stress, sex, and neural adaptation to a changing environment: Mechanisms of neuronal remodeling. Ann N Y Acad Sci. 2010;1204(Suppl):E38–E59. doi: 10.1111/j.1749-6632.2010.05568.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Duman RS, Monteggia LM. A neurotrophic model for stress-related mood disorders. Biol Psychiatry. 2006;59(12):1116–1127. doi: 10.1016/j.biopsych.2006.02.013. [DOI] [PubMed] [Google Scholar]
- 3.McEwen BS. Brain on stress: How the social environment gets under the skin. Proc Natl Acad Sci USA. 2012;109(Suppl 2):17180–17185. doi: 10.1073/pnas.1121254109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hara Y, Yuk F, Puri R, Janssen WGM, Rapp PR, Morrison JH. Presynaptic mitochondrial morphology in monkey prefrontal cortex correlates with working memory and is improved with estrogen treatment. Proc Natl Acad Sci USA. 2014;111:486–491. doi: 10.1073/pnas.1311310110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wallace DC. Colloquium paper: Bioenergetics, the origins of complexity, and the ascent of man. Proc Natl Acad Sci USA. 2010;107(Suppl 2):8947–8953. doi: 10.1073/pnas.0914635107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chan DC. Fusion and fission: Interlinked processes critical for mitochondrial health. Annu Rev Genet. 2012;46:265–287. doi: 10.1146/annurev-genet-110410-132529. [DOI] [PubMed] [Google Scholar]
- 7.Nilsen J, Diaz Brinton R. Mechanism of estrogen-mediated neuroprotection: Regulation of mitochondrial calcium and Bcl-2 expression. Proc Natl Acad Sci USA. 2003;100(5):2842–2847. doi: 10.1073/pnas.0438041100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Du J, et al. Dynamic regulation of mitochondrial function by glucocorticoids. Proc Natl Acad Sci USA. 2009;106(9):3543–3548. doi: 10.1073/pnas.0812671106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Picard M, Turnbull DM. Linking the metabolic state and mitochondrial DNA in chronic disease, health, and aging. Diabetes. 2013;62(3):672–678. doi: 10.2337/db12-1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Liu X, Hajnóczky G. Altered fusion dynamics underlie unique morphological changes in mitochondria during hypoxia-reoxygenation stress. Cell Death Differ. 2011;18(10):1561–1572. doi: 10.1038/cdd.2011.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ahmad T, et al. Computational classification of mitochondrial shapes reflects stress and redox state. Cell Death Dis. 2013;4:e461. doi: 10.1038/cddis.2012.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ivannikov MV, Sugimori M, Llinás RR. Synaptic vesicle exocytosis in hippocampal synaptosomes correlates directly with total mitochondrial volume. J Mol Neurosci. 2013;49(1):223–230. doi: 10.1007/s12031-012-9848-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rettberg JR, Yao J, Brinton RD. Estrogen: A master reulator of bioenergetic systems in the brain and body [published online ahead of print August 29, 2013] Front Neuroendocrinol. 2013 doi: 10.1016/j.yfrne.2013.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vos M, Lauwers E, Verstreken P. Synaptic mitochondria in synaptic transmission and organization of vesicle pools in health and disease. Front Synaptic Neurosci. 2010;2:139. doi: 10.3389/fnsyn.2010.00139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sun T, Qiao H, Pan PY, Chen Y, Sheng ZH. Motile axonal mitochondria contribute to the variability of presynaptic strength. Cell Rep. 2013;4(3):413–419. doi: 10.1016/j.celrep.2013.06.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Courchet J, et al. Terminal axon branching is regulated by the LKB1-NUAK1 kinase pathway via presynaptic mitochondrial capture. Cell. 2013;153(7):1510–1525. doi: 10.1016/j.cell.2013.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sharpley MS, et al. Heteroplasmy of mouse mtDNA is genetically unstable and results in altered behavior and cognition. Cell. 2012;151(2):333–343. doi: 10.1016/j.cell.2012.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nasca C, et al. L-acetylcarnitine causes rapid antidepressant effects through the epigenetic induction of mGlu2 receptors. Proc Natl Acad Sci USA. 2013;110(12):4804–4809. doi: 10.1073/pnas.1216100110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mattson MP. Energy intake and exercise as determinants of brain health and vulnerability to injury and disease. Cell Metab. 2012;16(6):706–722. doi: 10.1016/j.cmet.2012.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Picard M, et al. Acute exercise remodels mitochondrial membrane interactions in mouse skeletal muscle. J Appl Physiol (1985) 2013;115(10):1562–1571. doi: 10.1152/japplphysiol.00819.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Steiner JL, Murphy EA, McClellan JL, Carmichael MD, Davis JM. Exercise training increases mitochondrial biogenesis in the brain. J Appl Physiol (1985) 2011;111(4):1066–1071. doi: 10.1152/japplphysiol.00343.2011. [DOI] [PubMed] [Google Scholar]