Abstract
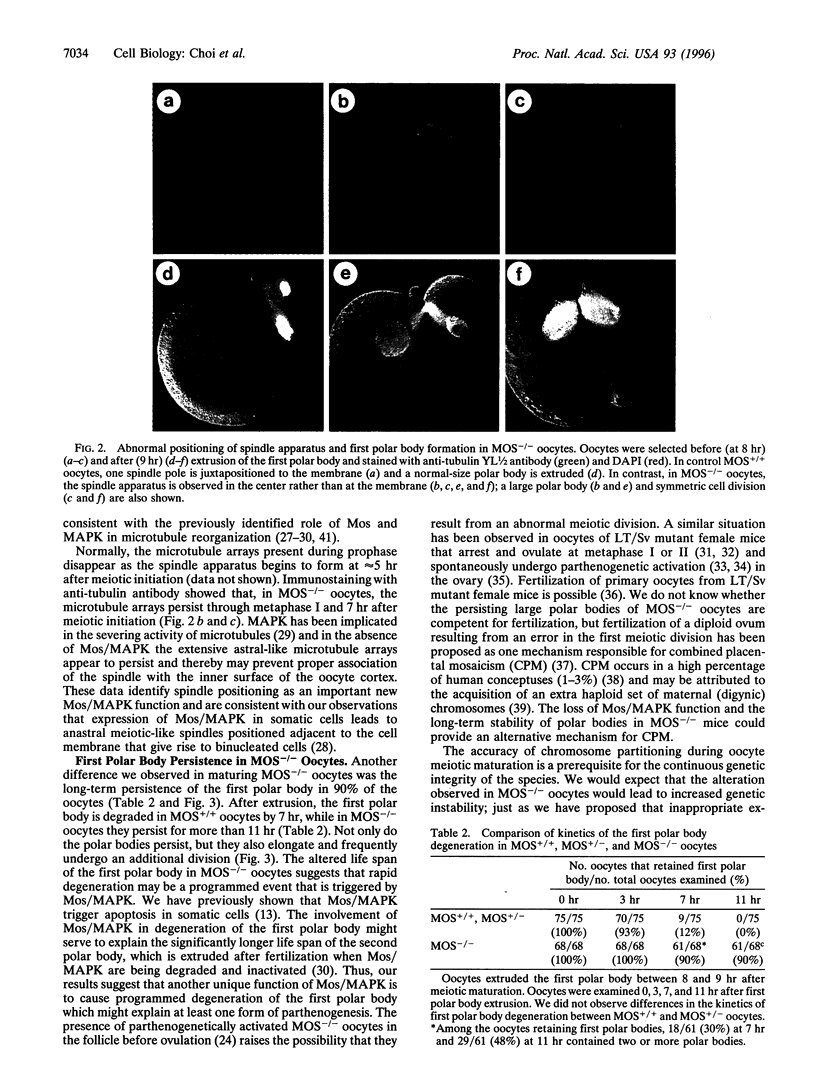
Mos is an upstream activator of mitogen-activated protein kinase (MAPK) and, in mouse oocytes, is responsible for metaphase II arrest. This activity has been likened to its function in Xenopus oocytes as a component of cytostatic factor. Thus, Mos-deficient female mice (MOS-/-) are less fertile and oocytes derived from these animals fail to arrest at metaphase II and undergo parthenogenetic activation [Colledge, W. H., Carlton, M. B. L., Udy, C. B. & Evans, M. J. (1994) Nature (London) 370, 65-68 and Hashimoto, N., Watanabe, N., Furuta. Y., Tamemoto, B., Sagata, N., Yokoyama, M., Okazaki, K., Nagayoshi, M., Takeda, N., Ikawa, Y. & Aizawa, S. (1994) Nature (London) 370, 68-71]. Here we show that maturing MOS-/- oocytes fail to activate MAPK throughout meiosis, while p34cdc2 kinase activity is normal until late in metaphase II when it decreases prematurely. Phenotypically, the first meiotic division of MOS-/- oocytes frequently resembles mitotic cleavage or produces an abnormally large polar body. In these oocytes, the spindle shape is altered and the spindle fails to translocate to the cortex, leading to the establishment of an altered cleavage plane. Moreover, the first polar body persists instead of degrading and sometimes undergoes an additional cleavage, thereby providing conditions for parthenogenesis. These studies identify meiotic spindle formation and programmed degradation of the first polar body as new and important roles for the Mos/MAPK pathway.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Colledge W. H., Carlton M. B., Udy G. B., Evans M. J. Disruption of c-mos causes parthenogenetic development of unfertilized mouse eggs. Nature. 1994 Jul 7;370(6484):65–68. doi: 10.1038/370065a0. [DOI] [PubMed] [Google Scholar]
- Ferrell J. E., Jr, Wu M., Gerhart J. C., Martin G. S. Cell cycle tyrosine phosphorylation of p34cdc2 and a microtubule-associated protein kinase homolog in Xenopus oocytes and eggs. Mol Cell Biol. 1991 Apr;11(4):1965–1971. doi: 10.1128/mcb.11.4.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukasawa K., Vande Woude G. F. Mos overexpression in Swiss 3T3 cells induces meiotic-like alterations of the mitotic spindle. Proc Natl Acad Sci U S A. 1995 Apr 11;92(8):3430–3434. doi: 10.1073/pnas.92.8.3430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotoh Y., Moriyama K., Matsuda S., Okumura E., Kishimoto T., Kawasaki H., Suzuki K., Yahara I., Sakai H., Nishida E. Xenopus M phase MAP kinase: isolation of its cDNA and activation by MPF. EMBO J. 1991 Sep;10(9):2661–2668. doi: 10.1002/j.1460-2075.1991.tb07809.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotoh Y., Nishida E., Matsuda S., Shiina N., Kosako H., Shiokawa K., Akiyama T., Ohta K., Sakai H. In vitro effects on microtubule dynamics of purified Xenopus M phase-activated MAP kinase. Nature. 1991 Jan 17;349(6306):251–254. doi: 10.1038/349251a0. [DOI] [PubMed] [Google Scholar]
- Haccard O., Jessus C., Cayla X., Goris J., Merlevede W., Ozon R. In vivo activation of a microtubule-associated protein kinase during meiotic maturation of the Xenopus oocyte. Eur J Biochem. 1990 Sep 24;192(3):633–642. doi: 10.1111/j.1432-1033.1990.tb19270.x. [DOI] [PubMed] [Google Scholar]
- Hashimoto N., Watanabe N., Furuta Y., Tamemoto H., Sagata N., Yokoyama M., Okazaki K., Nagayoshi M., Takeda N., Ikawa Y. Parthenogenetic activation of oocytes in c-mos-deficient mice. Nature. 1994 Jul 7;370(6484):68–71. doi: 10.1038/370068a0. [DOI] [PubMed] [Google Scholar]
- Jacobs P. A., Angell R. R., Buchanan I. M., Hassold T. J., Matsuyama A. M., Manuel B. The origin of human triploids. Ann Hum Genet. 1978 Jul;42(1):49–57. doi: 10.1111/j.1469-1809.1978.tb00930.x. [DOI] [PubMed] [Google Scholar]
- Kanki J. P., Donoghue D. J. Progression from meiosis I to meiosis II in Xenopus oocytes requires de novo translation of the mosxe protooncogene. Proc Natl Acad Sci U S A. 1991 Jul 1;88(13):5794–5798. doi: 10.1073/pnas.88.13.5794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kosako H., Gotoh Y., Nishida E. Requirement for the MAP kinase kinase/MAP kinase cascade in Xenopus oocyte maturation. EMBO J. 1994 May 1;13(9):2131–2138. doi: 10.1002/j.1460-2075.1994.tb06489.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mansour S. J., Matten W. T., Hermann A. S., Candia J. M., Rong S., Fukasawa K., Vande Woude G. F., Ahn N. G. Transformation of mammalian cells by constitutively active MAP kinase kinase. Science. 1994 Aug 12;265(5174):966–970. doi: 10.1126/science.8052857. [DOI] [PubMed] [Google Scholar]
- Masui Y., Markert C. L. Cytoplasmic control of nuclear behavior during meiotic maturation of frog oocytes. J Exp Zool. 1971 Jun;177(2):129–145. doi: 10.1002/jez.1401770202. [DOI] [PubMed] [Google Scholar]
- Maxwell S. A., Arlinghaus R. B. Serine kinase activity associated with Maloney murine sarcoma virus-124-encoded p37mos. Virology. 1985 May;143(1):321–333. doi: 10.1016/0042-6822(85)90119-9. [DOI] [PubMed] [Google Scholar]
- McFadden D. E., Kwong L. C., Yam I. Y., Langlois S. Parental origin of triploidy in human fetuses: evidence for genomic imprinting. Hum Genet. 1993 Nov;92(5):465–469. doi: 10.1007/BF00216452. [DOI] [PubMed] [Google Scholar]
- Nebreda A. R., Hunt T. The c-mos proto-oncogene protein kinase turns on and maintains the activity of MAP kinase, but not MPF, in cell-free extracts of Xenopus oocytes and eggs. EMBO J. 1993 May;12(5):1979–1986. doi: 10.1002/j.1460-2075.1993.tb05847.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niebuhr E. Triploidy in man. Cytogenetical and clinical aspects. Humangenetik. 1974 Feb 21;21(2):103–125. doi: 10.1007/BF00281030. [DOI] [PubMed] [Google Scholar]
- O'Neill G. T., Kaufman M. H. Ovulation and fertilization of primary and secondary oocytes in LT/Sv strain mice. Gamete Res. 1987 Sep;18(1):27–36. doi: 10.1002/mrd.1120180105. [DOI] [PubMed] [Google Scholar]
- Oskarsson M., McClements W. L., Blair D. G., Maizel J. V., Vande Woude G. F. Properties of a normal mouse cell DNA sequence (sarc) homologous to the src sequence of Moloney sarcoma virus. Science. 1980 Mar 14;207(4436):1222–1224. doi: 10.1126/science.6243788. [DOI] [PubMed] [Google Scholar]
- Pelech S. L., Sanghera J. S. MAP kinases: charting the regulatory pathways. Science. 1992 Sep 4;257(5075):1355–1356. doi: 10.1126/science.1382311. [DOI] [PubMed] [Google Scholar]
- Posada J., Cooper J. A. Requirements for phosphorylation of MAP kinase during meiosis in Xenopus oocytes. Science. 1992 Jan 10;255(5041):212–215. doi: 10.1126/science.1313186. [DOI] [PubMed] [Google Scholar]
- Posada J., Yew N., Ahn N. G., Vande Woude G. F., Cooper J. A. Mos stimulates MAP kinase in Xenopus oocytes and activates a MAP kinase kinase in vitro. Mol Cell Biol. 1993 Apr;13(4):2546–2553. doi: 10.1128/mcb.13.4.2546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sagata N., Daar I., Oskarsson M., Showalter S. D., Vande Woude G. F. The product of the mos proto-oncogene as a candidate "initiator" for oocyte maturation. Science. 1989 Aug 11;245(4918):643–646. doi: 10.1126/science.2474853. [DOI] [PubMed] [Google Scholar]
- Sagata N., Oskarsson M., Copeland T., Brumbaugh J., Vande Woude G. F. Function of c-mos proto-oncogene product in meiotic maturation in Xenopus oocytes. Nature. 1988 Oct 6;335(6190):519–525. doi: 10.1038/335519a0. [DOI] [PubMed] [Google Scholar]
- Sagata N., Watanabe N., Vande Woude G. F., Ikawa Y. The c-mos proto-oncogene product is a cytostatic factor responsible for meiotic arrest in vertebrate eggs. Nature. 1989 Nov 30;342(6249):512–518. doi: 10.1038/342512a0. [DOI] [PubMed] [Google Scholar]
- Shibuya E. K., Boulton T. G., Cobb M. H., Ruderman J. V. Activation of p42 MAP kinase and the release of oocytes from cell cycle arrest. EMBO J. 1992 Nov;11(11):3963–3975. doi: 10.1002/j.1460-2075.1992.tb05490.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shibuya E. K., Ruderman J. V. Mos induces the in vitro activation of mitogen-activated protein kinases in lysates of frog oocytes and mammalian somatic cells. Mol Biol Cell. 1993 Aug;4(8):781–790. doi: 10.1091/mbc.4.8.781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speirs S., Kaufman M. H. Effect of exogenous hormones on the ovulation of primary and secondary oocytes in LT/Sv strain mice. Gamete Res. 1988 Oct;21(2):179–184. doi: 10.1002/mrd.1120210208. [DOI] [PubMed] [Google Scholar]
- Stevens L. C., Varnum D. S. The development of teratomas from parthenogenetically activated ovarian mouse eggs. Dev Biol. 1974 Apr;37(2):369–380. doi: 10.1016/0012-1606(74)90155-9. [DOI] [PubMed] [Google Scholar]
- Tessarollo L., Vogel K. S., Palko M. E., Reid S. W., Parada L. F. Targeted mutation in the neurotrophin-3 gene results in loss of muscle sensory neurons. Proc Natl Acad Sci U S A. 1994 Dec 6;91(25):11844–11848. doi: 10.1073/pnas.91.25.11844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verlhac M. H., Kubiak J. Z., Clarke H. J., Maro B. Microtubule and chromatin behavior follow MAP kinase activity but not MPF activity during meiosis in mouse oocytes. Development. 1994 Apr;120(4):1017–1025. doi: 10.1242/dev.120.4.1017. [DOI] [PubMed] [Google Scholar]
- Verlhac M. H., Kubiak J. Z., Weber M., Géraud G., Colledge W. H., Evans M. J., Maro B. Mos is required for MAP kinase activation and is involved in microtubule organization during meiotic maturation in the mouse. Development. 1996 Mar;122(3):815–822. doi: 10.1242/dev.122.3.815. [DOI] [PubMed] [Google Scholar]
- Yew N., Mellini M. L., Vande Woude G. F. Meiotic initiation by the mos protein in Xenopus. Nature. 1992 Feb 13;355(6361):649–652. doi: 10.1038/355649a0. [DOI] [PubMed] [Google Scholar]
- Yew N., Oskarsson M., Daar I., Blair D. G., Vande Woude G. F. mos gene transforming efficiencies correlate with oocyte maturation and cytostatic factor activities. Mol Cell Biol. 1991 Feb;11(2):604–610. doi: 10.1128/mcb.11.2.604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou R. P., Oskarsson M., Paules R. S., Schulz N., Cleveland D., Vande Woude G. F. Ability of the c-mos product to associate with and phosphorylate tubulin. Science. 1991 Feb 8;251(4994):671–675. doi: 10.1126/science.1825142. [DOI] [PubMed] [Google Scholar]